Abstract
Previous studies have demonstrated an association of the NC_000012.12:g.53962605A > G, (rs2366152) single-nucleotide variant (SNV) situated in the long noncoding homeobox transcript antisense intergenic RNA (HOTAIR) gene with HPV16-related cervical cancer pathogenesis. However, little is known about the role of rs2366152 in cervical cancer progression and how oral birth control pills use, parity, menopausal status, and cigarette smoking influence the role of rs2366152 in cervical carcinogenesis. HRM analysis was used to determine the rs2366152 SNV prevalence in patients with cervical squamous cell carcinoma (SCC) (n = 470) and control group (n = 499) in a Polish Caucasian population. Logistic regression analyses were adjusted for age, using birth control pills, parity, menopausal status, and cigarette smoking. Our genetic studies revealed that the G/A vs. A/A (p = 0.031, p = 0.002) and G/A + G/G vs. A/A (p = 0.035, p = 0.003) genotypes of rs2366152 SNV were significantly related to the grade of differentiation G3 and tumor stage III, respectively. Moreover, cervical cancer risk increased among patients with rs2366152 SNV who smoked cigarettes and used birth control pills. We conclude that rs2366152 may promote the invasion and rapid growth of cervical SCC. Moreover, rs2366152 with cigarette smoking and using birth control pills can also be a risk factor for cervical cancerogenesis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cervical cancer (CC) is the fourth cause of cancer-related death among women globally and the fourth most common cancer globally, with approximately 604,000 newly diagnosed cancer and 342,000 deaths in 2020 (Sung et al. 2021). In Poland, current estimates indicate 3862 cases of CC yearly and 2137 deaths. CC arrays 6th in prevalence among women in Poland and 3rd among women aged 15 to 44 (Poland, Human Papillomavirus and Related Cancers, Fact Sheet 2023).
CC is preceded by precursor lesions, classified histologically as cervical intraepithelial neoplasia (CIN) (Kornovski et al. 2021). CC develops slowly and does not cause any symptoms for many years; clear signs usually indicate an advanced stage that is difficult or almost impossible to cure (Fowler et al. 2023). The most common cervical cancer type is squamous cell carcinoma (SCC), which is responsible for about 80% of all cervical malignancies, while approximately 10% of cervical cancers are adenocarcinoma (Tanaka et al. 2021; Fowler et al. 2023). The remaining are rare types of cancer, such as adenosquamous carcinoma, carcinoid, and non-epithelial tumors (sarcomas) (Tanaka et al. 2021; Fowler et al. 2023). SCC is characterized by low histological differentiation, extensive local infiltration, rapid growth, and a tendency to metastasise to lymph nodes, lungs, bone, liver, and other organs (Butt and Botha 2019; Zhou and Peng 2020; Fowler et al. 2023).
Human papillomavirus (HPV), particularly types 16 and 18, is believed to be the leading etiological agent of cervical cancerogenesis (Gutierrez-Xicotencatl et al. 2021). The HPV oncogenic potential is determined mainly by two virus genes encoding proteins E6 and E7 (Tomaić 2016). These proteins in viruses with high oncogenic potential are called oncoproteins. E6 and E7 oncoproteins interact with host cell cycle regulators, disrupting their function and leading to genetic instability, malignant transformation, and tumor development (Tomaić 2016). Carcinogenesis in the cervix is a multi-complex process in which, apart from oncogenic viruses, hereditary and environmental factors are also involved (Maciag and Villa 1999; Bowden et al. 2021). CC is a cancer whose development has a significant genetic background (Bowden et al. 2021). The disease risk is much higher in women with a first-degree relative with CC (Zoodsma et al. 2004). The genetic factors influencing CC development can be potentiated by coinfection with the human immunodeficiency virus or type 2 herpes simplex virus, high parity, pollutants, cigarette smoking, long-term, birth control pills use, and menopause status (Quinlan 2021; Tekalegn, et al. 2022).
The largest genome-wide association study (GWAS) meta-analysis recently revealed that the GSDMB, HLA-DRB1, LINC00339, CLPTM1L, CDC42, and PAX8/PAX8-AS1 genes related to the immune response, growth of reproductive tract, and apoptosis/proliferation play a significant role in the development of CC (Koel et al. 2023). Various previously studied gene variants did not achieve the GWAS significance threshold. However, they may affect the progression and invasion of cervical malignancies (Bahrami et al. 2018).
Previous studies demonstrated an association of the NC_000012.12:g.53962605A > G, (rs2366152) single-nucleotide variant (SNV) with HPV16-related cervical cancer pathogenesis (Saha et al. 2016). The rs2366152 is located in the long noncoding homeobox transcript antisense intergenic RNA (HOTAIR) gene, which is involved in developing many cancers (Liu et al. 2020).
The influence of rs2366152 in cervical malignancies progression is still unclear. Little is known about how menopausal status, birth control pills use, parity, and cigarette smoking modify the influence of rs2366152 in the development of CC.
We determined the rs2366152 SNV distribution in women with cervical SCC in the Caucasian population from Poland. We also determined the frequency of rs2366152 in the differentiation grades of SCC and stages I–IV. The distribution of rs2366152 genotypes was also assessed in patients and controls subdivided into groups of menopausal status birth control pills use, parity, and cigarette smoking.
Materials and methods
Cases and control group
There were 470 patients with established cervical SCC and 499 healthy women controls enrolled in this study.
The patient’s grade of differentiation and stage was assessed based on the International Federation of Gynecology and Obstetrics (FIGO) classification system and the World Health Organization (WHO) (Saleh et al. 2020) (Table 1). Clinical characteristics of patients were gained from cases enrolled between May 2016 and January 2021 at the Department of Radiotherapy of the Greater Poland Cancer Center in Poznań, Poland. The controls, healthy women were arbitrarily obtained during medical examinations at the Heliodor Swiecicki Clinical Hospital, Poznan, Poland (Table 1). Data on active cigarette smoking within the last 12 months, birth control pills use, menopausal status, and parity were collected from cases and healthy women. Both cervical SCC patients and healthy controls were Caucasian individuals from Poland. All participants provided written and oral consent. The research methodology was approved by the Local Ethical Committee of the Poznań University of Medical Sciences (reference number of ethical approval: 1010/07).
Methods
Genotype determination
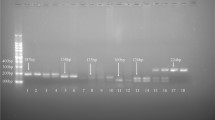
DNA was obtained from peripheral blood leucocytes via salting out cellular proteins. The primers were designated employing Beacon Designer 7.9 PREMIER Biosoft International San Francisco, CA. The 160 bp DNA fragment including the A > G rs2366152 SNV was amplified using the primers (forward 5′ TATATTCTGTGAGTTGTGTT 3′ and reverse 5′ ATCAGCAGATGGAGATTA′). The genotype of rs2366152 was determined via high-resolution melting (HRM) curve analysis using 5 × HOT FIREPol EvaGreen Mix no ROX (Solis BioDyne, Tartu, Estonia) with a LightCycler 480 system (Roche Diagnostics, Mannheim, Germany). Randomly, 10% of this variant’s patients and healthy control samples were checked by Sanger sequencing analyses. The agreement rate between HRM and Sanger sequencing results was 100%.
Statistical assessment
The genotype frequency in the women with cervical SCC and healthy controls and their genotype departure from Hardy-Weinberg (HW) equilibrium were assessed using a chi-squared test. The rs2366152 SNV was evaluated for risk factors of cervical SCC using the Cochran-Armitage p-trend test (ptrend). The chi-squared and Fisher exact tests were employed to find the divergencies in genotypic frequencies between the women with cervical SCC and healthy controls. The odds ratio (OR) and 95% confidence intervals (95% CI) were also determined. A logistic regression analysis was employed to adjust for menopausal status, using birth control pills, parity, tobacco smoking, and age. A p value greater than 0.05 means that no effect was observed.
Results
Prevalence of the rs2366152 SNV among all cases and healthy controls
The chi-squared test values of HW equilibrium were 0.052 and 0.057 for the patients and healthy controls, respectively. The assessment of the rs2366152 genotype presence in patients and controls is stated in Table 2. We did not find the statistical value ptrend = 0.850 calculated for the rs2366152 with all patients. However, the logistic regression analysis adjusted for the menopausal status, birth control pills use, parity, cigarette smoking, and age showed an influence of rs2366152 on the development of cervical SCC (Table 2). For G/A vs. A/A, the adjusted OR was 1.637 (95% CI 1.220–2.196, p = 0.001); for G/A + G/G vs. A/A, the adjusted OR was 1.522 (95% CI 1.153–2.010, p = 0.003). However, for G/G vs. A/A, we did not show a contribution of rs2366152; the adjusted OR was 1.168 (95% CI 0.704–1.935, p = 0.548).
The rs2366152 genotype differences among patients with differentiation grades and different tumor stages
On stratifying cases according to differentiation grade and tumor stage, we showed the influence of rs2366152 in grade G3 and stage III (Table 2). In patients with stage III, the p-trend value assessed for the rs2366152 was not statistically significant (ptrend = 0.625). However, the logistic regression analysis, adjusting for the menopausal status, birth control pills use, parity, cigarette smoking, and age in patients with stage III, showed a contribution of rs2366152 to cervical malignancies (Table 2). For G/A vs. A/A, the adjusted OR was 5.248 (95% CI 2.742–10.05, p = 0.002); for G/A + G/G vs. A/A, the adjusted OR was 4.311 (95% CI 2.473–7.516, p = 0.003). However, we did not show a statistical contribution of rs2366152 for G/G vs. A/A, and the adjusted OR was 2.012 (95% CI 0.943–4.291, p = 0.071) (Table 2).
In patients with differentiation grade G3, the p-trend value assessed for rs2366152 SNV was not statistically significant (ptrend = 0.940). However, in the same patient group, after adjusting for confounders, we showed the G/A vs. A/A genotype as risk cervical SCC, with an adjusted OR = 2.126 (95% CI = 1.601–4.264, p = 0.031) as well as for G/A + G/G vs. A/A with an adjusted OR = 1.929 (95% CI = 1.048–3.549, p = 0.035) (Table 2). In this group of patients, we did not show the risk effect of the G/G vs. A/A genotype with an adjusted OR = 1.027 (95% CI = 0.395–2.667, p = 0.357).
The logistic regression analysis did not indicate the contribution of rs2366152 to tumor stages I, II, and IV and the grade of differentiation grades G1, G2, and GX (Table 2).
The frequency of rs2366152 among women with cervical SCC and the control group with menopausal status, a history of birth control pills use, parity, and cigarette smoking
Stratification of patients for confounders showed the contribution of rs2366152 to patients with cigarette smoking and birth control pills (Table 3).
In patients with cigarette smoking, we observed an influence of rs2366152 with cervical SCC development. The age standardization OR for women with cigarette smoking for G/G vs. A/A was 3.023 (95% CI = 1.448–6.311, p = 0.015), for G/A vs. A/A was 2.178 (95% CI = 1.758–2.833, p = 0.043), and for A/G + G/G vs. A/A, the age standardization OR was 10.490 (95% CI = 5.322–20.67, p = 0.001).
The age standardization OR for women with birth control pills use for G/A vs. A/A was 1.627 (95% CI = 1.092–2.422, p = 0.017), and for A/G + G/G vs. A/A, the age standardization OR was 1.652 (95% CI = 1.127–2.424, p = 0.010). In this group, we did not show rs2366152 as a risk of cervical SCC for G/G vs. A/A was 1.894 (95% CI = 0.976–3.675, p = 0.059). We also did not find rs2366152 as a risk of cervical SCC in the patient’s group divided according to menopausal status and parity.
Discussion
HOTAIR is a non-protein-coding transcript longer than 200 nucleotides that belongs to an oncogenic-acting long noncoding RNA (lncRNA) (Tang and Hann 2018). HOTAIR is a trans-acting lncRNA, via interactions with various cellular partners, that governs gene expression processes on the epigenetic, transcriptional, or posttranscriptional level that regulate basic biochemical and cellular processes (Hajjari and Salavaty 2015; Tang and Hann 2018). HOTAIR is an oncogenic factor, and abnormal HOTAIR expressions are involved in the malignant transformation and development of at least 24 types of solid tumors (An and Liu 2022). HOTAIR lncRNA’s dysfunction promotes cancer cells’ proliferation, invasion, survival, metastasis, and drug resistance (He et al. 2022; Liang and Peng 2022; Wang et al. 2022). Deregulation of HOTAIR lncRNA expression can be a biomarker of poor prognosis and cancer progression for several solid tumors such as ovarian, gastric, and lung cancer (Shehata et al. 2020; Xin et al. 2021). Increased transcription of HOTAIR in various cancerous tissues is accompanied by shorter overall survival and disease-free survival, metastasis, and tumor resistance to chemo/radiotherapy (Tang et al. 2019; Lu et al. 2018; Raju et al. 2023). Recent studies revealed many HOTAIR sequence variations, mainly including SNVs in the HOTAIR gene (Liu et al. 2020). These genetic HOTAIR variants influence the expression and function of HOTAIR (Li et al. 2017). Most of the SNVs identified in the HOTAIR gene are responsible for acquiring oncotic HOTAIR activity, which supports the development of various solid tumors (Qi et al. 2016; Tian et al. 2016; Ge et al. 2017; Li et al. 2018; Moazeni-Roodi et al. 2020; Janaththani et al. 2021).
To date, it has been demonstrated that several HOTAIR gene SNPs are associated with the appearance and progression of lung, ovarian, breast, and gastric cancers, as well as cervical cancer (Qiu et al. 2017; Jin et al. 2017; Wang et al. 2019; Moazeni-Roodi et al. 2020; Ke et al. 2022; Liu et al. 2022, Yalınbaş Kaya et al. 2022).
The three SNVs in the HOTAIR gene, rs7958904, rs920778, and rs4759314, have been reported as risk factors appearance of cervical cancer in the Chinese population (Jin et al. 2017; Liu et al. 2022). Moreover, it has also been demonstrated that lncRNAs HOTAIR SNP rs920778 can predict cancer recurrence and worse overall survival (Weng et al. 2018). Additionally, Saha et al. (2016) demonstrated rs2366152G over-presentation in low HOTAIR-expressing HPV-positive cervical cancer cases compared to HPV-negative controls.
Our studies did not find an association of rs2366152 with the diagnosis of cervical SCC. Our observations agree with other genetic studies in the Chinese population, which also failed to present rs2366152 as a genetic risk factor for cervical SCC (Liu et al. 2022).
However, our genetic studies demonstrated the contribution of lncRNAs HOTAIR SNP rs2366152 to grade differentiation G3 and stages III of cervical malignancies. These findings may indicate that the G variant of s2366152 promotes the invasion of cervical malignancies cells to neighboring tissues and increase tumor growth compared to lower-grade cervical malignancies.
Evaluating confounding variables such as age, birth control pill use, parity, cigarette smoking, and menopause demonstrated that some were correlated with HOTAIR SNP rs2366152 in the diagnosis of cervical SCC. We observed a contribution of rs2366152 to cervical SCC in women with a positive history of using birth control pills and cigarette smoking. Our observation confirms previously demonstrated tobacco smoking and using birth control pills as environmental factors, which, alone or with genetic factors, may modulate the risk for the appearance of cervical SCC (Quinlan 2021; Tekalegn et al. 2022).
Our investigation is the first to present rs2366152 SNV as a new genetic risk factor for cervical malignant transformation in Polish women with a positive history of using birth control pills and cigarette smoking. We also find that the rs2366152 G variant supports the growth and invasion of malignant cells to surrounding tissue. To reinforce the value of our observations, these genetic studies should also be performed in other populations.
References
An X, Liu Y (2022) HOTAIR in solid tumors: emerging mechanisms and clinical strategies. Biomed Pharmacother 154:113594. https://doi.org/10.1016/j.biopha.2022.113594
Bahrami A, Hasanzadeh M, Shahidsales S et al (2018) Genetic susceptibility in cervical cancer: from bench to bedside. J Cell Physiol 233:1929–1939. https://doi.org/10.1002/jcp.26019
Bowden SJ, Bodinier B, Kalliala I et al (2021) Genetic variation in cervical preinvasive and invasive disease: a genome-wide association study. Lancet Oncol 22:548–557. https://doi.org/10.1016/S1470-2045(21)00028-0
Butt JL, Botha MH (2019) Bone involvement in patients with cervical carcinoma: a single-institution cohort study. Southern Afr J Gynaecol Oncol 11:31–37. https://doi.org/10.1080/20742835.2019.1702299
Fowler JR, Maani EV, Dunton CJ, Jack BW (2023) Cervical cancer. StatPearls Publishing, Treasure Island (FL)
Ge Y, Jiang R, Zhang M et al (2017) Analyzing 37,900 samples shows significant association between HOTAIR polymorphisms and cancer susceptibility: a meta-analysis. Int J Biol Markers 32:e231–e242. https://doi.org/10.5301/jbm.5000235
Gutierrez-Xicotencatl L, Pedroza-Saavedra A, Chihu-Amparan L et al (2021) Cellular functions of HPV16 E5 oncoprotein during oncogenic transformation. Mol Cancer Res 19:167–179. https://doi.org/10.1158/1541-7786.MCR-20-0491
Hajjari M, Salavaty A (2015) HOTAIR: an oncogenic long non-coding RNA in different cancers. Cancer Biol Med 12:1–9. https://doi.org/10.7497/j.issn.2095-3941.2015.0006
He W, Li D, Zhang X (2022) LncRNA HOTAIR promotes the proliferation and invasion/metastasis of breast cancer cells by targeting the miR-130a-3p/Suv39H1 axis. Biochem Biophys Rep 30:101279. https://doi.org/10.1016/j.bbrep.2022.101279
Janaththani P, Srinivasan SL, Batra J (2021) Long non-coding RNAs at the chromosomal risk loci identified by prostate and breast cancer GWAS. Genes (basel) 12:2028. https://doi.org/10.3390/genes12122028
Jin H, Lu X, Ni J et al (2017) HOTAIR rs7958904 polymorphism is associated with increased cervical cancer risk in a Chinese population. Sci Rep 7:3144. https://doi.org/10.1038/s41598-017-03174-1
Ke C, Feng X, Li J et al (2022) Association between long non-coding RNA HOTAIR polymorphism and lung cancer risk: a systematic review and meta-analysis. Exp Ther Med 24:540. https://doi.org/10.3892/etm.2022.11477
Koel M, Võsa U, Jõeloo M et al (2023) GWAS meta-analyses clarify the genetics of cervical phenotypes and inform risk stratification for cervical cancer. Hum Mol Genet 32:2103–2116. https://doi.org/10.1093/hmg/ddad043
Kornovski Y, Slavchev S, Kostov S et al (2021) Precancerous lesions of the cervix — aetiology, classification, diagnosis, prevention. Oncol Clin Pract 17:271–276. https://doi.org/10.5603/OCP.2021.0027
Li H, Tang XM, Liu Y et al (2017) Association of functional genetic variants of HOTAIR with hepatocellular carcinoma (HCC) susceptibility in a Chinese population. Cell Physiol Biochem 44:447–454. https://doi.org/10.1159/000485011
Li J, Cui Z, Li H et al (2018) Long non-coding RNA HOTAIR polymorphism and susceptibility to cancer: an updated meta-analysis. Environ Health Prev Med 23:8. https://doi.org/10.1186/s12199-018-0697-0
Liang H, Peng J (2022) LncRNA HOTAIR promotes proliferation, invasion and migration in NSCLC cells via the CCL22 signaling pathway. PLoS ONE 17:e0263997. https://doi.org/10.1371/journal.pone.0263997
Liu X, Zhao Y, Li Y et al (2020) Association between HOTAIR genetic polymorphisms and cancer susceptibility: a meta-analysis involving 122,832 subjects. Genomics 112:3036–3055. https://doi.org/10.1016/j.ygeno.2020.05.018
Liu Y, Zhang Q, Ni R (2022) Association between genetic variants (rs920778, rs4759314, and rs217727) in LncRNAs and cervical cancer susceptibility in Chinese population: a systematic review and meta-analysis. Front Genet 13:988207. https://doi.org/10.3389/fgene.2022.988207
Lu R, Zhang J, Zhang W et al (2018) Circulating HOTAIR expression predicts the clinical response to neoadjuvant chemotherapy in patients with breast cancer. Cancer Biomark 22:249–256. https://doi.org/10.3233/CBM-170874
Maciag PC, Villa LL (1999) Genetic susceptibility to HPV infection and cervical cancer. Braz J Med Biol Res 32:915–922. https://doi.org/10.1590/S0100-879X1999000700017
Moazeni-Roodi A, Aftabi S, Sarabandi S et al (2020) Genetic association between HOTAIR gene and the risk of cancer: an updated meta-analysis. J Genet 99:48. https://doi.org/10.1007/s12041-020-01214-w
Poland, Human Papillomavirus and Related Cancers, Fact Sheet (2023). https://hpvcentre.net/statistics/reports/POL_FS.pdf. Accessed 30 March 2023
Qi Q, Wang J, Huang B et al (2016) Association of HOTAIR polymorphisms rs4759314 and rs920778 with cancer susceptibility on the basis of ethnicity and cancer type. Oncotarget 7:38775–38784. https://doi.org/10.18632/oncotarget.9608
Qiu H, Wang X, Guo R et al (2017) HOTAIR rs920778 polymorphism is associated with ovarian cancer susceptibility and poor prognosis in a Chinese population. Future Oncol 13:347–355. https://doi.org/10.2217/fon-2016-0290
Quinlan JD (2021) Human papillomavirus: screening, testing, and prevention. Am Fam Phys 104:152–159
Raju GSR, Pavitra E, Bandaru SS et al (2023) HOTAIR: a potential metastatic, drug-resistant and prognostic regulator of breast cancer. Mol Cancer 22:65. https://doi.org/10.1186/s12943-023-01765-3
Saha SS, Roy Chowdhury R, Mondal NR et al (2016) Identification of genetic variation in the lncRNA HOTAIR associated with HPV16-related cervical cancer pathogenesis. Cell Oncol (Dordr) 39:559–572. https://doi.org/10.1007/s13402-016-0298-0
Saleh M, Virarkar M, Javadi S et al (2020) Cervical cancer: 2018 revised international federation of gynecology and obstetrics staging system and the role of imaging. AJR Am J Roentgenol 214:1182–1195. https://doi.org/10.2214/AJR.19.21819
Shehata AMF, Kamal Eldin SM, Osman NF, Helwa MA (2020) Deregulated expression of long non-coding RNA HOX transcript antisense RNA (HOTAIR) in Egyptian patients with multiple myeloma. Indian J Hematol Blood Transfus 36:271–276. https://doi.org/10.1007/s12288-019-01211-9
Sung H, Ferlay J, Siegel RL et al (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71:209–249. https://doi.org/10.3322/caac.21660
Tanaka LF, Schriefer D, Radde K et al (2021) Impact of opportunistic screening on squamous cell and adenocarcinoma of the cervix in Germany: a population-based case-control study. PLoS ONE 16:e0253801. https://doi.org/10.1371/journal.pone.0253801
Tang Q, Hann SS (2018) HOTAIR: an oncogenic long non-coding RNA in human cancer. Cell Physiol Biochem 47:893–913. https://doi.org/10.1159/000490131
Tang S, Zheng K, Tang Y et al (2019) Overexpression of serum exosomal HOTAIR is correlated with poor survival and poor response to chemotherapy in breast cancer patients. J Biosci 44:37. https://doi.org/10.1007/s12038-019-9861-y
Tekalegn Y, Sahiledengle B, Woldeyohannes D et al (2022) High parity is associated with increased risk of cervical cancer: systematic review and meta-analysis of case-control studies. Womens Health (Lond) 18:17455065221075904. https://doi.org/10.1177/17455065221075904
Tian T, Li C, Xiao J et al (2016) Quantitative assessment of the polymorphisms in the HOTAIR lncRNA and cancer risk: a meta-analysis of 8 case-control studies. PLoS ONE 11:e0152296. https://doi.org/10.1371/journal.pone.0152296
Tomaić V (2016) Functional roles of E6 and E7 oncoproteins in HPV-induced malignancies at diverse anatomical sites. Cancers (Basel) 8:95. https://doi.org/10.3390/cancers8100095
Wang B, Yuan F, Zhang F et al (2019) A systematic review and meta-analysis of the association between HOTAIR polymorphisms and susceptibility to breast cancer. Arch Med Sci 19:128–137. https://doi.org/10.5114/aoms.2019.87537
Wang BR, Chu DX, Cheng MY et al (2022) Progress of HOTAIR-microRNA in hepatocellular carcinoma. Hered Cancer Clin Pract 20:4. https://doi.org/10.1186/s13053-022-00210-8
Weng SL, Wu WJ, Hsiao YH et al (2018) Significant association of long non-coding RNAs HOTAIR genetic polymorphisms with cancer recurrence and patient survival in patients with uterine cervical cancer. Int J Med Sci 15:1312–1319. https://doi.org/10.7150/ijms.27505
Xin X, Li Q, Fang J, Zhao T (2021) LncRNA HOTAIR: a potential prognostic factor and therapeutic target in human cancers. Front Oncol 11:679244. https://doi.org/10.3389/fonc.2021.679244
Yalınbaş Kaya B, Peker B, Tuğrul F et al (2022) Association of HOTAIR rs1899663 G>T polymorphism with colorectal cancer in the Turkish population: a case-control study. Turk J Gastroenterol 33:689–695. https://doi.org/10.5152/tjg.2022.21836
Zhou S, Peng F (2020) Patterns of metastases in cervical cancer: a population-based study. Int J Clin Exp Pathol 13:1615–1623
Zoodsma M, Sijmons RH, de Vries EG, Zee AG (2004) Familial cervical cancer: case reports, review and clinical implications. Hered Cancer Clin Pract 2:99–105. https://doi.org/10.1186/1897-4287-2-2-99
Funding
The Poznań University of Medical Sciences supported this study.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study’s conception and design. Material preparation, data collection, and analysis were performed by SŁ, AS, AR, AP, and AM. The first draft of the manuscript was written by SŁ, PPJ, and ML, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Local Ethical Committee of the Poznań University of Medical (Date 2016/No 285 and 2016/No566).
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Competing interests
The authors no competing interests.
Additional information
Communicated by: Ewa Ziętkiewicz
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Łaźniak, S., Sowińska, A., Roszak, A. et al. Role of rs2366152 single-nucleotide variant located in the long noncoding RNA HOTAIR gene in the cervical cancer susceptibility in a Polish population. J Appl Genetics 65, 511–518 (2024). https://doi.org/10.1007/s13353-023-00822-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13353-023-00822-3