Abstract
Introduction
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder associated with memory, cognitive, and behavioral deficits, and brings significant economic burden on caregivers and healthcare systems. This study aims to estimate the long-term societal value of lecanemab plus standard of care (SoC) versus SoC alone, corresponding to a range of willingness-to-pay (WTP) thresholds based on the phase III CLARITY AD trial readouts from both the US payer and societal perspectives.
Methods
An evidence-based model was developed to simulate the effects of lecanemab on disease progression in early AD using interconnected predictive equations based on longitudinal clinical and biomarker data derived from the Alzheimer’s Disease Neuroimaging Initiative (ADNI). The model was informed with the results of the phase III CLARITY AD trial and published literature. Key model outcomes included patient life-years (LYs), quality-adjusted life-years (QALYs), and total costs of both the direct and indirect costs of patients and caregivers over a lifetime horizon.
Results
Patients treated with lecanemab plus SoC gained an additional 0.62 years of life versus SoC alone (6.23 years vs. 5.61 years). The mean time on lecanemab was 3.91 years, and the treatment was associated with an increase in patient QALYs of 0.61 and an increase in total QALYs of 0.64 when both patient and caregiver utilities were considered. The model estimated that the annual value of lecanemab for the US payer perspective was US$18,709–35,678 ($19,710–37,351 for societal perspective) at the WTP threshold of $100,000–200,000 per QALY gained, respectively. Scenario analyses of patient subgroups, time horizon, input sources, treatment stopping rules, and treatment dosing were conducted to explore the impact of alternative assumptions on the model results.
Conclusion
The economic study suggested that lecanemab plus SoC would improve health and humanistic (quality of life) outcomes and reduce economic burden for patients and caregivers in early AD.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Alzheimer’s disease is a progressive neurodegenerative disease which brings significant economic burden on caregivers and healthcare systems. |
Using a validated disease simulation model and data from the large, confirmatory phase III CLARITY AD trial, this study estimates the societal value of lecanemab plus standard of care (SoC) versus SoC alone, considering various willingness-to-pay (WTP) thresholds and both the perspectives of the US payer and society. |
While a broader range of values was considered, the severity-adjusted willingness-to-pay (WTP) threshold of $200,000 per quality-adjusted life-year (QALY) gained accurately reflects the societal value of lecanemab. |
Patients treated with lecanemab plus standard of care experienced better health outcomes (i.e., additional life-years, quality-adjusted life-years gained, and decreased costs) than those treated with standard of care alone over a lifetime time horizon from both US payer and societal perspectives. |
Predicted health economic outcome results provide a foundation for healthcare decision-makers to understand the potential clinical, economic, and societal value of lecanemab. |
Introduction
Alzheimer’s disease (AD) is a progressive neurodegenerative disorder associated with memory, cognitive, and behavioral deficits, and is the most common cause of dementia, accounting for 60–70% of all dementia cases. [1] Among Americans aged over 65 years, AD is the fifth leading cause of death. Approximately 6.5 million Americans aged over 65 years are estimated to have AD, and this number is predicted to reach 13.8 million by 2060. [2] Clinically, AD progresses from normal cognition to mild cognitive impairment (MCI), followed by dementia. As the disease progresses, the severity of the associated cognitive and functional deficits increases, escalating the burden on caregivers and healthcare systems. [3] In 2021, family members and other caregivers provided approximately 16 billion hours of unpaid care, valued at US$271 billion; this care is associated with additional costs, such as emotional distress and detrimental mental and physical care outcomes. [2] In 2022, the estimated direct costs of providing long-term care and hospital services to those aged ≥ 65 years with dementia were $321 billion. [2]
AD is characterized by two neuropathological hallmark features—the accumulation of abnormal protein deposits in the brain, including ß-amyloid (Aβ) plaques and neurofibrillary tangles. Biomarkers are important indicators of the presence and progression of a disease, and they play a crucial role in diagnosing and monitoring AD. In AD, two common types of biomarkers are used: positron emission tomography (PET) imaging and cerebrospinal fluid (CSF) analysis. PET imaging estimates the amount of amyloid and tau in the brain, while CSF analysis measures the soluble biomarkers in the cerebrospinal fluid. Both biomarkers help diagnose and monitor AD development and progression. [2, 4] Disease-modifying therapies (DMTs), i.e., treatments interfering with the pathological mechanisms underlying disease processes, are currently a subject receiving close review by stakeholders in the field of AD research, and studies are focusing on DMTs that target various primary and intermediate mechanisms, including therapies that affect Aβ or tau. [5] Over 120 agents are currently under investigation in more than 150 clinical trials in various phases; of these agents, 82.5% target the mechanisms underlying AD to achieve disease modification. [6]
Lecanemab, a humanized IgG1 monoclonal antibody targeting amyloid protofibrils, was investigated in a large, confirmatory phase III clinical trial (CLARITY AD; NCT03887455). [7] The CLARITY AD study was an 18-month, multicenter, double-blind, placebo-controlled trial investigating treatment with lecanemab in early AD patients aged 50–90 years, with evidence of amyloid positivity confirmed by PET imaging or CSF measurement. The trial results demonstrated that the individuals randomly assigned to receive lecanemab (10 mg/kg every two weeks) experienced greater reductions in brain amyloid levels and slower clinical decline in cognition and function scales [Clinical Dementia Rating Scale-Sum of Boxes (CDR-SB) and Alzheimer’s Disease Composite Score (ADCOMS)] compared to those given placebo after 18 months trial duration.
Recent studies have been published to assess the long-term health outcomes [8] of lecanemab as well as to estimate the potential value of lecanemab [9] based on the efficacy observed in the phase IIb trial (Study 201). [10] The objective of this study was to estimate the long-term societal value of lecanemab plus standard of care (SoC) versus SoC alone, corresponding to a range of willingness-to-pay (WTP) thresholds based on the phase III CLARITY AD trial readouts. Alzheimer’s disease is a progressive and degenerative condition that significantly impairs an individual's quality of life and daily functioning, and imposes a considerable economic burden on caregivers and the healthcare system. Therefore, higher WTP thresholds may be appropriate to adequately address the disease's substantial burden. [11] To achieve this goal, a previously developed evidence-based disease simulation model [12] was updated using data from the phase III CLARITY AD trial and the published literature to compare lecanemab plus SoC versus SoC alone both from the US payer and societal perspectives. [12]
Methods
Model Overview
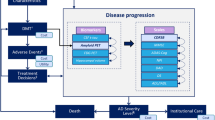
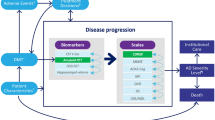
AD Archimedes condition-event (ACE) is a patient-level simulator that models the progression of AD based on complex interactions between the underlying pathological components (e.g., biomarkers measuring Aβ and tau levels) and clinical presentation of AD (e.g., patient-level scales measuring cognition, behavior, function, and dependence) to estimate the potential effects of DMTs on disease progression. [12, 13] AD ACE was developed based on a systemic literature review of economic modeling studies investigating AD, [14] the good modeling practice guidelines by the International Society of Pharmacoeconomics and Outcomes Research, [15] and a review of ongoing clinical trials investigating DMTs for AD. AD ACE has been validated using patient registries, clinical trials, and published literature, and has been shown to accurately predict and provide robust estimates of dementia, institutionalization, and mortality due to AD over a long-term horizon. [12, 13]
To simulate the effects of DMTs and interventions on disease progression in early AD, AD ACE estimates disease progression using interconnected predictive equations based on longitudinal clinical and biomarker data derived from the Alzheimer’s Disease Neuroimaging Initiative (ADNI). [16] The biomarkers extracted from the longitudinal ADNI dataset include CSF protein levels (Aβ1-42 and total tau), fluorodeoxyglucose-PET (assessing brain cell metabolic activity), and magnetic resonance imaging (MRI) (assessing hippocampal volume). Because the ADNI dataset does not effectively capture the more severe stages of AD, to simulate AD progression in more severe stages of AD, AD ACE relies on equations based on cognitive and behavioral scales derived from the Assessment of Health Economics in Alzheimer’s Disease II (AHEAD) study. [17, 18] This adjustment enhances the accuracy of the model and makes it more representative across the entire AD continuum and its varying levels of disease severity. Once a patient progresses to the moderate AD stage, as determined by a Mini Mental State Examination (MMSE) score below 15, representing a stage of moderately severe to severe Alzheimer’s, AD ACE switches from the ADNI equations to the AHEAD equations. Despite being derived independently from separate datasets, both the ADNI and AHEAD equations were found to provide similar predictions of disease progression rates across the range of mild to moderate AD. [12, 13] Compatibility and consistency in reported measures were carefully assessed and tested once switching between ADNI and AHEAD equations. A rigorous evaluation has been conducted following the transition from the ADNI to AHEAD equations to ensure that the reported measures remained consistent and compatible. [12, 13]
Furthermore, transitions between community and institutional care settings are captured by AD ACE as patients progress to more severe stages of AD. The detailed structure and equations of AD ACE have been previously published. [8, 9, 13, 19]
AD ACE includes the option to adopt either a payer or a societal perspective to capture the direct and indirect outcomes of AD over a time horizon of a lifetime. The key outcomes evaluated by the model include patient life-years (LYs), quality-adjusted life-years (QALYs), total costs, and incremental cost-effectiveness ratios. QALYs and costs are further stratified, and some components are optional. The total costs include both the direct and indirect costs of patients and caregivers in the community and institutional care settings, with indirect costs included only in the societal perspective reported separately. Furthermore, the overall QALYs are divided into patient QALYs, caregiver QALYs lost (optional), and QALYs lost due to amyloid-related imagining abnormalities-edema/effusion (ARIA-E) adverse events (AEs). Because the model accumulates outcomes over a lifetime time horizon, an annual discount rate of 3% was applied to all outcomes, following recommendations for cost-effectiveness analyses in the US. [20]
A population of patients in the early AD stage was selected for the base-case analysis. These patients had MCI due to AD or mild AD dementia with confirmed Aβ pathology. In total, ADNI included 1735 patient profiles, of which 260 were selected as inputs for AD ACE. This subgroup of patients was matched based on the inclusion criteria of the CLARITY AD trial as follows: an age of 50–85 years, an MMSE score greater than or equal to 22, and an amyloid PET standardized uptake value ratio (SUVr) of 1.1. [10] The mean baseline characteristics of the selected patients were highly consistent with those of the patients in the placebo and lecanemab groups in the phase III CLARITY AD trial (Table 1). In total, 2000 individual patient profiles were sampled from the selected subgroup of 260 ADNI patients with replacement. Then, the sampled profiles were simulated on the lecanemab plus SoC arm and SoC alone arm separately to capture the disease trajectory and treatment effect. Key patient subsets and various treatment stopping rules and dosing regimens were applied in the assessments.
To evaluate the influence of parameter uncertainty and the robustness of the results in different settings, scenario and sensitivity analyses were applied. In the scenario analyses, patient subsets were selected based on the base-case 260 ADNI profiles, and the impact of early or late treatment on the onset of AD dementia and progression to more severe disease was evaluated. Furthermore, the profiles were grouped by the baseline level of total tau (t-tau) in CSF to determine the impact of early or late treatment on the neurodegeneration level.
The CLARITY AD trial was conducted in accordance with the Declaration of Helsinki and the International Council for Harmonization and Good Clinical Practice guidelines, and was approved by the institutional review board or independent ethics committee at each site. All patients provided written informed consent. An independent interim monitoring committee was responsible for oversight and conduct of the interim analyses and response-adaptive randomization design to routinely evaluate the safety and review the futility analysis results.
This assessment relies on previously conducted studies and does not involve any new studies with human participants or animals conducted by the authors. The model parameters were primarily informed by published literature or the results of the CLARITY AD trial.
Model Inputs
Clinical Inputs
Disease Progression
AD ACE modeled the natural history of the progression of AD in patients receiving SoC using disease equations constructed based on longitudinal patient-level data from ADNI (i.e., early AD) [16] and published equations from AHEAD (i.e., more severe stages). [17, 18] To determine the patients’ disease severity at baseline and over time, AD ACE relied on the CDR-SB thresholds as follows: < 4.5, MCI due to AD; ≥ 4.5 to < 9.5, mild AD; ≥ 9.5 to < 16, moderate AD; and ≥ 16, severe AD. [21] Therefore, the proportion of patients with MCI due to AD at baseline should be comparable between AD ACE and the phase III CLARITY AD trial (i.e., the proportion of individuals with CDR-SB scores < 4.5 in AD ACE is comparable to that of individuals with CDR-Global scores = 0.5 in the CLARITY trial).
Mortality
To naturally increase the probability of death due to age in the model, the hazard ratios (HR) of age-specific mortality in the general US population [22] were applied to model mortality across all severity levels of AD. In the base-case settings, the excess mortality hazard derived from a previous population-based cohort study was then applied to individuals with mild to severe dementia as defined by their CDR-SB scores. [23] In the current analysis, MCI due to AD was considered to have no impact on the risk of mortality. The HRs applied in the scenario analyses were derived from a previous study. [24]
Institutionalization
The model applied estimates based on data from the Consortium to Establish a Registry for Alzheimer’s Disease [25] to determine the probability of transitioning from the community to institutionalized care by the severity level of AD. Patients with MCI due to AD were assumed to have no risk of institutionalization based on the available literature and minimal impact on the outcomes. In the scenario analyses, additional prevalence-based institutional data reflecting the proportion of patients institutionalized at each severity level of AD were further explored. [26]
Treatment Effect and Dosing of Lecanemab
Treatment with lecanemab is assumed to directly act on PET amyloid levels as a surrogate endpoint, which then influences other outcomes, [27, 28] since amyloid PET SUVr is a predictor in all disease equations used by AD ACE. AD ACE can model DMT targeting amyloid levels by evaluating the estimated amyloid PET SUVr outcomes of a simulated patient, and then projecting the course of disease progression for that patient’s lifetime to predict how the effect on amyloid PET SUVr will impact health–economic outcomes, such as LYs, QALYs, and total costs.
However, the primary outcome of the CLARITY AD trial was mean change from baseline in the CDR-SB at 18 months. To determine the appropriate treatment effect on amyloid PET SUVr needed to match the CDR-SB results from the trial, a calibration approach was used, in which the treatment effects on amyloid PET SUVr were tuned until the model result for CDR-SB matched the target values from the CLARITY AD. Since AD ACE applies treatment effects over time, the calibration process was applied to work back to the amyloid PET SUVr at each time interval. The calibrated reduction in the amyloid PET SUVr in a time interval influenced the values in subsequent time intervals for predicted amyloid PET SUVr, CDR-SB, and other AD biomarkers and scales. In the analysis, data regarding the change from baseline in the amyloid PET SUVr in the phase III CLARITY AD study were used as the basis for calibrating the mean amyloid PET SUVr reductions required at different time intervals. The calibrated reductions in amyloid PET SUVr resulted in predictions of CDR-SB that closely matched the changes from baseline in the CDR-SB observed in the trial during the first 18 months after treatment initiation (Fig. 1).
Furthermore, amyloid PET data from a model-based simulation study exploring the effect of continued treatment with lecanemab [29] were used to simulate the treatment effect beyond the time horizon of the CLARITY AD trial, until the mean amyloid level reached that observed in individuals with normal cognition in the ADNI dataset. [16] Then, the cognitively normal mean amyloid level in patients who remained on lecanemab was maintained by additional reduction in the amyloid PET level. This assumption was validated by clinical experts. The calibration process did not impact the default AD ACE equations, but only adjusted the estimated PET SUVr values over time.
In the base-case scenario, the model assumed that the patients receiving treatment were given 10 mg/kg of intravenous lecanemab biweekly, resulting in a 27% estimated change in the CDR-SB from baseline based on a conventional mixed model for repeated measures (MMRM). As MMRM cannot generate a confidence interval around ratios reflecting a percentage reduction or change from baseline, a variation of ± 15% was applied in the sensitivity analyses to evaluate uncertainty in the lecanemab treatment effect. This variation is consistent with the results across key randomization strata in the CLARITY AD trial [7] and a recently reported range of percentage reduction estimated using six different statistical approaches. [30] The potential impact of alternative treatment dosing regimens on the treatment effect during a maintenance phase was also evaluated in the scenario analyses. The treatment effect scenarios in the model after 18 months were based on the outcomes reported in a model-based simulation study investigating the effect of a long-term maintenance dosing regimen beyond the 18-month trial period. The study showed that less frequent dosing could prevent the re-accumulation of amyloid and maintain the treatment effect beyond the trial duration. [29] In each dosing regimen scenario, the mean amyloid reduction in the maintenance time intervals was recalibrated by adjusting the mean amyloid reductions estimated in the base-case setting.
Treatment Discontinuation
In the CLARITY AD trial, the risk of discontinuation was 18.8% (169 participants) in the lecanemab arm and 15.6% (140 participants) in the placebo arm throughout the 18-month trial period. [7] Study participants discontinued treatment due to AEs, opted to discontinue the treatment regimen, were lost to follow-up, withdrew consent, or discontinued for another unspecified reason. Adverse events leading to discontinuation of the trial agent occurred in 6.9% of the participants in the lecanemab arm and 2.9% of those in the placebo arm. [7] The overall annual risk of discontinuation, i.e., 13.0%, was used in the base-case analysis. Treatment discontinuation also occurred in the base case following progression to a moderate stage of AD, as determined by a CDR-SB score ≥ 9.5. In the sensitivity analyses, annual discontinuation risk levels of 10% and 20% were applied. In the scenario analyses, various treatment discontinuation rules based on fixed treatment durations of 1.5, 3, and 5 years were evaluated. Once a patient discontinued treatment, the mean calibrated amyloid reductions were no longer applied, and the changes in the amyloid level were estimated based on the risk equations for the natural history of disease progression. As a consequence of this approach, patients continued to experience a residual benefit of treatment even after discontinuation; however, this benefit gradually waned over time, such that eventually patients would be in a similar state as if they had not been treated.
AEs
The incidence rates of serious AEs were 14% in the lecanemab arm and 11.3% in the placebo arm. Infusion-related reactions (1.2% vs. 0 in the lecanemab arm and placebo arm, respectively), ARIA-E (0.8% vs. 0), atrial fibrillation (0.7% vs. 0.3%), syncope (0.7% vs. 0.1%), and angina pectoris (0.7% vs. 0) were the most commonly reported serious AEs. The overall incidence rates of AEs did not differ between the two arms. The most common AEs in the lecanemab arm were infusion-related reactions, ARIA with cerebral microhemorrhages, cerebral macrohemorrhages or superficial siderosis, headaches, and falls. Mild to moderate (grades 1–2, 96%) infusion reactions occurred mostly following the first dose (75%), and these patients positively responded to prophylactic treatment. In total, 113 (12.6%) cases of ARIA-E AEs were observed in 898 participants in the lecanemab group, with 25 (2.8%) symptomatic cases including headache, visual disturbance, and confusion. The ARIA-E events were mild to moderate (91%), mostly asymptotic (78%), reported during the first three months of the treatment (71%), and resolved within four months after detection (81%). Therefore, the model considered a 12.6% occurrence rate of ARIA-E adverse events in the first year, of which 22% were symptomatic. No treatment discontinuation due to ARIA-E AEs was considered in the model because these events only resulted in brief treatment interruptions in the phase III CLARITY AD trial.
Cost Inputs
This study evaluated the direct and indirect costs of community and residential care for patients and their caregivers. Sources providing cost data in the US across the full disease continuum are lacking; therefore, data from multiple sources were combined. The community-based care costs for patients with MCI and mild AD, including the direct medical and non-medical costs of healthcare resource use and indirect costs of caregiving, were obtained from GERAS-US, [31] a longitudinal, prospective cohort study adapted from GERAS I [32]. GERAS I evaluated the community-based costs of patients with mild, moderate, and severe dementia due to AD, and revealed that the costs increased along with the severity of the disease [32]. Both GERAS I and GERAS-US employed the MMSE scale to define the severity of AD [31, 32]. Because GERAS-US did not report the mean monthly costs of patients with moderate and severe dementia due to AD, the mean relative ratio between the estimated costs of mild AD and those of moderate and severe AD obtained from GERAS I was computed, and AD ACE applied this mean relative ratio to the community-based care costs for mild AD from GERAS-US to estimate the mean monthly costs. The computed mean relative ratios are consistent with previously reported findings regarding the average cost of care by disease stage (1.3 for mild to moderate AD and 2.0 for mild to severe AD) [19, 33] and the average ratios used by the Alzheimer’s Association (1.3 for mild to moderate AD and 2.0 for mild to severe AD) [34].
Genworth’s Cost of Care Survey tool [35] was used to obtain the direct non-medical costs in the residential setting. In 2021, the monthly median cost of a room in a residential care facility in the US was $8,477. Because this cost does not change as the severity of AD increases, the model did not adjust this cost. Other costs associated with residential care for both patients and caregivers were assumed to be the same as those for community-based care; however, regarding the indirect non-medical costs of caregiving, the model applied only 44% of the informal care giving cost in the community-based setting [36].
The patients receiving lecanemab further accrued monitoring costs, assumed to be the cost of five MRI scans in the first year. This approach aligns with the drug label recommendations and is easily accessible in typical clinical practice settings. In certain cases, individuals may require additional clinical or radiologic monitoring to ensure proper management of AEs; however, it is important to focus on the routine costs of disease management in economic evaluations. This is because including rare or unusual costs, such as those associated with extreme AE cases, may not accurately reflect the typical costs of the intervention or treatment being evaluated. Focusing on routine and typical costs allows for a more accurate representation of the true economic impact of the intervention on a broader population.
The unit costs were obtained from the Centers for Medicare and Medicaid Services (CMS) Physician Fee Schedule [37]. The estimates based on GERAS already included the symptomatic treatment costs, and thus, these costs were not included in the analysis. The medical resource use costs were obtained from the CMS database [37] and IBM® RED BOOK®. [38] The diagnostic and screening costs, i.e., CSF and PET scans, were not included in the base-case analysis due to variations in and uncertainties regarding reimbursement policies; however, these costs were evaluated in the scenario analyses. All costs were adjusted to 2022 US dollars according to the price index of personal consumer expenditures for healthcare by the US Bureau of Labor Statistics. [39] All cost categories are shown in Table 2.
Utilities
The patient utilities were obtained by conducting a fixed-effect meta-analysis of the studies included in a previously published systematic literature review [40]. The included studies defined the severity of AD using the MMSE, CDR-Global, Alzheimer’s Disease Assessment Scale-Cognitive Subscale, and Global Deterioration Scale. In the scenario analyses, alternative values based on the CDR-Global score obtained from Neumann et al. [41] were applied. The same utilities estimated based on the disease severity were applied in the community and institutionalized settings. For patients experiencing ARIA-E for 12 weeks, an estimated disutility due to headache of – 0.14 was applied [42]. Each patient was assumed to have one caregiver, and the caregiver disutilities were obtained from a previous study [43].
Results
Base-Case Analysis
Over a lifetime time horizon, patients treated with lecanemab plus SoC gained an additional 0.62 years of life versus SoC alone (6.23 years vs. 5.61 years). The mean time on lecanemab was 3.91 years, and the treatment was associated with an increase in patient QALYs of 0.61 and an increase in total QALYs of 0.64 when both patient and caregiver utilities were considered.
The total costs per patient associated with lecanemab plus SoC, excluding drug acquisition costs, decreased by $6,263 from a payer perspective (i.e., patient health and social care costs only) and $7,451 from the societal perspective (i.e., including caregiver healthcare and informal care costs) compared to SoC alone. From the payer perspective, the community care costs increased by $5,091 while the costs of residential care decreased by $12,439, resulting in a total decrease in care-related costs per patient of $7,349. When caregiver costs were considered, lecanemab plus SoC was associated with total incremental costs per patient of $7,671 and – $16,207 from community-based care and residential care, respectively, yielding a decrease in the total cost of care of $8,536 per patient. Lecanemab was associated with additional monitoring and ARIA-E management costs of $1,018 and $68, respectively. The model estimated that the annual societal value of lecanemab for the US payer perspective was $18,709–35,678 ($19,710–37,351 for societal perspective) for the WTP threshold of $100,000–200,000 per QALY gained, respectively (Table 3).
Scenario Analysis
Scenario analyses were conducted to explore the impact of alternative population subgroups, time horizons, input sources, treatment stopping rules, and treatment dosing on the model results. Incremental LYs, QALYs, total costs, and societal value estimates based on a WTP threshold of $200,000/QALY are presented in Table 4. In a subset of patients with MCI due to AD only, the model estimated higher incremental QALYs and lower incremental costs, resulting in a 2% (payer) and 3% (societal) increase in value compared to the base case. The estimated value in a subset of patients with mild AD dementia only decreased by 30%, and was associated with lower incremental QALYs compared to the base case. When comparing a younger population with a mean baseline age of 65 years, the model estimated additional incremental QALYs gained and a cost-saving over the lifetime yielding a higher value compared to the base-case scenario (i.e., payer, $45,763 vs. $35,678; societal, $48,472 vs. $37,351). The expected benefit associated with lecanemab was further observed in patients with MCI due to AD with a mean baseline age of 65 years (i.e., over 49% increase in total incremental QALYs yielding 30% increase in value for payer perspective and 48% increase in total incremental QALYs, yielding a 32% increase in value for societal perspective vs. base case, respectively). The scenario analysis also explored patient subsets stratified by the use of symptomatic drugs including acetylcholinesterase inhibitors (donepezil, rivastigmine, and galantamine) and memantine at baseline. Patients without symptomatic drug use at baseline had higher QALYs gained (payer, 0.68 vs. 0.61; societal, 0.71 vs. 0.64) compared to the overall population, yielding higher value estimates (payer, $37,069 vs. $35,678; societal, $39,345 vs. $37,351). When applying the treatment effect stratified by apolipoprotein E4 (APOE4) gene carrier status, the estimated value decreased by 6% for carriers and increased by 8% for non-carriers compared with the base-case. When patients were further divided into five quintiles based on their baseline CSF t-tau level, lecanemab produced better outcomes and a higher value in patients with lower t-tau at baseline. The value changed from 25% to − 19% and 29% to − 22% from the lowest to highest quintile of CSF t-tau versus the base case for the payer and societal perspectives, respectively.
Shorter time horizons resulted in lower incremental QALYs and less cost-savings, yielding lower predicted values compared to the lifetime results from both perspectives. Using lower mortality HRs derived from the Swedish dementia registry [24] resulted in 8% (payer) and 13% (societal) increases in value versus the base case. The model predicted lower incremental QALYs compared to the base case [41]. Results were slightly impacted by using a different risk of institutionalization and including screening costs.
The effect of treatment stopping rules and alternative treatment dosing were explored in different scenarios. In one scenario, treatment was stopped after 1.5 years with no residual benefit. Another scenario assumed the achieved amyloid reduction over a treatment duration of 1.5 years was maintained over a lifetime even after discontinuation. The model predicted lower QALYs gained and lower values in both cases. When patients were assumed to switch to a less frequent maintenance dosing schedule from biweekly dosing in the initial 1.5 years to once every 4 weeks in the maintenance phase assuming similar treatment effect to the base case, the estimated economic value increased by 44% for both payer and societal perspectives.
Sensitivity Analysis
A series of one-way sensitivity analyses were conducted to demonstrate the impact of varying the key parameters in the model. The results from the societal perspective at a WPT threshold of $200,000 are presented in a tornado diagram in Fig. 2. Varying the discount rate had the largest impact on the potential value of lecanemab. The predicted value decreased by 4.6% or increased by 9.8% when applying an annual discontinuation risk of 10% or 20%, respectively. Varying treatment effect as the percentage reduction in CDR-SB at 1.5 years by ± 15% of the base-case value of 27% changed the value by 5.8% and – 6.4%, respectively. The predicted value had a positive correlation with patient utilities for MCI due to AD and care costs for moderate and severe AD. Patient utility for moderate and severe AD, and care costs for MCI due to AD and mild AD were negatively associated with lecanemab value. Lower mortality HRs for severe AD resulted in higher estimated value for lecanemab. The remaining parameters tested resulted in a less than 1% change in value compared to the base case. The results of the one-way sensitivity analysis can be seen in Fig. 2.
Discussion
This modeling analysis used the AD ACE disease simulator to estimate the value of lecanemab plus SoC versus SoC alone for various WTP thresholds based on the phase III CLARITY AD trial readouts from both the US payer and societal perspectives. The flexible framework of AD ACE allowed further assessment on the influence of different key patient subsets, applying alternative treatment stopping rules and dosing regimens, as well as parameter uncertainty in this study.
The CLARITY AD trial provides compelling evidence of a clinical benefits of lecanemab. The treatment clears Aβ plaques, alters other disease biomarkers, and lessens clinical decline in individuals with early AD [7]. The observed treatment effect expanded with time on therapy, and indicated a disease-modifying effect. In this study, however, we assumed that the treatment effect would remain constant during the on-treatment period, in which patients were receiving lecanemab treatment, and continued at this same rate throughout the follow-up period. Treatment may be discontinued due to AEs or if the patient transitions to moderate AD dementia or a more severe stage. The aligns with the guidance and recommendations of the Alzheimer's Association working group, which consists of experienced and internationally recognized clinicians and researchers [44]. The group aimed to reassess the definition of a significant and meaningful benefit or slowing of ADs, while prioritizing the needs of patients and their families. According to the working group, if treatments are continued long-term—longer than the 18 months of a typical Phase III Alzheimer’s trial—and sustain their effectiveness at the same modest levels as in the clinical trial, they would be expected to show cumulative benefits that become larger, more readily apparent, and more meaningful over time. [44] Given that treatment effect expanded with time on therapy over the 18-month trial duration in the CLARITY AD, our modeling assumption may have been conservative.
In the base case, early AD stage patients with MCI due to AD or mild AD dementia had an additional 0.61 gain in QALYs (societal, 0.64) and $6,263 decrease in total non-treatment costs (societal, $7,451) when treated with lecanemab plus SoC versus SoC alone. For people treated with lecanemab, the total institutional care costs decreased by $12,439 (societal, $16,207) while the total community care costs increased by $5,091 (societal, $7,671) as treated patients spent more time in the community care over their lifetime. Compared to Study 201, fewer patients in the CLARITY AD trial discontinued treatment during the 18-month trial period, resulting in a longer mean time on treatment (3.91 years vs. 3.77 years in the previous analysis). At WTP threshold of $200,000 per QALY gained, the estimated annual value for lecanemab was $35,678 and $37,351 from the payer and societal perspectives, respectively. Considering the severity of ADs disease and its substantial burden, these findings provide strong evidence for the overall societal value of lecanemab.
The scenario analyses showed that lecanemab had a greater impact on total incremental costs and QALYs when initiated at earlier ages in patients with MCI due to AD dementia, and in patients earlier in their tau pathology (lower quintiles). The estimated QALYs gained ranged from 0.67 to 0.91 in the MCI due to AD subset versus 0.36 in the mild AD dementia subset, which was consistent with our previous findings based on Study 201 population characteristics and emphasizes the importance of early detection and treatment in AD [9]. A subset of patients with symptomatic drug use at baseline had lower QALYs gained yielding a lower value estimate compared to the overall patient population. APOE4 is a strong risk factor gene for developing AD, and carriers of APOE4 alleles were shown to have a higher risk and earlier onset of AD than non-carriers [45]. In the scenario where stratified treatment effect by APOE4 gene carrier status was explored, the predicted value was 6% lower for carriers and 8% higher for non-carriers compared to the base-case estimates.
Scenario analyses of treatment stopping rules and treatment dosing were conducted to explore the impact of alternative assumptions on the model results. The study findings indicated that a shorter time on treatment resulted in lower QALYs gained and less cost savings. A less frequent dosing regimen in the maintenance phase while assuming the same treatment effect as the base case increased the value by 44%.
A main update of this study compared to the previous preliminary assessment was that it used the recently published data from the large phase III CLARITY AD trial to assess the cost-effectiveness of lecanemab. To align with the primary outcome of the trial, a calibration process regarding the treatment effect on amyloid levels was carefully conducted (illustrated in Fig. 1) until the treatment effect observed in the model matched with the target values from the CLARITY AD study. This process continued beyond the time horizon of the CLARITY AD trial by using another study exploring the effect of continued treatment of lecanemab [29].
This study also has some limitations. Firstly, in this study, the amyloid PET level serves as a surrogate endpoint assumed to mediate or predict the effects of lecanemab on the key trial outcomes. If this conditional independence assumption is unwarranted, the mean cost and QALY estimates may be biased, and uncertainty may be underestimated [46]. The use of unsuitable surrogate endpoints and lack of accounting for uncertainty could impact future research efforts. However, lecanemab was shown to reduce brain amyloid levels and was associated with less decline on clinical measures of cognition and function than placebo in the large phase III CLARITY AD confirmatory trial. Next, while a strength of this model is the use of relevant studies to obtain data regarding key parameters, such as the mortality risk, costs, utility, and risk of institutionalization, uncertainty continues to exist in these parameters. Scenario and sensitivity analyses in our study aimed at reducing prediction uncertainty on model outputs. Furthermore, while the indirect treatment effects were assessed using CDR-SB, the behavior, dependence, function, and other domains were not examined. Moreover, disease severity, which was used to model patient utilities and the institutionalization risk, was estimated based on a single—but validated and widely used—measure (CDR-SB). The key assumptions underlying the model require further validation based on data derived from clinical trials with long-term follow-up periods and longitudinal real-world evidence. Such evidence will inevitably emerge following the market authorization of lecanemab, enabling the collection of real-world data. Finally, while studies of disease progression using ADNI have highlighted the pivotal role of regional Aβ and tau deposition in AD and have identified genetic factors underlying the disease, the restrict inclusion and exclusion criteria and lack of diversity in ethnocultural cohorts may have limited external validity [47]. Altogether, in this study, lecanemab exhibited the ability to delay AD onset, thereby increasing the number of disease-free years and reducing care-related costs, significantly benefiting not only patients and their caregivers but also society overall.
Conclusion
The economic analysis suggested that lecanemab plus SoC compared with SoC alone could lead to higher health and humanistic (quality of life) outcomes and lower economic burden for patients and caregivers in early AD. The findings of this study demonstrate the potential economic and societal value of lecanemab and can be used as a foundation for healthcare decision-making and health policy.
References
Huang LK, Chao SP, Hu CJ. Clinical trials of new drugs for Alzheimer disease. J Biomed Sci. 2020;27(1):18.
The Alzheimer’s Association. 2022 Alzheimer’s disease facts and figures. Alzheimers Dement. 2022;18(4):700–89.
Mank A, van Maurik IS, Rijnhart JJM, Bakker ED, Bouteloup V, Le Scouarnec L, et al. Development of multivariable prediction models for institutionalization and mortality in the full spectrum of Alzheimer’s disease. Alzheimers Res Ther. 2022;14(1):110.
Frisoni G. Structural imaging in the clinical diagnosis of Alzheimer's disease: problems and tools. BMJ Publishing Group Ltd; 2001;711–8.
Cummings J, Fox N. Defining disease modifying therapy for Alzheimer’s disease. J Prev Alzheimers Dis. 2017;4(2):109–15.
Cummings J, Lee G, Zhong K, Fonseca J, Taghva K. Alzheimer’s disease drug development pipeline: 2021. Alzheimers Dement (NY). 2021;7(1): e12179.
van Dyck CH, Swanson CJ, Aisen P, Bateman RJ, Chen C, Gee M, et al. Lecanemab in early Alzheimer’s disease. N Engl J Med. 2023;388(1):9–21.
Tahami Monfared AA, Tafazzoli A, Ye W, Chavan A, Zhang Q. Long-term health outcomes of lecanemab in patients with early Alzheimer’s disease using simulation modeling. Neurol Therapy. 2022;11(2):863–80.
Tahami Monfared AA, Tafazzoli A, Chavan A, Ye W, Zhang Q. The potential economic value of lecanemab in patients with early Alzheimer’s disease using simulation modeling. Neurol Therapy. 2022;11(3):1285–307.
Swanson CJ, Zhang Y, Dhadda S, Wang J, Kaplow J, Lai RYK, et al. A randomized, double-blind, phase 2b proof-of-concept clinical trial in early Alzheimer’s disease with lecanemab, an anti-Aβ protofibril antibody. Alzheimers Res Ther. 2021;13(1):80.
Lakdawalla DN, Phelps CE. Health technology assessment with risk aversion in health. J Health Econ. 2020;72: 102346.
Kansal AR, Tafazzoli A, Ishak KJ, Krotneva S. Alzheimer’s disease Archimedes condition-event simulator: development and validation. Alzheimers Dement (N Y). 2018;4:76–88.
Tafazzoli A, Weng J, Sutton K. Validating simulated cognition trajectories based on ADNI against trajectories from the National Alzheimer's Coordinating Center (NACC) dataset of Clinical Trials on Alzheimer's Disease (CTAD). 2018.
Hernandez L, Ozen A, DosSantos R, Getsios D. Systematic review of model-based economic evaluations of treatments for Alzheimer’s disease. Pharmacoeconomics. 2016;34(7):681–707.
Caro JJ, Briggs AH, Siebert U, Kuntz KM. Modeling good research practices—overview: a report of the ISPOR-SMDM modeling good research practices task force–1. Med Decis Making. 2012;32(5):667–77.
Alzheimer's disease neuroimaging initiative [Internet]. 2017. Available from: https://adni.loni.usc.edu/.
Getsios D, Blume S, Ishak KJ, Maclaine GD. Cost effectiveness of donepezil in the treatment of mild to moderate Alzheimer’s disease. Pharmacoeconomics. 2010;28(5):411–27.
Guo S, Getsios D, Revankar N, Xu P, Thompson G, Bobula J, et al. Evaluating disease-modifying agents: a simulation framework for Alzheimer’s disease. Pharmacoeconomics. 2014;32(11):1129–39.
Small GW, McDonnell DD, Brooks RL, Papadopoulos G. The impact of symptom severity on the cost of Alzheimer’s disease. J Am Geriatr Soc. 2002;50(2):321–7.
Sanders GD, Neumann PJ, Basu A, Brock DW, Feeny D, Krahn M, et al. Recommendations for conduct, methodological practices, and reporting of cost-effectiveness analyses: second panel on cost-effectiveness in health and medicine. JAMA. 2016;316(10):1093–103.
O’Bryant SE, Waring SC, Cullum CM, Hall J, Lacritz L, Massman PJ, et al. Staging dementia using clinical dementia rating scale sum of boxes scores: a Texas Alzheimer’s research consortium study. Arch Neurol. 2008;65(8):1091–5.
Arias E, Xu J, Kochanek KD. United States life tables, 2016. 2019.
Andersen K, Lolk A, Martinussen T, Kragh-Sørensen P. Very mild to severe dementia and mortality: a 14-year follow-up–the Odense study. Dement Geriatr Cogn Disord. 2010;29(1):61–7.
Wimo A, Handels R, Winblad B, Black CM, Johansson G, Salomonsson S, et al. Quantifying and describing the natural history and costs of Alzheimer’s disease and effects of hypothetical interventions. J Alzheimers Dis. 2020;75(3):891–902.
Neumann PJ, Hermann R, Kuntz K, Araki S, Duff S, Leon J, et al. Cost-effectiveness of donepezil in the treatment of mild or moderate Alzheimer’s disease. Neurology. 1999;52(6):1138.
Davis M, O’Connell T, Johnson S, Cline S, Merikle E, Martenyi F, et al. Estimating Alzheimer’s disease progression rates from normal cognition through mild cognitive impairment and stages of dementia. Curr Alzheimer Res. 2018;15(8):777–88.
Avgerinos KI, Ferrucci L, Kapogiannis D. Effects of monoclonal antibodies against amyloid-β on clinical and biomarker outcomes and adverse event risks: a systematic review and meta-analysis of phase III RCTs in Alzheimer’s disease. Ageing Res Rev. 2021;68: 101339.
Fletcher E, Filshtein TJ, Harvey D, Renaud A, Mungas D, DeCarli C, et al. Staging of amyloid β, t-tau, regional atrophy rates, and cognitive change in a nondemented cohort: results of serial mediation analyses. Alzheimer’s Demen Diagnos Assess Dis Monit. 2018;10:382–93.
McDade E, Cummings JL, Dhadda S, Swanson CJ, Reyderman L, Kanekiyo M, et al. Lecanemab in patients with early Alzheimer’s disease: detailed results on biomarker, cognitive, and clinical effects from the randomized and open-label extension of the phase 2 proof-of-concept study. Alzheimers Res Ther. 2022;14(1):191.
Dhadda S, Kanekiyo M, Li D, Swanson CJ, Irizarry M, Berry S, et al. Consistency of efficacy results across various clinical measures and statistical methods in the lecanemab phase 2 trial of early Alzheimer’s disease. Alzheimers Res Ther. 2022;14(1):182.
Robinson RL, Rentz DM, Andrews JS, Zagar A, Kim Y, Bruemmer V, et al. Costs of early stage Alzheimer’s disease in the United States: cross-sectional analysis of a prospective cohort study (GERAS-US). J Alzheimers Dis. 2020;75(2):437–50.
Wimo A, Reed CC, Dodel R, Belger M, Jones RW, Happich M, et al. The GERAS study: a prospective observational study of costs and resource use in community dwellers with Alzheimer’s disease in three European countries–study design and baseline findings. J Alzheimers Dis. 2013;36(2):385–99.
Leon J, Cheng C-K, Neumann PJ. Alzheimer’s disease care: costs and potential savings: caring for persons with Alzheimer’s disease in the community can save thousands of dollars, but at what cost to family caregivers? Health Aff. 1998;17(6):206–16.
The Alzheimer's Association. Changing the trajectory of Alzheimer’s disease: how a treatment by 2025 saves lives and dollars. Appendix A: Methodology. 2015.
Cost of Care Survey 2022 [Internet]. Available from: https://www.genworth.com/aging-and-you/finances/cost-of-care.html.
DiBenedetti DB, Slota C, Wronski SL, Vradenburg G, Comer M, Callahan LF, et al. Assessing what matters most to patients with or at risk for Alzheimer’s and care partners: a qualitative study evaluating symptoms, impacts, and outcomes. Alzheimer’s Res Ther. 2020;12(1):1–15.
Medicare Physician Fee Schedule. CY 2022 National Payment Amount by HCPCS Code 2022. [Internet]. 2022 [cited Apr 14, 2022]. Available from: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/PhysicianFeeSched.
IBM Micromedex RED BOOK [Internet]. [cited Apr 14, 2022]. Available from: https://www.ibm.com/products/micromedex-red-book.
Consumer price index [Internet]. [cited Apr 14, 2022]. Available from: https://www.bls.gov/cpi/.
Landeiro F, Mughal S, Walsh K, Nye E, Morton J, Williams H, et al. Health-related quality of life in people with predementia Alzheimer’s disease, mild cognitive impairment or dementia measured with preference-based instruments: a systematic literature review. Alzheimer’s Res Ther. 2020;12(1):1–14.
Neumann PJ, Kuntz KM, Leon J, Araki SS, Hermann RC, Hsu M-A, et al. Health utilities in Alzheimer's disease: a cross-sectional study of patients and caregivers. Medical care. 1999:27–32.
Xu R, Insinga RP, Golden W, Hu XH. EuroQol (EQ-5D) health utility scores for patients with migraine. Qual Life Res. 2011;20(4):601–8.
Mesterton J, Wimo A, Langworth S, Winblad B, Jonsson L. Cross sectional observational study on the societal costs of Alzheimer’s disease. Curr Alzheimer Res. 2010;7(4):358–67.
Petersen RC, Aisen PS, Andrews JS, Atri A, Matthews BR, Rentz DM, et al. Expectations and clinical meaningfulness of randomized controlled trials. Alzheimers Dement. 2023.
Monsell SE, Kukull WA, Roher AE, Maarouf CL, Serrano G, Beach TG, et al. Characterizing apolipoprotein E epsilon4 carriers and noncarriers with the clinical diagnosis of mild to moderate alzheimer dementia and minimal beta-amyloid peptide plaques. JAMA Neurol. 2015;72(10):1124–31.
Hawkins N, Richardson G, Sutton AJ, Cooper NJ, Griffiths C, Rogers A, et al. Surrogates, meta-analysis and cost-effectiveness modelling: a combined analytic approach. Health Econ. 2012;21(6):742–56.
Veitch DP, Weiner MW, Aisen PS, Beckett LA, DeCarli C, Green RC, et al. Using the Alzheimer’s disease neuroimaging initiative to improve early detection, diagnosis, and treatment of Alzheimer’s disease. Alzheimers Dement. 2022;18(4):824–57.
Acknowledgements
We thank the trial participants and their families for their generous contributions to this study. We could not have done this research without their invaluable support.
Funding
This study and the journal’s Rapid Service Fee was funded by Eisai Inc.
Medical Writing, Editorial, and Other Assistance
The authors would like to acknowledge Evidera’s medical writer, Ruth Sharf-Williams, and editor, Alison Danson, for proofreading and editorial services funded by Eisai Inc.
Author Contributions
Amir Abbas Tahami Monfared and Quanwu Zhang contributed to the initial study concept and were responsible for the overall study direction and planning. Henri Folse, Ameya Chavan and Aditya Sardesai contributed to the study conception and design. Model development and analysis were performed by Weicheng Ye and Kang Kang. All authors helped draft or critically revise the manuscript. All authors read and approved the final manuscript.
Disclosures
Amir Abbas Tahami Monfared and Quanwu Zhang are employees of Eisai Inc. Henri Folse, Ameya Chavan, Aditya Sardesai, Weicheng Ye, and Kang Kang are current employees of Evidera, a healthcare research firm that provides consulting and other research services to pharmaceutical, device, government, and non-government organizations. Evidera received funding from Eisai Inc. to conduct the study and develop this manuscript.
Compliance with Ethics Guidelines
The CLARITY AD trial (ClinicalTrials.gov identifier, NCT03887455) was conducted in accordance with the Declaration of Helsinki and the International Council for Harmonization and Good Clinical Practice guidelines and was approved by the institutional review board or independent ethics committee at each center. All patients provided written informed consent. An independent interim monitoring committee was responsible for oversight and conduct of the interim analyses and response adaptive randomization design to evaluate the safety routinely and reviewing futility analysis results. This assessment is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Data Availability
All data used for this study are provided in the manuscript. Additional details are available from the corresponding author on request.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Tahami Monfared, A.A., Ye, W., Sardesai, A. et al. Estimated Societal Value of Lecanemab in Patients with Early Alzheimer’s Disease Using Simulation Modeling. Neurol Ther 12, 795–814 (2023). https://doi.org/10.1007/s40120-023-00460-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40120-023-00460-1