Abstract
Introduction
Bloodstream infection (BSI) may occur after cardiac procedures, but this has rarely been investigated specifically in pediatric patients after percutaneous or surgical treatment for ventricular septal defect (VSD) or atrial septal defect (ASD) with recent data. The current study aimed to investigate the incidence, clinical features, and association with prognosis of BSI in this patient population.
Methods
Pediatric patients who received percutaneous or surgical procedure for VSD or ASD between 2010 and 2018 in a large children’s hospital in China were retrospectively enrolled via the Pediatric Intensive Care database, but only those who had blood culture records within 24 h after the procedure and who had no prior positive blood culture records were included. BSI after the procedure was identified by reviewing blood culture records, and baseline characteristics associated with BSI were explored by univariable logistic regression. In-hospital mortality and length of hospitalization were studied as prognostic outcomes and compared between patients with and without BSI.
Results
A total of 1340 pediatric patients were included. Among them, 46 (3.43%) patients had BSI within 24 h after the procedure, of which the majority (78.26%, 36/46) were caused by Gram-positive bacteria and 65.22% (30/46) had antibiotic-resistant organisms. Age [odds ratio (OR) 0.98 per 1-month increase, 95% confidence interval (CI) 0.97–1.00, P = 0.021] and antibiotic use within 72 h before the procedure (OR 1.81, 95% CI 1.00–3.26, P = 0.049) were statistically significantly associated with developing BSI. Compared with patients without BSI, there was no statistically significant difference in in-hospital mortality (0.00% versus 0.54%, P = 1.000), but patients with BSI had statistically significantly longer length of hospitalization (median 14.51 versus 12.94 days, P = 0.006), while the association was not statistically significant after adjustment for baseline characteristics by multivariable linear regression (β = 1.73, 95% CI −0.59 to 4.04, P = 0.144).
Conclusion
BSI is relatively uncommon in pediatric patients after procedures for VSD or ASD, but a younger age seems a risk factor. Developing BSI appears to be associated with increased length of hospitalization but not in-hospital mortality.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Bloodstream infection (BSI) may occur after cardiac procedures, but knowledge is limited about the occurrence of BSI after procedures for ventricular/atrial septal defect (VSD/ASD). |
The current study aimed to investigate the incidence, clinical features, and association with prognosis of BSI in pediatric patients after percutaneous or surgical treatment for VSD/ASD. |
What was learned from the study? |
BSI is relatively uncommon (about 3.5%) in pediatric patients after procedures for VSD or ASD, but a younger age seems to be a risk factor. |
Developing BSI appears to be associated with an increased length of hospitalization but not in-hospital mortality. |
Introduction
Bloodstream infection (BSI) is generally defined by the presence of viable bacterial or fungal microorganisms in the bloodstream confirmed by blood cultures, which include a wide variety of pathogens and clinical syndromes [1]. Although epidemiology of BSI from well-designed population-based surveillance is lacking, especially at a regional level and in resource-limited settings [2], the incidence of BSI is estimated to be similar to stroke or venous thrombosis (about 100–200 per 100,000 population) in the general population [3, 4], of which nosocomial BSI accounts for about 30–50% [5, 6]. This has become a heavy burden for healthcare, as the presence of BSI is often associated with both worse prognosis (including prolonged lengths of hospital stay and increased mortality risk) and increased cost, especially for critically ill patients [7,8,9].
Receiving invasive procedures is a well-established risk factor of nosocomial BSI, and therefore antimicrobial prophylaxis before or after a procedure is indicated in some situations [10,11,12,13]. However, there are also concerns about the inappropriateness of surgical antimicrobial prophylaxis [14,15,16], which may lead to unnecessary cost and contribute to the development and spread of antimicrobial resistance [17,18,19]. For cardiac surgery, routine antibiotic prophylaxis is generally recommended given the significant and large benefit observed in trials, although detailed recommendations on duration and timing varies between guidelines [20]; while for interventional cardiac catheterization, antibiotic prophylaxis is only recommended for patients at high risk of infective endocarditis [21, 22].
Cardiac surgery is one invasive procedure commonly received by pediatric patients, especially those with congenital cardiac diseases. A replacement for cardiac surgery, cardiac catheterization has gradually become a routine tool for diagnosis and intervention of congenital cardiac disease in childhood. Compared with research that focused on adult patients, evidence about BSI occurrence is rather limited for pediatric patients receiving cardiac surgery. Although BSI (especially infective endocarditis) seems to be a reasonable concern for cardiac catheterization, infection seems rare [23, 24]. However, since practices, patient profiles, and prevalent spectrum of pathogenic microorganisms may change over time or vary between regions, further investigations on this topic with recent data are warranted. Therefore, via a pediatric-specific database with recent (2010–2018) data from a large pediatric medical center in China, we conducted a study aiming to investigate the incidence, clinical features, and association with prognosis of BSI in pediatric patients after percutaneous or surgical treatment for ventricular septal defect (VSD) or atrial septal defect (ASD).
Methods
Data Source
The current study used data from the Pediatric Intensive Care (PIC) database (version 1.1.0) [25]. PIC is a pediatric-specific intensive care database, which contains information about children (aged 0–18 years) admitted to the critical care units between 2010 and 2018 in a comprehensive pediatric medical center (i.e., The Children’s Hospital, Zhejiang University School of Medicine) in China. Data used in the current study include medical records at hospital and intensive care unit (ICU) level, including information about operations, microbiological examination (including tests performed and sensitivities), medication prescription, and diagnoses made during the hospitalization.
The original project that developed the PIC database was approved by the Institutional Review Board (IRB) of the Children’s Hospital, Zhejiang University School of Medicine, and the requirement for individual patient consent was waived because the project did not impact clinical care, and all protected health information was deidentified [25].
After following the required procedures, our access to the PIC database was approved by the administrator of the PIC database on 19 August 2021. The current study was further approved by the IRB of the Guangdong Provincial People’s Hospital (no. KY-Q-2021-272-01). All the data used were deidentified and patient consent was waived. We complied with the Helsinki Declaration 1964 and its later amendments.
Study Population
With the PIC database, we screened the data for pediatric patients who received percutaneous or surgical intervention for VSD or ASD between 2010 and 2018, but only those who had blood culture records within 24 h after the procedure and who had no prior positive blood culture records (since hospital admission) were included. In detail, we first examined the information about operations (from operation rooms) and identify patients who had operation records of VSD or ASD (either closure or repair). The identification was based on free-text using “VSD” and “ASD” as the keywords. As the PIC database is an English–Chinese bilingual database, we also examined the original records in Chinese to further ensure the identified patients received percutaneous or surgical procedure for VSD or ASD during the hospitalizations. Patients who had more than one procedure record during the same hospitalizations were excluded, but those who received other procedure(s) together with the same procedure for VSD or ASD were included. We then examined the information about microbiological examination to identify and include patients who had blood culture records within 24 h after the procedure (according to the end time of the procedure). To exclude patients who had BSI before the procedure, we excluded patients who had blood culture records with positive results before the procedure.
BSI after Percutaneous or Surgical Procedure for VSD or ASD
BSI after the procedure was identified by reviewing the results of the blood culture records (within 24 h after the procedure). For positive results, details on types of organisms and results of antibiotic resistance test were collected. To better understand the cause of BSI, the types of organisms were categorized as Gram-positive cocci, Gram-positive bacilli, Gram-negative cocci, Gram-negative bacilli, fungus, and others.
Baseline Characteristics (Clinical Features)
The below information was collected from the relevant data and identified the following baseline characteristics: age, sex, ethnicity, (maximum) body temperature, (maximum) respiratory rate, (minimum) oxygen saturation, and antibiotic use within 72 h before the procedure, type of septal defect (ASD or VSD), guided by transesophageal echocardiography (or not), other procedure(s) received during the same procedure, duration of anesthesia, duration of the procedure, method of anesthesia, position for the procedure. We also collected all the diagnoses made during the hospitalizations of the included patients, in which data on diagnoses and procedures (mainly to identify specific types of congenital cardiac disease) were both used for the identification.
Prognostic Outcomes
We studied in-hospital mortality and length of hospitalization as prognostic outcomes, both of which were directly obtained from the hospitalization data.
Statistical Analysis
Summary statistics were presented as mean ± standard deviation or median (25–75% percentiles) for continuous variables according to whether the variables were normally distributed, and frequency (percentage) for categorical variables. Comparisons between two groups were examined by t-test or the Mann–Whitney U-test for continuous variables, and by the Chi-squared test or the Fisher's exact test for categorical variables (and for comparisons between three groups). The associations of each baseline characteristic with BSI after the procedure were examined by univariable logistic regression. The studied prognostic outcomes were compared between patients with and without BSI after the procedure, which was further evaluated by multivariable logistic regression (for in-hospital mortality) or linear regression (length of hospitalization). A P-value <0.05 was considered to indicate statistical significance. IBM SPSS Statistics for Windows (version 25.0. Armonk, NY: IBM Corp.) was used for the statistical analyses.
Results
Baseline Characteristics of the Study Population
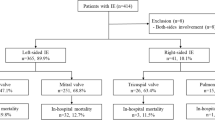
A total of 1340 pediatric patients who received percutaneous or surgical treatment for septal defect were included (Fig. 1). As presented in Table 1, the median age of the included patients were 15.51 (6.16–37.34) months, and 636 (47.46%) were male. The majority of the patients were Han (ethnicity, 99.48%), and were first admitted to the cardiac surgery department (99.03%, not shown in the table). About 38% (511/1340) of the patients received antibiotics within 72 h before the procedure, and the majority of them received only one type of antibiotics (95.85%, 490/511), of which cefradine was the most frequently prescribed (Supplementary Table 1).
According to the types of procedures the patients received, 691 (51.57%) patients received interventions for VSD, 463 (34.55%) for ASD, and 186 (13.88%) for both VDS and ASD, and only about 3% (39/1340) were guided by transesophageal echocardiography. The mean duration of anesthesia and the procedure were 173.20 ± 55.97 min and 116.77 ± 49.40 min, respectively, and about 20% (277/1340) received other procedures at the same time, in which closure of patent ductus arteriosus was the most commonly performed procedure (Supplementary Table 2). In terms of comorbidities, apart from VSD or ASD, patent ductus arteriosus (n = 132), tricuspid valve disease (n = 89), and patent foramen ovale (n = 63) were the most frequent comorbidities among the included patients (Supplementary Table 3).
Incidence and Clinical Features of BSI
The proportion of developing BSI within 24 h after the procedure was 3.43% (46/1340), and 2.24% (30/1340) for BSI with antibiotic-resistant organism. As presented in Table 2, among those who developed BSI, the majority (93.48%, 43/46) was caused by bacteria, of which Gram-positive bacteria was the most common (n = 36). Only one patient had multiple-organism infection (i.e., Streptococcus oralis and Neisseria flavescens), and Staphylococcus epidermidis (n = 16) and Staphylococcus hominis (n = 10) were the two most common organisms (Supplementary Table 4). There was no statistically significant difference in the types of organisms between septal defects (P all > 0.05, Supplementary Table 5). According to the results of the antibiotic sensitivity test (Supplementary Table 6), resistance to methicillin was rather common (25/27, 92.59%), followed by resistance to oxacillin (25/27, 92.59%), penicillin G (27/31, 87.10%), erythromycin (20/32, 62.50%), and sulfamethoxazole (13/32, 40.62%).
Baseline Characteristics Associated with BSI
Among the studied baseline characteristics, age [odds ratio (OR) 0.98 per 1-month increase, 95% confidence interval (CI) 0.97–1.00, P = 0.021], (maximum) respiratory rate within 72 h before the procedure (OR 1.03, 95% CI 1.00–1.06, P = 0.038), antibiotic use within 72 h before the procedure (OR 1.81, 95% CI 1.00–3.26, P = 0.049), and repair of total anomalous pulmonary venous connection (OR 8.36, 95% CI 1.69–41.39, P = 0.009) were found to be statistically significantly associated with developing BSI, while no statistically significant associations with other characteristics were observed (Table 3 and Supplementary Table 7). For BSI antibiotic-resistant organisms, age (OR 0.97 per 1-month increase, 95% CI 0.95–1.00, P = 0.021), (maximum) respiratory rate within 72 h before the procedure (OR 1.04, 95% CI 1.00–1.08, P = 0.045), patent foramen ovale repair (OR 3.26, 95% CI 1.10–9.65, P = 0.033), and comorbid other heart diseases (namely other heart diseases except for those presented in Supplementary Table 7 in detail, OR 3.93, 95% CI 1.14–13.56, P = 0.030) were potential predictors (Table 3 and Supplementary Table 7).
Association of BSI with Prognosis
The overall in-hospital mortality of the included patients was 0.52% (7/1340) with a median length of hospitalization of 12.97 (9.07–17.21) days. Compared with patients without BSI, there was no statistically significant difference in in-hospital mortality (0.00% versus 0.54%, P = 1.000), but patients with BSI showed statistically significantly longer lengths of hospitalization (median 14.51 versus 12.94 days, P = 0.006). A similar difference was observed between patients with BSI with antibiotic-resistant organism and those without (Table 4).
Results of the univariable linear regression indicated BSI was a risk factor of longer length hospitalization (β 2.71, 95% CI 0.20–5.21, P = 0.034). After adjusting for age, sex, type of septal defect, antibiotic use within 72 h before the procedure, and presence of other heart defects by multivariable linear regression, the association became nonsignificant (β 1.66, 95% CI −0.66 to 3.97, P = 0.160). For BSI with antibiotic-resistant organism, however, the associations estimated from either univariable or multivariable linear regression were nonsignificant (Table 5).
Discussion
The current study included a large cohort of pediatric patients who received percutaneous or surgical procedure for VSD or ASD, and investigated the occurrence of BSI immediately after the procedure, focusing on the incidence, clinical features, and association with prognosis of BSI in this pediatric patient population. The main findings of our study are as follows: (1) the incidence of BSI was about 3.5% in the study population we investigated; (2) Gram-positive cocci (mainly Staphylococcus epidermidis and Staphylococcus hominis) was the most prevalent spectrum of identified microorganisms, and antibiotic resistance was rather common; (3) some patient characteristics might serve as predictors of developing BSI after the procedure, including a younger age, increased respiratory rate, and prior antibiotic use (which may be better interpreted as potential concurrent active infection); (4) developing BSI after the procedure was not a significant risk factor for increased in-hospital mortality, but it appeared to be associated with a longer length of hospitalization.
These findings provide new epidemiological knowledge about the occurrence of BSI after percutaneous or surgical procedure for VSD or ASD in pediatric patients, and may also provide insights into antibiotic prophylaxis. Given that the risk of developing BSI after these procedures is relatively uncommon, and developing BSI does not seem to be associated with increased in-hospital mortality, our findings support the current guideline recommendation that antibiotic prophylaxis should only be considered for high-risk patients [21, 22]. We found that BSI appeared to be more prevalent in patients with some characteristics, such as a young age, increased respiratory rate, potential concurrent active infection (indicated for antibiotic use prior to the procedure), receiving repair of total anomalous pulmonary venous connection, or patent foramen ovale. Although these findings are only based on univariable regression analysis, which should be interpreted cautiously, they may serve as potential predictors to identify pediatric patients who are likely to develop BSI after the procedure and, accordingly, antibiotic prophylaxis may be considered, as we found developing BSI appeared to be associated with increased length of hospitalization. The findings on microorganisms and antibiotic resistance may also provide clues to choices of antibiotic, but generalizability should be taken into account since our findings were only based on data from a single center.
Research specifically investigating BSI in pediatric patients who received percutaneous or surgical procedures for VSD or ASD is rather limited, but there are some studies focused on infection in a similar patient population. Tweddell et al. [26] used a pediatric inpatient care database in the USA including a large cohort of pediatric patients who underwent cardiac surgery from 1997 to 2012, and found 1.6% of the patients had health care-associated infections, which was associated with younger age, heart failure, and acute kidney injury. Similarly, they found developing health care-associated infections was only independently associated with increased length of hospital stay and total charges for the hospital stay, but not with inpatient mortality. These results are consistent with ours, but it should be noted that, in addition to the different study periods (compared with ours), they identified health care-associated infections by diagnosis codes, which were not only limited to BSI, while in our study BSI was identified according to results of blood culture, which may be seen as a strength of our study. Elella et al. [27] reported a much higher incidence of BSI (8.6%) and mortality rate (11% in patients with BSI versus 2% in those without BSI) in pediatric patients who received cardiac surgery and were admitted to a pediatric cardiac intensive care unit (ICU) in 2007 from Saudi Arabia. This difference may be because the patients they included were at higher risk of developing BSI, as they were all percutaneously placed central lines and admitted to ICU. Another explanation may be the wide variety of cardiothoracic surgical procedures between studies, which was also found to be a risk factor (i.e., high surgical complexity) for BSI [27]. In our study, we also observed that receiving other procedures at the same time appears to increase risk of BSI, although the association was not statistically significant.
In terms of the spectrum of microbial causes, we found Gram-positive cocci (mainly Staphylococcus epidermidis and Staphylococcus hominis) was the most prevalent, which was consistent with most available investigations [28,29,30], although the study populations were not exactly the same. However, in the study by Elella et al. [27], Gram-negative organisms were the main causative organisms for BSI, which was different from our findings. A possible explanation is in the study by Elella et al. [27], all the patients received intravenous cefazolin before and after the cardiac surgery. This is similar to the findings of the study by Levy et al. [31], which investigated nosocomial infections after cardiac surgery in pediatric patients admitted to a pediatric cardiothoracic intensive care unit in Israel in 1999 and found Gram-negative bacilli (i.e., Klebsiella spp., Enterobacter spp., and Pseudomonas spp.) accounted for the majority of the causative organisms. In addition, the difference could also be related to different epidemiology of microorganisms among regions [32] and study periods. Nevertheless, strategies for antibiotic prophylaxis must consider local epidemiology and resistance patterns of organisms [33]. It should be noted that among the BSI we identified, about 70% were due to coagulase-negative staphylococci, which are also common contaminants in clinical specimens. Owing to limited clinical information in the database, we were unable to further distinguish a bacteremia from contamination, and therefore are at risk of overestimating the incidence of BSI. However, according to a sensitivity analysis we performed (which was not reported), the patients with coagulase-negative staphylococci BSI showed a similar length of hospitalization to those with non-coagulase-negative staphylococci BSI (median 14.58 and 14.45 days, respectively, versus 12.94 days for those without BSI). This suggests that at least not all the coagulase-negative staphylococci BSI we identified are contaminants. Furthermore, a potentially overestimated BSI incidence would not overturn our main finding that BSI is relatively uncommon in pediatric patients after procedures for VSD or ASD, which, therefore, relieves this concern to some extent (i.e., misclassification of BSI).
Some limitations should be noted in our study. First, the data was from a single center, which limits the generalizability of our findings. Second, the sample size may be not enough to evaluate the associations between BSI and prognostic outcomes, given the absolute risk of developing BSI or in-hospital mortality in the study population was relatively low. We did not observe an association between developing BSI and increased in-hospital mortality, but this should not be directly interpreted as developing BSI would not increase in-hospital mortality, as absence of evidence is not evidence of absence. Third, due to data limitation, the specific procedure types for VSD/ASD were uncertain, and were identified only by the recorded names of the procedures. In the study, we identified percutaneous or surgical treatment simply according to the presence of “closure” or “repair” (for transcatheter closure and surgical repair respectively, Supplementary Table 2). This is obviously a risk for misclassification, and for this reason, we did not perform subgroup analysis according to the type of procedure. Fourth, only patients who had records of blood culture within 24 h after the procedure were included, which might introduce selection bias. However, as the majority (1342/1569, about 85%) of patients who received percutaneous or surgical treatment for VSD or ASD had available postoperative blood culture records, this may not be a concern. Fifth, the database only provided limited clinical information, which hinders a clear interpretation of some of our findings. For example, we found about 40% patients received antibiotics within 72 h before the procedure, but detailed indications for prescribing antibiotics are unknown. This increased the concern about misclassification of BSI. In the investigation about baseline characteristics associated with BSI after the procedure, we found the patients with prior antibiotic use actually had a higher risk of BSI after the procedure than those without prior antibiotic use, suggesting that antibiotics might likely have been prescribed for an active infection. Other information such as hemodynamic/respiratory support or arterial lines was unavailable, raising concern about unmeasured confounding. Last, we only studied BSI within 24 h after the procedure, whether BSI developed at other time points was not studied. Owing to a lack of granular clinical data, we did not further investigate treatment the patients received after the procedure. Together with the above-mentioned limitations, our findings on the association between BSI and prognosis should be interpreted with caution. It might be more appropriate to interpret developing BSI as a potential predictor of longer hospital stay, instead of a causal interpretation. In addition, we only investigated two prognostic outcomes (in-hospital mortality and length of hospitalization), but other clinically relevant outcomes especially about the procedure itself were not studied due to lack of data. Further studies should address these limitations to confirm our findings.
Conclusions
BSI is relatively uncommon in pediatric patients after procedures for VSD or ASD, but a younger age seems to be a risk factor. Developing BSI appears to be associated with an increased length of hospitalization but not in-hospital mortality.
References
Viscoli C. Bloodstream infections: the peak of the iceberg. Virulence. 2016;7:248–51.
Kern WV, Rieg S. Burden of bacterial bloodstream infection-a brief update on epidemiology and significance of multidrug-resistant pathogens. Clin Microbiol Infect. 2020;26:151–7.
Scheres LJJ, Lijfering WM, Cannegieter SC. Current and future burden of venous thrombosis: not simply predictable. Res Pract Thromb Haemost. 2018;2:199–208.
Thrift AG, Thayabaranathan T, Howard G, et al. Global stroke statistics. Int J Stroke. 2017;12:13–32.
Laupland KB, Church DL. Population-based epidemiology and microbiology of community-onset bloodstream infections. Clin Microbiol Rev. 2014;27:647–64.
Goto M, Al-Hasan MN. Overall burden of bloodstream infection and nosocomial bloodstream infection in North America and Europe. Clin Microbiol Infect. 2013;19:501–9.
Harbarth S, Ferriere K, Hugonnet S, Ricou B, Suter P, Pittet D. Epidemiology and prognostic determinants of bloodstream infections in surgical intensive care. Arch Surg. 2002;137:1353–9 (discussion 1359).
Kallel H, Houcke S, Resiere D, et al. Epidemiology and prognosis of intensive care unit-acquired bloodstream infection. Am J Trop Med Hyg. 2020;103:508–14.
Jin L, Zhao C, Li H, Wang R, Wang Q, Wang H. Clinical profile, prognostic factors, and outcome prediction in hospitalized patients with bloodstream infection: results from a 10-year prospective multicenter study. Front Med (Lausanne). 2021;8: 629671.
Zhou HY, Yuan Z, Du YP. Prior use of four invasive procedures increases the risk of Acinetobacter baumannii nosocomial bacteremia among patients in intensive care units: a systematic review and meta-analysis. Int J Infect Dis. 2014;22:25–30.
Zhang X, Tong MM, Zhang MZ, Zhu HP. Risk factors of nosocomial bloodstream infections in surgical intensive care unit. Int J Clin Exp Med. 2015;8:16682–7.
Wenzel RP, Edmond MB. The impact of hospital-acquired bloodstream infections. Emerg Infect Dis J. 2001;7:174.
Bratzler DW, Hunt DR. The surgical infection prevention and surgical care improvement projects: national initiatives to improve outcomes for patients having surgery. Clin Infect Dis. 2006;43:322–30.
Ierano C, Thursky K, Marshall C, et al. Appropriateness of surgical antimicrobial prophylaxis practices in Australia. JAMA Netw Open. 2019;2: e1915003.
Pittalis S, Ferraro F, Piselli P, et al. Appropriateness of surgical antimicrobial prophylaxis in the Latium region of Italy, 2008: a multicenter study. Surg Infect (Larchmt). 2013;14:381–4.
Kaya S, Aktas S, Senbayrak S, et al. An evaluation of surgical prophylaxis procedures in Turkey: a multi-center point prevalence study. Eurasian J Med. 2016;48:24–8.
Mahmoudi L, Ghouchani M, Mahi-Birjand M, Bananzadeh A, Akbari A. Optimizing compliance with surgical antimicrobial prophylaxis guidelines in patients undergoing gastrointestinal surgery at a referral teaching hospital in southern Iran: clinical and economic impact. Infect Drug Resist. 2019;12:2437–44.
Kakolwa MA, Woodd SL, Aiken AM, et al. Overuse of antibiotics in maternity and neonatal wards, a descriptive report from public hospitals in Dar es Salaam, Tanzania. Antimicrob Resist Infect Control. 2021;10:142.
Karchmer AW. Nosocomial bloodstream infections: organisms, risk factors, and implications. Clin Infect Dis. 2000;31(Suppl 4):S139–43.
Lador A, Nasir H, Mansur N, et al. Antibiotic prophylaxis in cardiac surgery: systematic review and meta-analysis. J Antimicrob Chemother. 2012;67:541–50.
Wilson W, Taubert KA, Gewitz M, et al. Prevention of infective endocarditis: guidelines from the American Heart Association: a guideline from the American Heart Association Rheumatic Fever, Endocarditis, and Kawasaki Disease Committee, Council on Cardiovascular Disease in the Young, and the Council on Clinical Cardiology, Council on Cardiovascular Surgery and Anesthesia, and the Quality of Care and Outcomes Research Interdisciplinary Working Group. Circulation. 2007;116:1736–54.
Richey R, Wray D, Stokes T, Guideline DG. Prophylaxis against infective endocarditis: summary of NICE guidance. BMJ. 2008;336:770–1.
Cassidy SC, Schmidt KG, Van Hare GF, Stanger P, Teitel DF. Complications of pediatric cardiac catheterization: a 3-year study. J Am Coll Cardiol. 1992;19:1285–93.
Mehta R, Lee KJ, Chaturvedi R, Benson L. Complications of pediatric cardiac catheterization: a review in the current era. Catheter Cardiovasc Interv. 2008;72:278–85.
Zeng X, Yu G, Lu Y, et al. PIC, a paediatric-specific intensive care database. Sci Data. 2020;7:14.
Tweddell S, Loomba RS, Cooper DS, Benscoter AL. Health care-associated infections are associated with increased length of stay and cost but not mortality in children undergoing cardiac surgery. Congenit Heart Dis. 2019;14:785–90.
Abou Elella R, Najm HK, Balkhy H, Bullard L, Kabbani MS. Impact of bloodstream infection on the outcome of children undergoing cardiac surgery. Pediatr Cardiol. 2010;31:483–9.
Parajuli NP, Parajuli H, Pandit R, Shakya J, Khanal PR. Evaluating the trends of bloodstream infections among pediatric and adult patients at a teaching hospital of Kathmandu, Nepal: role of drug resistant pathogens. Can J Infect Dis Med Microbiol. 2017;2017:8763135.
Mendes RE, Sader HS, Castanheira M, Flamm RK. Distribution of main gram-positive pathogens causing bloodstream infections in United States and European hospitals during the SENTRY Antimicrobial Surveillance Program (2010–2016): concomitant analysis of oritavancin in vitro activity. J Chemother. 2018;30:280–9.
Zhu Q, Yue Y, Zhu L, et al. Epidemiology and microbiology of gram-positive bloodstream infections in a tertiary-care hospital in Beijing, China: a 6-year retrospective study. Antimicrob Resist Infect Control. 2018;7:107.
Levy I, Ovadia B, Erez E, et al. Nosocomial infections after cardiac surgery in infants and children: incidence and risk factors. J Hosp Infect. 2003;53:111–6.
Droz N, Hsia Y, Ellis S, Dramowski A, Sharland M, Basmaci R. Bacterial pathogens and resistance causing community acquired paediatric bloodstream infections in low- and middle-income countries: a systematic review and meta-analysis. Antimicrob Resist Infect Control. 2019;8:207.
Bratzler DW, Dellinger EP, Olsen KM, et al. Clinical practice guidelines for antimicrobial prophylaxis in surgery. Am J Health Syst Pharm. 2013;70:195–283.
Acknowledgements
We thank the researchers who made the Pediatric Intensive Care database freely and publicly accessible for conducting scientific research.
Funding
This study, including the journal’s Rapid Service Fees, was supported by the Shenzhen San-ming Project.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
All authors contributed to the material preparation and data analysis and/or interpretation. All authors read and approved the final manuscript and are accountable for accuracy and integrity of the data presented therein.
Disclosures
Qinchang Chen, Jinjin Yu, Pingchuan Huang, Yulu Huang, Qingui Chen, Zhiwei Zhang and Shushui Wang declare that they have no conflicts of interest in this work.
Compliance with Ethics Guidelines
The original project that developed the PIC database was approved by the Institutional Review Board (IRB) of the Children’s Hospital, Zhejiang University School of Medicine, and the requirement for individual patient consent was waived because the project did not impact clinical care, and all protected health information was deidentified (https://doi.org/10.1038/s41597-020-0355-4). After following the required procedures, our access to the PIC database was approved by the administrator of the PIC database on August 19, 2021. The current study was further approved by the IRB of the Guangdong Provincial People’s Hospital (No. KY-Q-2021-272-01). All the data used were de-identified and patient consent was waived. We complied with the Helsinki Declaration 1964, and its later amendments.
Data Availability
According to the access policy of the Pediatric Intensive Care database, sharing the data is not allowed, but the access to the database can be requested by following the formal procedures of the database (http://pic.nbscn.org/).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Chen, Q., Yu, J., Huang, P. et al. Incidence, Clinical Features, and Association with Prognosis of Bloodstream Infection in Pediatric Patients After Percutaneous or Surgical Treatment for Ventricular Septal Defect or Atrial Septal Defect: A Retrospective Cohort Study. Infect Dis Ther 11, 2219–2232 (2022). https://doi.org/10.1007/s40121-022-00702-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40121-022-00702-z