Abstract
Background
Repeated hospitalization is a predictor of outcomes in heart failure, indicating the presence of symptoms, a deteriorated condition at pre-admission, and worsened prognosis.
Objectives
The current database study aimed to understand the clinical and economic burden of repeated hospitalizations among patients with heart failure in Japan. The effect of repeated hospitalizations on the subsequent in-hospital mortality was the primary objective; economic burden of heart failure after discharge was investigated as a secondary outcome.
Methods
Between 2013 and 2018, administrative claims and discharge summary data of patients aged ≥ 20 years and diagnosed with heart failure were obtained from a Diagnosis Procedure Combination database maintained by Medical Data Vision. Hospitalization, mortality, and economic burden data were analyzed.
Results
This study included 49,094 patients. The mean length of the first hospital stay was 22.9 days. The in-hospital mortality rate was approximately 10%, with one to five repeated hospitalizations. The time interval between repeated hospitalizations for heart failure decreased with an increasing number of hospitalizations. In-hospital mortality did not increase even with an increasing number of hospitalizations. The mean heart failure-related healthcare cost per patient was ¥564,281 ± 990,447 (US$5178 ± 9,088), 67.3% of which was hospitalization costs. Among hospitalization costs, other costs were high, mainly for basic hospitalization fees (71.7%; ¥233,146/person-year).
Conclusions
Repeated hospitalization did not increase in-hospital mortality; however, it may shorten the intervals between heart failure-related hospitalizations, potentially caused by deterioration of the patient’s condition, and increase the clinical and economic burden on patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Repeated hospitalization was shown to be an important predictor of outcomes in patients with heart failure. |
The 2013–18 data showed that there was a 10% in-hospital mortality rate among patients who underwent one to five repeated hospitalizations. |
In-hospital mortality did not increase with the number of hospitalizations. |
1 Introduction
Heart failure (HF) increases dramatically with advancing age and has a prevalence of > 10% among individuals aged ≥ 70 years [1]. Japan has the largest growing elderly population worldwide [2], and a prolonged life expectancy [3]. Consequently, the number of patients with HF has been increasing rapidly, and it is expected to reach ~ 1.3 million by 2030 [4,5,6]. Thus, the management of HF is of extreme relevance and is expected to gain importance over time.
Several studies have revealed predictors of mortality and morbidity in HF [7]. Patients with HF tend to present with comorbidities, such as hypertension, diabetes mellitus, and chronic kidney disease [8,9,10,11], which both complicate treatment and increase morbidity and mortality. Guideline-directed medical therapy (GDMT) for HF includes treatment with angiotensin-converting enzyme inhibitors (ACEis), angiotensin II receptor blockers (ARBs), β-blockers, and mineralocorticoid receptor antagonists (MRAs) [12]. Recent data have indicated that lower doses (i.e., doses lower than the approved maintenance dose) at discharge are also being prescribed in several countries, including Japan [13]. Although dose increase and prognosis are related, adequate knowledge about the doses used in real-world settings is lacking.
Repeated hospitalization appears to be another important predictor of HF outcomes, as the need for hospitalization indicates a deteriorated condition at pre-admission and worsened prognosis [14]. Specifically, a comparison of groups of patients with or without frequent repeated hospitalization who were admitted to hospital for HF showed that repeated hospitalization was associated with a five-fold increased mortality risk [15]. Moreover, repeated hospitalizations have been identified as a general predictor of worse prognosis for patients with HF [8, 16].
Many studies have reported the substantial economic burden of HF [17,18,19]. A previous study from Japan reported that 44% of the overall hospitalization costs in acute major cardiovascular disease were attributable to hospitalizations for HF, and the cost of a one-time hospitalization is US$8284 [20, 21]. Research has shown that HF has one of the highest hospitalization rates [17], and the length of stay in Japan (> 20 days) is longer than in any other country [17], further increasing hospitalization costs. Additionally, it is reported that higher costs do not necessarily correlate with improved outcomes [22]. However, no studies have evaluated the HF-related medical expenses associated with long-term inpatient and outpatient treatment that may lead to repeated hospitalizations. Furthermore, it is not clear which factors contribute to increased or decreased HF-related medical expenses, including continuous hospitalization and outpatient care.
Understanding the HF burden in Japanese patients is necessary to estimate future global trends. Although patients with HF experience repeated hospitalizations, little is known about the longitudinal effect of repeated hospitalizations on the long-term condition and prognosis of patients with HF after discharge. Using a Japanese nationwide administrative hospital database to evaluate continuous patient data collected before and after hospital admission, we investigated the long-term economic and clinical burden of repeated hospitalizations for patients with HF. The effect of repeated hospitalizations on in-hospital mortality, along with factors affecting the number of hospitalizations, treatment patterns of GDMT after discharge, the economic burden of HF after discharge, and factors that affect economic burden after discharge were investigated.
2 Methods
2.1 Study Design and Data Sources
This was a retrospective observational study using administrative claims data and discharge summary data collected from an administrative database provided by Medical Data Vision Co., Ltd. (Tokyo, Japan) between 1 September, 2013, and 31 August, 2018. The study was approved by the Ethics Review Committee of the nonprofit corporation Clinical Research Promotion Network Japan (protocol numbers: CLCZ696BJP01 and CLCZ696BJP02).
The longitudinal database contains data collected from over 400 acute care hospitals (with an average of 358 beds) in Japan that are under the Diagnosis Procedure Combination/Per-Diem Payment System (DPC/PDPS). By August 2018, the database covered about 24.5 million patients treated in 369 DPC hospitals across the country, representing approximately 21% of the total DPC hospitals in Japan. This database has been widely used to research treatment patterns, epidemiology studies, and health economic outcomes research (https://en.mdv.co.jp/publication/).
The database-collected administrative claims information included detailed data for inpatient and outpatient visits (of previously hospitalized patients), patient demographics (e.g., age, sex), diagnoses (coded according to the International Classification of Diseases, Tenth Revision), medical procedures, drugs prescribed, and laboratory data (for approximately 10% of the hospitals), as well as other items such as cost. The discharge summary data included patient age and sex, height, objective of hospitalization, history of hospitalization, patient outcomes including in-hospital mortality, comorbidities at and during hospitalization, history of smoking, the determination of New York Heart Association functional class at hospitalization for HF, diagnoses recorded using International Classification of Diseases, Tenth Revision codes, drugs prescribed (during hospitalization, at discharge and outpatient visits), and other data items.
The index hospitalization was defined as the first HF-related hospital admission during the data collection period, and the index date was defined as the discharge date of the first HF-related hospitalization. For analysis of the in-hospital mortality of HF by the number of repeated hospitalizations, the baseline period was 180 days prior to the index date. The interval time was the time elapsed between two hospitalizations, indicating days alive and out of hospital.
When analyzing the economic burden of HF after discharge, the baseline period was defined as the time between the admission date for the first HF-related hospitalization and the discharge (index) date. Follow-up was for 365 days from the index date, including the discharge date.
2.2 Study Objectives
The primary objective of this study was to determine the effect of repeated hospitalizations on the subsequent in-hospital mortality. The secondary objectives were (1) to investigate the factors affecting the number of hospitalizations, (2) to describe the treatment patterns of GDMT of patients with HF in outpatient settings after discharge, (3) to describe the economic burden of HF after discharge, and (4) to investigate the factors affecting the economic burden after discharge. Only patients with repeat hospitalizations were included in this study.
2.3 Patients
For the primary objective, as well as secondary objectives 1 and 2, patients were included if they were aged ≥ 20 years on the index date; had the following index hospitalization diagnosis codes: International Classification of Diseases-10 code I50x (indicating diseases with the highest healthcare resource consumption) and DPC code 050130 (indicating resource-intensive hospitalizations associated with HF); had no hospitalization due to HF during the pre-index period; and had observable periods of ≥ 180 days before and ≥ 1 day after the index date. Patients were excluded if their index hospitalization outcome was death. Index hospitalizations that were scheduled admissions (for examination, education, or other purposes) were also excluded. This was achieved by including only the hospitalizations registered under DPC code 050130. Patients with no hospitalization during the baseline period were excluded as they were not admitted to the same hospital. The observable period was limited to ≥ 180 days before and ≥ 1 day after the index date so that patients with a short life expectancy were not excluded. The rationale behind the 180-day cut-off was to include patients who were as close to treatment naive as possible. Patients who were not hospitalized during the 180 days before the index administration were regarded as stable.
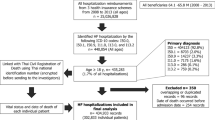
For the analysis of the economic burden of HF after discharge (secondary objectives 3 and 4), the same inclusion criteria were applied, but patients were excluded if they had observable periods that were shorter than the post-index period. A follow-up period of 365 days after the index date would allow the evaluation of the economic burden of HF on a cost per year basis. Patients could be enrolled for 1, 2, or 3 years. Figure 1 of the Electronic Supplementary Material shows the attrition flowchart for the analysis of in-hospital mortality of patients with HF and of HF-related healthcare costs.
2.4 Study Measures
The in-hospital mortality of patients with HF by number of repeated hospitalizations was analyzed as the primary outcome. Repeated hospitalization refers to any hospitalization (DPC code 050130 indicating resource-intensive hospitalizations associated with HF) that occurred after the index hospitalization. The number of HF-related hospitalizations during the study period, length of hospital stay, in-hospital mortality, and average intervals between HF-related hospitalizations were assessed. Baseline characteristic data included items that evaluated patients’ state of frailty at admission, such as the Charlson Comorbidity Index, given that frailty has been associated with increased mortality rates and healthcare utilization, including hospitalizations [23, 24]. Other baseline patient characteristics evaluated were sex, age, body mass index, New York Heart Association class, comorbidities, smoking history, activities of daily living (ADL) score at discharge, treatments during the index hospitalization, and drug prescriptions at discharge. These factors were used as covariates to analyze the association of patient baseline characteristics at the index hospitalization with repeated hospitalizations and were chosen because they have been linked to outcomes in previous studies [25, 26]. For the GDMT prescription pattern analysis, the median (interquartile range) daily doses of the drugs prescribed for chronic HF in Japan (ACEis [enalapril, lisinopril], ARBs [candesartan], β-blockers [bisoprolol, carvedilol], MRAs [spironolactone, eplerenone]) at the index date and the percentages of patients whose prescribed daily dose was lower than the minimum maintenance dose were evaluated.
For the analysis of HF-related healthcare costs, we calculated the annual health expenditure per patient after the index date. The annual health expenditures for the first, second, and third years were calculated for patients with a ≥ 3-year observable period after the index date. Factors (baseline characteristics of the patients at the index hospitalization) associated with HF-related health expenditure after discharge were evaluated. Based on the exchange rate on 2 December, 2019, ¥108.98 equaled US$1.
2.5 Statistical Analysis
To analyze factors (baseline characteristics of patients at the index hospitalization) associated with repeated hospitalizations 30 days after the index date, a Poisson regression analysis was used. For the analysis of the economic burden of HF after discharge, a multiple regression analysis (REG regression procedure in SAS) was conducted to evaluate the independent variables (baseline patient characteristics) and the dependent variable (logarithm of the HF-related health expenditure per patient during 365 days after the index date). A stepwise model was used to select the independent variables (slentry = 0.2, slstay = 0.2).
Categorical variables were reported as counts (n) and percentages (%) of total patients. Continuous variables were summarized as mean ± standard deviation (25th percentile, 75th percentile, interquartile range, and minimum, maximum).
A p-value of <0.05 was considered statistically significant. The statistical analysis in both studies was performed independently by Milliman Inc. (Tokyo, Japan) using SAS software (version 9.4; SAS Institute Inc., Cary, NC, USA) and Excel (version 2016; Microsoft Corporation, Redmond, WA, USA).
3 Results
3.1 Prognosis of Patients with HF by the Number of Repeated Hospitalizations
The demographic and clinical characteristics of 49,094 patients included in the analysis of prognosis of patients with HF by the number of repeated hospitalizations are shown in Table 1. The mean age was 79.6 ± 10.8 years, and 52.4% of patients (n = 25,747) were male. Patients aged > 80 years accounted for 55.0% (n = 27,019) of the analysis population. The most common comorbidity was hypertension (83.7%; n = 41,110). The mean length of index hospital stay was 22.9 ± 21.2 days. At discharge from the index hospitalization, 75.0% of patients (n = 36,816) were prescribed loop diuretics; 48.4% (n = 23,778), β-blockers; 36.1% (n = 17,715), MRAs; 30.8% (n = 15,129), ARBs; and 19.2% (n = 9408), ACEis. The mean observable period after the index date was 471.8 ± 413.6 days. The mean number of HF-related repeated hospitalizations and non-HF-related hospitalizations per patient during the observable period after the index date was 0.5 ± 0.0 and 1.0 ± 1.5, respectively.
Overall, 30.1% of patients (14,800/49,094) experienced HF-related repeated hospitalization. The distribution of HF-related repeated hospitalizations was right-skewed (Fig. 1a): 9659 patients underwent only one HF-related hospitalization, 2890 underwent two repeated hospitalizations, and only four underwent 14 repeated hospitalizations. Overall, the length of hospital stay remained relatively constant at approximately 22 days (Fig. 1b). The in-hospital mortality rate was also relatively constant, at approximately 10%, from the first to the fifth repeated hospitalization (Fig. 1c). The time between HF-related repeated hospitalizations seemed to decrease with increasing repeated hospitalizations (Fig. 1d).
Patients with the total number of heart failure (HF)-related repeated hospitalizations (a); and length of hospital stay (b), in-hospital mortality (c), and the time interval between repeated hospitalizations (d) by the order of HF-related repeated hospitalizations. In a, N denotes the number of patients who had the total number of HF-related repeated hospitalizations as indicated by the x-axis; thus, the total is exactly the number of patients included in this analysis (N = 49,098). In b–d, N denotes the number of patients at each turn of repeated hospitalization, and thus, the total number of patients will not add to 49,098. SD standard deviation. *The number ‘0’ denotes no re-hospitalization (i.e., patients had only one hospitalization). #The number ‘0’ denotes initial hospitalization
Multivariate analysis results for factors correlated with the number of HF-related hospitalizations are shown in Table 2. By the Poisson regression analysis, there was a positive correlation for age; male sex; body mass index <18.5 kg/m2; ADL score at discharge; comorbidities including diabetes mellitus, hyperuricemia, or ventricular tachycardia; Charlson Comorbidity Index; prescriptions at discharge of loop diuretics, tolvaptan, or inotropes; and thiazide diuretics. Dementia, squared Charlson Comorbidity Index, and prescription of MRA at discharge were correlated with the number of HF-related hospitalizations. In particular, ventricular tachycardia as a comorbidity and the use of inotrope at discharge were strongly correlated with repeated hospitalizations.
3.2 GDMT Prescription Patterns
Regarding GDMT, many patients prescribed ACEis (45.0%; n = 7874) and β-blockers (33.0%; n = 15,222) were receiving daily doses that were below the minimum maintenance dose at the time of discharge of the index hospitalization, while the majority (87.0%; n = 24,324) of patients were receiving ARBs at a daily dose greater than the minimum maintenance dose (i.e., within the recommended dose range) (Fig. 2).
Additionally, a large percentage of the patients prescribed MRAs (84.0%; n = 27,960) were receiving daily doses that were below the minimum maintenance dose (Fig. 2). Among patients who were prescribed a spironolactone 25-mg/day maintenance dose, 11.4% were receiving a dose lower than the approved maintenance dose (Table 3).
3.3 Economic Burden of HF After Discharge
A total of 42,830 patients were analyzed to investigate the economic burden of HF after discharge. The demographic and clinical characteristics were broadly similar to those of patients in the analysis for prognosis (Table 1 of the ESM). The mean observable period after the index date was 2.3 ± 1.0 years. The mean numbers of hospitalizations during the observable period after the index date were 0.7 ± 1.3 and 1.4 ± 1.8 for HF-related and non-HF-related hospitalizations, respectively.
The annual healthcare cost per patient, including both the HF-related and non-HF-related healthcare costs, was ¥1,513,926 per person-year (PY) [equivalent to 13,892 USD/PY], of which 62.7% (949,649 ¥/PY) was non-HF-related healthcare costs. The mean HF-related healthcare cost per patient was ¥564,281 ± 990,447 (US$5178 ± 9088), of which 67.3% was hospitalization costs.
Of the total HF-related hospitalization costs, 57.7% were other medical expenses (325,340 ¥/PY), of which basic hospitalization fees contributed the most at 71.7% (233,146 ¥/PY) (Fig. 3). However, the medical expenses per hospitalization were similar, with similar ratios of direct and other expenses (Fig. 4).
Table 4 shows the relationship between patient characteristics and HF-related expenditure per patient after the index date (multiple regression analysis). Factors significantly associated with higher HF-related expenditures were the length of hospital stay; presence of chronic obstructive pulmonary disease, valvular heart disease, atrial fibrillation, and hypertension; and the prescription of β-blockers and tolvaptan at discharge. Factors significantly associated with lower HF-related expenditures were the prescription of vasodilators other than carperitide and ACEi/ARBs at discharge. Particularly, the use of tolvaptan and comorbid hypertension were strongly associated with HF-related expenditure.
4 Discussion
Herein, we report the results of an administrative database study designed to estimate the burden of repeated hospitalizations on patients with HF in Japan. The distribution of repeated hospitalizations for HF was right-skewed, owing to a longer tail for increased frequency of hospitalizations. The overall length of index hospital stay remained relatively constant throughout the study period. Moreover, patients with an increasing number of repeated hospitalizations had shorter intervals between hospitalizations, suggesting a rapid deterioration and worsening condition of their disease over time.
Unlike the present findings, in previous studies [14, 27], repeated hospitalizations were found to predict mortality in the community population with HF. Other registries conducted in Japan reported relatively low but variable in-hospital mortality rates (1.3% in JASPER, 4.7% in WET‐HF, 5.6% in JCARE-CARD, and 6.4% in ATTEND) [28,29,30]. The reported in-hospital mortality rates for patients with HF-related hospitalization were 20.5% in Europe and 8.3% in the USA [5]. However, it is difficult to directly compare outcomes with previous studies conducted in other geographic regions, owing to dissimilarities in patient factors and healthcare regimens.
In our analysis, the in-hospital mortality rate remained fairly constant (approximately 10%) for fewer than five repeated hospitalizations during the study period, with some variations in small numbers of patients. One reason may be that we specifically measured the in-hospital mortality rate because the lack of a follow-up in this study precluded measurement of the mortality rate after discharge. Our study did not adjust for confounders related to hospitalization for HF; specifically, patients who were dead at index hospitalization were excluded. However, those who underwent frequent admissions to a hospital because of HF may tend to be vulnerable, and such patients might die in out-of-hospital settings. Previous studies have reported that although hospital stays tend to be longer among Japanese patients with HF compared with those in other regions [17], these longer hospital stays were linked to lower 1-year mortality and fewer readmissions [16, 30].
We found that patients with an increasing number of HF-related repeated hospitalizations were more likely to be younger, male, have several comorbidities, including diabetes mellitus, hyperuricemia, or ventricular tachycardia, be prescribed diuretics or inotropic drugs at discharge, and have high ADL scores, among others. These findings are aligned with the findings of a previous study [8] that characterized the “frequent HF admitter” phenotype (more than two admissions for HF per year) as younger male smokers with higher burdens of comorbidities, including a history of HF, chronic renal insufficiency, coronary artery disease, prior myocardial infarction, atrial fibrillation, dyslipidemias, diabetes mellitus, peripheral vascular disease, and chronic obstructive pulmonary disease. Further, frequent HF admitters were associated with adverse clinical outcomes and a higher risk of future admissions [8]. It is unclear why dementia was correlated with a reduced number of HF-related hospitalizations. However, such patients tend to stay in rehabilitation facilities for the elderly and are not generally admitted to hospitals equipped for critical care that are covered in the database.
Although no multicenter real-world studies to specifically examine drug dosing have yet been conducted in Japan, the results of our analysis demonstrated that GDMT was not adequately implemented. Many patients were prescribed ACEis (45.0%) and β-blockers (33.0%) at lower doses than the minimum maintenance dose. In contrast, 87.0% of patients prescribed ARBs were receiving daily doses greater than the minimum maintenance dose (i.e., within the recommended dose range). The reasons underlying the high proportion of ARB prescriptions above the maintenance dose are unclear. However, ARBs, rather than ACEis, tend to be prescribed for hypertensive patients in Japan, and the dosage could be up-titrated. Another potential reason for this finding is that ACEis are not up-titrated because of concern about adverse events, such as cough.
This study found that MRAs were prescribed at doses lower than the maintenance dose in 84.0% of patients. An interesting observation was that of patients prescribed spironolactone at a maintenance dose of 25 mg/day, only 11.4% were receiving a dose lower than the approved maintenance dose. The Japanese Circulation Society and the Japanese Heart Failure Society [31] recommend spironolactone 12.5 mg/day and eplerenone 25 mg/day as loading doses. This may explain why 11.4% of patients prescribed spironolactone 25 mg/day received a dose lower than the maintenance dose. Another possible reason for the differences in the maintenance doses used could be the availability of each drug, as drug stock-keeping units may be limited depending on the capacity/available space of each pharmacy and the proximity of each pharmacy to a hospital.
Real-world evidence obtained from patients hospitalized with HF in 36 countries has shown that adherence to guideline dosing recommendations for HF medication is associated with improved outcomes (reductions in cardiovascular events and lower mortality rates because of HF) [32]. Moreover, a randomized double-blind study with Japanese patients with chronic HF reported a dose-dependent improvement in risk reduction and HF-related hospitalization with a β-blocker [33]. Therefore, medication prescription should be monitored and increased in dosage, where possible, to mitigate the negative effects of suboptimal dosing. A recent publication has outlined a framework to promote physician awareness of subtarget dosing and to increase adherence to clinically recommended dosages with proven efficacy [34]. Overall, these findings are consistent with the underuse and underdosing described in other registries evaluating medication therapy for HF [35, 36].
Of the HF-related healthcare costs per patient, high other costs were observed, particularly for the basic hospitalization fees (71.7%). Furthermore, the costs of HF-related hospitalization (67.3%) were two-fold the costs of outpatient care (32.7%). Some studies have reported that inpatient costs are not associated with patient prognosis, but rather with physicians decisions and an indication of high-cost procedures, which are not necessarily related to improved patient outcomes [18, 20, 22].
In terms of outpatient costs, healthcare costs associated with HF treatment accounted for nearly half the total. These HF-related expenses could potentially be reduced by making more effective treatment choices. Current findings suggest that the use of ACEi/ARBs and MRAs, which are expected to improve the prognosis [37], was associated with a reduction in healthcare costs, whereas β-blockers, also considered to improve the prognosis in patients with HF [1, 38], were suggested to be associated with increased healthcare costs. As such, ACEi/ARB, MRA, and β-blocker use may be factors influencing healthcare costs. Previous studies have concluded that although prescription of some medications may increase immediate costs related to drug treatments, ultimately, these may result in lower overall total healthcare costs [39]. Therefore, healthcare costs in our analysis may have been elevated because the prognosis of patients with HF was not improved following treatment with inotropic drugs. Of note, the efficacy of short-term treatment with carperitide has not been widely validated for HF. In previous Japanese studies, this drug was not associated with lower in-hospital mortality rates, and patients who had a poor response to this drug showed poor prognosis and worsening of HF [40,41,42]. Thus, initial vasodilator drug choices must be carefully considered in order to avoid potential increases in healthcare costs.
It is also important to combine management approaches to reduce the number of repeated hospitalizations as well as HF-related healthcare costs. Some potential strategies may include the intensification of treatment and a stricter application of GDMT for HF. As the repeated hospitalizations observed in our analysis occurred frequently in young patients with high ADL scores, and HF is a progressive disease, these patients may be an appropriate target for the intensification of treatment.
4.1 Study Limitations
This study had several limitations. First, the Medical Data Vision database covered approximately 15% of acute hospitals in Japan at the time of the analysis, potentially resulting in selection bias. Second, missing echocardiography data precluded analyses of left ventricular ejection fraction (EF). Therefore, we were not able to distinguish from HF with reduced EF, HF with midrange EF, and HF with preserved EF, all of which have different pathophysiology. Third, mortality data of patients with HF after discharge could not be assessed, which precluded the assessment of a change in overall mortality. Analysis was limited by censoring of patients due to death or otherwise lost to follow-up. As each patient in the Medical Data Vision database is identified by an identification number individually assigned by each institution, medical treatment prescribed at other institutions is not traceable. Therefore, it is not possible to obtain information about hospitalization and outpatient visits at other facilities. Fourth, the outcomes in this study were limited to inpatient and outpatient visits in the facility where patients were hospitalized at the index hospitalization. Thus, the costs of hospitalization and outpatient visits to medical institutions other than those from the index hospitalization are not included in this analysis. Fifth, the database was limited in that insurance claims data may be prone to error because they are not collected specifically for research purposes; data relevant to the study outcomes may have been incomplete or inaccurate. For example, HF diagnosis and comorbidities were identified using diagnostic codes; these are subject to potential miscoding, which represents an inherent risk of information bias. Finally, new HF drugs were approved in Japan after the conclusion of the follow-up period of our study; therefore, the effect of these new drugs should be assessed in future studies.
5 Conclusions
The present results suggest that repeated hospitalizations for HF shorten the intervals between HF-related hospitalizations, which could reflect the deterioration or worsening of the patients’ HF status. However, the in-hospital mortality rates after discharge in the studied population remained relatively constant for fewer than five repeated hospitalizations during the observed period. Hospitalization costs accounted for most of the HF-related health expenditures per patient, and the proportion of other costs, such as basic hospitalization fees, was particularly high. Treatment intensifications that reduce HF-related hospitalizations may be essential in reducing the economic burden of HF after discharge. It is worth further investigating the effect of treatment intensification on repeated hospitalizations and associated costs in future interventional trials.
References
McMurray JJV, Adamopoulos S, Anker SD, Auricchio A, Böhm M, Dickstein K, et al. ESC guidelines for the diagnosis and treatment of acute and chronic heart failure 2012: the Task Force for the diagnosis and treatment of acute and chronic heart failure 2012 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association (HFA) of the ESC. Eur J Heart Fail. 2012;14:803–69. https://doi.org/10.1093/eurjhf/hfs105.
Kondo K. Progress in aging epidemiology in Japan: the JAGES project. J Epidemiol. 2016;26:331–6. https://doi.org/10.2188/jea.JE20160093.
United Nations. World population prospects: the 2017 revision, key findings and advance tables. 2017. https://population.un.org/wpp/Publications/Files/WPP2017_KeyFindings.pdf. Accessed 7 Jun 2021.
Ejiri K, Noriyasu T, Nakamura K, Ito H. Unprecedented crisis: heart failure hospitalizations in current or future Japan. J Cardiol. 2019;74:426–7. https://doi.org/10.1016/j.jjcc.2019.05.006.
Konishi M, Ishida J, Springer J, von Haehling S, Akashi YJ, Shimokawa H, et al. Heart failure epidemiology and novel treatments in Japan: facts and numbers. ESC Heart Fail. 2016;3:145–51. https://doi.org/10.1002/ehf2.12103.
Okura Y, Ramadan MM, Ohno Y, Mitsuma W, Tanaka K, Ito M, et al. Impending epidemic: future projection of heart failure in Japan to the year 2055. Circ J. 2008;72:489–91. https://doi.org/10.1253/circj.72.489.
Pocock SJ, Wang D, Pfeffer MA, Yusuf S, McMurray JJ, Swedberg KB, et al. Predictors of mortality and morbidity in patients with chronic heart failure. Eur Heart J. 2006;27:65–75. https://doi.org/10.1093/eurheartj/ehi555.
Go YY, Sellmair R, Allen JC Jr, Sahlén A, Bulluck H, Sim D, et al. Defining a ‘frequent admitter’ phenotype among patients with repeat heart failure admissions. Eur J Heart Fail. 2019;21:311–8. https://doi.org/10.1002/ejhf.1348.
Hamaguchi S, Kinugawa S, Goto D, Tsuchihashi-Makaya M, Yokota T, Yamada S, et al. Predictors of long-term adverse outcomes in elderly patients over 80 years hospitalized with heart failure: a report from the Japanese Cardiac Registry of Heart Failure in Cardiology (JCARE-CARD). Circ J. 2011;75:2403–10. https://doi.org/10.1253/circj.cj-11-0267.
Sato M, Sakata Y, Sato K, Nochioka K, Miura M, Abe R, et al. Clinical characteristics and prognostic factors in elderly patients with chronic heart failure: a report from the CHART-2 study. Int J Cardiol Heart Vasc. 2020;27: 100497. https://doi.org/10.1016/j.ijcha.2020.100497.
Ushigome R, Sakata Y, Nochioka K, Miyata S, Miura M, Tadaki S, et al. Temporal trends in clinical characteristics, management and prognosis of patients with symptomatic heart failure in Japan: report from the CHART studies. Circ J. 2015;79:2396–407. https://doi.org/10.1253/circj.CJ-15-0514.
Ponikowski P, Voors AA, Anker SD, Bueno H, Cleland JGF, Coats AJS, et al. 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: the Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur Heart J. 2016;37:2129–200. https://doi.org/10.1093/eurheartj/ehw128.
Akita K, Kohno T, Kohsaka S, Shiraishi Y, Nagatomo Y, Goda A, et al. Current use of guideline-based medical therapy in elderly patients admitted with acute heart failure with reduced ejection fraction and its impact on event-free survival. Int J Cardiol. 2017;235:162–8. https://doi.org/10.1016/j.ijcard.2017.02.070.
Setoguchi S, Stevenson LW, Schneeweiss S. Repeated hospitalizations predict mortality in the community population with heart failure. Am Heart J. 2007;154:260–6. https://doi.org/10.1016/j.ahj.2007.01.041.
Zomer AC, Vaartjes I, van der Velde ET, de Jong HMY, Konings TC, Wagenaar LJ, et al. Heart failure admissions in adults with congenital heart disease; risk factors and prognosis. Int J Cardiol. 2013;168:2487–93. https://doi.org/10.1016/j.ijcard.2013.03.003.
Akita K, Kohno T, Kohsaka S, Shiraishi Y, Nagatomo Y, Izumi Y, et al. Prognostic impact of previous hospitalization in acute heart failure patients. Circ J. 2019;83:1261–8. https://doi.org/10.1253/circj.CJ-18-1087.
Ambrosy AP, Fonarow GC, Butler J, Chioncel O, Greene SJ, Vaduganathan M, et al. The global health and economic burden of hospitalizations for heart failure: lessons learned from hospitalized heart failure registries. J Am Coll Cardiol. 2014;63:1123–33. https://doi.org/10.1016/j.jacc.2013.11.053.
Groeneveld PW, Medvedeva EL, Walker L, Segal AG, Menno DM, Epstein AJ. Association between spending and survival of chronic heart failure across Veterans Affairs medical centers. JAMA Netw Open. 2019;2: e197238. https://doi.org/10.1001/jamanetworkopen.2019.7238.
Ziaeian B, Sharma PP, Yu TC, Johnson KW, Fonarow GC. Factors associated with variations in hospital expenditures for acute heart failure in the United States. Am Heart J. 2015;169:282-9.e15. https://doi.org/10.1016/j.ahj.2014.11.007.
Sasaki N, Kunisawa S, Ikai H, Imanaka Y. Differences between determinants of in-hospital mortality and hospitalisation costs for patients with acute heart failure: a nationwide observational study from Japan. BMJ Open. 2017;7: e013753. https://doi.org/10.1136/bmjopen-2016-013753.
Kanaoka K, Okayama S, Nakai M, Sumita Y, Nishimura K, Kawakami R, et al. Hospitalization costs for patients with acute congestive heart failure in Japan. Circ J. 2019;83:1025–31. https://doi.org/10.1253/circj.CJ-18-1212.
Tsugawa Y, Jha AK, Newhouse JP, Zaslavsky AM, Jena AB. Variation in physician spending and association with patient outcomes. JAMA Intern Med. 2017;177:675–82. https://doi.org/10.1001/jamainternmed.2017.0059.
Gastelurrutia P, Lupón J, Altimir S, de Antonio M, González B, Cabanes R, et al. Fragility is a key determinant of survival in heart failure patients. Int J Cardiol. 2014;175:62–6. https://doi.org/10.1016/j.ijcard.2014.04.237.
Vitale C, Spoletini I, Rosano GM. Frailty in heart failure: implications for management. Card Fail Rev. 2018;4:104–6. https://doi.org/10.15420/cfr.2018.22.2.
Collier TJ, Pocock SJ, McMurray JJV, Zannad F, Krum H, van Veldhuisen DJ, et al. The impact of eplerenone at different levels of risk in patients with systolic heart failure and mild symptoms: insight from a novel risk score for prognosis derived from the EMPHASIS-HF trial. Eur Heart J. 2013;34:2823–9. https://doi.org/10.1093/eurheartj/eht247.
Pocock SJ, Ariti CA, McMurray JJV, Maggioni A, Køber L, Squire IB, et al. Predicting survival in heart failure: a risk score based on 39 372 patients from 30 studies. Eur Heart J. 2013;34:1404–13. https://doi.org/10.1093/eurheartj/ehs337.
Bello NA, Claggett B, Desai AS, McMurray JJV, Granger CB, Yusuf S, et al. Influence of previous heart failure hospitalization on cardiovascular events in patients with reduced and preserved ejection fraction. Circ Heart Fail. 2014;7:590–5. https://doi.org/10.1161/CIRCHEARTFAILURE.113.001281.
Hamaguchi S, Kinugawa S, Tsuchihashi-Makaya M, Goto D, Yamada S, Yokoshiki H, et al. Characteristics, management, and outcomes for patients during hospitalization due to worsening heart failure: a report from the Japanese Cardiac Registry of Heart Failure in Cardiology (JCARE-CARD). J Cardiol. 2013;62:95–101. https://doi.org/10.1016/j.jjcc.2013.03.009.
Nagai T, Yoshikawa T, Saito Y, Takeishi Y, Yamamoto K, Ogawa H, et al. Clinical characteristics, management, and outcomes of Japanese patients hospitalized for heart failure with preserved ejection fraction: a report from the Japanese Heart Failure Syndrome With Preserved Ejection Fraction (JASPER) registry. Circ J. 2018;82:1534–45. https://doi.org/10.1253/circj.CJ-18-0073.
Shiraishi Y, Kohsaka S, Sato N, Takano T, Kitai T, Yoshikawa T, et al. 9-year trend in the management of acute heart failure in Japan: a report from the National Consortium of Acute Heart Failure Registries. J Am Heart Assoc. 2018;7: e008687. https://doi.org/10.1161/JAHA.118.008687.
Tsutsui H, Isobe M, Ito H, Ito H, Okumura K, Ono M, et al. JCS 2017/JHFS 2017 guideline on diagnosis and treatment of acute and chronic heart failure: digest version. Circ J. 2019;83:2084–184. https://doi.org/10.1253/circj.CJ-19-0342.
Komajda M, Schöpe J, Wagenpfeil S, Tavazzi L, Böhm M, Ponikowski P, et al. Physicians’ guideline adherence is associated with long-term heart failure mortality in outpatients with heart failure with reduced ejection fraction: the QUALIFY international registry. Eur J Heart Fail. 2019;21:921–9. https://doi.org/10.1002/ejhf.1459.
Hori M, Sasayama S, Kitabatake A, Toyo-oka T, Handa S, Yokoyama M, et al. Low-dose carvedilol improves left ventricular function and reduces cardiovascular hospitalization in Japanese patients with chronic heart failure: the Multicenter Carvedilol Heart Failure Dose Assessment (MUCHA) trial. Am Heart J. 2004;147:324–30. https://doi.org/10.1016/j.ahj.2003.07.023.
Packer M, Metra M. Guideline-directed medical therapy for heart failure does not exist: a non-judgmental framework for describing the level of adherence to evidence-based drug treatments for patients with a reduced ejection fraction. Eur J Heart Fail. 2020;22:1759–67. https://doi.org/10.1002/ejhf.1857.
Greene SJ, Butler J, Albert NM, DeVore AD, Sharma PP, Duffy CI, et al. Medical therapy for heart failure with reduced ejection fraction: the CHAMP-HF registry. J Am Coll Cardiol. 2018;72:351–66. https://doi.org/10.1016/j.jacc.2018.04.070.
Teng TK, Tromp J, Tay WT, Anand I, Ouwerkerk W, Chopra V, et al. Prescribing patterns of evidence-based heart failure pharmacotherapy and outcomes in the ASIAN-HF registry: a cohort study. Lancet Glob Health. 2018;6:e1008–18. https://doi.org/10.1016/S2214-109X(18)30306-1.
Davidson A, Raviendran N, Murali CN, Myint PK. Managing heart failure with preserved ejection fraction. Ann Transl Med. 2020;8:395. https://doi.org/10.21037/atm.2020.03.18.
Yamamoto K. β-blocker therapy in heart failure with preserved ejection fraction: importance of dose and duration. J Cardiol. 2015;66:189–94. https://doi.org/10.1016/j.jjcc.2015.02.004.
Albert NM, Swindle JP, Buysman EK, Chang C. Lower hospitalization and healthcare costs with sacubitril/valsartan versus angiotensin-converting enzyme inhibitor or angiotensin-receptor blocker in a retrospective analysis of patients with heart failure. J Am Heart Assoc. 2019;8: e011089. https://doi.org/10.1161/JAHA.118.011089.
Matsue Y, Kagiyama N, Yoshida K, Kume T, Okura H, Suzuki M, et al. Carperitide is associated with increased in-hospital mortality in acute heart failure: a propensity score-matched analysis. J Card Fail. 2015;21:859–64. https://doi.org/10.1016/j.cardfail.2015.05.007.
Ogiso M, Isogai T, Okabe Y, Ito K, Tsuji M, Tanaka H. Effect of carperitide on in-hospital mortality of patients admitted for heart failure: propensity score analyses. Heart Vessels. 2017;32:916–25. https://doi.org/10.1007/s00380-017-0952-0.
Okuhara Y, Asakura M, Azuma K, Orihara Y, Nishimura K, Ando T, et al. Effects of early diuretic response to carperitide in acute decompensated heart failure treatment: a single-center retrospective study. PLoS ONE. 2018;13: e0199263. https://doi.org/10.1371/journal.pone.0199263.
Acknowledgements
The authors wish to thank Keyra Martinez Dunn, MD, of Edanz, Japan, for providing medical writing support, which was funded by Novartis Pharma K.K., Japan, through EMC K.K., Japan, in accordance with Good Publication Practice (GPP3) guidelines (http://www.ismpp.org/gpp3).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This research was supported by Novartis Pharma K.K. The sponsor had a role in the conduct of the study; in the collection, analysis, and interpretation of data; in the writing of the report; and in the decision to submit the article for publication. Funding for the Open Access fee was provided by the sponsor.
Conflict of Interest
S.E., Y.M., H.M., H.K., and N.O. are employees of Novartis Pharma K.K. A.K. and K.I. are employees of Milliman, Inc. T.Y. has no conflict of interest to declare.
Ethics Approval
This study was approved by the Ethics Review Committee of the nonprofit corporation Clinical Research Promotion Network Japan (protocol numbers: CLCZ696BJP01 and CLCZ696BJP02).
Consent to Participate
The requirement of informed consent was waived given that the database protects patient anonymity.
Consent for Publication
There was no requirement to obtain consent for publication given that the database protects patient anonymity.
Availability of Data and Material
The individual deidentified participant data, study protocol, and statistical analysis plan will be shared upon reasonable requests made directly to the corresponding author (shunsuke.eguchi@novartis.com). The data requester should provide a methodologically sound proposal when making the request. The requested data will be available immediately and up to 1 year following the publication of this article.
Code Availability
Not applicable.
Authors’ Contributions
All authors contributed to all of the following: (1) study design, conduct, or collection; (2) data analysis and interpretation, (3) writing or reviewing the manuscript, and (4) final approval of the manuscript for submission.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Eguchi, S., Morita, Y., Mitani, H. et al. Burden of Repeated Hospitalizations on Patients with Heart Failure: An Analysis of Administrative and Claims Data in Japan. Drugs - Real World Outcomes 9, 377–389 (2022). https://doi.org/10.1007/s40801-022-00315-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40801-022-00315-5