Abstract
Introduction
Patients with moderate-to-severe atopic dermatitis (AD) who are most likely to respond to the Janus kinase (JAK) 1/2 inhibitor baricitinib (BARI) are known to have an impacted body surface area (BSA) ≤ 40% and severe itch (numerical rating scale [NRS] ≥ 7], collectively termed ‘BARI itch-dominant’ patients. Our objective is to build on our previous work by providing a body region-specific, clinical characterization of the BARI itch-dominant patient at baseline and their response to BARI 4 mg.
Methods
BREEZE-AD7 was a phase 3 trial in adults with moderate-to-severe AD receiving placebo or 2 mg or 4 mg BARI in combination with topical corticosteroids. Assessing only data from BARI itch-dominant patients, we summarized the baseline characteristics and conducted body region-specific analyses on Eczema Area and Severity Index (EASI) data in order to report the response to placebo versus BARI 4 mg within this patient subtype.
Results
BARI 4 mg was highly effective across all body regions; at week 16, 75% improvement was seen in EASI scores (EASI75), and response rates with BARI 4 mg (head/neck, 58.3%; trunk, 69.2%; upper extremities, 61.5%; lower extremities, 87.5%) all exceeded those with placebo (head/neck: 37.5%; trunk, 40.6%; upper extremities, 18.8%; lower extremities, 40.6%) as well as the overall EASI75 rates of the intent-to-treat (ITT) population (BARI, 48.0%; placebo, 23.0%). At baseline, most BARI itch-dominant patients presented with involvement of all regions (mean regional BSA 22.7%–40.3%), highest in the head and neck, mean EASI region scores of 15.7–24.0, and considerably severe sign ratings (mean EASI sub-scores: 1.4–2.3, out of 3), especially for erythema.
Conclusion
BARI itch-dominant patients exhibit AD involvement across all body regions and considerable sign severity, especially erythema. In response to BARI 4 mg, EASI quickly improved across regions, substantially more so in this subtype than in the ITT population.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Despite the known heterogeneity in atopic dermatitis (AD) disease course and clinical presentation of signs and symptom, AD is still considered a singular disease and typically treated with a one-size-fits-all approach. A first step away from a one-size-fits-all approach to AD—and toward a more individualized understanding of treatment responses—was recently investigated for baricitinib (BARI) using machine learning; this previous work identified the patients most likely to benefit from BARI 4 mg, which are those with an affected body surface area (BSA) of up to 40% along with severe itch [Itch Numerical Rating Scale (NRS) scores ≥ 7], termed the ‘BARI itch-dominant’ subtype. |
The current study aimed to expand on the previous work by providing a detailed, Eczema Area and Severity Index (EASI)-based, body region-specific characterization of the BARI itch-dominant patient at baseline as well as in response to BARI 4 mg. |
What was learned from the study? |
The ‘BARI itch-dominant’ patient subtype presents at baseline with AD lesions spread across all body regions, although regional involvement (mean regional BSA) is highest in the head/neck; the associated mean EASI sign severity ratings are all consistently in the upper range, especially erythema. |
In response to BARI 4 mg, EASI severity ratings quickly improved across all body regions, substantially more than what was observed in the intent-to-treat (ITT) population. |
These findings highlight the need for clinicians to assess AD involvement across all body regions when making treatment decisions, and to be aware that a moderate BSA does not necessarily correspond to lower severity of skin lesions, particularly in patients with severe itch. |
Introduction
Atopic dermatitis (AD) is a chronic inflammatory skin disorder characterized by itch, eczema, excoriations, and lichenification of the skin [1, 2]. Standard practice has largely approached AD as a monolithic disease [3]; however, the clinical phenotypes of AD are known to be heterogeneous in both clinical signs and symptoms and response to treatment [4,5,6].
Recent work has contributed to the field’s understanding of AD clinical phenotypes using combined itch and lesion severity. Chovatiya and colleagues (2021) used these parameters (itch and lesion severity) to describe four AD phenotypes in a large sample of private practice patients (N = 592); the phenotypes were mild–moderate itch and lesions (MI–ML), mild–moderate itch and severe lesions (MI–SL), severe itch and mild–moderate lesions (SI–ML), and finally severe itch and lesions (SI–SL). While they found that the majority were ‘mild-to-moderate’ in both domains, the second-largest group—nearly a quarter of the sample—presented with severe itch accompanied by mild-to-moderate lesions (SI–ML). The authors labelled this subgroup ‘itch-dominant’ and noted a unique discrepancy between these patients and their healthcare providers (HCPs) in the recognition of their high disease burden [7]. Yet, optimal treatment for patients with varying levels of itch intensity and skin involvement remained to be determined. A first step away from a one-size-fits-all approach to a more individualized understanding of treatment response—one that accounts for different AD phenotypes—was recently investigated for the oral Janus kinase (JAK) inhibitor (JAKi) baricitinib (BARI) by utilizing a CART (classification and regression tree analysis)-based machine learning approach on data from the phase 3 topical corticosteroids (TCS) combination therapy trial BREEZE-AD7 [8].
BARI is an oral selective and reversible inhibitor of JAK 1 and 2 for the treatment of moderate-to-severe AD in adults as well as children and adolescents (2 years and older). While the 2-mg dose is reserved for special patient populations, the recommended starting dose is 4 mg [8, 9]. Among other systemic treatments, BARI is recommended as a first-line systemic therapy for patients not sufficiently controlled on topical therapies [10].
Using the above-mentioned machine learning approach, the patients most likely to benefit from BARI 4 mg were characterized as having an affected body surface area (BSA) of up to 40% along with severe itch (Itch Numerical Rating Scale [NRS] scores ≥ 7) [8], a subgroup of patients that is similar to the previously described itch-dominant phenotype [7]. The results of the machine learning-based analysis showed that moving from the intent-to-treat (ITT) population to this individualized approach led to increased rates of treatment response across signs and symptoms; for example, compared to 48% of the ITT population, approximately 70% of the BARI itch-dominant patients reached a 75% improvement in the Eczema Area and Severity Index score (EASI75), and likewise, compared to 44%, approximately 58% achieved a clinically relevant itch response (defined as a 4-point itch NRS improvement) [8].
Although the BARI itch-dominant subgroup of patients with AD is characterized by skin involvement of 40% or less affected BSA, which was identified as the main predictor for response [8], other skin measures might be preferred for clinical assessment. EASI is often the preferred instrument [11], as it along with the SCORing Atopic Dermatitis (SCORAD) index is recommended by the Harmonizing Outcome Measures for Eczema (HOME) initiative as one of the best-validated outcome measures for AD [12]. Furthermore, EASI can be used to assess four separate body regions, which may yield important information on lesion location, a factor known to moderate the impact on quality of life [13, 14]; plus, different body regions can exhibit different responses to the same treatment, e.g. [15, 16]. Therefore, in this new report, our aim is to provide a detailed, EASI-based, body region-specific characterization of the BARI itch-dominant patient at baseline and in response to BARI 4 mg.
Methods
BREEZE-AD7 was a TCS combination therapy trial (NCT03733301; N = 329) for which we have previously reported a detailed study design [17]. Protocols were approved by the institutional review board or ethics committee at each participating site. In brief, this was a 16-week, randomized, double-blind, placebo-controlled phase 3 trial. All studies included in this analysis were conducted in accordance with the ethical principles of the Declaration of Helsinki. Written informed consent was obtained from all eligible participants before undergoing study-related procedures. Stratified by baseline disease severity and geography, adults with moderate-to-severe AD and a history of inadequate response to topical treatments were randomized 1:1:1 to receive either once-daily placebo, BARI 2 mg, or BARI 4 mg while also being allowed the use of moderate- and/or low-potency TCS on active lesions. For trial eligibility, moderate-to-severe AD was defined by an EASI total score ≥ 16, a Validated Investigator’s Global Assessment of AD (vIGA-AD) score ≥ 3, and ≥ 10% BSA at baseline [17].
The current post hoc analysis evaluated data from BREEZE-AD7 participants who were receiving placebo (PBO) or BARI 2 mg or 4 mg and whose baseline profile included BSA up to 40% and an Itch NRS ≥ 7, i.e., the BARI itch-dominant subgroup [8]. We only report data from the PBO and BARI 4 mg groups. Baseline characteristics described in this analysis include the affected BSA (mean, median, and weighted), EASI signs sub-scores (excoriation, erythema, edema/papulation, and lichenification, each ranging from 0 to 3), and EASI region scores (head/neck, trunk, and upper and lower extremities, each ranging from 0 to 72), which are summarized as mean (± standard deviation [SD]). Weighted BSA was derived by multiplying the regional BSA (region-specific, i.e., %BSA affected by AD within the bounds of that body region) by the corresponding EASI multiplier to account for the region’s size relative to the whole body—0.4 for lower extremities, 0.3 for trunk, 0.2 for upper extremities, and 0.1 for the head/neck [18]. All data are presented separately by body region. A visualization of the baseline characteristics of the individuals randomized to BARI 4 mg is provided in Fig. 1.
Visualization of mean baseline characteristics of the baricitinib (BARI) itch-dominant patient subtype (body surface area [BSA] ≤ 40% and Itch numerical rating scale [NRS] score ≥ 7). The size of the coloured areas within each region represents the mean BSA, while the colour corresponds to the severity of EASI sign sub-scores in that region. Each homunculus represents one of the four EASI signs, with a cumulative representation on the right-hand side. BARI baricitinib, BSA body surface area, EASI Eczema Area and Severity Index, NRS numerical rating scale
The proportion of patients achieving EASI75 in each body region was evaluated over 16 weeks between treatment groups using logistic regression. Non-responder imputation was used for data missing due to discontinuation or rescue. The percent change from baseline (%CFB) in EASI scores for each body region (EASI region scores) was analysed using mixed-model repeated measures (MMRM). MMRM included treatment, geographic region, baseline disease severity (vIGA-AD), visit, and treatment-by-visit interaction as fixed categorical effects, and the baseline score and baseline score-by-visit interaction as fixed continuous effects. Data collected after the first rescue therapy date or permanent study drug discontinuation were excluded from the analysis, and no multiplicity was adjusted for the comparisons. Patients with a baseline score of 0 were not included in either %CFB or EASI75 analysis.
Results
Baseline Demographics and Clinical Characteristics of Patients with BSA ≤ 40%/Itch NRS ≥ 7
Detailed baseline demographics and characteristics have been published previously [8]. In brief, the patients with BSA ≤ 40%/Itch NRS ≥ 7 who were randomized to BARI 4 mg were of a mean (± SD) age of 34.1 (± 12.0) years, had mean disease duration of 26.6 years (± 13.0), and 64% had previously tried at least one other systemic therapy. The mean overall BSA affected by AD was 30.4% (± 6.9%), mean EASI total scores were 20.5 (± 3.3), and mean Itch NRS was 8.1 (± 0.8). A total of 35% had an EASI total score > 21, and the mean SCORAD value was 65.3 (± 9.6), both indicating a severe form of AD.
Baseline Disease Characteristics per Body Region in Patients with BSA ≤ 40%/Itch NRS ≥ 7
Body region-specific baseline characteristics of those randomized to either PBO (n = 32) or BARI 4 mg (n = 26) are reported in Table 1, with the latter group visualized in Fig. 1. Nearly all patients (92.3–100% BARI 4 mg and 100% PBO) exhibited involvement across all four body regions. Across both treatment groups, the mean regional BSA was highest in the head/neck region, reaching as high as 80% (PBO)/100% (BARI 4 mg) in some patients. This was followed by the upper extremities and the trunk, and then markedly lower in the lower extremities. When weighted using the region-specific EASI multiplier to adjust for the relative contribution to the total body [18], the mean weighted BSA was highest in the trunk and lower extremities. In other words, the trunk and lower extremities contributed more to the total BSA of up to 40% due to representing a larger portion of the skin surface area, but regional involvement was greatest in the head/neck area.
In every body region, across both treatment groups, the most relevant EASI clinical sign was erythema, with a mean score ranging from 1.7 to 2.3. Excoriation, edema/papulation, and lichenification, but not erythema, sub-scores tended to be higher in the upper extremities compared to other body regions. The mean EASI region scores were similarly high across the trunk (24.0 BARI 4 mg, 21.6 PBO), head/neck (23.3 BARI 4 mg, 21.6 PBO), and upper extremities (23.5 BARI, 23.4 PBO), and notably lower in the lower extremities (15.7 BARI 4 mg; 17.3 PBO). Maximal region scores ranged as high as 60 (out of 72) in the head/neck and upper extremities, as high as 44 in the trunk, and as high as 35 in the lower extremities.
Percent Change from Baseline (%CFB) in EASI Region Scores Over 16 Weeks for Patients with BSA ≤ 40%/Itch NRS ≥ 7 Receiving Either BARI 4 mg or PBO
The difference between BARI 4 mg and PBO in %CFB of EASI region scores rapidly became significant by week 1 in the upper extremities and by week 2 in all other body regions (Fig. 2). At the end of the study period at week 16, the greatest least square mean %CFB in response to BARI 4 mg was seen in the lower extremities at −81.9% (vs −41.9% PBO, P < 0.001), followed by the trunk at −69.1% (vs −43.4% PBO, P = 0.07), the head/neck at −65.0% (vs −39.2% PBO, P = 0.08), and the upper extremities at −59.9% (vs −31.7% PBO, P = 0.06). The response to BARI 4 mg appeared to plateau after week 4 in most body regions, except in the lower extremities, which continued to exhibit improvement through study end.
Percent change from baseline in Eczema Area and Severity Index (EASI) region scores, between baricitinib (BARI) 4 mg and placebo (PBO), by body region. Baseline EASI region scores are listed in the upper left-hand corner of each region-specific graph. Significant differences between BARI and PBO are marked on the graph (*P < 0.05; **P < 0.01; ***P < 0.001). BARI baricitinib, CFB change from baseline, EASI Eczema Area and Severity Index, N number, PBO placebo, TCS topical corticosteroids
EASI75 Response Rates Over 16 Weeks in Patients with BSA ≤ 40%/Itch NRS ≥ 7 Receiving Either BARI 4 mg or PBO
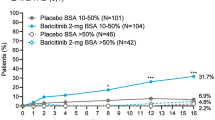
The pattern of EASI75 response rates was similar to that observed in %CFB of EASI region scores (Fig. 3). At the end of the study period, response rates to BARI 4 mg were highest in the lower extremities at 87.5% (vs 40.6% PBO, P < 0.001), followed by the trunk at 69.2% (vs 40.6% PBO, P < 0.05), the upper extremities at 61.5% (vs 18.8% PBO, P < 0.01), and the head/neck at 58.3% (vs 37.5% PBO, P = 0.18).
75% improvement in Eczema Area and Severity Index (EASI75) response rates, between baricitinib (BARI) 4 mg and placebo (PBO), by body region. Baseline EASI region scores are listed in the upper left-hand corner of each region-specific graph. Significant differences between BARI and PBO are marked on the graph (*P < 0.05; **P < 0.01; ***P < 0.001). BARI baricitinib, EASI Eczema Area and Severity Index, N number, PBO placebo, TCS topical corticosteroids
Discussion
BARI itch-dominant patients (BSA ≤ 40% and Itch NRS ≥ 7) who participated in BREEZE-AD7 presented with AD lesions across all body regions. Rather than predominant involvement of any specific region, approximately one fourth to one third of each body region was impacted in this patient type. All clinical signs of AD were seen across all body regions in most patients, with mean EASI sign severity ratings consistently in the upper range, particularly for erythema. The efficacy of BARI 4 mg + TCS (‘BARI 4 mg’) was also evident across all body regions, with significantly greater %CFB in EASI region scores in response to BARI 4 mg + TCS than to PBO + TCS (‘PBO’) seen early in every body region during the first 4 weeks of treatment. Together, these findings indicate that BARI itch-dominant patients present with BSA involvement across all four body regions and often severely impacted by AD clinical signs as assessed by EASI. Furthermore, our data demonstrate a robust response to BARI 4 mg across all body regions. All EASI75 region response rates in our analysis of this patient subtype (58.3–87.5%) were greater than the EASI75 overall response rate seen in the ITT population (48%) [17], and we observed a corresponding notable increase in the treatment response of the head/neck region (58.3%) relative to that of the ITT population (40.0%) [14]. These detailed insights further support individualized patient management and may ultimately help patients access therapies to which they are most likely to respond.
As previously reported [8], the BARI itch-dominant patient presents with a profound burden at baseline; the mean EASI total score is 20.5, with over one third of the sample exhibiting severe AD (score > 21) and some reaching as high as 30.5. Additionally, this patient subtype presents with severe itch (mean Itch NRS of 8.1) and severe SCORAD (mean rating of 65.3), despite having BSA limited up to 40%. This new analysis adds further granularity to our previous report by analysing body regions separately (see comprehensive visualization in Fig. 1). All body regions displayed involvement, particularly the trunk and upper extremities and notably less so the lower extremities. This presentation is unique from observations made at baseline with other available AD treatments; for example, phase III testing of the interleukin (IL)-4/IL-13 inhibitor dupilumab reported baseline regional EASI scores that were quite similar across all body regions (though still with some variation) [19]. This would suggest that the BARI itch-dominant patient can be distinguished by lesser involvement of the lower extremities, as the difference in regional EASI scores stemmed from the BSA rather than the sign severity; however, a recent real-world evidence (RWE) study in Japan proposed that high EASI region scores in the lower extremities at baseline are predictive of responding to BARI 4 mg [20], whereas our data indicate the opposite, a difference that may be due to the country from which patients originate. Across body regions, all clinical signs were rated severe, with erythema modestly prevailing. The baseline EASI sub-scores of this patient subtype are strikingly similar to those of the ITT population, even though the full population also includes patients with BSA above 40% and mean EASI scores of 30, some reaching as high as 40 [21]. These results therefore suggest that patients with AD with BSA limited up to 40% exhibit similar intensity of clinical signs as patients with higher BSA involvement, underlining the findings from machine learning analyses that did not identify baseline EASI but rather baseline BSA as the predicting factor for response to BARI 4 mg. This highlights the critical importance of HCPs evaluating the AD extent (i.e., BSA) across all body regions, alongside the patient’s itch-related burden, to determine whether the patient fits the BARI itch-dominant clinical description, which might aid patient selection and increase the likelihood that patients benefit from treatment.
In our analysis of data from the head/neck region, the most relevant clinical sign was erythema, followed by lichenification and edema/papulation. This region was unique for showing variation in the severity of AD signs and, when compared to the other regions, the head/neck had the highest region-specific skin involvement (i.e., regional BSA, unweighted). The mean EASI region score (BARI 4 mg: 23.3; PBO: 21.6) was in the severe range (> 21). In what is known from RWE to be a difficult-to-treat region, the EASI75 response rate following BARI 4 mg (58.3%), albeit lower than all other body regions examined in the BARI itch-dominant patients, profoundly exceeded the response observed in the head/neck of the ITT population (40.0%) [14]. Furthermore, the difference in head/neck response rates between the BARI itch-dominant patients who received BARI 4 mg vs PBO (20.8%) was greater than the corresponding difference seen in the ITT population (15.0%) [14]. However, while the previous analyses found the response rate of the ITT population to be significantly different from PBO, the heightened response rate and greater differentiation from PBO observed in the BARI itch-dominant subgroup only reached statistical significance at early time points, drawing attention to the limitations of our small sample size. Overall, this finding of an enhanced response in the head/neck region of BARI itch-dominant patients is promising, especially as this area is associated with a significant impact on quality of life as well as low responsivity to IL-4/IL-13 inhibitors [22, 24].
When affected BSA within each body region was considered in the context of the whole body via the application of region-specific EASI weights (i.e., weighted BSA), the trunk exhibited the highest relative level of skin area involvement. The trunk also had the highest overall EASI region scores, though all body regions presented with severe (> 21) mean EASI regional scores, except the lower extremities (BARI 4 mg: 15.7; PBO: 17.3), which were in the moderate range. However, the lower extremities, due to their 40% contribution to the body, accounted for one third of the total BSA. Across the trunk, upper extremities, and lower extremities, we observed a consistent pattern wherein the prevailing clinical sign was erythema, followed closely by edema/papulation, excoriation, and lichenification. All body regions exhibited rapid EASI improvement in response to BARI 4 mg, with %CFB in EASI region scores first differentiating significantly from PBO in the upper extremities at week 1, followed by all other regions at week 2. After 16 weeks, both the %CFB in EASI region scores and EASI75 regional response rates were greatest overall in the lower extremities. Similar to the head/neck region (58.3%), the range of EASI75 responses across the other body regions of the BARI itch-dominant patients (61.5% [upper extremities] to 87.5% [lower extremities]) all exceeded the overall EASI75 response rate in the ITT population (48%) [17]. In comparison to the ITT population, the BARI itch-dominant patients exhibited greater differentiation from PBO in EASI75 rates in every region. These findings indicate that by applying an individualized treatment approach, patients have a higher likelihood of achieving treatment response with BARI, across all body regions. This complements our previous work [8] by adding region-level detail and showing that patients who meet our selection criteria are most likely to benefit across all body regions.
Together with our previous work [8], this study provides a step toward individualized medicine for patients with AD. RWE is critical for confirming these findings, but there is a limited amount of RWE currently available regarding the effectiveness of BARI across specific body regions. A recent RWE study of AD patients in Germany receiving BARI 4 mg (N = 47) reported baseline EASI, BSA, SCORAD, and Itch NRS scores that were similar to those of the BARI itch-dominant patients [24]. This study aimed at examining clearance in sensitive body areas (head/neck, face, hands, plantar, and genitals), and the authors found significant decreases in involvement of all regions following BARI 4 mg [24]. For example, the head/neck region had been affected in 84% of patients at baseline, which decreased to 55.8% by study end at week 12; significant decreases were also seen in the face, and hand involvement went from 61 to 35% [24]. Similarly, in a recent multi-country survey of patients with AD initiating BARI in routine clinical practice (N = 170), patients reported baseline involvement, and subsequently clearance, across all body regions (arms, legs, trunk, hands, and head/neck) [25]. Importantly, the reported patient characteristics of this sample at the time of BARI initiation are in line with the BARI itch-dominant profile; the majority presented with a BSA below 10% and a high itch burden [26]. These participants indicated that the head/neck region was the most bothersome (e.g., had the greatest impact on quality of life), followed by hands and then arms and legs; of those who reported head/neck involvement at the start of treatment with BARI 4 mg, > 50% experienced a clearance of symptoms in this region and reported high treatment satisfaction by study end [25]. Currently ongoing observational studies such as AD-REAL (EUPAS37841), as well as AD registries, will be invaluable in providing further information on patients with a BSA up to 40% and severe itch (BARI itch-dominant).
This study has several limitations. First, it is a post hoc analysis limited to a 16-week study period, with a further-limited number of participants classified into the BARI itch-dominant subgroup. This may have reduced our ability to detect statistical differences and may have also resulted in the identification of statistical differences that are less clinically meaningful. Second, despite being a key measure for both research and routine clinical practice, EASI does not allow for further breakdown by anatomically distinct and clinically relevant subregions. For instance, the upper and lower extremities cannot be subdivided to assess hand or foot involvement, and the head/neck region cannot be subdivided to assess facial involvement. This rigidity is unfortunate given the evidence that specific subdivisions, such as the hands and feet, can exhibit responses that vary meaningfully from the overall EASI-defined region [24]. Specific studies to assess patients with this level of granularity may be necessary in the future.
Conclusion
In conclusion, the ‘BARI itch-dominant’ patients that have been identified as likely to benefit most from treatment with BARI 4 mg, i.e., those with severe itch and lesion extent of up to 40% of their body, present with AD involvement across all body regions and severe levels of AD signs, strikingly similar to those seen across the ITT population and particularly high for erythema. In response to BARI 4 mg, these patients exhibit enhanced efficacy relative to the ITT population, across all body regions. While our analyses did not find a predominantly impacted body region at baseline, this is likely related to the identification of BSA rather than EASI as a predictive factor for treatment response; additionally, our data clearly indicate that a moderate BSA does not necessarily correspond to reduced severity of the skin lesions, particularly in these patients with severe itch. This finding also emphasizes the need for clinicians to assess AD involvement across all body regions when making treatment decisions with patients. Our analysis provides important insights for dermatologists to help identify those most likely to benefit from BARI, and thus constitutes an important step toward an individualized approach to treatment of AD.
Data Availability
Lilly provides access to all individual participant data collected during the trial, after anonymization, with the exception of pharmacokinetic or genetic data. Data are available to request six months after the indication studied has been approved in the US and EU and after primary publication acceptance, whichever is later. No expiration date of data requests is currently set once data are made available. Access is provided after a proposal has been approved by an independent review committee identified for this purpose and after receipt of a signed data sharing agreement. Data and documents, including the study protocol, statistical analysis plan, clinical study report, blank or annotated case report forms, will be provided in a secure data sharing environment. For details on submitting a request, see the instructions provided at www.vivli.org.
References
Eichenfield LF, Tom WL, Chamlin SL, et al. Guidelines of care for the management of atopic dermatitis: 1. Diagnosis and assessment of atopic dermatitis. J Am Acad Dermatol. 2014;70:338–51.
Silverberg JI, Gelfand JM, Margolis DJ, et al. Patient burden and quality of life in atopic dermatitis in US adults: a population-based cross-sectional study. Ann Allergy Asthma Immunol. 2018;121:340–7.
Bieber T, D’Erme AM, Akdis CA, et al. Clinical phenotypes and endophenotypes of atopic dermatitis: where are we, and where should we go? J Allergy Clin Immunol. 2017;139:S58-64.
Bieber T. Atopic dermatitis: an expanding therapeutic pipeline for a complex disease. Nat Rev Drug Discov. 2022;21:21–40.
Chovatiya R, Silverberg JI. Evaluating the longitudinal course of atopic dermatitis: implications for clinical practice. Am J Clin Dermatol. 2022;23(4):459–68.
Ständer S. Atopic dermatitis. N Engl J Med. 2021;384(12):1136–43.
Chovatiya R, Lei D, Ahmed A, et al. Clinical phenotyping of atopic dermatitis using combined itch and lesional severity: a prospective observational study. Ann Allergy Asthma Immunol. 2021;127(1):83–90.
Thyssen JP, de Bruin-Weller M, Costanzo A, et al. Baseline body surface area and itch severity define response to baricitinib in patients with moderate-to-severe atopic dermatitis at week 16. Adv Ther. 2023;40:3574–87.
Olumiant. European Union Summary of Product Characteristics. Eli Lilly and Company. 2023. https://www.ema.europa.eu/en/documents/product-information/olumiant-eparproductinformation_en.pdf.
Wollenberg A, Kinberger M, Arents B, et al. European guideline (EuroGuiDerm) on atopic eczema: part I—systemic therapy. J Eur Acad Dermatol Venereol. 2022;36(9):1409–31.
Iannone M, Tonini G, Janowska A, et al. Definition of treatment goals in terms of clinician-reported disease severity and patient-reported outcomes in moderate-to-severe adult atopic dermatitis: a systematic review. Curr Med Res Opin. 2021;37(8):1295–301.
Schmitt J, Spuls PI, Thomas KS, et al. The Harmonising Outcome Measures for Eczema (HOME) statement to assess clinical signs of atopic eczema in trials. J Allergy Clin Immunol. 2014;134(4):800–7.
Lio PA, Wollenberg A, Thyssen JP, et al. Impact of atopic dermatitis lesion location on quality of life in adult patients in a real-world study. JDD. 2020;19(10):943–8.
Wollenberg A, Lio P, Kleyn E, et al. Improvement of head and neck symptoms in patients with atopic dermatitis treated with baricitinib based on five Phase III trials. Eur J Dermatol. 2022;32(4):522–9.
Gori N, Ippoliti E, Peris K, Chiricozzi A. Head and neck atopic dermatitis: still a challenging manifestation in the biologic era. Expert Opin Biol Ther. 2023;23(7):575–7.
Jo CE, Finstad A, Georgakopoulos JR, et al. Facial and neck erythema associated with dupilumab treatment: a systematic review. J Am Acad Dermatol. 2021;84(5):1339–47.
Reich K, Kabashima K, Peris K, et al. Efficacy and safety of baricitinib combined with topical corticosteroids for treatment of moderate to severe atopic dermatitis. JAMA Dermatol. 2020;156:1333–43.
Hanifin JM, Baghoomian W, Grinich E, et al. The eczema area and severity index—a practical guide. Dermatitis. 2022;33:187–92.
Blauvelt A, Rosmarin D, Bieber T, et al. Improvement of atopic dermatitis with dupilumab occurs equally well across different anatomical regions: data from phase III clinical trials. BJD. 2019;181:196–7.
Hagino T, Saeki H, Fujimoto E, et al. Efficacy and safety of baricitinib treatment for moderate to severe atopic dermatitis in real-world practice in Japan. J Dermatol. 2023;50:869–79.
Wollenberg A, Simon D, Kulthanan K, et al. Baricitinib treatment rapidly improves the four signs of atopic dermatitis assessed by Eczema Area and Severity Index (EASI) clinical subscores. J Eur Acad Dermatol Venereol. 2023. https://doi.org/10.1111/jdv.19669. (Epub ahead of print).
Ribero S, Puglisi B, Giura MT, et al. Head and neck severity index is associated to a significant worsening of quality of life in atopic dermatitis patients. Exp Derm. 2021;30(11):1717–8.
Vittrup I, Krogh NS, Larsen HHP, et al. A nationwide 104 weeks real-world study of dupilumab in adults with atopic dermatitis: ineffectiveness in head-and-neck dermatitis. J Eur Acad Dermatol Venereol. 2023;37:1046–55.
Maintz L, Schmitz M-T, Herrmann N, et al. Real-world data on short-term effectiveness, clinician- & patient-reported outcomes in patients with atopic dermatitis treated with baricitinib. Poster presented at EADV-Spring symposium, 18–20 May 2023, Seville, Spain.
Pinter A, Reguiai Z, Werfel T, et al. Baricitinib outcomes on atopic dermatitis lesion locations: results from a cross-sectional patient survey in France, Germany, and the United Kingdom. Poster presented at EADV symposium, 11–14 October 2023, Berlin, Germany.
Augustin M, Nosbaum A, Werfel T, et al. Treatment satisfaction of adult patients with moderate-to-severe atopic dermatitis treated with baricitinib in France, Germany, and the United Kingdom: Results from a cross-sectional patient survey. J Dermatol Treat. 2023;34(1):2276047.
Acknowledgements
Eli Lilly and Company would like to thank the clinical trial participants and their caregivers, without whom this work would not be possible. The authors would also like to thank Dr Inmaculada de la Torre MD, PhD, of Eli Lilly and Company, for her scientific input, contribution to the study design, and analysis interpretation.
Medical Writing and Editorial Support
Medical writing assistance in the preparation of this article was provided by Dominika Kennedy, PhD, of Eli Lilly and Company. Statistical analysis was performed by author Chunyuan Liu of Eli Lilly and Company. Quality review and support was provided by Certara Synchrogenix. The support for this assistance was funded by Eli Lilly and Company.
Funding
This study and the Dermatology & Therapy Rapid Service Fee are sponsored by Eli Lilly and Company.
Author information
Authors and Affiliations
Contributions
Jose-Manuel Carrascosa, Alessandra Narcisi, Toshifumi Nomura, Sonja Ständer, Christian Vestergaard, and Andreas Wollenberg contributed to the drafting and finalization of the manuscript. Silvia Sabatino, Susanne Grond, Uffe Koppelhus, and Mohamed Elrayes contributed to the concept and design as well as drafting and finalization of the manuscript. Yun-Fei Chen and Chunyuan Liu contributed to the statistical analysis as well as drafting and finalization of the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
Jose Manuel Carrascosa has received grants, consulting fees, speaker honoraria, travel support and/or served on an advisory board for AbbVie, Almirall, Bristol-Myers Squibb, Eli Lilly and Company, Galderma, Janssen, IB Pharma, LEO Pharma, Novartis, Pfizer, Sandoz, Sanofi, and UCB. Alessandra Narcisi has received consulting fees, speaker honoraria, and/or travel support from AbbVie, Almirall, Amgen, Boehringer, Eli Lilly and Company, LEO Pharma, Pfizer, Sanofi, and UCB. Toshifumi Nomura has received speaker honoraria from Eli Lilly and Company. Sonja Ständer has received grants, consulting fees, speaker honoraria, travel support, and/or served on an advisory board for AbbVie, Almirall, Beiersdorf, Bristol-Myers Squibb, Clexio, Eli Lilly and Company, Escient, FomF, Galderma, Grünenthal, Incyte, IntegrityCE, Kiniksa, Klinge Pharma, LEO Pharma, L’Oreal, MEDahead, Moroscience, Novartis, Pfizer, P.G. Unna Academy, Sanofi, TouchIME, Vifor, and WebMD. Christian Vestergaard has received consulting fees, speaker honoraria, and/or research support from AbbVie, Eli Lilly and Company, LEO Pharma, Novartis, and Sanofi. Silvia Sabatino, Susanne Grond, Uffe Koppelhus, Mohamed Elrayes, and Yun-Fei Chen are employees and minor shareholders of Eli Lilly and Company. Chunyuan Liu is an employee of TigerMed-BDM Inc. and under long-term contract as a statistical expert for Eli Lilly and Company. Andrea Wollenberg has received grants, consulting fees, and/or study support from AbbVie, Aileens, Almirall, Beiersdorf, Eli Lilly and Company, Galapagos, Galderma, Glenmark, GSK, Janssen, LEO Pharma, L’Oreal, MedImmune, MSD, Novartis, Pfizer, Pierre Fabre, Regeneron, Sanofi, and UCB. Chunyuan Liu is an employee of TigerMed-BDM Inc and under contract with Eli Lilly and Company. He is not a Lilly shareholder.
Ethical Approval
Protocols for all studies included in this analysis were approved by the Institutional Review Board or Ethics Committee at each participating site. All studies included in this analysis were conducted in accordance with the ethical principles of the Declaration of Helsinki. Written informed consent was obtained from all eligible participants before undergoing study-related procedures.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Carrascosa, JM., Narcisi, A., Nomura, T. et al. Baricitinib Improvement Across Regions in Atopic Dermatitis Patients with Baseline Body Surface Area up to 40% and Severe Itch. Dermatol Ther (Heidelb) 14, 1561–1573 (2024). https://doi.org/10.1007/s13555-024-01171-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13555-024-01171-7