Abstract
Background
Contrast-associated acute kidney injury (CA-AKI) is a common complication after coronary angiography (CAG), which brings a poor prognosis. But up to now, there were fewer studies to discuss the incidence of CA-AKI comprehensively. We comprehensively explore the incidence of CA-AKI after coronary angiography.
Methods
We searched Medline, Embase, and Cochrane Database of Systematic Reviews (to 30th June 2019). We evaluated the world’s incidence of the CA-AKI, and associated mortality, and to described geographic variations according to countries, regions, and economies. CA-AKI was defined as an increase in serum creatinine ≥ 0.5 mg/dl or ≥ 25% within 72 h. Random effects model meta-analyses and meta-regressions was performed to derive the sources of heterogeneity.
Results
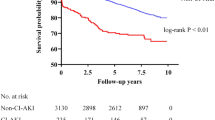
A total of 134 articles (1,211,106 participants) were included in our meta-analysis. Most studies originated from China, Japan, Turkey and United States, from upper middle income and high income countries. The pooled incidence of CA-AKI after coronary angiography was 12.8% (95% CI 11.7–13.9%), and the CA-AKI associated mortality was 20.2% (95% CI 10.7–29.7%). The incidence of CA-AKI and the CA-AKI associated mortality were not declined over time (Incidence rate change: 0.23% 95% CI − 0.050 to 0.510 p = 0.617; Mortality rate change: − 1.05% 95% CI − 3.070 to 0.970 p = 0.308, respectively).
Conclusion
CA-AKI was a universal complication in many regions, and the burden of CA-AKI remains severe. In clinical practice, physicians should pay more attention to the occurrence and active prevention and treatment of CA-AKI.
Graphic abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
As the burden of cardiovascular disease increases, cardiovascular diseases have become the world's leading cause of death, and the number of coronary angiography continues to rise [1]. Contrast-associated acute kidney injury (CA-AKI) is one of the major complications of coronary angiography (CAG), and the occurrence of CA-AKI is related to the toxicity of contrast agent, which leads to kidney function loss, with apoptosis and tubular necrosis [2, 3]. The high osmolality contrast agent and ionic contrast agent were related to the high risk of CA-AKI [4, 5]. At the same time, the risk of CA-AKI increases with each additional 100 ml of contrast agent [6]. In addition, renal ischemic injury caused by vasoactive substances such as endothelin, nitric oxide and prostaglandins may increase the risk of CA-AKI [7,8,9]. The occurrence of CA-AKI is significantly associated with prolonged hospitalization and with an increase in short and long-term mortality [10, 11].
Numerous studies have suggested that the incidence of CA-AKI ranged from 3 to 50% [12,13,14]. Chalikias et al. mentioned that the current incidence of CA-AKI was difficult to calculate due to the influence of many factors (CA-AKI definition, prevalence of comorbidity, including diabetes mellitus, pre-existing renal disease, heart failure, anemia) and was also related to clinical setting [15]. Furthermore, it has been confirmed in previous studies that age > 75 years, hypotension, intra-aortic balloon pump and hypoproteinemia are risk factors for CA-AKI [6, 16]. In this context, Hoste suggested that the current incidence of CA-AKI and the prognosis varied, depending on the diagnostic criteria and population characteristics [17]. Up to now, the incidence of CA-AKI around the world has not been systematically reviewed, and further research is needed to determine its stages of severity and associated mortality.
This meta-analysis was conducted to estimate the world incidence and mortality associated with CA-AKI after coronary angiography, and to describe variations according to countries, regions, comorbidities and economic aspects.
Materials and methods
Data sources and literature search
A literature search was performed in MEDLINE, Embase and the Cochrane Database with the searching terms “contrast-induced acute kidney injury”, “risk” and “acute kidney injury” (Supplemental Item 1). The search was limited to human studies written in English and published before 30th June, 2019 (PROSPERO register number: CRD42019121534).
Study selection
Studies were included if they fulfilled the following criteria: (1) studies reporting on the risk factors of CA-AKI and (2) retrospective and prospective observational studies. Studies were excluded if they were classified as (1) duplicate articles; (2) contained no precise definitions of CA-AKI; (3) randomized controlled trials, meta-analyses, case reports, editorials, animal studies. At least two authors independently assessed the citations retrieved in the electronic search and identified eligible studies.
Data extraction and quality assessment
At least two independent authors (ZB Lun and LW Liu) assessed each study by screening the title, abstract or full-text independently. Then these two reviewers extracted data on the study characteristics, the incidence of CA-AKI and associated potentially modifiable risk factors. Newcastle–Ottawa Scale was performed to evaluate the quality of the included studies. The authors resolved discrepancies in study selection, data extraction and assessment of study quality through discussion with an arbitrator (Y Liu).
Definitions and outcomes
According to the geographical scheme designed by the United Nations Statistics Division, countries are classified into different continents and world regions [18]. Countries’ economies were assessed according to three ranges of gross national income per capita derived from the World Bank’s classification of income of economies: lower middle, upper middle, and high income countries [19, 20]. Using the World Health Organization’s World Health Statistics data, countries are also classified according to the percentage of total national health expenditure to gross domestic product (GDP) [21].
We evaluated the incidence of CA-AKI by three definitions. The definition of CA-AKI was an increase in serum creatinine ≥ 0.5 mg/dl or ≥ 25% from baseline within 72 h after exposure to contrast. In addition, we also analyzed the incidence of two other definitions of CA-AKI, as reported in Supplementary material. The two definitions of CA-AKI were: an increase in serum creatinine ≥ 0.3 mg/dl or ≥ 50% from baseline within 72 h and the criteria of acute kidney injury network (AKI stage 1, ≥ 0.3 mg/dl absolute or 1.5 to 2.0-fold relative increase in serum creatinine; AKI stage 2, > 2- to threefold increase in serum creatinine; AKI stage 3, > threefold increase in serum creatinine or serum creatinine > 4.0 mg/dl with an acute increase of > 0.5 mg/dl) or RIFLE criteria (risk, injury, failure, loss of kidney function and end-stage kidney disease) [22] or Kidney-disease-improving-global outcomes[23]. The CA-AKI associated mortality was considered as CA-AKI associated all-cause mortality.
Statistics analysis
The pooled incidence rates of CA-AKI and mortality were assessed with random-effects or fixed-effects models, based on the heterogeneity of included studies. Heterogeneity was quantified by the Q statistic and I2 statistic, which describes the percentage of total variation across studies due to heterogeneity and not due to sampling error [24]. If I2 was > 50%, a random-effects model was used. Otherwise, the fixed-effects model was adopted [25]. Subgroup analyses and meta-regression were performed for the following subsets of studies: percutaneous coronary intervention related studies, CAG or percutaneous coronary intervention related studies, clinical comorbidities, CA-AKI definitions, countries, continents, latitude, countries income classification, countries total wealth. Funnel plot was used to assess publication bias [26]. Publication bias was considered significant when p < 0.05. All analyses were performed with STATA (version 13.0) and R software (version 3.6.1; R Core Team, Vienna, Austria).
Results
Study characteristics and quality assessment
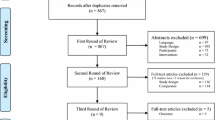
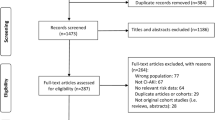
A total of 18,868 potentially relevant citations were identified and screened; 17,732 articles were retrieved for detailed evaluation, 134 of which fulfilled eligibility criteria (Supplemental References), representing 1.2 million patients from 24 countries worldwide (Fig. 1). Characteristics of all 134 studies are displayed in Supplemental Table 1. All studies were published in English and publication spanned 17 years. In the Table 1, most studies (23.2%) originated from Turkey (23 studies), followed by China (20 studies), United States (15 studies), Japan (10 studies), Italy (9 studies), Korea (7 studies). The top-three world zones where studies were conducted were Asia (67 studies), North America (15 studies), and Europe (14 studies). Most studies (48.5%) originated from high-income countries (48 studies) followed by upper middle income countries (47 studies). Most studies also originated from countries that spent over 5% of GDP on total health expenditure (94 studies). There were 97 studies (98.0%) that originated from countries located north of the equator. All studies were considered high-quality studies. Among them, 78 studies got 9 points, 45 studies got 8 points, and the rest got 7 points (Supplement Table 2).
Data synthesis
Pooled incidence rate of CA-AKI
We pooled the incidence rate of CA-AKI by 99 studies, the pooled rate of CA-AKI was 12.8% (95% CI 11.7–13.9%). In addition, the pooled incidence rate of CA-AKI in patients with percutaneous coronary intervention was 13.3% (95% CI 11.9–14.6%). When we study by clinical setting, the incidence rate in patients with chronic kidney disease, diabetes mellitus and ST-elevation myocardial infarction were 14.4%, 12.0% and 12.8%, respectively (Table 1). No publication bias was found, as confirmed by Egger’s tests (p = 0.07), and the funnel plot was shown in Fig. 2. As Table 2 knows, the pooled incidence rate of CA-AKI will not change with the growth of the year (rate change: 0.23%, 95% CI − 0.050 to 0.510, p = 0.103). At the same time, we found that compared with the incidence from 2002 to 2013, the incidence of CA-AKI from 2014 to 2019 did not increase (rate change: 0.08%, 95% CI − 0.1.490 to 3.070, p = 0.497).
The pooled incidence rate of other definitions of CA-AKI were 15.1% (95% CI 12.9–17.4%) and 16.0% (95% CI 12.6–19.5%), respectively (detailed in Supplementary Table 3).
Pooled CA-AKI Associated Mortality Rate
As shown in Table 3, the pooled CA-AKI associated mortality rate was 20.2% (95% CI 10.7–29.7%). As Table 2 knows, the pooled CA-AKI associated mortality rate will not change with the growth of the year (rate change: − 1.05%, 95% CI − 3.070 to 0.970, p = 0.308). Compared with the pooled CA-AKI associated mortality rate from 2002 to 2013, the mortality rate of CA-AKI from 2014 to 2019 did not increase (rate change: − 10.2%, 95% CI − 0.27.710 to 7.340, p = 0.497).
The pooled CA-AKI associated mortality of other two definitions were 20.6% (95% CI 9.1–32.1%) and 27.5% (95% CI 7.8–47.2%) (Supplementary Table 3).
Pooled incidence and mortality associated with CA-AKI and variability around the World
The incidence rate in Turkey and the United States was 14.7%, in China was 12.4%, in Japan was 14.5% and in Italy was 12.9%. The pooled incidence rate in Asia was higher than in Europe and North America (13.2% versus 12.7% and 10.4%). According to country income classification, the incidence of CA-AKI in high income countries was slightly lower than that of upper middle income countries (11.4% versus 14.1%). And the pooled incidence of CA-AKI in countries with spent over 10% GDP on total health expenditure was less than the countries with 5–10% GDP on total health expenditure (12.2% versus 13.1%). Mortality associated with CA-AKI in Turkey and the United States were 14.6% and 35.1%, China was 3.9%, Japan was 19.9%. Compared with upper middle income countries, the high income countries has higher mortality associated with CA-AKI (26.4% versus 10.0%). The pooled CA-AKI associated mortality in countries with spent over 10% GDP on total health expenditure was higher than the countries with 5–10% GDP on total health expenditure (28.7% versus 12.1%) (Tables 1, 3 and Fig. 3).
Discussion
Our meta-analysis is the first article with the aim to assess the global incidence of CA-AKI. Since 1946 (the earliest period of Medline retrieval), we have identified a total of 18,868 large cohort studies. The pooled incidence and mortality of CA-AKI after coronary angiography were 12.8% and 20.2%, respectively. Our results showed that the incidence was not significantly related to national economic conditions and the percentage of gross domestic product spent on total health expenditure. And, as time goes on, the incidence of CA-AKI has not changed.
We have pooled the incidence of CA-AKI through 99 studies and found that this is similar to the incidence reported in previous studies [27, 28]. And we also compared different definitions and found that the incidence of other definitions of CA-AKI was both higher than that defined an increase in serum creatinine ≥ 0.5 mg/dl or ≥ 25% from baseline within 72 h after exposure to contrast. Both Chen and Centola’s studies have confirmed that the incidence of CA-AKI is affected by the presence of different definitions [29, 30]. In addition, we also analyzed the incidence of patients with different complications. We found that the incidence of patients with ST-elevation myocardial infarction and chronic kidney disease were higher than the pooled incidence (12.8%), because previous studies have confirmed that ST-elevation myocardial infarction and chronic kidney disease are risk factors of CA-AKI [31, 32]. However, the incidence of CA-AKI in diabetic patients was lower than pooled incidence, which may be related to the small sample size.
We observed that the pooled incidence of CA-AKI in most countries was close to the incidence estimated by us. However, the incidence data reported in Germany, Brazil and Iran were significantly higher, which may be associated with the number of studies and patient characteristics. The patients included in the studies from Germany were elderly, and this was a risk factor for CA-AKI [33, 34]. The increased incidence in Brazil and Iran may be related to the small number of patients included. Because only two studies from Africa and South America were included, it is not possible to draw conclusions from these settings. The incidence rate was 13.2% in Asia, followed by Europe (12.7%) and North America (10.4%, 15 studies were from the United States). The incidence in high income countries was lower than upper middle income countries. Countries that spend more than 10% of GDP on health also have lower rates than countries that spend more than 5–10% of GDP. This may be related to the scale of national economic development and the performance of the medical and health security system. Moreover, a previous study suggested that AKI had a high incidence in intensive care unit in high income countries, making it difficult to prevent. Conversely, AKI in middle income countries often appears in rural health centers and hospitals as well as in large urban hospitals, and it is possible that it can be prevented through public health initiatives [17]. This may be one of the reasons for the difference. However, the opposite was true for CA-AKI associated mortality, which was higher in high income countries and in countries with high health expenditures as a percentage of GDP. The reason may be that the high mortality rate was at least partly due to other concomitant chronic diseases, which are more frequent in high income countries. Previous studies have confirmed that, in high income countries, patients with AKI were more likely to have risk factors that affect prognosis such as old age, heart failure, diabetes, and hypoproteinemia [35,36,37,38,39].
The incidence of CA-AKI is unclear, which may leads clinicians to underestimate CA-AKI. Our meta-analysis may therefore be of great significance to the future development and formulation of CA-AKI-related public health policies for the scientific community, government, and medical staff. Through meta-regression analysis, we can observe that the incidence and mortality of CA-AKI have not significantly changed since 2002. In addition, we also found that the incidence of CA-AKI from 2014 to 2019 did not change compared with the incidence from 2002 to 2013. Compared with the guidelines on myocardial revascularization of the European Society of Cardiology in 2010, the 2014 guidelines on myocardial revascularization of the European Society of Cardiology recommended that patients who undergo coronary angiography should be assessed for risk of CA-AKI [40, 41]. Moreover, the 2014 guidelines canceled the recommendation of ß-blockers, angiotensin-converting enzyme inhibitors or statins and reduced the level of evidence for N-acetylcysteine for chronic kidney disease patients. This may mean that the relevant prevention and treatment strategies of CA-AKI have not achieved actual benefits.
Our meta-analysis had several limitations. Firstly, the studies included in our analysis were targeted to the identification of risk factors of CA-AKI. However, our meta-study included as many as 134 articles, and the sample size was large. Secondly, we had included few studies from Africa and South America, which may lead to the lack of representativeness of results. Studies from the southern hemisphere were also very rare, and more studies are needed in these settings in the future. Thirdly, the various definitions of CA-AKI may have added to the heterogeneity. However, we analyzed the incidence of common definitions of CA-AKI, which makes our results equally representative. Our research compares morbidity and mortality in various countries, but international comparisons may vary depending upon geentic, environmental and social elements.
Conclusion
According to our metaanalysis, CA-AKI is a common complication in various regions of the world, and incidence and mortality are still high and did not decrease with time. This means that health care managers and and clinicians should pay more attention to CA-AKI to reduce its incidence and improve its prognosis.
Code availability
Not applicable.
Change history
17 June 2021
A Correction to this paper has been published: https://doi.org/10.1007/s40620-021-01090-2
References
Roth GA, Johnson C, Abajobir A et al (2017) Global, regional, and national burden of cardiovascular diseases for 10 causes, 1990 to 2015. J Am Coll Cardiol 70(1):1–25. https://doi.org/10.1016/j.jacc.2017.04.052
Tervahartiala P, Kivisaari L, Kivisaari R, Vehmas T, Virtanen I (1997) Structural changes in the renal proximal tubular cells induced by iodinated contrast media. Nephron 76(1):96–102. https://doi.org/10.1159/000190147
Guitterez NV, Diaz A, Timmis GC, O’Neill WW, Stevens MA, Sandberg KR, McCullough PA (2002) Determinants of serum creatinine trajectory in acute contrast nephropathy. J Interv Cardiol 15(5):349–354. https://doi.org/10.1111/j.1540-8183.2002.tb01067.x
Barrett BJ, Carlisle EJ (1993) Metaanalysis of the relative nephrotoxicity of high- and low-osmolality iodinated contrast media. Radiology 188(1):171–178. https://doi.org/10.1148/radiology.188.1.8511292
Rudnick MR, Goldfarb S, Wexler L, Ludbrook PA, Murphy MJ, Halpern EF, Hill JA, Winniford M, Cohen MB, VanFossen DB (1995) Nephrotoxicity of ionic and nonionic contrast media in 1196 patients: a randomized trial. The Iohexol cooperative study. Kidney Int 47(1):254–261. https://doi.org/10.1038/ki.1995.32
Mehran R, Aymong ED, Nikolsky E, Lasic Z, Iakovou I, Fahy M, Mintz GS, Lansky AJ, Moses JW, Stone GW, Leon MB, Dangas G (2004) A simple risk score for prediction of contrast-induced nephropathy after percutaneous coronary intervention: development and initial validation. J Am Coll Cardiol 44(7):1393–1399. https://doi.org/10.1016/j.jacc.2004.06.068
Briguori C, Quintavalle C, De Micco F, Condorelli G (2011) Nephrotoxicity of contrast media and protective effects of acetylcysteine. Arch Toxicol 85(3):165–173. https://doi.org/10.1007/s00204-010-0626-5
Keaney JJ, Hannon CM, Murray PT (2013) Contrast-induced acute kidney injury: how much contrast is safe? Nephrol Dial Transplant 28(6):1376–1383. https://doi.org/10.1093/ndt/gfs602
Romano G, Briguori C, Quintavalle C, Zanca C, Rivera NV, Colombo A, Condorelli G (2008) Contrast agents and renal cell apoptosis. Eur Heart J 29(20):2569–2576. https://doi.org/10.1093/eurheartj/ehn197
Mehran R, Dangas GD, Weisbord SD (2019) Contrast-associated acute kidney injury. N Engl J Med 380(22):2146–2155. https://doi.org/10.1056/NEJMra1805256
Sun G, Chen P, Wang K, Li H, Chen S, Liu J, He Y, Song F, Liu Y, Chen JY (2019) Contrast-induced nephropathy and long-term mortality after percutaneous coronary intervention in patients with acute myocardial infarction. Angiology 70(7):621–626. https://doi.org/10.1177/0003319718803677
Aubry P, Brillet G, Catella L, Schmidt A, Benard S (2016) Outcomes, risk factors and health burden of contrast-induced acute kidney injury: an observational study of one million hospitalizations with image-guided cardiovascular procedures. BMC Nephrol 17(1):167. https://doi.org/10.1186/s12882-016-0385-5
Okumura N, Hayashi M, Ishii H, Yoshikawa D, Yasuda Y, Goto M, Matsuo S, Oiso Y, Murohara T (2014) Novel preprocedural and acute-phase postprocedural predictive factors for contrast-induced kidney injury in CKD patients. Int J Cardiol 172(2):e293-296. https://doi.org/10.1016/j.ijcard.2013.12.193
Jiang W, Yu J, Xu J, Shen B, Wang Y, Luo Z, Wang C, Ding X, Teng J (2018) Impact of cardiac catheterization timing and contrast media dose on acute kidney injury after cardiac surgery. BMC Cardiovasc Disord 18(1):191. https://doi.org/10.1186/s12872-018-0928-8
Chalikias G, Drosos I, Tziakas DN (2016) Contrast-induced acute kidney injury: an update. Cardiovasc Drugs Ther 30(2):215–228. https://doi.org/10.1007/s10557-015-6635-0
Cirit M, Toprak O, Yesil M, Bayata S, Postaci N, Pupim L, Esi E (2006) Angiotensin-converting enzyme inhibitors as a risk factor for contrast-induced nephropathy. Nephron Clin Pract 104(1):c20-27. https://doi.org/10.1159/000093255
Hoste EAJ, Kellum JA, Selby NM, Zarbock A, Palevsky PM, Bagshaw SM, Goldstein SL, Cerda J, Chawla LS (2018) Global epidemiology and outcomes of acute kidney injury. Nat Rev Nephrol 14(10):607–625. https://doi.org/10.1038/s41581-018-0052-0
United Nations Statistics Division. Composition of macro geographical (continental) regions, geographical sub-regions, and selected economic and other groupings. http://millenniumindicators.un.org/unsd/methods/m49/m49regin.htm. Accessed 1 Sept 2020
TheWorld Bank. How we classify countries. http://data.worldbank.org/about/country-classifications. Accessed 1 Sept 2020
TheWorld Bank. World Development Indicators database. Gross national income per capita 2011, Atlas method and PPP. http://databank.worldbank.org/databank/download/GNIPC.pdf. Accessed 1 Sept 2020
World Health Organization. World Health Statistics 2012. http://www.who.int/gho/publications/world_health_statistics/2012/en/index.html. Accessed 1 Sept 2020
Bellomo R, Ronco C, Kellum JA, Mehta RL, Palevsky P, Acute Dialysis Quality Initiative w (2004) Acute renal failure—definition, outcome measures, animal models, fluid therapy and information technology needs: the Second International Consensus Conference of the Acute Dialysis Quality Initiative (ADQI) Group. Crit Care 8(4):R204-212. https://doi.org/10.1186/cc2872
Kellum JA, Lameire N, Group KAGW (2013) Diagnosis, evaluation, and management of acute kidney injury: a KDIGO summary (part 1). Crit Care 17(1):204. https://doi.org/10.1186/cc11454
Higgins JP, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327(7414):557–560. https://doi.org/10.1136/bmj.327.7414.557
Higgins JP, Thompson SG (2002) Quantifying heterogeneity in a meta-analysis. Stat Med 21(11):1539–1558. https://doi.org/10.1002/sim.1186
DerSimonian R, Laird N (1986) Meta-analysis in clinical trials. Control Clin Trials 7(3):177–188. https://doi.org/10.1016/0197-2456(86)90046-2
Barbieri L, Verdoia M, Schaffer A, Cassetti E, Marino P, Suryapranata H, De Luca G, Novara Atherosclerosis Study G (2015) Uric acid levels and the risk of contrast induced nephropathy in patients undergoing coronary angiography or PCI. Nutr Metab Cardiovasc Dis 25(2):181–186. https://doi.org/10.1016/j.numecd.2014.08.008
Nough H, Eghbal F, Soltani M, Nejafi F, Falahzadeh H, Fazel H, Sheikhvatan M (2013) Incidence and main determinants of contrast-induced nephropathy following coronary angiography or subsequent balloon angioplasty. Cardiorenal Med 3(2):128–135. https://doi.org/10.1159/000351981
Chen SQ, Liu Y, Smyth B, Li HL, Sun GL, Chen ZJ, Zhou YL (2019) Clinical implications of contrast-induced nephropathy in patients without baseline renal dysfunction undergoing coronary angiography. Heart Lung Circ 28(6):866–873. https://doi.org/10.1016/j.hlc.2018.04.291
Centola M, Lucreziotti S, Salerno-Uriarte D, Sponzilli C, Ferrante G, Acquaviva R, Castini D, Spina M, Lombardi F, Cozzolino M, Carugo S (2016) A comparison between two different definitions of contrast-induced acute kidney injury in patients with ST-segment elevation myocardial infarction undergoing primary percutaneous coronary intervention. Int J Cardiol 210:4–9. https://doi.org/10.1016/j.ijcard.2016.02.086
Han B, Li Y, Dong Z, Wan Q, Shen H, Li J, Wei M, Shen C (2018) Diastolic dysfunction predicts the risk of contrast-induced nephropathy and outcome post-emergency percutaneous coronary intervention in AMI patients with preserved ejection fraction. Heart Vessels 33(10):1149–1158. https://doi.org/10.1007/s00380-018-1165-x
Fan PC, Chen TH, Lee CC, Tsai TY, Chen YC, Chang CH (2018) ADVANCIS Score predicts acute kidney injury after percutaneous coronary intervention for acute coronary syndrome. Int J Med Sci 15(5):528–535. https://doi.org/10.7150/ijms.23064
Kanic V, Kompara G, Suran D, Tapajner A, Naji FH, Sinkovic A (2019) Acute kidney injury in patients with myocardial infarction undergoing percutaneous coronary intervention using radial versus femoral access. BMC Nephrol 20(1):28. https://doi.org/10.1186/s12882-019-1210-8
Ni Z, Liang Y, Xie N, Liu J, Sun G, Chen S, Ye J, He Y, Guo W, Tan N, Chen J, Liu Y, Chen Z, Wang S (2019) Simple pre-procedure risk stratification tool for contrast-induced nephropathy. J Thorac Dis 11(4):1597–1610. https://doi.org/10.21037/jtd.2019.04.69
Siew ED, Deger SM (2012) Recent advances in acute kidney injury epidemiology. Curr Opin Nephrol Hypertens 21(3):309–317. https://doi.org/10.1097/MNH.0b013e3283521d95
Roberts G, Phillips D, McCarthy R, Bolusani H, Mizen P, Hassan M, Hooper R, Saddler K, Hu M, Lodhi S, Toynton E, Geen J, Lodhi V, Grose C, Phillips A (2015) Acute kidney injury risk assessment at the hospital front door: what is the best measure of risk? Clin Kidney J 8(6):673–680. https://doi.org/10.1093/ckj/sfv080
Yu MY, Lee SW, Baek SH, Na KY, Chae DW, Chin HJ, Kim S (2017) Hypoalbuminemia at admission predicts the development of acute kidney injury in hospitalized patients: a retrospective cohort study. PLoS ONE 12(7):e0180750. https://doi.org/10.1371/journal.pone.0180750
Wiedermann CJ, Wiedermann W, Joannidis M (2010) Hypoalbuminemia and acute kidney injury: a meta-analysis of observational clinical studies. Intensive Care Med 36(10):1657–1665. https://doi.org/10.1007/s00134-010-1928-z
Wiedermann CJ, Wiedermann W, Joannidis M (2017) Causal relationship between hypoalbuminemia and acute kidney injury. World J Nephrol 6(4):176–187. https://doi.org/10.5527/wjn.v6.i4.176
Task Force on Myocardial Revascularization of the European Society of C, the European Association for Cardio-Thoracic S, European Association for Percutaneous Cardiovascular I, Wijns W, Kolh P, Danchin N, Di Mario C, Falk V, Folliguet T, Garg S, Huber K, James S, Knuuti J, Lopez-Sendon J, Marco J, Menicanti L, Ostojic M, Piepoli MF, Pirlet C, Pomar JL, Reifart N, Ribichini FL, Schalij MJ, Sergeant P, Serruys PW, Silber S, Sousa Uva M, Taggart D (2010) Guidelines on myocardial revascularization. Eur Heart J 31(20):2501–2555. https://doi.org/10.1093/eurheartj/ehq277
Authors/Task Force m, Windecker S, Kolh P, Alfonso F, Collet JP, Cremer J, Falk V, Filippatos G, Hamm C, Head SJ, Juni P, Kappetein AP, Kastrati A, Knuuti J, Landmesser U, Laufer G, Neumann FJ, Richter DJ, Schauerte P, Sousa Uva M, Stefanini GG, Taggart DP, Torracca L, Valgimigli M, Wijns W, Witkowski A (2014) 2014 ESC/EACTS Guidelines on myocardial revascularization: The Task Force on Myocardial Revascularization of the European Society of Cardiology (ESC) and the European Association for Cardio-Thoracic Surgery (EACTS) Developed with the special contribution of the European Association of Percutaneous Cardiovascular Interventions (EAPCI). Eur Heart J 35(37):2541–2619. https://doi.org/10.1093/eurheartj/ehu278
Funding
The study is supported by the National Natural Science Foundation of China (Grant no. 81670339 and 81970311), the Beijing Lisheng Cardiovascular Pilot Foundation (Grant no. LHJJ201612127), the Science and Technology Planning Project of Guangdong Province (Grant no. 2014B070706010), the Science and Technology Planning Project of Guangzhou (Grant no. 201704020124), and Dengfeng Project in Guangdong Province (DFJH201919 and DFJH2020026).
Author information
Authors and Affiliations
Contributions
ZBL, LWL, GZC and MY carried out the literature search and drafted the manuscript. ZBL, JL, JFY, JYC and YL were involved in the design of the study and performed the statistical analysis. BW, JJL, YQY, SQC and YBH assisted with the database searches and data extraction and examined the manuscript. EYC, JFY, JYC and YL conceived the study, participated in the study design and was instrumental in drafting the manuscript and critically reviewed the manuscript. All authors read and agreed to the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethics approval
This article does not contain any studies with human participants performed by any of the authors.
Consent to participate
Not applicable.
Consent for publication
All authors consent for publication.
Availability of data and material
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original article has been updated: Due to Affiliation 1 and 2 update.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lun, Z., Liu, L., Chen, G. et al. The global incidence and mortality of contrast-associated acute kidney injury following coronary angiography: a meta-analysis of 1.2 million patients. J Nephrol 34, 1479–1489 (2021). https://doi.org/10.1007/s40620-021-01021-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40620-021-01021-1