Abstract
Objective
To assess any differences and similarities in psoriatic arthritis (PsA) between sexes. Any possible differences of psoriasis and its potential impact on disease burden between sexes with PsA were also evaluated.
Methods
Cross-sectional analysis of two longitudinal PsA cohorts. The impact of psoriasis on the PtGA was evaluated. Patients were stratified in four groups based on BSA. The median PtGA was then compared between the four groups. Moreover, a multivariate linear regression analysis was performed in order to evaluate associations between PtGA and skin involvement, split by sexes.
Results
We enrolled 141 males and 131 females: PtGA, PtPnV, tender, swollen joint count, DAPSA, HAQ-DI, PsAID-12 were statistically significant higher in females (p ≤ 0.05). PASS “yes” was deemed more in males than in females and BSA was higher in males. MDA was present more in males than females. When the patients were stratified on BSA, median PtGA was not different between males and females with BSA = 0. Instead, in females with BSA > 0, a higher PtGA was observed compared to males with BSA > 0. There was not a statistically significant association between skin involvement and PtGA at linear regression analysis, even if a trend seems to be present in female.
Conclusions
Psoriasis is more present in males, but it seems to be related to a worse impact in females. In particular, a possible role of psoriasis as an influencing factor the PtGA was found. Moreover, female PsA patients tended to have more disease activity, worse function, and higher disease burden.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Understanding the different expression of disease in males and females is of fundamental importance in the management of this condition. |
Our study shows that BMI and BSA were higher in males, while PtGA, PtPnV, TJC, SJC, DAPSA, HAQ-DI, and PsAID-12 were higher in females. |
Prevalence of psoriasis is higher in males but it seems to have a higher impact on PtGA in females, suggesting the importance to take into account sex differences in the management of PsA patients. |
Introduction
Psoriatic arthritis (PsA) is a chronic inflammatory disease characterized by a variable clinical course [1, 2]. The achievement of the best possible disease control, such as disease remission or low disease activity, has been proposed as a treatment target, and is shown to be an achievable goal for PsA patients [3, 4]. In particular, the Disease Activity Score for Psoriatic Arthritis (DAPSA) [5] and Minimal Disease Activity (MDA) [6] are the two composite indices identified as treatment response criteria to capture the various disease states.
Beyond the real possibility to achieve good disease control in PsA patients, evidence suggests a different burden of disease and response to treatment between sexes in patients with spondyloarthritis [7,8,9] and PsA [10,11,12]. Although PsA is considered equal in prevalence between males and females, evidence showed that the burden of the disease is higher in females when compared to males [13, 14]. In particular, psoriasis, as a clinical feature of PsA, seems to be more prevalent in males, but with less impact on disease burden [15, 16]. Previous studies focused on differences between males and females in PsA, in terms of disease activity, functional impairment, and/or response to treatment [10,11,12,13,14]. However, to the best of our knowledge, few studies have investigated the differences in psoriasis and its impact on disease burden between sexes in PsA patients. Therefore, the aim of the present study was to assess any differences and similarities between sexes observed in two groups of PsA patients, as a further contribution to this intriguing topic. Moreover, a secondary aim was to evaluate any differences of psoriasis on disease burden between sexes with PsA.
Methods
The study protocol was carried out in compliance with the declaration of Helsinki; written consent was obtained from each participant. The study was approved by the Institutional Review Board of the University of Molise (protocol n. 0001-017-2021).
Patient Selection
In this cross-sectional analysis of two longitudinal cohorts, patients were enrolled at the Rheumatology Unit, Department of Medicine and Health Science-University of Molise, and at the Rheumatology Unit of University of Rome Tor Vergata. From February 1, 2022 until July 31, 2022, all PsA patients consecutively attending the rheumatology units were considered potentially eligible for the study.
Inclusion criteria were:
-
(1)
PsA classified with the ClASsification criteria for Psoriatic ARthritis (CASPAR) criteria [17],
-
2)
Age ≥ 18 years,
-
3)
Stable treatment with a conventional synthetic disease-modifying anti-rheumatic drugs (csDMARDs) or biological DMARDs (bDMARDs) for at least 6 months.
Data Collection
Patients’ data collection included a detailed medical history, physical examination, current use of medications, and laboratory assessment. Demographics and disease characteristics, including age, sex, body mass index (BMI) and disease duration, were recorded. The clinical assessment encompassed the number of tender (TJC) and swollen joints (SJC) (68/66), enthesitis by the Leeds Enthesitis Index (LEI) [18], and dactylitis. Psoriasis was quantified by the body surface area (BSA) [19]. The psoriasis onset age was also recorded, dividing patients in early onset psoriasis (EOP) (onset < 40 years) and late onset psoriasis (LOP) (≥ 40 years) [20].
The patient-reported outcomes (PROs) collected were: Health Assessment Questionnaire-Disability Index (HAQ-DI) [21], Patient Global Assessment (PtGA) [22], patient’s pain (PtPnV) assessed on numerical rating scale (NRS: 0–10 cm) and the Psoriatic Arthritis Impact of Disease 12-item (PsAID-12) [23]. PtGA was collected on NRS and comprises the global evaluation of psoriatic disease, with high values indicating worse status.
The Physician Global Assessment of disease activity (PGA), [22] and C reactive protein (CRP) were also collected.
We also collected the Patient Acceptable Symptom State (PASS) [24]. The global question assessing PASS was formulated as the following: ‘Think about all the ways your PsA has affected you during the last 48 h. If you were to remain in the next few months as you were during the last 48 h, would this be acceptable to you?’ The yes/no response was collected.
Finally, the DAPSA [5] and the MDA [6] were calculated as disease activity index and treatment target, respectively, and the presence of fibromyalgia (as a comorbidity) was also recorded. For the purpose of this study, no gender differences (referred to the characteristics of women, men, girls and boys that are socially constructed) were considered.
The study protocol was carried out in compliance with the Declaration of Helsinki; written consent was obtained from each participant. The study was approved by the Institutional Review Board of the University of Molise (protocol n. 0001-017-2021).
Statistical Analysis
Statistical analysis was performed using SPSS (version 27). All demographical and clinical characteristics were summarized by using descriptive statistics. Normally distributed variables were summarized by mean ± standard deviation (SD) and non-normally distributed variables by median and inter-quartile range (IQR).
Patients were divided into two groups according to sex. To compare these two groups, independent-sample t test, Mann–Whitney U test and χ-square test were performed, according with the data distribution. Moreover, to assess any changes of PtGA based on sex and BSA, patients were stratified in four groups as follow: females with BSA = 0, females with BSA > 0, males with BSA = 0, males with BSA > 0. The median PtGA was then compared within the four groups by using Kruskal–Wallis test.
Furthermore, each MDA domain (as categorical variable) was compared between males and females by using χ-square test. Finally, multivariate logistic regression analysis was performed in order to evaluate any association between the HAQ-DI and disease duration, also evaluating potential confounders, split by sexes. Goodness-of-fit was estimated using the adjusted R2. Odds ratio (OR) and confidence interval (CI) 95% were calculated when appropriate. Fibromyalgia was used as a control factor in the regression model n.2, only for female patients, due to its lower prevalence in male patients. A statistical significance level was defined as a two-tailed p value accepted at p ≤ 0.05.
Results
Descriptive Results
During the study period, 272 PsA (male 141, female 131) patients satisfying the inclusion criteria were enrolled.
There were not statistically significant differences, in terms of clinical and demographic features, between patients from the two centers (data not shown).
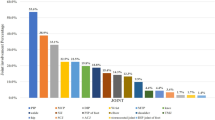
Table 1 shows the main clinical characteristics of the total enrolled patients divided by sex. Generally, in terms of differences, BMI and BSA were higher in males, while PtGA, PtPnV, TJC, SJC, DAPSA, HAQ-DI, and PsAID-12 were higher in females. The latter had a higher prevalence of fibromyalgia (F 19.2 vs. M 2.2%, p < 0.001). Moreover, MDA and an acceptable symptom state (PASS yes) were less likely present in females than males.
In terms of similarities, age, disease duration, psoriasis onset age (EOP and LOP), PsA patterns, dactylitis, LEI, CRP, PGA, and the current therapy were not different between the two sexes. However, even if there were no statistically significant differences, a higher percentage of females were on csDMARDs and tumor necrosis factor inhibitors (TNFi), while a higher number of male patients were on anti-IL-12/23 treatment.
Disease Activity, Function, and Impact of the Disease
Mean DAPSA was statistically significant higher in females. However, DAPSA is good for assessing peripheral arthritis, not other manifestations of PsA, and may not capture all the elements influencing disease reporting. Of note, when analyzing each DAPSA component, TJC, SJC, PtGA, and PtPnV were higher in females, despite CRP values were not different between the two groups, as previously mentioned.
Females, compared with males, reported higher mean pain [5 (± 2.78) vs. 4 (± 2.60), p = 0.003] and worst mean PtGA: [5.01 (± 2.51) vs. 3.99 ± 2.45, p < 0.001].
These data are also in keeping with worst function assessed by HAQ-DI, worst impact of the disease assessed by PsAID-12, and less presence of PASS yes ([69.5% in males and 47% in females, respectively, χ2 (1, n = 245) = 11.88, p < 0.001].
Finally, MDA, considered in the total population was present in 83/239 (34.7%), but when calculated by dividing the two sexes, this percentage was significantly different: 44% in males vs. 24.6% in females, [χ2 (1, n = 239) = 9.10, p = 0.003]. Moreover, going deeper into MDA domains, females, compared with males, less likely had PtPnV ≤ 1.5, HAQ-DI ≤ 0.5 and TJC ≤ 1; on the other hand, males less likely had a BSA ≤ 3 when compared with females (Table 2, Fig. 1).
Relationship Between Skin and Disease Burden
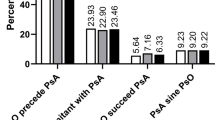
Mean (SD) BSA was higher in males: 2.16 (± 3.74) vs. 1.22 (± 2.26), p = 0.015. A possible role of psoriasis, measured as BSA, on the burden of the disease was analyzed in the two sexes. We found that PtGA was different in males and females with regard to BSA; in fact, when comparing median PtGA between males and females with BSA > 0, a statistically significant difference was found (p = 0.038), with females showing higher values of PtGA; therefore, when psoriasis was present, a sex difference in PtGA was observed. On the other hand, when comparing PtGA in females with BSA = 0 to males with BSA = 0, there was a trend to be worse in females, even if it was not statistically significant (median PtGA (IQR): 4 (3–6) and 3 (2–5), respectively). In the same way, when comparing PtGA in females with BSA = 0 to female with BSA > 0, there was a trend to be worse in females with BSA > 0, even if it was not statistically significant (median PtGA (IQR): 4 (3–6) and 6 (4–8), respectively). Moreover, males with BSA > 0 or BSA = 0 were also better than females with BSA > 0, and it was statistically significant in both cases (Fig. 2).
Comparison of PtGA based on sex and BSA. Patients were stratified in four groups based on sex and BSA category. Females with BSA = 0; females with BSA > 0; males with BSA = 0; males with BSA > 0. PtGA was compared among these groups using Kruskal–Wallis test. The table shows the p values for each comparison
These results could have been influenced by the sample size; however, these trends could be of some importance to distinguish the role of skin involvement in PtGA between males and females.
Finally, to evaluate any association between BSA and PtGA, independently by others confounding factors, we performed two multiple linear regression analysis, for males and females, respectively.
In male patients, the association between BSA > 0 and PtGA was not statistically significant (p = 0.169), when adjusted for other confounding factors (Table 3, model 1) (adjuster R2:0.21).
In female patients, the association between BSA > 0 and PtGA was not statistically significant, even if with a trend of significance (p = 0.074) was found (Table 3, model 2). Therefore, it could mean that when psoriasis is present in females, the mean value of PtGA tends to increase by 0.75, independently of articular involvement (TJC and SJC) and fibromyalgia (adjuster R2:0.25).
Discussion
The present study was aimed at evaluating the possible differences and similarities between males and females with PsA. The results of this study, based on two groups, showed that PsA might be similar in some characteristics while on some other aspects, such as disease burden, is different between male and female sexes. Among the various clinical manifestations of this multifaceted syndrome [2], we found that there is an unequal distribution of some clinical features such as dactylitis, enthesitis, and psoriasis onset age. On the other side, we found that TJC, SJC, disease activity level, and even the absence of achievement of MDA differed between the two sexes, being more prevalent in females [25]. These results are in keeping with previous data, reinforcing that the management of PsA between the two sexes is still un unmet need [26]. Consequently, a potential difference of the skin involvement between sexes could be one of the factors driving different choices in the management, even if psoriasis could be one of the “less problematic” features in rheumatological settings because it is generally not a major issue when compared to patients attending dermatological clinics and, possibly, perceived as not important as the musculoskeletal complaints [27]. Having said that, the presence of psoriasis may persist even in PsA patients achieving a condition of MDA, as previously shown [28]. Therefore, the impact of psoriasis might be one of the choices for a change in the treatment strategy.
The present article showed a worse psoriasis in males when compared to females in two PsA settings. This result has been previously reported [11], confirming that psoriasis could be more frequent in males. However, when evaluating the role of psoriasis in terms of disease burden, we found that psoriasis was a factor potentially influencing the PtGA, reinforcing that PtGA is able to capture differences between males and females when the skin involvement is present. In other words, the burden of psoriasis might have an impact on the global assessment, being higher in females and confirming other previous data. In fact, PtGA was already found as a reliable measure of patient’s global assessment [29]. Of note, a clear trend was present in PtGA in male patients in respect to females, but this could be confirmed in a large sample of PsA patients. The present results might contribute to better understanding any clinical sex differences in PsA and, potentially, providing some practical insights in the global management of this multifaceted condition [2]. Moreover, as another factor showing any differences in disease burden between the two sexes, we found that the achievement of a condition of acceptable symptom state was more frequent in male PsA patients. Finally, all these differences—clinical, functional, disease activity and disease burden—may help the physicians on different treatment strategy toward a personalized approach, as also recently shown [3, 30]. This is in keeping with other previous results [31], paving the way to a potential different treatment strategy sex driven.
PsA, per se, is not a sex-related disease but these results could support the concept that the burden of the disease is different when compared between the two sexes. However, as a potential weakness of the study, we evaluated only the extension of the skin disease more than severity one. In fact, BSA was the only assessment of psoriasis we performed, without evaluating other specific tools to assess the impact or the severity of psoriasis as usually dermatologist do.
Moreover, biological sex can influence PsA by affecting sex hormones, gene expression, immune function, pain mechanisms, and pharmacokinetics of medications, with treatment outcomes that may be influenced by behavior, adherence to medications, patient–physician interactions, pain reporting, social support, coping mechanisms, and access to care. This aspect is shared with other diseases such as rheumatoid arthritis and osteoarthritis [32, 33].
Conclusion
In conclusion, the present study showed that there are some differences and similarities between males and females with PsA. Psoriasis is more present in males but, in terms of disease burden, has a worse impact in females. Female PsA patients tended to have more disease activity, worse function, and higher disease burden.
A future research agenda on these differences between the two sexes should be addressed, including this topic on larger population studies.
References
FitzGerald O, Ogdie A, Chandran V, et al. Psoriatic arthritis. Nat Rev Dis Primers. 2021;7:59.
Lubrano E, Scriffignano S, Perrotta FM. Psoriatic arthritis, psoriatic disease, or psoriatic syndrome? J Rheumatol 201946:1428–30.
Gossec L, McGonagle D, Korotaeva T, et al. Minimal disease activity as a treatment target in psoriatic arthritis: a review of the literature. J Rheumatol. 2018;45:6–13.
Smolen JS, Schöls M, Braun J, et al. Treating axial spondyloarthritis and peripheral spondyloarthritis, especially psoriatic arthritis, to target: 2017 update of recommendations by an international task force. Ann Rheum Dis. 2018;77:3–17.
Schoels MM, Aletaha D, Alasti F, et al. Disease activity in psoriatic arthritis (PsA): defining remission and treatment success using the DAPSA score. Ann Rheum Dis. 2016;75:811–8.
Coates LC, Fransen J, Helliwel PS. Defining disease activity in psoriatic arthritis: a proposed objective target for treatment. Ann Rheum Dis. 2010;69:48–53.
Lubrano E, Perrotta FM, Manara M, et al. The sex influence on response to tumor necrosis factor-α inhibitors and remission in axial spondyloarthritis. J Rheumatol. 2018;45:195–201.
Mease PJ, McLean RR, Dube B, et al. Comparison of men and women with axial spondyloarthritis in the US-based Corrona Psoriatic Arthritis/Spondyloarthritis Registry. J Rheumatol. 2022;48:1528–36.
Chimenti MS, Alten R, D’Agostino MA, et al. Sex-associated and gender-associated differences in the diagnosis and management of axial spondyloarthritis: addressing the unmet needs of female patients. RMD Open. 2021;7: e001681.
Passia E, Vis M, Coates LC, et al. Sex-specific differences and how to handle them in early psoriatic arthritis. Arthritis Res Ther. 2022;24:22.
Coates LC, van der Horst-Bruinsma IE, Lubrano E, Beaver S, Drane E, Ufuktepe B, Ogdie AR. Sex-specific differences in patients with psoriatic arthritis: a systematic review. J Rheumatol. 2022. https://doi.org/10.3899/jrheum.220386.
Ramonda R, Lorenzin M, Carriero A, et al. Effectiveness and safety of secukinumab in 608 patients with psoriatic arthritis in real life: a 24-month prospective, multicentre study. RMD Open. 2021;7: e001519.
Eder L, Thavaneswaran A, Chandran V, Gladman DD. Gender difference in disease expression, radiographic damage and disability among patients with psoriatic arthritis. Ann Rheum Dis. 2013;72:578–82.
Duruöz MT, Gezer HH, Nas K, et al. Gender-related differences in disease activity and clinical features in patients with peripheral psoriatic arthritis: a multi-center study. Joint Bone Spine. 2021;88: 105177.
Lesuis N, Befrits R, Nyberg F, van Vollenhoven RF. Gender and the treatment of immune-mediated chronic inflammatory diseases: rheumatoid arthritis, inflammatory bowel disease and psoriasis: an observational study. BMC Med. 2012;10:82.
Schmid-Ott G, Künsebeck HW, Jäger B, et al. Significance of the stigmatization experience of psoriasis patients: a 1-year follow-up of the illness and its psychosocial consequences in men and women. Acta Derm Venereol. 2005;85:27–32.
Taylor W, Gladman D, Helliwell P, et al. CASPAR Study Group. Classification criteria for psoriatic arthritis: development of new criteria from a large international study. Arthritis Rheum. 2006;54:2665–73.
Healy PJ, Helliwell PS. Measuring clinical enthesitis in psoriatic arthritis: assessment of existing measures and development of an instrument specific to psoriatic arthritis. Arthritis Rheum. 2008;59:686–91.
Ashcroft DM, Wan Po AL, Williams HC, Griffiths CE. Clinical measures of disease severity and outcome in psoriasis: a critical appraisal of their quality. Br J Dermatol. 1999;141:185–91.
Henseler T, Christophers E. Psoriasis of early and late onset: characterization of two types of psoriasis vulgaris. J Am Acad Dermatol. 1985;13:450–6.
Ranza R, Marchesoni A, Calori G, et al. The Italian version of the functional disability index of the Health Assessment Questionnaire. A reliable instrument for multicenter studies on rheumatoid arthritis. Clin Exp Rheumatol. 1993;11:123–8.
Lubrano E, Perrotta FM, Parsons WJ, et al. Patient’s global assessment as an outcome measure for psoriatic arthritis in clinical practice: a surrogate for measuring low disease activity? J Rheumatol. 2015;42:2332–8.
Gossec L, de Wit M, Kiltz U, et al. A patient-derived and patient-reported outcome measure for assessing psoriatic arthritis: elaboration and preliminary validation of the Psoriatic Arthritis Impact of Disease (PsAID) questionnaire, a 13-country EULAR initiative. Ann Rheum Dis. 2014;73:1012–9.
Lubrano E, Scriffignano S, Azuaga AB, et al. Assessment of the Patient Acceptable Symptom State (PASS) in psoriatic arthritis: association with disease activity and quality of life indices. RMD Open. 2020;6: e001170.
Lubrano E, Scriffignano S, Perrotta FM. The “Climb” towards minimal disease activity in psoriatic arthritis. Rheumatol Ther. 2021;8:1443–50.
Gratacós J, Behrens F, Coates LC, et al. A 12-point recommendation framework to support advancement of the multidisciplinary care of psoriatic arthritis: a call to action. Joint Bone Spine. 2021;88: 105175.
Lubrano E, Delle Sedie A, Romanelli M, et al. Management of psoriatic arthritis in rheumatology and dermatology settings: sub-analysis of the Italian population from the international LOOP study. Clin Rheumatol. 2021;40:2251–62.
Lubrano E, Scriffignano S, Perrotta FM. Residual disease activity and associated factors in psoriatic arthritis. J Rheumatol. 2020;47:1490–5.
Cauli A, Gladman DD, Mathieu A, et al. Patient global assessment in psoriatic arthritis: a multicenter GRAPPA and OMERACT study. J Rheumatol. 2011;38:898–903.
Coates LC, Soriano ER, Corp N, et al. GRAPPA Treatment Recommendations domain subcommittees. Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA): updated treatment recommendations for psoriatic arthritis 2021. Nat Rev Rheumatol. 2022;18:465–79.
Orbai AM, Perin J, Gorlier C, et al. Determinants of patient-reported psoriatic arthritis impact of disease: an analysis of the association with sex in 458 patients from fourteen countries. Arthritis Care Res (Hoboken). 2020;72:1772–9.
Tschon M, Contartese D, Pagani S, Borsari V, Fini M. Gender and sex are key determinants in osteoarthritis not only confounding variables. A systematic review of clinical data. J Clin Med. 2021;10:3178.
Tengstrand B, Ahlmén M, Hafström I. The influence of sex on rheumatoid arthritis: a prospective study of onset and outcome after 2 years. J Rheumatol. 2004;31:214–22.
Acknowledgements
We want to thank the participants of the study.
Funding
No funding or sponsorship was received for this study or publication of this article. All authors had full access to all of the data in this study and take complete responsibility for the integrity of the data and accuracy of the data analysis.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Authorship Contributions
All authors have made substantial contributions to all of these sections: conception and design of the study, acquisition of data, analysis and interpretation of data, drafting the article, revising it critically for important intellectual content and final approval of the version to be submitted.
Disclosures
Ennio Lubrano, Silvia Scriffignano, Mauro Fatica, Paola Triggianese, Paola Conigliaro, Fabio Massimo Perrotta and Maria Sole Chimenti have nothing to disclose.
Compliance with Ethics Guidelines
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study. The study was approved by the Institutional Review Board of the University of Molise.
Data Availability
The datasets generated during and/or analyzed during the current study are not publicly available but are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Lubrano, E., Scriffignano, S., Fatica, M. et al. Psoriatic Arthritis in Males and Females: Differences and Similarities. Rheumatol Ther 10, 589–599 (2023). https://doi.org/10.1007/s40744-023-00535-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40744-023-00535-3