Abstract
Background
Reports on treatment patterns of biologic disease-modifying antirheumatic drugs (bDMARDs)/Janus kinase inhibitors (JAKi) for rheumatoid arthritis (RA) in clinical practice are still sparse in Japan, especially in combination with conventional synthetic DMARDs (csDMARDs).
Objectives
The aim of this study was to investigate treatment patterns of bDMARD/JAKi in the treatment of RA in real-world clinical practice in Japan.
Method
A retrospective cohort study was conducted using the Japanese Medical Data Vision health claims database. The inclusion criteria required a recorded diagnosis of RA, defined by ICD-10 codes, in patients aged 18 years and older on the index date. We analyzed 39,903 RA patients treated with DMARDs from 2008 to 2020.
Results
Among analyzed subjects, 10,196 patients (25.6%) were prescribed bDMARDs/JAKi in combination with csDMARDs, and 3067 patients (7.7%) were prescribed these drugs without csDMARDs. Among the bDMARDs/JAKi, tumor necrosis factor inhibitors (TNFi) were the most commonly prescribed DMARD overall, and also the most common first-line therapy, accounting for 60.0% or 45.5% of patients prescribed these drugs in combination with or without csDMARDs, respectively. Switching, temporary discontinuation (restarting with the same agents), and discontinuation of bDMARDs/JAKi were observed in 3150 (30.9%), 1379 (13.5%), and 2025 (19.9%) patients with csDMARDs, and in 849 (27.7%), 513 (16.7%), and 833 (27.2%) patients without csDMARDs, respectively.
Conclusions
Real-world treatment trajectories of bDMARDs/JAKi with and without csDMARDs was analyzed in RA patients in Japan between 2008 and 2020. TNFi were the predominant first-line therapy, and likely to be switched to different classes. Understanding the current treatment patterns, including discontinuation, is important to find an optimal treatment strategy for RA patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
This claims-based study to investigate real-world treatment pattern of biologic disease-modifying antirheumatic drugs (bDMARDs)/Janus kinase inhibitors (JAKi) in Japan between 2008 and 2020 indicated that 25.6% and 7.7% of the RA patients were prescribed these drugs in combination with and without conventional synthetic DMARDs (csDMARDs), respectively. |
Tumor necrosis factor inhibitors (TNFi) were the most commonly prescribed therapy among bDMARDs/JAKi throughout the study period. |
TNFi were used mainly as first-line therapy, and patients were likely to switch to a different class regardless of combination with csDMARDs. |
Temporary discontinuation and complete discontinuation of bDMARDs/JAKi was observed in 13.5% and 19.9% of the patients receiving combination with csDMARDs, and 16.7% and 27.2% of the patients without csDMARDs, respectively. |
1 Introduction
Rheumatoid arthritis (RA) is a chronic, inflammatory autoimmune joint disease that can cause cartilage and bone damage as well as disability, which may negatively impact patients’ ability to perform daily activities and reduce health-related quality of life [1, 2]. The global prevalence of RA between 1980 and 2019 is estimated to be 0.46% based on a recent systematic review [3].
RA therapies have slowly evolved over the last century. Previously, most were based on conventional synthetic disease-modifying antirheumatic drugs (csDMARDs). However, over the past 20 years, RA treatment has undergone an innovative improvement, including the approval of effective biologic and targeted synthetic DMARDs (b/tsDMARDs) [4, 5]. With these improvements, the recommended therapeutic strategy for RA has been a treat-to-target (T2T) approach, aiming to achieve and sustain disease remission [6]. With T2T, csDMARDs are recommended as first-line treatment, most commonly methotrexate; bDMARDs or tsDMARDs such as Janus kinase inhibitors (JAKi) are recommended for use in patients with inadequate response to csDMARDs [6]. Following this dramatic improvement in RA therapies, RA experts anticipate further therapeutic advances that include the differential use of therapeutic drugs and de-escalation of treatment after remission induction [7].
In Japan, a tumor necrosis factor inhibitor (TNFi) was approved for the indication of RA in 2003 as the first bDMARD introduced in real-world clinical practice, followed by an interleukin-6 inhibitor (IL-6i) and cytotoxic T lymphocyte-associated antigen-4 immunoglobulin fusion proteins (CTLA4-Ig), approved in 2008 and 2010, respectively [8,9,10]. These three classes of bDMARDs are currently clinically available, covered by insurance, and widely used in Japanese clinical practice [11]. An estimate based on the National Database of Health Insurance Claims and Specific Health Checkups of Japan (NDB) in 2017 shows that 22.9% of 825,772 patients with RA aged ≥ 16 years received treatment with bDMARDs, increasing to 50.9% in those aged 16–19 years, and decreasing to 13.7% in those aged ≥ 85 years [11]. In Japan, the first JAKi was approved for RA in 2013 [12]. Subsequently, JAKi are increasingly used as second-line treatment following methotrexate, and the total number of JAKi prescriptions reported increased approximately 8-fold during 2016–2019, based on an analysis of the Japan Medical Data Center (JMDC) claims database containing 10,399 patients with RA [13].
Recent availability of several different types of bDMARD/JAKi now provides opportunities for multiple therapeutic strategies in Japanese clinical practice, with different treatment patterns. In harmony with EULAR endorsement of T2T [6], Japanese clinical practice guidelines for RA management recommend the use of bDMARDs/JAKi, regardless of csDMARD use [14]. Reports on Japanese treatment patterns of bDMARD/JAKi use are still sparse, especially in combination with csDMARDs. We thus conducted a retrospective study of a claims-based database between April 2008 and August 2020, to investigate bDMARD/JAKi treatment patterns, with or without csDMARDs, in Japanese patients with RA. We also analyzed factors that contribute to initiation, switching, temporary discontinuation, and discontinuation of therapies with bDMARDs/JAKi.
2 Method
2.1 Study Design
This retrospective cohort study was conducted using data extracted from a health claims database in Japan. Data from between April 2008 and August 2020 were analyzed. The primary study objective was to describe treatment patterns using bDMARDs/JAKi in patients with RA. The secondary objective was to explore the determinants of first-line prescription of bDMARD/JAKi, as well as the determinants of switching, temporary discontinuation, and discontinuation of these drugs.
2.2 Data Source
The Japanese Medical Data Vision (MDV) health claims database (Medical Data Vision; Tokyo, Japan), a hospital-based database, recognized as one of the largest and most credible commercially available medical databases in Japan, was used in this study. The MDV database contains health insurance claims data, Diagnosis and Procedure Combination (DPC) data, and administrative data from over 400 hospitals covering over 30 million patients. This database was chosen for this study over other databases as it allows us to robustly evaluate data on elderly patients, as the mean age of RA patients is 60 years and over. Both inpatient and outpatient data for each patient are included in the MDV database unless the patient has stopped visiting the hospital; all data are unlinkable and anonymized. Diagnoses of patients are coded according to the International Classification of Diseases, Tenth Revision (ICD-10) coding scheme and indicated as either ‘confirmed’ or ‘suspected’. In this study, only patients with a ‘confirmed’ diagnosis of RA defined in the study cohort section were used after excluding ‘suspected’ patients who met the exclusion criteria shown below. The prescription data for TNFi, including etanercept, etanercept biosimilar (BS), infliximab, infliximab BS, adalimumab, adalimumab BS, golimumab, and certolizumab pegol; IL-6i including tocilizumab, sarilumab; CTLA4-Ig including abatacept; and JAKi, including tofacitinib, baricitinib, peficitinib, and upadacitinib were analyzed in this study.
2.3 Study Cohort
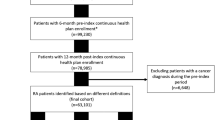
The inclusion criteria required patients to have a diagnosis of RA, defined by ICD-10 codes of M05 and M06, and to be at least 18 years of age on the index date. The index date was defined as the first date when a bDMARD/JAKi was prescribed after the first RA diagnosis between April 2008 and August 2020 in the DPC hospital. For patients who had no record of bDMARDs/JAKi (who were prescribed csDMARDs only), the index date was defined as the first prescription of csDMARD after the first diagnosis date. The exclusion criteria required patients who did not have data 3 months prior to the index date or 1 year after the index date to be excluded, as they would not have any data for baseline evaluation or endpoint assessment. In addition, patients diagnosed with ICD-10 codes for Crohn’s disease, ankylosing spondylitis, juvenile arthritis, psoriasis, ulcerative colitis and/or Behçet’s disease were excluded, even if the diagnosis was only a ‘suspected’ diagnosis. Patients who did not receive any prescription for antirheumatic drugs (including csDMARDs) or those who received a prescription before the first diagnosis date were also excluded. Patients who met all the inclusion criteria and none of the exclusion criteria were considered eligible. The csDMARDs included were methotrexate, sulfasalazine, bucillamine, iguratimod, actarit, leflunomide, penicillamine, gold salts, tacrolimus, mizoribine, lobenzarit, and auranofin. Concomitant use of csDMARDs was judged by the presence or absence of the record at the first prescription of bDMARD/JAKi. The patient flow diagram is shown in Supplementary Fig. 1 (see electronic supplementary material [ESM]). Among 380,328 patients diagnosed with RA during the period from 2008 to 2020, a total of 39,903 patients with RA were prescribed DMARDs and eligible for inclusion according to the selection criteria. Therefore, data of these 39,903 RA patients were analyzed.
2.4 Endpoints
The treatment patterns for bDMARDs/JAKi were evaluated using the following endpoints: (i) characteristics of patients prescribed bDMARDs/JAKi including age, sex, comorbidities, patient’s Charlson comorbidity index (CCI) score; (ii) change in the proportion of patients prescribed bDMARDs/JAKi from 2008 to 2020 by year; (iii) proportion of bDMARDs/JAKi prescribed as first- to third-line therapy; (iv) switching of the first-line bDMARDs/JAKi; and (v) proportion of patients undergoing temporary discontinuation or discontinuation of bDMARDs/JAKi. In this study, temporary discontinuation was defined as bDMARDs/JAKi suspension for at least 3 months, followed by re-prescription of the same drug during the study period. Discontinuation was defined as a period of >3 months after the last administration of bDMARDs/JAKi, without re-prescription during the study period. The determinants of the first-line prescription of bDMARD/JAKi, as well as the determinants of the switching, temporary discontinuation, and discontinuation of these drugs were identified as factors associated with an occurrence of each of the four events.
2.5 Statistical Analysis
Descriptive statistics used included number of patients, mean and standard deviation for continuous variables, and frequency distribution for categorical variables. The number and proportions of patients who were prescribed bDMARDs/JAKi during the study period were calculated by the group of a different type or a different drug of the same type, in combination with or without csDMARDs. A Sankey diagram was used to illustrate switching of first-line bDMARDs/JAKi to second- or third-line therapies, using R-studio and R software. To explore the determinants of first-line prescriptions, switching, temporary discontinuation, and discontinuation of bDMARDs/JAKi with follow-up period from 2008 to 2020, logistic regression model analysis was performed. Univariate analysis was applied for each candidate predictor. Thereafter, without model selection, multivariate analysis was applied for all candidate predictors. Age (reference: ≤ 60 years, vs 61–75 years and > 75 years), sex (reference: male, vs female), and CCI score (reference: < 2, vs 3–5 and > 5), and combination with csDMARDs (reference: with, vs without) were used as candidate predictors. The results were presented as odds ratios (ORs) and 95% confidence intervals (CIs). A p value < 0.05 was considered to be statistically significant in all analyses except for the univariate analysis. SAS version 9.4 (TS1M6) (SAS/STAT 15.1) was used for all analyses.
3 Results
3.1 Study Cohort
Among the 39,903 RA patients included in this study, 10,196 patients (25.6%) were prescribed bDMARDs, tsDMARDs, or JAKi in combination with csDMARDs; 3067 patients (7.7%) were prescribed bDMARDs/JAKi without csDMARDs; and 26,640 patients (66.8%) were prescribed csDMARDs only (Supplementary Fig. 1, see ESM).
Characteristics of the eligible patients are shown in Table 1. Among the 10,196 patients prescribed bDMARDs/JAKi with csDMARDs, TNFi, IL-6i, CTLA4-Ig, and JAKi were prescribed for 6116 patients (60.0%), 1725 patients (16.9%), 1997 patients (19.6%), and 358 patients (3.5%), respectively. Among the 3067 patients prescribed bDMARDs/JAKi without csDMARDs, TNFi, IL-6i, CTLA4-Ig, and JAKi were prescribed for 1395 patients (45.5%), 856 patients (27.9%), 689 patients (22.5%), and 127 patients (4.1%), respectively.
Among the 10,196 patients prescribed bDMARDs/JAKi with csDMARDs, switching, temporary discontinuation, and discontinuation were observed in 3150 patients (30.9%), 1379 patients (13.5%), and 2025 patients (19.9%), respectively. Among the 3067 patients prescribed bDMARDs/JAKi without csDMARDs, switching, temporary discontinuation, and discontinuation were also observed in 849 patients (27.7%), 513 patients (16.7%), and 833 patients (27.2%), respectively (Supplementary Table 1, see ESM).
3.2 Change in the Proportion of Patients Prescribed bDMARDs/JAKi
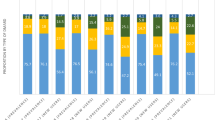
Figure 1 shows the change over time in the proportion of patients prescribed bDMARDs/JAKi in the study cohort during the period from April 2008 to August 2020. TNFi, IL-6i, CTLA4-Ig and JAKi were first introduced into real-world clinical practice in Japan in 2003, 2008, 2010, and 2013, respectively [8,9,10, 12]. Eight TNFi became clinically available with coverage by Japanese insurance, compared with just two IL-6i and one CTLA4-Ig. Prescription numbers for each bDMARD increased over time after introduction, although the count decreased in 2020 because of a short evaluation period for that year. JAKi prescriptions continued to increase after the introduction. Among bDMARDs/JAKi, TNFi were the most commonly prescribed treatment throughout this period.
Change in the proportion of patients prescribed bDMARDs/JAKi during the period from 2008 to 2020. Data are shown as the percentage of patients prescribed bDMARDs/JAKi, relative to all patients prescribed DMARDs in that year. Of note, the count decreased in 2020 because of a short evaluation period that year. bDMARDs biologic disease-modifying antirheumatic drugs, CTLA4-Ig cytotoxic T lymphocyte-associated antigen-4 immunoglobulin fusion proteins, IL-6i interleukin-6 inhibitor, JAKi Janus kinase inhibitor, TNFi tumor necrosis factor inhibitor
3.3 Proportion of the Prescribed bDMARDs/JAKi as a First- to Third-Line Therapy During 2008 to 2020
Regardless of whether these drugs were combined with csDMARDs or not, TNFi were most commonly prescribed as first-line bDMARDs/JAKi for patients with RA, respectively accounting for 60.0% or 45.5% of patients prescribed these drugs as first-line therapy (Fig. 2). TNFi were also the most commonly prescribed class of second- and third-line bDMARDs/JAKi. JAKi were more frequently prescribed as second- or third-line bDMARDs/JAKi compared with first-line prescription.
Proportion of the prescribed bDMARDs/JAKi as a first- to third-line therapy used during 2008 to 2020. bDMARDs biologic disease-modifying antirheumatic drugs, csDMARD conventional synthetic disease-modifying antirheumatic drug, CTLA4-Ig cytotoxic T lymphocyte-associated antigen-4 immunoglobulin fusion proteins, IL-6i interleukin-6 inhibitor, JAKi Janus kinase inhibitor, TNFi tumor necrosis factor inhibitor
3.4 Treatment Trajectories from the First bDMARD/JAKi
Switching of bDMARDs/JAKi was observed in 30.9% of patients also prescribed csDMARDs, and 27.7% of those without (Supplementary Table 1, see ESM). Sankey diagrams indicate that pathways for switching first-line bDMARDs/JAKi to a second- or third-line therapy varied, including switching to another drug class as well as switching to another drug within the same class of TNFi, IL-6i, or JAKi (Fig. 3). Supplementary Table 2 shows changes from first- to third-line therapy using bDMARD/JAKi, for each bDMARD (TNFi, IL-6i, and CTLA4-Ig) or JAKi, in addition to the number of patients prescribed each drug.
Sankey diagrams showing the switching of first-line bDMARDs/JAKi to second and third line during 2008 to 2020. bDMARDs biologic disease-modifying antirheumatic drugs, csDMARD conventional synthetic disease-modifying antirheumatic drug, CTLA4-Ig cytotoxic T lymphocyte-associated antigen-4 immunoglobulin fusion proteins, IL-6i interleukin-6 inhibitor, JAKi Janus kinase inhibitor, TNFi tumor necrosis factor inhibitor
3.5 Determinants of First-Line Prescription of bDMARDs/JAKi
Multivariate logistic regression analysis explored the determinants of first-line prescription of bDMARDs/JAKi, using the possible determinants of age, sex, and the patient’s CCI score (Table 2A). This analysis revealed that ages of 61–75 years and > 75 years were significantly negative determinants of the first-line prescription of TNFi (OR 0.72, 95% CI 0.66–0.80, p < 0.001 and OR 0.55, 95% CI 0.49–0.62, p < 0.001, respectively) and IL-6i (OR 0.80, 95% CI 0.71–0.90, p < 0.001 and OR 0.53, 95% CI 0.45–0.62, p < 0.001, respectively), and significantly positive determinants of first-line prescription of CTLA4-Ig (OR 2.23, 95% CI 1.96–2.54, p < 0.001 and OR 4.31, 95% CI 3.73–4.98, p < 0.001, respectively). Female sex was a significantly positive determinant of TNFi prescription (OR 1.11, 95% CI 1.01–1.22, p = 0.034), but a significantly negative determinant of IL-6i (OR 0.86, 95% CI 0.76–0.97, p = 0.014) and JAKi prescription (OR 0.76, 95% CI 0.60–0.96, p = 0.024). CCI scores of 3–5 and > 5 were significantly negative determinants of TNFi prescription (OR 0.68, 95% CI 0.62–0.75, p < 0.001 and OR 0.51, 95% CI 0.41–0.64, p < 0.001, respectively), and significantly positive determinants of IL-6i (OR 1.23, 95% CI 1.08–1.39, p = 0.001 and OR 1.57, 95% CI 1.21–2.04, p < 0.001, respectively) and CTLA4-Ig prescription (OR 1.45, 95% CI 1.29–1.63, p < 0.001 and OR 1.74, 95% CI 1.37–2.20, p < 0.001, respectively).
3.6 Determinants of the Switching of bDMARDs/JAKi
Multivariate logistic regression analysis using the determinants of age, sex, CCI score, and non-combination with csDMARDs (Table 2B) revealed that ages 61–75 years and >75 years were a significant negative determinant of switching of TNFi (OR 0.85, 95% CI 0.76–0.94, p = 0.002 and OR 0.80, 95% CI 0.69–0.91, p = 0.001, respectively) and CTLA4-Ig (OR 0.77, 95% CI 0.61–0.97, p = 0.028 and OR 0.50, 95% CI 0.39–0.65, p < 0.001, respectively). Female sex (OR 1.13, 95% CI 1.01–1.27, p = 0.039), and CCI score of > 5 (OR 1.50, 95% CI 1.14–1.97, p = 0.003) were significantly positive determinants of switching of TNFi. The prescription without csDMARDs was not significantly associated with switching of any bDMARDs/JAKi.
3.7 Analysis of Temporary Discontinuation of bDMARDs/JAKi
Proportions of patients who experienced temporary discontinuation of TNFi, IL-6i, CTLA4-Ig, and JAKi in combination with csDMARDs were 13.4%, 15.1%, 13.9%, and 6.1%, with temporary discontinuation periods (mean ± SD) of 291.0 ± 371.7, 314.7 ± 311.1, 256.0 ± 267.5, and 256.2 ± 194.1 days, respectively (Table 3A). Without csDMARDs, the proportions of temporary discontinuation of TNFi, IL-6i, CTLA4-Ig, and JAKi were 19.1%, 16.6%, 13.4%, and 10.2%, with temporary discontinuation periods (mean ± SD) of 306.0 ± 341.5, 284.1 ± 256.0, 323.3 ± 352.8, and 274.6 ± 167.3 days, respectively.
Multivariate logistic regression analysis was performed to investigate determinants of temporary discontinuation of bDMARDs/JAKi, examining age, sex, CCI score, and non-combination with csDMARDs (Supplementary Table 3, see ESM). Age > 75 years was a significant negative determinant of temporary discontinuation of TNFi (OR 0.66, 95% CI 0.54–0.80, p < 0.001) and IL-6i (OR 0.64, 95% CI 0.46–0.89, p = 0.009), and age 61–75 years was a significant negative determinant of temporary discontinuation of TNFi (OR 0.72, 95% CI 0.62–0.83, p < 0.001). Female sex was not significantly associated with temporary discontinuation of any bDMARD/JAKi. CCI score of > 5 was a significantly positive determinant of temporary discontinuation of JAK (OR 4.09, 95% CI 1.29–12.99, p = 0.017). Non-combination with csDMARDs was a significantly positive determinant of temporary discontinuation of TNFi (OR 1.57, 95% CI 1.34–1.83, p < 0.001).
3.8 Analysis of discontinuation of bDMARDs/JAKi
Proportions of patients who experienced discontinuation of TNFi, IL-6i, CTLA4-Ig, and JAKi when prescribed in combination with csDMARDs were 20.1%, 19.5%, 19.8%, and 17.0%, and discontinuation periods (mean ± SD) were 973.8 ± 716.0, 810.9 ± 631.7, 732.8 ± 518.9, and 577.8 ± 338.5 days, respectively (Table 3B). Proportions of patients who discontinued TNFi, IL-6i, CTLA4-Ig, and JAKi when prescribed without csDMARDs were 30.7%, 22.2%, 26.6%, and 25.2%, with discontinuation periods (mean ± SD) of 1077.7 ± 732.0, 818.1 ± 612.3, 889.2 ± 584.2, and 593.0 ± 294.3 days, respectively.
Multivariate logistic regression analysis revealed that age of 61–75 years was a significantly positive determinant of discontinuation of TNFi (OR 1.20, 95% CI 1.06–1.36, p = 0.004), and age of >75 years was a significantly positive determinant of discontinuation of TNFi (OR 1.43, 95% CI 1.22–1.67, p < 0.001) and IL-6i (OR 1.79, 95% CI 1.37–2.35) (Supplementary Table 4, see ESM). Female sex was a significantly negative determinant of discontinuation of TNFi (OR 0.79, 95% CI 0.69–0.90, p < 0.001). CCI score of 3–5 was a significantly positive determinant of discontinuation of TNFi (OR 1.16, 95% CI 1.01–1.33, p = 0.030). A prescription without csDMARDs was a significantly positive determinant of discontinuation of TNFi (OR 1.71, 95% CI 1.50–1.95, p < 0.001) and CTLA4-Ig (OR 1.43, 95% CI 1.16–1.75, p < 0.001).
4 Discussion
In the present study, using the MDV claims database, we reported the treatment patterns of bDMARDs and JAKi in clinical practice in Japan from 2008 to 2020, with or without csDMARDs. TNFi were the main first-line therapy, especially in combination with csDMARDs, and were likely to be switched to a different class regardless of combination with csDMARDs. Temporary discontinuation and discontinuation of bDMARDs/JAKi were also observed during the follow-up periods, irrespective of combination with csDMARDs.
Several articles have recently reported treatment patterns of bDMARDs/JAKi in real-world clinical practice in Japan [11, 13, 15]. Takabayashi et al. reported prescription trends and treatment retention of molecular-targeted drugs using the NDB from 2010 to 2016 [15]. A report by Kaneko et al. from the JMDC database demonstrated JAKi/bDMARD treatment patterns from 2016 to 2019 [13]. However, these reports did not fully examine treatment patterns related to the combination of advanced therapies with csDMARDs. In addition, our study using the MDV claims database covered the older RA patient population, which differed from the study by Kaneko et al. of a primarily working-age RA patient population (no enrollees aged ≥ 75 years) [13].
Our study results showed that 3.5% or 4.1% of patients were prescribed JAKi among bDMARD/JAKi with or without csDMARDs, respectively. It was a slightly higher rate than a previous report from NDB where JAKi were prescribed in 0.9% of the patients [11]. This may be caused by the difference in data source between MDV and NDB databases and in the analysis period between our study (2008‒2020) and the previous report from 2017. This might also suggest a recent increase in the use of JAKi. Among the study cohort of RA patients who were prescribed any DMARDs, the proportion of patients who were prescribed each bDMARD/JAKi generally continued to increase over time, after the introduction of each drug in Japan, suggesting that bDMARDs/JAKi play a key role in real-world clinical practice in Japan. Among different types of bDMARDs/JAKi available in clinical practice in Japan, TNFi were the most commonly prescribed drugs between 2008 and 2020. Accumulated evidence in Japanese RA patients, including post-marketing surveillance (PMS), supports increasing TNFi prescription numbers since 2008 [16, 17]. Among the bDMARDs/JAKi, TNFi were most commonly prescribed as first-line bDMARDs. In line with the prescribing information, TNFi were more commonly used in combination with csDMARDs, with more prescription of non-TNFi biologics (IL-6i, CTLA4-Ig, and JAKi) as monotherapy. This finding could be influenced by the guideline recommending an IL-6i and a JAKi to be used in patients who should not receive methotrexate/csDMARD [6, 14]. Regardless of combination with csDMARDs, JAKi were more frequently prescribed as second- or third-line bDMARDs/JAKi compared with first-line prescription. As in a previous report [13], the JAKi prescription numbers have continued to increase. However, as long-term safety of JAKi has not been fully established, the guidelines recommend consideration of JAKi use only after risk assessment, such as cardiovascular events and malignancies, regardless of concomitant use of csDMARDs [6, 14]. This may reflect our findings.
Approximately 30% of patients starting bDMARDs/JAKi therapy regardless of combination with csDMARDs are thought to switch to another bDMARDs or JAKi in real-world clinical practice in Japan. Sankey diagrams indicate varied switching pathways from first-line bDMARDs/JAKi to second- or third-line therapy, either switching to another drug class or within a class. Compared with intra-class switching, different class switching from TNFi was observed with second or third-line prescription, regardless of csDMARD combination. The guidelines suggest switching to non-TNFi biologics rather than another TNFi in RA patients failing TNFi therapy [6, 14]. This may explain our finding. We explored factors contributing to initiation and switching of therapies with bDMARDs/JAKi. Ages 61–75 years and > 75 years were significant negative determinants of first-line prescription of TNFi and IL-6i, and were otherwise a significant positive determinant of CTLA4-Ig use. A previous study using NDB Japan was consistent with our findings, indicating decreased TNFi or IL-6i use and increased CTLA4-Ig use with age [11]. Reasons for the difference in the treatment trend between TNFi/IL-6i and CTLA4-Ig are unknown, but some of the following evidence might indicate that this is a trend in treatment. Japanese studies have reported the risk of serious infection, especially in elderly patients with RA treated with TNFi or IL-6i [18, 19], while the 2015 ACR guidelines suggested the use of CTLA4-Ig in patients with previous serious infections, due to the lower risk of subsequent hospitalized infections among patients prescribed abatacept [2]. Interstitial lung disease (ILD) may occur more frequently among older RA patients, and CTLA4-Ig might have a lower risk of worsening pre-existing ILD [20]. In this study, a higher rate of chronic lung disease was seen in patients prescribed CTLA4-Ig, as compared with other bDMARDs/JAKi. In contrast, data from patients with older-onset RA (mean age 74.9 years) from a prospective registry study conducted in Japan indicate significant benefits from treatment initiated with non-biologic DMARDs, followed by TNFi or IL-6i, in order to achieve structural remission, functional remission, and low disease activity in clinical practice [21]. As suggested in the guideline, bDMARDs/JAKi should be used in elderly RA patients with particular consideration of safety, despite csDMARD therapy [14].
In the present study, temporary discontinuation and discontinuation of bDMARDs/JAKi were observed in patients both in combination with csDMARDs and without csDMARDs. Although reasons for temporary discontinuation and discontinuation were not available in this study, factors such as achievement of remission, adverse events, comorbidities, and treatment cost could be associated with this observation. The latest Japanese treatment guidelines suggest tapering of bDMARD/tsDMARD therapies in RA patients who maintain remission or low disease activity after glucocorticoid tapering [14]. Although, according to this recommendation, csDMARDs should not be stopped, we found some patients who experienced temporary discontinuation and discontinuation of bDMARDs/JAKi during the study period. In this study, multivariate analyses showed age 61–75 years and > 75 years were likely to be associated with discontinuation but not temporary discontinuation. In elderly patients, rheumatologists are required to consider patients’ safety carefully, including the presence of comorbidities such as ILD, infections, cardiovascular events, methotrexate toxicity, and effectiveness of re-treatment following discontinuation. A lower rate of temporary discontinuation was observed with the TNFi infliximab, possibly supported by the likelihood of infusion reactions with infliximab reintroduction [16, 22], a potential barrier to reintroduction. Although discontinuation of bDMARDs/JAKi ultimately leads to a flare, recent reports document discontinuation [23,24,25,26,27,28,29,30]. There may be various reasons for discontinuation; however, our study still provides real-life data on temporary discontinuation or discontinuation of bDMARDs/JAKi by drug. As the guidelines recommend, there needs to be a shared decision between rheumatologists and patients about treatment, taking into consideration disease activity, safety issues, and other patient factors, such as comorbidities and progression of structural damage.
Several limitations should be discussed. First, generalizability of the findings in our study should be interpreted with caution. Our data were generated in large Japanese DPC hospitals, we cannot rule out the possibility of a bias toward patients whose RA was more severe and thus were more likely to be treated with advanced therapies. The results are therefore not necessarily representative of RA treatment in Japan in daily practice. Second, regarding the definition of ‘first-line’ in this study, it is possible that patients in this cohort already started the first-line treatment in another hospital/clinic. Additionally, this MDV database does not allow RA patients that changed hospitals during the treatment to be followed. Third, we could not determine reasons for temporary discontinuation and discontinuation of treatment, potentially due to adverse events, lack of efficacy, or even clinical remission, because claims data analysis in general can utilize only a very limited set of medical information and parameters. Fourth, we explored the determinants of the first-line prescription, switching, temporary discontinuation, and discontinuation, using candidate predictors of age, sex, CCI score, and concomitant csDMARDs use. There may be other candidate predictors such as disease severity and RA duration, however, we could not evaluate these candidates because of the limitation of the study database. In addition, methotrexate was included in csDMARDs, because we focused on treatment pattern of bDMARDs/JAKi in this study. In the guideline, positioning of methotrexate differs from that of other csDMARDs and it is possibility that the difference between concomitant use of methotrexate and other csDMARDs could impact on the prescription of bDMARDs/JAKi. Fifth, determinants for prescription and the mean days of temporary discontinuation or discontinuation can be affected by available treatment options, prescription number, and follow-up period. Finally, sample sizes were heterogeneously distributed in each drug subclass, and were especially small in the JAKi group because of its recent launch in Japan. We anticipate future analysis of more RA patient data in real-world clinical practice in Japan, especially those including more patients treated with JAKi.
5 Conclusions
The real-world treatment trajectories of advanced therapies, with and without csDMARDs, were analyzed in Japanese RA patients using a claims-based database between 2008 and 2020. TNFi were the main first-line therapy, and likely to be switched to different classes. Understanding the current treatment patterns, including discontinuation, is important to find an optimal treatment strategy for RA patients.
References
Smolen JS, Aletaha D, McInnes IB. Rheumatoid arthritis. Lancet. 2016;388:2023–38. https://doi.org/10.1016/S0140-6736(16)30173-8.
Singh JA, Saag KG, Bridges SL Jr, Akl EA, Bannuru RR, Sullivan MC, et al. 2015 American College of Rheumatology guideline for the treatment of rheumatoid arthritis. Arthritis Rheumatol. 2016;68:1–26. https://doi.org/10.1002/art.39480.
Almutairi K, Nossent J, Preen D, Keen H, Inderjeeth C. The global prevalence of rheumatoid arthritis: a meta-analysis based on a systematic review. Rheumatol Int. 2021;41:863–77. https://doi.org/10.1007/s00296-020-04731-0.
Smolen JS. Insights into the treatment of rheumatoid arthritis: a paradigm in medicine. J Autoimmun. 2020;110: 102425. https://doi.org/10.1016/j.jaut.2020.102425.
McInnes IB, Schett G. Pathogenetic insights from the treatment of rheumatoid arthritis. Lancet. 2017;389:2328–37. https://doi.org/10.1016/S0140-6736(17)31472-1.
Smolen JS, Landewé RBM, Bergstra SA, Kerschbaumer A, Sepriano A, Aletaha D, et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2022 update. Ann Rheum Dis. 2023;82:3–18. https://doi.org/10.1136/ard-2022-223356.
Tanaka Y. Recent progress in treatments of rheumatoid arthritis: an overview of developments in biologics and small molecules, and remaining unmet needs. Rheumatology (Oxford). 2021;60:vi12–20. https://doi.org/10.1093/rheumatology/keab609.
Mitsubishi Tanabe Pharma Corporation (2022) REMICADE® for I.V. infusion interview form https://medical.mt-pharma.co.jp/di/file/if/f_rec.pdf. Accessed 31 Aug 2023 (in Japanese)
Chugai Pharmaceutical Co., Ltd. (2023) ACTEMRA® for intravenous infusion interview form https://chugai-pharm.jp/content/dam/chugai/product/act/div/if/doc/act_if.pdf. Accessed 31 Aug 2023 (in Japanese)
Bristol-Myers Squibb Company and Ono Pharmaceutical Co., Ltd. (2022) ORENCIA® for I.V. infusion interview form https://file.bmshealthcare.jp/bmshealthcare/pdf/interview/ORENCIAIV_InterviewForm.pdf. Accessed 31 Aug 2023 (in Japanese)
Nakajima A, Sakai R, Inoue E, Harigai M. Prevalence of patients with rheumatoid arthritis and age-stratified trends in clinical characteristics and treatment, based on the National Database of Health Insurance Claims and Specific Health Checkups of Japan. Int J Rheum Dis. 2020;23:1676–84. https://doi.org/10.1111/1756-185X.13974.
Pfizer Japan Inc. (2023) XELJANZ® tablets interview form https://www.pfizermedicalinformation.jp/ja-jp/system/files/content_files/xel01if_3.pdf. Accessed 31 Aug 2023 (in Japanese)
Kaneko Y, Sakurai M, Snijder R, Kokubo S, Kato D. A retrospective, longitudinal study of rheumatoid arthritis treatment patterns with Janus kinase inhibitors and other disease-modifying antirheumatic drugs in Japan. Mod Rheumatol. 2023;33:448–59. https://doi.org/10.1093/mr/roac046.
Kawahito Y, Morinobu A, Kaneko Y, Kohno M, Hirata S, Kishimoto M, et al. Drug treatment algorithm and recommendations from the 2020 update of the Japan College of Rheumatology clinical practice guidelines for the management of rheumatoid arthritis-secondary publication. Mod Rheumatol. 2023;33:21–35. https://doi.org/10.1093/mr/roac017.
Takabayashi K, Ando F, Ikeda K, Fujita S, Nakajima H, Hanaoka H, et al. Trend in prescription and treatment retention of molecular-targeted drugs in 121,131 Japanese patients with rheumatoid arthritis: A population-based real-world study. Mod Rheumatol. 2022;32:857–65. https://doi.org/10.1093/mr/roab126.
Takeuchi T, Tatsuki Y, Nogami Y, Ishiguro N, Tanaka Y, Yamanaka H, et al. Postmarketing surveillance of the safety profile of infliximab in 5000 Japanese patients with rheumatoid arthritis. Ann Rheum Dis. 2008;67:189–94. https://doi.org/10.1136/ard.2007.072967.
Koike T, Harigai M, Inokuma S, Inoue K, Ishiguro N, Ryu J, et al. Postmarketing surveillance of the safety and effectiveness of etanercept in Japan. J Rheumatol. 2009;36:898–906. https://doi.org/10.3899/jrheum.080791.
Komano Y, Tanaka M, Nanki T, Koike R, Sakai R, Kameda H, et al. Incidence and risk factors for serious infection in patients with rheumatoid arthritis treated with tumor necrosis factor inhibitors: a report from the Registry of Japanese Rheumatoid Arthritis Patients for Longterm Safety. J Rheumatol. 2011;38:1258–64. https://doi.org/10.3899/jrheum.101009.
Koike T, Harigai M, Inokuma S, Ishiguro N, Ryu J, Takeuchi T, et al. Postmarketing surveillance of tocilizumab for rheumatoid arthritis in Japan: interim analysis of 3881 patients. Ann Rheum Dis. 2011;70:2148–51. https://doi.org/10.1136/ard.2011.151092.
Nakashita T, Ando K, Takahashi K, Motojima S. Possible effect of abatacept on the progression of interstitial lung disease in rheumatoid arthritis patients. Respir Investig. 2016;54:376–9. https://doi.org/10.1016/j.resinv.2016.03.001.
Sugihara T, Ishizaki T, Hosoya T, Iga S, Yokoyama W, Hirano F, et al. Structural and functional outcomes of a therapeutic strategy targeting low disease activity in patients with elderly-onset rheumatoid arthritis: a prospective cohort study (CRANE). Rheumatology (Oxford). 2015;54:798–807. https://doi.org/10.1093/rheumatology/keu395.
Mitsubishi Tanabe Pharma Corporation (2022) REMICADE® for I.V. Infusion Package Insert https://medical.mt-pharma.co.jp/di/file/dc/rec.pdf. Accessed 31 Aug 2023 (in Japanese)
Yamanaka H, Nagaoka S, Lee SK, Bae SC, Kasama T, Kobayashi H, et al. Discontinuation of etanercept after achievement of sustained remission in patients with rheumatoid arthritis who initially had moderate disease activity-results from the ENCOURAGE study, a prospective, international, multicenter randomized study. Mod Rheumatol. 2016;26:651–61. https://doi.org/10.3109/14397595.2015.1123349.
Ghiti Moghadam M, Vonkeman HE, Ten Klooster PM, Tekstra J, van Schaardenburg D, Starmans-Kool M, et al. Stopping tumor necrosis factor inhibitor treatment in patients with established rheumatoid arthritis in remission or with stable low disease activity: a pragmatic multicenter, open-label randomized controlled trial. Arthritis Rheumatol. 2016;68:1810–7. https://doi.org/10.1002/art.39626.
Pavelka K, Akkoç N, Al-Maini M, Zerbini CAF, Karateev DE, Nasonov EL, Rahman MU, et al. Maintenance of remission with combination etanercept-DMARD therapy versus DMARDs alone in active rheumatoid arthritis: results of an international treat-to-target study conducted in regions with limited biologic access. Rheumatol Int. 2017;37:1469–79. https://doi.org/10.1007/s00296-017-3749-7.
Smolen JS, Emery P, Fleischmann R, van Vollenhoven RF, Pavelka K, Durez P, et al. Adjustment of therapy in rheumatoid arthritis on the basis of achievement of stable low disease activity with adalimumab plus methotrexate or methotrexate alone: the randomised controlled OPTIMA trial. Lancet. 2014;383:321–32. https://doi.org/10.1016/S0140-6736(13)61751-1.
Aguilar-Lozano L, Castillo-Ortiz JD, Vargas-Serafin C, Morales-Torres J, Sanchez-Ortiz A, Sandoval-Castro C, et al. Sustained clinical remission and rate of relapse after tocilizumab withdrawal in patients with rheumatoid arthritis. J Rheumatol. 2013;40:1069–73. https://doi.org/10.3899/jrheum.121427.
Kaneko Y, Kato M, Tanaka Y, Inoo M, Kobayashi-Haraoka H, Amano K, et al. Tocilizumab discontinuation after attaining remission in patients with rheumatoid arthritis who were treated with tocilizumab alone or in combination with methotrexate: results from a prospective randomised controlled study (the second year of the SURPRISE study). Ann Rheum Dis. 2018;77:1268–75. https://doi.org/10.1136/annrheumdis-2018-213416.
Takeuchi T, Matsubara T, Ohta S, Mukai M, Amano K, Tohma S, et al. Biologic-free remission of established rheumatoid arthritis after discontinuation of abatacept: a prospective, multicentre, observational study in Japan. Rheumatology (Oxford). 2015;54:683–91. https://doi.org/10.1093/rheumatology/keu338.
Mori S, Ueki Y. Outcomes of dose reduction, withdrawal, and restart of tofacitinib in patients with rheumatoid arthritis: a prospective observational study. Clin Rheumatol. 2019;38:3391–400. https://doi.org/10.1007/s10067-019-0472.
Acknowledgements
Medical writing support was provided by Yoshiaki Kita, PhD of Medical Professional Relations Inc., and was funded by Janssen Pharmaceutical K.K. The authors thank Seiji Yokoyama for supporting the study design and Shigefumi Tsukamoto (EPS Corp.) for performing the statistical analysis. Statistical analysis support was funded by Janssen Pharmaceutical K.K.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Janssen Pharmaceutical K.K. funded this study and participated in the study design, research, analysis, data collection, interpretation of data, review, and approval of the publication.
Competing interests
Masahiko Miyashiro and Hirohito Shimizu are employees of Janssen Pharma K.K., a wholly owned subsidiary of Johnson & Johnson. Teita Asano, Celine Miyazaki, Yutaka Ishii, and Junya Masuda are employees of Janssen Pharmaceutical K.K., a wholly owned subsidiary of Johnson & Johnson, and may hold stock and/or stock options in the company.
Ethics approval
The manuscript does not contain clinical studies or identifiable patient data.
Consent to participate
For this type of retrospective study, formal consent is not required.
Consent for publication
Not applicable.
Data availability
The datasets analyzed in this study are not publicly available because they were obtained from a commercial company, Medical Data Vision Co., Ltd., and used under license. However, data may be available from the corresponding author on reasonable request and with permission from Medical Data Vision Co., Ltd.
Code availability
Not applicable.
Author contributions
All authors met the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, participated in the monitoring of data quality, interpretation of study results, and in the drafting, critical revision, and approval of the final version of the manuscript. All authors had access to the study data, made the final decision about where to publish these data, and approved submission to this journal. All authors read and approved the final manuscript. The details are as follows: Conceptualization: all authors. Methodology: all authors. Formal analysis: Masahiko Miyashiro, Teita Asano, Yutaka Ishii, Hirohito Shimizu, Junya Masuda. Interpretation of data and writing – original draft: all authors. Funding acquisition and project administration: Yutaka Ishii, Celine Miyazaki, Junya Masuda.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Miyashiro, M., Asano, T., Ishii, Y. et al. Treatment Patterns of Biologic Disease-Modifying Antirheumatic Drugs and Janus Kinase Inhibitors in Patients with Rheumatoid Arthritis in Japan: A Claims-Based Cohort Study. Drugs - Real World Outcomes 11, 285–297 (2024). https://doi.org/10.1007/s40801-024-00423-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40801-024-00423-4