Abstract
Purpose of Review
Pioneering work in rodents has shown that the reactivation of recently acquired memories during sleep is a key mechanism underlying the beneficial effect of sleep on memory consolidation. In this review, we consider recent evidence of memory reactivation processes in human sleep.
Recent Findings
The precise temporal coupling of sleep spindles to slow oscillations during non-rapid eye movement sleep plays a central role in sleep-associated memory consolidation. Both correlational studies and studies directly manipulating oscillatory activity in the sleeping brain have confirmed that spindles coupled to slow oscillations are better predictors of memory than uncoupled spindles and that the greatest memory benefit comes when spindles are tightly coupled to the up-state of the slow oscillation. Recent evidence suggests that memory content is reactivated during sleep, with a functional benefit for memory performance after sleep. Reactivation events are time-locked around slow oscillation-spindle coupling events, as well as sharp-wave ripples in hippocampus.
Summary
Memory reactivation, which is facilitated by slow oscillation-spindle coupling events, can be observed during human sleep and shows promise as a prime mechanism underlying sleep’s beneficial effects on memory.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Seminal work in rodents has demonstrated a process of memory reactivation during sleep, especially non-rapid eye movement (NREM) sleep [1,2,3]. In these studies, reactivation is indicated by patterns of neuronal firing observed at learning being replayed during sleep in temporally compressed sequences [3,4,5]. Although primarily documented in hippocampus, memory replay also occurs in learning-related cortical sites [6], and co-ordinated replay of the same experience in both hippocampus and neocortex has been observed [7, 8]. Memory replay in sleep appears to play a fundamental role in consolidation, that is, the strengthening, stabilisation, and integration of newly acquired memories [2, 9, 10]. Evidence for this comes from studies showing that the occurrence of replay events during sleep is correlated with subsequent memory improvement [6] and others showing that the disruption of replay events through electrical stimulation and optogenetic techniques eliminates any sleep-related performance gain [11,12,13].
According to the influential Active Systems Consolidation (ASC) framework, coordinated memory reactivation between hippocampus and learning-related cortical areas underpins the overnight strengthening and integration of newly formed memory representations [14]. This hippocampal-cortical dialogue during sleep is believed to be driven by a finely tuned and hierarchical interplay of ~ 80–150 Hz hippocampal sharp-wave ripples, thalamocortical sleep spindles (~ 10–16 Hz), and global slow oscillations (SOs ~ 1 Hz) [15, 16] (Fig. 1A). In hippocampus, ripples encompassing reactivation events [4, 19] are nested into the excitable troughs of sleep spindles, which propagate to neocortex where they induce long-term potentiation (LTP) and synaptic plasticity in local circuits [20,21,22,23,24,25]. A subset of spindles are in turn nested into the up-states of SOs, periods of neuronal depolarisation and heightened communication throughout the brain, therefore providing a critical time window for spindle propagation across neocortex [26, 27•]. The precise temporal coupling of SOs, sleep spindles, and sharp wave ripples thus represents a physiological mechanism underpinning coordinated communication and reactivation between neural ensembles of hippocampus and neocortex [28,29,30].
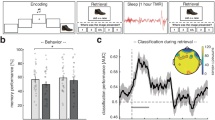
Mechanisms of memory consolidation during human non-rapid eye movement (NREM) sleep. A Cardinal neural oscillations of NREM sleep. Sharp wave ripples in hippocampus support memory replay. Ripples nest into the troughs of thalamocortical sleep spindles, which induce synaptic plasticity in learning-related cortical sites. Spindles are coupled to the excitable up-states of global slow oscillations (SOs). B Common metrics to quantify SO-spindle coupling in humans. Top occurrence: the amount of coupling, often expressed as the coupled spindle density (# coupled events/min) or % of spindles that are coupled. Bottom phase: the average phase of the SO that spindles are coupled to, typically expressed in degrees or radians (direction of the arrow in left-hand circular phase plot. Right: mapping of SO phase to circular plot). Spindles preferentially couple to the rising up-state or peak of the SO. Consistency: amount of variability in spindle coupling phase, often quantified as the mean vector length (length of the arrow in the circular phase plot). C Manipulations to enhance SO-spindle coupling. Left panel: pharmacology. Following administration of an oscillation-enhancing agent, the drug effect on SO-spindle characteristics and memory consolidation are contrasted to a placebo. Right panel: auditory stimulation. Following the online detection of an SO, two brief auditory clicks are presented in phase with SO up-states. Stimulation enhances the SO rhythm and phase-locked spindle activity. Figure reproduced from [17]. D Neural oscillations support memory reinstatement. Top: Following a TMR cue, there is a larger sleep spindle response for sounds associated with memories compared to non-memory control sounds. Bottom: Greater EEG pattern similarity for TMR cues corresponding to memories from the same vs different categories emerges ~ 2 s after the cue, in a time window is closely aligned to the evoked sleep spindle response. Figure reproduced from reference [18]. All figure reproductions made under a Creative Commons Attribution 4.0 International License
In humans, a wealth of behavioural evidence has accumulated to suggest that sleep facilitates the consolidation of both declarative (e.g. episodic) and non-declarative memories (e.g. motor skills) [31,32,33,34,35]. The purpose of this review is to address the current state of the literature in terms of the electrophysiological evidence of memory reactivation in human sleep. Our review is divided into three major parts. In the first part, we address the evidence for a role of NREM sleep neural oscillations, and their temporal coupling, in sleep-associated memory consolidation. In part two, we discuss research that has attempted to delineate memory reactivation in the sleeping brain. Finally, in part three, we focus on evidence that brings parts one and two together to link memory reactivation to the finely tuned temporal coupling of NREM sleep neural oscillations.
Neural Oscillations
Currently, the only way to directly measure ripples in human hippocampus is via invasive intracranial depth electrode (iEEG) recordings, typically only used with medication-resistant epilepsy patients. These studies have confirmed the nesting of ripples, spindles, and SOs in human [36] NREM sleep and have suggested that this coupling mediates information transfer between hippocampus and neocortex [37, 38]. Studies using non-invasive scalp EEG in healthy adults (which represents the vast majority of human work investigating NREM sleep neural oscillations) have reported analogous coupling between cortical SOs and spindles [39,40,41,42,43,44]. When interpreting the results of iEEG studies, it is important to consider that results come from a rare patient population who exhibit altered brain physiology and have been subject to chronic anti-convulsant medication use [45]. As such, caution should be taken when applying these results to the wider population.
Correlational Studies
Many studies have found that memory retention over sleep is correlated with the occurrence of sleep spindles (e.g. [46,47,48,49,50]). Meta-analysis has concluded that this association is robust across memory domains (declarative and non-declarative), spindle measures (e.g. spindle density, amplitude and power), and electrode locations (frontal, central, parietal) [51]. Sleep spindles have also been linked to the overnight, systems-level restructuring of recently formed memories, as indicated via measures of memory trace integration and hippocampal-cortical connectivity [52•]. Although encouraging, the meta-analysis found evidence of publication bias in the literature and noted issues of multiple comparisons and low statistical power [51] (see also [53, 54] for discussion of this topic). Furthermore, the largest study to date (N = 929) examining correlations between spindle activity and memory retention failed to find a significant relationship [55].
Because the temporal coupling between spindles and SOs is thought to play a major role in the memory function of sleep, more recent work has specifically examined the relationship between SO-coupled spindles and memory. Retention of word-pairs is uniquely associated with the density of SO-coupled spindles, and not uncoupled spindles [56•]. Similar findings have been reported in studies using non-declarative memory tasks [57, 58•], suggesting that SO-coupled spindles support consolidation across multiple memory domains. An interesting exception to this is one study examining emotional memory consolidation following a stress induction, where a negative association with the amount of SO-spindle coupling was found [59]. This highlights the need for more work to determine potential boundary conditions of SO-spindle mediated consolidation.
The precision of SO-spindle coupling is also relevant for memory consolidation. Several correlational studies have reported that memory retention in adults is optimal when spindles are consistently coupled close to the peak of the SO (Fig. 1B), with performance declining as the spindle coupling phase becomes less consistent and shifts further away from the SO peak [60,61,62,63,64,65,66,67,68].
Correlational studies have helped to establish an association between NREM sleep spindles (particularly those that are tightly coupled to the peak of the SO up-state) and overnight memory consolidation. Correlational studies cannot, however, determine a causal relationship. Evidence that SO-spindle coupling plays a causal role in memory consolidation has come from studies where these oscillations have been enhanced via experimental manipulation.
Enhancement of SO-Spindle Coupling
Certain pharmacological agents can be used to boost oscillatory activity during sleep (Fig. 1C). Relative to placebo, zolpidem (a GABA-a agonist) enhances the precision of SO-spindle coupling, with the magnitude of this enhancement predicting overnight memory retention [66, 67, 69]. Intriguingly, a different pattern emerges when a different GABA-a agonist, eszopiclone, is administered. While both drugs increase spindle activity, eszopiclone does not enhance memory consolidation [58•, 70]. Closer inspection reveals that, despite the increase in spindle activity, eszopiclone reduces the precision of SO-spindle coupling [58•]. These findings reveal that it is the coupling of SOs and spindles that is crucial for memory consolidation, rather than spindles in isolation, substantiating the correlational evidence above. It is important to note, however, that pharmacologically induced sleep spindles may differ from those generated endogenously. Furthermore, pharmaceuticals lead to widespread alterations in oscillatory activity during sleep [71, 72], raising issues of specificity regarding the influence of enhanced SO-spindle coupling on memory consolidation. Finally, from an intervention standpoint, long-term use of drugs such as zolpidem have been linked to cognitive impairment in some samples [73, 74] (though see [75]).
Non-invasive, non-pharmacological approaches provide an alternative method for enhancing SOs and spindles without influencing neural activity at other frequencies. Transcranial electrical stimulation, for example, has been used to boost SO and spindle activity [76, 77], shift spindle coupling closer to the SO peak [78], and, consequently, enhance memory performance [79]. Auditory stimulation protocols, where brief auditory clicks are delivered in phase with SO up-states (Fig. 1C), have also gained traction in recent years [17]. In a seminal study, auditory stimulation amplified the SO rhythm, enhanced SO-spindle coupling, and improved overnight memory retention, as compared to sham stimulation [80], effects that have been replicated in subsequent studies [17, 81, 82]. Current work assessing the boundary conditions of auditory stimulation (e.g. the types of memory that receive a stimulation benefit) [79, 83, 84] and optimal stimulation parameters (e.g. duration of stimulation) [85, 86] will serve to improve this method further.
Changes in SO-Spindle Coupling and Memory Consolidation Across Development
There are profound changes in sleep architecture and physiology across the lifespan [87, 88], and a growing number of studies have shown that developmental changes in the efficacy of sleep-associated memory consolidation co-vary with the maturation of SO-spindle coupling. In early childhood (~ 5–6 years old), the timing of SO-spindle coupling is less consistent than in adulthood [89]. Coupling consistency increases as children enter adolescence, with 14–18-year olds showing more consistent coupling than 5–11-year olds [90, 91]. Using a powerful longitudinal design, Hahn and colleagues showed that increases in the consistency of SO-spindle coupling across adolescence are associated with a stronger benefit of sleep for memory consolidation [92•]. As such, developmental changes in memory appear to track the maturation of SO-spindle timing.
While the temporal precision of SO-spindle coupling increases from childhood to young adulthood, the opposite occurs in older age [63, 65]. Similar to observations in early childhood, the consistency of SO-spindle coupling is reduced in older adults (~ 60–80 years old) compared to younger adults (~ 20–30 years old). Additionally, spindles start to couple earlier in the SO phase in older adults [63, 65]. Reduced SO-spindle coupling in older age is accompanied by a deterioration of overnight memory retention, with greater misalignment between the spindle and SO peak predicting worse performance [63, 65]. Relatedly, longitudinal work has shown that earlier coupling of spindles to the SO up-state is predictive of greater memory decline two years later [93]. These studies again suggest that precise coupling of sleep spindles to the SO peak is central to effective overnight memory processing.
Local Spindle Activity
A putative function of sleep spindles is to induce synaptic plasticity in learning-related cortical circuits [20, 21]. Therefore, SO-coupled spindle activity should be maximal at cortical sites that are engaged at learning. Studies addressing this question have employed tasks that recruit highly localised cortical areas during encoding and then examined whether spindle activity is enhanced across those areas during post-encoding sleep.
In declarative tasks, learning-related cortical areas show enhancements in spindle amplitude relative to control regions that are not recruited at encoding, as well as a shift in the timing of SO-coupled spindles, such that they emerge in closer proximity to the SO peak [94••, 95]. Similarly, in procedural motor learning tasks, SO-coupled spindles emerge more frequently in the hemisphere contralateral to the trained hand [57], with only SO-coupled spindles in this region (and not spindles occurring in isolation) correlating with overnight performance gains [57, 96]. Interestingly, recent work using a spatial memory task reported topographical overlap between cortical regions recruited at learning and expressions of spindle activity during subsequent sleep, with the extent of this encoding-spindle overlap predicting later memory performance. However, these effects were not observed when analyses were restricted to SO-coupled spindles, suggesting that uncoupled spindles in local cortical circuits support overnight memory processing under certain conditions [97]. Future work is necessary to integrate these findings with the SO-spindle coupling mechanisms emphasised by ASC theory.
Memory Reactivation
Endogenous Reactivation
A large body of evidence for memory reactivation in human sleep has come from neuroimaging studies demonstrating that neural activity patterns associated with learning re-emerge in the sleeping brain. A landmark study using positron emission tomography found that hippocampal responses observed during spatial navigation were observed again during post-learning sleep (but not pre-learning sleep) [98]. A multitude of subsequent neuroimaging studies have repeatedly demonstrated that patterns of brain activity associated with learning are reinstated during sleep, with the level of reinstatement correlating with post-sleep retention [99,100,101,102,103,104,105,106].
Similarly, electrophysiological methods have revealed that topographical patterns of cortical activity observed at learning are re-expressed during sleep [107, 108]. Work using multivariate classification approaches has reliably decoded from sleep EEG data the types of images viewed in a preceding learning session, consistent with memory reprocessing in sleep [109]. Interestingly, while reprocessing was detected throughout the night, only reprocessing during slow wave sleep (where SO-coupled spindles predominate) influenced later memory performance.
Targeted Memory Reactivation
Some of the strongest evidence that memory reactivation is a central mechanism of sleep-associated consolidation in humans has come from studies using an experimental technique known as targeted memory reactivation (TMR; Fig. 1D). In a typical TMR paradigm, sounds or odours are paired with to-be-learned information, and participants are re-exposed to a subset of these stimuli during sleep. This cueing procedure is intended to bias memory reactivation in the sleeping brain towards memories that are associated with the sound/odour cues [110]. Numerous studies have documented a behavioural benefit of TMR, showing improved memory retention for materials associated with the cues presented during sleep, as compared to the non-cued materials. This behavioural benefit has been confirmed by a recent meta-analysis [111].
The behavioural effects of TMR are usually accompanied by electrophysiological evidence of memory reprocessing in the sleeping brain. For instance, TMR cues often evoke a transient increase in spindle activity [95, 112,113,114,115,116,117,118,119,120,121,122], which is greater than that observed for previously unheard control sounds [18, 117, 120, 122], and predictive of later memory performance [112, 114, 116, 119, 122].
TMR-evoked changes in oscillatory activity often include increases in the theta band (~ 4–8 Hz), which precede those arising in the spindle band [112,113,114,115,116,117, 123, 124]. A notable study linked theta activity during pre-sleep retrieval with theta activity subsequently evoked by TMR, suggesting that oscillatory signatures of memory retrieval are reinstated upon the presentation of TMR cues in sleep [125]. While the ASC framework does not typically ascribe a role for theta oscillations in memory consolidation, adaptations to this framework have proposed complementary roles of theta and spindle oscillations, with the initial surge in theta activity reflecting memory reinstatement, and the subsequent increase in spindle activity supporting mnemonic stabilisation and transfer to neocortex [126]. Direct testing of this hypothesis is warranted in future work.
Recent work has combined the TMR protocol with multivariate decoding methods to provide further evidence of memory reinstatement during sleep. Cairney and colleagues showed that the categorical features (objects or scenes) of declarative memories cued via TMR can be reliably decoded during the evoked spindle response, with the magnitude of this effect predicting later memory performance [18]. Similar results have been found for motor skill learning, where researchers were able to differentiate TMR-evoked responses associated with left- and right-handed movements, but only for trials that were subsequently remembered after sleep [119]. Other work has successfully discriminated TMR-evoked patterns of brain activity associated with individual finger movements on a serial reaction time task [127], and of contextual details associated with encoded materials [128].
Neural Oscillations Support Memory Reactivation Events
SO-spindle coupling and memory reactivation are both integral to sleep-associated memory processing. Rodent work and experimental models, however, emphasise the importance of reactivation events that are tied to hippocampal sharp-wave ripples, which occur in synchrony with spindles and SOs [11, 28]. Hence, this next section will consider evidence from humans that memory reactivation is tied to this temporally coordinated oscillatory activity.
Spindles are most beneficial to memory when they are coupled to the rising phase of the SO. In line with this observation, the benefit of TMR has been shown to be greatest when cues are presented during the up-state of the SO [129, 130], though see [131]. Recent work has found that the decoding of categorical information from TMR-evoked neural activity is only possible when memory cues are presented during the SO up-state, with no evidence of reinstatement emerging when cues are presented in the SO down-state [132•]. This implies that reactivation is tied to the up-state of SOs.
A potential limitation of TMR is that reactivation events are externally cued and thus might not be reflective of memory reactivation that occurs endogenously. Recent work has tied endogenous memory reactivation to SO-spindle complexes [133••]. Here, the categorical features of previously encoded memories (objects or scenes) could be reliably discriminated near the peak of the ongoing SO, with the magnitude of categorical discrimination predicting the precision of SO-spindle coupling and subsequent retention performance [133••]. Taken together with the foregoing evidence from the TMR literature, this pioneering work supports the view that memory reactivation during SO-spindle complexes is a central mechanism of sleep-associated memory processing.
Using iEEG, Zhang et al. were able to detect spontaneous reactivation of recently learned information in hippocampus [134]. Although reactivation occurred continuously throughout both quiet rest and sleep, only reactivation that was tied to hippocampal ripples during sleep was beneficial to memory retention [134]. As such, the findings of this study are in line with rodent work linking hippocampal ripples in sleep to memory replay and, in turn, overnight consolidation.
A breakthrough study has reported evidence of memory reactivation in human sleep at the single neuron level. Recordings were obtained from the motor cortex of a patient performing a motor imagery task [135••]. During subsequent sleep, the neural sequences associated with learning were re-expressed at a rate significantly higher than chance [135••]. Consistent with rodent work, there was evidence that these reactivation events were temporally compressed and occurred most frequently during slow-wave sleep [135••]. Future work with multiple patients is now needed to link these events to behavioural expressions of memory consolidation and to assess how these reactivations are coupled to SO and spindle events.
Open Questions
Although much progress has been made in elucidating the mechanisms of memory reactivation during human NREM sleep, several open questions remain:
-
1)
Coupled and uncoupled spindles: Whereas SO-spindle coupling has been heavily implicated in memory consolidation, not all findings support this view, and various metrics of uncoupled spindles (e.g. synchronisation in local circuits [136, 137]) have been linked to overnight memory processing. Understanding the (complementary) mnemonic functions of coupled and uncoupled spindles is thus a crucial endeavour for future work in humans and will be integral to the advancement of ASC theory.
-
2)
Rapid eye movement (REM) sleep: Rodent studies have demonstrated that replay occurs in hippocampus during REM sleep and have highlighted REM sleep theta oscillations as a candidate mechanism for overnight memory processing [138, 139]. In humans, a limited number of studies have linked REM sleep theta oscillations to memory, especially for emotionally salient experiences [140,141,142], but REM theta stimulation does not lead to emotional memory enhancement [143]. A clearer characterisation of the neural oscillations that support REM-based consolidation will help further elucidate the memory function of sleep.
-
3)
Sleep-dependent consolidation? To what extent memory consolidation is dependent on the electrophysiological features of sleep remains unclear. Both SOs and hippocampal ripples occur during quiet resting wake [134, 144], during which spontaneous memory reactivations have been observed [145]. Moreover, recent work has shown that the memory benefits of quiet (vs active) wake are in some cases comparable to those of sleep [146, 147]. Spindles are, to our knowledge, uniquely sleep-dependent and might therefore be central to understanding the distinct roles of sleep and wake in mnemonic processing,
Conclusion
Research in non-human animals has demonstrated that memory reactivation, clocked by NREM sleep neural oscillations, is a key mechanistic driver of memory consolidation. Here, we reviewed the growing evidence for this process in humans. The precise temporal coupling of spindles to SOs predicts memory consolidation across multiple memory domains, and enhancement of these oscillations can lead to memory improvements. Triggering memory reactivations with external cues, combined with multivariate decoding approaches, provides a means of identifying mnemonic reinstatement in the sleeping brain. These reinstatements are closely tied to cardinal NREM sleep oscillations and are predictive of behavioural performance. Future challenges for the field will be to better characterise the memory functions of SO-coupled and uncoupled spindles, as well as the neural oscillations of REM sleep, to advance understanding of sleep’s unique contributions to memory.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Findlay G, Tononi G, Cirelli C. The evolving view of replay and its functions in wake and sleep. SLEEP Adv. 2020;1:zpab002.
Atherton LA, Dupret D, Mellor JR. Memory trace replay: the shaping of memory consolidation by neuromodulation. Trends Neurosci. 2015;38:560–70.
Robertson EM, Genzel L. Memories replayed: reactivating past successes and new dilemmas. Philos Trans R Soc B Biol Sci. 2020;375:20190226.
Wilson MA, McNaughton BL. Reactivation of hippocampal ensemble memories during sleep. Science. 1994;265:676–9.
Girardeau G, Zugaro M. Hippocampal ripples and memory consolidation. Curr Opin Neurobiol. 2011;21:452–9.
Ramanathan DS, Gulati T, Ganguly K. Sleep-dependent reactivation of ensembles in motor cortex promotes skill consolidation. PLOS Biol. 2015;13:e1002263.
Ji D, Wilson MA. Coordinated memory replay in the visual cortex and hippocampus during sleep. Nat Neurosci. 2007;10:100–7.
Wierzynski CM, Lubenov EV, Gu M, Siapas AG. State-dependent spike-timing relationships between hippocampal and prefrontal circuits during sleep. Neuron. 2009;61:587–96.
Foster DJ. Replay Comes of Age. Annu Rev Neurosci. 2017;40:581–602.
Lewis PA, Durrant SJ. Overlapping memory replay during sleep builds cognitive schemata. Trends Cogn Sci. 2011;15:343–51.
Kim J, Gulati T, Ganguly K. Competing roles of slow oscillations and delta waves in memory consolidation versus forgetting. Cell. 2019;179:514-526.e13.
Girardeau G, Benchenane K, Wiener SI, Buzsáki G, Zugaro MB. Selective suppression of hippocampal ripples impairs spatial memory. Nat Neurosci. 2009;12:1222–3.
Gridchyn I, Schoenenberger P, O’Neill J, Csicsvari J. Assembly-specific disruption of hippocampal replay leads to selective memory deficit. Neuron. 2020;106:291-300.e6.
Klinzing JG, Niethard N, Born J. Mechanisms of systems memory consolidation during sleep. Nat Neurosci. 2019;22:1598–610.
Rasch B, Born J. About sleep’s role in memory. Physiol Rev. 2013;93:681–766.
Born J, Wilhelm I. System consolidation of memory during sleep. Psychol Res. 2012;76:192–203.
Harrington MO, Cairney SA. Sounding it out: Auditory stimulation and overnight memory processing. Curr Sleep Med Rep. 2021;7:112–9.
Cairney SA, Guttesen AÁV, El Marj N, Staresina BP. Memory consolidation is linked to spindle-mediated information processing during sleep. Curr Biol. 2018;28:948–54.
Lee AK, Wilson MA. Memory of sequential experience in the hippocampus during slow wave sleep. Neuron. 2002;36:1183–94.
Fernandez LMJ, Luthi A. Sleep spindles: mechanisms and functions. Physiol Rev. 2020;100:805–68.
Peyrache A, Seibt J. A mechanism for learning with sleep spindles. Philos Trans R Soc B Biol Sci. 2020;375:20190230.
Seibt J, Richard CJ, Sigl-Glöckner J, Takahashi N, Kaplan DI, Doron G, et al. Cortical dendritic activity correlates with spindle-rich oscillations during sleep in rodents. Nat Commun. 2017;8:684.
Rosanova M, Ulrich D. Pattern-specific associative long-term potentiation induced by a sleep spindle-related spike train. J Neurosci. 2005;25:9398–405.
Timofeev I. Chapter 9. Neuronal plasticity and thalamocortical sleep and waking oscillations. In: Van Someren EJW, Van Der Werf YD, Roelfsema PR, Mansvelder HD, Lopes Da Silva FH, editors. Slow brain oscillations of sleep, resting state and vigilance, Elsevier; 2011. p. 121–44.
Steriade M. The corticothalamic system in sleep. Front Biosci J Virtual Libr. 2003;8:d878-899.
Steriade M, Nunez A, Amzica F. A novel slow (< 1 Hz) oscillation of neocortical neurons in vivo: depolarizing and hyperpolarizing components. J Neurosci. 1993;13:3252–65.
Niknazar H, Malerba P, Mednick SC. Slow oscillations promote long-range effective communication: the key for memory consolidation in a broken-down network. Proc Natl Acad Sci. 2022;119:e2122515119. Found heightened global communication across the brain during slow oscillation upstates, creating the conditions for memory reactivation to occur.
Latchoumane C-FV, Ngo H-VV, Born J, Shin H-S. Thalamic spindles promote memory formation during sleep through triple phase-locking of cortical, thalamic, and hippocampal rhythms. Neuron. 2017;95:424–35.
Maingret N, Girardeau G, Todorova R, Goutierre M, Zugaro M. Hippocampo-cortical coupling mediates memory consolidation during sleep. Nat Neurosci. 2016;19:959–64.
Oyanedel CN, Durán E, Niethard N, Inostroza M, Born J. Temporal associations between sleep slow oscillations, spindles and ripples. Eur J Neurosci. 2020;52:4762–78.
Hokett E, Arunmozhi A, Campbell J, Verhaeghen P, Duarte A. A systematic review and meta-analysis of individual differences in naturalistic sleep quality and episodic memory performance in young and older adults. Neurosci Biobehav Rev. 2021;127:675–88.
Berres S, Erdfelder E. The sleep benefit in episodic memory: an integrative review and a meta-analysis. Psychol Bull. 2021;147:1309–53.
Schmid D, Erlacher D, Klostermann A, Kredel R, Hossner E-J. Sleep-dependent motor memory consolidation in healthy adults: a meta-analysis. Neurosci Biobehav Rev. 2020;118:270–81.
Ashton JE, Harrington MO, Langthorne D, Ngo H-VV, Cairney SA. Sleep deprivation induces fragmented memory loss. Learn Mem. 2020;27:130–5.
Cairney SA, Lindsay S, Paller KA, Gaskell MG. Sleep preserves original and distorted memory traces. Cortex. 2018;99:39–44.
Staresina BP, Bergmann TO, Bonnefond M, van der Meij R, Jensen O, Deuker L, et al. Hierarchical nesting of slow oscillations, spindles and ripples in the human hippocampus during sleep. Nat Neurosci. 2015;18:1679–86.
Ngo H-V, Fell J, Staresina B. Sleep spindles mediate hippocampal-neocortical coupling during long-duration ripples. eLife. 2020;9:e57011.
Helfrich RF, Lendner JD, Mander BA, Guillen H, Paff M, Mnatsakanyan L, et al. Bidirectional prefrontal-hippocampal dynamics organize information transfer during sleep in humans. Nat Commun. 2019;10:3572.
Mölle M, Bergmann TO, Marshall L, Born J. Fast and slow spindles during the sleep slow oscillation: disparate coalescence and engagement in memory processing. Sleep. 2011;34:1411–21.
Mölle M, Marshall L, Gais S, Born J. Grouping of spindle activity during slow oscillations in human non-rapid eye movement sleep. J Neurosci. 2002;22:10941–7.
Cox R, Mylonas DS, Manoach DS, Stickgold R. Large-scale structure and individual fingerprints of locally coupled sleep oscillations. Sleep. 2018;41:zsy175.
Cox R, van Driel J, de Boer M, Talamini LM. Slow oscillations during sleep coordinate interregional communication in cortical networks. J Neurosci. 2014;34:16890–901.
Klinzing JG, Mölle M, Weber F, Supp G, Hipp JF, Engel AK, et al. Spindle activity phase-locked to sleep slow oscillations. Neuroimage. 2016;134:607–16.
Yordanova J, Kirov R, Verleger R, Kolev V. Dynamic coupling between slow waves and sleep spindles during slow wave sleep in humans is modulated by functional pre-sleep activation. Sci Rep. 2017;7:14496.
Whelan CD, Altmann A, Botía JA, Jahanshad N, Hibar DP, Absil J, et al. Structural brain abnormalities in the common epilepsies assessed in a worldwide ENIGMA study. Brain. 2018;141:391–408.
Schabus M, Gruber G, Parapatics S, Sauter C, Kloesch G, Anderer P, et al. Sleep spindles and their significance for declarative memory consolidation. Sleep. 2004;27:1479–85.
Lustenberger C, Wehrle F, Tüshaus L, Achermann P, Huber R. The multidimensional aspects of sleep spindles and their relationship to word-pair memory consolidation. Sleep. 2015;38:1093–103.
Gais S, Mölle M, Helms K, Born J. Learning-dependent increases in sleep spindle density. J Neurosci. 2002;22:6830–4.
Schmidt C, Peigneux P, Muto V, Schenkel M, Knoblauch V, Münch M, et al. Encoding difficulty promotes postlearning changes in sleep spindle activity during napping. J Neurosci. 2006;26:8976–82.
Tamminen J, Payne JD, Stickgold R, Wamsley EJ, Gaskell MG. Sleep spindle activity is associated with the integration of new memories and existing knowledge. J Neurosci. 2010;30:14356–60.
Kumral D, Matzerath A, Leonhart R, Schönauer M. Spindle-dependent memory consolidation in healthy adults: a meta-analysis. Neuropsychologia. 2023;189:108661.
Cowan E, Liu A, Henin S, Kothare S, Devinsky O, Davachi L. Sleep spindles promote the restructuring of memory representations in ventromedial prefrontal cortex through enhanced hippocampal–cortical functional connectivity. J Neurosci. 2020;40:1909–19. Showed that sleep spindles facilitate changes in the underlying neural memory traces.
Mantua J. Sleep physiology correlations and human memory consolidation: where do we go from here? Sleep. 2018;41:zsx04.
Cox R, Fell J. Analyzing human sleep EEG: a methodological primer with code implementation. Sleep Med Rev. 2020;54:101353.
Ackermann S, Hartmann F, Papassotiropoulos A, de Quervain DJ-F, Rasch B. No associations between interindividual differences in sleep parameters and episodic memory consolidation. Sleep. 2015;38:951–9.
Denis D, Mylonas D, Poskanzer C, Bursal V, Payne JD, Stickgold R. Sleep spindles preferentially consolidate weakly encoded memories. J Neurosci. 2021;41:4088–99. Showed that SO coupled spindles are a better predictor of declarative memory consolidation than uncoupled sleep spindles.
Solano A, Riquelme LA, Perez-Chada D, Della-Maggiore V. Motor learning promotes the coupling between fast spindles and slow oscillations locally over the contralateral motor network. Cereb Cortex. 2022;32:2493–507.
Mylonas D, Baran B, Demanuele C, Cox R, Vuper TC, Seicol BJ, et al. The effects of eszopiclone on sleep spindles and memory consolidation in schizophrenia: a randomized clinical trial. Neuropsychopharmacology. 2020;45:2189–97. Demonstrated that pharmacologically enhancing sleep spindles has no effect on memory if SO-spindle coupling is disrupted.
Denis D, Kim SY, Kark SM, Daley RT, Kensinger EA, Payne JD. Slow oscillation-spindle coupling is negatively associated with emotional memory formation following stress. Eur J Neurosci. 2022;55:2632–50.
Cross ZR, Helfrich RF, Corcoran AW, Kohler MJ, Coussens S, Zou-Williams L, et al. Spindle-slow oscillation coupling during sleep predicts sequence-based language learning. bioRxiv; 2021.
Hahn MA, Bothe K, Heib D, Schabus M, Helfrich RF, Hoedlmoser K. Slow oscillation-spindle coupling strength predicts real-life gross-motor learning in adolescents and adults. eLife. 2022;11:e66761
Halonen R, Kuula L, Antila M, Pesonen A-K. The Overnight retention of novel metaphors associates with slow oscillation–spindle coupling but not with respiratory phase at encoding. Front Behav Neurosci. 2021;15:712774.
Helfrich RF, Mander BA, Jagust WJ, Knight RT, Walker MP. Old brains come uncoupled in sleep: slow wave-spindle synchrony, brain atrophy, and forgetting. Neuron. 2018;97:221–30.
Mikutta C, Feige B, Maier JG, Hertenstein E, Holz J, Riemann D, et al. Phase-amplitude coupling of sleep slow oscillatory and spindle activity correlates with overnight memory consolidation. J Sleep Res. 2019;28:e12835.
Muehlroth BE, Sander MC, Fandakova Y, Grandy TH, Rasch B, Shing YL, et al. Precise slow oscillation-spindle coupling promotes memory consolidation in younger and older adults. Sci Rep. 2019;9:1940.
Niknazar M, Krishnan GP, Bazhenov M, Mednick SC. Coupling of thalamocortical sleep oscillations are important for memory consolidation in humans. PLoS ONE. 2015;10:e0144720.
Zhang J, Yetton B, Whitehurst LN, Naji M, Mednick SC. The effect of zolpidem on memory consolidation over a night of sleep. Sleep. 2020;43:zsaa084.
Bastian L, Samanta A, Ribeiro de Paula D, Weber FD, Schoenfeld R, Dresler M, et al. Spindle–slow oscillation coupling correlates with memory performance and connectivity changes in a hippocampal network after sleep. Hum Brain Mapp. 2022;43:3923–43.
Mednick SC, McDevitt EA, Walsh JK, Wamsley E, Paulus M, Kanady JC, et al. The critical role of sleep spindles in hippocampal-dependent memory: a pharmacology study. J Neurosci. 2013;33:4494–504.
Wamsley EJ, Shinn AK, Tucker MA, Ono KE, McKinley SK, Ely AV, et al. The effects of eszopiclone on sleep spindles and memory consolidation in schizophrenia: a randomized placebo-controlled trial. Sleep. 2013;36:1369–76.
Trachsel L, Dijk DJ, Brunner DP, Klene C, Borbély AA. Effect of zopiclone and midazolam on sleep and EEG spectra in a phase-advanced sleep schedule. Neuropsychopharmacology. 1990;3:11–8.
Lundahl J, Deacon S, Maurice D, Staner L. EEG spectral power density profiles during NREM sleep for gaboxadol and zolpidem in patients with primary insomnia. J Psychopharmacol (Oxf). 2012;26:1081–7.
Guo F, Yi L, Zhang W, Bian Z-J, Zhang Y-B. Association between Z drugs use and risk of cognitive impairment in middle-aged and older patients with chronic insomnia. Front Hum Neurosci. 2021;15:775144.
Leng Y, Stone KL, Yaffe K. Race differences in the association between sleep medication use and risk of dementia. J Alzheimers Dis JAD. 2023;91:1133–9.
Osler M, Jørgensen MB. Associations of benzodiazepines, Z-drugs, and other anxiolytics with subsequent dementia in patients with affective disorders: a nationwide cohort and nested case-control study. Am J Psychiatry. 2020;177:497–505.
Marshall L, Helgadóttir H, Mölle M, Born J. Boosting slow oscillations during sleep potentiates memory. Nature. 2006;444:610–3.
Paßmann S, Külzow N, Ladenbauer J, Antonenko D, Grittner U, Tamm S, et al. Boosting slow oscillatory activity using tDCS during early nocturnal slow wave sleep does not improve memory consolidation in healthy older adults. Brain Stimulat. 2016;9:730–9.
Ladenbauer J, Ladenbauer J, Külzow N, de Boor R, Avramova E, Grittner U, et al. Promoting sleep oscillations and their functional coupling by transcranial stimulation enhances memory consolidation in mild cognitive impairment. J Neurosci. 2017;37:7111–24.
Barham MP, Enticott PG, Conduit R, Lum JAG. Transcranial electrical stimulation during sleep enhances declarative (but not procedural) memory consolidation: Evidence from a meta-analysis. Neurosci Biobehav Rev. 2016;63:65–77.
Ngo H-VV, Martinetz T, Born J, Mölle M. Auditory closed-loop stimulation of the sleep slow oscillation enhances memory. Neuron. 2013;78:545–53.
Stanyer EC, Baniqued PDE, Awais M, Kouara L, Davies AG, Killan EC, et al. The impact of acoustic stimulation during sleep on memory and sleep architecture: a meta-analysis. J Sleep Res. 2022;31:e13385.
Wunderlin M, Züst MA, Hertenstein E, Fehér KD, Schneider CL, Klöppel S, et al. Modulating overnight memory consolidation by acoustic stimulation during slow-wave sleep: a systematic review and meta-analysis. Sleep. 2021;44:zsaa296.
Harrington MO, Ngo H-VV, Cairney SA. No benefit of auditory closed-loop stimulation on memory for semantically-incongruent associations. Neurobiol Learn Mem. 2021;183:107482.
Harlow TJ, Jané MB, Read HL, Chrobak JJ. Memory retention following acoustic stimulation in slow-wave sleep: A meta analytic review of replicability and measurement quality. Front in Sleep. 2023;2:1082253.
Ladenbauer J, Khakimova L, Malinowski R, Obst D, Tönnies E, Antonenko D, Obermayer K, Hanna J, Flöel A. Towards optimization of oscillatory stimulation during sleep. Neuromodulation. 2023;26:1592–601.
Navarrete M, Arthur S, Treder MS, Lewis PA. Ongoing neural oscillations predict the post-stimulus outcome of closed loop auditory stimulation during slow-wave sleep. Neuroimage. 2022;253:119055.
Kocevska D, Lysen TS, Dotinga A, Koopman-Verhoeff ME, Luijk MPCM, Antypa N, et al. Sleep characteristics across the lifespan in 1.1 million people from the Netherlands, United Kingdom and United States: a systematic review and meta-analysis. Nat Hum Behav. 2021;5:113–22.
Clawson BC, Durkin J, Aton SJ. Form and function of sleep spindles across the lifespan. Neural Plast. 2016;2016:6936381.
Joechner A-K, Wehmeier S, Werkle-Bergner M. Electrophysiological indicators of sleep-associated memory consolidation in 5- to 6-year-old children. Psychophysiology. 2021;58:e13829.
Joechner AK, Hahn MA, Gruber G, Hoedlmoser K, Werkle-Bergner M. Sleep spindle maturity promotes slow oscillation-spindle coupling across child and adolescent development. eLife. 2023;12:e83565
Kurz E-M, Zinke K, Born J. Sleep electroencephalogram (EEG) oscillations and associated memory processing during childhood and early adolescence. Dev Psychol. 2023;59(2):297–311.
Hahn MA, Heib D, Schabus M, Hoedlmoser K, Helfrich RF. Slow oscillation-spindle coupling predicts enhanced memory formation from childhood to adolescence. eLife. 2020;9:e53730. Longitudinal evidence that the development of robust SO-spindle coupling across adolescence tracks memory consolidation ability.
Chylinski D, Van Egroo M, Narbutas J, Muto V, Bahri MA, Berthomier C, et al. Timely coupling of sleep spindles and slow waves is linked to early amyloid-β burden and predicts memory decline. eLife. 2022;11:e78191.
Bar E, Marmelshtein A, Arzi A, Perl O, Livne E, Hizmi E, et al. Local targeted memory reactivation in human sleep. Curr Biol. 2020;30:1435-1446.e5. Showed that memory reactivation during sleep modulates SO-spindle coupling in cortical regions involved in initial learning.
Cox R, Hofman WF, de Boer M, Talamini LM. Local sleep spindle modulations in relation to specific memory cues. Neuroimage. 2014;99:103–10.
Nishida M, Walker MP. Daytime naps, motor memory consolidation and regionally specific sleep spindles. PLoS ONE. 2007;2:e341.
Petzka M, Chatburn A, Charest I, Balanos GM, Staresina BP. Sleep spindles track cortical learning patterns for memory consolidation. Curr Biol. 2022;32:2349–56.
Peigneux P, Laureys S, Fuchs S, Collette F, Perrin F, Reggers J, et al. Are spatial memories strengthened in the human hippocampus during slow wave sleep? Neuron. 2004;44:535–45.
Peigneux P, Laureys S, Fuchs S, Destrebecqz A, Collette F, Delbeuck X, et al. Learned material content and acquisition level modulate cerebral reactivation during posttraining rapid-eye-movements sleep. Neuroimage. 2003;20:125–34.
Bergmann TO, Mölle M, Diedrichs J, Born J, Siebner HR. Sleep spindle-related reactivation of category-specific cortical regions after learning face-scene associations. Neuroimage. 2012;59:2733–42.
Fogel S, Albouy G, King BR, Lungu O, Vien C, Bore A, et al. Reactivation or transformation? Motor memory consolidation associated with cerebral activation time-locked to sleep spindles. PLoS ONE. 2017;12:e0174755.
Boutin A, Pinsard B, Boré A, Carrier J, Fogel SM, Doyon J. Transient synchronization of hippocampo-striato-thalamo-cortical networks during sleep spindle oscillations induces motor memory consolidation. Neuroimage. 2018;169:419–30.
Shanahan LK, Gjorgieva E, Paller KA, Kahnt T, Gottfried JA. Odor-evoked category reactivation in human ventromedial prefrontal cortex during sleep promotes memory consolidation. eLife. 2018;7:e39681.
Sterpenich V, van Schie MKM, Catsiyannis M, Ramyead A, Perrig S, Yang H-D, et al. Reward biases spontaneous neural reactivation during sleep. Nat Commun. 2021;12:4162.
Deuker L, Olligs J, Fell J, Kranz TA, Mormann F, Montag C, et al. Memory consolidation by replay of stimulus-specific neural activity. J Neurosci. 2013;33:19373–83.
Vahdat S, Fogel S, Benali H, Doyon J. Network-wide reorganization of procedural memory during NREM sleep revealed by fMRI. eLife. 2017;6:e24987.
Piantoni G, Halgren E, Cash SS. Spatiotemporal characteristics of sleep spindles depend on cortical location. Neuroimage. 2017;146:236–45.
Murphy M, Stickgold R, Parr ME, Callahan C, Wamsley EJ. Recurrence of task-related electroencephalographic activity during post-training quiet rest and sleep. Sci Rep. 2018;8:5398.
Schönauer M, Alizadeh S, Jamalabadi H, Abraham A, Pawlizki A, Gais S. Decoding material-specific memory reprocessing during sleep in humans. Nat Commun. 2017;8:15404.
Bendor D, Wilson MA. Biasing the content of hippocampal replay during sleep. Nat Neurosci. 2012;15:1439–44.
Hu X, Cheng LY, Chiu MH, Paller KA. Promoting memory consolidation during sleep: a meta-analysis of targeted memory reactivation. Psychol Bull. 2020;146:218–44.
Antony JW, Piloto L, Wang M, Pacheco P, Norman KA, Paller KA. Sleep spindle refractoriness segregates periods of memory reactivation. Curr Biol. 2018;28:1736-1743.e4.
Farthouat J, Gilson M, Peigneux P. New evidence for the necessity of a silent plastic period during sleep for a memory benefit of targeted memory reactivation. Sleep Spindl Cortical States. 2017;1:14–26.
Groch S, Schreiner T, Rasch B, Huber R, Wilhelm I. Prior knowledge is essential for the beneficial effect of targeted memory reactivation during sleep. Sci Rep. 2017;7:39763.
Laventure S, Pinsard B, Lungu O, Carrier J, Fogel S, Benali H, et al. Beyond spindles: interactions between sleep spindles and boundary frequencies during cued reactivation of motor memory representations. Sleep. 2018;41:zsy142.
Lehmann M, Schreiner T, Seifritz E, Rasch B. Emotional arousal modulates oscillatory correlates of targeted memory reactivation during NREM, but not REM sleep. Sci Rep. 2016;6:39229.
Schechtman E, Antony JW, Lampe A, Wilson BJ, Norman KA, Paller KA. Multiple memories can be simultaneously reactivated during sleep as effectively as a single memory. Commun Biol. 2021;4:1–13.
Schreiner T, Lehmann M, Rasch B. Auditory feedback blocks memory benefits of cueing during sleep. Nat Commun. 2015;6:8729.
Wang B, Antony JW, Lurie S, Brooks PP, Paller KA, Norman KA. Targeted memory reactivation during sleep elicits neural signals related to learning content. J Neurosci. 2019;39:6728–36.
Guttesen A á V, Gaskell MG, Cairney SA. Delineating memory reactivation in sleep with verbal and non-verbal retrieval cues. bioRxiv; 2023; 2023.03.02.530762.
Yuksel C, Denis D, Coleman J, Oh A, Cox R, Morgan A, et al. Emotional memories are enhanced when reactivated in slow wave sleep, but impaired when reactivated in REM. bioRxiv; 2023; 2023.03.01.530661.
Denis D, Payne JD. Targeted memory reactivation during non-rapid eye movement sleep enhances neutral, but not negative, components of memory. bioRxiv; 2023; 2023.05.26.542120.
Joensen BH, Harrington MO, Berens SC, Cairney SA, Gaskell MG, Horner AJ. Targeted memory reactivation during sleep can induce forgetting of overlapping memories. Learn Mem. 2022;29:401–11.
Oyarzún JP, Morís J, Luque D, de Diego-Balaguer R, Fuentemilla L. Targeted memory reactivation during sleep adaptively promotes the strengthening or weakening of overlapping memories. J Neurosci. 2017;37:7748–58.
Schreiner T, Doeller CF, Jensen O, Rasch B, Staudigl T. Theta phase-coordinated memory reactivation reoccurs in a slow-oscillatory rhythm during NREM sleep. Cell Rep. 2018;25:296–301.
Antony JW, Schönauer M, Staresina BP, Cairney SA. Sleep spindles and memory reprocessing. Trends Neurosci. 2019;42:1–3.
Belal S, Cousins J, El-Deredy W, Parkes L, Schneider J, Tsujimura H, et al. Identification of memory reactivation during sleep by EEG classification. Neuroimage. 2018;176:203–14.
Schechtman E, Heilberg J, Paller KA. Memory consolidation during sleep involves context reinstatement in humans. Cell Rep. 2023;42:112331.
Göldi M, van Poppel EAM, Rasch B, Schreiner T. Increased neuronal signatures of targeted memory reactivation during slow-wave up states. Sci Rep. 2019;9:2715.
Shimizu RE, Connolly PM, Cellini N, Armstrong DM, Hernandez LT, Estrada R, et al. Closed-loop targeted memory reactivation during sleep improves spatial navigation. Front Hum Neurosci. 2018;12:28.
Batterink LJ, Creery JD, Paller KA. Phase of spontaneous slow oscillations during sleep influences memory-related processing of auditory cues. J Neurosci. 2016;36:1401–9.
Ngo H-VV, Staresina BP. Shaping overnight consolidation via slow-oscillation closed-loop targeted memory reactivation. Proc Natl Acad Sci. 2022;119:e2123428119. Found that playing memory cues during sleep only elicits a reactivation of memory content when cues are delivered in the excitable upstate of slow oscillations.
Schreiner T, Petzka M, Staudigl T, Staresina BP. Endogenous memory reactivation during sleep in humans is clocked by slow oscillation-spindle complexes. Nat Commun. 2021;12:3112. The first evidence in humans that endogenous memory reactivation occurs within SO-spindle events.
Zhang H, Fell J, Axmacher N. Electrophysiological mechanisms of human memory consolidation. Nat Commun. 2018;9:4103.
Rubin DB, Hosman T, Kelemen JN, Kapitonava A, Willett FR, Coughlin BF, et al. Learned motor patterns are replayed in human motor cortex during sleep. J Neurosci. 2022;42:5007–20. The first human evidence of single unit memory reactivation in the human motor cortex during the consolidation of a procedural memory task.
Boutin A, Doyon J. A sleep spindle framework for motor memory consolidation. Philos Trans R Soc B Biol Sci. 2020;375:20190232.
Solano A, Riquelme LA, Perez-Chada D, Della-Maggiore V. Visuomotor adaptation modulates the clustering of sleep spindles into trains. Front Neurosci. 2022;16:803387.
Louie K, Wilson MA. Temporally structured replay of awake hippocampal ensemble activity during rapid eye movement sleep. Neuron. 2001;29:145–56.
Hutchison IC, Rathore S. The role of REM sleep theta activity in emotional memory. Front Psychol. 2015;6:1439.
Nishida M, Pearsall J, Buckner RL, Walker MP. REM sleep, prefrontal theta, and the consolidation of human emotional memory. Cereb Cortex. 2009;19:1158–66.
Sopp MR, Michael T, Weeß H-G, Mecklinger A. Remembering specific features of emotional events across time: the role of REM sleep and prefrontal theta oscillations. Cogn Affect Behav Neurosci. 2017;17:1186–209.
Kim SY, Kark SM, Daley RT, Alger SE, Rebouças D, Kensinger EA, et al. Interactive effects of stress reactivity and rapid eye movement sleep theta activity on emotional memory formation. Hippocampus. 2020;30:829–41.
Harrington MO, Ashton JE, Ngo H-VV, Cairney SA. Phase-locked auditory stimulation of theta oscillations during rapid eye movement sleep. Sleep. 2020;44:zsaa227.
Brokaw K, Tishler W, Manceor S, Hamilton K, Gaulden A, Parr E, et al. Resting state EEG correlates of memory consolidation. Neurobiol Learn Mem. 2016;130:17–25.
Eichenlaub J-B, Jarosiewicz B, Saab J, Franco B, Kelemen J, Halgren E, et al. Replay of learned neural firing sequences during rest in human motor cortex. Cell Rep. 2020;31:107581.
Humiston GB, Tucker MA, Summer T, Wamsley EJ. Resting states and memory consolidation: a preregistered replication and meta-analysis. Sci Rep. 2019;9:1–9.
Wang SY, Baker KC, Culbreth JL, Tracy O, Arora M, Liu T, et al. ‘Sleep-dependent’ memory consolidation? Brief periods of post-training rest and sleep provide an equivalent benefit for both declarative and procedural memory. Learn Mem. 2021;28:195–203.
Funding
This project has received funding from the European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement No 101028886, awarded to DD, and from a Medical Research Council Career Development Award (MR/P020208/1), awarded to SAC.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Human and Animal Rights and Informed Consent
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The article will be published through the gold open access route so the article copyright will be with the authors. From the publication agreement: “Ownership of copyright in the Article will be vested in the name of the Author.”
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Denis, D., Cairney, S.A. Electrophysiological Mechanisms of Memory Consolidation in Human Non-rapid Eye Movement Sleep. Curr Sleep Medicine Rep 10, 181–190 (2024). https://doi.org/10.1007/s40675-024-00291-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40675-024-00291-y