Abstract
Introduction
A three-pronged approach to acne treatment combining an antibiotic, antimicrobial, and retinoid may be more efficacious than single/double treatments while potentially reducing antibiotic resistance. This study evaluated the efficacy and safety of the first fixed-dose, triple-combination topical acne product, clindamycin 1.2%/adapalene 0.15%/benzoyl peroxide (BPO) 3.1% gel (CAB) using pooled phase 3 data.
Methods
In two identical phase 3 (N = 183; N = 180), double-blind, 12-week studies, participants aged ≥ 9 years with moderate-to-severe acne were randomized 2:1 to receive once-daily CAB or vehicle gel. Endpoints included ≥ 2-grade reduction from baseline in Evaluator’s Global Severity Score and clear/almost clear skin (treatment success) and least-squares mean percent change from baseline in acne lesion counts. Treatment-emergent adverse events (TEAEs) and cutaneous safety/tolerability were evaluated.
Results
At week 12, 50.0% of participants achieved treatment success with CAB versus 22.6% with vehicle gel (P < 0.001). CAB resulted in > 70% reductions in inflammatory and noninflammatory lesions at week 12 (77.9% and 73.0%, respectively), which were significantly greater than vehicle (57.9% and 48.2%; P < 0.001, both). Most TEAEs were of mild-moderate severity, and < 3% of CAB-treated participants discontinued study/treatment because of AEs. Transient increases from baseline in scaling, erythema, itching, burning, and stinging were observed with CAB, but resolved back to or near baseline values by week 12.
Conclusions
The innovative fixed-dose, triple-combination clindamycin phosphate 1.2%/adapalene 0.15%/BPO 3.1% gel was efficacious and well tolerated in children, adolescents, and adults with moderate-to-severe acne. Half of participants achieved clear/almost clear skin by 12 weeks, rates not previously seen in clinical studies of other topical acne products.
Trial Registration
ClinicalTrials.gov identifier NCT04214639 and NCT04214652.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
A three-pronged approach to acne treatment using an antibiotic, antimicrobial, and a retinoid within an easy-to-use topical formulation could improve efficacy and treatment adherence. |
CAB polymeric mesh gel (clindamycin phosphate 1.2%/adapalene 0.15%/benzoyl peroxide 3.1%)—the first fixed-dose triple-combination acne topical product—demonstrated superior efficacy to vehicle and component dyads, with good safety/tolerability in a phase 2 and two phase 3 studies of moderate-to-severe acne. |
This post hoc analysis further evaluated the efficacy and safety of CAB gel using pooled phase 3 data. |
What was learned from the study? |
Results from this post hoc analysis of two pooled randomized, double-blind, vehicle-controlled clinical studies in participants with moderate-to-severe acne showed that half of participants treated with CAB gel achieved treatment success by week 12, with > 70% reductions in inflammatory and noninflammatory lesions. |
To our knowledge, observed acne improvements in these studies with CAB were greater than any FDA-approved topical acne treatment to date, making CAB an effective treatment option for children, adolescents, and adults with moderate-to-severe acne. |
Introduction
Acne vulgaris is the most common dermatologic disorder, affecting up to 50 million people in the USA annually [1]. Acne pathogenesis is multifactorial, and includes increased inflammation and sebum production, follicular proliferation of Cutibacterium acnes (formerly Propionibacterium acnes), and abnormal keratinization [2, 3]. As such, combination treatments that simultaneously target multiple pathological mechanisms may provide better efficacy compared to monotherapy [4]. Furthermore, combination treatments that are delivered in a fixed-dose formulation may improve patient adherence, and thereby treatment success, by reducing treatment complexity [5].
The most recently published acne treatment guidelines in the USA and the EU recommend combination treatment for most patients [2, 6]. Several topical dual-combination prescription products currently approved in the USA contain an antibiotic or a retinoid in combination with benzoyl peroxide (BPO). Clindamycin phosphate 1.2%/adapalene 0.15%/BPO 3.1% gel (CAB; Cabtreo®; Ortho Dermatologics) is the first triple-combination, fixed-dose topical acne treatment approved by the US Food and Drug Administration (FDA), and is indicated for use in patients 12 years of age and older [7]. In addition to being an antibiotic, clindamycin phosphate has anti-inflammatory effects on the skin [8, 9] while adapalene, a third-generation synthetic retinoid, modulates cellular proliferation, differentiation, and keratinization [10, 11]. BPO, a mainstay of topical acne products, is an antimicrobial agent with mild comedolytic and keratolytic activity [12, 13]. Importantly, combining an antibiotic such as clindamycin with BPO has the benefit of reducing the risk of antibiotic resistance, especially during prolonged use [2], which is a core goal of antibiotic stewardship recommended by the Centers for Disease Control and by the Scientific Panel on Antibiotic Use in Dermatology of the American Acne and Rosacea Society [14,15,16]. Overall, efficacy of CAB may be enhanced by its delivery of three active ingredients with overlapping mechanisms of action that simultaneously converge on multiple acne pathophysiological pathways (Fig. 1).
Pathogenesis of acne and treatment with triple-combination CAB gel. Active ingredients in CAB gel address three of the four pathogenic factors in acne: adapalene normalizes epithelial hyperproliferation; clindamycin and benzoyl peroxide reduce C. acnes viability; all three active ingredients have anti-inflammatory properties. Image ©2023. Courtesy of Ortho Dermatologics. BPO benzoyl peroxide, CAB clindamycin phosphate 1.2%/adapalene 0.15%/benzoyl peroxide 3.1%
In a phase 2 study of participants with moderate-to-severe acne, once-daily treatment with CAB was well tolerated and led to significantly greater acne improvements versus vehicle gel and three dyad component gels (BPO/adapalene, clindamycin phosphate/BPO, and clindamycin phosphate/adapalene) [17]. In two identical phase 3, randomized, double-blind, vehicle-controlled trials, 12 weeks of CAB treatment led to a significantly greater percentage of patients achieving treatment success and significantly greater reductions in inflammatory and noninflammatory lesions compared with vehicle [18]. Across all three clinical studies, acne lesions were reduced by > 70% and approximately 50% of CAB-treated participants achieved treatment success. In this manuscript, data from the phase 3 studies were pooled to more robustly examine the efficacy, quality of life impacts, safety, and tolerability of CAB gel in participants with moderate-to-severe acne.
Methods
Study Design and Participants
A detailed description of the study designs and methods of the two phase 3 studies (NCT04214639 and NCT04214652) has been published previously [18]. In brief, eligible participants were aged 9 years or older with moderate or severe facial acne (Evaluator’s Global Severity Score [EGSS] 3 or 4, see Table S1 in the Electronic Supplementary Material), ≥ 30 to ≤ 100 inflammatory facial lesions (papules/pustules/nodules), ≥ 35 to ≤ 150 noninflammatory facial lesions (closed/open comedones), and ≤ 2 facial nodules. Participants were randomized 2:1 to receive 12 weeks of once-daily CAB or vehicle gel. For optimal skin moisturization, cleaning, and protection, CeraVe® hydrating cleanser (L’Oréal, New York, NY), CeraVe® moisturizing lotion, and sunscreen were provided as needed.
These studies were carried out in accordance with principles of Good Clinical Practice (GCP) and the Declaration of Helsinki. Study protocols across all study centers were approved by the same central Institutional Review Board (Advarra IRB, Columbus, MD, USA). All participants or their legal guardians provided written informed consent before entering the clinical trial from which the data were derived. Additional informed consent was obtained from all individual participants for whom identifying information/photographs are included in this article.
Efficacy and Safety Evaluations
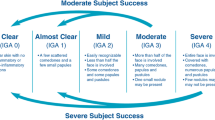
Participants were assessed at screening, baseline, and weeks 2, 4, 8, and 12. Efficacy assessments included facial inflammatory and noninflammatory lesion counts and treatment success, defined as the proportion of participants achieving a ≥ 2-grade reduction from baseline in facial EGSS and a score of 0 or 1 (clear or almost clear, see Table S1 in the Electronic Supplementary Material). At baseline and week 12, participants completed the Acne-Specific Quality of Life (Acne-QoL) questionnaire, which covers four domains: self-perception, role-emotional, role-social, and acne symptoms. Questions within each domain are scored from 0 (extremely) to 6 (not at all); an increase in score relative to baseline equates to an improvement in health-related quality of life [19].
Investigator assessments of cutaneous safety (scaling, erythema, hypopigmentation, hyperpigmentation) and participant assessments of tolerability (itching, burning, stinging) for the face were each scored at all post-screening visits using a 4-point scale (0 = none, 1 = mild, 2 = moderate, 3 = severe). Adverse events (AEs) were monitored throughout the studies.
Statistical Analysis
The intent-to-treat (ITT) population included all randomized participants in both studies. Those participants who were confirmed to have used the study drug at least once were included in the safety population. For both studies, co-primary endpoints consisted of the percentage of participants achieving treatment success at week 12 and absolute changes from baseline at week 12 in inflammatory and noninflammatory lesion counts. Secondary endpoints included least-squares mean percent changes in lesion counts from baseline at weeks 4, 8, and 12, and the percentage of participants achieving ≥ 2-grade reduction from baseline in EGSS at week 12.
Percent change from baseline in inflammatory and noninflammatory lesion counts was analyzed using analysis of covariance (ANCOVA), with factors of treatment group and analysis center and the respective baseline lesion count as a covariate; when significant skewness was observed, a nonparametric method was utilized in which data were rank transformed prior to analysis. Treatment success was analyzed using a logistic regression test (using Firth’s penalized likelihood) with factors of treatment group and analysis center. For Acne-QoL questionnaire responses, domain scores were transformed from 0–6 to 1–7 prior to ANCOVA with factors of treatment group and analysis center, and the respective baseline domain score as a covariate. For all efficacy assessments except Acne-QoL, multiple imputation was used to impute missing values using the Markov Chain Monte Carlo method. All statistical analyses were performed using SAS® version 9.4 or later. Statistical significance was based on two-tailed tests of the null hypothesis resulting in P ≤ 0.05.
Additionally, co-primary and select secondary endpoints (percent change in inflammatory and noninflammatory lesions; EGSS reduction of ≥ 2 grade from baseline) at week 12 were analyzed for the subgroup of participants aged 9–25 years. This age cutoff was chosen to assess efficacy in pediatric and adolescent participants, as acne in patients older than 25 years is typically considered “adult” acne [20, 21].
Cutaneous safety and tolerability assessments and AEs were summarized using descriptive statistics. Medical Dictionary for Regulatory Activities (MeDRA) terminology was used to classify AEs. No imputations were made for missing safety data.
Results
Participant Disposition and Demographics
A total of 363 participants were randomized to CAB or vehicle gel in the two trials (Fig. 2); all participants were included in both the ITT and safety populations. The most common reasons for study discontinuation were participant request and lost to follow-up.
Pooled participant disposition. A total of 363 participants were randomized to CAB or vehicle gel in the two trials; all participants were included in both the ITT and safety populations. aWithdrawal by a parent/guardian. bIncludes discontinuation due to COVID-19 disruption, lack of efficacy, and progressive disease. CAB clindamycin phosphate 1.2%/adapalene 0.15%/benzoyl peroxide 3.1%, ITT intent to treat
Demographics and baseline disease characteristics are shown in Table 1. The mean age was similar for the CAB and vehicle gel groups (approximately 20 years), with five participants aged < 12 years. Most participants were female (58.4%), White (73.6%), and had moderate acne at baseline (91.2%).
Efficacy and Quality of Life
At week 12, CAB was superior to vehicle gel on all efficacy assessments (Fig. 3, see Table S2 in the Electronic Supplementary Material). Half of participants treated with CAB achieved treatment success by week 12, compared with 22.6% of participants treated with vehicle (P < 0.001). Mean absolute reductions in inflammatory and noninflammatory lesions from baseline were significantly greater with CAB versus vehicle (inflammatory, 29.0 vs 21.0; noninflammatory, 35.3 vs 22.7; P < 0.001, each). Least-squares mean percent changes from baseline in inflammatory and noninflammatory lesion counts were also significantly greater with CAB versus vehicle gel as early as week 4 (P < 0.001, all), with reductions of > 70% achieved with CAB treatment by week 12 (Fig. 4). For participants in the 9–25 years subgroup, treatment success rates and lesion count reductions were similar to the overall participant population (see Table S3 in the Electronic Supplementary Material). Representative images showing acne improvements in participants treated with CAB are shown in Fig. 5.
Primary efficacy endpoints at week 12 (ITT population; pooled). At week 12, treatment success rates and mean absolute reductions in inflammatory and noninflammatory lesions from baseline were significantly greater with CAB versus vehicle gel. ***P < 0.001 vs vehicle. aTreatment success was defined as ≥ 2-grade reduction from baseline in Evaluator’s Global Severity Score and a score of 0 (clear) or 1 (almost clear). Multiple imputation with the Markov Chain Monte Carlo method used to impute missing values. CAB clindamycin phosphate 1.2%/adapalene 0.15%/benzoyl peroxide 3.1%, ITT intent to treat, LS least squares
Percent changes from baseline in inflammatory and noninflammatory lesion counts by visit (ITT population; pooled). Least-squares mean percent changes from baseline in inflammatory and noninflammatory lesion counts were significantly greater with CAB versus vehicle gel. ***P < 0.001 vs vehicle. Multiple imputation with the Markov Chain Monte Carlo method used to impute missing values. CAB clindamycin phosphate 1.2%/adapalene 0.15%/benzoyl peroxide 3.1% gel, ITT intent to treat, LS least squares

© 2023. Courtesy of Ortho Dermatologics Study Investigators. Part of this figure was previously published in Stein Gold LS et al., 2023 [18]. EGSS Evaluator’s Global Severity Score, CAB clindamycin phosphate 1.2%/adapalene 0.15%/benzoyl peroxide 3.1% gel, IL inflammatory lesions, NIL noninflammatory lesions
Acne improvements with CAB gel. Participant photos showing acne improvement with CAB gel; individual results may vary. Photographic images
For all four Acne-QoL domain scores, least-squares mean increases (improvements) from baseline at week 12 were significantly greater with CAB than with vehicle (range, CAB 5.2–8.9, vehicle 3.2–5.1; P < 0.001, all; Fig. 6, see Table S4 in the Electronic Supplementary Material).
Acne-Specific Quality of Life questionnaire at week 12 (ITT population; pooled). For all four Acne-QoL domain scores, least-squares mean increases (improvements) from baseline at week 12 were significantly greater with CAB than with vehicle. ***P < 0.001 vs vehicle. No imputation of missing values. Horizontal black lines indicate domain scores at baseline and arrows indicate changes from baseline. Range of possible scores are 5–35 for self-perception, role-emotional, and acne symptoms and 4–28 for role-social. Acne-QoL Acne-Specific Quality of Life questionnaire, CAB clindamycin phosphate 1.2%/adapalene 0.15%/benzoyl peroxide 3.1%, ITT intent to treat, LS least squares
Safety and Tolerability
Treatment-emergent adverse events (TEAEs) were generally mild or moderate and more common among participants treated with CAB (Table 2). The most common treatment-related TEAEs (≥ 2% of participants in any group) were erythema and application site pain, dryness, and irritation (see Table S5 in the Electronic Supplementary Material). No serious AEs were reported in either treatment arm. Discontinuations due to TEAEs were low among CAB-treated participants (< 3%).
Transient increases from baseline in investigator-assessed scaling and erythema and participant-assessed itching, burning, and stinging occurred with CAB treatment. Mean scores for these measures were highest at week 2 (Fig. 7), with substantial decreases at week 4, and resolution back to or near baseline values by week 8. Additionally, erythema decreased to below-baseline levels by week 4 (mean scores: baseline, 0.57; week 4, 0.55). No trends in investigator-assessed hyperpigmentation and hypopigmentation were observed in either treatment arm.
Cutaneous safety and tolerability (safety population; pooled). Mean increases from baseline in scaling, erythema, itching, burning, and stinging with CAB treatment were highest at week 2, with substantial decreases at week 4 and resolution back to or near baseline values by week 8. No imputation of missing values (N values shown for baseline only). Data not shown for hyperpigmentation and hypopigmentation as there were no trends in transient increases over time. BL baseline, CAB clindamycin phosphate 1.2%/adapalene 0.15%/benzoyl peroxide 3.1% gel, VEH vehicle
Discussion
This post hoc analysis of data pooled from two identical phase 3 studies evaluated the efficacy, safety, and tolerability of CAB—a recently approved, innovative fixed-dose, triple-combination clindamycin phosphate 1.2%/adapalene 0.15%/BPO 3.1% gel—in participants with moderate-to-severe acne. Compared with vehicle gel, CAB demonstrated superior efficacy on all three co-primary endpoints and was well tolerated. Half of CAB-treated participants achieved treatment success by week 12. In addition, CAB yielded > 50% reductions in inflammatory lesions as early as week 4, and > 70% reductions in inflammatory and noninflammatory lesions by week 12.
To our knowledge, observed acne improvements in these CAB studies were greater than any other FDA-approved topical acne treatment or systemic antibiotic to date, though patient populations and design may differ across studies. Specifically, percent reductions in facial acne lesions were greater with CAB gel (> 70%) compared with other topical monotherapies (range 25–66.2%) [22,23,24,25,26,27,28,29], topical dyads (21.9–68.7%) [30,31,32,33,34,35,36,37], or oral tetracyclines (43.1–52.2%) [38]. Further, when compared with other FDA-approved combination topical acne treatments with phase 3 studies that report treatment success at week 12, CAB was associated with treatment success in a greater percentage of participants (50% vs 21.5–39.9%) [39].
The results from this pooled analysis are consistent with a phase 2 study [17] (Fig. 8), and with a previous side-by-side analysis of these phase 3 trials [18], in which CAB treatment was superior to vehicle, and was associated with treatment success in around half of participants (range 49.6–52.5%) and with > 70% reductions in inflammatory and noninflammatory lesions (range 71.0–80.1%). In the phase 2 study, treatment efficacy with CAB was also superior to component dyads [17] (Fig. 8). In keeping with these findings, a meta-analysis of acne treatments found that triple-combination therapies like CAB that include an antibiotic (oral or topical), retinoid (topical), and BPO were among the top two most efficacious of all treatments assessed for both treatment success and total lesion count reductions, only second to oral isotretinoin [30].
Treatment successa and lesion reductions at week 12 across CAB phase 2 and phase 3 clinical studies (ITT populations). In a phase 2 study [17] and in this pooled analysis of the phase 3 trials, CAB treatment was superior to vehicle, with treatment success in around half of participants, and > 70% reductions in inflammatory and noninflammatory lesions. ***P ≤ 0.001 vs vehicle; ## P < 0.01, ###P ≤ 0.001 vs CAB gel. Values have been adjusted for multiple imputation. aDefined as percentage of participants achieving ≥ 2-grade reduction from baseline in Evaluator’s Global Severity Score and a score of 0 (clear) or 1 (almost clear). ADAP adapalene 0.15%, BPO benzoyl peroxide 3.1%, CLIN clindamycin phosphate 1.2%, CAB clindamycin phosphate 1.2%/adapalene 0.15%/benzoyl peroxide 3.1% gel, ITT intent to treat, LS least squares, VEH vehicle gel
Acne onset often follows hormonal changes during puberty, with trends toward earlier puberty associated with increasing acne prevalence among preteen children [40, 41]. In adolescents, acne can develop rapidly and present as more severe disease compared with adults [42]. Further, while patients aged > 18 years may be considered adults, the pathogenesis, presentation, and response to treatment may be distinct for those aged 18–24 years compared with those aged ≥ 25 years (“adult acne”) [43]. Pooling data from participants 9–25 years of age in the phase 3 studies of CAB enabled an analysis of efficacy in this subpopulation, which was similar to the overall population. When multiple imputation analyses were used, CAB treatment at week 12 was associated with 77.9% and 72.9% reductions in inflammatory and noninflammatory lesions in these younger patients, respectively (overall population 77.9% and 73.0%), and treatment success in 48.2% of participants (overall 50.0%). This suggests that CAB is highly efficacious regardless of any age-related acne differences. Moreover, delivering three ingredients in a simple, fixed-dose treatment regimen with once-daily application may additionally benefit treatment adherence, which has been shown to be significantly improved when patients are prescribed only one product versus two or three acne treatments [44]. This is especially compelling for patients aged < 25 years, among whom adherence is particularly low [45]. Notably, CAB gel is indicated for use in patients aged 12 years and older as a result of the limited number of participants aged < 12 years enrolled in these studies.
Combination treatment with multiple active ingredients carries the risk of increased tolerability concerns. However, in this analysis, CAB gel was found to be safe and well tolerated, consistent with previously published findings from the phase 2 and phase 3 studies [17, 18]. Further, in the phase 2 studies, cutaneous safety and tolerability assessments with CAB were better than BPO/adapalene and generally similar to clindamycin phosphate/adapalene, indicating that the additional product in the triple combination did not worsen tolerability [17]. In the phase 3 studies, mean tolerability scores with CAB treatment were all < 0.7 (1 = mild). Most TEAEs were of mild-to-moderate severity, and the rate of discontinuation due to AEs was low in the CAB group (< 3%). Several factors may contribute to CAB gel’s safety and tolerability profile. BPO and adapalene in this polymeric formulation have been micronized to allow for even distribution over the skin. This improves penetration into the pilosebaceous unit, which serves the dual function of enhancing tolerability and improving efficacy [10, 46]. Moisturizing agents within the gel vehicle may help reduce irritation common with many acne treatments containing BPO and/or retinoids. The anti-inflammatory properties of clindamycin may also provide a moderating effect on cutaneous irritation with BPO and adapalene [8]. Finally, the active ingredients are delivered in a pH-balanced, aqueous gel that contains the humectant propylene glycol and is free from preservatives, alcohol, occlusive agents, and surfactants.
Acne can negatively impact self-esteem, social relationships, and psychological well-being, resulting in a deleterious effect on patients’ quality of life [47]. Therefore, there is a need for treatments that are not only efficacious and safe but also improve quality of life. In the present analysis, CAB-treated participants had significantly greater increases from baseline at week 12 in all Acne-QoL domain scores compared with vehicle-treated participants, indicating greater improvement in quality of life.
Some limitations must be considered in the interpretation of these data. The 12-week treatment time course in these studies is short compared with treatment in the real world, which may require 6 months for maximum benefits to be seen [48]. However, the rapid and sustained effects of CAB on moderate-to-severe acne within the 12-week timeframe [49] suggests further improvements could occur with continued treatment, especially as both inflammatory and noninflammatory lesion counts continued to improve in the 4 weeks preceding the end of the study without evidence of plateauing efficacy. This pooled analysis also does not account for the fact that acne presentation and associated sequelae can vary across demographic factors such as skin type, age, and sex; this may be addressed by future post hoc analyses in subpopulations of interest. Additionally, interobserver bias and variation can be associated with global acne severity assessments such as the EGSS [31]. Finally, these phase 3 studies assessed safety and efficacy of CAB in participants with moderate-to-severe facial acne; this might limit the generalizability of these findings to patients with milder forms of acne or those with non-facial acne. Though the study monitored truncal acne, data on the trunk are not shown here as few patients chose to apply study drug to these areas (neck, upper chest, upper back, and shoulders).
Conclusion
In two pooled randomized, double-blind, vehicle-controlled clinical studies of participants with moderate-to-severe facial acne, CAB gel (clindamycin phosphate 1.2%/adapalene 0.15%/BPO 3.1%) demonstrated superior efficacy to vehicle after 12 weeks of once-daily treatment. CAB was associated with > 70% reductions from baseline in inflammatory and noninflammatory lesions, treatment success in 50% of participants, and a favorable safety and tolerability profile. These data demonstrate that CAB gel—the first fixed-dose, triple-combination topical treatment for acne—may be an effective treatment option for children, adolescents, and adults with moderate-to-severe acne.
Data Availability
The datasets generated during and/or analyzed during the current study are available upon reasonable request.
References
Bickers DR, Lim HW, Margolis D, et al. The burden of skin diseases: 2004 a joint project of the American Academy of Dermatology Association and the Society for Investigative Dermatology. J Am Acad Dermatol. 2006;55(3):490–500.
Zaenglein AL, Pathy AL, Schlosser BJ, et al. Guidelines of care for the management of acne vulgaris. J Am Acad Dermatol. 2016;74(5):945–73.e33.
Gollnick HP, Zouboulis CC, Akamatsu H, et al. Pathogenesis and pathogenesis related treatment of acne. J Dermatol. 1991;18(9):489–99.
Kircik LH. Synergy and its clinical relevance in topical ance therapy. J Clin Aesth Dermaol. 2011;4(11):30–3.
Moradi Tuchayi S, Alexander TM, Nadkarni A, Feldman SR. Interventions to increase adherence to acne treatment. Patient Prefer Adherence. 2016;10:2091–6.
Nast A, Dréno B, Bettoli V, et al. European evidence-based (S3) guideline for the treatment of acne - update 2016 - short version. J Eur Acad Dermatol Venereol. 2016;30(8):1261–8.
FDA approves Cabtreo™ (clindamycin phosphate, adapalene and benzoyl peroxide) topical gel for the treatment of acne vulgaris in patients twelve years of age and older. Bausch Health. Updated October 20, 2023. https://ir.bauschhealth.com/news-releases/2023/10-20-2023. Accessed 26 Oct 2023.
Del Rosso JQ, Schmidt NF. A review of the anti-inflammatory properties of clindamycin in the treatment of acne vulgaris. Cutis. 2010;85(1):15–24.
Armillei MK, Lomakin IB, Del Rosso JQ, et al. Scientific rationale and clinical basis for clindamycin use in the treatment of dermatologic disease. Antibiotics. 2024;13(3):270.
Baldwin H, Webster G, Stein Gold L, et al. 50 years of topical retinoids for acne: evolution of treatment. Am J Clin Dermatol. 2021;22(3):315–27.
Shroot B, Michel S. Pharmacology and chemistry of adapalene. J Am Acad Dermatol. 1997;36(6 Pt 2):S96–103.
Dutil M. Benzoyl peroxide: enhancing antibiotic efficacy in acne management. Skin Therapy Lett. 2010;15(10):5–7.
Waller JM, Dreher F, Behnam S, et al. “Keratolytic” properties of benzoyl peroxide and retinoic acid resemble salicylic acid in man. Skin Pharmacol Physiol. 2006;19(5):283–9.
Antibiotic stewardship statement for antibiotic guidelines – recommendations of the HICPAC. Centers for Disease Control and Prevention. Updated December 28, 2018. https://www.cdc.gov/hicpac/recommendations/antibiotic-stewardship-statement.html. Accessed 26 Oct 2023.
Del Rosso JQ, Rosen T, Thiboutot D, et al. Status report from the Scientific Panel on Antibiotic Use in Dermatology of the American Acne and Rosacea Society: Part 3: current perspectives on skin and soft tissue infections with emphasis on methicillin-resistant Staphylococcus aureus, commonly encountered scenarios when antibiotic use may not be needed, and concluding remarks on rational use of antibiotics in dermatology. J Clin Aesthet Dermatol. 2016;9(6):17–24.
Del Rosso JQ, Webster GF, Rosen T, et al. Status report from the Scientific Panel on Antibiotic Use in Dermatology of the American Acne and Rosacea Society: Part 1: antibiotic prescribing patterns, sources of antibiotic exposure, antibiotic consumption and emergence of antibiotic resistance, impact of alterations in antibiotic prescribing, and clinical sequelae of antibiotic use. J Clin Aesthet Dermatol. 2016;9(4):18–24.
Stein Gold L, Baldwin H, Kircik LH, et al. Efficacy and safety of a fixed-dose clindamycin phosphate 1.2%, benzoyl peroxide 3.1%, and adapalene 0.15% gel for moderate-to-severe acne: a randomized phase II study of the first triple-combination drug. Am J Clin Dermatol. 2022;23(1):93–104.
Stein Gold L, Lain E, Del Rosso JQ, et al. Clindamycin phosphate 1.2%/adapalene 0.15%/benzoyl peroxide 3.1% gel for moderate-to-severe acne: efficacy and safety results from two randomized phase 3 trials. J Am Acad Dermatol. 2023;89(5):927–35.
Martin AR, Lookingbill DP, Botek A, et al. Health-related quality of life among patients with facial acne—assessment of a new acne-specific questionnaire. Clin Exp Dermatol. 2001;26(5):380–5.
Dreno B, Thiboutot D, Layton AM, et al. Large-scale international study enhances understanding of an emerging acne population: adult females. J Eur Acad Dermatol Venereol. 2015;29(6):1096–106.
Skroza N, Tolino E, Mambrin A, et al. Adult acne versus adolescent acne: a retrospective study of 1,167 patients. J Clin Aesth Dermaol. 2018;11(1):21–5.
Clindagel® (clindamycin phosphate) Gel 1% [package insert]. Valeant, Bridgewater, NJ; 2017.
Tazorac® [package insert]. Allergan, Inc., Madison, NJ; 2018.
Altreno® [package insert]. Bausch Health Companies, Inc., Bridgewater, NJ; 1973.
Aklief® [package insert]. Galderma Laboratories, L.P., Fort Worth, TX; 2019.
Amzeeq® [package insert]. Vyne Pharmaceuticals, Inc., Bridgewater, NJ; 1971.
Arazlo® [package insert]. Bausch Health Companies, Inc., Bridgewater, NJ; 1997.
Winlevi® [package insert]. Cassiopea, Inc., Milan, Italy; 2020.
Differin® [package insert]. Galderma Laboratories, L.P., Fort Worth, TX; 1996.
Huang CY, Chang IJ, Bolick N, et al. Comparative efficacy of pharmacological treatments for acne vulgaris: a network meta-analysis of 221 randomized controlled trials. Ann Fam Med. 2023;21(4):358–69.
Tan JK, Fung K, Bulger L. Reliability of dermatologists in acne lesion counts and global assessments. J Cutan Med Surg. 2006;10(4):160–5.
Gold LS, Tan J, Cruz-Santana A, et al. A North American study of adapalene-benzoyl peroxide combination gel in the treatment of acne. Cutis. 2009;84(2):110–6.
Leyden JJ, Berger RS, Dunlap FE, et al. Comparison of the efficacy and safety of a combination topical gel formulation of benzoyl peroxide and clindamycin with benzoyl peroxide, clindamycin and vehicle gel in the treatments of acne vulgaris. Am J Clin Dermatol. 2001;2(1):33–9.
Pariser DM, Rich P, Cook-Bolden FE, Korotzer A. An aqueous gel fixed combination of clindamycin phosphate 1.2% and benzoyl peroxide 3.75% for the once-daily treatment of moderate to severe acne vulgaris. J Drugs Dermatol. 2014;13(9):1083–9.
Stein Gold L, Weiss J, Rueda MJ, et al. Moderate and severe inflammatory acne vulgaris effectively treated with single-agent therapy by a new fixed-dose combination adapalene 0.3 %/benzoyl peroxide 2.5 % gel: a randomized, double-blind, parallel-group, controlled study. Am J Clin Dermatol. 2016;17(3):293–303.
Thiboutot D, Zaenglein A, Weiss J, et al. An aqueous gel fixed combination of clindamycin phosphate 1.2% and benzoyl peroxide 2.5% for the once-daily treatment of moderate to severe acne vulgaris: assessment of efficacy and safety in 2813 patients. J Am Acad Dermatol. 2008;59(5):792–800.
Tschen EH, Katz HI, Jones TM, et al. A combination benzoyl peroxide and clindamycin topical gel compared with benzoyl peroxide, clindamycin phosphate, and vehicle in the treatment of acne vulgaris. Cutis. 2001;67(2):165–9.
Graber EM. Treating acne with the tetracycline class of antibiotics: a review. Dermatol Rev. 2021;2(6):321–30.
Feldman SR, Han G, Callender VD, et al. Benefit of topical combination therapy for acne: analyzing effect size using number needed to treat. J Drugs Dermatol. 2024;23(2):42–9.
Goldberg JL, Dabade TS, Davis SA, et al. Changing age of acne vulgaris visits: another sign of earlier puberty? Pediatr Dermatol. 2011;28(6):645–8.
Kumtornrut C, Yamauchi T, Koike S, et al. Androgens modulate keratinocyte differentiation indirectly through enhancing growth factor production from dermal fibroblasts. J Dermatol Sci. 2019;93(3):150–8.
Dréno B. Treatment of adult female acne: a new challenge. J Eur Acad Dermatol Venereol. 2015;29(Suppl 5):14–9.
Stein Gold L, Kircik L, Baldwin H, et al. Tazarotene 0.045% lotion for females with acne: analysis of two adult age groups. J Drugs Dermatol. 2022;21(6):587–95.
Anderson KL, Dothard EH, Huang KE, Feldman SR. Frequency of primary nonadherence to acne treatment. JAMA Dermatol. 2015;151(6):623–6.
Dréno B, Thiboutot D, Gollnick H, et al. Large-scale worldwide observational study of adherence with acne therapy. Int J Dermatol. 2010;49(4):448–56.
Kircik LH, Draelos ZD, Berson DS. Polymeric emulsion technology applied to tretinoin. J Drugs Dermatol. 2019;18(4):s148–54.
Tanghetti EA, Kawata AK, Daniels SR, et al. Understanding the burden of adult female acne. J Clin Aesthet Dermatol. 2014;7(2):22–30.
Eichenfield DZ, Sprague J, Eichenfield LF. Management of acne vulgaris: a review. JAMA. 2021;326(20):2055–67.
Harper JC, Kircik LH, Gold M, et al. Early and sustained acne lesion reductions with fixed-dose clindamycin phosphate 1.2%/adapalene 0.15%/benzoyl peroxide 3.1% gel. J Drugs Dermatol. 2024;23(3):125–31.
Acknowledgements
We thank the participants of the studies.
Medical Writing/Editorial Assistance.
Medical writing support was provided by Phillip Lewis, PhD and Kavitha Abiraman, PhD of Prescott Medical Communications Group, a Citrus Health Group, Inc. company (Chicago, IL), with financial support from Ortho Dermatologics. Ortho Dermatologics is a division of Bausch Health US, LLC.
Funding
The study and the Rapid Service Fee were funded by Ortho Dermatologics. Ortho Dermatologics is a division of Bausch Health US, LLC.
Author information
Authors and Affiliations
Contributions
Leon H. Kircik, Linda Stein Gold, Michael Gold, Jonathan S. Weiss, Julie C. Harper, James Q. Del Rosso, Christopher G. Bunick, Neal Bhatia, Emil A. Tanghetti, Lawrence F. Eichenfield, Hilary Baldwin, Zoe D. Draelos, Valerie D. Callender, George Han, Melinda J. Gooderham, Neil Sadick, Mary P. Lupo, Edward (Ted) Lain, and Wiliam Philip Werschler made substantial contributions to the conception or design of the work; drafted the work/revised it critically; approved the version to be published; and agree to be accountable for all aspects of this work.
Corresponding author
Ethics declarations
Conflict of Interest
Leon H. Kircik has served as either a consultant, speaker, advisor or an investigator for Allergan, Almirall, Epi Health, Galderma, Novartis, Ortho Dermatologics, and Sun Pharma. Linda Stein Gold has served as investigator/consultant or speaker for Ortho Dermatologics, LEO Pharma, Dermavant, Incyte, Novartis, AbbVie, Pfizer, Sun Pharma, UCB, Arcutis, and Lilly. Michael Gold has acted as an investigator, advisor, speaker, and consultant for Ortho Dermatologics. Jonathan S. Weiss is a consultant, speaker, advisor, and/or researcher for AbbVie, Ortho Dermatologics, Janssen Biotech, Dermira, Almirall, Brickell Biotech, DermTech, and Scynexis. Julie C. Harper has received honoraria from Aclaris, Almirall, BioPharmX, Cassiopea, Cutanea, Dermira, Foamix, Galderma, LaRoche-Posay, Ortho Dermatologics, and Sun Pharma. James Q. Del Rosso has served as a consultant, investigator, and/or speaker for Ortho Dermatologics, AbbVie, Amgen, Arcutis, Cutera, Dermavant, EPI Heath, Galderma, Incyte, JEM Health, La Roche-Posay, LEO Pharma, Lilly, L’Oreal, MC2 Therapeutics, Pfizer, Strata, Sun Pharma, and UCB. Christopher G. Bunick has served as an investigator for AbbVie, Almirall, Timber, and Palvella; a consultant for AbbVie, Almirall, Apogee, Arcutis, Eli Lilly, EPI Health/Novan, LEO Pharma, Novartis, Ortho Dermatologics, Pfizer, Sanofi-Regeneron, and UCB; and a speaker for and received honoraria from Allergan, Almirall, LEO Pharma, and UCB. Neal Bhatia has served as advisor, consultant, and investigator for AbbVie, Almirall, Biofrontera, BI, Brickell, BMS, EPI Health, Ferndale, Galderma, InCyte, ISDIN, J&J, LaRoche-Posay, LEO Pharma, Ortho Dermatologics, Regeneron, Sanofi, Sun Pharma, Verrica, and Vyne. Emil A. Tanghetti has served as speaker for Novartis, Ortho Dermatologics, Sun Pharma, Lilly, Galderma, AbbVie, and Dermira; served as a consultant/clinical studies for Hologic, Ortho Dermatologics, and Galderma; and is a stockholder for Accure. Lawrence F. Eichenfield has received honoraria for consulting services from AbbVie, BMS, Dermata, Dermira, Dermavant, Eli Lilly, Forte Pharma, Galderma, Incyte, J&J, Otsuka, Novartis, Pfizer, Regeneron Pharmaceuticals, Inc., Sanofi Genzyme, and Ortho Dermatologics; and study support (to institution) from AbbVie, Bausch Health, Dermira, Eli Lilly and Company, Galderma, Incyte, Pfizer, Regeneron Pharmaceuticals, Inc. and Sanofi Genzyme. Hilary Baldwin has served as advisor, investigator, and on speakers’ bureaus for Almirall, Cassiopea, Foamix, Galderma, Ortho Dermatologics, Sol Gel, and Sun Pharma. Zoe D. Draelos received funding from Ortho Dermatologics. Valerie D. Callender has served as an investigator, consultant, or speaker for Acne Store, Almirall, Aerolase, AbbVie, Allergan Aesthetics, Avava, Avita Medical, Beiersdorf, Cutera, Dermavant, Eli Lilly, Epi Health, Galderma, Janssen, Jeune Aesthetics, L’Oréal, Ortho Dermatologics, Pfizer, Prollineum, Regeneron, Scientis, Sente, SkinBetter science, SkinCeuticals, Symatese, UCB, and UpToDate. George Han is or has been an investigator, consultant/advisor, or speaker for AbbVie, Athenex, Boehringer Ingelheim, Bond Avillion, Bristol-Myers Squibb, Celgene, Eli Lilly, Novartis, Janssen, LEO Pharma, MC2, Ortho Dermatologics, PellePharm, Pfizer, Regeneron, Sanofi Genzyme, Sun Pharma, and UCB. Melinda J. Gooderham has served as investigator/consultant or speaker for AbbVie, Akros Pharma, Amgen, Arcutis Biotherapeutics, Aslan, Aristea, AnaptysBio, Bausch, BMS, Boehringer Ingelheim, Coherus BioSciences, Dermira, Dermavant, Eli Lilly, Galderma, GSK, Incyte, Inmagene, Janssen, Kyowa Kirin, LEO Pharma, MedImmune, Meiji, Merck, Moonlake, Nimbus, Novartis, Pfizer, Regeneron Pharmaceuticals, Inc., Reistone, Roche, Sanofi, Sun Pharma, Takeda, Tarsus, Union, UCB and Ventyx. Neil Sadick has served on advisory boards, as a consultant, investigator, speaker, and/or other and has received honoraria and/or grants/research funding from Almirall, Actavis, Allergan, Anacor Pharmaceuticals, Auxilium Pharmaceuticals, Bausch Health, Bayer, Biorasi, BTG, Carma Laboratories, Cassiopea, Celgene Corporation, Cutera, Cynosure, DUSA Pharmaceuticals, Eclipse Medical, Eli Lilly and Company, Endo International, EndyMed Medical, Ferndale Laboratories, Galderma, Gerson Lehrman Group, Hydropeptide, Merz Aesthetics, Neostrata, Novartis, Nutraceutical Wellness, Palomar Medical Technologies, Prescriber's Choice, Regeneron, Roche Laboratories, Samumed, Solta Medical, Storz Medical AG, Suneva Medical, Vanda Pharmaceuticals, and Venus Concept. Mary P. Lupo has served as a speaker, investigator, and/or on advisory boards for Ortho Dermatologics, Bausch Health US, Evolus, Revance, Eirion, Prolennium, Chemocentryx, Therapeutics Incorporated, Brickell, Endo, Cutera, In Mode, Revision, and Merz. Edward (Ted) Lain has served as investigator, consultant and/or speaker for Ortho Dermatologics, AbbVie, Almirall, Amgen, Arcutis, Dermavant, EPI Health, Galderma, Incyte, LEO Pharma, Novartis, Eli Lilly, Pfizer, Sun Pharma, UCB, Endo International, ChemoCentryx, Biorasi, Sirnaomics, Evelo Biosciences, Concert Pharmaceuticals, Cara Therapeutics, Castle Biosciences, Mindera, Biofrontera, Alfasigma, AiViva Biopharma, Anaptys Bio, Bausch Health, Dr Reddy’s, Trevi Therapeutics. William Philip Werschler has served as an investigator for Ortho Dermatologics.
Ethical Approval
These studies were carried out in accordance with principles of Good Clinical Practice (GCP) and the Declaration of Helsinki. Study protocols across all study centers were approved by the same central institutional review board (Advarra IRB, Columbus, MD, USA). All participants or their legal guardians provided written informed consent before entering the clinical trial from which the data were derived. Additional informed consent was obtained from all individual participants for whom identifying information/photographs are included in this article.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Kircik, L.H., Stein Gold, L., Gold, M. et al. Triple Combination Clindamycin Phosphate 1.2%/Adapalene 0.15%/Benzoyl Peroxide 3.1% for Acne: Efficacy and Safety from a Pooled Phase 3 Analysis. Dermatol Ther (Heidelb) 14, 1211–1227 (2024). https://doi.org/10.1007/s13555-024-01155-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13555-024-01155-7