Abstract
Introduction
Effective blood glucose control remains a constant problem in patients with type 2 diabetes (T2D), even if they are being properly treated with one or more currently available drugs. The present study was designed as a 3-year retrospective observational study to determine whether the use of either empagliflozin, a sodium-glucose co-transporter 2 (SGLT2) inhibitor, or insulin would provide any improvement in the control of the blood glucose levels in patients with T2D who were already being treated with a cocktail of three different oral antidiabetic drugs.
Methods
Adult patients with T2D were enrolled in this study if they exhibited suboptimal glycemic control (HbA1c 7.5–12.0%) despite being continuously treated for at least 3 months with metformin, dipeptidyl-peptidase 4 inhibitor, and glimepiride. Empagliflozin (25 mg/day, n = 154) or basal long-acting insulin (n = 147) was added as a fourth medication to the existing drug regimen. The major outcomes that were monitored in this study included the measurement of HbA1c, fasting plasma glucose (FPG), and general cardiometabolic and blood markers.
Results
After the addition of empagliflozin or basal insulin to the existing oral anti-diabetic agent (OAD) regimen, the baseline levels of HbA1c were reduced after month 36 in both the empagliflozin (8.9 ± 1.0% to 7.4 ± 0.8%, P < 0.01) and insulin (9.0 ± 1.4% to 8.0 ± .1.4%, P < 0.05) groups. The HbA1c reduction was higher in the empagliflozin group to the end of the 36-month study period (7.4 ± 0.8% vs. 8.0 ± 1.4%, empagliflozin vs. insulin, P < 0.05). FPG showed a similar trend in the early period but it was not maintained at the end of study. Body weight decreased (P < 0.01) from baseline (70.4 ± 12.3 kg) to month 36 (65.6 ± 11.4 kg) in the empagliflozin group but not the insulin group. At 36 months, the body weight in the empagliflozin group (65.6 ± 11.4 kg) was significantly lower (P < 0.01) than that in the insulin treatment group (70.0 ± 10.9 kg).
Conclusion
Empagliflozin was shown to perform as well as better than insulin when used as part of a quadruple drug regimen for regulating blood glucose levels in suboptimally controlled patients with T2D.
Clinical Trial Number
NCT 05103306 (ClinicalTrials.gov).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Although insulin remains the standard supplemental treatment prescribed to patients WITH T2D with inadequate glycemic control despite using three different OADs, not all patients are willing to inject themselves with insulin. For this reason, this study was designed to examine whether alternate oral antidiabetic drugs, specifically empagliflozin, can be prescribed to these patients to provide effective control of their glycemic index. |
What was learned from the study? |
The durability of the efficacy of empagliflozin as a fourth agent was maintained over 3 years, and empagliflozin showed similar or better glycemic control compared to basal long-acting insulin and yielded a body weight reduction. The addition of empagliflozin rather than basal insulin could become a viable treatment option for patients with T2D who have suboptimal glycemic control with multiple OADs. |
Introduction
In terms of the treatment of type 2 diabetes (T2D), the long-term durability of a single oral anti-diabetic agent (OAD) in maintaining adequate blood glucose reduces as the duration of diabetes increases because of progressive pancreatic β-cell dysfunction and insulin resistance [1]. There are a variety of drug regimens that can be employed to improve glucose control in T2D patients, including two or three OADs or injection therapy using either insulin or glucagon-like peptide 1 (GLP1). In particular, insulin injection provides an effective treatment modality to control hyperglycemia, with unlimited dose coverage. However, some T2D patients have a reluctance to initiate insulin injection due to their psychological fear of physical needle penetration or the potential to become hypoglycemic. Moreover, there is evidence that insulin injection therapy has limitations when used to achieve the glycemic goal and has, in some cases, lowered the quality of life in patients with T2D [2,3,4]. In a recent study that allowed patients to provide their opinions about insulin treatment via a questionnaire, many diabetic patients were found to suffer from negative perceptions such as personal failure, illness severity, and expected harm from the insulin injection, which led them to choose to find alternate OADs [5].
To respond these unmet needs, we performed a prior observational study to compare the results of using a quadruple drug regimen including empagliflozin to the results of using basal long-acting insulin in combination with three different OADs for 24 weeks. In that study, there was a marked improvement in glycemic control and reduced body weight in the empagliflozin group compared to the insulin glargine group [6].
In the present study, we extended our retrospective observational study of patients with inadequately controlled T2D to 3 years to evaluate the long-term efficacy of a quadruple drug combination that added either empagliflozin or basal long-acting insulin to the triple drug combination of metformin, glimepiride, and a dipeptidyl peptidase-4 (DPP-4) inhibitor. Our realtively long-term study was expected to overcome the limitations of conventional insulin-based treatment.
Methods
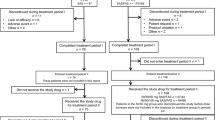
This retrospective observational study was conducted using previously eligible patients with T2D (aged 18 or older) that visited the Endocrinology Department at Chungbuk National University Hospital in South Korea between January 2015 and December 2021. The patients had to have shown inadequately controlled blood glucose levels at baseline (HbA1c levels between 7.5 and 12.0%) and had to have taken a triple drug cocktail consisting of metformin (2000 mg/day or their maximum tolerated dose), glimepiride (≥ 6 mg/day), and DPP-4 inhibitor (the maximum dose according to the local label; sitagliptin for 74 patients and vildagliptin for 80 patients) for at least 3 months beforehand. At the time of enrollment, all patients were encouraged to initiate long-acting insulin (Tresiba® or Lantus®) as their fourth drug to improve their glucose control. In the insulin group, the starting dose of long-acting insulin at baseline was determined by the blood glucose level, and was prescribed as between 10 and 20 units/day by the attending physician. At each scheduled visit, it was recommended that the patients should adjust their doses of insulin to achieve FPG levels of < 150 mg/dL or HbA1c < 7.5%. If those patients refused to use insulin, empagliflozin (25 mg/day) was added to their drug regimen. At this point, the patients were monitored by their attending physician for a period of 3 or more years. The exclusion criteria used in this study have been previously published by our group and were as follows [7]: (1) diagnosed with other types of diabetes, such as type 1 diabetes or secondary diabetes (e.g., chronic pancreatitis, pancreatectomy, steroid-induced diabetes mellitus, acromegaly, or Cushing’s syndrome); (2) ongoing anticancer therapy; (3) the use of any weight-reductive medications within 3 months before baseline measurements; (4) chronic hepatic disease (serum aspartate transaminase [AST] or alanine transaminase [ALT], 3 × upper limit of normal range) or renal impairment (serum creatinine level > 1.5 mg/dl for men or 1.4 mg/dl for women, estimated glomerular filtration rate [eGFR] < 60 mL/min/1.73 m2 of the body surface area); (5) use of any SGLT2 inhibitors for ≥ 7 consecutive days within 3 months prior to baseline measurements; and (6) the presence of symptoms reflecting severe hyperglycemia (polydipsia, polyuria, or polyphagia), weight loss, or ketosis. All patients were monitored at their 3-month follow-up appointments. If the patient exhibited an inability to maintain their normal glucose levels, the dose of glimepiride was decreased in the empagliflozin group or reduced insulin was prescribed for insulin-treated patients. Adherence to their regimen was determined by counting the number of pills remaining at each interval visit.
The study was carried out in accordance with the principles stated in the Declaration of Helsinki as revised in 2013 and the International Conference of Harmonization/Good Clinical Practice guidelines [8, 9]. An institutional review board at Chungbuk National University Hospital approved the study (no. 2021-05-009), and the need for written consent from patients to participate was waved because this study was performed retrospectively. This trial was registered with ClinicalTrials.gov, number NCT 05103306.
The primary efficacy outcome in this study was a change in the HbA1c levels at 3 years compared with baseline values. Secondary outcomes were: (1) changes in the fasting plasma glucose (FPG) levels; (2) changes in body weight and blood pressure (systolic and diastolic).
At baseline, the electronic medical records (EMRs) of all patients were reviewed, including comorbidities, family history of diabetes, duration of diabetes, smoking history, and alcohol consumption. At baseline, at 3 and 6 months, and then at every 6-month interval for the duration of the study period, each patient was measured for their blood pressure and body weight, and 12 h fasting blood sampling was performed. HbA1c (Bio-Rad Laboratories, Hercules, CA, USA), fasting insulin and C-peptide levels (Abbott, Lake Forest, IL, USA), plasma glucose, serum AST, ALT, lipid profiles, and urinary albumin-to-creatinine ratio (ACR) (TOSHIBA FX-8, Japan), and spot urine sediment (Sysmex, Japan) were measured as previously described [7] using the relevant monitoring equipment. The homeostasis model assessment of insulin resistance and beta-cell function was performed as previously described [10]. The eGFR was calculated using the Modification of Diet in Renal Disease formula [11].
Statistical analyses were done in SPSS for Windows (version 24.0, IBM Corp., Armonk, NY, USA). The required sample size was calculated for the 3-year outcome. A sample size of 146 patients per group was needed to achieve a 90% power to detect a difference in HbA1c of 0.5% with the two-sided t test at a significance level of 0.05, assuming a standard deviation of 1.2% and a 20% loss to follow-up.
Continuous variables and discrete variables are expressed as mean ± standard deviation or standard error and as number (%), respectively. The Kolmogorov–Smirnov test for normality was performed as an adequate statistical test for continuous variables. For the analysis of baseline characteristics, the independent Student’s t test and the χ2 test were performed. The median with the interquartile range was analyzed by the Mann–Whitney U test for fasting insulin, C-peptide, triglyceride, eGFR, and spot urine ACR due to a skewed distribution. Efficacy analyses based on the changes in HbA1c, FPG, blood pressure, and body weight between baseline and the time points at each visit were performed using the paired t-test. For tests of between-group differences, a two-sided significance level of 0.05 was used unless otherwise indicated. The last observation carried forward approach was used to impute missing data.
Results
A total of 301 patients were included in this study, and baseline characteristics are summarized in Table 1. No significant difference was calculated between the empagliflozin- and insulin-treated groups with respect to the mean (± SD) ages (57.7 ± 10.8 and 57.7 ± 14.8 years, respectively) and the mean duration of diabetes (12.2 ± 6.7 and 11.0 ± 9.6 years, respectively). There were a higher number of hypertensive patients in the empagliflozin group compared to the insulin group. Otherwise, laboratory parameters, including HbA1c, were comparable between groups.
After the addition of empagliflozin or basal long-acting insulin to the existing triple OAD drug regimen of the patients, HbA1c levels were significantly reduced over time from baseline to month 36 in both the empagliflozin (8.9 ± 1.0% to 7.4 ± 0.8%, P < 0.01) and insulin (9.0 ± 1.4% to 8.0 ± 0.1.4%, P < 0.05) groups. The reduction in HbA1c levels was significantly greater (P < 0.05) in the empagliflozin compared to the insulin group at the 36-month time point (Table 2). FPG levels exhibited a similar trend to HbA1c whereby the decrease was significantly higher (P < 0.01) in the empagliflozin group (118 ± 22.8 mg/dL) versus the insulin group (135 ± 40.2 mg/dL) at 3 months. After 6 months, FPG remained significantly lower (P < 0.05; 125.6 ± 31.8 mg/dL) in the empagliflozin versus the insulin group (138.4 ± 51.1 mg/dL), but this did not last through to the end of the study (Table 3).
With respect to body weight, a significant decrease from baseline to month 36 was observed in the empagliflozin group (70.4 ± 12.3 kg to 65.6 ± 11.4 kg, P < 0.01), but insulin-treated patients did not exhibit any measurable difference (68.2 ± 12.4 to 70.0 ± 10.9 kg, NS). Body weight at the end of month 36 showed a significant difference (P < 0.01) between the empagliflozin (65.6 ± 11.4 kg) and the insulin (70.0 ± 10.9 kg) groups (Fig. 1). In terms of blood pressure and lipid profiles, no significant difference was observed between the two treatment groups.
Discussion
A previous study by our group over a shorter 24-week study period showed that empagliflozin was more effective than basal insulin at reducing Hb1Ac in patients with T2D that were not even able to properly control their glycemia with triple OAD drug therapy which included metformin, glimepiride, and DPP4 inhibitors [6]. That initial observational study provided the rationale for the present study to pursue whether the effectiveness of empagliflozin could be maintained over a longer period of time (3 years in the present study) in this particular population with T2D. Since the standard treatment for this type of patient population with T2D is the injection of either insulin or glucagon-like peptide-1 (GLP-1) receptor agonist [12], our study was also designed to assess whether empagliflozin was more effective than insulin at managing glycemia. Alternate drug therapies to injection therapy are needed in patients that may be less compliant with this type of treatment, as there are a population of patients who have an aversion to injection therapy, which could have a negative long-time impact on their health [5].
The findings in the present study demonstrated that the effectiveness of empagliflozin was sustainable over a prolonged 3-year period, and that empagliflozin showed similar or even better glycemic control compared to the use of insulin. In addition to the glycemic control improvement, empagliflozin yielded a reduced body weight over the 3-year period, whereas a minimal change in weight was measured in the insulin users.
The FPG levels in the empagliflozin group were lower than those in the insulin group over the entire study period (Table 3). The elevated levels of FPG in the insulin group users may be attributed to various factors. First, some patients treated with insulin reported hypoglycemia-like symptoms—such as a sensation of frustration or hunger—when the FPG hovered near 150 mg/dL, so they preferred to maintain their FPG at > 200 mg/dL. Second, some patients in the insulin group self-regulated their insulin dosage by using an amount lower than that recommended by the physician due to their inherent fear of becoming hypoglycemic. Their fear may be region dependent due to differences in body type, since a prior study showed that a majority of east Asian patients with T2D are relatively lean and have a higher risk of hypoglycemia associated with insulin use than their Western counterparts [13]. Prior titration of basal insulin requires active participation by the health care provider and the willingness of the patient to comply with their medical guidance [14], so not using the recommended dosage could have led to the dysregulated levels of FPG.
In terms of adverse effects (AEs), hypoglycemia was the most commonly reported AE in both groups (12 cases in the empagliflozin group and 9 cases in the insulin group, respectively). The number of patients with nocturnal hypoglycemia was 3 cases in the empagliflozin group and 6 cases in the insulin group, and only 1 case of severe hypoglycemia was reported, which occurred in the insulin group. Four cases of genitourinary infection were reported—all in the empagliflozin group. A total of 22 patients were lost to follow-up: 12 patients in the empagliflozin group, including the death of 1 patient due to hepatic failure, and 10 patients in the insulin group, including the death of 1 patient due to prostatic cancer with lung metastasis.
As with all retrospective studies, a limitation of our study design and data was the relatively small population of patients chosen from our clinic. In our study, we selected 144 patients out of > 800 insulin users according to their propensity scores, which were based upon many of the biological factors described in the “Methods” section. There could also be selection bias due to the selection of empagliflozin for the patients that would not take insulin, although this is unlikely. With this in mind, it will be necessary to design a larger prospective multi-center study to confirm the present results within and outside of our geographical region.
Conclusions
The addition of empagliflozin rather than basal insulin as a fourth drug given to T2D patients with inadequate glucose control could be a viable therapeutic option to keep patients more willing to use their medication and to better control their glycemia, thus hopefully minimizing the future deterioration of other physiological functions.
References
Turner RC, Cull CA, Frighi V, Holman RR. Glycemic control with diet, sulfonylurea, metformin, or insulin in patients with type 2 diabetes mellitus: progressive requirement for multiple therapies (UKPDS 49). UK Prospective Diabetes Study (UKPDS) Group. JAMA. 1999;281(21):2005–12.
Groop LC, Pelkonen R, Koskimies S, Bottazzo GF, Doniach D. Secondary failure to treatment with oral antidiabetic agents in non-insulin-dependent diabetes. Diabetes Care. 1986;9(2):129–33.
Luigi FM, Lulu KL, Shaloo G, Ron P. Association of hypoglycemia severity with clinical, patient-reported and economic outcomes in US patients with type 2 diabetes using basal insulin. Diabetes Obes Metab. 2018;20:1156–65.
Brod M, Kongso JH, Lessard S, Christensen TL. Psychological insulin resistance: patient beliefs and implications for diabetes management. Qual Life Res. 2009;18(1):23–32.
Ku EJ, Lee DH, Jeon HJ, Park F, Oh TK. Psychometric analysis regarding the barriers to providing effective insulin treatment in type 2 diabetic patients. Diabetes Ther. 2021;12(1):159–70. https://doi.org/10.1007/s13300-020-00947-2.
Ku EJ, Lee DH, Jeon HJ, Oh TK. Effectiveness and safety of empagliflozin-based quadruple therapy compared with insulin glargine-based therapy in patients with inadequately controlled type 2 diabetes: an observational study in clinical practice. Diabetes Obes Metab. 2019;21(1):173–7. https://doi.org/10.1111/dom.13476.
Ku EJ, Lee DH, Jeon HJ, Oh TK. Long-term effectiveness and safety of quadruple combination therapy with empagliflozin versus dapagliflozin in patients with type 2 diabetes: 3-year prospective observational study. Diabetes Res Clin Prac. 2021. https://doi.org/10.1016/j.diabres109123.
World Medical Association. Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA. 2013;310(20):2191–4. https://doi.org/10.1001/jama.2013.281053.
ICH. ICH Harmonised Tripartite Guideline. Guideline for good clinical practice. J Postgrad Med. 2001;47(2):121–30.
Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985;28(7):412–9. https://doi.org/10.1007/BF00280883.
Fuiano G, Zoccali C, Bertoni E, et al. Guidelines for ambulatory monitoring of kidney transplant patients Adaptation of the Guidelines of the American Society of Transplantation (J Am Soc Nephrol 2000; 11 (S1): 86). G Ital Nefrol. 2004;21(Suppl 28):S11-50.
American Diabetes Association. 9. Pharmacologic approaches to glycemic treatment: standards of medical care in diabetes—2021. Diabetes Care. 2021;44(Suppl 1):S111–24.
Chan JCN, Bunnaq P, Chan SP, et al. Glycemic responses in Asian and non-Asian people with type 2 diabetes initiating insulin glargine 100 units/mL: a patients-level pooled anaysis of 16 randomized controlled trials. Diabetes Res Clin Prac. 2018;135:199–205.
Ji L, Zhang P, Zhu D, et al. Observational registry of basal insulin treatment (ORBIT) in patients with type 2 diabetes uncontrolled with oral antihyperglycemic drugs: real-life use of basal insulin in China. Diabetes Obes Metab. 2017;19:822–30.
Acknowledgements
Funding
This work was supported by funding for the academic research program of Chungbuk National University in 2022. The Rapid Service Fee was funded by the authors.
Medical Writing and/or Editorial Assistance
We would like to express our sincere thanks to Dr. Frank Park (University of Tennessee Health Science Center in Memphis) for his editing of the manuscript.
Authors Contributions
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this manuscript, had full access to the data in this study, take complete responsibility for the integrity of the data and accuracy of the data analysis, and give final approval for the version to be published. Tae Keun Oh is the guarantor taking full responsibility for this work, including the decision to submit and publish the manuscript. Eu Jeong Ku and Tae Keun Oh contributed to the conceptualization of the study protocol, collected and analyzed data, interpreted the results, wrote the initial draft, and revised the manuscript for important intellectual content. All authors provided final approval of the published version of the manuscript and agree to be accountable for all aspects of this work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Disclosures
Eu Jeong Ku and Tae Keun Oh have nothing to disclose.
Compliance with Ethics Guidelines
The study was carried out in accordance with the principles stated in the Declaration of Helsinki as revised in 2013 and the International Conference of Harmonization/Good Clinical Practice guidelines. An institutional review board at Chungbuk National University Hospital approved the study (no. 2021-05-009), and the need for written consent from patients to participate was waved because this study was performed retrospectively.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Ku, E.J., Oh, T.K. Long-Term Effectiveness of Quadruple Combination Therapy with Empagliflozin Versus Basal Long-Acting Insulin Therapy in Patients with Type 2 Diabetes: 3-Year Retrospective Observational Study. Diabetes Ther 14, 1471–1479 (2023). https://doi.org/10.1007/s13300-023-01437-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-023-01437-x