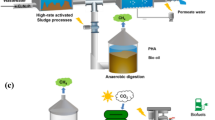
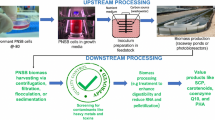
Abstract
Fruit juice industry wastewater is characterized by high chemical oxygen demand (COD) and low pH, which requires extensive energy if treated aerobically and can adversely affect aquatic organisms if discharged into water bodies without proper treatment. In this study, purple non-sulfur bacteria (PNSB) were employed to recover carbon through photoanaerobic treatment in the form of single-cell protein (SCP) suitable for aquaculture and rich in carotenoid and bacteriochlorophyll photopigments. The efficacy of PNSB was investigated using two types of wastewaters, citrus juice wastewater (CJW), and mixed juice wastewater (MJW) as substrates at different pH conditions (5.0–8.0). pH 8.0 showed the highest COD removal in CJW (89 ± 0.1%) and pH 7.0 in MJW (94 ± 1%), respectively. The biomass yields were 0.493 g-VSS·g-COD−1 and 0.397 g-VSS·g-COD−1 for CJW and MJW. SCP contents in CJW and MJW were 48 ± 1% and 40 ± 3%, respectively. Pigment analysis revealed that PNSB biomass contained carotenoids and bacteriochlorophyll. Carotenoids in CJW and MJW were 1354 ± 64 µg·g−1 and 1558 ± 218 µg·g−1 of biomass; bacteriochlorophyll contents were 2097 ± 110 µg·g−1 and 3100 ± 614 µg·g−1 of biomass, respectively. The microbial population showed diversity among the two substrates and pH conditions, with PNSB comprising potentially up to 51% of microbes in the culture. The results demonstrate that PNSB could be used as potential mediators to treat fruit juice wastewater, and produce SCP, and photopigments simultaneously.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
The fruit juice industry produces a large volume of wastewater through a series of sequential processes including washing, soaking, blanching, packing, and extraction [1]. These processes require plenty of fresh water which leads to the generation of excessive wastewater. Almost 10 L of wastewater is generated for 1 L of juice [2]. Fruit juice wastewater (FJW) is characterized by high chemical oxygen demand (COD), suspended solids, dissolved solids, low-nutrient (nitrogen and phosphorous) concentrations, and low pH [3]. Discharge of FJW without proper treatment can cause various environmental and ecological issues. Thus, FJW should be treated before discharging into the environment to meet regulatory standards [4, 5]. Several physicochemical methods such as flocculation, electro-coagulation, electro-oxidation, and membrane filtration have been developed to treat FJW [1, 6, 7]. However, their large-scale application is limited due to high-energy costs and a large volume of sludge generation.
Anaerobic biotreatment has been considered a mature technology to treat a variety of high-strength wastewaters including FJW. Anaerobic treatment of FJW is being recognized as an appropriate method as it is efficient and cost-effective [7]. Recent advancements have coupled this technology with single-cell protein (SCP) synthesis to drive additional economic benefits. SCP is a protein obtained from microbial biomass and can be used for human, animal, or aquaculture feed [8, 9]. SCP offers numerous environmental and ecological benefits over traditional protein sources, i.e., plants and animals [10]. Plant-based proteins lack essential amino acids such as lysine, cysteine, and methionine [11], while animal-based protein pollution associated with manure degradation and fertilizer supply is problematic. In contrast, nutrients are recovered and fixed in the biomass during the SCP synthesis process. Despite numerous advantages of SCP, their large-scale production is still limited due to high production costs.
In this perspective, purple non-sulfur bacteria (PNSB) can be used as potential mediators for the economical and sustainable production of SCP due to their unique metabolic features [12]. They are facultative anaerobes and can utilize complex organic materials as a carbon source. They can grow in imbalanced nutrient conditions and switch their growth mechanism between anoxyphotoheterotrophy and mixotrophic conditions to balance respiratory losses and govern high biomass productivity. The former photosynthetic mode of growth is their preferred metabolism and allows easy enrichment of this particular group of organisms in mixed cultures, avoiding costly sterilization and strain maintenance. Furthermore, PNSB biomass relative to many other microbial SCP sources contains value-added biomolecules such as carotenoids, bacteriochlorophyll, coenzyme Q10, and essential amino acids, which improve the nutritional value of SCP.
FJW contains a high concentration of organic matter, particularly sugars and organic acids, and its composition varies across different species of fruits. The concentration and types of natural organic compounds, preservatives, and other additives also vary between single-species fruit juice and mixed fruit juice [6]. FJW contains volatile fatty acids (VFAs), which are potentially used by PNSB as electron donors to carry out anoxygenic photosynthesis. FJW is characterized by acidic pH, but PNSB can grow across a wide pH range and naturally increase pH during photoheterotrophy. Thus, FJW could be considered a suitable substrate for PNSB.
This study aims to demonstrate the potential of PNSB to treat FJW and produce SCP simultaneously by evaluating their performance using citrus (orange) juice wastewater (CJW) and mixed fruit juice wastewater (MJW), as these are the first and third most consumed juice flavors [13] and provide significantly different juice composition. The effect of pH on PNSB growth and organic and inorganic removal was investigated. SCP, carotenoid, and bacteriochlorophyll contents were also determined in MJW and CJW. This is the first study to the authors’ knowledge involving PNSB for the treatment of FJW treatment.
2 Materials and methods
2.1 Wastewater collection and characterization
Wastewater samples were collected from a local fruit juice industry located in Qatar. The industry produces various juices including citrus and mixed fruit juices. The samples were collected in plastic bottles during the early flushing of the processing equipment and immediately stored at 4 °C. Major characteristics of the wastewater are given in Table 1.
2.2 PNSB growth conditions
Characterization showed that total COD (CODt) values of CJW and MJW were 6730 ± 141 mg·L−1 and 5590 ± 99 mg·L−1, respectively. Soluble COD (CODs) values were 5710 ± 28 and 5060 ± 14 mg·L−1 for CJW and MJW, respectively. The pH of CJW and MJW was 4.12 ± 0.05 and 3.85 ± 0.03, respectively. The literature review revealed that PNSB may not grow in undiluted FJW due to low pH and high concentrations of COD. Therefore, FJW was diluted with fresh water to neutralize the pH and bring down the COD value (1700–2000 mg·L−1) that could support PNSB growth [14,15,16]. Additional nutrients including NH4Cl, KH2PO4, vitamins, and trace metals as per media composition (ATCC-2672) [17] were added to the diluted FJW. Initial CODs values of CJW and MJW working solutions were 1950 ± 14 mg·L−1and 1703 ± 3 mg·L−1, respectively. The solutions were inoculated with a mixed culture of PNSB previously grown in the lab on another organic-rich industrial water source and their pH was adjusted to 5.0, 6.0, 7.0, and 8.0 by using 0.1 M NaOH and 0.1 M H2SO4. After pH adjustment, the solutions were transferred to 160-mL serum flasks. Nitrogen gas was purged for 2 min in the flasks to remove dissolved oxygen. The flasks were tightly sealed with a rubber stopper and aluminum rim by using a crimper. The flasks were incubated in an incubator shaker (Innova®44, New Brunswick) stirring continuously at 150 RPM at a temperature of 35 °C ± 1, under the illumination of 42 µmol·m−2·s−1. The illumination’s primary peak range was 640–660 nm. Periodic sampling was carried out from the flasks by removing the aluminum rim and rubber stopper. After sampling, the flasks were again purged with nitrogen, sealed, and kept in the incubator. PNSB growth was monitored by measuring absorbance using a UV–Vis spectrophotometer (UV-3600 Plus, Shimadzu). A full-spectrum (400–700 nm) absorbance test revealed that PNSB showed the maximum absorbance peak at 420 nm. Thus, this wavelength was selected to monitor their growth. Culture pH was measured by using a pH meter (Orion, Thermo Fisher Scientific). All analytical measurements were carried out in duplicates and reported with standard deviation.
2.3 Microbial community analysis
The 16 s rRNA were amplified using primers targeting the V2,4,8 and V3,6–7,9 variable regions from the Ion 16S™ Metagenomics Kit (as described by the manufacturer), using 1–3 ng of the microbial DNA. According to the manufacturer’s protocol, the amplified PCR products were extracted from a 2% agarose gel followed by purification using the AxyPrep DNA Gel Extraction Kit (Axygen BioScience, Inc., USA). Once the purified amplicons were pooled, Illumina adapters were added by ligation using the TruSeq DNA LT Sample Prep Kit. The amplicons were further amplified for 10 cycles to obtain adequate yield for sequencing. Finally, paired-end sequencing of the library was done on the MiSeq platform based on the standard protocol.
2.3.1 QIIME analysis
Reads obtained from the Illumina platform were paired-end (forward and reverse). Paired-end reads were “demultiplexed” using QIIME2 2019.10 [18]. Reads were quality filtered and denoised by truncating to 130 bps (base-pairs) and 290 bps to improve sequence quality, trimmed 13 bps and 20 bps to remove non-biological nucleotides like barcodes or linkers, and chimeras removed for both paired-end and single-end, respectively, and processed using QIIME2 “dada2” plugin to obtain the OTU (Operational Taxonomic Unit) representative sequences, read statistics, and OTU feature table files. The OTU representative sequences were assigned taxa based on SILVA 99% database [19]. Features that were represented by less than 2 reads as well as those assigned as chloroplast and mitochondria were removed. Further, the resulting OTU feature table was rarefied with a sampling depth of several sequences in the smallest-sized sample and only significant features were retained. Alpha-rarefaction curves for all samples were generated using QIIME2 “diversity” plugin. Phylogenetic Fast-tree was generated using MAFFT alignment by QIIME2 pipeline “phylogeny” using OTU representative sequences. The aforementioned phylogenetic tree was used to generate Beta diversity plots through QIIME2 diversity plugin.
2.4 Analytical determinations
Total suspended solids (TSS) and volatile suspended solids (VSS) were determined after filtering the samples with a glass fiber filter (Whatman, GF/C) following the standard method [20]. Sample COD was determined using HACH kits (21,258–51) and a HACH spectrophotometer (DR 1900). Total organic carbon (TOC) and inorganic carbon (IC) were measured by using TOC-L analyzer with TNM-1 module (Shimadzu). The samples were filtered through 0.45-μm polyethersulfone syringe filters before analyzing COD, TOC, TC, and IC, except when measuring CODt.
2.5 Elemental analysis
Lyophilized biomass samples containing 0.5–1.5-mg weight were loaded in small capsules and subjected to combustion at 980 °C in an elemental analyzer (EA 3000, EuroVector). Helium was used as a carrier gas with a rate of flow of 121 mL·min−1. Acetanilide was used to calibrate the instrument [21].
2.6 Protein extraction and measurement
The protein content of the biomass was analyzed by using a Modified Lowry Protein Assay Kit. Crude protein contents were estimated by multiplying the total nitrogen content measured through elemental analysis by a factor of 6.25 [22, 23]. It is viewed that this method can be an overestimate of crude protein content but is nevertheless generally accepted for an approximate measurement of crude protein.
2.7 Bacteriochlorophyll and carotenoids
Bacteriochlorophyll and carotenoids were first extracted using acetone as a solvent and measured by using a spectrophotometer at a wavelength of 473 nm and 771 nm following the method described in the literature [24]. The carotenoid and bacteriochlorophyll contents were calculated following Eqs. 1 and 2 below:
where A473 and A771 are the absorbances at 473 nm and 771 nm; L is the path length of the cuvette (cm); and W is the initial amount (g) of biomass sample, divided by the final volume (mL) of extracts obtained.
2.8 Statistical analysis
All determinations were carried out in duplicate and presented as average values with standard deviation. The difference between experimental treatments was analyzed by Student’s t-test at a significance level of p < 0.05 using JASP (version 0.14.1) software.
3 Results and discussion
3.1 PNSB growth
PNSB growth was monitored after adjusting the medium pH to 5.0, 6.0. 7.0, and 8.0. PNSB growth at the natural pH (without adjustment) of the FJW was not attempted, perhaps the pH was too low to support the growth of PNSB [14,15,16]. Results show that in CJW the cells started growing without any lag phase. Thus, it can be considered as a suitable substrate for the growth of microorganisms [25,26,27]. In all pH treatments, a steady increase in growth was observed until day 3. At pH 5.0 and pH 6.0, a decrease in growth was observed after 3 days of incubation, and then again, a slight increase after day 5. At higher pH (7.0, 8.0) growth was also slowed down after 3 days of incubation, but a gradual increase was observed until day 7. At this point, cultivation was stopped since the cells reached the early stationary phase (Fig. 1a). The highest absorbance (2.65 ± 0.01) was observed at pH 8.0 followed closely by pH 7.0 (2.63 ± 0.02). Absorbance at pH 6.0 (1.87 ± 0.44) and pH 5.0 (1.18 ± 0.06) were considerably less, indicating that acidic pH (5.0, 6.0) was relatively less favorable than neutral to slightly alkali conditions. One potential reason for the lower growth and the temporary reduction in biomass around days 3 to 5 is due to the equilibria of the organic acids being more towards the free acid forms, creating some inhibition on growth. However, pH profiles (“pH change” section) indicate that the fermentation of carbohydrates did not cause a further reduction in pH. Another possible reason could be the presence of methanogenic bacteria at acidic pH, which hampered PNSB growth. A mixed culture of PNSB was used in the study, and strict axenic techniques were not adopted to maintain PNSB abundance in the culture; therefore, the presence of other bacteria cannot be ruled out.
In MJW, the cells did not take time to adapt and showed an increase in growth until day 2 in all pH treatments; however, a slight decrease in growth on day 3 and then again an increase in growth was found after a day (Fig. 1b). This growth behavior is consistent with CJW treatment. Interestingly, pH 6.0 showed the highest absorbance (2.73 ± 0.01) followed by pH 7.0 (2.41 ± 0.47), pH 8.0 (2.29 ± 0.04), and pH 5.0 (2.20 ± 0.10). Absorbance at pH 6.0 was significantly higher (p < 0.05) than at pH 8.0, while at other pH values it was statistically non-significant due to high standard deviation. It turned out that pH 6.0 was the optimum pH while using MJW as a substrate. Microbial analysis indicated that 31% of fermentative bacteria belonging to the Clostridiacea family were present at this pH, providing further evidence for a co-operative interaction of PNSB with the fermentative bacteria.
VSS were determined to estimate the biomass production potential of PNSB. In CJW treatment, pH 8.0 showed the highest VSS (852 ± 115 mg·L−1) while pH 5.0 the lowest (278 ± 5 mg·L−1). VSS at pH 6.0 and 7.0 were 557 ± 125 mg·L−1 and 760 ± 96 mg·L−1, respectively (Fig. 2). VSS were closely correlated to absorbance values. In MJW, VSS also increased with an increase in pH but did not correlate well with absorbance measurements. VSS at pH 5.0, pH 6.0, pH 7.0, and pH 8.0 were (225 ± 35 mg·L−1), (175 ± 35 mg·L−1), (450 ± 141 mg·L−1), and (625 ± 35 mg·L−1), respectively. Biomass growth, represented by VSS, was not as rapid on MJW as it was on CJW. The difference in growth and the VSS can be attributed to the variation in the composition of the two substrates, i.e., MJW and CJW.
It is pertinent to note that the optimum initial pH for CJW and MJW was different. It indicated that the optimum initial pH depends on the substrate composition. CJW and MJW have different types and concentrations of carbon sources. For instance, citrus juice wastewater has a relatively higher content of citric and ascorbic acids which may be inhibitory at lower pH. Moreover, PNSB will follow different pathways to metabolize various carbon sources that may show pH selectivity based on the enzymes used. In addition, carbon sources have different reducing potentials that influence the pH and growth of PNSB [8, 13, 15].
3.2 pH change
CJW results showed a sharp decline in the pH on the first day of incubation in all of the treatments, except pH 6.0. Afterward, pH was stable over the entire growth period except for a slight increase in pH at the end. A decrease in the pH at the start of anaerobic incubation could be due to the fermentation of sugars and other organics in the juice or due to the photoheterotrophic growth of PNSB. They metabolize organic carbon present in CJW and produce CO2 that leads to a decrease in the pH [28, 29]. With time, the utilization of organic carbon in PNSB slows down resulting in less release of CO2, and thus, changes in the pH were not significant. An increase in the pH towards the end of the incubation period can be attributed to the photoautotrophic growth of PNSB. During photoautotrophic growth, the CO2 that was initially produced is consumed as a carbon source [12, 29]. The utilization of CO2 leads to an increase in the pH.
Initial ORP values in CJW for pH 8.0, pH 7.0, pH 6.0, and pH 5.0 were (− 26 ± 6 mV), (− 17 ± 7 mV), (18 ± 4 mV), and (104 ± 0 mV), respectively (Fig. 3c), indicating that the pH 8.0 treatment had the most reducing conditions, similar to pH 7.0. These values also suggest CO2 production rather than fermentation as the cause of the initial pH reduction, as the latter would require an ORP of less than − 100 mV. For the pH 5.0 treatment, ORP values reduced slightly in the first 3 days of incubation creating more reducing conditions. In other treatments (pH 6.0, 7.0, 8.0) there was a very slight increase in ORP values up to 5 days into the incubation, followed by a very slight decrease in the last few days. Overall, the ORP values were relatively stable. The pattern of change in ORP level under anaerobic conditions is in agreement with previous studies [13, 30].
Following a similar trend, MJW also showed a decrease in pH on the first 2 days, and then remained almost stable. The highest decrease in the pH was observed at pH 8.0, similar to CJW, and was followed by 7.0, 6.0, and 5.0. The ORP was relatively stable in the samples, decreasing very slightly at pH 8.0 and increasing slightly at pH 6.0. Overall, both wastewaters showed a similar trend of ORP change at different pH. For instance, at pH 5.0, both CJW and MJW showed a slight decrease in ORP, whereas at other pH values (6.0, 7.0, 8.0) they showed almost no change in the ORP, and both wastewaters showed similar ORP values for each pH condition.
3.3 Microbial community analysis
Microbial abundance was determined in CJW and MJW at the most acidic and alkaline pH conditions (6.0 and 8.0) to recognize the effect of pH change on the microbial community. The results showed a relatively high abundance of the Xanthobacteraceae family. Their abundance in CJW at pH 6.0 and pH 8.0 were 51% and 22%, respectively (Fig. 4). In MJW, Xanthobacteraceae abundance was 20% at pH 6.0 and 51% at pH 8.0. This family includes PNSB genera such as Rhodoplanes and Rhodopseudomonas [31, 32]. MJW also showed the presence of the Rhizobiaceae family, which includes some PNSB, and their abundance at pH 6.0 and pH 8.0 were 4% and 8%, respectively. Interestingly, the abundance of these organisms that are the most likely candidates of PNSB in the culture showed an opposite trend to the absorbance measurements taken at 420 nm (Fig. 1). This may be associated with the relatively low light intensity provided by the incubator shaker, so that as culture absorbance increases the available light to drive photoheterotrophy becomes more and more limiting, thereby reducing the dominance of this family. Further studies of these effects at high light intensity are needed in the future to confirm these effects.
Relative microbial abundance at family level in CJW and MJW at pH 6.0 and 8.0. The microbial population showing > 1% abundance was derived from the relative frequency barplot (up to 5 taxonomic levels) using QIIME2view. MiDAS 4.8.1 database [31] was used to know microbial taxonomy up to the genus level
Rhizobiaceae presence in CJW was almost negligible (< 0.2%). Pleomorphomonadaceae was found in all treatments; its highest abundance (9%) was observed in MJW at pH 6.0. In other treatments, it showed almost 2% abundance. A relatively high abundance of the anaerobic fermenters, Clostridiaceae, was found in CJW at pH 8.0 (18%) and in MJW at pH 6.0 (31%); in other samples, it was present only in a small fraction (1%). These organisms are anaerobic fermenters that are also nitrogen-fixing and frequently inhabit the same environments as PNSB [33]. Their presence seems to correlate well with absorbance growth measurements. Rikenellaceae was found at only pH 8.0 both in CJW and MJW. The presence of Enterobacteriaceae was dominant (22%) in CJW at pH 6.0 only, whereas its presence in other treatments was only 2.0 to 4.0% only. Sphingomonadaceae was also found in all samples but a small quantity only (0.1 to 4%). A large fraction (14 to 25%) of the samples remained un-assigned.
It is important to note that most of the identified microbial families such as Xanthobacteraceae, Pleomorphomonadaceae, Rhizobiaceae, and Sphingomonadaceae share the same class, i.e., Alphaproteobacteria. Clostridiaceae belongs to Firmicutes, Enterobacteriaceae to Gammaproteobacteria, and Rikenellaceae belongs to Bacteroidia class. Overall, the results show a reasonable abundance of potential PNSB in both feedstocks. The presence of other organisms was expected as a mixed microbial culture, dominated by PNSB, was used as an inoculum. Furthermore, the results demonstrate that the microbial population largely depends on the type of feedstock and the culture pH.
3.4 Carbon utilization
3.4.1 Total organic carbon and inorganic carbon
The initial TOC and IC values in CJW were 635 ± 45 mg·L−1 and 481 ± 9 mg·L−1, respectively. TOC and IC removals at different pH treatments were determined. In CJW, TOC removal increased with an increase in pH. TOC removals at pH 5.0, 6.0, 7.0, and 8.0 were (259 ± 7 mg·L−1), (329 ± 47 mg·L−1), (371 ± 12 mg·L−1), and (422 ± 13 mg·L−1), respectively (Fig. 5). TOC removal was closely correlated to cell growth. The highest TOC removal at pH 8.0 resulted in the highest cell density (VSS) at this pH. IC removal showed an opposite trend to TOC removal. pH 5.0 showed the highest IC removal (452 ± 2 mg·L−1), whereas the lowest IC removal (119 ± 19 mg·L−1) was found at pH 8.0. High IC removal at low pH was perhaps due to the degradation of bicarbonate, whereas at pH 7.0 and 8.0 its degradation was very small [34, 35]. PNSB display diverse metabolic pathways to degrade a variety of carbon sources. They remove carbon through the pentose phosphate pathway and tricarboxylic acid cycle (TCA). PNSB generate energy during anoxygenic photosynthesis that drives the removal of organic carbon [29].
Initial TOC and IC in MJW were 692 ± 55 mg·L−1 and 597 ± 5 mg·L−1, respectively. In MJW, pH 7.0 showed the highest TOC removal (357 ± 0 mg·L−1). pH 5.0, 6.0, and 8.0 showed TOC removals of (259 ± 33 mg·L−1), (227 ± 16 mg·L−1), and (274 ± 30 mg·L−1), respectively. IC removals in MJW at pH 5.0, 6.0, 7.0, and 8.0 were (115 ± 55 mg·L−1), (344 ± 11 mg·L−1), (154 ± 0 mg·L−1), and (36 ± 25 mg·L−1) consistent with CJW. Unlike CJW, the TOC removal in MJW did not correspond closely to biomass growth.
These contradicting results provide an understanding that TOC assimilation and conversion mechanisms do not necessarily depend on the cell culture density, but also on the feedstock composition and pH. One explanation for the high TOC removal at pH 7 in MJW, despite a lower VSS, is the conversion of some carbon to polyhydroxyalkanoates. While not tested, this would also explain the relatively low conversion to CO2. Microbial population analysis (“Microbial community analysis” section) revealed that the microbial abundance in the systems with the two feedstocks was different, but also showed some similarities. Thus, a change in microbial abundance that would alter carbon utilization metabolism showed varied behavior of TOC removal between different treatments. In CJW, carbon yield values (g-Cbiomass·g-Consumed−1) were 0.82, 0.70, 0.45, and 0.24 at pH 8.0, 7.0, 6.0, and 5.0 respectively, assuming a cell biomass formula of C5H7O2N. The corresponding yield (g-Cbiomass·g-CConsumed−1) values in MJW were 1.06, 0.46, 0.16, and 0.32. In both treatments, the carbon yield increased with an increase in the pH with only an exception at pH 6.0 in MJW.
3.4.2 COD removal
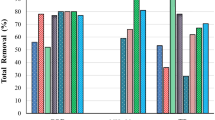
COD removal is an important parameter to determine the efficiency of the wastewater treatment system, where COD assimilation generally drives microbial biomass growth. CJW treatment results showed that the COD removal was dependent on the pH; removal increased with an increase in the pH. COD removals in CJW at pH 5.0, 6.0, 7.0, and 8.0 were 709 ± 65 mg·L−1, 1501 ± 25 mg·L−1, 1665 ± 8 mg·L−1, and 1729 ± 3 mg·L−1, respectively, and showed a similar trend to biomass growth (VSS) measurements observed earlier. These removals correlated to COD removal efficiencies of 36 ± 3.3%, 77 ± 1.3, 85 ± 0.4%, and 89 ± 0.1%, respectively. In comparison, COD removals in MJW at pH 5.0, 6.0, 7.0, and 8.0 were 661 ± 48 mg·L−1, 593 ± 14 mg·L−1, 1599 ± 11 mg·L−1, and 1575 ± 17 mg·L−1, respectively. These translate to removal efficiencies of 39 ± 3%, 35 ± 0.8, 94 ± 1%, and 92 ± 1% (Fig. 6). MJW COD removals also showed similar trends to biomass growth (VSS). The COD removal trend shows that it increased with an increase in the pH and the pH of 8.0 turned out as the optimum pH for CJW while for MJW COD removal efficiency was statistically similar (p > 0.05) at pH 7.0 and 8.0. High COD removal at elevated pH could be due to increased activity of PNSB and reduced inhibition of the overall biomass culture from unionized volatile fatty acids. These provide positive feedback generating more biomass and therefore faster COD degradation rates.
3.4.3 Nitrogen removal
Nitrogen is a key component to drive the growth of the microorganisms and the utilization of carbon. Total nitrogen (TN) removal was measured in CJW and MJW treatments (Fig. 7). TN removal in CJW was higher than the MJW at all pH levels. TN removal increased with an increase in the pH. In CJW, the highest TN removal was found at pH 8.0 (70 ± 3.13 mg·L−1) followed by pH 7.0 (64 ± 1 mg·L−1), pH 6.0 (43 ± 12 mg·L−1), and pH 5.0 (15.5 ± 4 mg·L−1). In MJW, TN removal at pH 8.0, 7.0, 6.0, and 5.0 were 19 ± 3 mg·L−1, 23.6 ± 3 mg·L−1, 21 ± 0 mg·L−1, and 26.3 ± 0 mg·L−1, respectively. Higher TN removal in CJW than the MJW is because the former one showed higher growth and the cell density. An increase in TN removal with pH was consistent with growth profile of the PNSB—as the growth increased with an increase in the pH.
3.5 Biomass yield
The biomass yields in CJW at pH 5.0, 6.0, 7.0, and 8.0 were 0.393 ± 0.01, 0.371 ± 0.08, 0.456 ± 0.06, and 0.493 ± 0.07 g-VSS·g-COD−1. It implies that an increase in pH enhances the metabolic ability of PNSB to consume and assimilate COD into microbial biomass. However, in MJW this trend of COD assimilation was not followed. Biomass yield showed higher values at low and high pH, with pH 8 being optimal. The specific biomass yields on MJW were 0.340 ± 0.05, 0.295 ± 0.06, 0.281 ± 0.09, and 0.397 ± 0.02 g-VSS·g-COD−1 at pH 5.0, 6.0, 7.0, and 8.0 (Fig. 8). The yield values were not statistically different from each other (p > 0.05). The results show that at the same pH condition in CJW and MJW, PNSB shows a different trend of COD removal and biomass yield. It is likely due to variations in the composition of the two wastewaters that contain different types and concentrations of organic compounds, nutrients, and other growth factors.
Previous studies demonstrate the biomass yield of PNSB using different wastewaters as feedstocks. The biomass yields using domestic wastewater, meat-processing wastewater, food-processing wastewater, aquaculture wastewater, and anaerobic digestate were 0.95, 0.7, 0.6, 0.6, and 0.2 g-CODbiomass·g-COD−1, respectively. On a g-VSS·g-COD−1 basis, the yield (theoretical) is 0.66 for domestic wastewater, 0.49 for meat-processing wastewater, 0.42 for food-processing and aquaculture wastewater, and 0.14 for anaerobic digestate [12, 14, 16, 36,37,38]. Overall, the yields obtained in this study were comparable with the reported studies on photoanaerobic growth cultures. The maximum yield obtained in this study of 0.49 g-VSS·g-COD−1 through anaerobic treatment was comparable with aerobic growth systems. In typical aerobic heterotrophic culture, the biomass yield is 0.45 g-VSS·g-COD−1 translating to 0.63 g-CODbiomass·g-COD−1 [39]. A relatively low biomass yield obtained in this study could be due to the non-ideal composition of the FJW wastewater as a growth medium. However, the most likely reason is the low light intensity in the incubator shaker, leading to a relatively low enrichment level of PNSB as discussed in the “Microbial community analysis” section.
3.6 Biochemical composition
3.6.1 Single-cell protein
SCP contents were determined by using Lowry assay as well as through elemental analysis. Protein contents determined with the Lowry assay were 48 ± 1% and 40 ± 3% in CJW and MJW, respectively. Whereas crude protein estimates in CJW and MJW were 64.5 ± 2.1% and 60 ± 3.8%, respectively. The higher estimate of crude protein content than measured by the Lowry assay could be due to the multiplication factor of 6.25 not being valid for this particular culture/substrate. For an accurate measurement of crude protein, a correlation factor between total nitrogen and protein content of FJW PNSB enriched mixed cultures should be developed. Nevertheless, this study demonstrates that FJW is a useful substrate for PNSB to produce a moderate level of SCP. It is important to note that nitrogen was sufficient at the end of the test, with the lowest residual total nitrogen value being 19 ± 5 at pH 8.0 for CJW and 59 ± 0.05 at pH 8.0 in MJW. The SCP contents obtained in this study are comparable with the previous studies. Hulsen et al. [37] reported 60% of crude protein using PNSB and agro-industrial wastewater as a substrate at pH 6.7. Honda et al. [39] achieved an SCP content of 56–68% using simulated anaerobic wastewater as a feedstock for Rhodopseudomonas palustris. The pH of the wastewater was adjusted to 7.0. Similarly, Getha et al. [40] reported 72–74% crude protein, and Kim et al. [41] achieved 61% protein while growing PNSB at pH 5.5 using MYC media. Although different types of substrates, culture conditions, and PNSB species were employed across the studies making direct comparison difficult, these results indicate that FJW is a suitable substrate for SCP production despite its high sugar and VFA contents. This is the first report of PNSB conversion of FJW into SCP. Further studies should be carried out to optimize the SCP yield and composition to realize its final use in a biorefinery scheme.
3.6.2 Photopigments
In addition to SCP, the PNSB biomass also contains photopigments such as carotenoids and bacteriochlorophyll. They provide protection against photodamage, enhance growth performance, and more importantly improve the quality of SCP. These photopigments are essential components of SCP to qualify as high-quality feed [42]. Photopigments have been used as supplements for shrimp and animal feed [42,43,44]. In CJW, bacteriochlorophyll and carotenoid contents were 2097 ± 110 and 1354 ± 64 µg·g−1. In comparison, the PNSB in MJW showed bacteriochlorophyll of 3100 ± 614, and carotenoids of 1558 ± 218 µg·g−1 (Table 2). There was no statistically significant difference (α = 0.05) in the photopigment production between the two FJW substrates. Carotenoid contents achieved in this study were comparable with previous studies. Kuo et al. [45] reported 1783 µg·g−1 carotenoids by feeding NS medium to Rhodopseudomonas palustris. These results indicate that the real, complex FJW is a suitable source for carotenoid production, given that the study carried out by Kuo et al. [45] utilized pure media and a single strain of PNSB. A typical carotenoid contents in PNSB biomass range from 0.5 to 13 mg·g−1 of dry biomass [46, 47] experimented the cultivation of fairy shrimp with three different types of PNSB species including Rhodobacter sphaeroides, Rhodopseudomonas palustris, and Rhodopseudomonas faecalis. Fairy shrimp fed with Rhodopseudomonas faecalis showed the highest growth performance under larval and adult stages. PNSB showed a higher survival rate of the shrimp than using commercial dried spirulina. The study concluded that PNSB could as an alternative feed for fairy shrimp growth. In another study, Alloul et al. [48] tested PNSB as a feed for the shrimp. The results indicated that the shrimp fed with PNSB biomass showed higher shrimp weight and feed conversion ratio than the commercial diet. Moreover, PNSB-fed shrimp culture showed higher tolerance against ammonia than the commercial diet. Based on these studies, PNSB can be suggested as an alternative and value-added feed ingredient for aquaculture. One possible issue in using FJW as a substrate for PNSB biomass production and its subsequent use as an SCP feed could be the economic sustainability. FJW does not contain essential nutrients such as nitrogen, phosphorous, and trace nutrients (minerals and vitamins). A sustainable and cost-effective supply of these nutrients is challenging. One possibility is to explore other wastewaters containing plenty of nutrients that could be used as a substrate for PNSB and displace the need for trace and other nutrient addition. To this end, few studies demonstrated the use of real industrial wastewater as a feedstock for SCP production without the addition of nutrients. Hulsen et al. [49] investigated the use of piggery- and chicken-processing wastewaters as PNSB substrates without the addition of any nutrients. PNSB biomass showed 50–65% (depending on the wastewater) of SCP contents. In another study, Vethathirri et al. [50] utilized soybean-processing wastewater as a microbial substrate, without nutrient addition, to produce SCP that could meet aquaculture feed requirements.
3.6.3 Elemental analysis
The element composition of PNSB biomass showed that the carbon, hydrogen, nitrogen, and sulfur contents in CJW were 49.8 ± 2.3%, 7.53 ± 0.21%, 11.9 ± 1.1%, and 11.17 ± 3.71%, respectively. The PNSB grown with MJW showed an elemental composition containing carbon 52.7 ± 4.0%, hydrogen 8.1 ± 1.2%, and nitrogen 13.14 ± 0.9%. The C/N ratios of CJW and MJW biomasses were 4.20 ± 0.22 and 4.0 ± 0.22, respectively. The results indicate that both treatments showed almost similar elemental composition. They showed a relatively high nitrogen percentage (11–13%) that corresponds to high protein content. CHN content is in agreement with other microbial compositions such as algae [51, 52]. A direct comparison of elemental composition data of this study with the PNSB was not possible due to the lack of literature studies on PNSB.
4 Conclusions
This study investigated the use of PNSB for the treatment of juice industry wastewater and the conversion of soluble organic contaminants into valuable protein-containing solids that can be utilized for animal/aquaculture feed purposes. CJW and MJW were used as substrates at different pH values. It was found that PNSB were able to utilize both substrates effectively. The substrate utilization, COD removal, organic carbon removal, and the VSS were pH dependent. pH 8.0 was the most suitable to achieve high treatment performance and biomass yield. Moreover, PNSB response to the substrates was dependent on the juice composition. At the optimum pH, cellular protein contents in CJW and MJW were 48 ± 1% and 40 ± 3%. PNSB accumulated an appreciable amount of carotenoids and bacteriochlorophyll while using CJW and MJW. Based on these results, it is concluded that PNSB-based SCP is a worthy biorefinery application to treat FJW. This study indicates that low biomass yield and additional supplements of the nutrients (trace and minerals) are the major limitations to the sustainable treatment of FJW using PNSB. Further investigations are required to explore the cheap source of the nutrients and to carry out process optimization studies to improve biomass yield.
Data availability
Not applicable.
References
Tawfik A, El-Kamah H (2012) Treatment of fruit-juice industry wastewater in a two-stage anaerobic hybrid (AH) reactor system followed by a sequencing batch reactor (SBR). Environ Technol 33:429–436. https://doi.org/10.1080/09593330.2011.579178
Sáenz de Miera B, Souza de Oliveira A, Baeza Herrera J et al (2019) Treatment and valorization of fruit juice wastewater by aqueous phase reforming: effect of pH, organic load and salinity. J Clean Prod 252:119849. https://doi.org/10.1016/j.jclepro.2019.119849
Can OT (2014) COD removal from fruit-juice production wastewater by electrooxidation electrocoagulation and electro-Fenton processes. Desalt Water Treat 52:65–73. https://doi.org/10.1080/19443994.2013.781545
Amor C, Lucas M, Pirra A et al (2012) Treatment of concentrated fruit juice wastewater by the combination of biological and chemical processes. J Environ Sci Health. Part A Toxic/Hazard Subst Environ Eng 47:1809–1817. https://doi.org/10.1080/10934529.2012.689244
TeKippe RJ (1972) Treatment and disposal of citrus fruit processing wastes. J Water Pollut Control Fed 44:2001–2012
Akbay HEG, Akarsu C (2017) Kumbur, H (2018) Treatment of fruit juice concentrate wastewater by electrocoagulation: optimization of COD removal. Intl Adv Res Eng J 1:53–57
Zema D, Calabrò P, Folino A et al (2019) Wastewater management in citrus processing industries: an overview of advantages and limits. Water 11:2481. https://doi.org/10.3390/w11122481
Meng F, Yang A, Wang H et al (2018) One-step treatment and resource recovery of high-concentration non-toxic organic wastewater by photosynthetic bacteria. Bioresour Technol 251:121–127. https://doi.org/10.1016/j.biortech.2017.12.002
Anupama, Ravindra, P (2000) Value-added food: single cell protein. Biotechnol Adv 18:459-479. https://doi.org/10.1016/S0734-9750(00)00045-8
Matassa S, Batstone DJ, Hülsen T et al (2015) Can direct conversion of used nitrogen to new feed and protein help feed the world? Environ Sci Technol 49:5247–5254. https://doi.org/10.1021/es505432w
Van der Spiegel M, Noordam MY, Van der Fels-Klerx HJ (2013) Safety of novel protein sources (insects, microalgae, seaweed, duckweed, and rapeseed) and legislative aspects for their application in food and feed production. Compr Rev Food Sci Food Safe 12:662–678. https://doi.org/10.1111/1541-4337.12032
Hulsen T, Batstone DJ, Keller J (2014) Phototrophic bacteria for nutrient recovery from domestic wastewater. Water Res 50:18–26. https://doi.org/10.1016/j.watres.2013.10.051
FONA, https://www.fona.com/wp-content/themes/fona/migrated-files/Juice%20Category%20Report%200917.pdf, FONA International, 2017, Geneva, 2017
Carlozzi P, Di Lorenzo T, Ghanotakis DF et al (2020) Effects of pH, temperature and salinity on P3HB synthesis culturing the marine Rhodovulum sulfidophilum DSM-1374. Appl Microbiol Biotechnol 104:2007–2015. https://doi.org/10.1007/s00253-020-10352-1
Kantachote D, Torpee S, Umsakul K (2005) The potential use of anoxygenic phototrophic bacteria for treating latex rubber sheet wastewater. Electron J Biotechnol 8:3–8. https://doi.org/10.2225/vol8-issue3-fulltext-8
Inglesby A, Beatty D, Fisher A (2012) Rhodopseudomonas palustris purple bacteria fed Arthrospira maxima cyanobacteria: demonstration of application in microbial fuel cells. RSC Adv 2:4829–4838. https://doi.org/10.1039/C2RA20264F
ATCC 2672 https://www.atcc.org/~/media/A457A0AE8C604A12B5597E5EB1A6583A.ashx.
Bolyen E, Rideout JR, Dillon MR et al (2019) Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nature Biotechnol 37:852–857. https://doi.org/10.1038/s41587-019-0209-9
Yilmaz P, Parfrey LW, Yarza P et al (2014) The SILVA and “All-species Living Tree Project (LTP)” taxonomic frameworks. Nucleic Acids Res 42:D643-648. https://doi.org/10.1093/nar/gkt1209
Greenberg AE, Clesceri LS, Eaton AD (1992) Standard methods for the examination of water and wastewater. American Public Health Association, Washington DC
Barbarino E, Lourenço SO (2009) Comparison of CHN analysis and Hach acid digestion to quantify total nitrogen in marine organisms. Limnol Oceanogr Methods 7:751–760. https://doi.org/10.4319/lom.2009.7.751
Eding EH, Kamstra A, Verreth JAJ et al (2006) Design and operation of nitrifying trickling filters in recirculating aquaculture: a review. Aquacult Eng 34:234–260. https://doi.org/10.1016/j.aquaeng.2005.09.007
Ponsano EHG, Pinto MF, Garcia Neto M et al (2004) Performance and color of broilers fed diets containing Rhodocyclus gelatinosus biomass. Brazil J Poult Sci 6:237–242. https://doi.org/10.1590/S1516-635X2004000400008
Zhou Q, Zhang P, Zhang G (2014) Biomass and pigments production in photosynthetic bacteria wastewater treatment: effects of light sources. Bioresour Technol 179C:505–509. https://doi.org/10.1016/j.biortech.2014.12.077
Brotosudarmo THP, Limantara, L, Heriyanto et al (2015) Adaptation of the photosynthetic unit of purple bacteria to changes of light illumination intensities. Procedia Chem 14:414-421. https://doi.org/10.1016/j.proche.2015.03.056
Cerruti M, Stevens B, Ebrahimi S et al (2020) Enrichment and aggregation of purple non-sulfur bacteria in a mixed-culture sequencing-batch photobioreactor for biological nutrient removal from wastewater. Front Bioeng Biotechnol 8:557234. https://doi.org/10.3389/fbioe.2020.557234
Izu K, Nakajima F, Yamamoto K et al (2001) Aeration conditions affecting growth of purple nonsulfur bacteria in an organic wastewater treatment process. Syst Appl Microbiol 24:294–302. https://doi.org/10.1078/0723-2020-00027
Kantachote D, Torpee S, Umsakul K (2005) The potential use of anoxygenic phototrophic bacteria for treating latex rubber sheet wastewater. Elect J Biotechnol 8:3–8. https://doi.org/10.2225/vol8-issue3-fulltext-8
Sepúlveda-Muñoz CA, de Godos I, Puyol D et al (2020) A systematic optimization of piggery wastewater treatment with purple phototrophic bacteria. Chemosphere 253:126621. https://doi.org/10.1016/j.chemosphere.2020.126621
Wu C, Zhou Y, Wang P et al (2015) Improving hydrolysis acidification by limited aeration in the pretreatment of petrochemical wastewater. Bioresour Technol 194:256–262. https://doi.org/10.1016/j.biortech.2015.06.072
Dueholm MS, Nierychlo M, Andersen KS et al (2022) MiDAS 4: A global catalog of full-length 16S rRNA gene sequences and taxonomy for studies of bacterial communities in wastewater treatment plants. Nat Commun 13:1908. https://doi.org/10.1038/s41467-022-29438-7
Nierychlo M, Andersen KS, Xu Y et al (2020) MiDAS 3: an ecosystem-specific reference database, taxonomy and knowledge platform for activated sludge and anaerobic digesters reveals species-level microbiome composition of activated sludge. Water Res 182:115955. https://doi.org/10.1016/j.watres.2020.115955
Pepe-Ranney C, Koechli C, Potrafka R et al (2016) Non-cyanobacterial diazotrophs mediate dinitrogen fixation in biological soil crusts during early crust formation. The ISME J 10:287–298. https://doi.org/10.1038/ismej.2015.106
Liu J, Chu X, Zhu L et al (2010) Simultaneous aqueous-phase reforming and KOH carbonation to produce COx-free hydrogen in a single reactor. Chemsuschem 3:803–806. https://doi.org/10.1002/cssc.201000093
Xu Y, Tian Z, Wen G et al (2006) Production of COx-free hydrogen by alkali enhanced hydrothermal catalytic reforming of biomass-derived alcohols. Chem Lett 35:216–217. https://doi.org/10.1246/cl.2006.216
Hulsen T, Barry EM, Lu Y et al (2016) Domestic wastewater treatment with purple phototrophic bacteria using a novel continuous photo anaerobic membrane bioreactor. Water Res 100:486–495. https://doi.org/10.1016/j.watres.2016.04.061
Hülsen T, Hsieh K, Lu Y et al (2018) Simultaneous treatment and single cell protein production from agri-industrial wastewaters using purple phototrophic bacteria or microalgae — a comparison. Bioresour Technol 254:214–223. https://doi.org/10.1016/j.biortech.2018.01.032
Capson-Tojo G, Batstone DJ, Grassino M et al (2020) Purple phototrophic bacteria for resource recovery: challenges and opportunities. Biotechnol Adv 43:107567. https://doi.org/10.1016/j.biotechadv.2020.107567
Honda R, Fukushi K, Yamamoto K (2006) Optimization of wastewater feeding for single-cell protein production in an anaerobic wastewater treatment process utilizing purple non-sulfur bacteria in mixed culture condition. J Biotechnol 125:565–573. https://doi.org/10.1016/j.jbiotec.2006.03.022
Getha K, Chong V, Vikineswary S (1998) Potential use of the phototrophic bacteria, Rhodopseudomonas palustris, as an aquaculture feed. Asian Fish Sci 10:223–232
Kim JK, Lee B-K (2000) Mass production of Rhodopseudomonas palustris as a diet for aquaculture. Aquacult Eng 23:281–293. https://doi.org/10.1016/S0144-8609(00)00057-1
Saejung C, Salasook P (2018) Recycling of sugar industry wastewater for single-cell protein production with supplemental carotenoids. Environ Technol 41:1–35. https://doi.org/10.1080/09593330.2018.1491633
Chumpol S, Kantachote D, Nitoda T et al (2018) Administration of purple nonsulfur bacteria as single cell protein by mixing with shrimp feed to enhance growth, immune response and survival in white shrimp (Litopenaeus vannamei) cultivation. Aquaculture 489:85–95. https://doi.org/10.1016/j.aquaculture.2018.02.009
Wang W, Sun J, Liu C et al (2017) Application of immunostimulants in aquaculture: current knowledge and future perspectives. Aquaculture Res 48:1–23. https://doi.org/10.1111/are.13161
Kuo F-S, Chien Y-H, Chen C-J (2012) Effects of light sources on growth and carotenoid content of photosynthetic bacteria Rhodopseudomonas palustris. Bioresour Technol 113:315–318. https://doi.org/10.1016/j.biortech.2012.01.087
Rashid N, Onwusogh U, Mackey HR (2022) Exploring the metabolic features of purple non-sulfur bacteria for waste carbon utilization and single-cell protein synthesis. Biomass Convers Biorefinery. https://doi.org/10.1007/s13399-022-03273-8
Saejung C, Chaiyarat A, Sanoamuang L-o (2021) Optimization of three anoxygenic photosynthetic bacteria as feed to enhance growth, survival, and water quality in fairy shrimp (Streptocephalus sirindhornae) cultivation. Aquaculture 534:736288. https://doi.org/10.1016/j.aquaculture.2020.736288
Alloul A, Wille M, Lucenti P et al (2021) Purple bacteria as added-value protein ingredient in shrimp feed: Penaeus vannamei growth performance, and tolerance against Vibrio and ammonia stress. Aquaculture 530:735788. https://doi.org/10.1016/j.aquaculture.2020.735788
Hulsen T, Stegman S, Batstone DJ et al (2022) Naturally illuminated photobioreactors for resource recovery from piggery and chicken-processing wastewaters utilising purple phototrophic bacteria. Water Res 214:118194. https://doi.org/10.1016/j.watres.2022.118194
Vethathirri RS, Santillan E, Thi SS et al (2022) Microbial community-based production of single cell protein from soybean-processing wastewater of variable chemical composition.bioRxiv:2022.2008.2002.502426. https://doi.org/10.1101/2022.08.02.502426v1
Zhang H, Gong T, Li J et al (2022) Study on the effect of spray drying process on the quality of microalgal biomass: a comprehensive biocomposition analysis of spray-dried S. acuminatus biomass. BioEnergy Res 15:320–333. https://doi.org/10.1007/s12155-021-10343-8
Nayak M, Karemore A, Sen R (2016) Sustainable valorization of flue gas CO2 and wastewater for the production of microalgal biomass as a biofuel feedstock in closed and open reactor systems. RSC Adv 6:91111. https://doi.org/10.1039/c6ra17899e
Funding
Open Access funding provided by the Qatar National Library. Financial support was provided by the Ministry of Municipality and Environment to enable this work through grant MME01-0910–190,029.
Author information
Authors and Affiliations
Contributions
Naim Rashid: experimental design, execution, writing — original draft and editing. Mirna N. Abdelnabi: microbial analysis. Annette Shoba Vincent: writing — review and editing, funding acquisition. Hamish R. Mackey: writing — review and editing, funding acquisition, supervision.
Corresponding author
Ethics declarations
Ethics approval
The authors declare that the manuscript has not been published previously.
Consent to participate
All the authors voluntarily participated in this research study.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rashid, N., Abdelnabi, M.N.A., Vincent, A.S. et al. Simultaneous treatment of fruit juice industry wastewater and single-cell protein synthesis using purple non-sulfur bacteria. Biomass Conv. Bioref. 13, 16321–16332 (2023). https://doi.org/10.1007/s13399-022-03669-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-022-03669-6