Abstract
Introduction
Reliable quality of life (QoL) measures and utility values are needed for patients with type 2 diabetes mellitus (T2DM) with a variety of comorbid conditions to help facilitate cost-effectiveness modeling. This study aimed to evaluate the Diabetes Treatment-Related Quality of Life (DTR-QOL) and EuroQol 5-dimension 5-level (EQ-5D-5L) questionnaires in patients with T2DM with and without diabetes complications and comorbidities in Japan.
Methods
This was an observational survey study involving 1000 patients with T2DM, at least 20 years old, receiving treatment at Nara University Hospital or Takamura Internal Medicine Clinic in Japan. Patients completed the DTR-QOL and EQ-5D-5L questionnaires and clinicians completed an accompanying case report form. The DTR-QOL and EQ-5D-5L are scored on a scale of 0–100 and 0–1, with 100 and 1 representing the best possible scores, respectively.
Results
Out of 1000 recruited patients, 978 were included in the final analysis. Patients reported an average EQ-5D-5L value of 0.92 ± 0.11. Utility values corresponded to the degree of severity of health conditions while few differences were observed when stratified by the HbA1c 7% threshold, age, or BMI level, nor did the values correspond to the degree of clinical risk factors. Patients reported an average total DTR-QOL score of 79.26 ± 13.26. The DTR-QOL was sensitive to detect differences in patients with T2DM with a variety of complications and comorbidities, risk factors, and treatments.
Conclusion
This is the largest study to report QOL values for patients with diabetes in Japan and the first to include a variety of comorbid diabetic conditions. These findings may be useful for cost-effectiveness modeling of patients with T2DM in Japan.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Reliable quality of life (QoL) measures and utility values are needed for patients with type 2 diabetes mellitus (T2DM) with a variety of comorbid conditions to help facilitate cost-effectiveness modeling. |
This study aimed to evaluate the Diabetes Treatment-Related Quality of Life (DTR-QOL) and EuroQol 5-dimension 5-level (EQ-5D-5L) questionnaires in patients with T2DM with and without comorbidities in Japan. |
What was learned from the study? |
Patients reported an average EQ-5D-5L value of 0.92 ± 0.11. Utility values corresponded to the degree of severity of health conditions while little differences were observed when stratified by the HbA1c 7% threshold, age, or BMI level, nor did the values correspond to the degree of clinical risk factors. Patients reported an average total DTR-QOL score of 79.26 ± 13.26. |
The DTR-QOL was sensitive to detect differences in patients with T2DM with a variety of complications, risk factors, and treatments. These findings may be useful for cost-effectiveness modeling of patients with T2DM in Japan. |
Digital Features
This article is published with digital features to facilitate understanding of the article. You can access the digital features on the article’s associated Figshare page. To view digital features for this article go to https://doi.org/10.6084/m9.figshare.13013909.
Introduction
Type 2 diabetes mellitus (T2DM) is an endocrine disease, which occurs when a patient’s blood glucose level is too high and it is estimated to be prevalent in more than 10 million people in Japan [1]. Chronic in nature, T2DM is a disease that requires regular drug therapies along with continuous diet and exercise therapy. Without appropriate treatment and care, diabetes causes serious complications and comorbidities, such as neuropathy, retinopathy, or kidney diseases and further progresses to advanced conditions such as coronary heart disease, stroke, foot amputation, or kidney failure [2]. It is also known that patients with diabetes have a greater risk of depression than those without diabetes [3]. Complications and comorbidities can also have profound effects on patients’ ability to manage their self-care which ultimately affects treatment adherence and effectiveness outcomes. Such complications and comorbidities can significantly influence patients’ perception of health, greatly restrict one’s daily activities, and reduce quality of life (QoL). With a wide variety of comorbid conditions linked with T2DM, it is imperative to investigate the impact of each condition on patients’ QoL accordingly.
Generation of QoL values is also important from a cost-effectiveness analysis (CEA) perspective. In Japan, up until April 2019, cost-effectiveness was not a key evaluation factor for reimbursement or pricing decisions. However, this changed after the Japanese Ministry of Health, Labour and Welfare (MHLW) implemented a CEA-based health technology assessment (HTA) for pharmaceuticals and medical devices [4]. Now there is a growing need for local utility data to populate cost-effectiveness models.
In order to derive utility values, generic instruments, such as the EuroQol five-dimension questionnaire (EQ-5D), are widely used. The EQ-5D is a preferred measure for MHLW and other HTA organizations in the world, as it allows comparison of utilities across various indications [5, 6]. Despite its benefits, previous research has shown that for some conditions, important aspects of a disease or symptoms may not be captured solely through the five dimensions of the EQ-5D [7]. As the content of the utility measure has a significant impact on the accurate measurement of treatment effects [8, 9], capturing unique patient experiences in combination with the generic measures is generally endorsed by regulatory authorities.
For the diabetes indication, there are several Japanese studies reporting utility values [10,11,12]; however, the data is not segmented by various demographic characteristics nor detailed sequelae of diabetes and, to date, there are no findings utilizing the EQ-5D five-level version (EQ-5D-5L) in Japan. Diabetes-specific instruments to measure QoL have also been developed [13]. One validated QoL instrument for Japanese patients with diabetes is the Diabetes Therapy-Related QoL (DTR-QOL) questionnaire. It was developed to measure the impact of treatment on patient QoL, including patients’ comfort and satisfaction [14].
This study aimed to collect utility values associated with various comorbidities and complications of patients with T2DM utilizing EQ-5D-5L, a general QoL measure, and DTR-QOL, a disease-specific measure.
Methods
This was an observational survey study involving 1000 patients diagnosed with T2DM who were 20 years old or older, and receiving diabetes care at Nara University Hospital or Takamura Internal Medicine Clinic. Patients with any serious or unstable psychiatric disorders (such as drug addiction), disorders that prevented physical participation in the study, or those with learning and/or cognitive difficulties that prevent him/her reading and understanding questionnaires (e.g., dementia) were excluded from this study.
Patients completed the paper-based DTR-QOL and EQ-5D-5L questionnaires and recorded self-reported hypoglycemic events, neuropathic symptoms, and patient characteristics. Clinicians completed a brief case report form on medical history for each patient.
The general utility measure used in this study was the Japanese validated version of the EQ-5D-5L. The EQ-5D-5L measures the patient’s overall health state in a descriptive system of health-related QoL states consisting of five dimensions (mobility, self-care, usual activities, pain/discomfort, and anxiety/depression) each of which can takes one of five responses ranging from no problems to extreme problems [15]. The EQ-5D-5L results were converted to health utility scores using the Japan tariff [16]. The EQ-5D-5L is scored on a scale of negative 1 to 1, with 1 representing perfect health, 0 representing death, and − 1 representing worse than death. In addition, a visual analogue scale (VAS) rated current health state between 0 and 100 [16].
The disease-specific measure selected for this study was the DTR-QOL questionnaire. DTR-QOL consists of 29 questions, developed and validated to assess QoL and daily life impact of patients with diabetes in Japan [14]. This tool has been used in previous studies to measure QoL of patients with diabetes and was utilized for the first time in the IDegLira DUAL II Japan phase 3 trial in Japanese patients with T2DM, with results reported in 2019 [17, 18]. DTR-QOL consists of four domains (usual activities, anxiety and dissatisfaction, hypoglycemia, and satisfaction) and 29 questions, each of which can takes one of seven responses. The DTR-QOL is scored on a scale of 0–100, with 100 representing the best possible score.
Patient characteristics, DTR-QOL, and EQ-5D-5L values were reported descriptively. Mean, median, and standard deviation were reported for continuous variables and number/percentage for categorical variables. The comparison of psychometric properties of the instruments was not a focus of this study. The main focus was rather to derive utility values and QoL scores to inform researchers.
As an exploratory objective, significance testing of patient characteristics, complications, and comorbid conditions was conducted with one-way analysis of variance (ANOVA) tests and a stepwise linear regression model was constructed to understand the factors influencing EQ-5D-5L and DTR-QOL values. In order to control for a potentially inflated type error I due to multiple testing, a Bonferroni correction was applied which yielded a corrected significance threshold of p ≤ 0.001. All data analyses were performed using statistics software SAS version 9.4.
This study was approved by the Nara University institutional review board committee (ID #1665) and an independent ethics committee (Network Japan; ID #17NNP0030) prior to participant recruitment. It was performed in accordance with the Helsinki Declaration of 1964 and its later amendments. All participants provided informed consent to participate in the study.
Results
Out of 1000 recruited patients, 21 respondents provided incomplete data and one did not meet the eligibility criteria. In total 978 patients completed both the EQ-5D-5L and DTR-QOL and were included in the final analysis. Sixty-two percent of patients had at least one comorbid condition, with neuropathic disorder being the most commonly reported comorbidity. Stage 1 nephropathy was also frequently observed within this population; however, as this is commonly defined as a pre-nephropathy condition, it was not counted as a comorbid condition in this study [19]. The female population accounted for 42.4% of the total population and the mean age was 62.6 ± 11.30 with the highest proportion of patients from the 60–69 years old age group (Table 1).
Patients’ perceived health conditions were queried using a self-developed case report form (CRF). Less than 20% of the total population perceived their health conditions as “poor” or “very poor”. Similar trends were observed in the distribution of each clinical condition, where the majority of the population reported between “fair” to “excellent” health (Table 2).
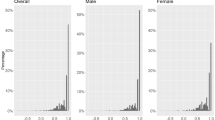
As an EQ-5D-5L score of 0 and 1, respectively, denotes “death” and “perfect health”, the overall mean of the EQ-5D-5L score in this study was close to perfect health (0.92 ± 0.11) despite 62% having T2DM with one or more comorbid conditions (Table 3). Patients without any comorbid conditions had an average utility value of 0.95 ± 0.09, while patients with a comorbid condition had a utility of 0.91 ± 0.12. The VAS score for all patients was 76.5 ± 15.1, where 0 and 100 each representing the worst possible condition and the best possible condition. Despite the large sample, the distribution of responses to the EQ-5D-5L and the VAS score were highly skewed towards better health; 60.33% of patients had a perfect EQ-5D-5L score (1.0) (Figs. 1, 2).
The DTR-QOL and EQ-5D-5L were able to differentiate across patient-reported health conditions (Table 3). By types of comorbid condition, EQ-5D-5L scores corresponded consistently to the severity of health conditions, where less severe groups reported higher scores. The lowest utilities were reported for gangrene of the foot (n = 6; 0.61) and blindness (n = 1; 0.10). Utility value for severe hypoglycemia was reported in two patients with an average score of 0.89 (Table 3). In terms of treatment mode of administration, the EQ-5D-5L utility values were higher for oral treatment patients compared to the injectable user group (0.94 vs 0.87). Few differences were observed in utility values when stratified by the HbA1c 7% threshold, age, or BMI level, nor did the values correspond to the degree of clinical risk factors (Fig. 3)
In order to better interpret the results, several exploratory regression models were constructed. Factors most strongly associated with EQ-5D-5L scores included neuropathy, diabetic foot with gangrene, and peripheral arterial disease. The average DTR-QOL score was 79.26 ± 13.85 out of a total score of 100 (Table 3). Similar to the EQ-5D-5L utility findings, gangrene of the foot (61.66) and blindness (57.14) had the lowest scores.
Values of the DTR-QOL total score demonstrated the largest differences with therapy type, where patients on diet and exercise had an average score of 83.21 compared to a score of 70.45 for the injectable therapy group (Fig. 4). Statistically significant differences were observed in the DTR-QOL total score utility values when stratified by the HbA1c 7% threshold, type of therapy, age, or BMI level, whereas this was only the case for the EQ-5D-5L values for BMI and type of therapy (Figs. 3, 4). This confirms the sensitivity of the DTR-QOL questionnaire to detect differences in key elements of diabetes therapy.
Discussion
This study examined the patient characteristics, utility values for various comorbid conditions using EQ-5D-5L, and disease-specific patient-reported outcomes using DTR-QOL for patients with type 2 diabetes who are currently undergoing treatment in Japan.
Our findings reported an average utility value close to a perfect score (0.92) within a range of 0.32–1.00 with 60.33% reporting perfect health (1.00), despite almost two-thirds of patients having one or more comorbid condition or complication. A previous study in Japan found similarly high scores for patients with diabetes even across different measures such as EQ-5D-3L and EQ-5D-5L [20]. Compared to existing literature outside of Japan, the utility values were much higher [21]. For example, in a UK study published in 2002, the overall score for those without any comorbid condition was 0.785, and 0.505 for those with an amputation, while our study reported a score of 0.780 for foot amputation patients [22]. In previous literature from Japan among patients with T2DM assessed with EQ-5D-3L, the overall utility score ranged between 0.846 and 0.930. The majority of these patients did not report any problems on the questionnaire. There was also a similar difference between EQ-5D scores and EQ-VAS scores in comparison to the difference in the present study (0.12 vs 0.14) [11]. These findings illustrate that Japan utility values are relatively higher than studies in other settings. This may imply that Japanese patients tend to perceive their health in a better condition compared to the perceptions of patients in other countries.
Alternatively, the generic and broad nature of the five items of the EQ-5D-5L may lack sensitivity in the Japanese context for the concerns most salient to the T2DM indication in comparison to DTR-QOL, which was developed specifically to address patients with T2DM. The average DTR-QOL score was sensitive to detect differences in patients with T2DM with a variety of complications, risk factors, and treatment, in part because of the items related to T2DM-specific treatment and related anxieties. This finding re-emphasizes the importance of utilizing a disease-specific measure in addition to a generic measure, especially for evidence generation related to disease-specific regulatory submission. Studies have also shown that the utility values and DTR-QOL scores corresponded to the degree of severity. For example, patients with stage 1 nephropathy reported a higher utility score than stage 4 patients, and patients with foot amputation had a lower score than those without one [23]. This finding is an important reference amid the emerging needs for local utility data in preparation for CEA submissions.
In contrast to the DTR-QOL questionnaire, the EQ-5D-5L measure was only able to demonstrate statistical significance for type of therapy and BMI level, as presented in Figs. 3 and 4. For example, the lower age group tended to have a lower QoL value by 0.1 points, while those with an HbA1C level under 7% had a higher utility value by 0.1 points than the 7% or more group. However, the DTR-QOL total scores stratified by HbA1c level, type of therapy, age, and BMI level were all found to be statistically different—indicating sensitivity to detect differences in QoL with these factors.
Monitoring glycemic control and body weight or BMI is fundamental in the treatment guidelines by the Japan Diabetes Society [24]. The DTR-QOL is able to detect differences in these factors as well as treatment type which are a focus of day-to-day patient impact and can subsequently affect longer-term complications. The DTR-QOL is also able to detect differences in microvascular complications, similar to the EQ-5D-5L. The EQ-5D-5L is a more generic QoL measure, and therefore may be better suited for evaluating the maintenance of QoL compared to patients without diabetes, the final objective of treatment. Therefore, these measures appear to complement each other, with the DTR-QOL covering the fundamental factors associated with diabetes and its treatment through detecting the impact of microvascular and atherosclerotic complications.
The EQ-5D-5L has better coverage of these complications and overall QoL for comparison to other patient populations. A large prospective observational study of patients with diabetes in 17 countries found considerable heterogeneity in self-rated health in different geographies across several QoL-related domains. Japanese patients were found to have the lowest levels of patient engagement and empowerment among all countries studied [25]. Patients with low engagement, defined as low awareness of diabetes-related pathologies and the need to take preventative measures, may not express concerns about their conditions which could negatively impact sensitivity of some QoL scales to detect differences among Japanese patients with different complications and comorbidities. Our exploratory analysis results suggested that neuropathy, diabetic foot with gangrene, and peripheral arterial disease were most strongly associated with the levels of utility values. Further investigation of factors that are associated with utility value levels would benefit development of cost-effectiveness models.
A limitation of this study was that the population investigated appears to be relatively healthy, limiting our ability to assess the impact of more severe complications. Extensive testing with inferential statistics was outside the scope of this study. Psychometric analysis and comparison of the measures used in this study would be valuable, requiring further investigation. Another limitation was that this study did not account for timing of complications as patients may have adjusted or acclimated to their severe consequences which would be reflected in the relatively small decrement in utility. Also, for certain conditions such as severe hypoglycemia, it was difficult to generalize the utility score because of the small sample size (n = 2). So even in a study of this size, critical utility values for CEAs within diabetes could not all be identified.
Conclusions
This is the first study to our knowledge to report QoL values in a variety of comorbid diabetic conditions in Japan by utilizing the EQ-5D-5L and DTR-QOL questionnaires on a large sample of patients. Comparison of the results identifies disease-specific characteristics that a generic QoL measure cannot fully capture. Values derived from the EQ-5D-5L can be utilized by researchers involved in CEA as a benchmark as most the values associated with complications and comorbidities were not available until now. Practicing clinicians can utilize the EQ-5D-5L to compare their patients to those without diabetes to glean insights into T2DM impairments or utilize the DTR-QOL to monitor the day-to-day impact from treatments which can subsequently affect longer-term outcomes. In addition to gaining insights into the patients’ perspective, these findings may also be useful for cost-effectiveness modeling of patients with T2DM in Japan to meet the newly developed HTA requirements.
References
Ministry of Health Labour Welfare. National health and nutrition survey results. Japan 2017. https://www.mhlw.go.jp/stf/houdou/0000177189.html. Accessed 22 May 2019.
National Institute of Diabetes and Digestive and Kidney Diseases. Preventing diabetes problems. USA 2019. https://www.niddk.nih.gov/health-information/diabetes/overview/preventing-problems. Accessed 27 May 2019.
Holt RI, de Groot M, Golden SH. Diabetes and depression. Curr Diab Rep. 2014;14(6):014–0491.
Ministry of Health Labour Welfare. 17th Chuo-shakai hokeniryou-kyougikai Cost-effectiveness Analysis subcommittee, Drug pricing subcommittee. Tokyo 2019. https://www.mhlw.go.jp/stf/shingi2/0000211220_00013.html. Accessed 17 July 2019.
EuroQol. EuroQol—a new facility for the measurement of health-related quality of life. Health Policy. 1990;16(3):199–208.
National Institute of Public Health. Guideline for preparing cost-effectiveness evaluation to the Central Social Insurance Medical Council. Core2 Health. 2019. https://c2h.niph.go.jp/tools/guideline/guideline_en.pdf. Accessed 22 July 2019.
Swinburn P, Lloyd A, Boye KS, Edson-Heredia E, Bowman L, Janssen B. Development of a disease-specific version of the EQ-5D-5L for use in patients suffering from psoriasis: lessons learned from a feasibility study in the UK. Value Health. 2013;16(8):1156–62.
Kind P, Lafata JE, Matuszewski K, Raisch D. The use of QALYs in clinical and patient decision-making: issues and prospects. Value Health. 2009;12(Suppl 1):S27–30.
McGrath C, Rofail D, Gargon E, Abetz L. Using qualitative methods to inform the trade-off between content validity and consistency in utility assessment: the example of type 2 diabetes and Alzheimer’s disease. Health Qual Life Out. 2010;8:23.
Ohsawa I, Ishida T, Oshida Y, Yamanouchi K, Sato Y. Subjective health values of individuals with diabetes in Japan: comparison of utility values with the SF-36 scores. Diabetes Res Clin Pract. 2003;62(1):9–16.
Sakamaki H, Ikeda S, Ikegami N, et al. Measurement of HRQL using EQ-5D in patients with type 2 diabetes mellitus in Japan. Value Health. 2006;9(1):47–53.
Takahara M, Katakami N, Shiraiwa T, et al. Evaluation of health utility values for diabetic complications, treatment regimens, glycemic control and other subjective symptoms in diabetic patients using the EQ-5D-5L. Acta Diabetol. 2019;56(3):309–19.
Nair R, Kachan P. Outcome tools for diabetes-specific quality of life: study performed in a private family practice clinic. Can Fam Physician. 2017;63(6):e310–5.
Ishii H. Development and psychometric validation of the Diabetes Therapy-Related QOL (DTR-QOL) questionnaire. J Med Econ. 2012;15(3):556–63.
EuroQol. EQ-5D-5L about. The Netherlands 2019. https://euroqol.org/eq-5d-instruments/eq-5d-5l-about/. Accessed 17 Oct 2019.
Ikeda S, Shiroiwa K, Igarashi A, et al. Developing a Japanese version of the EQ-5D-5L value set. Health Med Sci. 2015;64:47–55 (in Japanese).
Clinicaltrials.gov. A double-blinded trial comparing the efficacy and safety of insulin degludec/liraglutide and insulin degludec both in combination with metformin in Japanese subjects with type 2 diabetes mellitus inadequately controlled with basal or pre-mix/combination insulin therapy and oral anti-diabetic drugs (DUAL™ II Japan). 2019. https://clinicaltrials.gov/ct2/show/NCT02911948?term=DUAL+II+Japan&rank=1. Accessed 28 Nov 2019.
Crawford B, Takamura S, Nishioka Y, Langer J, Watanabe M, Kim H. Quality of life and utility values for cost-effectiveness modeling in Japanese patients with type 2 diabetes. Value Health. 2018;21:S139.
Haneda M, Utsunomiya K, Koya D, et al. A new classification of diabetic nephropathy 2014: a report from joint committee on diabetic nephropathy. J Diabetes Investig. 2015;6(2):242–6.
Shiroiwa T, Fukuda T, Ikeda S, et al. Japanese population norms for preference-based measures: EQ-5D-3L, EQ-5D-5L, and SF-6D. Qual Life Res. 2016;25(3):707–19.
Beaudet A, Clegg J, Thuresson PO, Lloyd A, McEwan P. Review of utility values for economic modeling in type 2 diabetes. Value Health. 2014;17(4):462–70.
Clarke P, Gray A, Holman R. Estimating utility values for health states of type 2 diabetic patients using the EQ-5D (UKPDS 62). Med Decis Mak. 2002;22(4):340–9.
Fukuhara S, Lopes AA, Bragg-Gresham JL, et al. Health-related quality of life among dialysis patients on three continents: the Dialysis Outcomes and Practice Patterns Study. Kidney Int. 2003;64(5):1903–10.
Japan Diabetes Society. Treatment guide for diabetes. Tokyo 2016. http://www.fa.kyorin.co.jp/jds/uploads/Treatment_Guide_for_Diabetes_2016-2017.pdf.
Nicolucci A, Kovacs Burns K, Holt RI, et al. Diabetes Attitudes, Wishes and Needs second study (DAWN2™): cross-national benchmarking of diabetes-related psychosocial outcomes for people with diabetes [published correction appears in Diabet Med. 2013 Oct;30(10):1266]. Diabet Med. 2013;30(7):767–777.
Acknowledgements
Funding
This study was sponsored, and the journal’s rapid service fee funded, by Novo Nordisk Pharma Ltd.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Disclosures
Hitoshi Ishii has received lecture fees from Novo Nordisk Pharma, Eli Lilly, Sanofi, Daiichi Sankyo, Sumitomo Dainippon Pharma, Takeda Pharmaceutical, Mitsubishi Tanabe Pharma, Nippon Boehringer Ingelheim, MSD, Ono Pharmaceutical. Hiroshi Takamura has no disclosures. Yuichi Nishioka has received consulting fees from Novo Nordisk Pharma Ltd. Hyunchung Ray Kim and Bruce Crawford are employees of Syneos Health which was contracted to conduct this study. Michihisa Watanabe and Jakob Langer are employees of Novo Nordisk Pharma Ltd.
Compliance with Ethics Guidelines
This study was approved by the Nara University institutional review board committee (ID #1665) and an independent ethics committee (Network Japan; ID #17NNP0030) prior to participant recruitment. It was performed in accordance with the Helsinki Declaration of 1964 and its later amendments. All participants provided informed consent to participate in the study. The authors would like to thank the study participants for their cooperation.
Data Availability
The datasets generated during and/or analyzed during the current study are not publicly available due to the wording of the informed consent form (ICF).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Ishii, H., Takamura, H., Nishioka, Y. et al. Quality of Life and Utility Values for Cost-Effectiveness Modeling in Japanese Patients with Type 2 Diabetes. Diabetes Ther 11, 2931–2943 (2020). https://doi.org/10.1007/s13300-020-00938-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-020-00938-3