Abstract
Introduction
Weight loss has been identified as a key strategy for improving glycemic and metabolic outcomes in people with type 2 diabetes (T2D). However, the long-term, real-world impact of weight loss on these outcomes remains unclear. This study aimed to investigate (1) the association between weight loss and glycemic control, (2) association between weight loss and metabolic parameters, and (3) predictors of weight loss and how weight change trajectory varies based on index body mass index (BMI).
Methods
A retrospective, longitudinal cohort study using the linked IQVIA Ambulatory electronic medical records and PharMetrics® Plus databases was performed from January 1, 2010 through December 31, 2019 in adults with T2D. Participants were categorized into 1-year and 5-year follow-up cohorts based on their observed weight change over time. Longitudinal values for vital signs and laboratory parameters, including BMI, weight, glycated hemoglobin (HbA1c), and metabolic parameters (liver enzymes and cholesterol), were reported at index date and every 6 months post index date. Multivariable logistic regression analysis was used to evaluate the factors associated with weight loss.
Results
Of 1,493,964 people evaluated, 1,061,354 (71%) and 308,320 (20.6%) were classified into the 1-year and 5-year follow-up cohorts. Average HbA1c reductions of 1.2% and 0.5% were observed among people who lost ≥ 15% of index weight in the 1-year and 5-year follow-up cohorts, respectively. Higher weight loss percentages were associated with numerically greater improvements in metabolic parameters. The presence of bariatric surgery and higher index BMIs were identified as the strongest predictors of ≥ 15% and ≥ 10% weight loss in both follow-up cohorts.
Conclusion
Results from this study suggest that modest and sustained weight loss can lead to clinically meaningful improvements in glycemic and metabolic parameters among people with T2D. These findings highlight the importance of weight management in managing T2D and preventing its associated complications.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
There is limited real-world evidence on long-term impact of weight loss on glycemic control and metabolic parameters in people with type 2 diabetes (T2D). |
The current study assesses both the short-term (1-year) and long-term (5-year) impact of weight loss in people with T2D using US claims data linked with electronic medical records (EMR). |
What was learned from the study? |
The key findings highlight the positive effects of weight loss in people with T2D and overweight or obesity and indicate that modest and sustained weight loss can lead to clinically meaningful improvements in glycemic and metabolic parameters. |
In view of these results, health care providers may consider personalizing weight-centric approaches for their patients for better management of T2D and prevention of its associated complications. |
Introduction
Over the past 2 decades, there has been a substantial increase in the prevalence of diabetes, with approximately 37 million people being affected in the USA [1]. Obesity is a strong predictor and risk factor for type 2 diabetes (T2D) and is associated with insulin resistance and hyperglycemia [2]. Previous studies have established the role of weight loss in effective management of T2D [3,4,5,6]. A systematic literature review and meta-analysis of prospective trials studying the effects of energy-reduced diets, anti-obesity drugs, and bariatric surgery showed that weight loss in people with T2D and overweight or obesity is accompanied by a reduction in glycated hemoglobin (HbA1c) levels [7].
A modest and sustained weight loss of 5–10% of total body weight in people with T2D and overweight or obesity was shown to improve glycemic control and reduce dependency on glucose-lowering medications. An even greater weight loss can further lower HbA1c and fasting glucose, and promote sustained remission of diabetes for at least 2 years [8]. For people with body mass index (BMI) > 35 kg/m2 who do not achieve durable weight loss nor improvement in comorbidities with nonsurgical methods, bariatric surgery has been shown to result in weight loss of as much as 60–70% of excess body weight, with about 60–80% of people with obesity showing T2D remission following the surgery [9].
Inflammation has also been identified as one of the key risk factors contributing to the development of T2D [2, 10, 11]. The chronic inflammatory microenvironment established by obesity and T2D in people with these conditions results in various metabolic disorders associated with weight gain [12,13,14,15], including diabetic dyslipidemia, characterized by high low-density lipoprotein (LDL) cholesterol and triglycerides, and low high-density lipoprotein (HDL) cholesterol, comprising a major cardiovascular risk factor [16]; and elevated plasma levels of liver enzymes, such as aspartate aminotransferase (AST) and alanine transaminase (ALT) [17]. Furthermore, weight loss in people with obesity was shown to reduce serum levels of inflammatory markers, subsequently improving insulin sensitivity [18,19,20].
Given the association between obesity and T2D, treatment guidelines from the American Diabetes Association, the American Association of Clinical Endocrinology, and the American College of Endocrinology recommend weight loss through lifestyle interventions [4, 21] or nonsurgical energy restriction in people with T2D and a BMI ≥ 25 kg/m2 [21]. Furthermore, the guidelines recommended newer therapies such as sodium-glucose co-transporter 2 inhibitors (SGLT2i), glucose-dependent insulinotropic polypeptide (GIP)/glucagon-like peptide 1 (GLP-1) receptor agonist (RA), and GLP-1 RAs for weight-centric management of T2D. In addition to their ability to improve glycemic control, these therapies have also been shown to promote weight loss [2], leading to a paradigm shift towards better management of T2D.
However, there is limited real-world evidence on weight loss in people with T2D and its impact on long-term glycemic control and metabolic parameters. The current study assessed both the short-term and long-term impact of weight loss in people with T2D using US claims data linked with electronic medical records (EMR). Changes in glycemic and metabolic parameters were assessed in both 1-year and 5-year follow-up cohorts across weight-change categories. In addition, the study also assessed the factors influencing weight loss and the impact of index BMI on the participants’ weight-loss trajectory.
Methods
Study Design and Data Source
This retrospective, longitudinal cohort study was conducted using data from the IQVIA Ambulatory EMR-US database and the PharMetrics® Plus commercial claims database. The IQVIA Ambulatory EMR database is a large real-world EMR database in the USA, containing data for more than 76 million people from over 100,000 physicians in over 800 practices. A subset of people in the EMR database also have health insurance claims information available in the PharMetrics Plus database, which contains information for more than 150 million individuals across the USA. The linked IQVIA Ambulatory EMR and PharMetrics Plus databases encompass de-identified data for over 12 million people. This study was conducted from January 1, 2010 through December 31, 2019, and the study selection period was from January 1, 2010 through December 31, 2018.
Selection Criteria
This study included all people in the EMR database who had at least one diagnosis of T2D during the study selection period and at least two recorded weight values. The index date was defined as the date of the first observed T2D diagnosis. Participants were aged ≥ 18 years on the index date and had at least one weight value recorded in the database on or after this date. This weight value was designated as the index weight, and the corresponding date was the index weight date. In addition to the index weight, participants were also required to have another weight value recorded in the database ≥ 90 days after the index weight date. Lastly, eligible participants had ≥ 12 months of continuous health plan enrollment data post index weight date and were followed from index weight date until either disenrollment from the health plan or the last available physician follow-up visit (whichever came earlier). Participants were excluded if they had a diagnosis of any of the conditions listed in Supplementary Table S1.
Study Population
The study population was categorized into two non-mutually exclusive cohorts based on observed weight change over time: (1) participants with weight values observed at 1-year (± 90 days) follow-up from the index weight date, and (2) participants with weight values observed at 5-year (± 90 days) follow-up from the index weight date. Both cohorts were further stratified by weight changes, and the following categories were identified: weight increase of > 3%; no weight change (i.e., weight within ± 3% of index weight value); weight decrease of > 3% to < 5%; weight decrease of ≥ 5% to < 10%; weight decrease of ≥ 10% to < 15%; and weight decrease of ≥ 15%.
Study Variables
Demographic data were collected relative to the index weight date. This included variables such as age, sex, race, and geographic location (US census region). Longitudinal values for vital signs and laboratory parameters, including BMI, weight, HbA1c, proportion of participants achieving HbA1c < 7%, and metabolic parameters, were reported for each 6-month period following the index weight date. Baseline or index BMI was reported based on the following categories: normal weight (18.5 to < 25 kg/m2), overweight (25 to < 30 kg/m2), class 1 obesity (30 to < 35 kg/m2), class 2 obesity (35 to < 40 kg/m2), and class 3 obesity (≥ 40 kg/m2). The metabolic parameters evaluated in this study were LDL cholesterol, HDL cholesterol, triglycerides, total cholesterol, fasting blood glucose (FBG), ALT, and AST. All the data were obtained from the EMR database. Overall, comorbidity burden was summarized using the Charlson Comorbidity Index (CCI) score [22], and diabetes severity was assessed using the Diabetes Complications Severity Index (DCSI) score [23]. Both scores were calculated and reported for each participant during the first year of follow-up. Additionally, the percentages of participants with nonalcoholic steatohepatitis, sleep apnea, polycystic ovary syndrome, and obesity were reported. The number and percentages of participants who underwent bariatric surgery and who received antidepressants, oral corticosteroids, and/or weight loss medications for each year of follow-up were also reported.
Statistical Analysis
The data were analyzed using descriptive statistics and reported separately for each cohort. Continuous variables were summarized using means (standard deviation), and categorical variables were summarized using numbers and percentages. A multivariable logistic regression analysis was performed to evaluate the factors associated with weight loss as a function of a set of covariates (demographics, clinical characteristics, and baseline BMI) to control for potential confounding factors. In this analysis, the likelihood of a participant achieving a specified weight loss threshold (i.e., ≥ 10% and ≥ 15% weight loss at 1-year or 5-year follow-up) was estimated, and the associated odds ratios (OR) and 95% confidence intervals (CI) were calculated. Statistical significance was defined as a p value of < 0.05. Imputations were not applied for missing data, either for participants or variables. Only observed data were reported at each timepoint. All data were analyzed using SAS Studio.
Compliance with Ethics Guidelines
This study was conducted in accordance with the ethical principles that have their origin in the Declaration of Helsinki and that are consistent with Good Pharmacoepidemiology Practices and applicable laws and regulations of the country or countries where the study was conducted, as appropriate. Participants’ data were deidentified to protect privacy. A formal Consent to Release Information form was not required since this was an observational study that used previously collected data and did not impose any form of intervention.
Results
Demographics and Clinical Characteristics
A total of 1,493,964 people with T2D met the study selection criteria, of which 1,061,354 (71%) were in the 1-year follow-up cohort and 308,320 (20.6%) in the 5-year follow-up cohort (Fig. S1). Table 1 presents a summary of demographic and baseline characteristics for both cohorts. The mean age was similar across all weight-change categories in each of the cohorts. The lowest mean age (58.7 years) was observed among participants with ≥ 15% weight loss in the 1-year follow-up period. Conversely, in the 5-year follow-up period, the lowest mean age (59.2 years) was observed among participants with weight increase > 3%. For the cohorts, large decreases in body weight (i.e., 10% or higher) occurred in < 5% of participants during 1-year follow-up and < 15% of participants during 5-year follow-up. There were no substantial differences among the weight-change categories in both follow-up periods for the following variables: race, geographic region, weight loss medications, CCI, or DCSI scores.
Obesity and sleep apnea were the most commonly diagnosed comorbidities of interest across all weight-change categories. Moreover, these comorbidities were most prevalent among those who experienced greater weight loss percentages. Furthermore, a numerically higher prevalence of bariatric surgery was also observed among people who had a weight loss of ≥ 15% (1-year follow-up cohort: 6.8% vs. 0.2–1.0% for the remaining weight-change categories; 5-year follow-up cohort: 4.0% vs. 0.3–0.7% for the remaining weight-change categories). Overall, the percentage of participants who underwent bariatric surgery tended to increase among patients with greater weight loss in both follow-up cohorts.
Changes in Longitudinal Weight
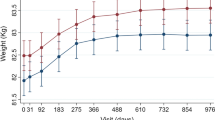
In the 1-year follow-up cohort, participants with the greatest weight loss, i.e., ≥ 15%, had a 23% (25.9 kg) reduction in weight from the index weight date (Fig. 1a). A similar pattern was observed in the 5-year follow-up cohort, that is, participants with ≥ 15% weight loss had a 22% (22.9 kg) reduction in weight from the index weight date (Fig. 1b). Overall, participants with numerically greater weight loss in follow-up tended to have a higher index weight in both follow-up cohorts.
Association Between Weight Loss and Glycemic Parameters
During both 1-year and 5-year follow-up periods, the largest decrease in HbA1c was observed among participants who lost ≥ 15% of their index weight: 1.2% and 0.5%, respectively (Fig. 2a, b). The percentage of participants with HbA1c < 7% at weight index date was relatively similar across weight-change categories (Fig. 3). The percentage of participants who attained HbA1c < 7% in the 1-year follow-up cohort was numerically highest among participants with ≥ 10% to < 15% and ≥ 15% weight loss (74% and 77%, respectively, vs. 48–65% for the remaining weight-change categories). A similar pattern was observed for the 5-year follow-up cohort, that is, 56% and 64%, respectively, vs. 40–50% for the remaining weight-change categories. Overall, increasing weight loss percentages were associated with numerically greater reductions in HbA1c levels and a higher proportion of participants achieving HbA1c targets.
In the 1-year follow-up cohort, those who lost ≥ 15% of their index weight had the largest mean reduction in FBG levels (26.3 mg/dL; Fig. 4a). However, in the 5-year follow-up cohort, a mean increase of 3.6 mg/dL was observed within this category (Fig. 4b).
Association Between Weight Change and Metabolic Parameters
In the 1-year follow-up cohort, reductions in LDL (5.4–9.6 mg/dL) and total cholesterol (6.4–13.8 mg/dL) plateaued beyond ≥ 10% weight loss from the index weight date (Fig. 5a, b). In addition, the weight-change categories in the 5-year follow-up cohort (Fig. 6a, b) revealed a similar plateauing effect beyond ≥ 10% weight loss for LDL (9.9–11.9 mg/dL) and total cholesterol (11.6–15.0 mg/dL). Numerically greater improvements in HDL, triglycerides, ALT, and AST levels were associated with increasing weight loss percentages, among both 1-year and 5-year follow-up cohorts.
The increase from the index date in HDL ranged from 0.4 to 5.0 mg/dL in the 1-year cohort and 0.6–4.5 mg/dL in the 5-year follow-up cohort (Figs. 5c, 6c). The reduction from the index date in triglycerides ranged from 5.3 to 53.0 mg/dL in the 1-year cohort and 0.3–40.4 mg/dL in the 5-year follow-up cohorts (Figs. 5d, 6d). The level of HDL cholesterol was lower than baseline (an undesirable change) among participants who had a weight increase of > 3% at both 1-year and 5-year follow-up. Total reduction in liver enzymes from index date to follow-up ranged from 0.4 to 9.7 IU/L in both follow-up cohorts (Figs. 5e, f, 6e, f).
Predictors of Weight Loss
A total of 1,049,618 and 307,455 people were categorized into the five index BMI categories for the 1-year and 5-year follow-up cohorts, respectively (Fig. 7a, b). Index BMI data for the remaining participants were unknown or < 18.5 kg/m2 and are not included in this analysis. The percentage of participants who experienced ≥ 15% weight loss in the 1-year follow-up cohort increased from 0.6% to 4.2% as the index BMI increased. Similarly, this percentage increased from 2.3% to 11.0% in the 5-year follow-up cohort.
The multivariable logistic regression analysis indicated that among all BMI categories, BMI ≥ 35 kg/m2 was the strongest predictor of ≥ 15% weight loss in the 1-year and 5-year follow-up cohorts (1-year follow-up cohort: OR 4.8, 95% CI 4.4–5.3; 5-year follow-up cohort: OR 5.3, 95% CI 4.9–5.8; p < 0.001 for both; Table 2). All other covariates held constant, the strongest predictor of ≥ 15% and ≥ 10% weight loss in both follow-up cohorts was the presence of bariatric surgery (1-year follow-up cohort: OR 17.0, 95% CI 15.8–18.3 and OR 9.6, 95% CI 8.9–10.2, respectively; 5-year follow-up cohort: OR 8.5, 95% CI 7.7–9.4 and OR 4.9, 95% CI 4.5–5.4, respectively; p < 0.001 for all).
Additionally, female sex and the use of antidepressants were factors that were positively associated with weight loss in both follow-up cohorts. In the 1-year follow-up cohort, the use of weight loss medications, sulfonylureas, and dipeptidyl peptidase 4 (DPP4) inhibitors were significantly associated with ≥ 10% and ≥ 15% weight loss. However, this association was not significant in the 5-year follow-up cohort, except for DPP4 inhibitors in the ≥ 15% weight loss category (Table 2). The use of GLP-1 RA was not associated with either ≥ 10% or ≥ 15% weight loss. The combined effect of sex, BMI, and age (as interactive predictors) on weight loss was also evaluated, and it was observed that women aged < 65 years with BMI ≥ 35 kg/m2 were significantly more likely to achieve ≥ 10% and ≥ 15% weight loss in the 1-year follow-up cohort. In the 5-year follow-up cohort, women aged > 75 years with BMI ≥ 35 kg/m2 had the highest likelihood of weight loss among all evaluated interactive predictors. Data for all interactive predictors of ≥ 10% and ≥ 15% weight loss are provided in Supplementary Table S2.
Discussion
To date, most studies have reported only the short-term effects of weight loss in people with T2D and overweight or obesity, most of them in relation to specific antihyperglycemic treatment classes [2, 24,25,26]. Studies examining the long-term effect of weight loss in this population are limited. The current study aimed to assess the long-term effects of weight loss among people with T2D in the USA using real-world data derived from an EMR database and linked health care claims data. It is noteworthy that the improvements in glycemic and metabolic parameters observed in the study population are independent of antihyperglycemic medications used.
Association Between Weight Loss and Glycemic Parameters
HbA1c remained consistent across the weight-change categories at the index date and subsequently decreased along the categories with increasing weight loss in both 1-year and 5-year follow-up cohorts. The higher the weight loss, the greater the improvement in HbA1c. There was an incremental increase in the percentage of participants with HbA1c < 7.0% in the highest weight loss category in the 1-year follow-up cohort. In line with our findings, Gummesson et al. [7] reported a direct association between weight loss and HbA1c reduction with a 0.1% reduction in mean HbA1c with every 1 kg of weight lost. In addition, the study reported that similar degrees of weight loss resulted in greater HbA1c reduction in individuals with poor glycemic control than their counterparts with better glycemic control at baseline. Further, in a retrospective cohort study, Shantha et al. [27] reported an intentional weight loss of 4.5%, 8.7%, and 10.3% resulting in a corresponding reduction in HbA1c levels by 0.5%, 1.0%, and 1.5% at 5.6, 8.7, and 10.1 months, respectively. In contrast, it was observed in our study that the improvement in glycemic control resulting from weight loss in the 5-year follow-up cohort was less than that in the 1-year follow-up cohort across the weight-change categories. Also, a modest weight loss of 5–10% in the 5-year follow-up cohort did not translate into a clinically meaningful reduction in HbA1c. This underscores the fact that weight loss alone, despite being a critical component, does not bring about glycemic control, as diabetes involves a complex interplay of various physiological, therapeutic, and lifestyle factors [28] that might have varied over the course of the study period.
Association Between Weight Loss and Metabolic Parameters
A weight loss of ≥ 10% resulted in greater improvements in LDL and total cholesterol from the index date to the 1-year follow-up. This finding is consistent with that of Wing et al. [6], which showed greater odds of clinically significant improvements in lipid profiles in individuals with a weight loss of 10–15% of body weight. There was an improvement in LDL levels in the 1-year follow-up cohort that persisted across the weight-change categories in the 5-year follow-up cohort. The trend observed in LDL level agrees with the Action for Health in Diabetes (Look AHEAD) study that reported a positive association between the magnitude of weight loss and improvement in lipids, except for LDL [10]. Furthermore, HDL and triglyceride levels also improved with increasing categorical weight loss in the 1-year and 5-year follow-up cohorts. Blonde et al. [29] reported highest improvement in liver enzymes with greater reductions in body weight with exenatide once weekly therapy. In the current study, regardless of antihyperglycemic medications, a similar trend was observed in AST and ALT levels across the weight-change categories in both 1-year and 5-year cohorts.
Predictors of Weight Loss
In the current study, the highest weight loss was noted in participants in the higher index BMI categories. These findings corroborate those of Raynor et al. [30] who assessed the effect of intensive lifestyle interventions on intentional weight loss in relation to baseline BMI in people with T2D and overweight and suggested that a greater amount of weight loss was associated with a higher baseline BMI. However, Deng et al. [24] reported that baseline BMI had no effect on weight loss in people on exenatide monotherapy. Further, in a study conducted by Bonora et al. [31] to evaluate the impact of dulaglutide on weight loss in people with T2D, it was reported that those with higher baseline BMI experienced numerically greater absolute weight loss. However, the mean percentage weight loss between the BMI subgroups was similar, indicating that weight loss is independent of baseline BMI.
A greater percentage of participants experiencing a weight loss of ≥ 15% of index weight in the 1-year and 5-year follow-up cohorts were reported to have undergone bariatric surgery, which is commonly recommended for the effective remission and prevention of T2D in adults with a higher BMI showing no sustained weight loss and improvement in comorbidities with nonsurgical methods [8, 32, 33].
An increased likelihood of achieving a weight loss of ≥ 10% and ≥ 15% was observed among those receiving antidepressants, DPP4 inhibitors, and sulfonylureas at the 1-year follow-up and those receiving insulin at the 5-year follow-up, in contrast to the usual weight loss or weight gain effects of these drugs [34, 35], warranting further investigation. GLP-1 RA use was noted in 5–7% of the participants; however, it was not associated with weight loss in both cohorts. This may be linked to the approved doses of GLP-1RAs at the time of this study, which might have been insufficient to facilitate substantial degrees of weight loss. Anti-obesity medications were found to be significant predictors of weight loss in the 1-year follow-up cohort but not in the 5-year follow-up cohort. This could be attributed to the fact that to achieve and sustain weight loss and manage obesity in the long run, it should be treated as a chronic condition, warranting continued treatment. Often these medications should be given in combination with adjuvant lifestyle or behavioral-based interventions [36], which emphasizes the need for a shift in the treatment paradigm of chronic management of obesity and thus novel anti-obesity medications that can produce sustained weight loss in the long run. Another reason for the lack of significant predictors of weight loss in the 5-year follow-up cohort could be the limited number of claims on the long-term use of anti-obesity medications in our study cohorts.
This study highlights some clinically meaningful implications of weight loss on long-term outcomes in people with T2D and overweight or obesity through analysis of real-world data in the USA. These findings reinforce the importance of weight management in people with T2D and provide additional information to support clinical decision-making. Furthermore, assessing predictors that influence weight loss can help mitigate disease burden.
Limitations
Retrospective database studies are subject to several limitations, and the findings of this study should be viewed in the context of such limitations. There is a possibility of missing diagnosis data as a result of either physician choice or lack of reporting by the patient. Also, the information in the EMR database on the date of onset of T2D and medication adherence is limited. The patient records came from a well-insured population; therefore, the reported results may not be generalizable to the entire US population with T2D. In addition, other demographics variables that potentially affect glycemic control, such as race/ethnicity, region, socioeconomic status, or duration of diabetes, were not available for this analysis. Finally, the findings of this study indicate associations but not causation.
Conclusion
The key findings of this study showed that participants with a higher index BMI (≥ 30 kg/m2) had greater weight loss percentages and the subsequent largest reduction in HbA1c. Modest and sustained weight loss resulted in improved metabolic parameters, which in turn could translate into better cardiometabolic outcomes. In light of the results highlighting the positive effects of weight loss in people with T2D, health care providers may consider personalizing weight-centric approaches for better management of T2D for their patients.
Data Availability
All data generated or analyzed during this study are included in this published article/as supplementary information files.
References
CDC. National Diabetes Statistics Report, 2022. 2022 [updated Oct 25, 2022]. https://www.cdc.gov/diabetes/data/statistics-report/index.html. Accessed Jan 31, 2023.
Brown E, Wilding JPH, Barber TM, Alam U, Cuthbertson DJ. Weight loss variability with SGLT2 inhibitors and GLP-1 receptor agonists in type 2 diabetes mellitus and obesity: mechanistic possibilities. Obes Rev. 2019;20(6):816–28. https://doi.org/10.1111/obr.12841.
Franz MJ, Boucher JL, Rutten-Ramos S, VanWormer JJ. Lifestyle weight-loss intervention outcomes in overweight and obese adults with type 2 diabetes: a systematic review and meta-analysis of randomized clinical trials. J Acad Nutr Diet. 2015;115(9):1447–63. https://doi.org/10.1016/j.jand.2015.02.031.
Garber AJ, Abrahamson MJ, Barzilay JI, et al. Consensus statement by the American Association of Clinical Endocrinologists and American College of Endocrinlogy on the comprehensive type 2 diabetes management algorithm—2018 executive summary. Endocr Pract. 2018;24(1):91–120. https://doi.org/10.4158/cs-2017-0153.
Wing RR. Long-term effects of a lifestyle intervention on weight and cardiovascular risk factors in individuals with type 2 diabetes mellitus: four-year results of the Look AHEAD trial. Arch Intern Med. 2010;170(17):1566–75. https://doi.org/10.1001/archinternmed.2010.334.
Wing RR, Lang W, Wadden TA, et al. Benefits of modest weight loss in improving cardiovascular risk factors in overweight and obese individuals with type 2 diabetes. Diabetes Care. 2011;34(7):1481–6. https://doi.org/10.2337/dc10-2415.
Gummesson A, Nyman E, Knutsson M, Karpefors M. Effect of weight reduction on glycated haemoglobin in weight loss trials in patients with type 2 diabetes. Diabetes Obes Metab. 2017;19(9):1295–305. https://doi.org/10.1111/dom.12971.
ElSayed NA, Aleppo G, Aroda VR, et al. 8. Obesity and weight management for the prevention and treatment of type 2 diabetes: standards of care in diabetes—2023. Diabetes Care. 2022;46(Suppl_1):S128–S39.https://doi.org/10.2337/dc23-S008
Schroeder R, Harrison TD, McGraw SL. Treatment of adult obesity with bariatric surgery. Am Fam Physician. 2016;93(1):31–7.
Madsen LR, Baggesen LM, Richelsen B, Thomsen RW. Effect of Roux-en-Y gastric bypass surgery on diabetes remission and complications in individuals with type 2 diabetes: a Danish population-based matched cohort study. Diabetologia. 2019;62(4):611–20. https://doi.org/10.1007/s00125-019-4816-2.
Panunzi S, De Gaetano A, Carnicelli A, Mingrone G. Predictors of remission of diabetes mellitus in severely obese individuals undergoing bariatric surgery: do BMI or procedure choice matter? A meta-analysis. Ann Surg. 2015;261(3):459–67. https://doi.org/10.1097/sla.0000000000000863.
Aktas G, Kocak MZ, Taslamacioglu Duman T, et al. Mean platelet volume (MPV) as an inflammatory marker in type 2 diabetes mellitus and obesity. Bali Med J. 2018. https://doi.org/10.15562/bmj.v7i3.806.
Bakhshimoghaddam F, Razmi H, Malihi R, Mansoori A, Ahangarpour A. The association between the dietary inflammatory index and gestational diabetes mellitus: a systematic review of observational studies. Clin Nutr ESPEN. 2023;57:606–12. https://doi.org/10.1016/j.clnesp.2023.08.007.
Rohm TV, Meier DT, Olefsky JM, Donath MY. Inflammation in obesity, diabetes, and related disorders. Immunity. 2022;55(1):31–55. https://doi.org/10.1016/j.immuni.2021.12.013.
Tsalamandris S, Antonopoulos AS, Oikonomou E, et al. The role of inflammation in diabetes: current concepts and future perspectives. Eur Cardiol. 2019;14(1):50–9. https://doi.org/10.15420/ecr.2018.33.1.
Athyros VG, Doumas M, Imprialos KP, et al. Diabetes and lipid metabolism. Hormones. 2018;17(1):61–7. https://doi.org/10.1007/s42000-018-0014-8.
Liu C, Shao M, Lu L, Zhao C, Qiu L, Liu Z. Obesity, insulin resistance and their interaction on liver enzymes. PLoS ONE. 2021;16(4):e0249299. https://doi.org/10.1371/journal.pone.0249299.
Chen S-B, Lee Y-C, Ser K-H, et al. Serum C-reactive protein and white blood cell count in morbidly obese surgical patients. Obes Surg. 2009;19(4):461–6. https://doi.org/10.1007/s11695-008-9619-3.
Martos R, Valle M, Morales RM, Cañete R, Gascón F, Urbano MM. Changes in body mass index are associated with changes in inflammatory and endothelial dysfunction biomarkers in obese prepubertal children after 9 months of body mass index SD score loss. Metabolism. 2009;58(8):1153–60. https://doi.org/10.1016/j.metabol.2009.03.017.
Orea Soler I, Illán Gómez F, Gonzálvez Ortega M, et al. Soluble intercellular adhesion molecule-1 and C reactive protein after bariatric surgery. Endocrinol Nutr. 2010;57(3):90–4. https://doi.org/10.1016/j.endonu.2010.01.008.
Davies MJ, Aroda VR, Collins BS, et al. Management of hyperglycaemia in type 2 diabetes, 2022. A consensus report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetologia. 2022;65(12):1925–66. https://doi.org/10.1007/s00125-022-05787-2.
Charlson ME, Charlson RE, Peterson JC, Marinopoulos SS, Briggs WM, Hollenberg JP. The Charlson comorbidity index is adapted to predict costs of chronic disease in primary care patients. J Clin Epidemiol. 2008;61(12):1234–40. https://doi.org/10.1016/j.jclinepi.2008.01.006.
Young BA, Lin E, Von Korff M, et al. Diabetes complications severity index and risk of mortality, hospitalization, and healthcare utilization. Am J Manag Care. 2008;14(1):15–23.
Deng H, Lin S, Yang X, et al. Effect of baseline body mass index on glycemic control and weight change with exenatide monotherapy in Chinese drug-naïve type 2 diabetic patients. J Diabetes. 2019;11(7):509–18. https://doi.org/10.1111/1753-0407.12883.
Horton ES, Silberman C, Davis KL, Berria R. Weight loss, glycemic control, and changes in cardiovascular biomarkers in patients with type 2 diabetes receiving incretin therapies or insulin in a large cohort database. Diabetes Care. 2010;33(8):1759–65. https://doi.org/10.2337/dc09-2062.
Rondanelli M, Perna S, Astrone P, Grugnetti A, Solerte SB, Guido D. Twenty-four-week effects of liraglutide on body composition, adherence to appetite, and lipid profile in overweight and obese patients with type 2 diabetes mellitus. Patient Prefer Adher. 2016;10:407–13. https://doi.org/10.2147/PPA.S97383.
Shantha GP, Kumar AA, Kahan S, Cheskin LJ. Association between glycosylated hemoglobin and intentional weight loss in overweight and obese patients with type 2 diabetes mellitus: a retrospective cohort study. Diabetes Educ. 2012;38(3):417–26. https://doi.org/10.1177/0145721712443293.
Van Gaal L, Scheen A. Weight management in type 2 diabetes: current and emerging approaches to treatment. Diabetes Care. 2015;38(6):1161–72. https://doi.org/10.2337/dc14-1630.
Blonde L, Pencek R, MacConell L. Association among weight change, glycemic control, and markers of cardiovascular risk with exenatide once weekly: a pooled analysis of patients with type 2 diabetes. Cardiovasc Diabetol. 2015;14(1):12. https://doi.org/10.1186/s12933-014-0171-2.
Raynor HA, Jeffery RW, Ruggiero AM, Clark JM, Delahanty LM, Look AHEAD (Action for Health in Diabetes) Research Group. Weight loss strategies associated with BMI in overweight adults with type 2 diabetes at entry into the look AHEAD (Action for Health in Diabetes) Trial. Diabetes Care. 2008;31(7):1299–304. https://doi.org/10.2337/dc07-2295.
Bonora E, Frias JP, Tinahones FJ, et al. Effect of dulaglutide 3.0 and 4.5 mg on weight in patients with type 2 diabetes: exploratory analyses of AWARD-11. Diabetes Obes Metab. 2021;23(10):2242–50. https://doi.org/10.1111/dom.14465.
Adams TD, Davidson LE, Litwin SE, et al. Weight and metabolic outcomes 12 years after gastric bypass. N Engl J Med. 2017;377(12):1143–55. https://doi.org/10.1056/NEJMoa1700459.
Qi QYD, Playfair J, Brown WA, Burton P, O’Brien PE, Wentworth JM. Long-term impact of weight loss for people with overweight but not obesity, and with type 2 diabetes: ten-year outcomes of a randomised trial of gastric band surgery. Diabetes Obes Metab. 2023. https://doi.org/10.1111/dom.14992.
Apovian CM, Okemah J, O’Neil PM. Body weight considerations in the management of type 2 diabetes. Adv Ther. 2019;36(1):44–58. https://doi.org/10.1007/s12325-018-0824-8.
Ghusn W, Hurtado MD, Acosta A. Weight-centric treatment of type 2 diabetes mellitus. Obes Pillars. 2022;4: 100045. https://doi.org/10.1016/j.obpill.2022.100045.
Tak YJ, Lee SY. Anti-obesity drugs: long-term efficacy and safety: an updated review. World J Mens Health. 2021;39(2):208–21.
Acknowledgements
We are grateful for the participants and their parents/guardian, without whom this research could not have been done.
Medical Writing, Editorial, and Other Assistance
We thank Meena Ravuri and Mythili Ananth of Eli Lilly and Company for providing medical writing and editorial support throughout the manuscript (funded by Eli Lilly Services India Private Limited). We thank Brian Calingaert of RTI health solutions for his assistance with the data analysis.
Funding
This study and Rapid Service Fee were funded by Eli Lilly and Company.
Author information
Authors and Affiliations
Contributions
Shraddha Shinde and Vivian Thuyanh Thieu conceptualized the work. Juliana Meyers contributed to acquisition of data. Anita YM Kwan, Katherine Houghton and David Schapiro were involved in the analysis of data. Vivian Thuyanh Thieu was involved in drafting the work. All authors contributed to design of the work, data interpretation and the critical revision of the work for important intellectual content.
Corresponding author
Ethics declarations
Conflict of interest
Shraddha Shinde, Vivian Thuyanh Thieu, Anita YM Kwan and David Schapiro are employees and shareholders of Eli Lilly and Company. Katherine Houghton and Juliana Meyers are employees of RTI Health Solutions. RTI Health Solutions received funding from Eli Lilly and Company to conduct this study.
Ethical approval
This study was conducted in accordance with the ethical principles that have their origin in the Declaration of Helsinki and that are consistent with Good Pharmacoepidemiology Practices and applicable laws and regulations of the country or countries where the study was conducted, as appropriate. Participants’ data were deidentified to protect privacy. A formal Consent to Release Information form was not required since this was an observational study that used previously collected data and did not impose any form of intervention.
Additional information
Prior Presentation: A portion of the results included in this manuscript was previously presented as poster presentations at the American Diabetes Association’s 82nd Scientific Sessions and the Obesity Society’s ObesityWeek 2022 conference. The corresponding abstracts were published in the journals Diabetes and Obesity: (a) The relationship between weight loss and HbA1c in people with type 2 diabetes. Diabetes 1 June 2022;71(Suppl_1):952–P. (b) Special Issue: Abstracts from the 40th Annual Meeting of the Obesity Society at ObesityWeek® November 1–4, 2022. Obesity. 21 November 2022;30(Suppl 1):55–293.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Shinde, S., Thieu, V.T., Kwan, A.Y.M. et al. Impact of Weight Change on Glycemic Control and Metabolic Parameters in T2D: A Retrospective US Study Based on Real-World Data. Diabetes Ther 15, 409–426 (2024). https://doi.org/10.1007/s13300-023-01511-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13300-023-01511-4