Abstract
Purpose of Review
Probiotics intake may be considered beneficial by prospective and pregnant mothers, but their effects on offspring development are incompletely understood. The purpose of this review was to examine recent pre-clinical and clinical studies to understand how maternal probiotics exposure affects offspring health outcomes.
Recent Findings
Effects were investigated in the context of supporting offspring growth, intestinal health, and gut microbiota, preventing allergic diseases, supporting neurodevelopment, and preventing metabolic disorders in pre-clinical and clinical studies. Most human studies focused on infancy outcomes, whereas pre-clinical studies also examined outcomes at adolescence and young adulthood. While still understudied, both pre-clinical and clinical studies propose epigenetic modifications as an underlying mechanism. Optimal timing of intervention remains unclear.
Summary
Administration of selected probiotics to mothers has programming potential for sustaining life-long health of offspring. Administration protocols, specific windows of susceptibility, and individual-specific responses need to be further studied.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Probiotics are defined as “live microorganisms that, when administered in adequate amounts, confer a health benefit on the host” [1]. Probiotics can be delivered in various modalities such as dietary supplements, food products, or drugs, targeting individuals across the entire life spectrum. Probiotics stem from various taxa including both prokaryotic and eukaryotic microorganisms and display common and/or strain-specific benefits such as regulation of intestinal transit, competitive exclusion of pathogens, vitamin and specific bioactive synthesis, intestinal barrier enhancement, or immunomodulation [1]. The gut ecosystem is a determinant of health and a main target of probiotics administration. Established in early life, altered dynamics in this process are associated with disease later in life, such as asthma [2], allergy [3], eczema [3], obesity [4, 5], and susceptibility to infection [6]. Several probiotics have been studied for their role in early stages of life and ability to sustain health in mothers and children. Substantiated benefits in the context of eczema for lactobacilli alone or in combination with Bifidobacterium species administered to mothers or infants [7] have been translated into guidelines by the World Allergy Organization in 2015 [8]. These guidelines indicate that probiotic consumption during pregnancy might be beneficial for pregnant or breastfeeding women at high risk for having a child who develops allergies. Additional benefits of maternally administered probiotics include improvement of metabolic parameters (insulin levels and insulin resistance, very low density lipoprotein and total cholesterol concentration) in gestational diabetes mellitus (GDM) [9], reduced rectal and vaginal Group B Streptococci colonization before parturition (important to prevent offspring mortality caused by Early Onset Group B Streptococcus disease) [10], and reduced incidence of mastitis [11]. These studies are sparse and heterogeneous in terms of probiotics usage, vehicle, and dosage. Because maternal exposures such as metabolic syndrome and infection are associated with offspring outcomes [12, 13], it is likely that offspring can also be affected by maternal use of probiotics. In fact, maternal probiotics administration was shown to result in temporary colonization of the offspring and/or modulation of the offspring microbiota [14, 15].
In a recent meta-analysis, probiotic consumption during pregnancy and/or lactation was shown to be generally safe for pregnant mothers in terms of gastrointestinal symptoms, tachycardia, vaginal discharge, eczema, and headache [16]. Only one probiotic mix containing Lacticaseibacillus rhamnosus GR-1 and Limosilactobacillus reuteri RC-14 was associated with an increased risk of vaginal discharge and changes in stool consistency after consumption during the first and second trimesters of pregnancy [17]. Direct probiotic administration to infants between birth and 2 years of age has also been reported as safe. This was shown through a systematic review that stratified the incidence of an adverse effect according to the infant’s health condition (healthy, low-birth weight, dermatitis, diarrhea, or formula-fed) [18]. The utilization of probiotics during these sensitive periods of life could therefore be considered to support infant health. A survey conducted in Canada suggests that the public already utilizes probiotics as 50.8% of women with a child aged 2 years or younger reported giving a probiotic product to their infant. Responses came from 413 mothers enrolled in the Alberta Pregnancy Outcomes and Nutrition (APrON) study in 2012 [19].
It is known that an altered maternal microbiota during pregnancy can affect offspring microbiota establishment, immune development, and metabolic health throughout life [20,21,22]. Thus, interventions targeting the maternal microbiota, such as probiotics, may have the potential to program offspring health. The purpose of this review was to examine recent literature from pre-clinical and clinical studies on the effects of probiotic administration started during the pre-conception period or pregnancy on offspring outcomes.
Search Strategy and Selection Criteria
PubMed, Embase, and Cochrane Library were searched for original research articles including pre-clinical (animal) and clinical studies and meta-analyses over the last 5 years (January 2017–March 2022). The search terms used were as follows: “probiotics”, “gestation”, “pregnancy”, “lactation”, “pre-conception” or “preconception”, “pre-mating” or “premating”, “pre-conceptional”, combined or not with “offspring”, “infant”, “baby” and complemented with manual inspection of reference lists of the selected articles. Articles were selected if they included a probiotic intervention starting at pre-conception and/or during pregnancy and assessed offspring outcomes. Studies of probiotics administered in combination with other ingredients were included. We included studies using substantiated probiotics strains, according to the Food and Agriculture Organization of the United Nations (FAO) guidelines [23], as well as strains for which research is ongoing, especially at the clinical stage.
Articles were screened to retrieve information about the probiotic strains, dose, vehicle, and supplementation period and this information is reported in tabulated form according to the Population, Interventions, Comparisons, Outcomes and Study designs (PICOS) elements [24] and grouped as pre-clinical (Table 1) or clinical (Table 2) studies. Findings are discussed according to 6 offspring outcomes identified across the studies: (1) growth and anthropometric indices at birth (19 articles); (2) intestinal barrier and gut health (14 articles); (3) neurodevelopment and anxiety-like behavior (8 articles); (4) allergic diseases (12 articles); (5) metabolic disorders (14 articles); and (6) intestinal microbiota (18 articles).
Probiotics in the Context of the Developmental Origins of Health and Disease
The Developmental Origins of Health and Disease (DOHaD) paradigm is based on the principle of developmental plasticity, which refers to the phenomenon by which “a given genotype can give rise to a range of different physiological or morphological states in response to various environmental exposures throughout development.” The term programming refers to a stimulus introduced at a “critical” or “sensitive” period, which causes long-term consequences for an organism [25]. A key principle of DOHaD is the existence of “windows of opportunity”, i.e., the prenatal stages of life (from pre-conception to embryonic and fetal stages and birth), infancy, and adolescence [25]. These time-sensitive life stages of exposure lead to tissue-specific effects thought to optimize or alter one’s biological potentials, and promote long-term health or a disease state [26, 27]. For example, undernutrition during pregnancy induces structure and functional remodeling in the fetus preserving brain development and prioritizing survival, negatively impacting development of other functions such as glucose metabolism and insulin sensitivity [26]. With time, as the evolutionary advantage of developmental plasticity begins to decrease, an individual’s ability to adapt to positive or negative environmental challenges becomes more limited [26]. Historically, the area of DOHaD research has primarily focused on overnutrition or undernutrition, linked with the manifestation of non-communicable diseases in later life, such as obesity or diabetes mellitus. Prenatal and early infancy exposures have been specifically studied at the macro- and micro-nutrient levels, with offspring outcomes ranging from glucose homeostasis to blood pressure [28]. In utero epigenetic modifications may be underlying mechanisms [29]. The gut microbiota has been proposed to regulate host gene expression epigenetically [30, 31], for example, via DNA methylation [32] with implications for offspring disease susceptibility [33]. The intestinal microbiota is a dynamic and interactive across-kingdom ecosystem composed of characteristic microbial communities co-evolving with their host [34]. While what constitutes a health-compatible microbiota remains elusive [35], many taxa have been identified whose altered representation is associated with diseases. Specifically, in infancy, microbiota variation is a predictor of overweight [5], asthma, and allergy [3]. The microbiota is seeded by the maternal microbiota [36] and continues to develop during infancy [37] thus going through sequential stages of plasticity that encompass pregnancy and are susceptible to programming, while being a determinant of life-long health [38]. In this context, maternally administered probiotics can be used as a dietary intervention that targets the offspring intestinal ecosystem. In utero and early life exposure to probiotics may affect growth and gut health [39,40,41], suggesting that specific windows of opportunities may exist for probiotic administration.
Growth and Anthropometric Indices at Birth
Probiotics have a long history of use to support growth of farm animals [42, 43]. Investigation into the underlying mechanisms suggests that they play a role in hormone metabolism. For example, certain Lactiplantibacillus plantarum strains support Drosophila melanogaster larval growth via target of rapamycin (TOR)-dependent mechanisms and hormonal growth signaling [44]. Lactiplantibacillus plantarum strains also support systemic growth in undernourished mice via growth hormone sensitivity enhancement and increased tissue insulin-like growth factor 1 activity [45]. Previous studies investigating the impact of maternal probiotics intake on offspring growth have largely focused on metabolic or allergic diseases as main outcomes [39, 46]. In recent studies in overweight women and women diagnosed with GDM, mid- or late-pregnancy supplementation with several mixtures of Lactobacillus, Bifidobacterium, and Streptococcus species had no effect on infant anthropometrics at birth [47,48,49,50,51]. Recently, several studies were also completed in healthy participants. As shown in Table 2, supplementation with various probiotic mixes starting at the beginning of pregnancy, at the second trimester, or at the end of pregnancy had no impact on neonatal birth weight, body mass index, birth length, femur length, or head circumference [52, 53, 54•, 55, 56•]. These findings align with a recent meta-analysis investigating infant birth weight following exposure to Lacticaseibacillus rhamnosus strains alone or in mixtures with Streptococcus or Bifidobacterium strains started during mid- or late-pregnancy [57]. Sex-specific effects have not been comprehensively studied.
Birth anthropometrics are important prognostic markers of healthy growth. Long-term effects of maternal probiotics on offspring growth during infancy and until adulthood have not been investigated in clinical studies; however, some studies have been conducted in animals. In healthy rodent models, the probiotic strains Limosilactobacillus fermentum CECT5716 and Lacticaseibacillus rhamnosus GG supplemented during pregnancy and lactation did not impact offspring body weight at weaning [58, 59]. Though supplementation with Bacillus subtilis PB6 or A and B from late pregnancy until weaning led to increased body weight in healthy piglet models (male and female combined) [60, 61]. Similarly, maternal intake of Lacticaseibacillus rhamnosus GG from late pregnancy until birth, combined with neonatal administration during the first five days of life, increased offspring weight during the four weeks after birth compared to the control mice that received the inactivated strain [62]. Findings were combined from male and female piglets. Interestingly, supplementation with Lactobacillus helveticus NS8 at the end of gestation in a healthy rat model led to a decreased body weight from late adolescence (51 days of life) until adulthood (76 and 86 days of life), without influencing the weight difference between males and females [63]. These findings suggest that the duration of the follow-up measurements is an important consideration to fully determine the effect of maternal probiotics exposure on offspring growth.
Intestinal Barrier and Gut Health
The intestinal barrier is critical to host health and a recognized target for preventative and therapeutic strategies [64]. The gut microbiota contributes to the protection of the epithelium from luminal pathogens and antigens, both physically and chemically [65], and supports the development of the infant immune system [3]. The intestinal barrier and the microbiota co-evolve in early life and reciprocally influence each other, resulting in the establishment of a mature intestinal ecosystem [66]. Probiotics have long been recognized to sustain the intestinal barrier, including in early life [40, 41]. Additionally, studies have proposed that the intestinal barrier, including the tight junction and the toll-like receptor associated pathways, is under epigenetic regulation [33]. Interestingly, in piglets, a recognized relevant model for pediatric nutritional studies assessing intestinal outcomes [67], maternal probiotics supplementation with Bacillus altitudinis WIT588 starting during late pregnancy increases intestinal crypt depth and villus length at postnatal day 34 [68]. In mice, supplementation with Lacticaseibacillus rhamnosus GG increases cell proliferation and differentiation and tight junction protein expression at postnatal day 21 [59, 62]. These findings suggest that maternal probiotic supplementation supports offspring intestinal digestive and absorptive functions in infancy [69]. Sex effects were not examined in these studies.
The long-term impact of this Lacticaseibacillus rhamnosus GG supplementation was investigated at 8 months of age when mice displayed increased goblet cell numbers and tight junction gene expression [59]. This was accompanied by increased antioxidant enzyme activities [59], and this increase was also demonstrated in the adult progeny of mini-sows that had received a Lactiplantibacillus plantarum B90 and Saccharomyces cerevisiae P11 mix, alone or with xylooligosaccharides, since the beginning of gestation [69]. This enhanced antioxidant capacity may help reduce ageing-associated oxidative stress and was associated with an increased jejunal villus height at 65 days [70]. Overall, findings from these pre-clinical studies suggest that maternal probiotic supplementation may help maintain gut barrier integrity in early life and adulthood and may improve antioxidant status. Indeed, recent studies support preventative effects of maternally administered probiotics in offspring with impaired intestinal barrier. In sows, maternal intake of Enterococcus faecium DSM7134 and Lactiplantibacillus plantarum CAM6 was shown to reduce diarrhea incidence in offspring [71, 72]. In a clinical trial, Limosilactobacillus reuteri LR92 supplementation to healthy mothers starting at late pregnancy and until birth led to a decreased colic frequency and severity in infants until 5 months of age, although the influence of sex on this effect was not reported [54•]. Interestingly, a recent study in mice showed that maternal administration of a mix of three Limosilactobacillus reuteri strains protected female offspring from dextran sodium sulfate-induced colitis [73••], although no protective effects were seen in male offspring. Taken together, these findings suggest that administration of selected probiotics during pregnancy may beneficially prevent intestinal inflammation in the offspring. More studies are required to understand sex-differences in responses and underlying mechanisms.
Neurodevelopment and Anxiety-Like Behavior
The gut ecosystem, including the microbiota, and the peripheral and central nervous system entertain a continuous bi-directional communication that is typically referred to as the gut-brain axis. Underlying mechanisms include endocrine pathways through cortisol and the hypothalamic–pituitary–adrenal axis, immunomodulation through cytokines, and interaction with the vagus nerve and the enteric nervous system [74]. The effects of probiotics on the gut-brain axis have been recently reviewed; Mörkl et al. demonstrated that probiotics may be therapeutically beneficial in the context of depression, but not schizophrenia, while data for anxiety are lacking [74]. Interestingly, these conditions may be rooted into neurodevelopment in early life and environmental exposures during stages of plasticity [75]. Moreover, the gut microbiota has been shown to foster fetal thalamocortical axonogenesis [76]. Thus, probiotic exposure during early stages of life, including in utero, may have preventative potential. In fact, one study in mice found that maternal supplementation of Lactococcus lactis (strain not disclosed) from 10.5 days of gestation increased blood vessel numbers and size in the cortical plate and cortical neurons density in offspring of both sexes at postnatal day 1, while the numbers of mitotic neural progenitor cells were increased in females only [77]. Interestingly, effects were investigated in the context of anxiety, which is known to appear early among psychiatric disorders [75]. At 10 weeks of age, females but not males from the above study had a higher activity level in bright zones and reduced fearful behavior [77]. Similarly, adolescent offspring, especially females, of rats that received Lactobacillus helveticus NS8 in late pregnancy spent more time in open spaces compared to controls [63]. No effects on cognition and behavior were seen in 11 years old children born at risk for allergic disease and exposed to Lacticaseibacillus rhamnosus HN001 or Bifidobacterium animalis subsp. lactis HN019 since late gestation and until 2 years of age [78]. Interestingly, these benefits of probiotics are also seen in the context of obesity. Administration of a multi-strain probiotic to mouse dams since conception prevented obesity-induced anxiety-like behavior in offspring at weaning and at adulthood in both sexes [79••]. Thus, findings from these recent pre-clinical studies suggest that maternal probiotic intake may reduce anxiety-like behavior in the next generation, potentially through modulation of neurodevelopment processes in utero and early life. Moreover, these studies suggest that effects may be sex-specific. Since psychiatric disorders manifest differently between sexes and are more prevalent and damaging in females compared to males [80], more studies should be conducted to understand if maternally administered probiotics hold potential for sex-targeted clinical applications.
Allergic Diseases
The effect of probiotic supplementation during prenatal and early life on prevention of allergic diseases has been extensively studied, leading to the release of recommendations for populations at risk [7]. These guidelines are the same for both sexes, and previous studies largely focused on Lactobacillus species, alone or in combination with Bifidobacterium. Recent literature has continued investigating the mechanisms behind probiotic preventative effects. Studies in mice show that probiotics, typically administered starting at mid gestation, act through immune-mediated mechanisms (details in Table 1), in both models of asthma [81] and atopic dermatitis [82], and these effects may be strain-specific [82].
Interestingly, studies in healthy women receiving probiotics from 36 weeks of gestation to 3 months of lactation showed a reduction in infant atopic dermatitis incidence, which was associated with decreased proportion of Th22 but independent from Th1/Th2 balance [83, 84]. A clinical study of women with a family history of treated allergic diseases receiving Limosilactobacillus reuteri ATCC 55730 during late pregnancy reported a modulation in DNA methylation of CD4 + T cells genes related to immune maturation and allergy development in infants at birth, suggesting that epigenetic modifications may mediate the preventative effects of probiotics in this context [85]. Further studies are needed to decipher the exact mechanisms. Long-term effects were also investigated in clinical studies. While combined maternal and infant supplementation with a probiotic mix (Lactobacillus salivarius CUL61, Lactobacillus paracasei CUL08, Bifidobacterium animalis subsp. lactis CUL34, Bifidobacterium bifidum CUL20) had no effects on asthma or eczema prevalence in 5-year-old children [86], the administration of another probiotic mix (Lacticaseibacillus rhamnosus GG and LC705, Bifidobacterium breve Bb99, Propionibacterium freudenreichii ssp. shermanii JS) or of Lacticaseibacillus rhamnosus HN001 to the mother and infant decreased the lifetime prevalence of eczema and atopic sensitization [87, 88] and food allergy [88] in 10- and 11-year-old children, respectively. Probiotic preventive effects on upper respiratory tract infections [88] and eczema [89] were also reported in populations subgroups of caesarean-delivered infants, after 10 and 13 years, respectively. Of importance, Lacticaseibacillus rhamnosus HN001 was found not to have the same effects on infant eczema, wheeze, and atopic sensitization when given to the mother alone without infant supplementation [87, 90•], thus calling for more studies investigating the timing of probiotic administration.
Metabolic Disorders
Maternal nutritional status is a determinant of offspring metabolic health and both pre-clinical and clinical studies demonstrate that various components of the metabolic syndrome are susceptible to fetal programming [91]. The study of maternal probiotics administration and effects on offspring metabolic health is still in its infancy, with most human studies focused solely on maternal outcomes [47,48,49, 92]. In pre-clinical studies, offspring have been evaluated at weaning and adulthood, with hypertension and risk for cardiovascular disease being the most studied conditions [93,94,95,96]. Interestingly, the only pre-clinical study that we found administering probiotic since pre-conception identified sex- and time-specific (weaning versus young adults) benefits in offspring and stronger evidence for reduced cardiovascular dysfunction in female offspring [93]. Most other pre-clinical studies have been conducted in males only (Table 1).
A study in normal-weight women supplemented with Lacticaseibacillus rhamnosus GG and Bifidobacterium lactis Bb-12 from the 4th week of pregnancy until birth showed a reduction in DNA methylation of obesity and weight gain-related genes in offspring, suggesting again that epigenetics might be a mechanism underlying probiotics programming effects [56•]. As pre-clinical studies revealed that significant weight differences are observable at weaning and adulthood, although males and females were not studied separately, it would be interesting to assess whether maternal probiotics effects on offspring growth are also observable during infancy and puberty, and in a sex-specific manner.
Intestinal Microbiota
Modulation of the microbiota is not a prerequisite for probiotics effects. In addition, resilience of an established gut microbiota makes it difficult to modify using exogenous microorganisms, though microbiota susceptibility is likely higher during early stages of plasticity. A recent meta-analysis found that maternal exposures including body mass index, drug-induced alteration of the microbiota, and probiotics administration influence the offspring intestinal microbiota [97]. In animal models, maternal probiotic supplementation had different effects on the offspring gut microbiota composition and structure, depending on the species administered and the time of administration [58, 59, 62, 69,70,71, 73••, 81, 82, 93, 95, 96, 98,99,100]. Different effects were also observed in various regions of the intestinal tract [69]. Interestingly, co-administration of probiotic Lactiplantibacillus plantarum B90 and Saccharomyces cerevisiae P11 with prebiotic xylooligosaccharides modified the probiotic effect on microbiota outcomes of piglets [69]. Probiotics with and without prebiotic increased Bacteroidetes and Bifidobacterium jejunal relative abundance, while only co-administration also increased Firmicutes and Lactobacillus [69]. Most human studies of maternally administered probiotics did not assess infant microbiota. For those that did, maternal probiotic intake was found to transiently increase the relative abundance of administered probiotic species or strain in the offspring [83, 101], regardless of the participants being healthy or at risk for allergic diseases (Table 2). Pre-clinical studies are also the only studies assessing microbial outcomes long-term, including effects on microbial diversity and/or composition at puberty in healthy mice [98] and mice born to dams exposed to an obesogenic diet [93], as well as at young adulthood [93, 95, 96]. Interestingly, some of these studies investigated microbiota outcomes in offspring of both sexes. This is relevant because there is evidence for sex-specific microbiota taxa as early as 2 weeks after birth [102], which likely become more pronounced during puberty [103]. Some of the probiotics studied showed sex-dependent effects on microbiota structure and taxa relative abundance [73••, 93]. This may be a result of hormonal interactions or of the offspring microbiota sex-driven effects. The latter is aligned with the finding that the infant baseline microbiota may determine the success of a maternal probiotic intervention aimed at preventing atopic dermatitis [83].
Notably, none of these studies used metagenomic approaches and thus it remains unknown if probiotics modulated microbial function. This is important because some pre-clinical studies found that microbial metabolites were altered in response to probiotics, including increased fecal short chain fatty acids in response to Lacticaseibacillus rhamnosus [62, 96] and decreased plasma trimethylamine N-oxide (TMAO) in response to Lacticaseibacillus casei [96]. Short-chain fatty acids such as acetate, propionate, and, in particular, butyrate are bacterial metabolites produced by fermentation of dietary fibers, and help maintain intestinal homeostasis [104]. TMAO is produced by liver oxidation of the intestinal microbial metabolite trimethylamine and is a marker of cardiovascular diseases [105]. It will be important for future studies to expand on these analyses to determine if programming effects of probiotics manifest at the functional level and to determine the quality and quantity of the microbial metabolites to which the host becomes exposed.
Conclusions
Here, we have reviewed recent pre-clinical and clinical studies investigating programming effects of maternally administered probiotics on offspring health. Studies published during the past 5 years expand on previous knowledge in the context of growth and gut health and additionally describe effects in the context of the metabolic syndrome and behavior (Fig. 1). Underlying mechanisms are also starting to be investigated. The study of probiotics in the context of DOHaD is in its infancy and it is not currently possible to make recommendations for clinical practice beyond allergic diseases. Pre-clinical studies are essential to investigate maternal programming effects of multiple outcomes in both male and female offspring in strictly controlled conditions and to elucidate molecular mechanisms. To encourage transparency and reproducibility of studies among research groups, it is important that these studies systematically report study details, including probiotic strain and dose along with timing and duration of probiotic exposure; the Animal Research: Reporting of In Vivo Experiments (ARRIVE) guidelines offer comprehensive recommendations for reporting [106].
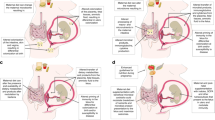
Offspring health outcomes programmed by maternal probiotic intake started prenatally. Previous knowledge (until 2017) included growth, gut health and intestinal barrier, allergic diseases, and gut microbiota findings. In addition to these outcomes, recent studies described effects on metabolic disorders, and neurodevelopment and anxiety. Clinical and pre-clinical findings are indicated in bold and italics, respectively. Icons depict findings assessed in infants or animal models (mice, rats, rabbits, or pigs). Created with BioRender.com
While most clinical studies initiated the administration of probiotics towards the end of pregnancy and continued until 3 months of lactation, pre-clinical studies started at the beginning of pregnancy and continued until weaning. There are no studies that isolated the pregnancy period, and only one mouse and one clinical study began supplementation at pre-conception; it would be important to understand which are the windows of susceptibility for probiotics to positively affect the offspring. Offspring characteristics, including sex, can also play a role. The male sex is more susceptible to in utero programming [107] and different placental DNA methylation patterns have been observed for male and female infants [108]. Interestingly, four studies reviewed here proposed DNA methylation as an underlying mechanism for programming effects of probiotics [56•, 84, 100, 109•]. Effects could also be mediated by the microbiota, with preventative benefits in inflammation [73••]. Interestingly, a clinical study of probiotics administered during late pregnancy and lactation for the prevention of atopic dermatitis found that individual microbiota characteristics, in this case, representation of Bifidobacterium dentium, could determine the effects of the probiotic intervention [83]. It would be important to study the programming effects of probiotics in infants at risk for altered microbial maturation patterns, for example very low birth weight or malnourished infants. Microbial maturation continues throughout adolescence and studies have in fact found that probiotic effects might appear during puberty and adulthood [62, 87,88,89, 94,95,96]. Beyond allergy-related outcomes, current human studies report findings at birth or in infants up to 2 years of age; longer trials will provide a more comprehensive understanding of the programming potential of probiotics. Finally, the studies reviewed were performed in Northern Europe, Asia, and Oceania calling for more trials to be conducted in America and Africa and to encompass ethnically diverse populations.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Hill C, Guarner F, Reid G, et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol. 2014;11:506–14.
Arrieta M-C, Stiemsma LT, Dimitriu PA, et al. Early infancy microbial and metabolic alterations affect risk of childhood asthma. Sci Transl Med. 2015;7:307ra152.
Tamburini S, Shen N, Wu HC, Clemente JC. The microbiome in early life: implications for health outcomes. Nat Med. 2016;22:713–22.
Azad MB, Bridgman SL, Becker AB, Kozyrskyj AL. Infant antibiotic exposure and the development of childhood overweight and central adiposity. Int J Obes. 2014;38:1290–8.
Kalliomäki M, Carmen Collado M, Salminen S, Isolauri E. Early differences in fecal microbiota composition in children may predict overweight. Am J Clin Nutr. 2008;87:534–8.
Stewart C, Marrs E, Magorrian S, Nelson A, Lanyon C, Perry J, Embleton N, Cummings S, Berrington J. The preterm gut microbiota: changes associated with necrotizing enterocolitis and infection. Acta Paediatr. 2012;101:1121–7.
Zuccotti G, Meneghin F, Aceti A, et al. Probiotics for prevention of atopic diseases in infants: systematic review and meta-analysis. Allergy. 2015;70:1356–71.
Fiocchi A, Pawankar R, Cuello-Garcia C, et al. World Allergy Organization-McMaster University Guidelines for Allergic Disease Prevention (GLAD-P): Probiotics. World Allergy Organ J. 2015;8:1–13.
Zhou L, Ding C, Wu J, Chen X, Ng DM, Wang H, Zhang Y, Shi N. Probiotics and synbiotics show clinical efficacy in treating gestational diabetes mellitus: a meta-analysis. Prim Care Diabetes. 2021;15:937–47.
Hanson L, VandeVusse L, Malloy E, Garnier-Villarreal M, Watson L, Fial A, Forgie M, Nardini K, Safdar N. Probiotic interventions to reduce antepartum Group B streptococcus colonization: a systematic review and meta-analysis. Midwifery. 2022;105:103208.
Jiménez E, Manzano S, Schlembach D, Arciszewski K, Martin R, Ben Amor K, Roelofs M, Knol J, Rodríguez JM, Abou-Dakn M. Ligilactobacillus salivarius PS2 supplementation during pregnancy and lactation prevents mastitis: a randomised controlled trial. Microorganisms. 2021;9:1933.
Christensen JJ, Retterstøl K, Godang K, Roland MCP, Qvigstad E, Bollerslev J, Ueland T, Henriksen T, Holven KB. LDL cholesterol in early pregnancy and offspring cardiovascular disease risk factors. J Clin Lipidol. 2016;10:1369–78.
Schuchat A. Group B streptococcus. The Lancet. 1999;353:51–6.
Schultz M, Göttl C, Young RJ, Iwen P, Vanderhoof JA. Administration of oral probiotic bacteria to pregnant women causes temporary infantile colonization. J Pediatr Gastroenterol Nutr. 2004;38:293–7.
Gueimonde M, Sakata S, Kalliomäki M, Isolauri E, Benno Y, Salminen S. Effect of maternal consumption of lactobacillus GG on transfer and establishment of fecal bifidobacterial microbiota in neonates. J Pediatr Gastroenterol Nutr. 2006;42:166–70.
Sheyholislami H, Connor KL. Are probiotics and prebiotics safe for use during pregnancy and lactation? A systematic review and meta-analysis Nutrients. 2021;13:2382.
Gille C, Böer B, Marschal M, et al. Effect of probiotics on vaginal health in pregnancy. EFFPRO, a randomized controlled trial. Am J Obstet Gynecol. 2016;215:608.
van den Nieuwboer M, Claassen E, Morelli L, Guarner F, Brummer RJ. Probiotic and synbiotic safety in infants under two years of age. Benef Microbes. 2014;5:45–60.
Bridgman SL, Azad MB, Field CJ, Letourneau N, Johnston DW, Kaplan BJ, Kozyrskyj AL. Maternal perspectives on the use of probiotics in infants: a cross-sectional survey. BMC Complement Altern Med. 2014;14:366.
Nyangahu DD, Lennard KS, Brown BP, et al. Disruption of maternal gut microbiota during gestation alters offspring microbiota and immunity. Microbiome. 2018;6:124.
Gonzalez-Perez G, Hicks AL, Tekieli TM, Radens CM, Williams BL, Lamousé-Smith ESN. Maternal antibiotic treatment impacts development of the neonatal intestinal microbiome and antiviral immunity. J Immunol. 2016;196:3768–79.
Mueller NT, Whyatt R, Hoepner L, Oberfield S, Dominguez-Bello MG, Widen EM, Hassoun A, Perera F, Rundle A. Prenatal exposure to antibiotics, cesarean section and risk of childhood obesity. Int J Obes (Lond). 2015;39:665–70.
Food and Agricultural Organization of the United Nations, World Health Organization. Probiotics in food: Health and nutritional properties and guidelines for evaluation - Report of a Joint FAO/WHO Expert Consultation on Evaluation of Health and Nutritional Properties of Probiotics in Food including Powder Milk with Live Lactic Acid Bacteria. Rome, Italy: FAO/WHO; 2006.
The Center for Reviews and Dissemination (CRD). Systematic Reviews: CRD’s guidance for undertaking reviews in health care. York: York Associates International; 2008.
Lucas A. Programming by early nutrition in man. Ciba Found Symp. 1991;156:38–50 discussion 50–55.
Calkins K, Devaskar SU. Fetal origins of adult disease. Curr Probl Pediatr Adolesc Health Care. 2011;41:158–76.
Lucas A. Role of nutritional programming in determining adult morbidity. Arch Dis Child. 1994;71:288–90.
Langley-Evans SC. Nutrition in early life and the programming of adult disease: a review. J Hum Nutr Diet. 2015;28:1–14.
Sánchez-Hernández D, Poon AN, Kubant R, et al. High vitamin A intake during pregnancy modifies dopaminergic reward system and decreases preference for sucrose in Wistar rat offspring. J Nutr Biochem. 2016;27:104–11.
Woo V, Alenghat T. Epigenetic regulation by gut microbiota. Gut Microbes. 2022;14:2022407.
Miro-Blanch J, Yanes O. Epigenetic regulation at the interplay between gut microbiota and host metabolism. Front Genet. 2019;10:638.
Takahashi K, Sugi Y, Nakano K, Tsuda M, Kurihara K, Hosono A, Kaminogawa S. Epigenetic control of the host gene by commensal bacteria in large intestinal epithelial cells. J Biol Chem. 2011;286:35755–62.
Cortese R, Lu L, Yu Y, Ruden D, Claud EC. Epigenome-Microbiome crosstalk: a potential new paradigm influencing neonatal susceptibility to disease. Epigenetics. 2016;11:205–15.
Berg G, Rybakova D, Fischer D, et al. Microbiome definition re-visited: old concepts and new challenges. Microbiome. 2020;8:103.
McBurney MI, Davis C, Fraser CM, Schneeman BO, Huttenhower C, Verbeke K, Walter J, Latulippe ME. Establishing what constitutes a healthy human gut microbiome: state of the science, regulatory considerations, and future directions. J Nutr. 2019;149:1882–95.
Ferretti P, Pasolli E, Tett A, et al. Mother-to-infant microbial transmission from different body sites shapes the developing infant gut microbiome. Cell Host Microbe. 2018;24:133-145.e5.
Derrien M, Alvarez A-S, de Vos WM. The gut microbiota in the first decade of life. Trends Microbiol. 2019;27:997–1010.
Sarkar A, Yoo JY, Valeria Ozorio Dutra S, Morgan KH, Groer M. The association between early-life gut microbiota and long-term health and diseases. J Clin Med. 2021. https://doi.org/10.3390/jcm10030459.
Luoto R, Kalliomäki M, Laitinen K, Isolauri E. The impact of perinatal probiotic intervention on the development of overweight and obesity: follow-up study from birth to 10 years. Int J Obes. 2010;34:1531–7.
Gareau MG, Wine E, Reardon C, Sherman PM. Probiotics prevent death caused by Citrobacter rodentium infection in neonatal mice. J Infect Dis. 2010;201:81–91.
Fåk F, Ahrné S, Molin G, Jeppsson B, Weström B. Maternal consumption of Lactobacillus plantarum 299v affects gastrointestinal growth and function in the suckling rat. Br J Nutr. 2008;100:332–8.
Bernardeau M, Vernoux J-P. Overview of differences between microbial feed additives and probiotics for food regarding regulation, growth promotion effects and health properties and consequences for extrapolation of farm animal results to humans. Clin Microbiol Infect. 2013;19:321–30.
Angelakis E. Weight gain by gut microbiota manipulation in productive animals. Microb Pathog. 2017;106:162–70.
Storelli G, Defaye A, Erkosar B, Hols P, Royet J, Leulier F. Lactobacillus plantarum promotes Drosophila systemic growth by modulating hormonal signals through TOR-dependent nutrient sensing. Cell Metab. 2011;14:403–14.
Schwarzer M, Makki K, Storelli G, et al. Lactobacillus plantarum strain maintains growth of infant mice during chronic undernutrition. Science. 2016;351:854–7.
Luoto R, Laitinen K, Nermes M, Isolauri E. Impact of maternal probiotic-supplemented dietary counselling on pregnancy outcome and prenatal and postnatal growth: a double-blind, placebo-controlled study. Br J Nutr. 2010;103:1792–9.
Okesene-Gafa KAM, Li M, McKinlay CJD, et al. Effect of antenatal dietary interventions in maternal obesity on pregnancy weight-gain and birthweight: Healthy Mums and Babies (HUMBA) randomized trial. Am J Obstet Gynecol. 2019;221:152.e1-152.e13.
Halkjær SI, de Knegt VE, Lo B, et al. Multistrain probiotic increases the gut microbiota diversity in obese pregnant women: results from a randomized, double-blind placebo-controlled study. Curr Dev Nutr. 2020;4:nzaa095.
Asgharian H, Homayouni-Rad A, Mirghafourvand M, Mohammad-Alizadeh-Charandabi S. Effect of probiotic yoghurt on plasma glucose in overweight and obese pregnant women: a randomized controlled clinical trial. Eur J Nutr. 2020;59:205–15.
Shahriari A, Karimi E, Shahriari M, Aslani N, Khooshideh M, Arab A. The effect of probiotic supplementation on the risk of gestational diabetes mellitus among high-risk pregnant women: a parallel double-blind, randomized, placebo-controlled clinical trial. Biomed Pharmacother. 2021;141:111915.
Kijmanawat A, Panburana P, Reutrakul S, Tangshewinsirikul C. Effects of probiotic supplements on insulin resistance in gestational diabetes mellitus: A double-blind randomized controlled trial. J Diabetes Investig. 2019;10:163–70.
Chen Y, Li Z, Tye KD, et al. Probiotic supplementation during human pregnancy affects the gut microbiota and immune status. Front Cell Infect Microbiol. 2019;9:254.
Yang P, Li Z, Tye KD, Chen Y, Lu T, He Z, Zhou J, Xiao X. Effects of an orally supplemented probiotic on the autophagy protein LC3 and Beclin1 in placentas undergoing spontaneous delivery during normal pregnancy. BMC Pregnancy Childbirth. 2020;20:216.
• Pourmirzaiee MA, Famouri F, Moazeni W, Hassanzadeh A, Hajihashemi M. The efficacy of the prenatal administration of Lactobacillus reuteri LR92 DSM 26866 on the prevention of infantile colic: a randomized control trial. Eur J Pediatr. 2020;179:1619–26. This study adds to current knowledge on postnatal administration of probiotics to mitigate colic and suggests that maternal prenatal probiotic intake may also have potential.
Wickens KL, Barthow CA, Murphy R, et al. Early pregnancy probiotic supplementation with Lactobacillus rhamnosus HN001 may reduce the prevalence of gestational diabetes mellitus: a randomised controlled trial. Br J Nutr. 2017;117:804–13.
• Vähämiko S, Laiho A, Lund R, Isolauri E, Salminen S, Laitinen K. The impact of probiotic supplementation during pregnancy on DNA methylation of obesity-related genes in mothers and their children. Eur J Nutr. 2019;58:367–77. This paper provides clinical mechanistic evidence underlying the programming effects by probiotics in the context of obesity.
Pérez-Castillo ÍM, Fernández-Castillo R, Lasserrot-Cuadrado A, Gallo-Vallejo JL, Rojas-Carvajal AM, Aguilar-Cordero MJ. Reporting of perinatal outcomes in probiotic randomized controlled trials. A systematic review and meta-analysis Nutrients. 2021;13:256.
Azagra-Boronat I, Tres A, Massot-Cladera M, Franch À, Castell M, Guardiola F, Pérez-Cano FJ, Rodríguez-Lagunas MJ. Lactobacillus fermentum CECT5716 supplementation in rats during pregnancy and lactation impacts maternal and offspring lipid profile, immune system and microbiota. Cells. 2020;9:575.
Liu T, Song X, An Y, et al. Lactobacillus rhamnosus GG colonization in early life ameliorates inflammaging of offspring by activating SIRT1/AMPK/PGC-1α pathway. Oxid Med Cell Longev. 2021;2021:3328505.
Han L, Azad MdAK, Huang P, Wang W, Zhang W, Blachier F, Kong X. Maternal supplementation with different probiotic mixture from late pregnancy to day 21 postpartum: consequences for litter size, plasma and colostrum parameters, and fecal microbiota and metabolites in sows. Front Vet Sci. 2022;9:726276.
Zhang Q, Li J, Cao M, et al. Dietary supplementation of Bacillus subtilis PB6 improves sow reproductive performance and reduces piglet birth intervals. Anim Nutr. 2020;6:278–87.
Zhou B, Jin G, Pang X, et al. Lactobacillus rhamnosus GG colonization in early life regulates gut-brain axis and relieves anxiety-like behavior in adulthood. Pharmacol Res. 2022;177:106090.
Niu Y, Liang S, Wang T, Hu X, Li W, Wu X, Jin F. Pre-Gestational intake of Lactobacillus helveticus NS8 has anxiolytic effects in adolescent Sprague Dawley offspring. Brain Behav. 2020. https://doi.org/10.1002/brb3.1714.
Bischoff SC, Barbara G, Buurman W, Ockhuizen T, Schulzke J-D, Serino M, Tilg H, Watson A, Wells JM. Intestinal permeability–a new target for disease prevention and therapy. BMC Gastroenterol. 2014;14:189.
Martens EC, Neumann M, Desai MS. Interactions of commensal and pathogenic microorganisms with the intestinal mucosal barrier. Nat Rev Microbiol. 2018;16:457–70.
Rokhsefat S, Lin A, Comelli EM. Mucin–microbiota interaction during postnatal maturation of the intestinal ecosystem: clinical implications. Dig Dis Sci. 2016;61:1473–86.
Sciascia Q, Daş G, Metges CC. REVIEW: The pig as a model for humans: effects of nutritional factors on intestinal function and health1. J Anim Sci. 2016;94:441–52.
Crespo-Piazuelo D, Gardiner GE, Ranjitkar S, Bouwhuis MA, Ham R, Phelan JP, Marsh A, Lawlor PG. Maternal supplementation with Bacillus altitudinis spores improves porcine offspring growth performance and carcass weight. Br J Nutr. 2022;127:403–20.
Wang K, Kong X, Azad MAK, Zhu Q, Xiong L, Zheng Y, Hu Z, Yin Y, He Q. Maternal probiotic or synbiotic supplementation modulates jejunal and colonic antioxidant capacity, mitochondrial function, and microbial abundance in Bama mini-piglets. Oxid Med Cell Longev. 2021;2021:6618874.
Wang K, Hu C, Tang W, Azad MAK, Zhu Q, He Q, Kong X. The enhancement of intestinal immunity in offspring piglets by maternal probiotic or synbiotic supplementation is associated with the alteration of gut microbiota. Front Nutr. 2021;8:686053.
Lan R, Kim I. Enterococcus faecium supplementation in sows during gestation and lactation improves the performance of sucking piglets. Vet Med Sci. 2019;6:92–9.
Betancur C, Martínez Y, Tellez-Isaias G, Castillo R, Ding X. Effect of oral administration with Lactobacillus plantarum CAM6 strain on sows during gestation-lactation and the derived impact on their progeny performance. Mediators Inflamm. 2021;2021:6615960.
•• Krishna M, Engevik M, Queliza K, Britto S, Shah R, Ruan W, Wang H, Versalovic J, Kellermayer R. Maternal Lactobacillus reuteri supplementation shifts the intestinal microbiome in mice and provides protection from experimental colitis in female offspring. FASEB Bioadv. 2021;4:109–20. This study considers individual susceptibility to colitis and shows that maternal probiotics may mitigate its effects in adult offspring. It is also one of the few studies assessing sex-specific responses.
Mörkl S, Butler MI, Holl A, Cryan JF, Dinan TG. Probiotics and the microbiota-gut-brain axis: focus on psychiatry. Curr Nutr Rep. 2020;9:171–82.
Leonardo ED, Hen R. Anxiety as a developmental disorder. Neuropsychopharmacology. 2008;33:134–40.
Vuong HE, Pronovost GN, Williams DW, Coley EJL, Siegler EL, Qiu A, Kazantsev M, Wilson CJ, Rendon T, Hsiao EY. The maternal microbiome modulates fetal neurodevelopment in mice. Nature. 2020;586:281–6.
Surzenko N, Pjetri E, Munson CA, Friday WB, Hauser J, Mitchell ES. Prenatal exposure to the probiotic Lactococcus lactis decreases anxiety-like behavior and modulates cortical cytoarchitecture in a sex specific manner. PLoS ONE. 2020;15:e0223395.
Slykerman RF, Kang J, Van Zyl N, et al. Effect of early probiotic supplementation on childhood cognition, behaviour and mood a randomised, placebo-controlled trial. Acta Paediatr. 2018;107:2172–8.
•• Radford-Smith DE, Probert F, Burnet PWJ, Anthony DC. Modifying the maternal microbiota alters the gut–brain metabolome and prevents emotional dysfunction in the adult offspring of obese dams. Proc Natl Acad Sci USA. 2022;119:e2108581119. One of the few papers connecting gut microbiota, metabolic syndrome, and brain function and the first to target this tripartite interaction in the context of DOHaD and the use probiotics as preventative therapeutics.
McLean CP, Asnaani A, Litz BT, Hofmann SG. Gender differences in anxiety disorders: prevalence, course of illness, comorbidity and burden of illness. J Psychiatr Res. 2011;45:1027–35.
Terada-Ikeda C, Kitabatake M, Hiraku A, Kato K, Yasui S, Imakita N, Ouji-Sageshima N, Iwabuchi N, Hamada K, Ito T. Maternal supplementation with Bifidobacterium breve M-16V prevents their offspring from allergic airway inflammation accelerated by the prenatal exposure to an air pollutant aerosol. PLoS ONE. 2020;15:e0238923.
Zhao Y, Qi C, Li X, et al. Prevention of atopic dermatitis in mice by Lactobacillus reuteri Fn041 through induction of regulatory T cells and modulation of the gut microbiota. Mol Nutr Food Res. 2022;66:2100699.
Avershina E, Cabrera Rubio R, Lundgård K, Perez Martinez G, Collado MC, Storrø O, Øien T, Dotterud CK, Johnsen R, Rudi K. Effect of probiotics in prevention of atopic dermatitis is dependent on the intrinsic microbiota at early infancy. J Allergy Clin Immunol. 2017;139:1399–402.
Rø ADB, Simpson MR, Rø TB, Storrø O, Johnsen R, Videm V, Øien T. Reduced Th22 cell proportion and prevention of atopic dermatitis in infants following maternal probiotic supplementation. Clin Exp Allergy. 2017;47:1014–21.
Forsberg A, Huoman J, Söderholm S, Bhai Mehta R, Nilsson L, Abrahamsson TR, Ernerudh J, Gustafsson M, Jenmalm MC. Pre- and postnatal Lactobacillus reuteri treatment alters DNA methylation of infant T helper cells. Pediatr Allergy Immunol. 2020;31:544–53.
Davies G, Jordan S, Brooks CJ, Thayer D, Storey M, Morgan G, Allen S, Garaiova I, Plummer S, Gravenor M. Long term extension of a randomised controlled trial of probiotics using electronic health records. Sci Rep. 2018;8:7668.
Wickens K, Barthow C, Mitchell EA, Kang J, van Zyl N, Purdie G, Stanley T, Fitzharris P, Murphy R, Crane J. Effects of Lactobacillus rhamnosus HN001 in early life on the cumulative prevalence of allergic disease to 11 years. Pediatr Allergy Immunol. 2018;29:808–14.
Peldan P, Kukkonen AK, Savilahti E, Kuitunen M. Perinatal probiotics decreased eczema up to 10 years of age, but at 5–10 years, allergic rhino-conjunctivitis was increased. Clin Exp Allergy. 2017;47:975–9.
Kallio S, Kukkonen AK, Savilahti E, Kuitunen M. Perinatal probiotic intervention prevented allergic disease in a Caesarean-delivered subgroup at 13-year follow-up. Clin Exp Allergy. 2019;49:506–15.
• Wickens K, Barthow C, Mitchell EA, et al. Maternal supplementation alone with Lactobacillus rhamnosus HN001 during pregnancy and breastfeeding does not reduce infant eczema. Pediatr Allergy Immunol. 2018;29:296–302. This paper contrasts administration of probiotics during pregnancy and breastfeeding to mother only versus mothers and their offspring, thus contributing essential knowledge to determine optimal probiotic administration protocols to program specific offspring outcomes.
de Gusmão Correia ML, Volpato AM, Águila MB, Mandarim-de-Lacerda CA. Developmental origins of health and disease: experimental and human evidence of fetal programming for metabolic syndrome. J Hum Hypertens. 2012;26:405–19.
Callaway LK, McIntyre HD, Barrett HL, et al. Probiotics for the prevention of gestational diabetes mellitus in overweight and obese women: findings from the SPRING double-blind randomized controlled trial. Diabetes Care. 2019;42:364–71.
Guo Y, Wang Z, Chen L, Tang L, Wen S, Liu Y, Yuan J. Diet induced maternal obesity affects offspring gut microbiota and persists into young adulthood. Food Funct. 2018;9:4317–27.
Guimarães KSDL, Braga VDA, Noronha SISRD, et al. Lactiplantibacillus plantarum WJL administration during pregnancy and lactation improves lipid profile, insulin sensitivity and gut microbiota diversity in dyslipidemic dams and protects male offspring against cardiovascular dysfunction in later life. Food Funct. 2020;11:8939–50.
Hsu C-N, Lin Y-J, Hou C-Y, Tain Y-L. Maternal administration of probiotic or prebiotic prevents male adult rat offspring against developmental programming of hypertension induced by high fructose consumption in pregnancy and lactation. Nutrients. 2018;10:E1229.
Hsu C-N, Hou C-Y, Chan JYH, Lee C-T, Tain Y-L. hypertension programmed by perinatal high-fat diet: effect of maternal gut microbiota-targeted therapy. Nutrients. 2019;11:2908.
Grech A, Collins CE, Holmes A, Lal R, Duncanson K, Taylor R, Gordon A. Maternal exposures and the infant gut microbiome: a systematic review with meta-analysis. Gut Microbes. 2021;13:1897210.
Qi Y, Yu L, Tian F, Zhao J, Zhang H, Chen W, Zhai Q. A. muciniphila supplementation in mice during pregnancy and lactation affects the maternal intestinal microenvironment. Nutrients. 2022;14:390.
Liu L, Zeng D, Yang M, et al. Probiotic Clostridium butyricum improves the growth performance, immune function, and gut microbiota of weaning rex rabbits. Probiotics & Antimicro Prot. 2019;11:1278–92.
Xiao J, Wang T, Xu Y, Gu X, Li D, Niu K, Wang T, Zhao J, Zhou R, Wang H-L. Long-term probiotic intervention mitigates memory dysfunction through a novel H3K27me3-based mechanism in lead-exposed rats. Transl Psychiatry. 2020;10:25.
Korpela K, Salonen A, Vepsäläinen O, et al. Probiotic supplementation restores normal microbiota composition and function in antibiotic-treated and in caesarean-born infants. Microbiome. 2018;6:182.
Chen J, Li H, Hird SM, Chen M-H, Xu W, Maas K, Cong X. Sex differences in gut microbial development of preterm infant twins in early life: a longitudinal analysis. Front Cell Infect Microbiol. 2021;11:671074.
Org E, Mehrabian M, Parks BW, Shipkova P, Liu X, Drake TA, Lusis AJ. Sex differences and hormonal effects on gut microbiota composition in mice. Gut Microbes. 2016;7:313–22.
Wong JMW, de Souza R, Kendall CWC, Emam A, Jenkins DJA. Colonic health: fermentation and short chain fatty acids. J Clin Gastroenterol. 2006;40:235–43.
Yang S, Li X, Yang F, et al. Gut microbiota-dependent marker TMAO in promoting cardiovascular disease: inflammation mechanism, clinical prognostic, and potential as a therapeutic target. Front Pharmacol. 2019;10:1360.
The ARRIVE guidelines 2.0. In: ARRIVE Guidelines. https://arriveguidelines.org/arrive-guidelines. Accessed 14 Oct 2021
Eriksson JG, Kajantie E, Osmond C, Thornburg K, Barker DJP. Boys live dangerously in the womb. Am J Hum Biol. 2010;22:330–5.
Martin E, Smeester L, Bommarito PA, Grace MR, Boggess K, Kuban K, Karagas MR, Marsit CJ, O’Shea TM, Fry RC. Sexual epigenetic dimorphism in the human placenta: implications for susceptibility during the prenatal period. Epigenomics. 2017;9:267–78.
• Huoman J, Martínez-Enguita D, Olsson E, Ernerudh J, Nilsson L, Duchén K, Gustafsson M, Jenmalm MC. Combined prenatal Lactobacillus reuteri and ω-3 supplementation synergistically modulates DNA methylation in neonatal T helper cells. Clin Epigenetics. 2021;13:135. This paper shows that omega-3 fatty acids and L. reuteri have a synergetic effect on DNA methylation in neonates, suggesting that co-administration of probiotics with other supplements may be a strategy to amplify effects.
Cruz LX, Hirsch CD, de Moura MQ, de Avila LFC, Martins LHR, Klafke GB, Conceição FR, Berne MEA, Scaini CJ. Saccharomyces boulardii reduces the vertical transmission of Toxocara canis larvae in mice. J Helminthol. 2021;95:e11.
Menegat MB, DeRouchey JM, Woodworth JC, Dritz SS, Tokach MD, Goodband RD. Effects of Bacillus subtilis C-3102 on sow and progeny performance, fecal consistency, and fecal microbes during gestation, lactation, and nursery periods. J Anim Sci. 2019;97:3920–37.
Melandri M, Aiudi GG, Caira M, Alonge S. A Biotic Support During Pregnancy to Strengthen the Gastrointestinal Performance in Puppies. Front Vet Sci. 2020;7:417.
Lu J, Lu L, Yu Y, Baranowski J, Claud EC. Maternal administration of probiotics promotes brain development and protects offspring’s brain from postnatal inflammatory insults in C57/BL6J mice. Sci Rep. 2020;10:8178.
Kar F, Hacioglu C, Kar E, Donmez DB, Kanbak G. Probiotics ameliorates LPS induced neuroinflammation injury on Aβ 1–42, APP, γ-β secretase and BDNF levels in maternal gut microbiota and fetal neurodevelopment processes. Metab Brain Dis. 2022. https://doi.org/10.1007/s11011-022-00964-z.
Godfrey KM, Barton SJ, El-Heis S, Kenealy T, Nield H, Baker PN, Chong YS, Cutfield W, Chan S-Y. Myo-inositol, probiotics, and micronutrient supplementation from preconception for glycemia in pregnancy: NiPPeR international multicenter double-blind randomized controlled trial. Diabetes Care. 2021;44:1091–9.
Mantaring J, Benyacoub J, Destura R, Pecquet S, Vidal K, Volger S, Guinto V. Effect of maternal supplement beverage with and without probiotics during pregnancy and lactation on maternal and infant health: a randomized controlled trial in the Philippines. BMC Pregnancy Childbirth. 2018;18:193.
Schei K, Avershina E, Øien T, Rudi K, Follestad T, Salamati S, Ødegård RA. Early gut mycobiota and mother-offspring transfer. Microbiome. 2017;5:107.
Dewanto NEF, Firmansyah A, Sungkar A, Dharmasetiawani N, Sastroasmoro S, Kresno SB, Suradi R, Bardosono S, Prasetyo D. The effect of Bifidobacterium animalis lactis HNO19 supplementation among pregnant and lactating women on interleukin-8 level in breast milk and infant’s gut mucosal integrity. Medical Journal of Indonesia. 2017;26:204–11.
Forsberg A, Abrahamsson TR, Nilsson L, Ernerudh J, Duchén K, Jenmalm MC. Changes in peripheral immune populations during pregnancy and modulation by probiotics and ω-3 fatty acids. Sci Rep. 2020;10:18723.
Pellonperä O, Mokkala K, Houttu N, Vahlberg T, Koivuniemi E, Tertti K, Rönnemaa T, Laitinen K. Efficacy of fish oil and/or probiotic intervention on the incidence of gestational diabetes mellitus in an at-risk group of overweight and obese women: a randomized, placebo-controlled, double-blind clinical trial. Diabetes Care. 2019;42:1009–17.
Sahhaf Ebrahimi F, Homayouni Rad A, Mosen M, Abbasalizadeh F, Tabrizi A, Khalili L. Effect of L. acidophilus and B. lactis on blood glucose in women with gestational diabetes mellitus: a randomized placebo-controlled trial. Diabetol Metab Syndr. 2019;11:75.
Funding
Work in the laboratory of EMC is supported by the Natural Sciences and Engineering Research Council of Canada (NSERC) (Grant # RGPIN-2019–06100) and the Canadian Institutes of Health Research (CIHR). EMC was the recipient of the Lawson Family Chair in Microbiome Nutrition Research. SES was funded by NSERC Graduate Scholarships-Master’s (NSERC CGS M) and Ontario Graduate Scholarship (OGS).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
EMC has received research support from Lallemand Health Solutions and Ocean Spray and has received consultant fees or speaker or travel support from Danone and Lallemand Health Solutions.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Functional Foods
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cuinat, C., Stinson, S.E., Ward, W.E. et al. Maternal Intake of Probiotics to Program Offspring Health. Curr Nutr Rep 11, 537–562 (2022). https://doi.org/10.1007/s13668-022-00429-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13668-022-00429-w