Abstract
Background
Long-acting injectable (LAI) antipsychotics can reduce relapse, hospitalization, and costs in patients with schizophrenia. However, real-world evidence assessing the impact of treatment with LAIs in Germany is limited.
Objective
To provide updated evidence on the impact of LAI initiation on hospitalization rates and therapy costs.
Methods
Using a mirror-image design, claims data of 850 German patients with schizophrenia who initiated treatment with LAIs during 2013–2015 was retrospectively analyzed. For the included patients, costs and resource utilization were compared for the 12 months before the index date (first initiation of LAI) and the 12 months after the index date. Annual treatment costs, hospitalization rates, ambulatory visits, sick leaves and medical aids were assessed. Two models were used to evaluate hospitalization and its costs. In model 1, hospitalization during the index date (first LAI prescription in 2013–2015) was allocated to the “pre-” time interval, while in model 2 it was neither attributed to the pre- nor to the post-index date. Regression analysis was performed to identify patients who benefited the most in terms of cost reduction from LAI initiation.
Results
Medication costs were significantly higher post-switching to LAI compared with pre-switching period (€3832 vs €799; p < 0.001). In model 1, number of hospitalizations, days hospitalized, and associated costs were significantly lower post-switching compared with pre-switching (2.3 vs 2.6; 59.2 vs 73.4; and €5355 vs €11,908, respectively; all p < 0.001). Similar results were obtained for costs in model 2 (€5355 vs €10,276; p < 0.001). Mean total costs reduced significantly from pre-switching to post-switching period in model 1 (€13,776 vs €10,418; p < 0.001). Patients with characteristics such as higher number of non-psychiatric and psychiatric inpatient stays during the pre-index period (all p < 0.05) benefited the most from cost reduction after LAI initiation.
Conclusion
In this cohort of German patients with schizophrenia, treatment initiation with LAI resulted in reduced hospitalization rates and total costs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Usage of long-acting injectable (LAI) antipsychotics increased the medication costs but reduced the number of hospitalizations, duration of hospitalization, and its costs. This resulted in a net reduction in total costs. |
LAIs should be used more frequently in the treatment of schizophrenia in Germany to reduce the economic burden of schizophrenia. |
1 Introduction
Schizophrenia is a severe mental disorder, characterized by profound disruptions in thinking, language, perception, and the sense of self [1]. According to the Global Burden of Disease 2016 study, the prevalence of schizophrenia in Germany is 0.29% [2]. Schizophrenia adversely affects cognitive performance [3], thus impairing daily activities, work productivity, and social functioning [4], and increasing the economic burden. In 2008, the annual economic burden of schizophrenia for German society was estimated to be €9.6–€13.5 billion [5].
Hospitalization is a driver of healthcare costs, contributing to 43% of the total direct medical costs of treating German patients with schizophrenia [5]. Moreover, relapses in schizophrenia have been shown to increase healthcare resource utilization (HRU) and associated costs as well as reduce the quality of life [6, 7]. Antipsychotic usage can reduce the adverse influence of schizophrenia on a patient’s life. However, poor adherence due to young age, low education level, alcohol and substance abuse, poor cognition, lack of awareness or insight about schizophrenia, and a complex medication regime [8], result in relapse and rehospitalization as well as increased costs [9,10,11]. Therefore, treatment strategies that improve compliance as well as reduce relapse and rehospitalization are critical to successfully manage schizophrenia.
Long-acting injectable (LAI) antipsychotics have been shown to increase medication adherence [12, 13], reduce relapse and rehospitalization [13,14,15], and be more cost effective [16,17,18] compared with oral antipsychotics (OAP). However, in Germany, second-generation antipsychotic LAIs are prescribed less due to their high costs [19]. Although savings in HRU can offset the pharmacy costs of LAI usage, the cost effectiveness could vary based on the drug prices and procedural costs in individual countries. Hospitalization rates and costs among LAI users in Germany have been mostly assessed in clinical trials [20,21,22,23]. A few studies have utilized model-based approaches [16, 17, 24] to estimate the aforementioned outcomes; however, they used pre-2012 data for the model or compared a single LAI to other antipsychotics [25]. The current study utilized a large German claims database in order to provide updated evidence regarding the impact of LAI initiation.
2 Methods
2.1 Data Source
De-identified records were obtained from a claims database provided by Team Gesundheit, Essen, Germany. The database encompasses healthcare resource utilization of approximately 4.2 million insured individuals in Germany and contains detailed electronic records of health insurance claim information on inpatient, outpatient, and prescription drug data at the individual member level. These data originated from different company health insurance funds (Betriebskrankenkassen), which are part of the German statutory health insurance scheme. Membership in the statutory health insurance scheme is compulsory for 87% of the German population. The remaining 13% of the population is privately insured. The database is extensively utilized for health services research [26, 27].
2.2 Study Population
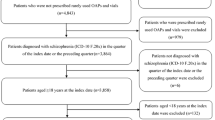
Patients who were diagnosed with schizophrenia according to ICD-10-GM (International Classification of Diseases German Modification, 10th Revision) code F20.x (Supplementary Table 1) from 2012 to 2015 were included in this retrospective cohort study. The diagnosis could have been made in the in-patient setting (primary and secondary diagnosis) or in the out-patient setting (verified diagnosis). Next, all patients who had prescriptions of the pre-defined LAIs in the timeframe from 2013 to 2015 but did not have a preceding LAI prescription documented in the prior 365 days, were identified. The index date was defined as the first prescription of a LAI. The following LAIs were identified: fluphenazine, haloperidol, flupentixol, zuclopenthixol, fluspirilene, olanzapine, risperidone, aripiprazole, and paliperidone. Patients were required to have an ICD-10 F20.x diagnosis in the quarter of index date or in the preceding quarter, be aged ≥ 18 years at the index date and have at least 365 days of continuous enrollment prior to and after the index date. Patients who were first prescribed a LAI in 2012 and those who were identified with certain comorbidities such as attention deficit/hyperactivity disorder (ICD-10 F90.x-F98.x); epilepsy (ICD-10 G09.x, G40.x, G41.x, I69.4, and O99.3); or dementia (ICD-10 F0.x, G30.x, G31.x, G10.x, G20.X, B22.0, and E75.6) in the quarter of the index date or in the preceding quarter were excluded (Fig. 1) as patients with these comorbidities often receive off-label antipsychotic medications [28].
2.3 Covariates
Baseline covariates of interest included age, gender, common comorbidities, and the Charlson Comorbidity Index (CCI) [29]. The CCI included 19 comorbidities as given in Supplementary Table 2 (myocardial infarction, congestive heart failure, peripheral vascular disease, cerebrovascular disease, dementia, chronic pulmonary disease, rheumatic disease, peptic ulcer disease, mild liver disease, diabetes without chronic complication, diabetes with chronic complication, hemiplegia or paraplegia, renal disease, tumors without metastasis, lymphoma, leukemia, moderate or severe liver disease, metastatic solid tumors, and AIDS/HIV) and assigned a weight between 1 and 6 to each comorbidity. Higher CCI indicates a greater morbidity of the patient.
2.4 Outcomes
HRU and costs for the 12 months before and after the index date were compared using a mirror-image design (Fig. 2). The evaluated outcomes included number of hospitalizations, length of stay and associated costs; number of outpatient office visits and associated costs; drug acquisition costs; cost of medical aids; duration of unemployment in months and work status change; and number and duration of sick leave periods and associated costs that were covered by health insurance. In Germany, during an employee’s illness, the agreed salary wage continues to be paid for a period of 42 days by the employer. After that, health insurance covers sick pay for up to 546 days. Patients get up to 90% of their net salary, with a cap of €103.25 per day.
In order to assess the robustness of our results, we followed the method from Spill et al. [30] and tested two models to decipher whether hospitalization of patients during their index LAI prescription should be allocated to the pre- or post-index period. In model 1, a hospitalization during the index date was allocated to the “pre-” time interval, while in model 2 it was neither attributed to the pre- nor to the post-index date.
2.5 Statistical Analysis
This mirror-image study compares outcomes before and after the index date. Means and standard deviations (SD) for continuous variables and counts and percentages for categorical variables were calculated. Differences were tested for statistical significance using Wilcoxon rank tests for non-normally distributed variables. A p value of ≤ 0.05 was considered statistically significant.
Regression analysis with the ordinary least squares method was performed to identify patients who have benefited most in terms of cost reduction from LAI initiation. The model comprised variables such as age; gender; insurance status; HRU during pre-index period (number of different agents, number of psychiatric and non-psychiatric inpatient and outpatient stays, and number of office visits); and whether or not a consulting psychiatric interview was conducted. The analysis was undertaken using SAS software (ver. 9.4; SAS Institute Inc. Cary, NC, USA).
3 Results
3.1 Population Characteristics
Among 23,188 patients diagnosed with schizophrenia, 850 were eligible for the study (Fig. 1). Among the 850 included patients, at index date, 27.1% received paliperidone palmitate), 20.7% received risperidone decanoate, and 15.2% received flupentixol decanoate. The overall mean (SD) age was 45 (15) years with substantial variation across different LAIs. In general, patients on first-generation antipsychotics were older and had more comorbidities than those treated with second-generation antipsychotics. Specifically, patients prescribed olanzapine and haloperidol were the youngest and oldest in the sample, respectively. Overall, 45.4% of the patients were female, with distribution varying from 17.4% for olanzapine to 69.7% for fluspirilene. More than one-third of the overall sample were retired (38.9%), with the distribution of retired patients varying from 24.8% for the paliperidone cohort to 55.0% for the flupentixol cohort. Chronic pulmonary disease (16.9%) and diabetes without chronic complications (12.4%) were among the more common comorbidities. Common addiction-related comorbidities were mental and behavioral disorders due to use of tobacco (24.2%), alcohol (18.5%), and cannabinoids (16.1%) (Table 1). We observed a huge degree of heterogeneity between the different LAIs regarding patient characteristics, making comparisons across different LAIs difficult. Although outcomes are reported on a compound base as well, we want to remind the reader that the design of the study is to assess the effect of treatment initiation with a LAI and not to compare different LAIs with each other.
3.2 Outcomes
3.2.1 Medications and Associated Costs
Overall, the mean number of agents used, antipsychotic costs, and medication costs were significantly higher in the post-index period compared with pre-index (2.3 vs 1.4; €3458 vs €508; and €3832 vs €799, respectively; p < 0.001 for all). The lowest increase in both antipsychotic and medication costs in the post-index period was observed for fluspirilene (up to 1.1-fold) and the highest increase was for olanzapine (up to 17.8-fold, Table 2). Overall, risperidone was the most prescribed agent in both the pre-index (24.1%) and post-index (33.1%) periods, while paliperidone palmitate was the most prescribed LAI in the post-index period (30.8%, Supplementary Table 3).
3.2.2 Hospitalization and Associated Costs
Two models were tested to evaluate hospitalizations and associated costs. In model 1 the overall mean number of all hospitalizations, number of days hospitalized, and costs were significantly lower in the post-index period compared with pre-index (2.3 vs 2.6; 59.2 vs 73.4; and €5355 vs €11,908, respectively; p < 0.001 for all). Similar reductions were observed for psychiatric and non-psychiatric inpatients, although only the paliperidone palmitate group showed significant changes in the latter (p < 0.05). Significant increases were also observed across all the parameters for psychiatric outpatients (p < 0.001 for all, Table 3).
In model 2, overall there were no significant changes in the mean number of all hospitalizations and number of days hospitalized. Costs were significantly lower in the post-index period compared with pre-index (€5355 vs €10,276; p < 0.001). Overall, psychiatric inpatients showed a significant decrease while psychiatric outpatients showed a significant increase (p < 0.001 for all) across parameters. Similar to model 1, only the paliperidone group showed significant changes in non-psychiatric inpatients (p < 0.05, Table 4). Supplementary Table 4 shows the changes in the number of patients with at least one hospitalization.
The decrease in hospitalization was especially triggered by a reduced number of psychiatric inpatient stays. The number of patients with at least one psychiatric inpatient stay was reduced by half from 60.1 to 31.2% in model 1 (Supplementary Table 4). Therefore, the number of days spent hospitalized in a psychiatric institution decreased from a mean of 47 days during pre-index period to 18.5 days during post-index period (Table 3). These results are supported by model 2 where a reduction of 23.4% in patients with at least one psychiatric inpatient stay (Supplementary Table 4) and a reduction of the number of days spent hospitalized by 21.8 days (Table 4).
3.2.3 Ambulatory Healthcare Resource Utilization and Associated Costs
The mean overall number of visits and costs increased significantly from pre-index to post-index period (19.4 vs 26.0 and €581 vs €739; p < 0.001 for all). Differentiating by specialty, a significant increase in the mean number of visits was observed for general practitioners, neurologists and psychiatrists, laboratory medicine, psychological psychotherapists, and unknown (p < 0.05 for all; Table 5). Supplementary Table 5 shows the changes in the number of patients with these visits.
3.2.4 Sick Leave and Sickness Benefits
Overall, the number of patients with sick leave periods and sickness benefit periods decreased from pre-index to post-index (20.6% vs 17.6% and 13.6% vs 11.6%). The overall mean number and duration of sick leave periods also significantly decreased from pre-index to post-index (0.6 vs 0.5 and 20.6 vs 16.9; p < 0.01 for all). However, group-wise significant changes were observed only in the olanzapine, risperidone, and paliperidone groups (p < 0.05 for all). No significant changes were observed in the mean number and duration of sickness benefit periods and sickness benefit costs (Table 6).
3.2.5 Employment, Aids, and Associated Costs
Across the pre- and post-index periods, the overall number of unemployed patients (26.3% vs 27.1%) and mean length of unemployment (2.4 months) were similar. Compared to typical antipsychotic users, a greater number of atypical antipsychotic LAI users were unemployed (11.9% vs 34.5%) and for a longer duration (1.1 vs 3.2 months). Comparison of the number of patients who switched from unemployment to employment and vice versa also showed similar results (Supplementary Table 6).
Overall, fewer patients required aids in the post-index period compared with pre-index (0.6% vs 1.2%). The mean number of aids and associated costs significantly decreased from the pre-index to post-index (p < 0.05 for all; Supplementary Table 7).
3.2.6 Therapies
Overall, the mean number of times biperiden; lorazepam; and psychiatric interview, consulting (single treatment) was used was greater in the post-index period compared with pre-index (0.6 vs 0.4, 0.6 vs 0.4, and 4.7 vs 3.1; p < 0.001 for all). Elaborate diagnostics usage decreased significantly from pre-index to post-index (0.1 vs 0.0, p < 0.001). The overall mean number of times standard and intensive adult treatment was used also decreased significantly from pre-index to post-index (2.7 vs 1.3 and 1.6 vs 0.7, p < 0.001 for all). All the atypical antipsychotics showed a significant decrease compared with less than half of the typical antipsychotics. Similar reductions from pre-index to post-index period was observed for treatment area usage. Supplementary Table 8 shows the above results and changes in the number of patients utilizing the services.
3.2.7 Total Costs
As total costs include the associated costs of hospitalizations, total costs were also affected by the two models. Figure 3 depicts the total costs as a composite of individual costs for models 1 and 2, respectively. In model 1, the overall mean total costs reduced significantly from pre-index to post-index (€13,776 vs €10,418; p < 0.001). Group-wise, significant cost reduction was observed for haloperidol (€12,131 vs €4971; p = 0.009), flupentixol (€11,571 vs €7504; p = 0.014), risperidone (€15,725 vs 12,973; p = 0.049), and paliperidone palmitate (€17,166 vs €13,499; p = 0.001); with haloperidol showing the greatest reduction (2.4-fold). Compared to pre-index costs, the post-index costs increased for fluspirilene users (€3906 vs €4914; p = 0.005). In model 2, only haloperidol showed a significant decrease (€11,534 vs €4971; p = 0.024) while fluspirilene showed a significant increase (€3882 vs €4914; p = 0.004) in total costs.
Total annual costs (€) during the 12-month pre-index interval compared to post-index interval. Two models were used to evaluate hospitalization costs; hence, the other costs show the same value irrespective of the model. Significant differences were observed between pre-index and post-index periods for model 1 total costs (p < 0.001), model 1 and 2 hospitalization costs (p < 0.001 for both), medication and antipsychotics costs (p < 0.001 for both), ambulatory visit costs (p < 0.001), and medical aids costs (p = 0.014)
3.3 Beneficial Characteristics for LAI Initiation
More non-psychiatric (p = 0.008) and psychiatric (p < 0.001) inpatient stays during the pre-index period were present in patients who benefitted the most from cost reduction after LAI initiation (Table 7). The estimated parameter suggests that each stay in a psychiatric hospital before the index date is associated with a post-index date cost reduction of €3304 after LAI initiation.
4 Discussion
This retrospective German claims-based study assessed the impact of LAI initiation among patients with schizophrenia in terms of HRU and costs. Multiple typical and atypical antipsychotic LAIs were evaluated. The number and duration of hospitalizations decreased after LAI initiation during the post-index period. Although LAI initiation increased medication costs, antipsychotic costs, and ambulatory visit costs, the corresponding decrease in hospitalization costs resulted in decreased total costs.
Switching to LAI decreased the number and duration of hospitalizations by 12% and 19%, respectively. The reduction was mainly driven by a decrease in psychiatric inpatient stays. These results are consistent with other mirror-image studies conducted in cohorts of less than 200 patients [30,31,32], a cohort of almost 2000 patients [33], and a meta-analysis of 15 mirror-image studies [34] that showed a significant decrease in hospitalization after patients switched to LAI. Particularly strong evidence was reported in a Spanish 10-year mirror-image study conducted in more than 300 patients that linked LAI administration with significant reductions in number of hospitalized patients and number of hospitalizations due to relapse [35]. Several real-world non-mirror-image studies also reported lower hospitalization on usage of LAIs compared with OAPs [14, 36,37,38,39,40].
Switching to LAIs in the current study resulted in total annual savings of €3358 (model 1) and €1726 (model 2), consequent to the psychiatric inpatient stay-driven reduction in hospitalization and resultant costs. The resultant savings are similar to another German mirror-image study wherein 119 patients were switched from an OAP to a risperidone LAI (RLAI). Here, net savings after a year of switching to RLAI were €3812 [30]. Studies in other populations have also reported savings after LAI initiation. Two Canadian mirror-image studies evaluated the effect of LAI usage for 12 months on healthcare costs. Chawla et al [32] analyzed data of 44 patients in a community center hospital and reported pre-LAI initiation hospital costs of $1,169,600 and post-LAI initiation hospital and LAI costs to be $363,801, resulting in net savings of $18,314 per patient per year. On the other hand, Lachaine et al [33] used the Régie de l’assurance maladie du Québec database to assess data of 1992 patients and reported net savings of CAD11,292 per year. A similar analysis by Peng et al [41] in the USA showed savings of $3228 per patient after 6 months of LAI initiation in 147 patients with schizophrenia. Cost analysis using models also show the benefit of switching to LAIs. Laux et al [17] used a discrete event simulation model to compare the costs of RLAI with oral olanzapine over a 5-year period in Germany, and reported that LAI initiation reduces costs by €6096. Another German study by Zeidler et al [16] estimated the cost effectiveness of paliperidone LAI (PLAI) using a Markov decision analytic model over a 5-year period. Here, the incremental cost-effectiveness ratio (€/gained quality-adjusted life-years) of PLAI was €69,659 and €23,183 compared with oral atypical and oral typical agents, respectively. A budget impact analysis also estimated enormous savings amounting to > 8000 million yen ($68 million) for the Japanese healthcare system after the market introduction of PLAI [42]. These data represent only direct costs; the indirect costs of preventing relapse were not assessed.
Compound-wise analysis reveals that significant overall cost-reductions were associated with haloperidol (€ − 7160), and flupentixol (€ − 4068) among the first-generation antipsychotics; and with risperidone (€ − 2753), and paliperidone palmitate (€ − 3667) among the second-generation antipsychotics. However, patient populations treated with first- and second-generation antipsychotics are different from each other as indicated by lower total pre-index costs. Those pre-index cost differences probably reflect a high degree of heterogeneity in disease severity. Therefore, comparisons across compounds should be treated with caution.
Multiple European studies have associated LAI usage with lower relapses, symptomatic improvement, increased personal recovery, and lower suicidal ideation and suicide attempts [20, 43, 44]. Interestingly, Corigliano et al [44] reported that patients in the early stages of schizophrenia (≤ 5 years of schizophrenia) seemed to benefit more from LAI initiation than chronic patients (> 5 years of schizophrenia). Similar effects were noted by Brown et al [45] on administering PLAI once a month and once every 3 months to patients with various durations of schizophrenia. Early LAI initiation was also associated with lower hospitalization and costs in the USA [46]. Although we could not observe these effects as the data were not stratified based on disease duration, future studies could certainly assess this outcome.
In current clinical practice, LAIs are often reserved for non-adherent patients, and psychiatrists themselves avoid prescribing LAIs to patients with first-episode schizophrenia [47]. While some patients prefer LAIs over OAPs due to increased relapse prevention [48] as well as better quality of life [49], others report being coerced into undergoing treatment with LAIs [50]; with pain at the injection site, misconceptions regarding LAIs, and lack of awareness also being reported as reasons for low LAI uptake [51, 52]. Bridges et al [53] recently quantified the effect of schizophrenia treatment goals on a patient’s treatment plan. In this study, patients who had functional goals such as improving social relationships, increasing interest in work, and experiencing full range of emotions were more likely to prefer LAIs over OAPs. While the current study along with existing evidence suggests reduced HRU and financial burden on LAI initiation, successful treatment of schizophrenia also depends on the patient’s comfort. Therefore, we suggest that treatment decisions should incorporate patient preference and treatment goals in addition to other clinical factors and costs.
4.1 Limitations
There are certain limitations in the current study. Since claims data were used to identify a diagnosis of schizophrenia, it is possible that misclassification or coding errors could have occurred. Moreover, claims data insufficiently reflect the severity of a disease and other influencing circumstances. Claims data were also used to identify antipsychotic utilization; however, the data only support receipt and payment and actual consumption is only presumed. Nevertheless, since LAIs are generally administered by a healthcare professional, a claim for a LAI provides confidence that the effective dose was administered to the patient. Finally, indirect costs were excluded from the analyses as this study concentrated on the payer’s perspective. However, inclusion of indirect costs would most likely result in lower total costs for LAIs compared with OAPs as LAIs cause fewer relapses and would have probably reduced loss of productivity more than OAPs.
5 Conclusion
Patients with schizophrenia who switched to treatment with LAI antipsychotics had fewer hospitalizations and clinic visits compared with their treatment period before the switch. Although treatment with LAIs increases the medication and ambulatory visit costs, the corresponding reduction in hospitalization costs results in overall total cost reduction. The average total costs considering all the LAIs together reduced by 24.4% and 14.2% as per models 1 and 2, respectively. This is one of the first claims database analysis for Germany that studied the impact of treatment initiation with LAIs on costs in a large cohort of patients with schizophrenia. In line with results from other countries, we found significant savings from a health insurance point of view that calls for a wider use of LAIs in Germany. Since treatment of schizophrenia is a long-term engagement, future research could perform an extended follow-up and report the impact of treatment with LAIs.
References
Schizophrenia. World Health Organization. https://www.who.int/mental_health/management/schizophrenia/en/. Accessed 16 Aug 2019.
Global Burden of Disease 2016. Institute for Health Metrics and Evaluation, Seattle, WA, USA. 2016. https://gbd2016.healthdata.org/gbd-search/. Accessed 16 Aug 2019.
Fioravanti M, Bianchi V, Cinti ME. Cognitive deficits in schizophrenia: an updated metanalysis of the scientific evidence. BMC Psychiatry. 2012;12:64. https://doi.org/10.1186/1471-244X-12-64.
Kitchen H, Rofail D, Heron L, Sacco P. Cognitive impairment associated with schizophrenia: a review of the humanistic burden. Adv Ther. 2012;29(2):148–62. https://doi.org/10.1007/s12325-012-0001-4.
Frey S. The economic burden of schizophrenia in Germany: a population-based retrospective cohort study using genetic matching. Eur Psychiatry. 2014;29(8):479–89. https://doi.org/10.1016/j.eurpsy.2014.04.003.
Lafeuille MH, Gravel J, Lefebvre P, Fastenau J, Muser E, Doshi D, et al. Patterns of relapse and associated cost burden in schizophrenia patients receiving atypical antipsychotics. J Med Econ. 2013;16(11):1290–9. https://doi.org/10.3111/13696998.2013.841705.
Almond S, Knapp M, Francois C, Toumi M, Brugha T. Relapse in schizophrenia: costs, clinical outcomes and quality of life. Br J Psychiatry. 2004;184:346–51. https://doi.org/10.1192/bjp.184.4.346.
Ljungdalh PM. Non-adherence to pharmacological treatment in schizophrenia and schizophrenia spectrum disorders—an updated systematic literature review. Eur J Psychiatry. 2017;31(4):172–86.
Morken G, Widen JH, Grawe RW. Non-adherence to antipsychotic medication, relapse and rehospitalisation in recent-onset schizophrenia. BMC Psychiatry. 2008;8:32. https://doi.org/10.1186/1471-244X-8-32.
Knapp M, King D, Pugner K, Lapuerta P. Non-adherence to antipsychotic medication regimens: associations with resource use and costs. Br J Psychiatry. 2004;184:509–16. https://doi.org/10.1192/bjp.184.6.509.
Novick D, Haro JM, Suarez D, Perez V, Dittmann RW, Haddad PM. Predictors and clinical consequences of non-adherence with antipsychotic medication in the outpatient treatment of schizophrenia. Psychiatry Res. 2010;176(2–3):109–13. https://doi.org/10.1016/j.psychres.2009.05.004.
Greene M, Yan T, Chang E, Hartry A, Touya M, Broder MS. Medication adherence and discontinuation of long-acting injectable versus oral antipsychotics in patients with schizophrenia or bipolar disorder. J Med Econ. 2018;21(2):127–34. https://doi.org/10.1080/13696998.2017.1379412.
Marcus SC, Zummo J, Pettit AR, Stoddard J, Doshi JA. Antipsychotic adherence and rehospitalization in schizophrenia patients receiving oral versus long-acting injectable antipsychotics following hospital discharge. J Manag Care Spec Pharm. 2015;21(9):754–68. https://doi.org/10.18553/jmcp.2015.21.9.754.
Tiihonen J, Mittendorfer-Rutz E, Majak M, Mehtala J, Hoti F, Jedenius E, et al. Real-world effectiveness of antipsychotic treatments in a nationwide cohort of 29823 patients with schizophrenia. JAMA Psychiatry. 2017;74(7):686–93. https://doi.org/10.1001/jamapsychiatry.2017.1322.
Taipale H, Mehtala J, Tanskanen A, Tiihonen J. Comparative effectiveness of antipsychotic drugs for rehospitalization in schizophrenia-a nationwide study with 20-year follow-up. Schizophr Bull. 2018;44(6):1381–7. https://doi.org/10.1093/schbul/sbx176.
Zeidler J, Mahlich J, Greiner W, Heres S. Cost effectiveness of paliperidone palmitate for the treatment of schizophrenia in Germany. Appl Health Econ Health Policy. 2013;11(5):509–21. https://doi.org/10.1007/s40258-013-0050-0.
Laux G, Heeg B, van Hout BA, Mehnert A. Costs and effects of long-acting risperidone compared with oral atypical and conventional depot formulations in Germany. Pharmacoeconomics. 2005;23(Suppl 1):49–61. https://doi.org/10.2165/00019053-200523001-00005.
Achilla E, McCrone P. The cost effectiveness of long-acting/extended-release antipsychotics for the treatment of schizophrenia: a systematic review of economic evaluations. Appl Health Econ Health Policy. 2013;11(2):95–106. https://doi.org/10.1007/s40258-013-0016-2.
Heres S, Hamann J, Kissling W, Leucht S. Attitudes of psychiatrists toward antipsychotic depot medication. J Clin Psychiatry. 2006;67(12):1948–53.
Schreiner A, Aadamsoo K, Altamura AC, Franco M, Gorwood P, Neznanov NG, et al. Paliperidone palmitate versus oral antipsychotics in recently diagnosed schizophrenia. Schizophr Res. 2015;169(1–3):393–9. https://doi.org/10.1016/j.schres.2015.08.015.
Schreiner A, Svensson A, Wapenaar R, Cherubin P, Princet P, Serazetdinova L, et al. Long-acting injectable risperidone and oral antipsychotics in patients with schizophrenia: results from a prospective, 1-year, non-interventional study (InORS). World J Biol Psychiatry. 2014;15(7):534–45. https://doi.org/10.3109/15622975.2014.902990.
Gaebel W, Schreiner A, Bergmans P, de Arce R, Rouillon F, Cordes J, et al. Relapse prevention in schizophrenia and schizoaffective disorder with risperidone long-acting injectable vs quetiapine: results of a long-term, open-label, randomized clinical trial. Neuropsychopharmacology. 2010;35(12):2367–77. https://doi.org/10.1038/npp.2010.111.
Schmauss M, Sacchetti E, Kahn JP, Medori R. Efficacy and safety of risperidone long-acting injectable in stable psychotic patients previously treated with oral risperidone. Int Clin Psychopharmacol. 2007;22(2):85–92. https://doi.org/10.1097/YIC.0b013e3280119ddb.
Frey S, Linder R, Juckel G, Stargardt T. Cost-effectiveness of long-acting injectable risperidone versus flupentixol decanoate in the treatment of schizophrenia: a Markov model parameterized using administrative data. Eur J Health Econ. 2014;15(2):133–42. https://doi.org/10.1007/s10198-013-0460-9.
Potempa C, Rychlik R. Hospitalization rates and resource utilization of schizophrenic patients switched from oral antipsychotics to aripiprazole-depot in Germany. Health Econ Rev. 2018;8(1):30. https://doi.org/10.1186/s13561-018-0215-5.
Clouth J, Liepa AM, Moeser G, Friedel H, Bernzen M, Trojan J, et al. Hepatocellular carcinoma after prior sorafenib treatment: incidence, healthcare utilisation and costs from German statutory health insurance claims data. Health Econ Rev. 2018;8(1):18. https://doi.org/10.1186/s13561-018-0199-1.
Mahlich J, Alba A, Hadad LE, Leisten MK, Peitsch WK. Drug survival of biological therapies for psoriasis treatment in germany and associated costs: a retrospective claims database analysis. Adv Ther. 2019;36(7):1684–99. https://doi.org/10.1007/s12325-019-00969-8.
Haw C, Stubbs J. Off-label use of antipsychotics: are we mad? Expert Opin Drug Saf. 2007;6(5):533–45. https://doi.org/10.1517/14740338.6.5.533.
Charlson ME, Pompei P, Ales KL, MacKenzie CR. A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 1987;40(5):373–83.
Spill B, Konoppa S, Kissling W, Maino K, Messer T, Heres S. Long-term observation of patients successfully switched to risperidone long-acting injectable: a retrospective, naturalistic 18-month mirror-image study of hospitalization rates and therapy costs. Int J Psychiatry Clin Pract. 2010;14(1):53–62. https://doi.org/10.3109/13651500903311821.
Willis M, Svensson M, Lothgren M, Eriksson B, Berntsson A, Persson U. The impact on schizophrenia-related hospital utilization and costs of switching to long-acting risperidone injections in Sweden. Eur J Health Econ. 2010;11(6):585–94. https://doi.org/10.1007/s10198-009-0215-9.
Chawla K, Bell M, Chawla B. Long acting injectable versus oral antipsychotics in reducing hospitalization outcomes in schizophrenia: a mirror-image study. Int J Emerg Ment Health. 2017;19(4):377. https://doi.org/10.4172/1522-4821.1000377.
Lachaine J, Lapierre ME, Abdalla N, Rouleau A, Stip E. Impact of switching to long-acting injectable antipsychotics on health services use in the treatment of schizophrenia. Can J Psychiatry. 2015;60(3 Suppl 2):S40–7.
Kishimoto T, Nitta M, Borenstein M, Kane JM, Correll CU. Long-acting injectable versus oral antipsychotics in schizophrenia: a systematic review and meta-analysis of mirror-image studies. J Clin Psychiatry. 2013;74(10):957–65. https://doi.org/10.4088/JCP.13r08440.
Diaz-Fernandez S, Frias-Ortiz DF, Fernandez-Miranda JJ. Mirror image study (10 years of follow-up and 10 of standard pre-treatment) of psychiatric hospitalizations of patients with severe schizophrenia treated in a community-based, case-managed programme. Rev Psiquiatr Salud Ment. 2019. https://doi.org/10.1016/j.rpsm.2019.04.004.
Cheung S, Hamuro Y, Mahlich J, Nakahara T, Sruamsiri R, Tsukazawa S. Drug utilization of Japanese patients diagnosed with schizophrenia: an administrative database analysis. Clin Drug Investig. 2017;37(6):559–69. https://doi.org/10.1007/s40261-017-0517-0.
Tiihonen J, Haukka J, Taylor M, Haddad PM, Patel MX, Korhonen P. A nationwide cohort study of oral and depot antipsychotics after first hospitalization for schizophrenia. Am J Psychiatry. 2011;168(6):603–9. https://doi.org/10.1176/appi.ajp.2011.10081224.
Grimaldi-Bensouda L, Rouillon F, Astruc B, Rossignol M, Benichou J, Falissard B, et al. Does long-acting injectable risperidone make a difference to the real-life treatment of schizophrenia? Results of the Cohort for the General study of Schizophrenia (CGS). Schizophr Res. 2012;134(2–3):187–94. https://doi.org/10.1016/j.schres.2011.10.022.
Olivares JM, Rodriguez-Morales A, Diels J, Povey M, Jacobs A, Zhao Z, et al. Long-term outcomes in patients with schizophrenia treated with risperidone long-acting injection or oral antipsychotics in Spain: results from the electronic Schizophrenia Treatment Adherence Registry (e-STAR). Eur Psychiatry. 2009;24(5):287–96. https://doi.org/10.1016/j.eurpsy.2008.12.002.
Stip E, Lachaine J. Real-world effectiveness of long-acting antipsychotic treatments in a nationwide cohort of 3957 patients with schizophrenia, schizoaffective disorder and other diagnoses in Quebec. Ther Adv Psychopharmacol. 2018;8(11):287–301. https://doi.org/10.1177/2045125318782694.
Peng X, Ascher-Svanum H, Faries D, Conley RR, Schuh KJ. Decline in hospitalization risk and health care cost after initiation of depot antipsychotics in the treatment of schizophrenia. Clinicoecon Outcomes Res. 2011;3:9–14. https://doi.org/10.2147/CEOR.S16061.
Mahlich J, Nishi M, Saito Y. Modeling the budget impact of long-acting injectable paliperidone palmitate in the treatment of schizophrenia in Japan. Clinicoecon Outcomes Res. 2015;7:267–72. https://doi.org/10.2147/CEOR.S85514.
Diaz-Fernandez S, Frias-Ortiz DF, Fernandez-Miranda JJ. Suicide attempts in people with schizophrenia before and after participating in an intensive case managed community program: a 20-year follow-up. Psychiatry Res. 2019. https://doi.org/10.1016/j.psychres.2019.112479.
Corigliano V, Comparelli A, Mancinelli I, Montalbani B, Lamis DA, De Carolis A, et al. Long-acting injectable second-generation antipsychotics improve negative symptoms and suicidal ideation in recent diagnosed schizophrenia patients: a 1-year follow-up pilot study. Schizophr Res Treatment. 2018;2018:4834135. https://doi.org/10.1155/2018/4834135.
Brown B, Turkoz I, Mancevski B, Mathews M. Evaluation of paliperidone palmitate long-acting injectable antipsychotic therapy as an early treatment option in patients with schizophrenia. Early Interv Psychiatry. 2019. https://doi.org/10.1111/eip.12868.
Munday J, Greene M, Chang E, Hartry A, Yan T, Broder MS. Early initiation of long-acting injectable antipsychotic treatment is associated with lower hospitalization rates and healthcare costs in patients with schizophrenia: real-world evidence from US claims data. Curr Med Res Opin. 2019;35(7):1231–9. https://doi.org/10.1080/03007995.2019.1571295.
Altamura AC, Aguglia E, Bassi M, Bogetto F, Cappellari L, De Giorgi S, et al. Rethinking the role of long-acting atypical antipsychotics in the community setting. Int Clin Psychopharmacol. 2012;27(6):336–49. https://doi.org/10.1097/YIC.0b013e328357727a.
Heres S, Schmitz FS, Leucht S, Pajonk FG. The attitude of patients towards antipsychotic depot treatment. Int Clin Psychopharmacol. 2007;22(5):275–82. https://doi.org/10.1097/YIC.0b013e3280c28424.
Pietrini F, D’Anna G, Tatini L, Talamba GA, Andrisano C, Calderani E, et al. Changes in attitude towards LAI antipsychotic maintenance treatment: a two-year follow-up study. Eur Psychiatry. 2018;53:58–65. https://doi.org/10.1016/j.eurpsy.2018.06.002.
Patel MX, de Zoysa N, Bernadt M, Bindman J, David AS. Are depot antipsychotics more coercive than tablets? The patient’s perspective. J Psychopharmacol. 2010;24(10):1483–9. https://doi.org/10.1177/0269881109103133.
Patel MX, De Zoysa N, Bernadt M, David A. Depot and oral antipsychotics: patient preferences and attitudes are not the same thing. J Psychopharmacol. 2009;23(7):789–96. https://doi.org/10.1177/0269881108092124.
Das AK, Malik A, Haddad PM. A qualitative study of the attitudes of patients in an early intervention service towards antipsychotic long-acting injections. Ther Adv Psychopharmacol. 2014;4(5):179–85. https://doi.org/10.1177/2045125314542098.
Bridges JF, Beusterien K, Heres S, Such P, Sanchez-Covisa J, Nylander AG, et al. Quantifying the treatment goals of people recently diagnosed with schizophrenia using best-worst scaling. Patient Prefer Adherence. 2018;12:63–70. https://doi.org/10.2147/PPA.S152870.
Acknowledgements
Open Access funding provided by Projekt DEAL. We thank Leo J. Philip Tharappel from Siro Clinpharm Pvt Ltd. For providing medical writing and editorial assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The study was funded by Janssen-Cilag, Neuss, Germany.
Conflict of interest
Jörg Mahlich, Kerstin Olbrich, and Antonie Wimmer are employees of Janssen-Cilag. Adrian Wilk is an employee of Team Gesundheit GmbH which received funding for this study from Janssen-Cilag. Claus Wolff-Menzler has no conflict of interest to declare.
Ethics approval
Not required as this study used anonymized German claims data.
Informed consent
Not applicable.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Mahlich, J., Olbrich, K., Wilk, A. et al. Hospitalization Rates and Therapy Costs of German Schizophrenia Patients Who are Initiated on Long-Acting Injectable Medication: A Mirror-Image Study. Clin Drug Investig 40, 355–375 (2020). https://doi.org/10.1007/s40261-020-00900-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40261-020-00900-y