Abstract
Elevated low-density lipoprotein cholesterol (LDL-C) is unanimously recognized as a major modifiable risk factor related to the development of atherosclerotic cardiovascular disease (ASCVD). Consistent evidence confirms that reducing LDL-C is associated with reduction of major adverse cardiovascular events (MACEs), with benefits proportionally related to initial individual CV risk and absolute reduction of LDL-C levels. The recent European guidelines on cardiovascular prevention have proposed a revised approach in cardiovascular risk evaluation, taking into account a renewed consideration of the interaction between risk factors and possible confounding factors (e.g., age). Although for patients considered to be at high and very high cardiovascular risk the need for stringent risk factors treatment is clearly stated, for those who are at low-to-moderate cardiovascular risk the issue is more debated. For those latter subjects, current guidelines indicate that risk factor treatment is generally not necessary, unless the impact of CV risk modifiers, lifetime CV risk and treatment benefit may be substantial. In addition, despite the estimated low-to-moderate short-term CV risk, the early appearance of even mild LDL-C level elevations may contribute to impair long-term CV prognosis. Therefore, encouraging the achievement of desired LDL-C goals through tailored conservative lifestyle changes and, if necessary, pharmacologic strategies should not be excluded categorically in all low-to-moderate risk individuals. In this review, we summarize the most recent evidence that may influence the choice to treat or not to treat LDL-C elevations in subjects at low-to-moderate risk and the suggested therapeutic tools aimed at achieving the recommended LDL-C goals.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Atherosclerotic cardiovascular disease (ASCVD) remains one of the main cause of morbidity and mortality worldwide. Elevation of plasma LDL-cholesterol (LDL-C) and apolipoprotein (apo) B-containing lipoproteins levels are causally related to ASCVD, due to their pathophysiological role in atherogenesis [1]. Evidence from patients with familial hypercholesterolemia has strongly supported the concept that lifetime exposure to elevated LDL-C values is unequivocally related to the early development of ASCVD [2]. Also, during the last decades, epidemiological data and Mendelian randomization studies have confirmed that lower LDL-C levels are associated with improved cardiovascular prognosis; furthermore, randomized clinical trials have further supported the overwhelming evidence that greater LDL-C lowering reduces ASCVD risk both in primary and secondary prevention [3, 4]. The new European cardiovascular prevention guidelines emphasize the importance of the latter concept, indicating dyslipidemias as one of the primary risk factor in which is possible to intervene with extremely important beneficial effects on CV prognosis [5]. In order to define the goals of any ASCVD prevention strategy and to choose the appropriate LDL-C-targeted intervention, it is essential to have a precise estimate of the subject global CV risk. Thanks to the recently revised ASCVD stratification models, it is possible to obtain a 10-year CV risk estimation for geographic areas at different CV risk, to extend risk estimation to different age groups (i.e., older adults), to improve CV risk measures by using non-HDL-C levels, and to provide cumulative CV risk estimates instead of CV risk mortality only. Moreover, the possibility to improve CV risk classification in younger individuals, by using lifetime CV risk estimates, further helps to define specific LDL-C goals and to select the appropriate therapeutic strategy depending on the degree of the LDL-C lowering required.
Despite the assumption that the lower the LDL-C values the better the ASCVD risk improvement is particularly stressed and widely accepted for the secondary CV prevention, the choice to treat or not low-to-moderate CV risk subjects and the preferred goals for risk factors treatment are often controversial and require a more complex balance of benefits and downsides [6,7,8]. Hence, individualization, stepwise approach, correct communication and decision sharing are integral part of the updated recommendations in lower risk categories. The aim of this review is to present the recommended tools and criteria to define the low-to-moderate risk subject and the current cholesterol-lowering strategies to be preferred in this cardiovascular risk category.
2 Cardiovascular Risk Scoring and Risk Categories
The ASCVD risk consists in the likelihood to develop a fatal or non-fatal cardiovascular event over a defined timeframe and it depends on the possible prognostic interaction between several CV risk and protective factors. ASCVD risk groups should be considered as a “continuum” and not a set of sharply separated categories, although indicative thresholds are required for choosing individual targets of intervention. The conditions that affect cardiovascular risk can be divided into modifiable and non-modifiable. Non-modifiable are essentially determined by genetics and cannot be changed through lifestyle or therapy. They consist of ethnicity [9], familiarity and age [5]. Modifiable risk factors, on the other hand, are susceptible to intervention and have been unanimously identified as cigarette smoking, blood pressure (BP) elevation, diabetes mellitus (DM), adiposity and rising in apo-B containing lipoproteins [5]. Individual cardiovascular risk is also influenced by the possible presence of protective factors, although their actual benefit remains debated. Among these, the one most studied is HDL cholesterol (HDL-C) levels, which are inversely associated with cardiovascular risk [10]. However, Mendelian randomization studies or randomized controlled trials did not suggest that increasing HDL-cholesterol (HDL-C) can result in a reduction in cardiovascular risk [11]. Similarly, the protective role of estrogen, pointed to as evidence for the lower incidence of ASCVD in young women, is still debated [12].
Many risk evaluating systems have been designed for CV risk assessment, in particular for those defined “apparently healthy people”, namely without DM, chronic kidney disease (CKD), familial hypercholesterolemia (FH) and ASCVD.
In the august 2021, new guidelines for cardiovascular prevention have been released [5], in which the Systematic Coronary Risk Estimation (SCORE) algorithm, thought for risk assessment in primary prevention, has been updated. The new edited SCORE2 introduces innovations to overcome some critical issues highlighted in the previous one (SCORE), helping to estimate more appropriately a patient's cardiovascular risk. As in the past edition, SCORE2 is calibrated by geographic areas based on specific national cardiovascular mortality data. Country-specific CV risk is stratified into four categories (low, moderate, high and very high) and not two (low and high) as in SCORE. Some countries have therefore seen their CV risk reclassified, such as Italy, which is now considered a moderate-risk country.
Firstly, SCORE2 estimates 10-year risk of total CV events, considering also non-fatal in addition to fatal events. This adjustment makes the algorithm able to estimate the burden connected to morbidity as well as mortality. Second, the new risk score is applicable to a wider age group, extended if compared to the previous model beyond to 70 years old. In addition, the lipid variable used in SCORE2 is no longer total cholesterol, but non-HDL-C, a parameter more realistically close to the plasma concentration of Apo-B containing lipoproteins and better correlated to long-term CVD risk [13]. Furthermore, in the different age ranges (< 50; 50–69; ≥ 70 years old), different CV risk estimates may define the same subject risk category; thus, a low-to-moderate risk subject will be defined by an estimated CV risk < 2.5%, < 5% or < 7.5% in subjects aged < 50, 50–69 or ≥ 70 year, respectively. Using the SCORE algorithm in people aged more than 65 years old, overestimation of ASCVD-related mortality might result despite relatively low levels of the individual CV risk factors, possibly encouraging the achievement of more “aggressive” risk factors goals in this age group. Indeed, CV risk estimate might be inaccurate in older people if we equate the impact of the different CV risk factors in all age’s categories [14]. In order to contrast CV risk overestimation and possible risk factors overtreatment in the elderly, the new Systematic Coronary Risk Estimation—Older Persons (SCORE-OP) has been proposed. The algorithm, available for over 70-y people, is calibrated considering the weaker association between CV risk factors and ASCVD observed with aging as well as the competing risk.
Another limitation observed using SCORE algorithm was related to risk estimation in younger people; in this latter category, a low age-related absolute risk can be estimated even in the presence of an isolated elevation of an individual risk factor, which may suggest subsequent harmful consequences during life span [15]. To obviate this issue, the recommendation for younger adults is to use a lifetime CV risk tool. The LIFE-CVD model, validated for low- and moderate-risk countries, analyzes the benefit in terms of disease-free years gained for every ~40 mg/dL (1 mmol/L) reduction of LDL-C levels. This gain is estimated differentially according to the presence or absence of the following risk factors: smoking habit, systolic blood pressure, non-HDL-C levels, age, and sex. This tool not only may provide a guide for the choice of the most appropriate treatment in younger subjects at apparently lower CV risk, but may also support the communication with the patient aimed at sharing the most appropriate treatment strategy. Thanks to the valorization of this tool, the awareness around the lifelong action of an individual risk factor might be improved and the importance of early intervention, supporting treatment decisions, further implemented.
3 Cardiovascular Disease Risk Categories: The Low-to-Moderate Risk Subject
As mentioned above, regarding primary ASCVD prevention, the population may be roughly divided into two broad categories, according to the presence or absence of specific comorbidities, including chronic renal disease (CKD), familial hypercholesterolemia (FH) and type 2 diabetes mellitus. In the apparently healthy population, without the mentioned comorbidities, the likelihood of developing a major adverse cardiovascular event (MACE) at 10 years, estimated through the SCORE2 and SCORE-OP risk charts, allows placing the patient in a specific risk category depending on the exposure to different risk factors and according to the age group they belong to.
A 10-year CV risk is considered very high when SCORE2/OP is ≥ 7.5% in individuals younger than 50 years, ≥ 10% in those aged between 50 and 69 years, ≥ 15% in individuals older than 70 years. In contrast, high cardiovascular risk occurs when SCORE2/OP is 2.5% to < 7.5% in individuals younger than 50 years, between 5 and 10% in those aged 50 to 69 years, 7.5% to < 15% in those older than 70 years. Finally, as previously stated, in patients at low-to-moderate 10-year cardiovascular risk, SCORE2/OP will be < 2.5% in individuals younger than 50 years, < 5% in individuals aged 50–69 years, < 7.5% in individuals older than 70 years. A separate discussion should be made for patients with type 2 diabetes mellitus over 40 years of age who have a short-lasting disease (< 10 years), in the absence of both overt target organ damage (TOD) and additional cardiovascular risk factors. These patients, although not eligible for SCORE2/OP algorithm, must be considered at moderate cardiovascular risk according to ESC guidelines.
In order to better define the individual cardiovascular risk and to make a more convinced decision on the goals to be achieved and the treatment measures to be taken, the new European guidelines on cardiovascular prevention have expanded the range of possible modifiers of CV risk [5]. Among these, markers of organ damage such as carotid atherosclerotic plaque (defined as ultrasound evidence of an intima-media thickness (IMT) ≥ 1.5 mm or as a wall thickening ≥ 50% of the surrounding vessel wall), left ventricular hypertrophy, microalbuminuria or estimated glomerular filtration rate (eGFR) reduction (i.e < 60 mL/min/1.73 m2) are certainly of relevant importance. Although these variables are not included in the SCORE2/OP algorithms, their correlation with cardiovascular morbidity and mortality is well established [16]. In the complex assessment of cardiovascular risk there are also other non-traditional risk modifiers. Family history and genetics, ethnicity, psychosocial and socioeconomic factors, clinical scenarios that determine frailty, are all important influencing elements.
While in the high and very high CV risk patients the treatment of risk factors is recommended or at least to be considered in a large proportion of them, in the patient at low-to-moderate cardiovascular risk, the therapeutic approach is controversial. In this latter category, there is the tendency to limit pharmacological treatment to selected cases in which lifestyle modification are unable to achieve the CV risk goals and possibly according to additional opportunity issues.
4 Cholesterol-Lowering in Low-to-Moderate Risk Subjects: Rationale and Strategies
In consideration of the causal role of LDL-C in ASCVD and the evidence of a linear relationship between LDL-C and ASCVD risk reductions, cholesterol-lowering treatment constitutes the cornerstone of any CV prevention strategy [5, 17]. The clinical benefit that can be obtained with cholesterol-lowering therapy depends both on the initial individual CV risk and on the absolute reduction of LDL-C that is achieved; thus, the intensity of the treatment must be modulated as to obtain more ambitious goals (lower LDL-C levels) in patients at higher CV risk. Consistent with this and with the results of several clinical trials, and in accordance with the principle "the lower the better" (i.e., the greater the absolute reduction in LDL-C and the greater the benefit in terms of CV risk reduction), the recommended LDL-C goals in patients at high and very high CV risk are achieving LDL-C values < 70 mg/dL and < 55 mg/dL, respectively, in addition to the decrease in LDL-C ≥ 50% from baseline [5, 6].
Importantly, the relationship between LDL-C and ASCVD risk is strongly amplified when duration of follow-up and cholesterol-lowering treatment is longer. Therefore, the cumulative lifelong exposure to LDL-C plays a crucial role in influencing ASCVD risk [1]. Accordingly, even mild elevations in plasma LDL-C in young adults (LDL-C ≥ 100 mg/dL) are associated with a significant long-term increased risk for ASCVD and related mortality [18, 19]. Therefore, even mild elevation of plasma LDL-C levels in subjects at lower short-term CV risk might exert an unfavorable impact on long-term CV risk. This assumption is clearly evidenced by the discrepancy in estimated risk from 10-year ASCVD risk and lifetime CV risk scores in younger adults with early elevations in plasma LDL-C. In addition, plasma LDL-C level lowering is associated with ASCVD events reduction in young adults at low CV risk [20]. Thus, with respect to LDL-C, along with the statement “the lower the better” for patients at higher CV risk, “the earlier the better, for longer” statement should be also considered for patients at lower estimated CV risk. Importantly, 2019 ESC/EAS guidelines recommend an LDL-C goal of less than 116 mg/dL for low-risk subjects; LDL-C < 100 mg/dL has been recommended by the more recent guidelines in low-to-moderate risk subjects, thus further underlining the need of a tailored and effective cholesterol-lowering strategy for this patients’ categories.
In order to ensure the achievement of the recommended therapeutic goals, the 2021 cardiovascular disease prevention guideline propose a stepwise approach in the choice and possibly intensification of treatment [5]. Despite the general rule proposed by these guidelines is to reserve a conservative approach based on lifestyle intervention in low-to-moderate risk individuals, risk modifiers, lifetime CV risk and treatment benefit, along with patient preferences should be considered in order to integrate the latter conservative approach with the 2019 ESC/EAS recommendations regarding the tailored LDL-C target-driven intervention strategy [6].
5 Lifestyle Interventions Affecting Cholesterol Levels
5.1 Diet
Higher consumption of fruit and vegetables, legumes, nuts, vegetable oils, fish and whole grains, along with the replacement of animal fats with PUFAs, a lower intake of trans-fatty acids, red and processed meats, refined carbohydrates, and salt, are associated with a lower incidence of CV events [21]. In detail, regarding fruit and vegetable consumption, a 4% reduction in fatal cardiovascular events was observed for each daily serving of fruits (i.e. 77 g) and vegetables (i.e. 80 g) [22]. The new ESC guidelines on cardiovascular prevention recommend a minimum fruit and vegetable consumption of 2-3 servings (200 g) per day in each category [5]. Overall, the impact of healthy diets on plasma LDL-C levels is variable and strongly influenced by adherence and long-term persistence. In this context, dietary fibers, particularly in substitution of saturated fat, has beneficial effects on plasma lipid profile [23].
5.2 Physical Activity
Physical activity and particularly aerobic exercise has been found to improve CV prognosis by favorably influencing several CV risk factors, including dyslipidemias [21, 22]. Studies focusing on the relationship between physical activity and HDL-C have reported that HDL-C levels are more sensitive to aerobic exercise than LDL-C [22]. This is of particular importance in relation to the evidence showing that HDLs exert anti-atherosclerotic effects and physical activity improves HDL function and HDL-C levels as well [24, 25]. The effect of physical activity on LDL-C is generally inconsistent, although plasma levels of the highly atherogenic small-dense LDL particles are reduced by aerobic exercise, and the average size of LDLs is increased [26]. These beneficial effects of physical activity on lipid profile are generally paralleled by a significant reduction in fasting and post-prandial [27] triglyceride levels when applied to patients with elevated triglyceride levels [22].
5.3 Body Weight Management
Altered body composition, commonly understood as obesity (body mass index (BMI) ≥30 kg/m2) or overweight (BMI 25-29 kg/m2) influences cardiovascular risk [28]. Irrespective of the composition of the weight loss strategy, reducing body weight was associated with a significant decrease TC, LDL-C VLDL-C and triglyceride levels; however, there was only a modest correlation between extent of weight loss and changes in lipid levels [29]. Weight loss was overall associated with an average LDL-C lowering of 0.2 mmol/L and TG lowering of 0.13 mmol/L [30]. Among different macronutrient composition contributing to weight loss, low carbohydrate diets were associated with greater triglyceride than LDL-C level reductions [31].
5.4 Nutraceuticals
Although outcome studies demonstrating that cholesterol-lowering nutraceuticals could improve CV prognosis are still lacking (except for the case of xuezhikang), the use of selected safe and effective nutraceutical has been proposed as to improve the quality of the cholesterol-lowering treatment, possibly increasing the chance to achieve LDL-C goal in clinical practice [5]. More importantly, the 2019 ESC/EAS guidelines [6], along with a number of position statements and consensus documents [32, 33], support the possible use of some cholesterol-lowering nutraceuticals in combination with additional lifestyle interventions in order to amplify their respective cholesterol-lowering impact. A recent International Lipid Expert Panel (ILEP) position paper reported that some nutraceuticals with an evidence for a lipid lowering impact (e.g., red yeast rice, beberine, curcumin, isoflavones) may also produce improvement in markers of systemic inflammation [34] and arterial function [35].
6 Drugs Affecting Cholesterol Levels in Lower Risk Subjects
Although drug treatment does not represent the first line therapeutic choice for subjects at low-to-moderate CV risk, this latter approach might be considered in specific conditions in order to achieve the recommended LDL-C goals. In particular, for individuals in which lifestyle changes show moderate success at reducing LDL-C, as indicated by LDL-C > 115 mg/dL in low risk subjects and by LDL-C > 100 mg/dL in moderate risk subjects, preferably every 12 weeks lipid profile monitoring and enhanced educational interventions might be appropriate. When adherence to lifestyle changes is poor and LDL-C levels still persist significantly higher than recommended goals, nutraceuticals (e.g., single agent or combination formulations) and/or low-dose statin therapy and/or ezetimibe (especially in those with statin intolerance or not willing to use statins) in combination with continued lifestyle intervention might be considered with the aim to achieve the recommended LDL-C goals [36]. It is important to underline that albeit statins and ezetimibe represent the more effective cholesterol-lowering drug strategies for selected low-to-moderate risk subjects, their cholesterol-lowering efficacy should be always balanced with the risk to benefit ratio in such a lower risk category of subjects. In addition, further beneficial effects (e.g., anti-inflammatory, endothelium protective, etc.) have been related to statin and ezetimibe therapy [37,38,39].
While the availability of bempedoic acid might possibly represent an additional pharmacological tool for subjects at different CV risk, especially those with statin-related side effects, the use of the more potent Proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors (i.e., anti-PCSK9 antibodies and inclisiran) is currently recommended for the highest risk patients categories.
Statins represent the cornerstone of cholesterol-lowering treatment both in the primary and secondary prevention of ASCVD. Through the inhibition of 3-hydroxy-3-methyl-glutaryl-CoA reductase (HMG-CoA), they reduce cholesterol synthesis, increase liver expression of the LDL receptor and the uptake of Apo-B containing lipoproteins. These effects result in the significant reduction of plasma LDL-C levels and, to a lesser extent, of triglyceridemia.
According to the type, dose and their cholesterol-lowering efficacy, statins are classified in low intensity, moderate and high intensity (Table 1). The efficacy of statins in reducing LDL-C levels translates into a significant prevention of ASCVD events and deaths [3, 40, 41]. Particularly, a 1 mmol/L (~ 40 mg/dL) reduction of LDL-C during statin treatment is estimated to result in a reduction of major ASCVD events by approximately 20–25% [8].
The clinical efficacy of statins may be conditioned by poor treatment adherence, which is greatly responsible for the inability to achieve the recommended LDL-C goals, especially in the long-term [42]. Statin intolerance is often reported as a major determinant of poor treatment adherence and persistence, although the most recent evidence has resized these phenomena. Accordingly, a recent meta-analysis of 176 studies with 4.143.517 patients showed that the worldwide prevalence of statin intolerance, defined according to the criteria of the International Lipid Expert Panel (ILEP), the National Lipid Association (NLA), and the Luso-Latin American Consortium (LLAC), is 9.1% [43]. Among the reported adverse effects, the most frequent are statin associated muscle symptoms (i.e., statin-associated muscle symptoms abbreviated as “SAMS”, ranging from myalgia to the rare rhabdomyolysis), the increase in liver transaminase levels and new-onset diabetes mellitus [5, 6]. Nocebo effect has been found to be a significant contributor to SAMS [43]. Irrespective of the nature of statin intolerance, statin treatment discontinuation is associated with increased risk of first or recurrent ASCVD events [44].
Other than statins, ezetimibe, a drug reducing the intestinal cholesterol absorption, represents an additional valuable therapeutic tool to reduce LDL-C levels. The efficacy in reducing LDL-C, supported by indirect evidence from genetic studies [45], is also confirmed by a large number of randomized clinical trials. Although with an interpersonal variability partially linked to the individual intestinal cholesterol absorption capacity, when administered as a monotherapy, ezetimibe is able to reduce LDL-C by 15–22% [46]. Greater LDL-C lowering efficacy (approximately 21–27%) is achievable when ezetimibe is administered in combination with statin treatment [6]. A proposed explanation for such an evidence is related to the enhanced intestinal cholesterol absorption associated with statin therapy, which may be better antagonized by ezetimibe. Although the LDL-C reductions elicited by ezetimibe are generally modest compared with statins, it should be also considered that even a small LDL-C reduction that is sustained over a long period of time may provide a benefit in terms of ASCVD risk reduction. Accordingly, mild LDL-C reduction was paralleled by a significant reduction of ASCVD risk [47, 48].
Current guidelines recommend the use of ezetimibe in combination with statins when the LDL-C goal is not achieved by using appropriated doses of high intensity statins. In addition, the use of ezetimibe should be considered as an alternative to statins in patients with confirmed statin intolerance, possibly in association with PCSK9 inhibitors, depending on patients ASCVD risk and the distance from the recommended LDL-C goal.
Importantly, the efficacy of the mentioned major lipid lowering drugs does not change in patients with lower baseline LDL-C levels [49]. This evidence, along with the recognized deleterious impact of long-term exposure to even mild LDL-C elevations [19], further support the possible use of these drug categories in selected subjects at low-to-moderate risk.
7 Improving Cardiovascular Risk Management
Optimizing cardiovascular risk management is a complex challenge in which treatment adherence is the most important goal. Complexity of treatment regimens, psychosocial factors, and poor doctor-patient communication are the three main variables that negatively affect adherence to treatment. The problem of treatment adherence is of greater significance in primary cardiovascular prevention than in secondary, as it affects up to 50 percent of patients [50]. A frequent reason for poor treatment adherence lies in statin intolerance, which is more often driven by subjective muscle complaints, termed statin-associated muscle symptoms (SAMS), than by laboratory or clinical changes [51]. Although scaled down in recent literature [43], intolerance to statin treatment remains a major problem. The use of alternative drugs such as approved PCSK9 inhibitors in the treatment of patients at high cardiovascular risk is a useful tool to overcome this issue. Some studies have shown that quality of life and treatment adherence improve after starting treatment with anti-PCSK9 [52, 53]. The use of new drugs, burdened with fewer side effects, and characterized by relative simplicity of treatment regimens, may also play a role in low to moderate risk patients in the future. Of paramount importance remains the ability to communicate to the patient the potential long-term benefits of behavioral and drug therapy, involving the patient in the decision-making process.
8 Conclusions
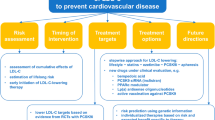
The binomial of elevation in plasma LDL-C levels and exposure to low-to-moderate CV risk is highly represented in the general population and may account for a significant increase in population attributable risk, due to both elevated frequency of this metabolic alteration and the recognized causal association between cumulative exposure to LDL-C and ASCVD risk. Based on this, early identification of subjects with even mild LDL-C elevations and appropriate 10-year CV risk stratification, possibly integrated with lifetime CV estimates in younger individuals, represent the cornerstones of CV prevention in these risk categories. The decision-making process for cholesterol-lowering therapy in patients at lower CV risk could be based on the distance of the "current" LDL-C level from the recommended LDL-C goal as a key information to plan the more appropriate prevention strategy (Fig. 1). General and specific lifestyle interventions should always represent the background of treatment of dyslipidemia in patients at low-to-moderate risk; in the condition in which the difficulty of achieving therapeutic goals with lifestyle changes is foreseeable, safe and effective cholesterol-lowering nutraceuticals might improve LDL-C goals achievement. Low dose statin therapy and/or ezetimibe might be additional therapeutic tools to be considered in case of failure to achieve the recommended LDL-C goals; however, patients’ preferences, risk to benefit ratio and pharmaco-economic issues should be considered in lower risk individuals. Additional lines of pharmacological cholesterol-lowering therapy need to be evaluated in consideration of the above-mentioned issues and specific prescription/reimbursement criteria.
References
Ference BA, Ginsberg HN, Graham I, Ray KK, Packard CJ, Bruckert E, Hegele RA, Krauss RM, Raal FJ, Schunkert H, Watts GF, Borén J, Fazio S, Horton JD, Masana L, Nicholls SJ, Nordestgaard BG, van de Sluis B, Taskinen MR, Tokgözoglu L, Catapano AL. Low-density lipoproteins cause atherosclerotic cardiovascular disease. 1. Evidence from genetic, epidemiologic, and clinical studies. A consensus statement from the European Atherosclerosis Society Consensus Panel. Eur Heart J. 2017;38(32):2459–72. https://doi.org/10.1093/eurheartj/ehx144.
Austin MA, Hutter CM, Zimmern RL, Humphries SE. Familial hypercholesterolemia and coronary heart disease: a HuGE association review. Am J Epidemiol. 2004;160(5):421–9. https://doi.org/10.1093/aje/kwh237.
Cholesterol Treatment Trialists’ (CTT) Collaboration, Baigent C, Blackwell L, Emberson J, Holland LE, Reith C, Bhala N, Peto R, Barnes EH, Keech A, Simes J, Collins R. Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170,000 participants in 26 randomised trials. Lancet (Lond, Engl). 2010;376(9753):1670–81. https://doi.org/10.1016/S0140-6736(10)61350-5.
Stamler J, Vaccaro O, Neaton JD, Wentworth D. Diabetes, other risk factors, and 12-yr cardiovascular mortality for men screened in the Multiple Risk Factor Intervention Trial. Diabetes Care. 1993;16(2):434–44. https://doi.org/10.2337/diacare.16.2.434.
Visseren F, Mach F, Smulders YM, Carballo D, Koskinas KC, Bäck M, Benetos A, Biffi A, Boavida JM, Capodanno D, Cosyns B, Crawford C, Davos CH, Desormais I, Di Angelantonio E, Franco OH, Halvorsen S, Hobbs F, Hollander M, Jankowska EA, ESC Scientific Document Group et al. 2021 ESC Guidelines on cardiovascular disease prevention in clinical practice. Eur J Prevent Cardiol 2022;29(1):5–115. https://doi.org/10.1093/eurjpc/zwab154.
Authors/Task Force Members, ESC Committee for Practice Guidelines (CPG), & ESC National Cardiac Societies. 2019 ESC/EAS guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Atherosclerosis. 2019;290:140–205. https://doi.org/10.1016/j.atherosclerosis.2019.08.014.
Stone NJ, Grundy SM. The 2018 AHA/ACC/Multi-Society Cholesterol guidelines: looking at past, present and future. Prog Cardiovasc Dis. 2019;62(5):375–83. https://doi.org/10.1016/j.pcad.2019.11.005.
Cholesterol Treatment Trialists’ (CTT) Collaborators, Mihaylova B, Emberson J, Blackwell L, Keech A, Simes J, Barnes EH, Voysey M, Gray A, Collins R, Baigent C. The effects of lowering LDL cholesterol with statin therapy in people at low risk of vascular disease: meta-analysis of individual data from 27 randomised trials. Lancet (Lond, Engl). 2012;380(9841):581–90. https://doi.org/10.1016/S0140-6736(12)60367-5.
Hippisley-Cox J, Coupland C, Brindle P. Development and validation of QRISK3 risk prediction algorithms to estimate future risk of cardiovascular disease: pro- spective cohort study. BMJ. 2017;357: j2099.
Madsen CM, Varbo A, Nordestgaard BG. Extreme high high-density lipoprotein cholesterol is paradoxically associated with high mortality in men and women: two prospective cohort studies. Eur Heart J. 2017;38(2478):2486.
Voight BF, et al. Plasma HDL cholesterol and risk of myocardial infarction: a mendelian randomisation study. Lancet (Lond, Engl). 2012;380(9841):572–80. https://doi.org/10.1016/S0140-6736(12)60312-2.
Mauvais-Jarvis F, Bairey Merz N, Barnes PJ, Brinton RD, Carrero JJ, DeMeo DL, De Vries GJ, Epperson CN, Govindan R, Klein SL, Lonardo A, Maki PM, McCullough LD, Regitz-Zagrosek V, Regensteiner JG, Rubin JB, Sandberg K, Suzuki A. Sex and gender: modifiers of health, disease, and medicine. Lancet (Lond, Engl). 2020;396(10250):565–82. https://doi.org/10.1016/S0140-6736(20)31561-0.
Pencina KM, Thanassoulis G, Wilkins JT, Vasan RS, Navar AM, Peterson ED, Pencina MJ, Sniderman AD. Trajectories of Non-HDL cholesterol across midlife: implications for cardiovascular prevention. J Am Coll Cardiol. 2019;74(1):70–9. https://doi.org/10.1016/j.jacc.2019.04.047.
Cooney MT, Selmer R, Lindman A, Tverdal A, Menotti A, Thomsen T, DeBacker G, De Bacquer D, Tell GS, Njolstad I, Graham IM, SCORE and CONOR investigators. Cardiovascular risk estimation in older persons: SCORE OP. Eur J Prevent Cardiol. 2016;23(10):1093–103. https://doi.org/10.1177/2047487315588390.
Berry JD, Dyer A, Cai X, Garside DB, Ning H, Thomas A, Greenland P, Van Horn L, Tracy RP, Lloyd-Jones DM. Lifetime risks of cardiovascular disease. N Engl J Med. 2012;366(4):321–9. https://doi.org/10.1056/NEJMoa1012848.
Volpe M, Battistoni A, Tocci G, Rosei EA, Catapano AL, Coppo R, del Prato S, Gentile S, Mannarino E, Novo S, Prisco D, Mancia G. Cardiovascular risk assessment beyond Systemic Coronary Risk Estimation: a role for organ damage markers. J Hypertens. 2012;30(6):1056–64. https://doi.org/10.1097/HJH.0b013e3283525715.
Cholesterol Treatment Trialists’ (CTT) Collaboration, Fulcher J, O’Connell R, Voysey M, Emberson J, Blackwell L, Mihaylova B, Simes J, Collins R, Kirby A, Colhoun H, Braunwald E, La Rosa J, Pedersen TR, Tonkin A, Davis B, Sleight P, Franzosi MG, Baigent C, Keech A. Efficacy and safety of LDL-lowering therapy among men and women: meta-analysis of individual data from 174,000 participants in 27 randomised trials. Lancet (Lond, Engl). 2015;385(9976):1397–405. https://doi.org/10.1016/S0140-6736(14)61368-4.
Zhang Y, Vittinghoff E, Pletcher MJ, Allen NB, Zeki-Al Hazzouri A, Yaffe K, Balte PP, Alonso A, Newman AB, Ives DG, Rana JS, Lloyd-Jones D, Vasan RS, Bibbins-Domingo K, Gooding HC, de Ferranti SD, Oelsner EC, Moran AE. Associations of blood pressure and cholesterol levels during young adulthood with later cardiovascular events. J Am Coll Cardiol. 2019;74(3):330–41. https://doi.org/10.1016/j.jacc.2019.03.529.
Abdullah SM, Defina LF, Leonard D, Barlow CE, Radford NB, Willis BL, Rohatgi A, McGuire DK, de Lemos JA, Grundy SM, Berry JD, Khera A. Long-term association of low-density lipoprotein cholesterol with cardiovascular mortality in individuals at low 10-year risk of atherosclerotic cardiovascular disease. Circulation. 2018;138(21):2315–25. https://doi.org/10.1161/CIRCULATIONAHA.118.034273.
Jeong SM, Choi S, Kim K, Kim SM, Lee G, Park SY, Kim YY, Son JS, Yun JM, Park SM. Effect of change in total cholesterol levels on cardiovascular disease among young adults. J Am Heart Assoc. 2018;7(12): e008819. https://doi.org/10.1161/JAHA.118.008819.
Milan A, Tosello F, Fabbri A, Vairo A, Leone D, Chiarlo M, Covella M, Veglio F. Arterial stiffness: from physiology to clinical implications. High Blood Pressure Cardiovasc Prevent. 2011;18(1):1–12. https://doi.org/10.2165/11588020-000000000-00000.
Wang Y, Xu D. Effects of aerobic exercise on lipids and lipoproteins. Lipids Health Dis. 2017;16(1):132. https://doi.org/10.1186/s12944-017-0515-5.
Brown L, Rosner B, Willett WW, Sacks FM. Cholesterol-lowering effects of dietary fiber: a meta-analysis. Am J Clin Nutr. 1999;69(1):30–42. https://doi.org/10.1093/ajcn/69.1.30.
Lupattelli G, Marchesi S, Lombardini R, Siepi D, Bagaglia F, Pirro M, Ciuffetti G, Schillaci G, Mannarino E. Mechanisms of high-density lipoprotein cholesterol effects on the endothelial function in hyperlipemia. Metabol Clin Exp. 2003;52(9):1191–5. https://doi.org/10.1016/s0026-0495(03)00157-4.
Lupattelli G, Marchesi S, Roscini AR, Siepi D, Gemelli F, Pirro M, Sinzinger H, Schillaci G, Mannarino E. Direct association between high-density lipoprotein cholesterol and endothelial function in hyperlipemia. Am J Cardiol. 2002;90(6):648–50. https://doi.org/10.1016/s0002-9149(02)02575-4.
Varady KA, St-Pierre AC, Lamarche B, Jones PJ. Effect of plant sterols and endurance training on LDL particle size and distribution in previously sedentary hypercholesterolemic adults. Eur J Clin Nutr. 2005;59:518–25.
Mestet ML, Plaisance EP, Ratcliff LA, et al. Aerobic exercise and postprandial lipemia in men with the metabolic syndrome. Med Sci Sports Exerc. 2008;40(12):2105–11.
Emerging Risk Factors Collaboration, Wormser D, Kaptoge S, et al. Separate and combined associations of body-mass index and abdominal adiposity with cardiovascular disease: collaborative analysis of 58 prospective studies. Lancet. 2011;377(9771):1085–95. https://doi.org/10.1016/S0140-6736(11)60105-0.
Dattilo AM, Kris-Etherton PM. Effects of weight reduction on blood lipids and lipoproteins: a meta-analysis. Am J Clin Nutr. 1992;56(2):320–8. https://doi.org/10.1093/ajcn/56.2.320.
Zomer E, Gurusamy K, Leach R, Trimmer C, Lobstein T, Morris S, James WP, Finer N. Interventions that cause weight loss and the impact on cardiovascular risk factors: a systematic review and meta-analysis. Obesity Rev. 2016;17(10):1001–11. https://doi.org/10.1111/obr.12433.
Nordmann AJ, Nordmann A, Briel M, Keller U, Yancy WS Jr, Brehm BJ, Bucher HC. Effects of low-carbohydrate vs low-fat diets on weight loss and cardiovascular risk factors: a meta-analysis of randomized controlled trials. Arch Intern Med. 2006;166(3):285–93. https://doi.org/10.1001/archinte.166.3.285.
Mannarino MR, Ministrini S, Pirro M. Nutraceuticals for the treatment of hypercholesterolemia. Eur J Intern Med. 2014;25(7):592–9. https://doi.org/10.1016/j.ejim.2014.06.008.
Pirro M, Vetrani C, Bianchi C, Mannarino MR, Bernini F, Rivellese AA. Joint position statement on “Nutraceuticals for the treatment of hypercholesterolemia” of the Italian Society of Diabetology (SID) and of the Italian Society for the Study of Arteriosclerosis (SISA). Nutr Metab Cardiovasc Dis. 2017;27(1):2–17. https://doi.org/10.1016/j.numecd.2016.11.122.
Ruscica M, Penson PE, Ferri N, Sirtori CR, Pirro M, Mancini G, Sattar N, Toth PP, Sahebkar A, Lavie CJ, Wong ND, Banach M, International Lipid Expert Panel (ILEP) and International Lipid Expert Panel Experts (alphabetically). Impact of nutraceuticals on markers of systemic inflammation: potential relevance to cardiovascular diseases—a position paper from the International Lipid Expert Panel (ILEP). Progress Cardiovasc Dis. 2021;67:40–52. https://doi.org/10.1016/j.pcad.2021.06.010.
Bianconi V, Mannarino MR, Sahebkar A, Cosentino T, Pirro M. Cholesterol-lowering nutraceuticals affecting vascular function and cardiovascular disease risk. Curr Cardiol Rep. 2018;20(7):53. https://doi.org/10.1007/s11886-018-0994-7.
Penson PE, Pirro M, Banach M. LDL-C: lower is better for longer-even at low risk. BMC Med. 2020;18(1):320. https://doi.org/10.1186/s12916-020-01792-7.
Pirro M, Schillaci G, Romagno PF, Mannarino MR, Bagaglia F, Razzi R, Pasqualini L, Vaudo G, Mannarino E. Influence of short-term rosuvastatin therapy on endothelial progenitor cells and endothelial function. J Cardiovasc Pharmacol Ther. 2009;14(1):14–21. https://doi.org/10.1177/1074248408331021.
Pirro M, Schillaci G, Savarese G, Gemelli F, Vaudo G, Siepi D, Bagaglia F, Mannarino E. Low-grade systemic inflammation impairs arterial stiffness in newly diagnosed hypercholesterolaemia. Eur J Clin Invest. 2004;34(5):335–41. https://doi.org/10.1111/j.1365-2362.2004.01345.x.
Guimarães ES, Cerda A, Dorea EL, Bernik M, Gusukuma MC, Pinto GA, Fajardo CM, Hirata MH, Hirata R. Effects of short-term add-on ezetimibe to statin treatment on expression of adipokines and inflammatory markers in diabetic and dyslipidemic patients. Cardiovasc Therapeut. 2017;35:6. https://doi.org/10.1111/1755-5922.12307.
Collins R, Reith C, Emberson J, Armitage J, Baigent C, Blackwell L, Blumenthal R, Danesh J, Smith GD, DeMets D, Evans S, Law M, MacMahon S, Martin S, Neal B, Poulter N, Preiss D, Ridker P, Roberts I, Rodgers A, Peto R, et al. Interpretation of the evidence for the efficacy and safety of statin therapy. Lancet (Lond, Engl). 2016;388(10059):2532–61. https://doi.org/10.1016/S0140-6736(16)31357-5.
Mills EJ, Rachlis B, Wu P, Devereaux PJ, Arora P, Perri D. Primary prevention of cardiovascular mortality and events with statin treatments: a network meta-analysis involving more than 65,000 patients. J Am Coll Cardiol. 2008;52(22):1769–81. https://doi.org/10.1016/j.jacc.2008.08.039.
Boekholdt SM, Hovingh GK, Mora S, Arsenault BJ, Amarenco P, Pedersen TR, LaRosa JC, Waters DD, DeMicco DA, Simes RJ, Keech AC, Colquhoun D, Hitman GA, Betteridge DJ, Clearfield MB, Downs JR, Colhoun HM, Gotto AM Jr, Ridker PM, Grundy SM, Kastelein JJ. Very low levels of atherogenic lipoproteins and the risk for cardiovascular events: a meta-analysis of statin trials. J Am Coll Cardiol. 2014;64(5):485–94. https://doi.org/10.1016/j.jacc.2014.02.615.
Bytyçi I, Penson PE, Mikhailidis DP, Wong ND, Hernandez AV, Sahebkar A, Thompson PD, Mazidi M, Rysz J, Pella D, Reiner Ž, Toth PP, Banach M. Prevalence of statin intolerance: a meta-analysis. Eur Heart J. 2022;2022:chac015. https://doi.org/10.1093/eurheartj/ehac015.
Serban MC, Colantonio LD, Manthripragada AD, Monda KL, Bittner VA, Banach M, Chen L, Huang L, Dent R, Kent ST, Muntner P, Rosenson RS. Statin intolerance and risk of coronary heart events and all-cause mortality following myocardial infarction. J Am Coll Cardiol. 2017;69(11):1386–95. https://doi.org/10.1016/j.jacc.2016.12.036.
Myocardial Infarction Genetics Consortium Investigators, Stitziel NO, Won HH, Morrison AC, Peloso GM, Do R, Lange LA, Fontanillas P, Gupta N, Duga S, Goel A, Farrall M, Saleheen D, Ferrario P, König I, Asselta R, Merlini PA, Marziliano N, Notarangelo MF, Schick U, Kathiresan S, et al. Inactivating mutations in NPC1L1 and protection from coronary heart disease. N Engl J Med. 2014;371(22):2072–82. https://doi.org/10.1056/NEJMoa1405386.
Pandor A, Ara RM, Tumur I, Wilkinson AJ, Paisley S, Duenas A, Durrington PN, Chilcott J. Ezetimibe monotherapy for cholesterol lowering in 2,722 people: systematic review and meta-analysis of randomized controlled trials. J Intern Med. 2009;265(5):568–80. https://doi.org/10.1111/j.1365-2796.2008.02062.x.
Cannon CP, Blazing MA, Giugliano RP, McCagg A, White JA, Theroux P, Darius H, Lewis BS, Ophuis TO, Jukema JW, De Ferrari GM, Ruzyllo W, De Lucca P, Im K, Bohula EA, Reist C, Wiviott SD, Tershakovec AM, Musliner TA, Braunwald E, IMPROVE-IT Investigators, et al. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med. 2015;372(25):2387–97. https://doi.org/10.1056/NEJMoa1410489.
Ouchi Y, Sasaki J, Arai H, Yokote K, Harada K, Katayama Y, Urabe T, Uchida Y, Hayashi M, Yokota N, Nishida H, Otonari T, Arai T, Sakuma I, Sakabe K, Yamamoto M, Kobayashi T, Oikawa S, Yamashita S, Rakugi H, Ito H, et al. Ezetimibe lipid-lowering trial on prevention of atherosclerotic cardiovascular disease in 75 or Older (EWTOPIA 75): a randomized controlled trial. Circulation. 2019;140(12):992–1003. https://doi.org/10.1161/CIRCULATIONAHA.118.039415.
Marcusa DP, Giugliano RP, Park JG, de Lemos JA, Cannon CP, Sabatine MS. Association of baseline low-density lipoprotein cholesterol and percentage low-density lipoprotein cholesterol reduction with statins, ezetimibe, and PCSK9 inhibition. JAMA Cardiol. 2020;6(5):1–5. https://doi.org/10.1001/jamacardio.2020.6184.
Naderi SH, Bestwick JP, Wald DS. Adherence to drugs that prevent cardiovas- cular disease: meta-analysis on 376,162 patients. Am J Med. 2012;125:882–881.
Guyton JR, Bays HE, Grundy SM, Jacobson TA. The national lipid association statin intolerance panel an assessment by the statin intolerance panel: 2014 update. J Clin Lipidol. 2014;8:S72–81.
Cesaro A, Gragnano F, Fimiani F, et al. Impact of PCSK9 inhibitors on the quality of life of patients at high cardiovascular risk. Eur J Prev Cardiol. 2020;27(5):556–8. https://doi.org/10.1177/2047487319839179.
Gragnano F, Natale F, Concilio C, et al. Adherence to proprotein convertase subtilisin/kexin 9 inhibitors in high cardiovascular risk patients: an Italian single-center experience. J Cardiovasc Med (Hagerstown). 2018;19(2):75–7. https://doi.org/10.2459/JCM.0000000000000611.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Funding
Open access funding provided by Università degli Studi di Perugia within the CRUI-CARE Agreement. The authors did not receive support from any organization for the submitted work.
Ethical declaration
This study did not need any ethical approvals or informed consent.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Figorilli, F., Mannarino, M.R., Bianconi, V. et al. Cholesterol-Lowering Therapy in Patients at Low-to-Moderate Cardiovascular Risk. High Blood Press Cardiovasc Prev 29, 327–336 (2022). https://doi.org/10.1007/s40292-022-00529-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40292-022-00529-2