Abstract
Introduction
While incidence rates of vancomycin-resistant Enterococcus faecium have remained comparatively low in Japan, there have been increasing reports of more vancomycin-resistant Enterococcus (VRE) outbreaks, requiring costly measures to contain. Increased incidence of VRE in Japan may lead to more frequent and harder to contain outbreaks with current control measures, causing a significant burden to the healthcare system in Japan. This study aimed to demonstrate the clinical and economic burden of vancomycin-resistant E. faecium infections to the Japanese healthcare system and the impact of increasing rates of vancomycin resistance.
Methods
A de novo deterministic analytic model was developed to assess the health economic outcomes of treating hospital-acquired VRE infections; patients are treated according to a two-line treatment strategy, dependent on their resistance status. The model considers hospitalisation costs and the additional cost of infection control. Scenarios investigated the current burden of VRE infections and the additional burden of increased incidence of VRE. Outcomes were assessed over a 1-year and 10-year time horizon from a healthcare payer’s perspective in a Japanese setting. Quality-adjusted life years (QALYs) were valued with a willingness-to-pay threshold of ¥5,000,000 ($38,023), and costs and benefits were discounted at a rate of 2%.
Results
Current VRE incidence levels in enterococcal infections in Japan equates to ¥130,209,933,636 ($996,204,669) in associated costs and a loss of 185,361 life years (LYs) and 165,934 QALYs over 10 years. A three-fold increase (1.83%) is associated with an additional ¥4,745,059,504 ($36,084,651) in total costs on top of the current cost burden as well as an additional loss of 683 LYs over a lifetime, corresponding to 616 QALYs lost.
Conclusion
Despite low incidence rates, VRE infections already represent a substantial economic burden to the Japanese healthcare system. The substantial increase in costs associated with a higher incidence of VRE infections could result in a significant economic challenge for Japan.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Why carry out this study? |
Vancomycin-resistant Enterococcus (VRE) are a high priority pathogen due to their ability to develop and transfer resistance mechanisms and survive for prolonged periods on inanimate surfaces. |
VRE outbreaks in hospitals require costly countermeasures that aim to identify and prevent transmission to contain the outbreak. |
Vancomycin-resistant Enterococcus faecium incidence in Japan has remained low compared with other countries, with vancomycin resistance in 1.4% of E. faecium isolates compared with 16.8% in Europe and 82.1% in the US. |
This study aims to demonstrate the clinical and economic burden of vancomycin-resistant E. faecium to the Japanese healthcare system and the impact of increasing rates of vancomycin resistance. |
What was learned from the study? |
VRE infections in Japan pose a considerable clinical and economic burden to the healthcare system, despite the relatively low incidence; an increase in resistance rates would exacerbate this burden further with additional costs associated with containing VRE outbreaks contributing to the majority of economic burden. |
This analysis can be utilised to inform Japanese decision-makers of the significant impact on the healthcare system if VRE infections are not effectively controlled and the importance of preventative action against antimicrobial resistance (AMR). |
Introduction
Antimicrobial resistance (AMR) is a growing threat to public health worldwide that is associated with high morbidity and mortality [1, 2]. The emergence of multi-drug-resistant infections, combined with inadequate infection prevention and control measures, and insufficient development of new antimicrobials has resulted in increasingly difficult-to-treat and untreatable infections [1]. The World Health Organization (WHO) has developed a global action plan to address AMR worldwide. The plan, adopted in 2015, aims to ensure the effective treatment and prevention of infectious diseases by implementing country-specific strategies [3]. In 2016, the Japanese government devised the National Action Plan on AMR 2016–2020, outlining countermeasures against AMR across six key areas: surveillance and monitoring, public awareness and education, and infection prevention and control [4]. The Japanese National Action Plan targets were based on national surveillance data from the Japan Nosocomial Infections Surveillance (JANIS) database [2]. Data from the JANIS database will also support Japan’s and the WHO’s aims for improved surveillance of AMR and monitoring the impact AMR countermeasures [2, 5]. Gram-positive bacteria include a broad range of resistant pathogens that pose a serious concern due to their genetic ability to acquire and develop resistance mechanisms [6]. These include vancomycin-resistant Enterococcus (VRE), which has been identified as a high priority pathogen by the WHO in its global list of pathogens for research and development [7], reported as a ‘serious threat’ to human health by the Centers for Disease Control and Prevention (CDC) [8] and included in the Japanese priority pathogen list [2, 9].
The incidence of VRE infections has increased since it was first reported in the 1980s and has become a global cause for concern, in particular in hospital settings [10, 11]. Enterococci are facultative anaerobic gram-positive cocci responsible for causing infections including urinary tract infection (UTI), intra-abdominal infection (IAI), bacteraemia and endocarditis [12, 13]. Enterococci are normally commensal organisms to the human gastrointestinal tract, and VRE rarely causes infections when it enters the body of healthy individuals. However, VRE can cause severe infections such as sepsis and peritonitis in patients with cancer or serious underlying diseases. In addition, most antimicrobial agents used for VRE infections in clinical practice are often ineffective; as Enterococci are part of the normal microbiota, it is challenging to eradicate VRE colonisation, enabling patients to act as VRE reservoirs [14]. Enterococci are intrinsically resistant to a number of antimicrobial classes and have shown capacity to rapidly acquire and transfer multi-drug resistance mechanisms including vancomycin resistance [15]. Additionally, the ability of these organisms to survive on inanimate surfaces for several months increases the risk of transmission, which is an added challenge within a hospital environment [16]. The Enterococcus genus is made up of > 50 different species of Enterococci, though Enterococcus faecalis and Enterococcus faecium most commonly cause human infection [13]. While E. faecalis is more pathogenic than E. faecium, E. faecium is responsible for most VRE infections because of its higher resistance levels [17, 18]. Thus, E. faecium presents a bigger threat in the treatment of infectious diseases. VRE infections are associated with both increased mortality relative to Enterococci infections and greater healthcare costs and length of hospital stay [19, 20]. In addition to the risk of VRE outbreaks, there is a risk that resistance may be transmitted to other gram-positive pathogens, for example, methicillin-resistant Staphylococcus aureus [7, 10].
In Japan, the first VRE infections were identified in 1996. Since 2013 the major species causing VRE infections in Japan continues to be E. faecium [21]. Incidence has remained low compared with other counties, with vancomycin resistance in 1.4% of E. faecium isolates compared with 16.8% in Europe and 82.1% in the US [22,23,24]. Between 2016 and 2020, the European Centre for Disease Prevention and Control (ECDC) via the European Antimicrobial Resistance Surveillance Network (EARS-Net) and the Central Asian and European Surveillance of Antimicrobial Resistance (CAESAR) identified a significant increase in vancomycin resistance in E. faecium, from 11.6% to 16.8% [23]. Unlike many other resistant bacterial pathogens, vancomycin-resistant E.faecium followed no distinct geographical patterns [23]. The main mechanism of vancomycin resistance involves alterations of the portion of the cell wall that vancomycin binds to; the vanA and vanB operons have been identified to be responsible for most VRE outbreaks in humans [25]. Over the past 10 years, newly emerged resistant clones have outcompeted and replaced existing clones, for example vanA ST1421 in Australia and vanB ST192 in Germany [26, 27]. Such strains like ST1421 and related endemic multilocus sequence typing non-typable VRE have been responsible for causing a multi-jurisdictional outbreak in Australia and have spread worldwide [28,29,30]. A study of the global dissemination of VRE clones indicated a regional link for the distribution of E. faecium between Japan and Australia [31]. Indeed, the first outbreak of the ST1421 strain occurred in Aomori Japan in 2018 leading to 500 cases across 14 hospitals [32]. Since this first outbreak there have been other reports of large nosocomial VRE outbreaks in Japan [33,34,35]. VRE outbreaks in Japan are increasing in frequency in acute-care hospitals and subsequently spreading into satellite hospitals with long-term care beds [36]. The importance of infection control measures to control AMR in healthcare associated infections have been recognised for decades [37]. However, due to additional transmission risks associated with VRE infections, additional countermeasures such as contact precaution, personal protective equipment, environmental hygiene, on-admission surveillance (including environmental culture) and antimicrobial stewardship have been adopted by hospitals to contain outbreaks [37, 38]. Despite control measures being associated with increased cost, they have been shown to be cost-effective because of the high monetary burden associated with VRE outbreaks [20]. Nevertheless, if vancomycin resistance in Japan increases to rates similar to those observed in Europe (16.8%) [23] and the US (82.1%) [24], outbreaks may become more frequent and harder to contain with current control measures, causing a significant burden to healthcare providers in Japan.
This study aims to demonstrate the clinical and economic burden of vancomycin-resistant E. faecium to the Japanese healthcare system and the impact of increasing rates of vancomycin resistance.
Methods
In this study a de novo deterministic analytic model of VRE infections in the Japanese setting was built to investigate the current burden of VRE infections as well as the additional burden of alternative scenarios where the incidence of VRE is increased. The methodology behind the treatment pathway component of this model follows the same principles as shown in previous studies [39,40,41]. The model considers patients at a population level who have a hospital-acquired infection (HAI) caused by the gram-positive pathogen E. faecium, including UTIs, bloodstream infections (BSIs) and IAIs. The model estimated both clinical and economic outcomes over a 1-year and 10-year time horizon from a healthcare payer’s perspective in Japan. This study is reported in line with the recommendations of the Consolidated Health Economic Evaluation Reporting Standards 2022 (CHEERS 2022) statement [42].
Model Structure
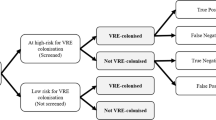
The model structure allows a treatment pathway of up to two lines of treatment. Treatments include ampicillin (ABPC), daptomycin (DAP), linezolid (LZD) and vancomycin (VCM). As VRE rates and drugs reimbursed or indications differ between Japan and other countries, the treatment pathway was defined based on Japanese clinical guidelines and local expert opinion to best represent clinical practice in Japan [43]. The local expert (TM) is a physician specialising in infectious diseases who works in a university hospital in Japan. It is assumed that APBC and VCM susceptibility status is known for all patients. For LZD and DAP, susceptibility status is assumed to be 100% as the resistance levels of these treatments are < 0.1% in Japan [22]. Therefore, grouping patients according to susceptibility to LZD and DAP is not considered. The treatment pathway is specific to patients’ indication and susceptibility to ABPC and VCM. The treatment pathway best representative of each patient profile, informed by the Japanese clinical guidelines and local expert opinion [43], is presented in Fig. 1.
Model schematic: Patients with Enterococcus faecium infections are treated according to the treatment regimen based on their indication and susceptibility. The deterministic treatment pathway estimates outcomes for cost, utility and survival based on the probabilities of successful or unsuccessful treatment associated with each treatment and indication. ABPC = ampicillin, BSI = bloodstream infection, DAP = daptomycin, IAI = intra-abdominal infection, LZD = linezolid, UTI = urinary tract infection. *Alternative treatment strategy used to treat patients with bloodstream infections
Upon entering the appropriate deterministic treatment pathway (Fig. 1), all patients receive the first-line antimicrobial treatment according to their specified treatment pathway. Patients can have three responses to treatment: (1) cured through successful treatment or the infection naturally resolves, (2) die from infection or (3) remain infected because of unsuccessful treatment. Patients who remain infected progress onto the second-line antimicrobial in their appropriate treatment pathway. When all treatment options are exhausted and a patient is still infected, it is assumed that these patients will die 3 days after their last treatment, based on local expert opinion.
Japanese-specific inputs were applied in the model to estimate the clinical (hospital length of stay [LOS], defined daily dose [DDD] of antimicrobials, quality-adjusted life years [QALYs] and life years [LY]) and economic burden (hospitalisation costs and monetary burden) of VRE infections in Japan. Costs were converted from Japanese yen to US dollars using the 2022 annual exchange rate, as published by the Organization for Economic Cooperation and Development, of US $1 = ¥131.498 [44].
Model Settings
The total annual infected population in Japan of 40,565 patients (mean: 75 years, median: 78 years) is based on estimates from Medical Data Vision (MDV), a Japanese medical claims database [45], and JANIS data [46], with the probability of E. faecium within each indication (UTI, BSI and IAI) informed by published literature [47,48,49]. Since 2003, data from over 40 million patients attending Japanese public and private healthcare facilities have been captured through the MDV. For the purpose of this analysis, data were extracted from patients aged 15 years and older with a diagnosis in a relevant indication: UTI, BSI and IAI. Population inputs reflecting the distribution of patients by indication and susceptibility to ABPC and VCM are presented in Table 1, and calculations are detailed in Tables S1, S2 and S3.
Key clinical inputs including mortality, utility and treatment efficacy inputs are presented in Table 2. Utility (infected) parameters are aligned to the utility of an infected patient specific to each modelled indication (UTI, BSI and IAI) to estimate QALYs lost due to infection. Parameters for utility of patients no longer infected is aligned to the general population in Japan at the average age of the infected population (75 years) [45]. Life expectancy was determined using Japanese life tables matched to the modelled population age [50].
The model considers the daily cost associated with indication-specific hospitalisations and the additional cost for preventing the spread of VRE infections, such as testing uninfected patients in the same ward as VRE-infected patients (testing costs for infected patients are included in the daily hospitalisation costs), cost of contact precautions incurred in addition to standard precautions for both infected patients and uninfected patients, cost of private room charges lost per infected patient per day and cost of environmental cultures per infected patient. These additional costs, as presented in Table 3, were considered to capture the additional burden of containing VRE infections. The outcome of the additional costs of uninfected patients is calculated considering the number of patients per VRE-infected patient (by hospital type) and the percentage of these hospitals in Japan, as described in Table S5 in the supplementary material.
Inputs related to LOS have been informed by expert opinion (Table 3). For non-VRE patients, the LOS associated with a successful treatment (per therapy line) for BSI and IAI is assumed to be 14 days and 7 days for UTI. For VRE patients it was assumed that patients would not be discharged immediately after the infection clears as a precautionary measure; therefore, the LOS associated with a successful treatment (per therapy line) for BSI and IAI is assumed to be 20 days and 10 days for UTI. The model assumes that patients will switch treatment after 4 days if the treatment is unsuccessful, if an alternative is available, and the LOS associated with mortality is assumed to be an additional 3 days, based on the local expert opinion.
Analysis
The burden of VRE in enterococcal infections with the current resistance level of 0.61% and two alternative scenarios—1.83% reflecting three times the current level (Scenario 1) and 6.10% reflecting ten times the current level in Japan (Scenario 2)—were evaluated at a 1-year and 10-year time horizon. Current resistance is based on unpublished JANIS 2020 data of blood samples only [46]. A willingness-to-pay threshold of ¥5,000,000 ($38,023) per QALY was considered and a discount rate of 2% was applied to costs and benefits in line with guidelines for cost-effective evaluations [66, 67].
Sensitivity Analysis
A one-way sensitivity analysis (OWSA) was undertaken on Scenario 1 where outcomes for additional costs and QALYs were reported; sensitivity analyses were also conducted for outcomes of current resistance levels and Scenario 2 in the supplementary materials. Key inputs were adjusted by ± 20% and the discount rate was excluded.
Ethics Approval
Ethics approval was not required for this study; the analysis in this article is based on previously publicly available data and does not involve any new studies of human or animal subjects performed by any of the authors.
Results
Cumulative outcomes demonstrating the clinical and economic burden with the current VRE infection rate and two scenario analyses exploring alternative levels of VRE in enterococcal infections in Japan at a 10-year time horizon are presented in Table 4; 1-year outcomes are presented in Table S7 in the supplementary material.
Clinical and Economic Burden of Current VRE Infection Levels (0.61%)
Over 1 year, it is estimated that the current VRE incidence in enterococcal infections in Japan is associated with 460,473 hospital bed days, an additional 455,116 DDDs and 1418 deaths. Additionally, an estimated 18,536 LYs and 16,620 QALYs will be lost. This equates to ¥14,012,109,996 ($106,557,560) in hospitalisation costs and a total cost of ¥14,236,257,761 ($108,262,162). The additional cost of infection control (testing, contact precautions, environmental cultures and lost private room fees) for VRE infected patients and uninfected patients in same ward as VRE patient accounted for ¥94,890,482 ($721,611) and ¥129,257,284 ($982,960) of the total costs, respectively. Over 10 years, the current incidence of VRE in enterococcal infections is estimated to be associated with 14,180 deaths, 4,604,728 hospital bed days and 4,551,160 DDDs, as well as a loss of 185,361 LYs and 165,934 QALYs. This relates to hospitalisation costs of ¥128,159,797,554 ($974,614,044) and a total cost of ¥130,209,933,636 ($990,204,669). The additional cost of infection control (VRE-infected patients and uninfected patients in same ward as VRE patient) accounted for ¥867,902,474 ($6,600,119) and ¥1,182,233,608 ($8,990,506) of the total costs, respectively.
Clinical and Economic Burden of VRE Infection Levels Increased to 1.83% (Scenario 1)
With an increased VRE incidence in enterococcal infections three times that of the current level (1.83%), over 10 years it is estimated to cost an additional ¥4,745,059,504 ($36,084,651) in total costs compared to the costs associated with current VRE incidence. The increased cost of infection control (VRE-infected patients and uninfected patients in the same ward as a VRE patient) accounted for the majority of the additional costs (¥1,735,804,949 and ¥2,364,467,215, respectively) (Fig. 2). This increase in VRE incidence results in an additional 23,174 hospital bed days; 964 DDDs (Fig. 3) will be required and an additional 52 deaths will occur. Furthermore, an estimated additional 683 LYs will be lost over a lifetime, which corresponds to 616 QALYs lost, as shown in Fig. 3. Over a lifetime, this equates to an additional monetary burden of approximately ¥7,823,023,155 ($59,491,575) at a willingness-to-pay threshold of ¥5,000,000 ($38,023) per QALY (Fig. 4).
Clinical and Economic Burden of VRE Infection Levels in Enterococcal Infections Increased to 6.10% (Scenario 2)
A ten-fold increase to the current VRE incidence in enterococcal infections from 0.61 to 6.10% results in an estimated additional ¥21,352,767,766 ($162,380,932) in total costs compared with the costs associated with current VRE incidence. The additional cost of infection control (VRE-infected patients and uninfected patients in the same ward as a VRE patient) accounted for the majority of the additional costs (¥7,811,122,268 and ¥10,640,102,469, respectively) (Fig. 2). This equates to an additional 104,281 hospital bed days and 4337 DDDs (Fig. 3) as well as an additional 235 deaths over a 10-year time horizon. An additional 3075 LYs will also be lost over a lifetime, corresponding to 2770 QALYs lost (Fig. 3). Over a lifetime, this equates to an additional monetary burden of approximately ¥35,203,604,197 ($267,712,088) at a willingness-to-pay threshold of ¥5,000,000 ($38,023) per QALY (Fig. 4).
Sensitivity Analysis
Adjusting model inputs ± 20% during an OWSA conducted for Scenario 1 (1.83%) demonstrated that the cost of testing per patient has the greatest impact on the outcome for additional costs, which ranged from ¥4,272,166,060 ($32,488,449) to ¥5,217,952,947 ($39,680,854). Hospital length of stay for successful VRE treatment (IAI) and private room costs were also shown to be drivers of cost. Varying these changed additional cost outputs to range from ¥4,359,422,895 ($33,152,009) to ¥5,130,696,093 ($39,017,294) and ¥4,455,266,987 ($33,880,873) to ¥5,034,852,020 ($38,288,430), respectively (Fig. 5). Treatment efficacy was shown to have the largest impact on QALYs lost as demonstrated in Fig. 6. VCM efficacy ranges from − 1720 to 2531 and DAP efficacy from 2533 to − 1273. See Figure S1, S2, S3, and S4 in the supplementary material for the OWSA of current VRE resistance level (0.61%) and Scenario 2 (6.10%).
OWSA on additional costs when current VRE incidence in enterococcal infections in Japan are increased to 1.83%. ABPC = ampicillin, BSI = bloodstream infection, DAP = daptomycin, IAI = intra-abdominal infection, LOS = length of stay, LZD linezolid, OWSA = one-way sensitivity analysis, UTI = urinary tract infection, VCM = vancomycin, VRE = vancomycin-resistant Enterococci
OWSA on QALYs when current VRE incidence in enterococcal infections in Japan are increased to 1.83%. ABPC = ampicillin, BSI = bloodstream infection, DAP = daptomycin, IAI = intra-abdominal infection, LOS = length of stay, LZD = linezolid, OWSA = one-way sensitivity analysis, QALY = quality-adjusted life year, UTI = urinary tract infection, VCM = vancomycin, VRE = vancomycin-resistant Enterococci
Discussion
This study demonstrates that the threat of increased VRE infection rates in Japan would result in significant additional clinical and economic burden to healthcare providers in the long term. To the authors’ knowledge, this is the first deterministic analysis to assess current burden and also predict the clinical and economic impact of increased VRE infection incidence in Japan. Despite low incidence of VRE infections in Japan, a substantial clinical and economic burden to the healthcare system already exists. The analyses showed the current incidence of VRE in enterococcal infections over 10 years equates to ¥130,209,933,636 ($996,204,669) in total healthcare costs and a loss of 185,361 LYs and 165,934 QALYs. A ten-fold increase (6.10%) in current levels of VRE in enterococcal infections—in line with VRE infection rates seen in Europe and the US—is associated with an additional ¥21,352,767,766 ($162,380,932) in total healthcare costs on top of the current cost burden as well as an additional loss of 3075 LYs over a lifetime, corresponding to 2770 QALYs lost.
This study highlights VRE outbreaks in hospitals are associated with considerable economic burden, where the majority of total additional costs associated with increased VRE incidence are attributed to costs associated with containing infections (testing, contact precautions, environmental cultures and lost private room fees). These findings are supported by research conducted by Morii et al. (2020), who estimated the total cost of containing an AMR HAI outbreak in a Japanese hospital to be $678,000, per outbreak, including surveillance, cleaning and decontamination, and disposal and repurchasing [68]. However, the authors also showed that hospitals experienced productivity losses which were much greater than the containment costs, costing $4.62 million [68]. Whilst our analysis considered losses for private room fees, Morii et al. showed the length of bed occupation was not significantly associated with productivity losses, suggesting that there are considerable additional costs associated with VRE outbreaks faced by hospitals not considered in our analysis.
Due to the high-risk nature of VRE with its ability to survive on inanimate surfaces for prolonged periods of time and transfer multi-drug resistance mechanisms, despite high costs containment countermeasures including contact precaution and isolation, personal protective equipment, environmental hygiene and additional testing and screening are critical. Previous investigations report these countermeasures are advantageous against VRE outbreaks [69]. Mac et al. (2019) showed that screening and isolation measures are able to prevent VRE colonisation and transmission in the hospital setting compared with the absence of such measures [70]. However, if an outbreak is not appropriately controlled by these measures and spreads beyond an isolated group of patients, eradication becomes even more difficult and expensive [71]. While preventative measures such as testing and isolation are associated with high costs, they have been shown to be cost-effective compared with the economic impact of large-scale VRE outbreaks on healthcare systems [70]. In Japan, hospitals are reimbursed for the cost of infection control; however, no additional costs can be charged even in the case of an outbreak. Therefore, many of the costs associated with containing a VRE outbreak are taken as out-of-pocket costs from the hospital’s budget; more frequent and larger outbreaks associated with increase VRE incidence will exert considerable strain on hospital budgets displacing funding for other treatments and services. This reinforces the need for effective prevention and control measures against multi-drug-resistant infections such as VRE despite the current low incidence rates. If appropriate measures are not implemented, considering the substantial burden realised even with low incidence rates, the escalation of VRE infection rates could be catastrophic to the Japanese healthcare system.
As with any study of its kind, the analyses are subject to some limitations. As this is a deterministic model, the incidence of infection is assumed to be constant throughout the modelled population as well as the level of VRE within the population. Consequently, this may not be fully representative of changes in VRE levels. The model is limited to exploring two lines of treatment to best represent E. faecium infections for the indications of interest resulting in a possible underestimation of VRE infections caused by other classes of Enterococci. Additionally, the model does not consider change in future populations and demographics. Finally, as mentioned earlier, the model does not consider broader healthcare and societal costs, for example, opportunity costs associated with an increased number of patients in isolation, reducing ward capacity and increased hospital staff care requirements, and the social impact of isolation on emotional wellbeing of patients. Therefore, the impact of increased VRE levels is potentially far greater than demonstrated in this study.
Conclusion
This study demonstrates the clinical and economic burden of increased vancomycin-resistant E. faecium infection rates in Japan. Despite low incidence rates of VRE at present, a significant burden exists to the Japanese healthcare system. If resistance rises to the levels seen in Europe and the US, with the additional costs associated with containing a VRE outbreak, this could become a significant economic challenge for the Japanese healthcare system. This analysis can be utilised to inform Japanese healthcare decision-makers of the significant impact on the healthcare system if VRE infections are not effectively controlled as well as to highlight the importance of preventative action against AMR.
Change history
27 August 2023
A Correction to this paper has been published: https://doi.org/10.1007/s40121-023-00853-7
References
Frieri M, Kumar K, Boutin A. Antibiotic resistance. J Infect Public Health. 2017;10(4):369–78.
Kajihara T, Yahara K, Hirabayashi A, Shibayama K, Sugai M. Japan nosocomial infections surveillance (JANIS): current status, international collaboration, and future directions for a comprehensive antimicrobial resistance surveillance system. Jpn J Infect Dis. 2021;74(2):87–96.
World Health Organization. Global action plan on antimicrobial resistance 2015. Available from: https://www.who.int/publications/i/item/9789241509763. Accessed 1 Apr 2023.
The Government of Japan. National Action Plan on Antimicrobial Resistance (AMR) 2016–2020. Available from: https://www.mhlw.go.jp/file/06-Seisakujouhou-10900000-Kenkoukyoku/0000138942.pdf. Accessed 1 Apr 2023.
World Health Organization. Global Antimicrobial Resistance and Use Surveillance System (GLASS) report: 2022. Available from: https://www.who.int/initiatives/glass. Accessed 1 Apr 2023.
Jubeh B, Breijyeh Z, Karaman R. Resistance of gram-positive bacteria to current antibacterial agents and overcoming approaches. Molecules. 2020. https://doi.org/10.3390/molecules25122888.
Tacconelli E, Carrara E, Savoldi A, Harbarth S, Mendelson M, Monnet DL, et al. Discovery, research, and development of new antibiotics: the WHO priority list of antibiotic-resistant bacteria and tuberculosis. Lancet Infect Dis. 2018;18(3):318–27.
Centers for Disease Control and Prevention. Antibiotic resistance threats in the United States 2019. Available from: https://www.cdc.gov/drugresistance/pdf/threats-report/2019-ar-threats-report-508.pdf. Accessed 1 Apr 2023.
Japan Agency for Medical Research and Development (AMED). Priority Pathogens Lists for R&D of New Antibiotics (2021 version). 2022. Available from: https://id3catalyst.jp/apid/en/list.html. Accessed 1 Apr 2023.
Cetinkaya Y, Falk P, Mayhall CG. Vancomycin-resistant enterococci. Clin Microbiol Rev. 2000;13(4):686–707.
Ye JJ, Shie SS, Cheng CW, Yang JH, Huang PY, Wu TS, et al. Clinical characteristics and treatment outcomes of vancomycin-resistant Enterococcus faecium bacteremia. J Microbiol Immunol Infect. 2018;51(6):705–16.
Levitus M, Rewane A, Perera TB. Vancomycin-resistant enterococci. StatPearls: StatPearls Publishing; 2022.
Miller WR, Murray BE, Rice LB, Arias CA. Resistance in vancomycin-resistant enterococci. Infect Dis Clin North Am. 2020;34(4):751–71.
Willems RPJ, van Dijk K, Vehreschild M, Biehl LM, Ket JCF, Remmelzwaal S, et al. Incidence of infection with multidrug-resistant Gram-negative bacteria and vancomycin-resistant enterococci in carriers: a systematic review and meta-regression analysis. Lancet Infect Dis. 2023. https://doi.org/10.1016/S1473-3099(22)00811-8.
Gorrie C, Higgs C, Carter G, Stinear TP, Howden B. Genomics of vancomycin-resistant Enterococcus faecium. Microb Genom. 2019;5(7): e000283.
Kramer A, Schwebke I, Kampf G. How long do nosocomial pathogens persist on inanimate surfaces? A systematic review. BMC Infect Dis. 2006. https://doi.org/10.1186/1471-2334-6-130.
Sievert DM, Ricks P, Edwards JR, Schneider A, Patel J, Srinivasan A, et al. Antimicrobial-resistant pathogens associated with healthcare-associated infections: summary of data reported to the National Healthcare Safety Network at the Centers for Disease Control and Prevention, 2009–2010. Infect Control Hosp Epidemiol. 2013;34(1):1–14.
French GL. Enterococci and vancomycin resistance. Clin Infect Dis. 1998;27(Suppl 1):S75-83.
DiazGranados CA, Zimmer SM, Mitchel K, Jernigan JA. Comparison of mortality associated with vancomycin-resistant and vancomycin-susceptible enterococcal bloodstream infections: a meta-analysis. Clin Infect Dis. 2005;41(3):327–33.
MacDougall C, Johnstone J, Prematunge C, Adomako K, Nadolny E, Truong E, et al. Economic evaluation of vancomycin-resistant enterococci (VRE) control practices: a systematic review. J Hosp Infect. 2020;105(1):53–63.
National Institute of Infectious Diseases. Infectious Agents Surveillance Report (IASR) 2021. 42(8):155–167. Available from: https://www.niid.go.jp/niid/ja/vre-m/vre-iasrtpc/10589-498t.html. Accessed 1 Apr 2023.
Japan Nosocomial Infections Surveillance. Annual Open Report 2020 (All Facilities). Available from: https://janis.mhlw.go.jp/english/report/index.html. Accessed 1 Apr 2023.
European Centre for Disease Prevention and Control. Surveillance of antimicrobial resistance in Europe, 2020 data. Available from: https://www.ecdc.europa.eu/sites/default/files/documents/Surveillance-antimicrobial-resistance-in-Europe-2020.pdf. Accessed 1 Apr 2023.
Weiner-Lastinger LM, Abner S, Edwards JR, Kallen AJ, Karlsson M, Magill SS, et al. Antimicrobial-resistant pathogens associated with adult healthcare-associated infections: summary of data reported to the National Healthcare Safety Network, 2015–2017. Infect Control Hosp Epidemiol. 2020;41(1):1–18.
Ahmed MO, Baptiste KE. Vancomycin-resistant enterococci: a review of antimicrobial resistance mechanisms and perspectives of human and animal health. Microb Drug Resist. 2018;24(5):590–606.
van Hal SJ, Beukers AG, Timms VJ, Ellem JA, Taylor P, Maley MW, et al. Relentless spread and adaptation of non-typeable vanA vancomycin-resistant Enterococcus faecium: a genome-wide investigation. J Antimicrob Chemother. 2018;73(6):1487–91.
Bender JK, Kalmbach A, Fleige C, Klare I, Fuchs S, Werner G. Population structure and acquisition of the vanB resistance determinant in German clinical isolates of Enterococcus faecium ST192. Sci Rep. 2016;6:21847.
Carter GP, Buultjens AH, Ballard SA, Baines SL, Tomita T, Strachan J, et al. Emergence of endemic MLST non-typeable vancomycin-resistant Enterococcus faecium. J Antimicrob Chemother. 2016;71(12):3367–71.
Lemonidis K, Salih TS, Dancer SJ, Hunter IS, Tucker NP. Emergence of an Australian-like pstS-null vancomycin resistant Enterococcus faecium clone in Scotland. PLoS One. 2019;14(6): e0218185.
Kim HM, Chung DR, Cho SY, Huh K, Kang CI, Peck KR. Emergence of vancomycin-resistant Enterococcus faecium ST1421 lacking the pstS gene in Korea. Eur J Clin Microbiol Infect Dis. 2020;39(7):1349–56.
van Hal SJ, Willems RJL, Gouliouris T, Ballard SA, Coque TM, Hammerum AM, et al. The global dissemination of hospital clones of Enterococcus faecium. Genome Med. 2021;13(1):52.
Saito N, Kitazawa J, Horiuchi H, Yamamoto T, Kimura M, Inoue F, et al. Interhospital transmission of vancomycin-resistant Enterococcus faecium in Aomori, Japan. Antimicrob Resist Infect Control. 2022;11(1):99.
Kitagawa D, Komatsu M, Nakamura A, Suzuki S, Oka M, Masuo K, et al. Nosocomial infections caused by vancomycin-resistant Enterococcus in a Japanese general hospital and molecular genetic analysis. J Infect Chemother. 2021;27(12):1689–93.
Takakura S. Vancomycin-resistant enterococci. Rinsho Byori. 2016;64(11):1243–8.
Matsushima A, Takakura S, Yamamoto M, Matsumura Y, Shirano M, Nagao M, et al. Regional spread and control of vancomycin-resistant Enterococcus faecium and Enterococcus faecalis in Kyoto, Japan. Eur J Clin Microbiol Infect Dis. 2012;31(6):1095–100.
National Institute of Infectious Diseases. Infectious. Infectious Agents Surveillance Report (IASR). 2021. 42(5):100–101. Available from: https://www.niid.go.jp/niid/ja/vre-m/vre-iasrs/10264-495p01.html. Accessed 1 Apr 2023.
Muto CA, Jernigan JA, Ostrowsky BE, Richet HM, Jarvis WR, Boyce JM, et al. SHEA guideline for preventing nosocomial transmission of multidrug-resistant strains of Staphylococcus aureus and enterococcus. Infect Control Hosp Epidemiol. 2003;24(5):362–86.
Siegel JD, Rhinehart E, Jackson M, Chiarello L. Management of multidrug-resistant organisms in health care settings, 2006. Am J Infect Control. 2007;35(10 Suppl 2):S165–93.
Gordon J, Darlington O, McEwan P, Lumley M, Taie A, Hicks M, et al. Estimating the value of new antimicrobials in the context of antimicrobial resistance: development and application of a dynamic disease transmission model. Pharmacoeconomics. 2020;38(8):857–69.
Matsumoto T, Darlington O, Miller R, Gordon J, McEwan P, Ohashi T, et al. Estimating the economic and clinical value of reducing antimicrobial resistance to three gram-negative pathogens in Japan. J Health Econ Outcomes Res. 2021;8(2):64–75.
Matsumoto T, Yuasa A, Miller R, Pritchard C, Ohashi T, Taie A, et al. Estimating the economic and clinical value of introducing ceftazidime/avibactam into antimicrobial practice in Japan: a dynamic modelling study. Pharmacoecon Open. 2023;7(1):65–76.
Husereau D, Drummond M, Augustovski F, de Bekker-Grob E, Briggs AH, Carswell C, et al. Consolidated health economic evaluation reporting standards 2022 (CHEERS 2022) statement: updated reporting guidance for health economic evaluations. Value Health. 2022;25(1):3–9.
JAID/JSC Infectious Disease Treatment Guide Guideline Formulation Committee. JAID/JSC Infectious Disease Treatment Guide = The JAID/JSC guide to clinical management of infectious diseases. 2019.
Organization for Economic Cooperation and Development. OECD Data Exchange rates 2022. Available from: https://data.oecd.org/conversion/exchange-rates.htm. Accessed 1 Apr 2023.
Medical Data Vision. Available from: https://en.mdv.co.jp/. Accessed 1 Apr 2023.
Ministry of Health Labour and Welfare, Japan Nosocomial Infections Surveillance. Data on file (Patient identifiers were de-identified by each facility before submission to JANIS. approval number: 0628–4). 2020.
Huang L, Huang C, Yan Y, Sun L, Li H. Urinary tract infection etiological profiles and antibiotic resistance patterns varied among different age categories: a retrospective study from a tertiary general hospital during a 12-year period. Front Microbiol. 2021;12: 813145.
Liu C, Xu M, Li X, Dong H, Ming L. Trends in antimicrobial resistance in bloodstream infections at a large tertiary-care hospital in China: a 10-year retrospective study (2010–2019). J Glob Antimicrob Resist. 2022;29:413–9.
Zhang J, Zhao C, Chen H, Li H, Wang Q, Wang Z, et al. A multicenter epidemiology study on the risk factors and clinical outcomes of nosocomial intra-abdominal infections in China: results from the Chinese Antimicrobial Resistance Surveillance of Nosocomial Infections (CARES) 2007–2016. Infect Drug Resist. 2018;11:2311–9.
National Institute of Population and Social Security Research. Life tables 2020 (Age 75–79). Available from: https://www.ipss.go.jp/p-toukei/JMD/00/STATS/bltper_5x1.txt. Accessed 1 Apr 2023.
Wozniak TM, Dyda A, Lee X. The increased length of hospital stay and mortality associated with community-associated infections in Australia. Open Forum Infect Dis. 2022;9(5):ofac133.
Pinholt M, Ostergaard C, Arpi M, Bruun NE, Schønheyder HC, Gradel KO, et al. Incidence, clinical characteristics and 30-day mortality of enterococcal bacteraemia in Denmark 2006–2009: a population-based cohort study. Clin Microbiol Infect. 2014;20(2):145–51.
Morvan AC, Hengy B, Garrouste-Orgeas M, Ruckly S, Forel JM, Argaud L, et al. Impact of species and antibiotic therapy of enterococcal peritonitis on 30-day mortality in critical care-an analysis of the OUTCOMEREA database. Crit Care. 2019;23(1):307.
Shiroiwa T, Noto S, Fukuda T. Japanese population norms of EQ-5D-5L and health utilities index mark 3: disutility catalog by disease and symptom in community settings. Value Health. 2021;24(8):1193–202.
Ernst EJ, Ernst ME, Hoehns JD, Bergus GR. Women’s quality of life is decreased by acute cystitis and antibiotic adverse effects associated with treatment. Health Qual Life Outcomes. 2005;3:45.
Pliakos EE, Andreatos N, Shehadeh F, Ziakas PD, Mylonakis E. The cost-effectiveness of rapid diagnostic testing for the diagnosis of bloodstream infections with or without antimicrobial stewardship. Clin Microbiol Rev. 2018. https://doi.org/10.1128/CMR.00095-17.
Brasel KJ, Borgstrom DC, Weigelt JA. Cost-utility analysis of contaminated appendectomy wounds. J Am Coll Surg. 1997;184(1):23–30.
Wingler MJ, Patel NR, King ST, Wagner JL, Barber KE, Stover KR. Linezolid for the treatment of urinary tract infections caused by vancomycin-resistant enterococci. Pharmacy (Basel). 2021. https://doi.org/10.3390/pharmacy9040175.
Twilla JD, Finch CK, Usery JB, Gelfand MS, Hudson JQ, Broyles JE. Vancomycin-resistant Enterococcus bacteremia: an evaluation of treatment with linezolid or daptomycin. J Hosp Med. 2012;7(3):243–8.
Quickert S, Würstle S, Reuken PA, Hagel S, Schneider J, Schmid RM, et al. Real-world effectiveness of piperacillin/tazobactam with and without linezolid for spontaneous bacterial peritonitis. Dig Dis. 2022;40(6):777–86.
Naber KG, Eisenstein BI, Tally F. Daptomycin versus ciprofloxacin in the treatment of complicated urinary tract infection due to gram-positive bacteria. Infect Dis Clin Pract. 2004;12:322–7.
Piano S, Fasolato S, Salinas F, Romano A, Tonon M, Morando F, et al. The empirical antibiotic treatment of nosocomial spontaneous bacterial peritonitis: results of a randomized, controlled clinical trial. Hepatology. 2016;63(4):1299–309.
Ministry of Health Labour and Welfare. Diagnostic Procedure Classification (DPC) electronic score table 2022. Available from: https://www.mhlw.go.jp/stf/seisakunitsuite/bunya/0000198757_00004.html. Accessed 1 Apr 2023.
Ministry of Health Labour and Welfare. Japan Medical Fee Points 2022. Available from: https://www.mhlw.go.jp/stf/seisakunitsuite/bunya/0000188411_00037.html. Accessed 1 Apr 2023.
Trick WE, Temple RS, Chen D, Wright MO, Solomon SL, Peterson LR. Patient colonization and environmental contamination by vancomycin-resistant enterococci in a rehabilitation facility. Arch Phys Med Rehabil. 2002;83(7):899–902.
Center for Outcomes Research and Economic Evaluation for Health. Guideline for Preparing Cost-Effectiveness Evaluation to the Central Social Insurance Medical Council. Version 3.0. 2022. Available from: https://c2h.niph.go.jp/tools/guideline/guideline_en.pdf. Accessed 1 Apr 2023.
Hasegawa M, Komoto S, Shiroiwa T, Fukuda T. Formal implementation of cost-effectiveness evaluations in Japan: a unique health technology assessment system. Value Health. 2020;23(1):43–51.
Morii D, Tomono K, Imanaka Y. Economic impact of antimicrobial-resistant bacteria outbreaks on Japanese hospitals. Am J Infect Control. 2020;48(10):1195–9.
Escaut L, Bouam S, Frank-Soltysiak M, Rudant E, Saliba F, Kassis N, et al. Eradication of an outbreak of vancomycin-resistant Enterococcus (VRE): the cost of a failure in the systematic screening. Antimicrob Resist Infect Control. 2013;2(1):18.
Mac S, Fitzpatrick T, Johnstone J, Sander B. Vancomycin-resistant enterococci (VRE) screening and isolation in the general medicine ward: a cost-effectiveness analysis. Antimicrob Resist Infect Control. 2019;8:168.
Lai KK, Kelley AL, Melvin ZS, Belliveau PP, Fontecchio SA. Failure to eradicate vancomycin-resistant enterococci in a university hospital and the cost of barrier precautions. Infect Control Hosp Epidemiol. 1998;19(9):647–52.
Acknowledgements
The MDV data were extracted and tabulated by Linghua Xu, Pfizer Japan Inc.
Funding
This work was supported by Pfizer Japan Inc., including the journal’s Rapid Service Fee.
Medical Writing Assistance
The authors’ heartfelt appreciation goes to Chloe Hembury and James Dennis of Health Economics and Outcomes Research Ltd. for providing medical writing/editorial support, which was funded by Pfizer Japan Inc. in accordance with Good Publication Practice (GPP3) guidelines (http://www.ismpp.org/gpp3).
Authorship
All authors met all four requirements for authorship as outlined by the International Committee of Medical Journal Editors (ICMJE). All authors read and approved the final version of the manuscript.
Author Contributions
Motoyuki Sugai, Akira Yuasa and Jason P Gordon conceptualised and designed the study. Ryan L Miller and Vasileios Vasilopoulos were responsible for data analysis. Hitomi Kurosu and Amer Taie provided expert guidance to that end. Tetsuya Mmatsumoto ensured applicability of the model and analysis to the Japanese clinical setting and provided expert guidance to that end. All authors contributed to interpretation of the results, preparation and review of the manuscript, and approval of the final manuscript for publication.
Disclosures
The authors declare the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: Motoyuki Sugai and Hitomi Kurosu have no conflicts of interest. Akira Yuasa is a full-time employee of Pfizer Japan Inc. Jason P Gordon, Ryan L Miller and Vasileios Vasilopoulos are employees of Health Economics and Outcomes Research Ltd., which received funding from Pfizer Japan Inc. to undertake the research outlined in this study. Amer Taie is a full-time employee of Pfizer R&D UK Limited. Akira Yuasa and Amer Taie hold stocks and stock options from Pfizer Inc. Tetsuya Matsumoto has been on the speakers’ bureau for Pfizer Japan Inc., KYORIN Pharmaceutical Co., Ltd., and MSD K.K.
Compliance with Ethics Guidelines
Ethics approval was not required for this study; the analysis in this article is based on previously publicly available data and does not involve any new studies of human or animal subjects performed by any of the authors.
Data Availability
The anonymised patient data underlying this manuscript are derived from the Medical Data Vision database and the national surveillance program (Japan Nosocomial Infections Surveillance) and cannot be made available by the authors. The datasets generated and/or analysed during the current study are available from the corresponding author on reasonable request. Except for confidential data associated with modelling and data analysis, all data are available from the corresponding author upon reasonable request.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Sugai, M., Yuasa, A., Miller, R.L. et al. An Economic Evaluation Estimating the Clinical and Economic Burden of Increased Vancomycin-Resistant Enterococcus faecium Infection Incidence in Japan. Infect Dis Ther 12, 1695–1713 (2023). https://doi.org/10.1007/s40121-023-00826-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40121-023-00826-w