Abstract
Activation of biomass before pyrolysis with various chemicals significantly affects the surface area and porosity, chemical composition, and formation and distribution of functional groups on the surface of the biochar produced. For this purpose, raw tea waste (RTW) was mixed with potassium nitrate (KNO3), potassium sodium carbonate (NaKCO3), and sodium borohydride (NaBH4) in solid form and pyrolyzed at 500 °C for 1 h. The effects of the chemical activators on biomass char formation were investigated using DTA-TGA and DSC. Compared to conventional pyrolysis, carbonate, nitrate, and hydrides increase the gasification of biochar by catalyzing the decomposition of cellulose and lignin. The effect of NaBH4 on graphitization and deoxidation of carbon is higher than that of carbonates and nitrides. In addition, all prepared biochar samples were characterized by XRD, SEM, FT-IR, elemental analysis, and N2 adsorption–desorption. While treatment of RTW with KNO3 and NaKCO3 increased the hydroxylation of the biochar, treatment with NaBH4 decreased hydroxylation by increasing dehydrogenation and dehydroxylation. Increasing boron content led to hydroxylation of the material with hydratation of NaBO2. The surface area and pore distribution results showed that nitrates and carbonates have insignificant effect on the surface area of biochar, while NaBH4 almost doubles the surface area and total pore volume of biochar by forming hydrogen.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Biochar has excellent surface area and strong adsorption capabilitie, which therefore has been employed extensively in water and soil remediation as an environmentally beneficial material [1]. The features of biochar’s surface will determine how it is used to reduce pollution. The primary method for enhancing biochar’s surface characteristics during preparation and after preperation is activation. Activated biochar has gained increasing attention in recent years due to its well-developed porous structures, large surface area, and wide spectrum of surface functional groups. Biochar can be activated in two main ways: physically and chemically. Due to its ability to concurrently combine carbonization and activation and its potential to produce biochar with superior porous structures than physical activation, chemical activation has recived increasing attention [2]. A variety of chemicals, including ZnCl2 [3], KOH [4, 5], NaOH[6], H3PO4 [5, 7], and NH3·H2O[8], can be used to activate biochar. Chemical activation could increase the removal of water and volatile compounds from biochar through the degradation of aliphatic side chains and aromatic C-blocks [9] and the reaction of the activator with carbon [10], reduce tar formation [9], and promote deoxygenation, dehydrogenation, and aromatization reactions [11]. The chemical surface properties of biochar, such as the composition of the functional groups on the surface, composition, the aromaticity of the surface, the degree of crystallization, and porosity, are primarily altered by some of these activators.
In addition to the above chemicals, biomass can also be activated by chemical compounds such as carbonate, nitride, and hydride. Among the chemicals, potassium carbonate (K2CO3) is a very good chemical activator for design of textural structure of the biomass because it is nontoxic and relatively inexpensive [12, 13]. The K2CO3 solution was mixed with straw or cellulosic material; K2CO3 at about 700 °C reacts with carbon to form potassium and carbon monoxide via K2CO3 + 2C → 2 K + 3CO [14]. By intercalation over carbon layers at a temperature lower than 600 °C, the metallic K could further increase the porosity and expand the pores. Removal of the tar-containing material in the pores opens the pores and micro- and mesopores are formed [15]. Zhu et al. reported that the aromatic, hydrophobic, and nonpolar properties of the biochar were enhanced after activation with potassium carbonate to produce an aromatized non-polar surface, and the aromatic properties were enhanced with an increase in the pyrolysis temperature [16]. According to Mai et al. [17], activation with potassium carbonate can produce carbons with a high proportion of structural defects and porosity evenly distributed over micro- and mesopores. In addition, various researchers [18,19,20,21,22,23,24] have reported using biochar activated by K2CO3 with specific surface areas ranging from 500 to 3000 m2/g in supercapacitors, lithium-ion batteries, electrochemical hydrogen storage, and other fields. For KNO3 activation, Wongmat et al. reported similar results with KHCO3 [25].
Activation of biomass with boron precursors before and after carbonization alters the electronic and chemical structure of the carbon surface through changes in the electron transfer rate in the structure and the formation of defects and active sites [26]. In addition, the addition of boron to carbon structure leads to an increase in the mechanical and thermal stability of the composite [27]. For this purpose, boric acid is often used as boron precursor [28,29,30,31,32,33]. On the other hand, sodium borohydride (NaBH4) is generally known as a reducing chemical and a hydrogen carrier. NaBH4 is generally used to produce hydrogen by reaction with methanol, and there are few studies on its use as a chemical activator. The main objective of the study is to investigate in detail how the structure and composition of biochar produced from raw tea waste with predominantly cellulosic and hemicellulosic structures changes when activated by carbonates, nitrates, and hydrides. Therefore, in this study, the production mechanism of biochar from raw tea waste activated with NaBH4 along with NaKCO3 and KNO3 was investigated using DSC and DTA-TGA. In addition, the physicochemical and chemical properties of the produced biochar were investigated using elemental analysis, SEM–EDS, XRD, FT-IR, elemental analysis, and N2 adsorption and desorption. The effects of the activators on the pyrolysis mechanism and the properties of the biochar were evaluated in detail.
2 Materials and methods
2.1 Chemicals
Raw tea waste (RTW) was obtained from the Rize region of Turkey. The waste was crushed with a ball mill, and the particle size was adjusted with sieves ranging from 0.25 to 0.5 mm. Before treatment, 50 g of RTW was washed with 1 L of distilled water to remove impurities such as dust and soil on the surface of the RTW coming from the tea factories and dried in an oven. The chemicals, potassium nitrate (KNO3, ≥ 99.0%, Merck (CAS No:
7757–79-1)), potassium sodium carbonate (NaKCO3·6H2O, ≥ 99.0%, Chemical Aid), and sodium borohydride (NaBH4, ≥ 98.0%, Merck (CAS No:16940–66-2)), used for the treatment of RTW were purchased from companies and used without purification.
2.2 The production and treatment of biochar
Five grams of RTW was placed in the ceramic curicible and sealed with a lid. The sample was placed in the muffle furnace (Protherm) and heated to 500 °C with a ramp of 10 °C/min and kept for 1 h. Since the decomposition of RTW, which consists of hemicellulose, cellulose, and lignin, takes place at around 280–500 °C [7], the pyrolysis temperature was set at 500 °C. After pyrolysis, the sample was washed with distilled water to remove activators that did not enter the structure, chemicals such as tar remaining on the surface, and to open the pores of the biochar. The washing process continued until the color of the distilled water became clear and dried in the oven. The sample was labeled as BC.
For the chemical treatment, NaKCO3, KNO3, and NaBH4 were mixed with RTW at a ratio of 1:5 (chemical:biomass) in a mortar and pyrolyzed with BC under the same conditions. The ratio of chemical to biomass was set to 1:5 based on the data reported in the literature [34, 35] for KOH/biomass in order to keep the biochar yield high due to the chemical modification.
Also, the different ratios of RTW:NaBH4 (5:1, 3:2, 3:3) were pyrolyzed under the same conditions. The chemically activated biochar produced was designated BC-x–y, where x and y indicate the activating chemical and the ratio of RTW and the activating chemical, respectively.
2.3 Characterization
X-ray diffraction (XRD) of the biochar samples was measured using the XRD RIGAKU MINIFLEX 600 device for Cu-Kβ radiation with a wavelength of λ = 1.5 Å at 40 kW 30 mA conditions in the range of 5–90 of 2θ. Biochar morphology was visualized by field emission scanning electron microscopy (FESEM) with a Tescan® Mira3 XMU (Brno, Czech Republic) instrument using scattering electron (SE) and back-scattering electron (BSE) detectors with 5-nm gold plating result. The elemental composition of the biochar samples was determined using an energy-dispersive X-ray spectroscopy instrument (EDS, Oxford Instruments Inca, X-Act/51-ADD0013) connected to the Tescan® Mira3 XMU FESEM instrument and by an elemental analyzer (LECO, CHNS-932), and oxygen (O) content of samples was determined by mass balance. The carbonization of biochar was measured by differential scanning calorimeter (DSC) (DSC-60 Plus/60A, Schimadzu Corparation, Japan) and thermogravimetry differential thermal analysis (TG–DTA Shimadzu 60, Japan) under the nitrogen atmosphere.
FT-IR (Fourier transform infrared) analysis was applied to the samples to determine the functional groups of the biochar. Samples were analyzed using a BrukerAplha FT-IR instrument. For the analysis, each sample of 0.15 g KBr and 0.0015 g sample was weighed and prepared in tablet form and analyzed for their functional groups. The spectra were recorded in the wavelength range of 400–4000 cm−1. The surface and pore characteristics of the samples were determined by N2 adsorption–desorption using Quantachrome Autosorb1C analyzer (FL, USA).
3 Results and discussion
3.1 DSC and TG–DTA
The DSC and TG–DTA curves of RTW and chemically activated RTW are shown in Fig. 1. For the conventional pyrolysis of RTW, four different DSC peaks can be seen in Fig. 1 a. As reported in [36], RTW consists of cellulose, hemicellulose, and lignin, which are decomposed during pyrolysis. The distinct negative peak at 88.8 °C in Fig. 1 a is due to the removal of water from RTW. The chemical activation of RTW prior to pyrolysis shifted the maximum of the dehydration peak of RTW. The dehydration maxima of KNO3, NaKCO3, and NaBH4 are determined to be 107, 92, and 101 °C, respectively. The differences between the peak maxima could be related to the formation of pores during activation, so that the removal of water from the pores requires a slightly higher temperature. The decomposition peaks of hemicellulose, cellulose and lignin in RTW were determined in the range of 140–300 °C, around 340 °C, and above 430 °C, respectively, as reported in [37, 38]. In the DTA, these decomposition peaks are observed as a large positive peak between 30 and 500 °C (Fig. 1a). The results obtained from the TG data show that the percentage weight loss in this temperature range is 11%.
In the pyrolysis of a mixture of KNO3 and RTW, three negative peaks were measured at 107, 131, and 330 °C and a prominent positive peak at 395 °C. The peaks at 131 °C and 330 °C indicate the transition from α to β and the melting of KNO3, respectively [39]. The peak at 395 °C could be due to the reaction of cellulose with KNO3. The DTA curves agree with those of DSC, and the total mass loss after addition of KNO3 was calculated to be 62%. Compared to RTW, the addition of KNO3 significantly increases the gasification of biomass. This is consistent with the results of Wongmat and Wagner [25], reporting that the presence of KNO3 reduces the activation energy of wheat straw decomposition.
Figure 1 c illustrates the effect of NaKCO3 on the pyrolysis of RTW. The DSC shows a sharp negative peak at 92 °C, which is due to the dehydration of water from RTW and NaKCO3. Water evaporation from Na2CO3 was measured in the range of 80.1–101.3 °C [40], which is consistent with peak maximum. A broad negative peak in the range of 150–250 °C is due to the decomposition of hemicellulose. In addition, two positive peaks at 333 and 425 °C are due to the decomposition reaction of cellulose and lignin in the presence of NaKCO3. Compared to conventional pyrolysis, the alkali salt treatment of RTW slightly lowers the decomposition temperature of cellulose and lignin. The results are consistent with the results of Wei et al. for the production of bamboo biochar with Na2CO3 [41]. The TGA results showed that the weight loss of biomass in the presence of NaKCO3 is similar to that of KNO3.
The pyrolysis of RTW in the presence of NaBH4 is shown in Fig. 1 d. Four negative peaks and two positive peaks as well as a positive peak shoulder were observed in the decomposition of NaBH4 with biomass. The first negative peak at 48 °C could be due to the removal of volatile compounds by the reaction of NaBH4 and biomass. The negative peak at 101 °C is due to the removal of water and hydrogen from biochar and NaBH4. The negative peak at 265 °C may be due to the decomposition of hemicellulose. The peak at 331 °C and the shoulder at 300 °C are due to the decomposition of cellulose with NaBH4. The peak at 540 °C is due to the decomposition of NaBH4 [42]. The TGA results show a mass loss of 57% due to reaction of RTW with NaBH4. The comparison with the RTW of RTW-NaBH4 mixture shows that the addition of NaBH4 increases the gasification of biomass by removing oxygen species from hemicellulose and cellulose in the presence of NaBH4.
3.2 XRD results
XRD pattern of biochar samples produced from RTW by conventional pyrolysis and by activation with KNO3, NaKCO3 and NaBH4 is shown in Fig. 2. All biochar samples showed a distinct peak at 23.5° of 2θ and a broad peak at 42.9 of 2θ, corresponding to the amorphous carbon and graphitic carbon [43], respectively. Except KNO3 activation, the biochar showed a distict peak at 7.5 of 2θ via formation of carbonaceous peaks. Moreover, the diffraction peaks detected at 2θ = 23° and 44.5° are related to the graphitization of RTW biochar with activation [44]. The highest graphitization was obtained with NaBH4 activation. The increasing degree of graphitization of the material in the presence of NaBH4 was explained by the replacement of the sp2 and sp3 structure by the incorporation of small radius boron into the carbon lattice [45, 46].
3.3 Morphology and composition of biochars
The SEM images of the biochar samples are shown in Fig. 3. Conventional pyrolysis of RTW leads to the formation of a layered and foliated structure. The composition of the biochar samples is shown in Table 1 and 2. The biochar produced by conventional pyrolysis contains 45.4% C, 5.5% H, 2.7% N, 0.1% S, and 46.3% O. In addition, the EDS analysis in Fig. 3 b also detected Mg (0.8%), Al (2.2%), and K (0.8%), the proportions of which were less than 1%, as shown in Table 2. When the RTW was treated with KNO3, carbonization and dehydroxylation of the biochar increased along with gasification. This could be due to a reduction in the activation energy of RTW decomposition. Namely, the reaction of carbon with KNO3 resulted in KNO2 and CO2 formation (C + 2KNO3→KNO2 + CO2) [47], causing the removal of oxygen and the formation of pores in the leaf structure in Fig. 3 a via removal of gases. In addition, the decrease in the oxygen content of BC-KNO3 in Table 1 confirms the removal of oxygen by KNO3 treatment.
Prior to pyrolysis, treatment of RTW with NaKCO3 resulted in increased char formation (Table 1) compared to conventional pyrolysis and KNO3 treatment. In addition, Na and K formed during decomposition of NaKCO3 (2NaKCO3+ C → 2Na + 2 K + 3CO2) [48] cannot be removed from the carbon structure by washing and were measured to be 0.4 and 1.4%, respectively, in Table 2. Treatment with NaKCO3 facilitates the diffusion of sodium and potassium ions into the biomass structure and the breaking of hydrogen bonds. This shifts the decomposition of biomass to lower temperatures, as shown in Fig. 1 c. In addition, the removal of CO2 produced during NaKCO3 decomposition leads to the formation of a lace-like carbonaceous structure (Fig. 3a).
Treatment with NaBH4 of RTW leads to increasing carbonization and deoxygenation due to the reducing effect of NaBH4 in Table 1 and 2. In addition, the reaction NaBH4 + 2H2O → NaBO2 + 4H2 [49, 50] occurs during pyrolysis, leading to dehydration of the biochar, as shown by the FT-IR results (Fig. 4). The boron content of BC was found to be 9.5%, 39.2%, and 39. 8% at 5:1, 3:2, and 3:3 of RTW: NaBH4, respectively. The boron content of BC at 3:2 and 3:3 of RTW:NaBH4 is close, which could be due to the fact that boron is removed during washing or the BC cannot be loaded above a certain ratio. Moreover, the carbon and oxygen content of BC decreased with increasing reduction activity of boron. According to Yang et al. [51], boron doping might result in the efficient usage of carbon electrons for O2 reduction. Consequently, it is thought that doping might change the electronic structure of the carbon materail and add positively charged sites, which play important roles in boosting the oxygen reduction activity. Similar to this, Pan et al. reported that B might change O = C reduction to O–C and produce functional groups called –O–B–O during the pyrolysis process [31].
The SEM images of BC treated with NaBH4 are shown in Fig. 3 a at two different magnifications. With increasing boron loading, the fibers of the RTW open and break up the layered structure into flaky material of different sizes. This is due to the reshaping of the structural morphology of the biochar with the removal of oxygen by dehydration.
3.4 FT-IR
The FT-IR spectrum of the biochar produced by conventional and chemically activated pyrolysis is shown in Fig. 4. The peak at 3314 cm−1 corresponding to the N–H stretch [52] is observed in the BC samples treated with KNO3 and NaKCO3. This may be due to the presence of –NH groups in RTW. The N–H concentration is low in BC and BC-NaBH4 because some of these groups were removed during pyrolysis, as can be seen from the elemental composition of the samples in Table 1.
The band at 3196 cm−1 for all biochar samples can be assigned to water molecules with weak H-bonding [53]. The weak band at 3047 cm−1 observed in BC can be assigned to –CH stretching vibrations [54].
The aromatic C = C vibration is responsible for the peak at 1580 cm−1 [55]. Irrespective of activation chemical, the sharp aromatic C = C peak was observed in all samples.
The band at 1280 cm−1 corresponds to the stretching vibrations of the B–O bonds of “boroxol rings” [56]. The sharp B–O peak was observed for BC treated with NaBH4 as a result of the formation of B–O species via hydroxylation reaction of NaBH4 [49, 50]. The bands at 1280 cm−1 in BC treated with KNO3 and NaKCO3 were related to C–O functional groups [57]. Similarly, the band at 1262 cm−1 were assgined to phenolic –OH stretching or aromatic CO–[58, 59]. This peak was observed in all BC samples except for NaBH4. The absence of –OH groups or CO groups in BC-NaBH4 might be related to dehydroxylation and reduction of BC with NaBH4. The peak at 1244 cm−1 is due to the C–O–C in cellulose chain [60]. For this, significant peaks were observed in biochar treated with NaBH4 although shoulders with very low intensity appear in the RTW pyrolyzed in the presence of KNO3 and NaKCO3.
A band at 1085 cm−1 was observed in biochar treated with KNO3, which is due to more cyclic carbohydrates remain intact after pyrolysis [61]. In other treatments, the intensity of this peak is either very low or absent. This can be attributed to the low efficacy of KNO3 on pyrolysis of RTW. The band at 875 cm−1 [62] detected on all BC samples correspond to formation of carbonates [62].
The peaks observed at 819 cm−1 can be ascribed to out-of-plane C–H bending, indicating the presence of hydrogen atoms in aromatic rings [63]. The peak at 565 cm−1 is assigned to stretching vibrational absorption peak of –OH [64].
FT-IR spectrum of BC samples prepared with mixing at different ratios of RTW and NaBH4 are shown in Fig. 4 b. The highest band intensities at 1572 cm−1 for boron-doped biochar samples indicate B–O stretching vibrations due to either B–OH or NaBO2 formation [65]. The intensity of these peaks increases with increasing boron content. In addition, the peak at 1429 cm−1 detected in BC with highest boron can be attributed to B–O–H bond [66] formed via rehydroxylation of boron oxides. This can be due to the hydratation of NaBO2 formed at higher NaBH4 amounts [67].
3.5 Surface area and pore characteristics
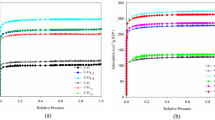
Figure 5 shows the N2 adsorption–desorption and pore characteristics of BC samples produced in the presence and absence of chemical activators. According to the IUPAC classification, the isotherms of biochar samples correspond to the type II [68], indicating the formation of microporous and mesoporous, allowing the formation of multiple adsorption layers with increasing P/P°. The surface area and pore properties of the samples were calculated from the isotherms of N2 adsorption–desorption and are listed in Table 3. Compared to conventional pyrolysis, the treatment with KNO3 and NaKCO3 has only an insignificant effect on the surface area and pore volume of the biochar, but only on the pore distribution (see Fig. 5). Although KNO3 increases the average pore diameter of the biochar, NaKCO3 has an insignificant effect. NaBH4, on the other hand, increases the surface area and pore volume as the average pore diameter decreases. Increasing NaBH4 content has only a negligible effect on surface area and pore volume, as shown in Fig. 6. The highest surface area and pore volume were found to be 158.6 m2/g and 0.22 cm3/g, respectively, for BC-NaBH4 = 3:3. The effect of the chemical activators on the surface area and pore diameter could be related to the concentration and nature of the gas formed during pyrolysis. Indeed, dehydration and/or dehydroxylation of biomass in the presence of activators has a stronger effect on pore formation and carbonization than decarboxylation, as shown in the pore size distribution (Figs. 5 and 6) and the elemental composition (Table 1) of the material.
4 Conclusion
The production of biochar from raw tea waste (RTW) by conventional pyrolysis and in the presence of chemical activators containing potassium nitrate (KNO3), potassium sodium carbonate (NaKCO3), and sodium borohydride (NaBH4) was studied using DTA-TG and DSC. All three activators increased gasification of RTW and enabled the degradation of hemicellulose, cellulose, and lignin. Activation with KNO3 and NaKCO3 increased the hydroxylation of the biochar and only insignificantly affects surface area and pore properties of the biochar. However, treatment with NaBH4 of RTW resulted into a decrease in OH groups and formation of NaBO2 and H2. Increasing the NaBH4 content resulted in rehydroxylation of the biochar with the hydration of NaBO2. Moreover, the surface area and total pore volume increased twofold in the presence of NaBH4 during pyrolysis.
The comparison of the activation chemicals showed that NaBH4 can be a suitable activation chemical to produce a material with low oxygen and OH content and larger surface area. The results indicate that depending on the application such as agriculture, adsorption, catalyst, and biological application, the chemical activation compound of biomass can be selected.
Data availability
Not applicable.
References
Ahmad M, Rajapaksha AU, Lim JE et al (2014) Biochar as a sorbent for contaminant management in soil and water: a review. Chemosphere 99:19–33. https://doi.org/10.1016/j.chemosphere.2013.10.071
Yahya MA, Al-Qodah Z, Ngah CWZ (2015) Agricultural bio-waste materials as potential sustainable precursors used for activated carbon production: a review. Renew Sustain Energy Rev 46:218–235. https://doi.org/10.1016/J.RSER.2015.02.051
Nguyen TB, Truong QM, Chen CW et al (2022) Mesoporous and adsorption behavior of algal biochar prepared via sequential hydrothermal carbonization and ZnCl2 activation. Bioresour Technol 346:126351. https://doi.org/10.1016/J.BIORTECH.2021.126351
Zhang C, Sun S, He S, Wu C (2022) Direct air capture of CO2 by KOH-activated bamboo biochar. J Energy Inst 105:399–405. https://doi.org/10.1016/J.JOEI.2022.10.017
Ateş A (2023) The effect of microwave and ultrasound activation on the characteristics of biochar produced from tea waste in the presence of H3PO4 and KOH. Biomass Convers Biorefinery 13:9075–9094. https://doi.org/10.1007/s13399-021-01838-7
Acemioğlu B (2022) Removal of a reactive dye using NaOH-activated biochar prepared from peanut shell by pyrolysis process. Int J Coal Prep Util 42:671–693. https://doi.org/10.1080/19392699.2019.1644326
Ateş A, Mert Y, Timko MT (2023) Evaluation of characteristics of raw tea waste-derived adsorbents for removal of metals from aqueous medium. Biomass Convers Biorefinery 13:7811–7826. https://doi.org/10.1007/s13399-021-01721-5
Zhang Y, Jiao X, Liu N et al (2020) Enhanced removal of aqueous Cr(VI) by a green synthesized nanoscale zero-valent iron supported on oak wood biochar. Chemosphere 245:125542. https://doi.org/10.1016/J.CHEMOSPHERE.2019.125542
Akhil D, Lakshmi D, Kartik A et al (2021) Production, characterization, activation and environmental applications of engineered biochar: a review. Environ Chem Lett 19:2261–2297. https://doi.org/10.1007/s10311-020-01167-7
Chen W, Gong M, Li K et al (2020) Insight into KOH activation mechanism during biomass pyrolysis: chemical reactions between O-containing groups and KOH. Appl Energy 278:115730. https://doi.org/10.1016/J.APENERGY.2020.115730
Fu Z, Sun K, Fan H et al (2022) Understanding evolution of the products and emissions during chemical activation of furfural residue with varied potassium salts. J Clean Prod 357:131936. https://doi.org/10.1016/J.JCLEPRO.2022.131936
Güzel F, Sayğılı H, Akkaya Sayğılı G et al (2017) Optimal oxidation with nitric acid of biochar derived from pyrolysis of weeds and its application in removal of hazardous dye methylene blue from aqueous solution. J Clean Prod 144:260–265. https://doi.org/10.1016/j.jclepro.2017.01.029
Wang L, Sun F, Hao F et al (2020) A green trace K2CO3 induced catalytic activation strategy for developing coal-converted activated carbon as advanced candidate for CO2 adsorption and supercapacitors. Chem Eng J 383:123205. https://doi.org/10.1016/j.cej.2019.123205
McKee DW, Spiro CL, Lamby EJ (1984) The inhibition of graphite oxidation by phosphorus additives. Carbon N Y 22:285–290. https://doi.org/10.1016/0008-6223(84)90172-6
Nishimura M, Iwasaki S, Horio M (2009) The role of potassium carbonate on cellulose pyrolysis. J Taiwan Inst Chem Eng 40:630–637. https://doi.org/10.1016/J.JTICE.2009.05.005
Zhu L, Zhao N, Tong L, Lv Y (2018) Structural and adsorption characteristics of potassium carbonate activated biochar. RSC Adv 8:21012–21019. https://doi.org/10.1039/c8ra03335h
Mai T-T, Vu D-L, Huynh D-C et al (2019) Cost-effective porous carbon materials synthesized by carbonizing rice husk and K2CO3 activation and their application for lithium-sulfur batteries. J Sci Adv Mater Devices 4:223–229. https://doi.org/10.1016/j.jsamd.2019.04.009
Demir M, Doguscu M (2022) Preparation of porous carbons using NaOH, K2CO3, Na2CO3 and Na2S2O3 activating agents and their supercapacitor application: a comparative study. Chemistry Select 7:e202104295. https://doi.org/10.1002/slct.202104295
Mu J, Li Q, Kong X et al (2019) Characterization of hierarchical porous carbons made from bean curd via K2CO3 activation as a supercapacitor electrode. ChemElectroChem 6:4022–4030. https://doi.org/10.1002/celc.201900962
Arie AA, Kristianto H, Cengiz EC, Demir-Cakan R (2020) Preparation of salacca peel-based porous carbons by K 2 CO 3 activation method as cathode materials for LiS battery. Carbon Lett 30:207–213. https://doi.org/10.1007/s42823-019-00085-1
Ma B, Huang Y, Nie Z et al (2019) Facile synthesis of Camellia oleifera shell-derived hard carbon as an anode material for lithium-ion batteries. RSC Adv 9:20424–20431. https://doi.org/10.1039/C9RA03345A
Xi Y, Wang Y, Yang D et al (2019) K2CO3 activation enhancing the graphitization of porous lignin carbon derived from enzymatic hydrolysis lignin for high performance lithium-ion storage. J Alloys Compd 785:706–714. https://doi.org/10.1016/j.jallcom.2019.01.039
Seifi H, Masoum S, Tafreshi SAH et al (2020) Highly porous carbon from microalga, Chlorella vulgaris, as an electrochemical hydrogen storage material. J Electrochem Soc 167:120525. https://doi.org/10.1149/1945-7111/abaf73
Lv L, Huang S, Zhou C, Ma W (2023) Biochar activated by potassium carbonate to load organic phase change material: better performance and environmental friendliness. Ind Crops Prod 204:117184. https://doi.org/10.1016/j.indcrop.2023.117184
Wongmat Y, Wagner DR (2022) Effect of potassium salts on biochar pyrolysis. Energies 15(16):5779. https://doi.org/10.3390/en15165779
Lin Z, Wang X (2013) Nanostructure engineering and doping of conjugated carbon nitride semiconductors for hydrogen photosynthesis. Angew Chemie 125:1779–1782. https://doi.org/10.1002/ange.201209017
Zhang J, Koubaa A, Xing D et al (2020) Improving lignocellulose thermal stability by chemical modification with boric acid for incorporating into polyamide. Mater Des 191:108589. https://doi.org/10.1016/j.matdes.2020.108589
Zhang X, Gang DD, Zhang J et al (2022) Insight into the activation mechanisms of biochar by boric acid and its application for the removal of sulfamethoxazole. J Hazard Mater 424:127333. https://doi.org/10.1016/j.jhazmat.2021.127333
Dou J, Cheng J, Lu Z et al (2022) Biochar co-doped with nitrogen and boron switching the free radical based peroxydisulfate activation into the electron-transfer dominated nonradical process. Appl Catal B Environ 301:120832. https://doi.org/10.1016/j.apcatb.2021.120832
Gao S, Wang Z, Wang H et al (2022) Peroxydisulfate activation using B-doped biochar for the degradation of oxytetracycline in water. Appl Surf Sci 599:153917. https://doi.org/10.1016/j.apsusc.2022.153917
Pan G, Wei J, Xu M et al (2023) Insight into boron-doped biochar as efficient metal-free catalyst for peroxymonosulfate activation: ımportant role of-OBO-moieties. J Hazard Mater 445:130479. https://doi.org/10.1016/j.jhazmat.2022.130479
Choong Z-Y, Gasim MF, Zhou T et al (2023) The influence of B heteroatom concentrations on the physiochemical properties of N, B-co-doped biochar for peroxymonosulfate activation in ciprofloxacin removal. J Water Process Eng 51:103468. https://doi.org/10.1016/j.jwpe.2022.103468
Ateş A, Aydemir B, Öksüz KE (2023) Investigation of physicochemical and biological properties of boron-doped biochar. Biomass Convers Biorefin. https://doi.org/10.1007/s13399-023-04567-1
Sousa É, Rocha L, Jaria G et al (2020) Microwave-driven production of carbon adsorbents from biomass waste and their application to the removal of antibiotics from water. Sociedade Portuguesa de Qu, Lisboa. https://digital.csic.es/bitstream/10261/234873/1/834814.pdf
Yakaboylu GA, Jiang C, Yumak T et al (2021) Engineered hierarchical porous carbons for supercapacitor applications through chemical pretreatment and activation of biomass precursors. Renew Energy 163:276–287. https://doi.org/10.1016/j.renene.2020.08.092
Uzun BB, Apaydin-Varol E, Ateş F et al (2010) Synthetic fuel production from tea waste: characterisation of bio-oil and bio-char. Fuel 89:176–184. https://doi.org/10.1016/j.fuel.2009.08.040
Yang H, Yan R, Chen H et al (2007) Characteristics of hemicellulose, cellulose and lignin pyrolysis. Fuel 86:1781–1788. https://doi.org/10.1016/j.fuel.2006.12.013
Weber K, Quicker P (2018) Properties of biochar. Fuel 217:240–261. https://doi.org/10.1016/j.fuel.2017.12.054
Bene O, Konings RJM, Wurzer S et al (2010) A DSC study of the NaNO3–KNO3 system using an innovative encapsulation technique. Thermochim Acta 509:62–66. https://doi.org/10.1016/J.TCA.2010.06.003
Chen X, Lu K, Xiao Y et al (2022) Investigation on the ınhibition of aluminum dust explosion by sodium bicarbonate and ıts solid product sodium carbonate. ACS Omega 7:617–628. https://doi.org/10.1021/acsomega.1c05224
Wei Y, Shen C, Xie J, Bu Q (2020) Study on reaction mechanism of superior bamboo biochar catalyst production by molten alkali carbonates pyrolysis and its application for cellulose hydrolysis. Sci Total Environ 712:136435. https://doi.org/10.1016/j.scitotenv.2019.136435
Xi S, Zhang P, Fu Y et al (2020) Hydrogen release: thermodynamic and kinetic studies of NaBH4 activated by different zeolite nanoparticles. Energy Fuels 34:10218–10224. https://doi.org/10.1021/acs.energyfuels.0c01995
Li K, Ma S, Xu S et al (2021) The mechanism changes during bisphenol A degradation in three iron functionalized biochar/peroxymonosulfate systems: The crucial roles of iron contents and graphitized carbon layers. J Hazard Mater 404:124145. https://doi.org/10.1016/j.jhazmat.2020.124145
Mondal S, Derebe AT, Wang K (2018) Surface functionalized carbon microspheres for the recovery of copper ion from refinery wastewater. Korean J Chem Eng 35:147–152. https://doi.org/10.1007/s11814-017-0245-4
Burgess JS, Acharya CK, Lizarazo J et al (2008) Boron-doped carbon powders formed at 1000 C and one atmosphere. Carbon N Y 46:1711–1717. https://doi.org/10.1016/j.carbon.2008.07.022
Yu J, Feng H, Tang L et al (2020) Metal-free carbon materials for persulfate-based advanced oxidation process: microstructure, property and tailoring. Prog Mater Sci 111:100654. https://doi.org/10.1016/j.pmatsci.2020.100654
Xin G, Wang Y, Zang J et al (2016) Temperature tuned carbon morphologies derived from flexible graphite sheets in KNO3 molten salt. Carbon N Y 98:221–224. https://doi.org/10.1016/j.carbon.2015.11.016
Sun J, Lu Y, Yang H et al (2018) Rechargeable Na-CO2 batteries starting from cathode of Na2CO3 and carbon nanotubes. Research 2018:1–9. https://doi.org/10.1155/2018/6914626
Marrero-Alfonso EY, Gray JR, Davis TA, Matthews MA (2007) Minimizing water utilization in hydrolysis of sodium borohydride: the role of sodium metaborate hydrates. Int J Hydrogen Energy 32:4723–4730. https://doi.org/10.1016/j.ijhydene.2007.08.014
Laversenne L, Goutaudier C, Chiriac R et al (2008) Hydrogen storage in borohydrides comparison of hydrolysis conditions of LiBH 4, NaBH 4 and KBH 4. J Therm Anal Calorim 94:785–790. https://doi.org/10.1007/s10973-008-9073-4
Yang L, Jiang S, Zhao Y et al (2011) Boron-doped carbon nanotubes as metal-free electrocatalysts for the oxygen reduction reaction. Angew Chemie Int Ed 50:7132–7135. https://doi.org/10.1002/anie.201101287
Kim JH, Choi SH, Park MG et al (2022) Biodegradation of polyurethane by Japanese carpenter bee gut-associated symbionts Xanthomonas sp. HY-71, and its potential application on bioconversion. Environ Technol Innov 28:102822. https://doi.org/10.1016/J.ETI.2022.102822
Nawrocka A, Krekora M, Niewiadomski Z, Miś A (2018) FTIR studies of gluten matrix dehydration after fibre polysaccharide addition. Food Chem 252:198–206. https://doi.org/10.1016/J.FOODCHEM.2018.01.110
Krishnakumar V, Surumbarkuzhali N, Muthunatesan S (2009) Scaled quantum chemical studies on the vibrational spectra of 4-bromo benzonitrile. Spectrochim Acta Part A Mol Biomol Spectrosc 71:1810–1813. https://doi.org/10.1016/J.SAA.2008.06.037
Kim KH, Kim J-Y, Cho T-S, Choi JW (2012) Influence of pyrolysis temperature on physicochemical properties of biochar obtained from the fast pyrolysis of pitch pine (Pinus rigida). Bioresour Technol 118:158–162. https://doi.org/10.1016/j.biortech.2012.04.094
Feng N, Zheng A, Wang Q et al (2011) Boron environments in B-doped and (B, N)-codoped TiO2 photocatalysts: a combined solid-state NMR and theoretical calculation study. J Phys Chem C 115:2709–2719. https://doi.org/10.1021/jp108008a
Melo CA, Junior FHS, Bisinoti MC et al (2017) Transforming sugarcane bagasse and vinasse wastes into hydrochar in the presence of phosphoric acid: an evaluation of nutrient contents and structural properties. Waste Biomass Valorization 8:1139–1151. https://doi.org/10.1007/s12649-016-9664-4
Chen X, Chen G, Chen L et al (2011) Adsorption of copper and zinc by biochars produced from pyrolysis of hardwood and corn straw in aqueous solution. Bioresour Technol 102:8877–8884. https://doi.org/10.1016/j.biortech.2011.06.078
Kumar M, Shukla SK, Upadhyay SN, Mishra PK (2020) Analysis of thermal degradation of banana (Musa balbisiana) trunk biomass waste using iso-conversional models. Bioresour Technol 310:123393. https://doi.org/10.1016/j.biortech.2020.123393
Mothé C, de Miranda I (2009) Characterization of sugarcane and coconut fibers by thermal analysis and FTIR. J Therm Anal Calorim 97:661–665. https://doi.org/10.1007/s10973-009-0346-3
Quosai P, Anstey A, Mohanty AK, Misra M (2018) Characterization of biocarbon generated by high-and low-temperature pyrolysis of soy hulls and coffee chaff: For polymer composite applications. R Soc open Sci 5:171970. https://doi.org/10.1098/rsos.171970
Shaw CL (2022) An evaluation of the infrared 630 cm−1 OH libration band in bone mineral as evidence of fire in the archaeological record. J Archaeol Sci Reports 46:103655. https://doi.org/10.1016/J.JASREP.2022.103655
Singh RK, Ruj B, Sadhukhan AK, Gupta P (2020) A TG-FTIR investigation on the co-pyrolysis of the waste HDPE, PP, PS and PET under high heating conditions. J Energy Inst 93:1020–1035. https://doi.org/10.1016/j.joei.2019.09.003
Lianke Z, Yang W, Weida W et al (2018) The preparation of biochar-supported nano-hydroxyapatite and its adsorption of Pb2+. Acta Sci Circumstantiae 37:3492–3501. https://doi.org/10.16085/j.issn.1000-6613.2017-2213
Bethell DE, Sheppard N (1955) The infra-red spectrum and structure of boric acid. Trans Faraday Soc 51:9–15. https://doi.org/10.1039/TF9555100009
Yang Q, Sha J, Wang L et al (2005) Crystalline boron oxide nanowires on silicon substrate. Phys E Low-dimensional Syst Nanostruct 27:319–324. https://doi.org/10.1016/j.physe.2004.12.015
Andrieux J, Laversenne L, Krol O et al (2012) Revision of the NaBO2–H2O phase diagram for optimized yield in the H2 generation through NaBH4 hydrolysis. Int J Hydrogen Energy 37:5798–5810. https://doi.org/10.1016/j.ijhydene.2011.12.106
Thommes M, Kaneko K, Neimark AV et al (2015) Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl Chem 87:1051–1069. https://doi.org/10.1515/pac-2014-1117
Acknowledgements
Ayten Ateş thanks Sivas Cumhuriyet University Scientific Research Foundation (CUBAP) under grant number M-2021-835 for providing financial assistance for this project.
Funding
Open access funding provided by the Scientific and Technological Research Council of Türkiye (TÜBİTAK). Project M-2021–835 of Sivas Cumhuriyet University-Scientific Research (CUBAP) fund provided funding for this work.
Author information
Authors and Affiliations
Contributions
Ayten Ates: conceptualization; investigation; methodology; writing—original draft; review and editing; funding acquisition.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Competing interests
The author declares no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
ATEŞ, A. Investigation of physicochemical and chemical properties of biochar activated with carbonate, nitrate, and borohydride. Biomass Conv. Bioref. (2024). https://doi.org/10.1007/s13399-024-05323-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13399-024-05323-9