Abstract
Background
There has been considerable focus on the use of psychotropic agents in people living with dementia in long-term care. However, psychotropic use often commences well before transitioning to long-term care.
Objectives
To synthesize the available literature to identify factors associated with psychotropic medication use in people living with dementia in the community.
Methods
This PROSPERO-registered review reports findings from a comprehensive search of Embase, PsycINFO, and PubMed (including MEDLINE) databases according to predefined inclusion and exclusion criteria (2010–2022). Inclusion criteria were original prospective or retrospective design research papers enrolling people diagnosed with dementia utilizing a psychotropic medication and living at home. Quality and risk of bias was assessed Newcastle–Ottawa Quality Assessment Scale. The last search was conducted in November 2022. Thematic analysis was used to synthesize the emergent factors identified, and a meta-analysis was undertaken on suitable data.
Results
The search identified 619 articles. After review and exclusions, 39 articles were included for synthesis, including 1,338,737 people. The majority of papers (67%) were rated as low risk of bias and corresponding good quality. Thematic analysis suggested associations between psychotropic prescribing and patient and environmental factors, with little data concerning carer and prescriber factors. Such factors included age (< 75 years, > 90 years), sex, more advanced functional decline, and living alone. Meta-analysis identified significant associations between psychotropic use and respite (temporary full-time care or hospitalization) and comorbid psychiatric illness.
Conclusions
While it is clear from this review that there remains a significant lack of clarity as to the reasons why these medications are being utilized in this population, this review provides greater insight and understanding into the context of psychotropic use. The study has highlighted an opportunity for further targeted research to be conducted and provides a much-needed context for this to occur.
PROSPERO Registration Number
CRD42021286322.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Psychotropic medication is utilized frequently for people with dementia living in the community. |
Factors identified in this review provide opportunities for targeted intervention and further research. |
Practical strategies for reflecting on psychotropic prescribing and optimizing psychotropic use are suggested. |
1 Introduction
Globally, the number of people living with dementia is predicted to increase from approximately 50 million in 2019 to 130 million in 2050 [1]. Approximately 60–70% of people with dementia live at home, while the remainder receives formal residential care of various types [2]. Given these exponentially increasing rates and rising healthcare costs, it is essential to understand how and what drives the management of this population’s needs (physical, social, psychological, and behavioral) and whether resources and approaches are effective, appropriate, and cost efficient. This is all the more salient given the failings of aged care systems worldwide in meeting such needs [3,4,5].
Psychotropic medications, defined for the purpose of this paper as antipsychotics, antidepressants, anxiolytic/hypnotics (benzodiazepines and Z-drugs), anticonvulsants, and opioid medications, are frequently prescribed to people living with dementia [6]. Emerging literature has demonstrated that psychotropic prescribing in this population often begins in the community and is continued upon transition to long-term care, although the reasons for this are unclear [7].
Prevalence figures on psychotropic use in people living with dementia in the community range from 10 to over 50%, depending on the class of medication being used [8, 9]. These figures are alarmingly high given the burden of potential side effects and increased mortality risk associated with these medications in the absence of significant benefits [6, 10]. Hitherto, there has been extensive focus on psychotropic medication use in long-term care settings to implement quality measures and minimize inappropriate use in this setting [11,12,13]. However, there has been little comparative research into psychotropic medication use in people living with dementia in the community.
Critical work is being done in parallel to understand changed behaviors (also referred to as behavioral and psychological symptoms of dementia), the target symptoms for psychotropic treatment [14, 15]. Understanding the context of psychotropic use in this population is essential to improving their care and outcomes. By understanding the context of psychotropic use, further in-depth qualitative studies and targeted programs can be implemented to develop quality prescribing initiatives in this setting.
2 Objective
The primary objective of this systematic review and meta-analysis was to address the following research question: what factors (outcome) are associated with the use of psychotropic medication (intervention) in people living with dementia (population) in the community (context)?
3 Methods
3.1 Search Strategy
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines were used [16]. The review protocol was submitted to PROSPERO and published online in December 2021 (PROSPERO registration number: CRD42021286322).
An initial search of the Embase, PsycINFO, and PubMed (including MEDLINE) databases was conducted by two raters (KL and CP) in September 2021 using medical subject headings or free text (keywords) to reflect different indexing in databases. A final search was done in November 2022 for any new material. Nonpeer-reviewed literature, abstracts, gray literature, and opinion pieces were excluded because of the potential risk of bias and variable quality [17].
The search dates were January 2010 to November 2022, with 2010 chosen to avoid publications that focused on the effects of legislative changes concerning psychotropics and dementia, most of which occurred before 2009. The search strategy can be accessed in the supplementary material (S1).
Hand searches of the included publications’ bibliographies were also conducted to identify additional relevant records. All search results were entered into EndNote version 20.2.1 (Clarivate, PA) to identify duplicates and organize material.
Inclusion criteria were retrospective or prospective research papers involving people living with dementia in the community using psychotropic medications, compared with people living with dementia in the community not using psychotropic medications. Exclusion criteria were reviews, commentaries, abstracts, and gray literature. Studies that only reported prevalence without associated factors or those primarily designed to investigate legislative changes in prescribing were excluded. Populations in which people with or without dementia were included, where subset analysis was not possible for the population with dementia only. Populations in which a transition from community living to full-time care occurred were included if the community-specific period could be evaluated separately. An English language criterion was applied for the first search; however, this was expanded to include all language papers, which did not identify any additional papers missed in the subsequent search. Papers involving opioid use were identified on the basis of a clear exclusion for opioid appropriate pain conditions. Two people (KL and CP) were involved in defining the search terms. The database search was conducted by one individual (KL) and the input of an experienced research librarian.
Two raters (KL and CP) independently reviewed papers and discussed all papers identified for exclusion. The first round of reviews was conducted on the titles and abstracts. Any papers that were the subject of a disagreement between raters regarding exclusion were then reviewed in full in the next review round and discussed until a consensus was reached or the third reviewer (AW) provided adjudication. In the event of multiple reports relating to the same population and outcome, the most recently published report was selected. Data from the selected studies were extracted systematically into an Excel spreadsheet by KL. The data collected included generalized descriptive data, conceptual data subject to thematic analysis, and quantitative data for purposes of the meta-analysis. The data and spreadsheet were reviewed and independently verified by CP for accuracy.
3.2 Quality and Risk of Bias Assessment
Formal quality and risk of bias assessments were conducted independently by two researchers using a tool developed by Hoy et al. [18] for studies involving prevalence and the Newcastle–Ottawa Quality Assessment Scale for case-control and cohort studies [19]. A low risk of bias was indicated by the use of validated diagnostic criteria for dementia and consistent data input, such as mandatory population registries, clinical trials, or medical administration databases. Studies assessed as having a high risk of bias used less vigorous diagnostic criteria for dementia, used specific population groups, or had high dropout rates or poor comparability leading to less generalizability of results.
3.3 Data Synthesis and Analysis
Data from the selected studies were extracted systematically including country, sample size, participant demographics, diagnosis, objectives, outcome measures, quality, and bias rating.
Data were synthesized inductively, using thematic analysis to capture and interrogate data that were not amenable to meta-analysis. Thematic analysis is a validated and extensively utilized tool for the synthesis of data obtained from systematic reviews, particularly to synthesize data not amenable to meta-analyses. [20] A standard thematic analysis approach was adopted [20, 21]. Specifically, the reports/findings of all identified papers were examined and subjected to “free” or “open coding” whereby discrete concepts or patterns in relation to psychotropic prescribing were identified from the papers. These codes were then organized and grouped into “axial codes” by looking for similarities and differences between codes. Finally, higher order, abstract, or analytical themes were identified. This process was undertaken independently by a first coder (KL), followed by a second coder (CP), and then consensually.
Comprehensive Meta-Analysis Software version 2.2.027 (Biostat, NJ) was used to calculate raw data to event rates and pooled, adjusted event rates for different modifier subanalyses. Variables collected included psychotropic drug class, participant demographics, use of/no use of psychotropic medication by class, allowance for temporary respite care, and psychiatric comorbidity. A random-effects model was chosen over a fixed-effects model because the rates would likely vary between populations of different ethnicities, nationalities, and respite allowances with different mixes of psychiatric and dementia diagnoses, sex ratios, care levels, and health needs. Types of temporary respite accommodation outside the usual home were combined for analysis given the small numbers and overarching implications of any out of home care for psychotropic prescribing. In the event of missing data, we excluded the paper for meta-analytic purposes and therefore no extrapolation was included. Meta-analyses were based on separate psychotropic classes because it was not possible to cross-reference incidences of multiple or previous drug use, as most studies were cross-sectional and observational. Within-group heterogeneity was assessed using the I2 statistic, and between-group heterogeneity was assessed using the Q-value statistic. Potential publication bias was assessed using Egger et al.’s regression intercept test [22]. Duval and Tweedie’s trim and fill method was used to examine and impute possible missing samples [23]. Data were analyzed using IBM SPSS Statistics 25.0 for descriptive statistics, and forest plots were produced using Comprehensive Meta-Analysis Software. Relevant authors were contacted via email to obtain or augment data; however, the additional data obtained were insufficient for the purpose of inclusion in the meta-analysis.
4 Results
4.1 Search Findings
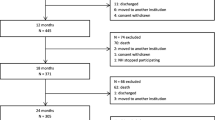
A PRISMA flow diagram of the study selection process is shown in Fig. 1. A total of 39 studies were included after inclusion and exclusion criteria were applied.
The study characteristics, key findings, and risk of bias are summarized in Table 1.
The sample size varied from 173 to 1,169,894, with a total sample size of 1,338,737 people living with dementia in the community. Of the studies, 17 included various analyses from a single population cohort, the Finnish Medication and Alzheimer’s disease (MEDALZ) study database [24]. The MEDALZ study population was only counted once in the total subject sample of this review. However, each MEDALZ study cohort paper investigated a different outcome within this cohort and was included individually in the synthesis. Three large US studies based on Medicare administrative data were included; however, the population numbers were only included once to minimize the risk of over-inflation.
The country of origin of the studies included Finland (n = 18), the USA (n = 11), Canada (n = 3), Germany (n = 2), Italy (n = 1), Norway (n = 1), Sweden (n = 1), the Netherlands (n = 1), and France (n = 1). The participants’ ages ranged from 37 to 100 years. One study specifically focused on people with younger onset dementia (ages < 65 years) [25], and one specifically on males [26].
The majority (n = 26; 67%) of the studies were rated as having a low risk of bias and corresponding good quality, and 13 (33%) studies were rated as having a moderate risk and fair quality. None were rated as having a high risk of bias or low quality. Agreement between independent assessors was high, with 90% agreement across the studies. All studies used quantitative or mixed methodologies. Five were prospective cohort studies, 29 were retrospective cohort studies, four were cross-sectional studies, and one was a nested case-control study.
4.2 Thematic Analysis and Synthesis
Following an inductive process and thematic analysis, factors common to the use of all psychotropic medications and factors associated with using specific classes of psychotropic medications were identified.
The thematic trends that emerged from the analysis are summarized for both psychotropic medications in general and the different classes of psychotropic medications in Fig. 2. When combined, these general and class-specific factors were grouped into higher-order themes: (1) patient related, (2) illness related, (3) environmental, (4) carer related, and (5) prescriber related or prescribing trends. Thematic analysis did not identify differences in outcome themes when studies were grouped according to type (retrospective/prospective longitudinal cohort or cross-sectional).
4.2.1 Theme 1: Patient-Related Factors
4.2.1.1 Sex
Women were more commonly prescribed antidepressants, anxiolytics/hypnotics, and opioids than men, who received more antipsychotic and anticonvulsant medications. Six of the seven papers reporting on anxiolytic/hypnotic medications and sex found a significantly greater percentage of females prescribed anxiolytics/hypnotics than males [8, 27,28,29,30,31]. The majority of these papers (5/6) were retrospective longitudinal cohort studies of good quality and low risk of bias. One paper [25], a prospective longitudinal cohort study of good quality and low risk of bias, found no sex difference in their population of young onset people with dementia. Of the four papers that reported the use of opioids, all identified a significant difference in favor of females [8, 27, 28, 32]. Three out of the four studies were rated as good quality and low risk of bias. Eight of the ten papers that reported sex and antidepressant use found greater female use of antidepressants [8, 27, 30, 33,34,35,36,37]. Seven of these were longitudinal cohort studies (six retrospective and one prospective) and one was a cross-sectional analysis of a prospective cohort). Five of the eight studies were good quality and low risk of bias. The two remaining papers found no difference between males and females [25, 38]. Of these two papers, one represented a unique younger onset dementia population [25], while the other investigated carer and care recipient factors rather than sex, which may have influenced the outcomes of this finding [38]. Both were of good quality and low risk of bias. One was a prospective longitudinal cohort, the other was a cross-sectional analysis of a retrospective cohort. Antipsychotics were utilized more frequently in males compared with females in six papers [27, 36, 39,40,41,42]. Of these two were retrospective cross-sectional analyses, two were prospective longitudinal cohorts, and two were retrospective longitudinal cohorts. Two papers, both retrospective longitudinal cohorts, reported greater use in females [8, 43] and one, a retrospective longitudinal cohort, identified that females were prescribed antipsychotics more commonly prior to diagnosis compared with males [44]. The use of anticonvulsant drugs was reported in one study, where males used anticonvulsants significantly more than females (p < 0.001) [45].
4.2.1.2 Ethnicity
There was sparse data relating to ethnicity. One paper [46] investigated this as the primary objective, identifying that anxiolytic use was more common in African American people with dementia, while antipsychotics were used more frequently in non-Hispanic white people. No consistent trend was identified in the other available papers reporting on ethnicity as secondary outcomes [8, 38, 42].
4.2.1.3 Age
There were 20 papers available for synthesizing age and psychotropic use. There was increased use of psychotropics in older individuals (> 80 years) before dementia diagnosis and increased association with psychotropic use in younger individuals (< 75 years) immediately following diagnosis [9, 28, 44]. All three of these studies were of good quality and low risk of bias. A second trend was identified for “very old” people (defined in the paper as > 90 years) in whom the use of psychotropics appeared to be higher than those < 90 years old [47]. Younger individuals (< 75 years) received more antidepressant, antipsychotic, and anticonvulsant medications [8, 37, 42, 48], while older individuals were prescribed more benzodiazepines and opioids [27, 28, 32, 49]. There was a consistent trend for younger individuals to have more psychotropic polypharmacy [9, 50, 51]. These findings were consistent across both the longitudinal and cross-sectional studies.
4.2.1.4 Psychiatric Comorbidity
The use of psychotropic polypharmacy was significantly associated with having a psychiatric history in four papers [9, 28, 50, 51]. Although there was no significant association between a history of mental disorders and antipsychotic use in Eichler et al.’s study, sample numbers were low, with only 140 participants included in the factor analysis, of whom 22 were on antipsychotic medication [52].
A history of depression and antidepressant use was associated with increased use of antipsychotics [9, 40, 41, 44, 50, 51, 53, 54]. The use of benzodiazepines and/or opioids was significantly associated with a history of prior substance abuse [28, 29, 32].
Highly significant associations with prior diagnoses of psychiatric illness (schizophrenia, bipolar disorder, or depression) were also demonstrated for long-term benzodiazepine use (p < 0.0001) [31] and high-dose antipsychotic use (p < 0.0001) [55]. Anticonvulsant use was also significantly associated with depression, schizophrenia, and bipolar disorders (p < 0.001 for all), although only formally investigated in one study [45].
4.2.1.5 Pain
Only two studies specifically investigated pain in relation to psychotropic use. Grace et al. [38] reported a significant association between pain and antipsychotic use. Hamina et al. [56] identified that opioid initiation triggered an immediate increase in benzodiazepine and antipsychotic use, with a nonsignificant decrease in the use of these agents up to 6 months post-opioid initiation.
4.2.2 Theme 2: Illness-Related Factors
4.2.2.1 Severity of Dementia
Antipsychotic use did not increase in parallel with the severity of cognitive impairment. Antipsychotic use was significantly associated with moderately severe dementia (as opposed to mild or severe) in all three papers that reported on this [48, 52, 57]. One paper found no association between cognition (measured by the Mini-Mental State Examination) and antipsychotic use; however, Mini-Mental State Examination scores were not categorized as mild, moderate, or severe, and a cumulative value was used, which may have influenced the findings [58]. Two papers reported a significant association between antipsychotic use and greater severity on the Clinical Dementia Rating sum of boxes scale [42, 58]. No trend was identified between cognitive functioning and other classes of psychotropics. All the papers above were rated as moderate risk of bias apart from one [48], which was rated a low risk of bias. Three of the studies were cross-sectional in nature [42, 48, 52] and two were longitudinal cohorts of up to 1 year follow-up [57, 58].
4.2.2.2 Functional Ability/Activities of Daily Living Independence
There was a significant association between antipsychotic use and greater impairment in activities of daily living across the six studies that reported on this [38, 40, 41, 48, 52, 58].
4.2.2.3 Neuropsychiatric Symptoms
Antipsychotic use was most closely associated with Neuropsychiatric Inventory (NPI) scores. Six of eight studies demonstrated a significant association between increasing NPI scores and antipsychotic use [26, 42, 48, 54, 57, 59], while two did not [25, 58]. No consistent trend was identified regarding NPI scores and antidepressant or anxiolytic/hypnotic medication use.
4.2.3 Theme 3: Environmental Factors
4.2.3.1 Socioeconomic
Lower socioeconomic status was generally associated with the use of benzodiazepines, antipsychotics, and opioids [8, 28, 32, 40,41,42, 45, 48, 50]. Bargagli et al. [39] found that lower socioeconomic status was associated with the use of the typical antipsychotics specifically, while Grace et al. [38] found no association between psychotropic use and income adequacy overall; however, individual psychotropic class analysis was not available. There was no consistent trend in antidepressant use and socioeconomic status in two studies [33, 34].
4.2.3.2 Living Alone
All four papers that investigated this association demonstrated a positive association between psychotropic use and living alone. Two of these papers reported a statistically significant association [35, 48]. Eichler et al. [52] reported a trend, but following multivariate analysis, no significant difference; however, the numbers in this study were small and may have influenced the outcome. Arbus et al. [33] reported a numerically greater percentage of those living alone using antidepressants; however, no p value was reported.
4.2.3.3 Hospitalization
Two papers investigated the association between recent hospitalizations and either antipsychotic [60] or antidepressant initiation [53]; both reported a significant positive association.
4.2.4 Theme 4: Carer-Related Factors
Only three papers investigated carer-related factors in psychotropic use. In Grace et al.’s study [38], greater caregiver vigilance was associated with significantly greater anxiolytic/hypnotic use (p ≤ 0.05), and greater caregiver dementia knowledge was associated with greater antidepressant use (p = 0.02), while greater caregiver confidence was associated with significantly less use of anxiolytics (p = 0.04). Rhee et al. [48] reported significantly lower rates of depression in carers of people living with dementia being treated with antipsychotics (p < 0.001), while people with dementia were significantly less likely to be taking an antipsychotic if the caregiver was depressed (p = 0.005). There was no significant association between antidepressant use and caregiver burden reported by Arbus et al. [33].
4.2.5 Theme 5: Prescriber-Related Factors
4.2.5.1 Concomitant Psychotropic Use
Definitions of polypharmacy differed between studies. Orsel et al. [9] reported a prevalence of 18.3% of two or more psychotropic agents prescribed concomitantly, while Maust et al. [50] found that 13.9% were prescribed three or more central nervous system-active medications for at least 30 days. The use of three antipsychotic agents was reported in 3.9% of the sample in a study by Aigbogun et al. [57].
4.2.5.2 Time Course of Psychotropic Use
The first 6 months after dementia diagnosis was identified as a risk factor for initiating psychotropic medication. Although psychotropic use was already evident years before dementia diagnoses, the incidence of use tended to peak between diagnosis and 6 months, especially for antipsychotics and antidepressants [37, 44]. Once initiated, psychotropic use appeared to continue beyond 6 months into years, with long-term psychotropic exposure occurring for all psychotropic medication classes [9, 25, 30,31,32, 37, 45, 56, 58, 61]. This was consistent across study types.
4.3 Meta-analysis
The range of meta-analyses able to be performed was limited to available published appropriate data and that obtained on request. The following analyses were conducted in eligible included studies: antipsychotic use and psychiatric comorbidity; antipsychotic use and respite allowance; antipsychotic use and sex; antidepressant use and psychiatric comorbidity; antidepressant use and respite allowance; anxiolytic/hypnotic use and respite allowance; and study country and antipsychotic, antidepressant, and opioid use.
There were insufficient data to conduct meta-analyses for opioid or anticonvulsant use. Two authors provided raw data; however, this was insufficient for a meta-analysis of age or gender associations [15, 25].
4.3.1 Antipsychotic Treatment
Figure 3 shows the event rates and 95% confidence intervals (CIs) for the prevalence of any antipsychotic treatment in people living with dementia grouped by allowances for respite (i.e., whether the study overtly limited or excluded any time in either temporary facility-based care or hospitalization). There was a high degree of within-group heterogeneity, defined as an I2 value above 50%. (Q = 9308; df(Q) = 16; p < 0.001; I2 = 99.8; see Table S1). There was evidence of publication bias, as indicated by Egger’s test (t = 2.48; df = 15; p < 0.05). Trim and fill analysis did not identify any of the included studies that might have been biased toward higher rates of antipsychotic use.
There was a significant association between the prevalence of antipsychotic medication and allowances for respite (temporary facility-based care/hospitalization) (see Fig. 3 and Table S1). Kester et al.’s study [40] was excluded from this analysis because this variable was not stated. Antipsychotic use was lower in studies excluding any temporary respite or hospitalization (OR 0.140; 95% CI 0.100, 0.192; N = 13) compared with those with respite allowances [OR 0.217, 95% CI 0.173, 0.268; N = 3; q = 4.85; df(Q) = 1; p = 0.028].
Subgroup analysis indicated significant differences in antipsychotic use and the presence or absence of a psychiatric diagnosis (see Table S1). Antipsychotic rates were significantly lower in those without a comorbid psychiatric diagnosis (OR 0.082; 95% CI 0.058, 0.114; N = 4) compared with those with a comorbid psychiatric diagnosis (OR 0.180; 95% CI 0.124, 0.253; N = 6) or studies that did not report this variable [OR 0.182; 95% CI 0.136, 0.238; N = 7; q = 15.02; df(Q) = 2; p < 0.001]. There were no significant differences in antipsychotic rates and the country where studies were conducted (USA versus non-USA; see Table S1) or antipsychotic rates and gender (OR 1.034; 95% CI .931, 1.124; p = 0.433; N = 10 studies).
4.3.2 Antidepressant Treatment
Figure 4 shows the event rates and 95% CIs for the prevalence of any antidepressant medication in people living with dementia grouped by respite allowance as above. There was a high degree of within-group heterogeneity [Q = 16960; df(Q) = 9; p < 0.001; I2 = 99.9; see Table S2]. There was no evidence of publication bias, as indicated by Egger’s test (t = 1.68; df = 8; p = 0.13). Trim and fill analysis did not identify any of the included studies that might have been biased toward higher rates of antidepressant use.
There was no significant difference between the prevalence of antidepressant treatment and respite allowances (see Fig. 4 and Table S2). Those studies that precluded respite allowances had similar rates of antidepressant drug use (OR 0.330; 95% CI 0.303, 0.358; N = 7) as those allowing temporary respite care [OR 0.390; 95% CI 0.226, 0.584; N = 3; q = 0.42; df(q) = 1; p = 0.519]. Subgroup analyses indicated no significant differences in antidepressant drug use and the presence or absence of a comorbid psychiatric diagnosis (p = 0.976) or country where the study was conducted (p = 0.408; see Table S2).
4.3.3 Anxiolytic/Hypnotic Treatment
Figure 5 shows the event rates and 95% CIs for the prevalence of any sedative/hypnotic medication in people living with dementia grouped by respite allowance. There was a high degree of within-group heterogeneity [Q = 5316; df(Q) = 8; p < 0.001; I2 = 99.9; see Table S3]. There was no evidence of publication bias, as indicated by Egger’s test (t = 0.87; df = 7; p = 0.414). Trim and fill analysis did not identify any of the included studies that might have been biased toward higher rates of anxiolytic/hypnotic use. There were no significant differences between the prevalence of anxiolytic/hypnotic use and respite allowances or the country where the study was conducted (see Fig. 5 and Table S3).
5 Discussion
This study is, to our knowledge, unique because it is the first systematic review and meta-analysis of factors associated with psychotropic use in people with dementia living in the community. Although there was heterogeneity identified, thematic analysis did not identify differences in outcome when studies were grouped according to type. In the majority of studies, the risk of bias was low or moderate. Five broad categories of factors emerged from the thematic analysis: (1) patient-related (age, sex, comorbidities), (2) illness-related (severity, functional impairment), (3) environmental (living alone, periods of full-time care), (4) carer-related, and (5) prescriber-related trends. Meta-analysis key findings included significant associations between psychotropic use and psychiatric comorbidity and allowances for respite, specifically for antipsychotics.
Regarding patient factors, age and sex emerged as key factors in the thematic synthesis. Findings concerning age have important implications for symptom interpretations. From the thematic synthesis, ages < 75 years and > 90 years were associated with increased use of psychotropics in general. This was more clearly reflected in class-specific analyses, with antidepressants, antipsychotics, and anticonvulsants being used in younger individuals and benzodiazepines and opioids in older individuals. It is difficult to interpret these findings given the lack of information about reasons for prescribing and the variability of age group definitions. However, there may be a demographic bias concerning risk assessments and behavioral expressions that influence the selection of medications, warranting empirical examination. Moreover, given the normalization or expectation of depression associated with aging [62], it is not surprising that older patients are less likely to be prescribed antidepressants.
Although thematic synthesis identified a trend toward males receiving antipsychotics more than females, this was not demonstrated in the meta-analysis of ten studies with adequate data. Several reasons could account for this, including the limited data amenable to meta-analysis; limited information about the timing, type, or dose of antipsychotic being used; and reasons for use, all of which have sex-specific implications.
The need for psychotropic medication is very much in the eye of the beholder (or perhaps the prescriber), often driven by the degree to which behavior or symptoms are considered problems (i.e., obvious or disruptive) or, conversely, significant and worthy of treatment; a finding consistent with that observed in long-term care [63]. The most common reasons for prescribing antipsychotic medications were excessive motor activity, anxiety, agitation, and even apathy, rather than delusions, hallucinations, or severe aggression. The reasons for these associations remain speculative but could reflect a symptom description/classification issue [14], failure to identify or misidentification of the causes of behaviors (i.e., unmet need, pain, delirium, or illness) [64], or lack of skills and resources available to manage these behaviors [65].
The most robust illness-related finding regarding psychotropic use in this population was a decline in functional ability. The direction of causality is unclear here—that is, whether psychotropic medications cause functional decline or functional decline precipitates use. A hypothetical mechanism for this is a possible relationship between functional decline and care demands, unmet needs of the recipient, and the consequent effects on carer stress [66]. Elucidating what it is about functional decline, independent of cognitive decline or neuropsychiatric symptoms, remains to be investigated and would provide a critical piece of information needed to optimize interventions and support.
Regarding environmental factors and patterns of prescribing, an apparent prescribing trajectory emerged from the literature, with psychotropic prescribing often starting years before dementia diagnoses. Once people are on this trajectory, they appear to continue receiving psychotropic medications, often for prolonged periods. This suggests that the culture of prescribing is initiated before or around the diagnosis period and then continued and increased during certain times throughout the illness. This correlates with other studies suggesting that a dementia diagnosis is associated with the prescribing of psychotropic medications [7, 67].
Points of contact with healthcare providers and different care settings were identified as another environmental factor associated with increased psychotropic use emerging in both the thematic synthesis and meta-analysis. Such points of contact included periods of hospitalization and respite in temporary care. Possible explanations are that temporary time in residential care or hospitalization could relate to acute illness and delirium [68], increased changed behaviors [69], and/or care needs or carer burnout [70]. Again, this is hypothetical and needs empirical testing, but identifying this might help raise awareness among clinicians to avoid the pitfalls of starting new psychotropic medications that are then continued over time, an ideal opportunity for intervention, improved multidisciplinary collaboration, and medication review practices [71, 72].
Living alone was also associated with psychotropic use, potentially pointing to unmet needs being treated pharmacologically. These needs could relate to difficulties navigating services and accessing community supports [73] and increased rates of hospitalization and placement [74].
This review identified the dearth of information regarding carer and caring dyad influences on psychotropic use in this setting. What little literature was available suggested that carer symptomatology [38] and medication use [75] can influence prescribing in the care recipient. Therefore, this relationship deserves greater attention, begging the question: whom are we treating, and are there alternative approaches to lowering the carer burden and supporting carers rather than medicating the person living with dementia?
Despite the availability of guidelines to manage behavioral symptoms in the context of dementia, the implementation and applicability of these to clinical practice remain suboptimal [76]. By addressing identified gaps in understanding the factors driving psychotropic medication use, we will be better able to design and implement interventions to support the needs of this population and their families/carers.
Notwithstanding the gaps in the results relating to the prescriber, some practical questions emerged from this review that might raise awareness and inform prescribers when assessing psychotropic use and prescribing for their patients with dementia in this setting as described in Table 2.
While the focus of quality prescribing has hitherto targeted residential care settings, there is an equal imperative to promote quality prescribing initiatives for those living in the community. This review has shone a light on home-based care as a target for quality prescribing interventions and as an available opportunity for people living with dementia.
This study’s limitations relate mostly to the type of data available, limiting meta-analysis. Further, crude rates of antipsychotic prescribing do not provide any information on the potential appropriateness of use; hence, all possible prevalence studies were not included in this systematic review. This paper was not designed to determine causality, but rather to examine potential factors that were associated with use of psychotropics to provide a greater understanding as to how and why these medications are used for more targeted future research and targeted quality interventions. Five of the 39 studies were cross-sectional, rendering limitations not only in causality but in temporality. The nature of the illness is complex, and while assumptions can be made, there may be multiple confounders influencing the use of psychotropics at an individual level for which data is unavailable. Gray literature was not included due to the potential risk of quality and publication bias. This study had a strong Western bias because no studies were from Asia or Africa.
6 Conclusions
In this systematic review and meta-analysis, we have identified a range of factors associated with the use of psychotropic medication in people living with dementia in the community The reviewed studies suggest that while psychotropic prescribing is prevalent in this population group, a clear understanding as to the reasons why these medications are being used is not available from existing literature. While associated factors can be identified for specific psychotropic classes, there is a clear need to develop targeted research and programs to truly understand what is driving this medication use. While some psychotropic medication use may very well be appropriate and needed in some circumstances (e.g., for treatment of major depression or psychotic symptoms), it would seem that, from the analysis, there is use occurring in circumstances and for reasons that are ill defined or potentially inappropriate. This study has highlighted the very clear need for further targeted research to be conducted. The identified factors provide a much-needed context for this research and future quality improvement programs to be implemented in a targeted way.
Qualitative studies that capture the voice of the person living with dementia at home and their carers, as well as examining aspects of their relationship, are urgently needed to better understand these findings.
References
GBD 2019 Dementia Forecasting Collaborators. Estimation of the global prevalence of dementia in 2019 and forecasted prevalence in 2050: an analysis for the Global Burden of Disease Study 2019. Lancet Public Health. 2019;7(2):e105–25. https://doi.org/10.1016/s2468-2667(21)00249-8.
Alzheimer’s Association. 2017 Alzheimer’s disease facts and figures. Alzheimer’s Association. 2017. https://www.alz.org/media/images/2017-facts-and-figures.pdf. Accessed 05 May 2022.
Royal Commission into Aged Care Quality and Safety. Final report: care, dignity and respect. Commonwealth of Australia. Vol. X. 2021. https://agedcare.royalcommission.gov.au/publications/final-report. Accessed 08 Sep 2022.
Bynum JPW. The long reach of Alzheimer’s disease: patients, practice, and policy. Health Aff. 2014;33(4):534–40. https://doi.org/10.1377/hlthaff.2013.1247.
Johnson S, Bacsu J, Abeykoon H, McIntosh T, Jeffrey B, Novik N. No place like home: a systematic review of home care for older adults in Canada. Can J Aging. 2018;37(4):400–19. https://doi.org/10.1017/s0714980818000375.
Maust DT. Improving psychotropic medication use among persons with dementia. Int Psychogeriatr. 2016;28(10):1587–8. https://doi.org/10.1017/s1041610216001253.
Joling KJ, Koppel MT, van Hout HPJ, Onwuteaka-Philipsen BD, Francke AL, Verheij RA, et al. Psychotropic drug prescription rates in primary care for people with dementia from recorded diagnosis onwards. Int J Geriatr Psychiatry. 2021;36(3):443–51. https://doi.org/10.1002/gps.5442.
Maust DT, Strominger J, Bynum JP, Langa KM, Gerlach LB, Zivin K, et al. Prevalence of psychotropic and opioid prescription fills among community-dwelling older adults with dementia in the US. J Am Med Assoc. 2020;324(7):706–9. https://doi.org/10.1001/jama.2020.8519.
Orsel K, Taipale H, Tolppanen A-M, Koponen M, Tanskanen A, Tiihonen J, et al. Psychotropic drugs use and psychotropic polypharmacy among persons with Alzheimer’s disease. Eur Neuropsychopharmacol. 2018;28(11):1260–9. https://doi.org/10.1016/j.euroneuro.2018.04.005.
Maust DT, Myra Kim H, Seyfried LS, Chiang C, Kavanagh J, Schneider LS, et al. Antipsychotics, other psychotropics, and the risk of death in patients with dementia: number needed to harm. J Am Med Assoc Psychiatry. 2015;72(5):438–45. https://doi.org/10.1001/jamapsychiatry.2014.3018.
Peisah C, Strukovski J-A, Wijeratne C, Mulholland R, Luscombe G, Brodaty H. The development and testing of the quality use of medications in dementia (QUM-D): a tool for quality prescribing for behavioral and psychological symptoms of dementia (BPSD). Int Psychogeriatr. 2015;27(8):1313–22. https://doi.org/10.1017/s1041610214002816.
Westbury JL, Gee P, Ling T, Brown DT, Franks KH, Bindoff I, et al. RedUSe: reducing antipsychotic and benzodiazepine prescribing in residential aged care facilities. Med J Aust. 2018;208(9):398–403. https://doi.org/10.5694/mja17.00857.
Westbury J, Gee P, Ling T, Kitsos A, Peterson G. More action needed: psychotropic prescribing in Australian residential aged care. Aust N Z J Psychiatry. 2019;53(2):136–47. https://doi.org/10.1177/0004867418758919.
Maust DT, Kales HC. Bringing precision medicine to the management of BPSD. Am J Geriatr Psychiatry. 2017;25(5):469–70. https://doi.org/10.1016/j.jagp.2017.02.010.
Maust DT, Langa KM, Blow FC, Kales HC. Psychotropic use and associated neuropsychiatric symptoms among patients with dementia in the USA. Int J Geriatr Psychiatry. 2017;32(2):164–74. https://doi.org/10.1002/gps.4452.
Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6(7):e1000097. https://doi.org/10.1371/journal.pmed.1000097.
Martin JLR, Pérez V, Sacristán M, Álvarez E. Is grey literature essential for a better control of publication bias in psychiatry? An example from three meta-analyses of schizophrenia. Eur Psychiatry. 2005;20(8):550–3. https://doi.org/10.1016/j.eurpsy.2005.03.011.
Hoy D, Brooks P, Woolf A, Blyth F, March L, Bain C. Assessing risk of bias in prevalence studies: modification of an existing tool and evidence of interrater agreement. J Clin Epidemiol. 2012;65(9):934–9. https://doi.org/10.1016/j.jclinepi.2011.11.014.
Wells GA, Shea B, O’Connell D, Peterson J, Welch V, Losos M, et al. The Newcastle-Ottawa scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Canada: Ottawa Hospital Research Institute; 2014.
Thomas J, Harden A. Methods for the thematic synthesis of qualitative research in systematic reviews. BMC Med Res Methodol. 2008;8:45. https://doi.org/10.1186/1471-2288-8-45.
Corbin J, Strauss A. Basics of qualitative research: techniques and procedures for developing grounded theory. 4th ed. Los Angeles: SAGE; 2015.
Egger M, Smith GD, Philips AN. Meta-analysis: principles and procedures. BMJ. 1997;315(7121):1533–7. https://doi.org/10.1136/bmj.315.7121.1533.
Duval S, Tweedie R. A nonparametric “trim and fill” method of accounting for publication bias in meta-analysis. J Am Stat Assoc. 2000;95:89–98. https://doi.org/10.1080/01621459.2000.10473905.
Tolppanen AM, Taipale H, Koponen M, Lavikainen P, Tanskanen A, Tiihonen J. Cohort profile: the Finnish Medication and Alzheimer’s disease (MEDALZ) study. BMJ Open. 2016;6(7):e012100. https://doi.org/10.1136/bmjopen-2016-012100.
Gerritsen AJA, Bakker C, Bruls E, Verhey FRJ, Pijnenburg YAL, Millenaar JK, et al. Psychotropic drug use in community-dwelling people with young-onset dementia: two-year course and determinants. Aging Ment Health. 2021;25(1):179–86. https://doi.org/10.1080/13607863.2019.1691145.
Sapra M, Varma A, Sethi R, Vahia I, Chowdhury M, Kim K, et al. Utilization of antipsychotics in ambulatory elderly with dementia in an outpatient setting. Fed Pract. 2012;29(1):29–33.
Efjestad AS, Ihle-Hansen H, Hjellyik V, Engedal K, Blix HS. Sex differences in psychotropic and analgesic drug use before and after initiating treatment with acetylcholinesterase inhibitors. PLoS ONE. 2021;16(9):e0243804. https://doi.org/10.1371/journal.pone.0243804.
Karttunen N, Taipale H, Hamina A, Taskanen A, Tiihonen J, Tolppanen A-M, et al. Concomitant use of benzodiazepines and opioids in community-dwelling older people with or without Alzheimer’s disease—a nationwide register-based study in Finland. Int J Geriatr Psychiatry. 2019;34(2):280–8. https://doi.org/10.1002/gps.5018.
Saarelainen L, Taipale H, Koponen M, Tanskanen A, Tolppanen A-M, Tiihonen J, et al. The incidence of benzodiazepine and related drug use in persons with and without Alzheimer’s disease. J Alzheimer’s Dis. 2016;49(3):809–18. https://doi.org/10.3233/jad-150630.
Sivananthan SN, Lavergne MR, McGrail KM. Caring for dementia: a population-based study examining variations in guideline-consistent medical care. Alzheimer’s Dement. 2015;11(8):906–16. https://doi.org/10.1016/j.jalz.2015.02.008.
Taipale H, Koponen M, Tanskanen A, Tolppanen A-M, Tiihonen J, Hartikainen S. Long-term use of benzodiazepines and related drugs among community-dwelling individuals with and without Alzheimer’s disease. Int Clin Psychopharmacol. 2015;30(4):202–8. https://doi.org/10.1097/yic.0000000000000080.
Hamina A, Taipale H, Tanskanen A, Tolppanen A-M, Karttunen N, Pylkkänen L, et al. Long-term use of opioids for nonmalignant pain among community-dwelling persons with and without Alzheimer disease in Finland: a nationwide register-based study. Pain. 2017;158(2):252–60. https://doi.org/10.1097/j.pain.0000000000000752.
Arbus C, Gardette V, Bui E, Cantet C, Anrieu S, Nourhashémi F, et al. Antidepressant use in Alzheimer’s disease patients: results of the REAL.FR cohort. Int Psychogeriatr. 2010;22(1):120–8. https://doi.org/10.1017/s1041610209990780.
Bhattacharjee S, Oh YM, Reiman EM, Burke WJ. Prevalence, patterns, and predictors of depression treatment among community-dwelling elderly individuals with dementia in the United States. Am J Geriatr Psychiatry. 2017;25(7):803–13. https://doi.org/10.1016/j.jagp.2017.03.003.
Cermakova P, Nelson M, Secnik J, Garcia-Ptacek S, Johnell K, Fastbom J, et al. Living alone with Alzheimer’s disease: data from SveDem, the Swedish Dementia Registry. J Alzheimer’s Dis. 2017;58(4):1265–72. https://doi.org/10.3233/jad-170102.
Drummond N, McCleary L, Freiheit E, Molnar F, Dalziel W, Cohen C, et al. Antidepressant and antipsychotic prescribing in primary care for people with dementia. Can Fam Physician. 2018;64:e488–97. https://www.cfp.ca/content/cfp/64/11/e488.full.pdf
Puranen A, Taipale H, Koponen M, Tanskanen A, Tolppanen A-M, Tiihonen J, et al. Incidence of antidepressant use in community-dwelling persons with and without Alzheimer’s disease: 13-year follow-up. Int J Geriatr Psychiatry. 2017;32(1):94–101. https://doi.org/10.1002/gps.4450.
Grace EL, Burgio LD, Allen RS, DeCoster J, Aiello AE, Algase DL. Caregiver and care recipient characteristics as predictors of psychotropic medication use in community-dwelling dementia patients. Aging Ment Health. 2016;20(12):1297–304. https://doi.org/10.1080/13607863.2015.1075960.
Bargagli AM, Cascini S, Agabiti N, Kirchmayer U, Marino C, Davoli M. Determinants of antipsychotic drugs prescription among community-living older adults with dementia: a population-based study using health information systems in the Lazio region, Italy. Clin Interv Aging. 2019;14:2071–83. https://doi.org/10.2147/cia.s218641.
Kester R, Unützer J, Hogan D, Huang H. Antipsychotic prescribing patterns in a Medicare Advantage population of older individuals with dementia. J Ment Health. 2017;26(2):167–71. https://doi.org/10.1080/09638237.2016.1244720.
Nili M, Shen C, Sambamoorthi U. Low-value care: antipsychotic medication use among community-dwelling Medicare beneficiaries with Alzheimer’s disease and related dementias and without severe mental illness. Aging Ment Health. 2020;24(3):504–10. https://doi.org/10.1080/13607863.2018.1544211.
Xiong GL, Filshtein T, Beckett LA, Hinton L. Antipsychotic use in a diverse population with dementia: a retrospective review of the National Alzheimer’s Coordinating Center database. J Neuropsychiatry Clinic Neurosci. 2015;27(4):326–32. https://doi.org/10.1176/appi.neuropsych.15010020.
Laitinen M-L, Bell JS, Lavikainen P, Lönnroos E, Sulkava R, Hartikainen S. Nationwide study of antipsychotic use among community-dwelling persons with Alzheimer’s disease in Finland. Int Psychogeriatr. 2011;23(10):1623–31. https://doi.org/10.1017/s1041610211001621.
Koponen M, Tolppanen A-M, Taipale H, Tanskanen A, Tiihonen J, Johnell K, et al. Incidence of antipsychotic use in relation to diagnosis of Alzheimer’s disease among community-dwelling persons. Br J Psychiatry. 2015;207(5):444–9. https://doi.org/10.1192/bjp.bp.114.162834.
Sarycheva T, Taipale H, Lavikainen P, Tiihonen J, Tanskanen A, Hartikainen S, et al. Incidence and prevalence of antiepileptic medication use in community-dwelling persons with and without Alzheimer’s disease. J Alzheimer’s Dis. 2018;66(1):387–95. https://doi.org/10.3233/jad-180594.
Grace EL, Allen RS, Ivey K, Knapp SM, Burgio LD. Racial and ethnic differences in psychotropic medication use among community-dwelling persons with dementia in the United States. Aging Ment Health. 2018;22(4):458–67. https://doi.org/10.1080/13607863.2017.1286451.
Taipale H, Koponen M, Tanskanen A, Tolppanen A-M, Tiihonen J, Hartikainen S. Drug use in persons with and without Alzheimer’s disease aged 90 years or more. Age Ageing. 2016;45(6):900–4. https://doi.org/10.1093/ageing/afw141.
Rhee Y, Csernansky JG, Emanuel LL, Chang C-G, Shega JW. Psychotropic medication burden and factors associated with antipsychotic use: an analysis of a population-based sample of community-dwelling older persons with dementia. J Am Geriatr Soc. 2011;59(11):2100–7. https://doi.org/10.1111/j.1532-5415.2011.03660.x.
Taipale H, Koponen M, Tanskanen A, Tolppanen A-M, Tiihonen J, Hartikainen S. High prevalence of psychotropic drug use among persons with and without Alzheimer’s disease in Finnish nationwide cohort. Eur Neuropsychopharmacol. 2014;24(11):1729–37. https://doi.org/10.1016/j.euroneuro.2014.10.004.
Maust DT, Strominger J, Myra Kim H, Langa KM, Bynum JPW, Chang C-H, et al. Prevalence of central nervous system—active polypharmacy among older adults with dementia in the US. J Am Med Assoc. 2021;325(10):952–61. https://doi.org/10.1001/jama.2021.1195.
Taipale H, Koponen M, Tanskanen A, Tolppanen A-M, Tiihonen J, Hartikainen S. Antipsychotic polypharmacy among a nationwide sample of community-dwelling persons with Alzheimer’s disease. J Alzheimer’s Dis. 2014;41(4):1223–8. https://doi.org/10.3233/jad-140282.
Eichler T, Wucherer D, Thyrian JR, Kilimann I, Hertel J, Nichalowsky B, et al. Antipsychotic drug treatment in ambulatory dementia care: prevalence and correlates. J Alzheimer’s Dis. 2015;43(4):13–11. https://doi.org/10.3233/jad-141554.
Hakala A, Tolppanen A-M, Koponen M, Taipale H, Tanskanen A, Hartikainen S, et al. Does recent hospitalization increase antipsychotic initiation among community dwellers with Alzheimer’s disease? J Am Med Dir Assoc. 2021;22(7):1543–7. https://doi.org/10.1016/j.jamda.2020.12.021.
Rios S, Perlman CM, Costa A, Heckman G, Hirdes JP, Mitchell L. Antipsychotics and dementia in Canada: a retrospective cross-sectional study of four health sectors. BMC Geriatr. 2017;17(1):244. https://doi.org/10.1186/s12877-017-0636-8.
Taipale H, Koponen M, Tanskanen A, Tolppanen A-M, Tiihonen J, Hartikainen S. Antipsychotic doses among community-dwelling persons with Alzheimer disease in Finland. J Clin Psychopharmacol. 2014;34(4):435–40. https://doi.org/10.1097/jcp.0000000000000133.
Hamina A, Lavikainen P, Tanskanen A, Tolppanen A-M, Tiihonen J, Hartikainen S, et al. Impact of opioid initiation on antipsychotic and benzodiazepine and related drug use among persons with Alzheimer’s disease. Int Psychogeriatr. 2018;30(7):947–56. https://doi.org/10.1017/s1041610217002897.
Aigbogun MS, Cloutier M, Gauthier-Loiselle M, Guerin A, Ladouceur M, Baker RA, et al. Real-world treatment patterns and characteristics among patients with agitation and dementia in the United States: findings from a large, observational, retrospective chart review. J Alzheimer’s Dis. 2020;77(3):1181–94. https://doi.org/10.3233/jad-200127.
Törmälehto S, Martikainen J, Bell JS, Hallikainen I, Koivisto AM. Use of psychotropic medications in relation to neuropsychiatric symptoms, cognition and functional performance in Alzheimer’s disease over a three-year period: Kuopio ALSOVA study. Int Psychogeriatr. 2017;29(10):1723–33. https://doi.org/10.1017/s1041610217001090.
Teipel SJ, Thyrian JR, Hertel J, Eichler T, Wucherer D, Michalowsky B, et al. Neuropsychiatric symptoms in people screened positive for dementia in primary care. Int Psychogeriatr. 2015;27(1):39–48. https://doi.org/10.1017/s1041610214001987.
Tarvainen A, Hartikainen S, Taipale H, Tanskanen A, Koponen M, Tolppanen A-M. Association of recent hospitalisation with antidepressant initiation among community dwellers with Alzheimer’s disease. Int J Geriatr Psychiatry. 2021;36(7):1075–84. https://doi.org/10.1002/gps.5505.
Koponen M, Taipale H, Tanskanen A, Tolppanen A-M, Tiihonen J, Ahonen R, et al. Long-term use of antipsychotics among community-dwelling persons with Alzheimer’s disease: a nationwide register-based study. Eur Neuropsychopharmacol. 2015;25(10):1706–13. https://doi.org/10.1016/j.euroneuro.2015.07.008.
Lyons A, Alba B, Heywood W, Fileborn B, Minichiello V, Barrett C, et al. Experiences of ageism and the mental health of older adults. Aging Ment Health. 2018;22(11):1456–64. https://doi.org/10.1080/13607863.2017.1364347.
Resnick B, Kolanowski A, Van Haitsma K, Galik E, Boltz M, Ellis J, et al. Current psychotropic medication use and contributing factors among nursing home residents with cognitive impairment. Clinic Nurs Res. 2021;30(1):59–69. https://doi.org/10.1177/1054773819838678.
Scuteri D, Vulnera M, Piro B, Bossio RB, Morrone LA, Sandrini G, et al. Pattern of treatment of behavioural and psychological symptoms of dementia and pain: evidence on pharmacoutilization from a large real-world sample and from a centre for cognitive disturbances and dementia. Eur J Clin Pharmacol. 2021;77:241–9. https://doi.org/10.1007/s00228-020-02995-w.
Braun A, Trivedi DP, Dickenson A, Hamilton L, Goodman C, Gage H, et al. Managing behavioural and psychological symptoms in community dwelling older people with dementia: 2. a systematic review of qualitative studies. Dementia 2019;18(7-8);2950–70. https://doi.org/10.1177/1471301218762856
Razani J, Kakos B, Orieta-Barbalace C, Wong JT, Casas R, Lu P, et al. Predicting caregiver burden from daily functional abilities of patients with mild dementia. J Am Geriatr Soc. 2007;55(9):1415–20. https://doi.org/10.1111/j.1532-5415.2007.01307.x.
Gnjidic D, Agogo GO, Ramsey CM, Moga DC, Allore H. The impact of dementia diagnosis on patterns of potentially inappropriate medication use among older adults. J Gerontol Ser A Biol Sci Med Sci. 2018;73(10):1410–7. https://doi.org/10.1093/gerona/gly078.
Fong TG, Davis D, Growdon ME, Albuquerque A, Inouye SK. The interface between delirium and dementia in elderly adults. Lancet Neurol. 2015;14(8):823–32. https://doi.org/10.1016/s1474-4422(15)00101-5.
Toot S, Swinson T, Devine M, Challis D, Orrell M. Causes of nursing home placement for older people with dementia: a systematic review and meta-analysis. Int Psychogeriatr. 2017;29(2):195–208. https://doi.org/10.1017/s1041610216001654.
Maayan N, Soares-Weiser K, Lee H. Respite care for people with dementia and their carers. Cochrane Database Syst Rev. 2014;16(1):CD004396. https://doi.org/10.1002/14651858.cd004396.pub3.
Brunero S, Wand APF, Lamont S, John L. A point prevalence study of the use of psychotropic medication in an acute general hospital. Int Psychogeriatr. 2016;28(6):967–75. https://doi.org/10.1017/s104161021500232x.
Tumusiime WA, Hardman CJ, Breen JL. Antipsychotic prescribing in people admitted to hospital with dementia or delirium. Australas J Ageing. 2022;41(2):258–64. https://doi.org/10.1111/ajag.13017.
Read S, Hu B, Wittenberg R, Brimblecombe N, Robinson L, Banerjee S. A longitudinal study of functional unmet need among people with dementia. J Alzheimer’s Dis. 2021;84(2):705–16. https://doi.org/10.3233/jad-210724.
Connolly D, Garvey J, McKee G. Factors associated with ADL/IADL disability in community dwelling older adults in the Irish longitudinal study on ageing (TILDA). Disabil Rehabil. 2017;39(8):809–16. https://doi.org/10.3109/09638288.2016.1161848.
Thorpe JM, Thorpe CT, Kennelty KA, Gellad WF, Schulz R. The impact of family caregivers on potentially inappropriate medication use in noninstitutionalized older adults with dementia. Am J Geriatr Pharmacother. 2012;10(4):230–41. https://doi.org/10.1016/j.amjopharm.2012.05.001.
Ma H, Lu X, Zhou A, Wang F, Zuo X, Zhan M, et al. Clinical practice guidelines for the management of behavioral and psychological symptoms of dementia: a systematic review with AGREE II. Front Neurol. 2022;25(13):799723. https://doi.org/10.3389/fneur.2022.799723.
Acknowledgements
Thanks to the two authors who provided data in response to our request. Thanks to our research librarian, Jennifer Whitfield of UNSW.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions.
Conflicts of interest
The authors have no conflicts of interest to declare.
Data availability statement
Raw data is available on request from the corresponding author.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Code availability
Not applicable.
Author contributions
KL designed the study, conducted the research, wrote the manuscript, and revised the manuscript following editorial and peer review following discussion and agreement with the authorship group. CP designed the study, conducted the research, and assisted in writing of the manuscript. AW and JB were involved in design of the study, review of the data, and writing and review of the manuscript. GH was involved in statistical planning and conduct of the meta-analysis, and writing and review of the manuscript. All authors have read the revised version of the manuscript and are accountable for the final contents.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Loftus, K.L., Wand, A.P.F., Breen, J.L. et al. Factors Associated with Psychotropic Medication Use in People Living with Dementia in the Community: A Systematic Review and Meta-Analysis. Drugs Aging 40, 1053–1084 (2023). https://doi.org/10.1007/s40266-023-01070-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40266-023-01070-0