Abstract
Osteoarthritis (OA), a prevalent chronic disease among the elderly, presents a complex pathogenesis and currently lacks effective treatment. Traditional observational studies are time-consuming, labor-intensive, susceptible to confounding factors, and cannot establish causal relationships. Mendelian randomization (MR) analysis, leveraging genetic variation to assess causal associations between exposures and outcomes, offers a cost-effective and efficient alternative. Over the past decade, large-scale genome-wide association studies have identified numerous genetic variants linked to OA risk factors, facilitating MR study design. In this review, we systematically identified 52 MR studies meeting specific criteria and evaluated their quality, exploring the impact of lifestyle, nutrition, comorbidities, circulating metabolites, plasma proteins, and other health factors on OA risk. We discuss the results and potential mechanisms of MR findings, addressing conflicting evidence based on existing literature and our prior research. With the ongoing expansion of genome-wide association data, we anticipate MR’s role in future OA studies to broaden, particularly in drug development research using targeted MR approaches. We thus aim for this paper to offer valuable insights for researchers and clinicians in related fields.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoarthritis (OA) is a common degenerative disease that primarily affects the elderly [1]. The pain and joint deformities associated with OA are significant contributors to disability and diminished quality of life in this age group [2]. The prevalence of osteoarthritis is increasing annually and is expected to increase by at least 50% overall by 2050 [3]. This has led to a significant rise in the costs associated with treatment and rehabilitation, posing a substantial public health burden and challenge [4,5,6,7]. Bone degeneration is a multifaceted process primarily influenced by genetic and environmental factors [8, 9]. Observational studies offer unparalleled advantages in conducting extensive epidemiological investigations and exploring causes. Nevertheless, in conventional observational analyses, it is challenging to fully address confounding factors [10]. Simultaneously, definitive proof of causal relationships between exposure factors and diseases remains elusive [11]. Findings on osteoarthritis in traditional observational studies are often disputed or conflicting, mainly due to ethical constraints and technical challenges. As a result, investigating the pathogenic factors and treatment strategies for osteoarthritis through Mendelian randomization (MR) analyses has emerged as a viable approach.
MR analysis is an observational study that uses genetic variation to make inferences about potential causal relationships between exposures (risk factors) and phenotypes [12, 13]. The genetic variants associated to exposure are randomly assigned at conception and are not influenced by factors such as acquired lifestyle [14]. Researchers consider MR to be akin to an observational randomized controlled trial, as it helps mitigate confounding factors and allows for the inference of causal associations found in observational studies [15]. Over the past 20 years, MR analyses have significantly advanced, expanding to investigate a wide range of topics. Initially focused on exploring risk factors for disease, these analyses now encompass studies on drug mechanisms of action and other relevant areas [14, 16, 17]. MR analysis mitigates ethical concerns by utilizing publicly available genetic variant data. These genetic variants are linked to exposure factors but remain unaffected by lifestyle or socioeconomic variables. The technical validity of Mendelian randomization studies has been extensively demonstrated across various degenerative conditions such as Alzheimer’s disease [18], Parkinson’s disease [19], and OA [20].
The core assumptions of MR include the relevance assumption, meaning the genetic variants selected must have a strong association with the exposure; the independence assumption, meaning the genetic variants are not influenced by any measured or unmeasured confounders; and the exclusion restriction assumption, meaning the selected genetic variants can only affect the outcome through the exposure pathway (Fig. 1). In order for causal inferences between exposure and outcome to be valid, all three assumptions must be fully satisfied [21, 22]. The main methods used in MR analyses include single-sample MR analysis and two-sample MR analysis [23]. In single-sample MR, both exposure- and outcome-related genetic variants come from the same sample, allowing for the assessment of causality within a single population. On the other hand, two-sample MR analysis utilizes genetic variants for exposure and outcome from separate independent populations, which can increase the statistical efficacy of the method by utilizing existing pooled data from large-scale GWAS consortia [24, 25]. Various large biological databases currently offer genetic variation data, including the UK Biobank, which can support single-sample or two-sample MR analyses effectively. Moreover, a growing number of disease GWAS studies are enriching genetic data for multi-ethnic and multi-population MR analyses, significantly boosting both the quantity and quality of current MR studies [25, 26].
Basic principles and core assumptions of Mendelian randomization (MR). The relevance assumption asserts that genetic variation is strongly correlated with exposure. The independence assumption indicates that genetic variation is independent of potential confounding factors. Lastly, the exclusion restriction assumption proposes that genetic variation influences the outcome solely through its effect on exposure
As in all specialties, exploring disease risk factors is necessary but often costly. MR analysis serves as a cost-effective and reliable research method that has significantly contributed to the body of evidence in OA research. Nevertheless, the number of risk factors for OA uncovered by MR is rapidly increasing, and certain causal associations appear to contradict available evidence, thereby complicating result interpretation. This review provides a comprehensive summary of MR studies related to OA published to date. A thorough search on PubMed (through December 2023), using specific keywords such as ‘Mendelian randomization,’ ‘osteoarthritis,’ ‘degenerative osteoarthropathy,’ and ‘OA,’ was conducted to identify relevant studies. The analysis encompasses a review of published reports on MR analyses in OA diseases, highlighting research progress and the challenges encountered.
Results
Description of included studies
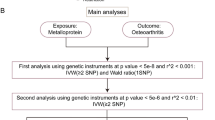
After applying several inclusion criteria, a total of 52 studies were included in this review (Fig. 2) [27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78]. Since there are no standardized quality assessment criteria, we followed the approach of several previous reviews and assessed the quality of these studies by examining their hypothesis validation [79, 80] (etable 1–3). Additionally, we summarized the main findings of these studies and indicated the source of SNPs for each study (etable 4). With a few exceptions, most studies used data from the UK Biobank, the largest human genetic database to date. Thus, MR studies of OA risk have been conducted mainly using genetic information from European populations. Factors revealed by MR to be associated with OA risk mainly include lifestyle, nutrition, comorbidities, circulating metabolites, plasma proteins, and other health factors. In several studies, OA has also been found to have a causal effect on other diseases when considered as an exposure factor. These associations were mainly found in reverse causality analyses of comorbidity MR studies, and we believe that these factors are also noteworthy for OA risk.
Lifestyle and nutritional factors
The occurrence and development of osteoarthritis are influenced by both genetic and environmental factors [8, 9]. Numerous studies have highlighted the increasing incidence of osteoarthritis, which is closely linked to modern lifestyle changes [81](Table 1). Ni et al. reported a potential causal association between sleep and osteoarthritis, suggesting that insomnia or short sleep duration may increase the risk of osteoarthritis [27]. This finding is consistent with previous cross-sectional and case-control studies [82, 83], despite an incomplete understanding of the underlying biological mechanisms. Animal studies have reported that sleep deprivation can induce a cytokine storm-like syndrome in mammals, significantly weakening the body’s immunity [84]. Another perspective suggests that insufficient melatonin secretion due to sleep disorders may be a potential mechanism for these associations [85]. Melatonin is believed to affect OA by regulating proinflammatory factors, cartilage-degrading enzymes, and other inflammatory mediators [86]. Nevertheless, these insights currently lack validation in populations. Future exploration in large prospective cohorts will help identify these causal relationships and potential mechanisms.
The impact of exercise on osteoarthritis has been a topic of debate. Upon analyzing different exercise intensities, it was discovered that varying levels of exercise intensity had contrasting effects on osteoarthritis risk. Low-intensity exercise, as predicted genetically, was found to be associated with a higher risk of osteoarthritis [30], while moderate-intensity activities (such as housework and brisk walking) were associated with a reduced risk [28, 29]. In contrast to multiple intensities of exercise, a completely inactive lifestyle, such as sedentary behavior and prolonged television viewing, can significantly increase the risk of all osteoarthritis [31]. Although MR analysis revealed multiple causal associations between exercise and OA, observational studies remain skeptical of these associations [87], suggesting that the underlying mechanisms may be complex and unclear for a long time.
The debate surrounding the dietary impact of coffee and tea remains contentious. Various observational studies have highlighted a potential link between coffee and tea consumption and a lower risk of certain diseases, such as cancer, cardiovascular disease, and osteoarthritis [88,89,90]. However, a recent Mendelian randomization (MR) study suggested a possible association between genetically predicted coffee intake and a heightened risk of osteoarthritis [32], while tea consumption was found to significantly elevate the overall risk of osteoarthritis [33]. Such contradictory results may arise from study design, where observational studies are more likely to have biases. For example, our previous work found a dose-dependent, nonlinear association between coffee and tea consumption and bone health, and this association differed significantly by gender, coffee type, and other subgroups [91]. For these causal associations revealed by MR, some studies have reported that caffeine, the main active substance in coffee and tea, and its metabolites in vivo can directly influence the anabolic actions of chondrocytes [92, 93]. Moreover, it has been proposed that caffeine intake is associated with the inflammatory cytokines IL-1 and TNF-α [94]. Furthermore, consistent findings indicated that smoking and alcohol consumption were more prominently linked to an increased risk of osteoarthritis, with both behaviors significantly raising the risk across all types of osteoarthritis [34, 35].
Comorbidities
Comorbidities play a crucial role in predicting the onset and prognosis of osteoarthritis in research [95]. Observational studies have shown that the majority of osteoarthritis patients have at least one comorbid disease, often related to the cardiovascular and digestive systems [96, 97]. Recent MR studies have identified causal links between osteoarthritis and various comorbidities, although some studies present conflicting evidence. For example, several studies by Zhao et al. noted a causal association between genetically predicted risk of osteoarthritis and an increased risk of stroke [36,37,38], while Cai et al. suggested a causal association between ischemic stroke and osteoarthritis [39]. Additionally, Cai et al. identified a positive causal relationship between genetically predicted osteoarthritis and Parkinson’s disease [39]. Furthermore, regarding cardiovascular disease, the current MR evidence is contradictory. Wang et al. reported conflicting evidence, with hip osteoarthritis linked to an increased risk of heart failure, while coronary artery disease appeared to have a protective effect on knee osteoarthritis risk [37]. Xu et al. noted a causal association between myocardial infarction and reduced osteoarthritis risk [40]. Yin et al. highlighted a causal effect of osteoarthritis on atrial fibrillation risk but a protective effect against coronary atherosclerosis [41]. Other studies have shown that genetically determined osteoporosis may reduce osteoarthritis risk [42], while osteoarthritis could increase the risk of type 2 diabetes [44], gastroesophageal reflux disease [45], and bladder cancer [46], but no inverse association was found [43]. Moreover, allergic diseases like allergic rhinitis and asthma have been genetically linked to an increased risk of osteoarthritis [47](Table 2).
It is worth noting that caution should be exercised in interpreting causal associations between osteoarthritis and comorbidities. Some studies have suggested that the association between OA and the risk of cardiovascular disease can be explained by changes in the amount of exercise, but this view is not widely supported [98, 99]. In addition to potential confounders and study quality [45], co-inheritance, on the other hand, may explain some of the causal associations between diseases [100]. Although more mechanistic research evidence is needed to validate the association between osteoarthritis and comorbidities, this suggestive MR evidence could provide new directions for osteoarthritis research and consider more comorbidities in diagnosis and treatment.
Circulating metabolites and proteins
Osteoarthritis is a multifaceted process involving metabolism, nutrition, and inflammation [101,102,103].While traditional observational studies have examined various peripheral biomarkers and established their potential link to the risk of osteoarthritis, most of these markers are assessed at specific time points in both cross-sectional and cohort studies. This makes it challenging to determine the association between longitudinal trends over time and osteoarthritis. Consequently, there has been a growing interest in MR studies in recent years, which aim to investigate the causal relationship between genetically predicted levels of peripheral markers and the risk of osteoarthritis.
The role of metabolism in the progression of bone and joint health has been extensively studied, with abnormal metabolites potentially acting as predictive markers for osteoarthritis and even exacerbating the disease [101, 104]. Recent advancements in large-scale next-generation sequencer technology and GWAS analysis have facilitated the study of metabolism-related Mendelian randomization analysis. Through 11 MR studies, causal relationships between thousands of metabolites and the risk of osteoarthritis have been investigated [48,49,50,51,52,53,54,55,56,57,58](Table 3). Eleven metabolites were found to have a causal association with an increased risk of osteoarthritis, including isovaleryl carnitine, taurocholate, kynurenine, acetaminophen 4-sulfate, homocysteine, serum iron, transferrin saturation, insulin-like growth factor-1, serum copper, and serum zinc. On the other hand, 10 metabolites were associated with a reduced risk of osteoarthritis, such as 1-linolenoylglycerophosphorylcholine, arginine, alanine (Ala), omega-3 fatty acids, omega-6 fatty acids, LDL cholesterol, transferrin, folate, vitamin B12, and serum calcium. Potential mechanistic explanations include direct effects of metabolites on chondrocytes, or altering the inflammatory state, which in turn affects the metabolic activity of cartilage. For example, nitric oxide, a metabolite produced in vivo from arginine, has strong antioxidant and anti-inflammatory effects, and oxidative stress has been widely implicated in the progression of OA [105, 106]. In contrast, kynurenine, a major degradation product of tryptophan, inhibits chondrocyte cell proliferation in a dose-dependent manner [107]. Furthermore, activation of the kynurenine-aromatic hydrocarbon receptor axis impairs chondrogenesis and chondroprotection in mesenchymal stromal cells [108]. Similarly, one of the components of Omega-6 fatty acids, arachidonic acid (AA), has been identified as a precursor to a variety of potent pro-inflammatory mediators. Consequently, Omega-6 fatty acid levels are significantly correlated with chronic inflammation in the body [109, 110].
Although a number of other metabolites have been found to be causally related to OA, the underlying mechanisms remain unclear. For instance, two separate MR studies have shown a link between increased LDL cholesterol levels and a reduced risk of osteoarthritis, contradicting previous observational studies [52, 53]. It has been suggested that LDL can reduce APOA1 levels and serum amyloid A protein in human primary chondrocytes and fibroblast-like synoviocytes, thereby alleviating joint inflammation [111]. However, we remain cautious about this hypothesis. While the specific mechanisms of action of these metabolites on osteoarthritis were not determined by the researchers, their findings offer valuable insights for future studies.
Research has demonstrated a strong association between diseases such as thyroid disorders and gonadal dysfunction with the development of osteoarthritis [112, 113]. Twelve studies have identified potential causal links between 14 protein and hormone molecular biomarkers and the risk of osteoarthritis [48, 58,59,60,61,62,63,64,65,66,67, 78](Table 4). Eight markers were found to be associated with an increased risk of osteoarthritis, including serum testosterone, serum dihydrotestosterone, sex hormone-binding globulin, glycosylated hemoglobin (HbA1c), insulin-like growth factor-binding protein 4, lipocalin, leptin, and resistin. On the other hand, six markers were genetically predictive of causality and linked to a reduced risk of osteoarthritis, such as X-11,423-O-sulfo-L-tyrosine, ADpSGEGDFXAEGGGVR, parathyroid hormone, and retinol. Some studies provide further support for existing observational evidence. For instance, elevated levels of parathyroid hormone have been shown to be causally linked to a decreased risk of osteoarthritis [60]. Previous research has demonstrated that parathyroid hormone-related proteins inhibit the hypertrophic differentiation of chondrocytes, which is beneficial for repairing cartilage damage and osteoarthritis [114]. In addition, animal studies have confirmed that PTH slows the progression of cartilage degeneration in OA mice by reducing the number of mast cells in the subchondral bone and maintaining its microstructure [115]. Mendelian randomization analyses also support this genetic correlation, offering potential targets for future interventions.
The association between glycosylated hemoglobin (HbA1c) and insulin-like growth factor-binding protein 4 with an increased risk of osteoarthritis validates the link between diabetes and osteoarthritis risk [65, 66]. Potential mechanisms include local toxicity from high glucose exposure, increased expression of cytokines and protein hydrolases, and accumulation of advanced glycation end products (AGEs). Previous studies have reported that high glucose levels promote cartilage degeneration by increasing the expression of cyclooxygenase−2 (COX−2) and MMP−13, while decreasing the synthesis of type II collagen and peroxisome proliferator-activated receptor γ (PPARγ) [116]. Rasheed et al. further showed that AGEs induce IL−6 and IL−8 expression in human OA chondrocytes through the receptor for AGEs (RAGE) activation pathway, leading to oxidative stress [117]. We believe that the effects of hyperglycemia on OA are multifaceted due to its ability to induce a wide range of metabolic disorders.
MR reveals a causal relationship between adipokines, such as adipose transport proteins, leptin, and resistin, and the risk of osteoarthritis; however, the underlying mechanisms are more complex. Zhao et al. reported that high-dose leptin induces cell cycle arrest and senescence in chondrogenic progenitor cells through activation of the p53/p21 pathway and inhibition of the Sirt1 pathway, thereby promoting OA [118]. Moreover, Feng et al. reviewed the mechanisms of lipocalin’s effects on OA, suggesting that lipocalin may regulate chondrocyte autophagy and promote cellular pyroptosis in OA through multiple pathways [119]. On the other hand, it has been noted that while adipokines may trigger chondrocyte apoptosis, they primarily act as inflammatory mediators [120].
Numerous studies have confirmed the presence of chronic low-grade inflammation in the progression of osteoarthritis [121]. Traditional observational studies have shown that elevated levels of systemic and local inflammation play a role in the progression of osteoarthritis. Additionally, abnormal peripheral inflammatory factors have been found to be somewhat predictive of the trajectory of osteoarthritis [122]. Two MR studies have demonstrated a causal link between genetically predicted levels of inflammatory factors and the risk of osteoarthritis [66, 78]. The expression of immune-related CD25-associated traits has been linked to a lower risk of osteoarthritis [69]. Additionally, lesser-known factors such as macrophage inflammatory protein−1β and tumor necrosis factor beta have also shown potential in reducing the risk of osteoarthritis [68](Table 5). By extrapolating the association between these genetically predicted inflammatory factors and osteoarthritis risk, we can gain a deeper understanding of the biological mechanisms underlying inflammation in osteoarthritis.
Other health factors
Due to the intricate pathogenesis of osteoarthritis, numerous MR studies have investigated potential connections between osteoarthritis and various health indicators such as childhood obesity, sarcopenia, body mass index, age at menstruation and childbearing, blood pressure, bone density, and gut flora [70,71,72,73,74,75,76,77](Table 6). The association between childhood obesity and adult health has been supported by numerous studies, including those focusing on osteoarthritis. A 25-year cohort study revealed a significant link between childhood overweight and knee pain, stiffness, and dysfunction in adulthood [123]. MR studies have confirmed a genetically predictable causal relationship between muscle loss or underweight and significant metabolic disturbances [70], which can lead to deterioration in general health. While the association with osteoarthritis has not received much attention [124], MR studies have found a significant causal link between genetically determined muscle loss and an increased risk of all osteoarthritis [71]. An abnormal rise in basal metabolism is often indicative of endocrine disruption or nutritional imbalance, a systemic alteration that has been associated with the risk for a variety of diseases [125]. Several previous MR studies have found a causal association between genetically predicted elevations in basal metabolic rate and osteoporosis and several cancers [126, 127]. Similarly, in osteoarthritis, MR studies have validated genetically predicted causal connections [73]. Further research is needed to understand the mechanisms and clinical implications of protective factors like age at menarche/first childbearing and gut flora in osteoarthritis [71, 77].
Interestingly, conflicting evidence was found in multiple MR studies regarding the relationship between bone mineral density (BMD) and osteoarthritis. Funck et al. reported a positive causal relationship between increased femoral neck BMD and increased risk of osteoarthritis, with each standard deviation increase in BMD increasing the risk of overall osteoarthritis, hip osteoarthritis, and knee osteoarthritis by 14%, 22%, and 18%, respectively [74]. Jiang et al. supported this finding by showing that genetically higher whole-body BMD increased the risk of hip osteoarthritis [76]. In addition, Liu et al. also noted that osteoporosis (decreased BMD) had a genetically predicted protective effect against osteoarthritis [42]. Some studies have proposed the hypothesis that increased bone density is associated with subchondral bone sclerosis and increased bone resorption [128, 129]. We are cautious about this hypothesis. In contrast, Qu et al. found that genetically reduced BMD in specific areas was associated with an increased risk of osteoarthritis [75]. The potential mechanistic explanation for this association is that bone loss in the subchondral bone in patients with OP leads to articular surface collapse and uneven articular cartilage stress, resulting in secondary osteophyte proliferation and cartilage damage [130]. Overall, the association between BMD and OA remains controversial. We believe that these conflicting results stem primarily from differences in the instrumental variables selected for MR studies.
Future research directions
Current MR analyses provide extensive evidence of OA risk, at least in a statistically significant manner. Regarding the need for additional MR studies, we believe that future research should focus on MR analyses that help develop treatment options for OA, such as drug-targeted MR.
Drug-targeted MR studies, an emerging research method, aim to evaluate whether changing exposure through therapeutic manipulation of a target can lead to expected outcomes [131,132,133]. In the field of osteoarthritis drug development, MR analysis is being used to investigate therapeutic mechanisms and identify specific targets. For instance, metformin, a well-known drug used for diabetes treatment, has shown protective effects against osteoarthritis [134, 135]. Research indicates that metformin may protect cartilage by targeting cells or activating specific receptors [136, 137]. Drug-targeted MR analyses have further confirmed that metformin targets AMP-activated protein kinase (AMPK) and growth differentiation factor 15 (GDF-15) have genetically predicted protective effects against osteoarthritis [138]. This strengthens the evidence for developing targeted therapies for osteoarthritis.
Combining MR with other methodologies, such as multi-omics analysis or MR-based multi-omics data analysis, can significantly improve result confidence. Additionally, given that most MR analyses are currently conducted using genetic data from European populations, there is a need to perform them in other ethnicities and subpopulations in the future to enhance result generalizability. Finally, causal associations revealed by MR should not solely rely on statistical significance but should be complemented with experimental studies to validate the functional roles of identified genetic variants and pathways in OA pathogenesis.
Limitations
However, we should be aware that MR analysis has its limitations in advancing disease research, particularly in reporting or interpreting MR results [139]. Common pitfalls include chain imbalance [13], horizontal pleiotropy [140], vertical pleiotropy [141], weak instrumental variables [142], and population stratification [143]. It is essential to carefully consider these shortcomings to ensure more accurate results when conducting or interpreting MR analyses. In addition, we should also note that observational studies have limitations that may deviate from real-world scenarios, even if the analysis process and interpretation of the results are fully compliant with norms. For example, in a drug-targeted MR analysis, the authors found a causal association between genetic variants in PCSK9 inhibitors and statins and the risk of cognitive impairment [144]. Ference et al. argued that the results of this study were unreliable in the real world, as PCSK9 monoclonal antibodies do not cross the blood-brain barrier, and also contradicted previous MR reports [145]. It is crucial to recognize that while observational studies can guide research direction and provide a broader perspective, statistically significant associations may not always align with actual outcomes.
Conclusion
In conclusion, MR analysis offers valuable insights into OA risk research. MR’s research model, based on genetic information, minimizes confounding factors and ethical concerns. In the exploration of OA risk, MR can efficiently and rapidly reveal potential associations and provide causal evidence. The application of drug-targeted MR further enhances the utility of MR analysis in OA therapeutic research, offering precise guidance for drug development. Looking ahead, there is a growing trend towards integrating MR with other methods, which can enhance result confidence. Additionally, adhering to rigorous research methodology and reporting standards is crucial for improving the quality and reliability of MR studies in the field of OA.
Data availability
No datasets were generated or analysed during the current study.
References
He X, Hu W, Zhang Y et al (2023) Cellular senescence in skeletal disease: mechanisms and treatment. Cell Mol Biol Lett 28(1):88
Rothrauff B, Tang Q, Wang J et al (2022) Osteoarthritis is positively associated with self-reported sleep trouble in older adults. Aging Clin Exp Res 34(11):2835–2843
Global regional, national burden of osteoarthritis (2023) 1990–2020 and projections to 2050: a systematic analysis for the global burden of Disease Study 2021. Lancet Rheumatol 5(9):e508–e522
Costăchescu B, Niculescu AG, Teleanu RI et al (2022) Recent advances in managing spinal intervertebral discs degeneration. Int J Mol Sci. 23 (12)
Clynes MA, Harvey NC, Curtis EM et al (2020) The epidemiology of osteoporosis. Br Med Bull 133(1):105–117
Dong Y, Kang H, Peng R et al (2022) Global, Regional, and National Burden of Low Bone Mineral Density from 1990 to 2019: results from the global burden of Disease Study 2019. Front Endocrinol (Lausanne) 13:870905
Safiri S, Kolahi AA, Smith E et al (2020) Global, regional and national burden of osteoarthritis 1990–2017: a systematic analysis of the global burden of Disease Study 2017. Ann Rheum Dis 79(6):819–828
Wang F, Cai F, Shi R et al (2016) Aging and age related stresses: a senescence mechanism of intervertebral disc degeneration. Osteoarthritis Cartilage 24(3):398–408
Ikegawa S (2013) The genetics of common degenerative skeletal disorders: osteoarthritis and degenerative disc disease. Annu Rev Genomics Hum Genet 14:245–256
D’Onofrio BM, Sjölander A, Lahey BB et al (2020) Accounting for confounding in Observational studies. Annu Rev Clin Psychol 16:25–48
Harper S (2019) A future for Observational Epidemiology: clarity, credibility, transparency. Am J Epidemiol 188(5):840–845
Emdin CA, Khera AV, Kathiresan S (2017) Mendelian Randomization Jama 318(19):1925–1926
Lawlor DA, Harbord RM, Sterne JA et al (2008) Mendelian randomization: using genes as instruments for making causal inferences in epidemiology. Stat Med 27(8):1133–1163
Smith GD, Ebrahim S (2003) Mendelian randomization’: can genetic epidemiology contribute to understanding environmental determinants of disease? Int J Epidemiol 32(1):1–22
Haycock PC, Burgess S, Wade KH et al (2016) Best (but oft-forgotten) practices: the design, analysis, and interpretation of mendelian randomization studies. Am J Clin Nutr 103(4):965–978
Bovijn J, Lindgren CM, Holmes MV (2020) Genetic variants mimicking therapeutic inhibition of IL-6 receptor signaling and risk of COVID-19. Lancet Rheumatol 2(11):e658–e659
Skrivankova VW, Richmond RC, Woolf BAR et al (2021) Strengthening the reporting of Observational studies in Epidemiology using mendelian randomization: the STROBE-MR Statement. JAMA 326(16):1614–1621
Anderson EL, Howe LD, Wade KH et al (2020) Education, intelligence and Alzheimer’s disease: evidence from a multivariable two-sample mendelian randomization study. Int J Epidemiol 49(4):1163–1172
Bottigliengo D, Foco L, Seibler P et al (2022) A mendelian randomization study investigating the causal role of inflammation on Parkinson’s disease. Brain 145(10):3444–3453
Ho J, Mak CCH, Sharma V et al (2022) Mendelian randomization studies of lifestyle-related risk factors for Osteoarthritis: a PRISMA Review and Meta-Analysis. Int J Mol Sci 23:19
Davies NM, Holmes MV, Davey Smith G (2018) Reading Mendelian randomisation studies: a guide, glossary, and checklist for clinicians. Bmj. 362, k601
Pierce BL, Ahsan H, Vanderweele TJ (2011) Power and instrument strength requirements for mendelian randomization studies using multiple genetic variants. Int J Epidemiol 40(3):740–752
Gala H, Tomlinson I (2020) The use of mendelian randomisation to identify causal cancer risk factors: promise and limitations. J Pathol 250(5):541–554
Larsson SC, Butterworth AS, Burgess S (2023) Mendelian randomization for cardiovascular diseases: principles and applications. Eur Heart J 44(47):4913–4924
Burgess S, Davies NM, Thompson SG (2016) Bias due to participant overlap in two-sample mendelian randomization. Genet Epidemiol 40(7):597–608
Burgess S, Scott RA, Timpson NJ et al (2015) Using published data in mendelian randomization: a blueprint for efficient identification of causal risk factors. Eur J Epidemiol 30(7):543–552
Ni J, Zhou W, Cen H et al (2022) Evidence for causal effects of sleep disturbances on risk for osteoarthritis: a univariable and multivariable mendelian randomization study. Osteoarthritis Cartilage 30(3):443–450
Li X, Wang S, Liu W et al (2023) Causal effect of physical activity and sedentary behaviors on the risk of osteoarthritis: a univariate and multivariate mendelian randomization study. Sci Rep 13(1):19410
Qiu P, Wu J, Kui L et al (2023) Causal effects of walking pace on osteoarthritis: a two-sample mendelian randomization study. Front Genet 14:1266158
Wang B, Liu Y, Zhang YC et al (2023) Assessment of causal effects of physical activity on the risk of osteoarthritis: a two-sample mendelian randomization study. BMC Med Genomics 16(1):237
Cao Z, Li Q, Li Y et al (2023) Causal association of leisure sedentary behavior with arthritis: a mendelian randomization analysis. Semin Arthritis Rheum 59:152171
Zhang Y, Fan J, Chen L et al (2021) Causal Association of Coffee Consumption and total, knee, hip and self-reported osteoarthritis: a mendelian randomization study. Front Endocrinol (Lausanne) 12:768529
Li G, Zhang Z, Liu Y (2022) Genetically predicted tea intake increases the risk of osteoarthritis: a mendelian randomization study. Front Genet 13:1004392
Wen MT, Liang XZ, Luo D et al (2023) Plasma lipids, alcohol intake frequency and risk of Osteoarthritis: a mendelian randomization study. BMC Public Health 23(1):1327
Ni J, Wang P, Yin KJ et al (2022) Does smoking protect against developing osteoarthritis? Evidence from a genetically informed perspective. Semin Arthritis Rheum 55:152013
Zhao H, Zhu J, Ju L et al (2022) Osteoarthritis & stroke: a bidirectional mendelian randomization study. Osteoarthritis Cartilage 30(10):1390–1397
Wang Z, Kang C, Xu P et al (2022) Osteoarthritis and cardiovascular disease: a mendelian randomization study. Front Cardiovasc Med 9:1025063
Shen Y, Li F, Cao L et al (2022) Hip osteoarthritis and the risk of Lacunar Stroke: a two-sample mendelian randomization study. Genes (Basel). 13 (9)
Cai Y, Zhang G, Liang J et al (2021) Causal relationships between Osteoarthritis and Senile Central nerve system dysfunction: a bidirectional two-sample mendelian randomization study. Front Aging Neurosci 13:793023
Xu H, Ling Y, Jiang H et al (2022) Osteoarthritis, coronary artery disease, and myocardial infarction: a mendelian randomization study. Front Cardiovasc Med 9:892742
Yin M, Xu W, Pang J et al (2023) Causal relationship between osteoarthritis with atrial fibrillation and coronary atherosclerosis: a bidirectional mendelian randomization study of European ancestry. Front Cardiovasc Med 10:1213672
Lin L, Luo P, Yang M et al (2022) Causal relationship between osteoporosis and osteoarthritis: a two-sample mendelian randomized study. Front Endocrinol (Lausanne) 13:1011246
Cui Z, Feng H, He B et al (2020) Type 2 diabetes and glycemic traits are not causal factors of Osteoarthritis: a two-sample mendelian randomization analysis. Front Genet 11:597876
Xing X, Wang Y, Pan F et al (2023) Osteoarthritis and risk of type 2 diabetes: a two-sample mendelian randomization analysis. J Diabetes 15(11):987–993
Xu H, Wei J, Chen D et al (2023) Assessing causality between osteoarthritis and gastrointestinal disorders: a mendelian randomization study. Sci Rep 13(1):19603
Zhang X, Wen Z, Xing Z et al (2023) The causal relationship between osteoarthritis and bladder cancer: a mendelian randomization study. Cancer Med. 13 (1)
Baker MC, Robinson WH, Ostrom Q (2023) Genetic association between atopic disease and osteoarthritis. Osteoarthritis Cartilage 32(2):220–225
Gu Y, Jin Q, Hu J et al (2023) Causality of genetically determined metabolites and metabolic pathways on osteoarthritis: a two-sample mendelian randomization study. J Transl Med 21(1):357
Cui ZY, Feng H, He BC et al (2023) Causal associations between serum amino acid levels and osteoarthritis: a mendelian randomization study. Osteoarthritis Cartilage 31(8):1121–1131
Hong H, Chen L, Zhong Y et al (2023) Associations of Homocysteine, Folate, and Vitamin B12 with Osteoarthritis: A Mendelian Randomization Study. Nutrients. 15 (7)
Li X, Lu Z, Qi Y et al (2023) The Role of Polyunsaturated Fatty Acids in Osteoarthritis: Insights from a Mendelian Randomization Study. Nutrients. 15 (22)
Hindy G, Åkesson KE, Melander O et al (2019) Cardiometabolic Polygenic Risk scores and osteoarthritis outcomes: a mendelian randomization study using data from the Malmö Diet and Cancer Study and the UK Biobank. Arthritis Rheumatol 71(6):925–934
Meng H, Jiang L, Song Z et al (2022) Causal Associations of Circulating Lipids with Osteoarthritis: A Bidirectional Mendelian Randomization Study. Nutrients. 14 (7)
Ruan G, Ying Y, Lu S et al (2023) The effect of systemic iron status on osteoarthritis: a mendelian randomization study. Front Genet 14:1122955
Xu J, Zhang S, Tian Y et al (2022) Genetic Causal Association between Iron Status and Osteoarthritis: A Two-Sample Mendelian Randomization. Nutrients. 14 (18)
Hartley A, Sanderson E, Paternoster L et al (2021) Mendelian randomization provides evidence for a causal effect of higher serum IGF-1 concentration on risk of hip and knee osteoarthritis. Rheumatology (Oxford) 60(4):1676–1686
Zhou J, Liu C, Sun Y et al (2021) Genetically predicted circulating levels of copper and zinc are associated with osteoarthritis but not with rheumatoid arthritis. Osteoarthritis Cartilage 29(7):1029–1035
Qu Z, Yang F, Hong J et al (2021) Causal relationship of serum nutritional factors with osteoarthritis: a mendelian randomization study. Rheumatology (Oxford) 60(5):2383–2390
Qu Z, Yang F, Yan Y et al (2021) A mendelian randomization study on the role of serum parathyroid hormone and 25-hydroxyvitamin D in osteoarthritis. Osteoarthritis Cartilage 29(9):1282–1290
Huang G, Zhong Y, Li W et al (2021) Causal relationship between parathyroid hormone and the risk of Osteoarthritis: a mendelian randomization study. Front Genet 12:686939
Yan YS, Qu Z, Yu DQ et al (2021) Sex steroids and osteoarthritis: a mendelian randomization study. Front Endocrinol (Lausanne) 12:683226
Qu Z, Huang J, Yang F et al (2020) Sex hormone-binding globulin and arthritis: a mendelian randomization study. Arthritis Res Ther 22(1):118
Huang G, Li W, Zhong Y et al (2023) Mendelian randomization to evaluate the causal relationship between liver enzymes and the risk of six specific bone and joint-related diseases. Front Immunol 14:1195553
Huang L, Xie Y, Jin T et al (2023) Diet-derived circulating antioxidants and risk of knee osteoarthritis, hip osteoarthritis and rheumatoid arthritis: a two-sample mendelian randomization study. Front Med (Lausanne) 10:1147365
Chen L, Jia C, Yang H (2023) Causal effect of higher glycated hemoglobin (HbA1c) levels on knee osteoarthritis risk: a mendelian randomization study. Rheumatol Ther 10(1):239–247
Han BX, Yan SS, Yu H et al (2023) Causal effects of plasma proteome on osteoporosis and osteoarthritis. Calcif Tissue Int 112(3):350–358
Fan J, Zhu J, Sun L et al (2021) Causal association of adipokines with osteoarthritis: a mendelian randomization study. Rheumatology (Oxford) 60(6):2808–2815
Su D, Ai Y, Zhu G et al (2023) Genetically predicted circulating levels of cytokines and the risk of osteoarthritis: a mendelian randomization study. Front Genet 14:1131198
Luo H, Zhu Y, Guo B et al (2023) Causal relationships between CD25 on immune cells and hip osteoarthritis. Front Immunol 14:1247710
Cao Z, Wu Y, Li Q et al (2022) A causal relationship between childhood obesity and risk of osteoarthritis: results from a two-sample mendelian randomization analysis. Ann Med 54(1):1636–1645
Yang J, Liu P, Wang S et al (2023) Causal relationship between Sarcopenia and osteoarthritis: a bi-directional two-sample mendelian randomized study. Eur J Med Res 28(1):327
Wang B, Wu J, Li H et al (2022) Using genetic instruments to estimate the causal effect of hormonal reproductive factors on osteoarthritis. Front Public Health 10:941067
Zhou J, Wei P, Yi F et al (2023) The association between basal metabolic rate and osteoarthritis: a mendelian randomization study. BMC Med Genomics 16(1):258
Funck-Brentano T, Nethander M, Movérare-Skrtic S et al (2019) Causal factors for knee, hip, and Hand Osteoarthritis: a mendelian randomization study in the UK Biobank. Arthritis Rheumatol 71(10):1634–1641
Qu Y, Chen S, Han M et al (2023) Osteoporosis and osteoarthritis: a bi-directional mendelian randomization study. Arthritis Res Ther 25(1):242
Jiang L, Jiang Y, Wang A et al (2022) The causal association between bone mineral density and risk of osteoarthritis: a mendelian randomization study. Front Endocrinol (Lausanne) 13:1021083
Yu XH, Yang YQ, Cao RR et al (2021) The causal role of gut microbiota in development of osteoarthritis. Osteoarthritis Cartilage 29(12):1741–1750
Huang G, Li W, Kan H et al (2023) Genetic influences of the effect of circulating inflammatory cytokines on osteoarthritis in humans. Osteoarthritis Cartilage 31(8):1047–1055
van de Luitgaarden IAT, van Oort S, Bouman EJ et al (2022) Alcohol consumption in relation to cardiovascular diseases and mortality: a systematic review of mendelian randomization studies. Eur J Epidemiol 37(7):655–669
Alhassan E, Nguyen K, Hochberg MC et al (2024) Causal factors for Osteoarthritis: a scoping review of mendelian randomization studies. Arthritis Care Res (Hoboken) 76(3):366–375
Berenbaum F, Wallace IJ, Lieberman DE et al (2018) Modern-day environmental factors in the pathogenesis of osteoarthritis. Nat Rev Rheumatol 14(11):674–681
Park HM, Kwon YJ, Kim HS et al (2019) Relationship between Sleep Duration and Osteoarthritis in Middle-aged and older women: a Nationwide Population-based study. J Clin Med. 8 (3)
Jacob L, Smith L, Konrad M et al (2021) Association between sleep disorders and osteoarthritis: a case-control study of 351,932 adults in the UK. J Sleep Res. 30 (6), e13367
Sang D, Lin K, Yang Y et al Prolonged sleep deprivation induces a cytokine-storm-like syndrome in mammals. Cell 2023, 186 (25), 5500–5516e5521
Jahanban-Esfahlan R, Mehrzadi S, Reiter RJ et al (2018) Melatonin in regulation of inflammatory pathways in rheumatoid arthritis and osteoarthritis: involvement of circadian clock genes. Br J Pharmacol 175(16):3230–3238
Hossain FM, Hong Y, Jin Y et al (2019) Physiological and pathological role of circadian hormones in Osteoarthritis: dose-dependent or Time-Dependent? J Clin Med 8:9
Gates LS, Perry TA, Golightly YM et al (2022) Recreational physical activity and risk of incident knee osteoarthritis: an International Meta-Analysis of Individual participant-Level Data. Arthritis Rheumatol 74(4):612–622
Lim CO, Park HJ, Koo BM et al (2022) Analysis of correlation between the consumption of beverages and the risk of radiographic knee osteoarthritis in Korean people: a cross-sectional study using the Fifth Korea National Health and Nutrition Examination Survey (KNHANES V-1, 2). Med (Baltim). 101 (37), e30105
Chen Y, Zhang Y, Zhang M et al (2022) Consumption of coffee and tea with all-cause and cause-specific mortality: a prospective cohort study. BMC Med 20(1):449
Chieng D, Kistler PM (2022) Coffee and tea on cardiovascular disease (CVD) prevention. Trends Cardiovasc Med 32(7):399–405
Zhang S, Wu S, Xia B et al (2024) Association of coffee and tea consumption with osteoporosis risk: A prospective study from the UK biobank. Bone. 117135
Guillan-Fresco M, Franco-Trepat E, Alonso-Perez A et al (2020) Caffeine, a risk factor for Osteoarthritis and Longitudinal Bone Growth Inhibition. J Clin Med. 9 (4)
Choi H, Choi Y, Kim J et al (2017) Longitudinal bone growth is impaired by direct involvement of caffeine with chondrocyte differentiation in the growth plate. J Anat 230(1):117–127
Kuo CL, Liu ST, Chang YL et al (2018) Zac1 regulates IL-11 expression in osteoarthritis. Oncotarget 9(65):32478–32495
Kadam UT, Croft PR (2007) Clinical comorbidity in osteoarthritis: associations with physical function in older patients in family practice. J Rheumatol 34(9):1899–1904
King LK (2023) Osteoarthritis and comorbidity: time for action. Osteoarthritis Cartilage 31(4):423–424
Swain S, Sarmanova A, Coupland C et al (2020) Comorbidities in Osteoarthritis: a systematic review and Meta-analysis of Observational studies. Arthritis Care Res (Hoboken) 72(7):991–1000
Jacob L, Tanislav C, Kostev K (2021) Osteoarthritis and incidence of stroke and transient ischemic attack in 320,136 adults followed in general practices in the United Kingdom. Joint Bone Spine 88(2):105104
Booth FW, Roberts CK, Laye MJ (2012) Lack of exercise is a major cause of chronic diseases. Compr Physiol 2(2):1143–1211
Arruda AL, Hartley A, Katsoula G et al (2023) Genetic underpinning of the comorbidity between type 2 diabetes and osteoarthritis. Am J Hum Genet 110(8):1304–1318
Jansen NEJ, Molendijk E, Schiphof D et al (2023) Metabolic syndrome and the progression of knee osteoarthritis on MRI. Osteoarthritis Cartilage
Kluzek S, Newton JL, Arden NK (2015) Is osteoarthritis a metabolic disorder? Br Med Bull 115(1):111–121
Zhang S, Wang D, Zhao J et al (2024) Metabolic syndrome increases osteoarthritis risk: findings from the UK Biobank prospective cohort study. BMC Public Health 24(1):233
Valdes AM (2020) Metabolic syndrome and osteoarthritis pain: common molecular mechanisms and potential therapeutic implications. Osteoarthritis Cartilage 28(1):7–9
Zhai G (2021) Clinical relevance of biochemical and metabolic changes in osteoarthritis. Adv Clin Chem 101:95–120
Zhang S, Wang L, Kang Y et al (2023) Nanomaterial-based reactive oxygen species scavengers for osteoarthritis therapy. Acta Biomater 162:1–19
Logters TT, Laryea MD, Jager M et al (2010) Kynurenine inhibits chondrocyte proliferation and is increased in synovial fluid of patients with septic arthritis. J Orthop Res 28(11):1490–1496
Wang X, Zhao Y, Li S et al (2023) Activation of the kynurenine-aryl hydrocarbon receptor axis impairs the chondrogenic and chondroprotective effects of human umbilical cord-derived mesenchymal stromal cells in osteoarthritis rats. Hum Cell 36(1):163–177
Bersch-Ferreira AC, Sampaio GR, Gehringer MO et al (2017) Association between polyunsaturated fatty acids and inflammatory markers in patients in secondary prevention of cardiovascular disease. Nutrition 37:30–36
Innes JK, Calder PC (2018) Omega-6 fatty acids and inflammation. Prostaglandins Leukot Essent Fat Acids 132:41–48
de Seny D, Cobraiville G, Charlier E et al (2015) Apolipoprotein-A1 as a damage-associated molecular patterns protein in osteoarthritis: ex vivo and in vitro pro-inflammatory properties. PLoS ONE. 10 (4), e0122904
Chen S, Sun X, Zhou G et al (2022) Association between sensitivity to thyroid hormone indices and the risk of osteoarthritis: an NHANES study. Eur J Med Res 27(1):114
Hussain SM, Cicuttini FM, Alyousef B et al (2018) Female hormonal factors and osteoarthritis of the knee, hip and hand: a narrative review. Climacteric 21(2):132–139
Zhang W, Chen J, Zhang S et al (2012) Inhibitory function of parathyroid hormone-related protein on chondrocyte hypertrophy: the implication for articular cartilage repair. Arthritis Res Ther 14(4):221
Morita Y, Ito H, Ishikawa M et al (2018) Subchondral bone fragility with meniscal tear accelerates and parathyroid hormone decelerates articular cartilage degeneration in rat osteoarthritis model. J Orthop Res 36(7):1959–1968
Cannata F, Vadala G, Ambrosio L et al (2020) Osteoarthritis and type 2 diabetes: from pathogenetic factors to therapeutic intervention. Diabetes Metab Res Rev. 36 (3), e3254
Rasheed Z, Akhtar N, Haqqi TM (2011) Advanced glycation end products induce the expression of interleukin-6 and interleukin-8 by receptor for advanced glycation end product-mediated activation of mitogen-activated protein kinases and nuclear factor-kappab in human osteoarthritis chondrocytes. Rheumatology (Oxford) 50(5):838–851
Zhao X, Dong Y, Zhang J et al (2016) Leptin changes differentiation fate and induces senescence in chondrogenic progenitor cells. Cell Death Dis. 7 (4), e2188
Feng X, Xiao J, Bai L (2022) Role of adiponectin in osteoarthritis. Front Cell Dev Biol 10:992764
Wang T, He C (2018) Pro-inflammatory cytokines: the link between obesity and osteoarthritis. Cytokine Growth Factor Rev 44:38–50
Knights AJ, Redding SJ, Maerz T (2023) Inflammation in osteoarthritis: the latest progress and ongoing challenges. Curr Opin Rheumatol 35(2):128–134
Pan F, Tian J, Cicuttini F et al (2022) Prospective Association between Inflammatory Markers and knee cartilage volume loss and Pain Trajectory. Pain Ther 11(1):107–119
Antony B, Jones G, Venn A et al (2015) Association between childhood overweight measures and adulthood knee pain, stiffness and dysfunction: a 25-year cohort study. Ann Rheum Dis 74(4):711–717
Biolo G, Cederholm T, Muscaritoli M (2014) Muscle contractile and metabolic dysfunction is a common feature of Sarcopenia of aging and chronic diseases: from sarcopenic obesity to cachexia. Clin Nutr 33(5):737–748
Zheng L, Zhang Z, Sheng P et al (2021) The role of metabolism in chondrocyte dysfunction and the progression of osteoarthritis. Ageing Res Rev 66:101249
Wu E, Ni J, Tao L et al (2022) A bidirectional mendelian randomization study supports the causal effects of a high basal metabolic rate on colorectal cancer risk. PLoS ONE. 17 (8), e0273452
Hemani G, Tilling K, Davey Smith G (2017) Orienting the causal relationship between imprecisely measured traits using GWAS summary data. PLoS Genet. 13 (11), e1007081
Hardcastle SA, Gregson CL, Deere KC et al (2013) High bone mass is associated with an increased prevalence of joint replacement: a case-control study. Rheumatology (Oxford) 52(6):1042–1051
Hardcastle SA, Dieppe P, Gregson CL et al (2014) Osteophytes, enthesophytes, and high bone mass: a bone-forming triad with potential relevance in osteoarthritis. Arthritis Rheumatol 66(9):2429–2439
Lajeunesse D, Reboul P (2003) Subchondral bone in osteoarthritis: a biologic link with articular cartilage leading to abnormal remodeling. Curr Opin Rheumatol 15(5):628–633
Georgakis MK, Gill D (2021) Mendelian randomization studies in stroke: exploration of risk factors and drug targets with Human Genetic Data. Stroke 52(9):2992–3003
Lin J, Zhou J, Xu Y (2023) Potential drug targets for multiple sclerosis identified through mendelian randomization analysis. Brain 146(8):3364–3372
Reay WR, Cairns MJ (2021) Advancing the use of genome-wide association studies for drug repurposing. Nat Rev Genet 22(10):658–671
Song Y, Wu Z, Zhao P (2022) The effects of metformin in the treatment of osteoarthritis: current perspectives. Front Pharmacol 13:952560
Baker MC, Sheth K, Liu Y et al (2023) Development of osteoarthritis in adults with type 2 diabetes treated with metformin vs a Sulfonylurea. JAMA Netw Open. 6 (3), e233646
Li J, Zhang B, Liu WX et al (2020) Metformin limits osteoarthritis development and progression through activation of AMPK signalling. Ann Rheum Dis 79(5):635–645
Li D, Ruan G, Zhang Y et al (2023) Metformin attenuates osteoarthritis by targeting chondrocytes, synovial macrophages and adipocytes. Rheumatology (Oxford) 62(4):1652–1661
Zhang Y, Li D, Zhu Z et al (2022) Evaluating the impact of metformin targets on the risk of osteoarthritis: a mendelian randomization study. Osteoarthritis Cartilage 30(11):1506–1514
Davey Smith G, Hemani G (2014) Mendelian randomization: genetic anchors for causal inference in epidemiological studies. Hum Mol Genet 23(R1):R89–98
Verbanck M, Chen CY, Neale B et al (2018) Detection of widespread horizontal pleiotropy in causal relationships inferred from mendelian randomization between complex traits and diseases. Nat Genet 50(5):693–698
Wang Z, Emmerich A, Pillon NJ et al (2022) Genome-wide association analyses of physical activity and sedentary behavior provide insights into underlying mechanisms and roles in disease prevention. Nat Genet 54(9):1332–1344
Burgess S, Thompson SG (2011) Bias in causal estimates from mendelian randomization studies with weak instruments. Stat Med 30(11):1312–1323
Wacholder S, Rothman N, Caporaso N (2000) Population stratification in epidemiologic studies of common genetic variants and cancer: quantification of bias. J Natl Cancer Inst 92(14):1151–1158
Rosoff DB, Bell AS, Jung J et al (2022) Mendelian randomization study of PCSK9 and HMG-CoA reductase inhibition and cognitive function. J Am Coll Cardiol 80(7):653–662
Ference BA (2022) Interpreting the clinical implications of drug-target mendelian randomization studies. J Am Coll Cardiol 80(7):663–665
Acknowledgements
This study was supported by the Program for Youth Innovation in Future Medicine, Chongqing Medical University (no.w0155) and the Natural Science Foundation of Chongqing (no. 2023NSCQ-MSX0742).
Author information
Authors and Affiliations
Contributions
X.L and S.Z provided direction and guidance throughout the preparation of this manuscript. Q.L and S.Z conducted the literature review, drafted. Q.Y, Y.D and H.Y helped a lot in the collection of materials and beautification of tables. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Luo, Q., Zhang, S., Yang, Q. et al. Causal factors for osteoarthritis risk revealed by mendelian randomization analysis. Aging Clin Exp Res 36, 176 (2024). https://doi.org/10.1007/s40520-024-02812-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40520-024-02812-9