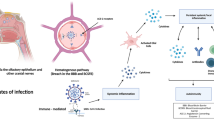
Abstract
While COVID-19 is no longer the “hot-topic” it was 2 years ago, its prevalence and impact are still significant. In 2022, the CDC estimated that over 90% of children from 6 months to 17 years old have had COVID-19 infection. While most children have limited and mild symptoms, a substantial subset experiences significant neurological manifestations and/or complications which may lead to long-lasting morbidity or even mortality. Such neurological manifestations of SARS-CoV-2 include acute encephalitis, seizures, central demyelinating disease, cerebrovascular events, peripheral neurological disorders, and chronic symptoms in the setting of long COVID, which may affect up to 25% of infected children and adolescents. Given the high prevalence of COVID-19 in the general and pediatric population, it is essential for clinicians to understand the full breadth of its potential effects. In this article, we review common neurological manifestations and sequelae of SARS-CoV-2 in the pediatric population and describe their prevalence, timing, and associated neuroimaging findings.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
We have long known that numerous viruses have the potential to cause various neuropathology in both children and adults. Neurological diseases such as encephalitis, meningitis, acute cerebellar ataxia, vasculopathy, and acute flaccid myelitis have all been directly related to viral etiologies (Costa & Sato, 2020; Grose et al., 2023; McGill et al., 2017; Murphy et al., 2021; Parvez & Ohtsuki, 2022). Thus, it is not surprising that SARS-CoV-2 infection also has the potential to affect the nervous system. What has been surprising, and challenging, is some of the unique and rarer neurological manifestations of this virus. Furthermore, given the relative novelty and prevalence of SARS-CoV-2, it is imperative that clinicians across disciplines are familiar with its neurological manifestations and that we continue to study potential long-term neurological sequelae in those with history of SARS-CoV-2 infection. In this article, we will review SARS-CoV2 in children and adolescents with particular attention to its neurological and neuroimaging manifestations and implications. Table 1 summarizes the studies that were included in this review. It should be noted we do not address the neuropsychological impacts of SARS-CoV-2 infection in this review, as this topic is covered in depth in article by Koturba et al., in this issue.
Background
SARS-CoV-2 is a highly infectious novel coronavirus that was first identified in December 2019 in Wuhan, China. The virus is responsible for the COVID-19 pandemic, which has since spread globally. As of July 2023, there have been over 750 million confirmed cases and almost 7 million deaths worldwide, though this number is likely a gross underestimate as most cases go unreported (Global COVID-19 Tracker | KFF, n.d.). As of late 2022, the CDC estimates that 91.9% of children 6 months to 17 years of age have had a SARS-CoV-2 infection (CDC, 2020). SARS-CoV-2 can change over time due to genomic mutations leading to different viral variants. Because of this, individuals, including children, may be susceptible to repeated infections, though preexisting immunity from prior infection, immunization, or both appears to be protective against most severe disease, hospitalization, and death.
The virus is primarily spread through person-to-person transmission by the respiratory route most often after close (within 6 feet) contact. Children of all ages can transmit the virus, and transmission can occur from both symptomatic and asymptomatic individuals. Respiratory droplets released when an infected person talks, coughs, or sneezes can infect another through inhalation or by droplets landing on exposed mucus membranes. The virus can also spread if one touches a surface contaminated with the virus and then transfers the virus to the mouth, nose, or eyes, though contaminated surfaces are not thought to play a major role in transmission. Longer distance airborne transmission is also possible but thought to play a much smaller role. The contagious period lasts from 1 to 2 days before symptom onset and is highest earlier in infection with infectiousness declining with time; most are no longer contagious after 10 days.
While older adults and those with underlying health conditions are at a higher risk of severe illness and death, children may also develop severe disease. Children with underlying medical conditions and those under 1 year of age seem to be at highest risk though severe disease is not exclusive to these groups. Children most at risk for severe disease include those with underlying neurologic conditions as well as those with chronic pulmonary and cardiovascular disease, obesity, diabetes, and those with other medical complexities (Woodruff et al., 2022). Fortunately, childhood death from COVID-19 is relatively rare with recent estimates of the crude death rate at 1 per 100,000 for children 0–19 years of age in the U.S (Flaxman et al., 2023).
Symptoms of SARS-CoV-2 infection in children range from being asymptomatic to causing severe disease, hospitalization, and death, but most infections in children are asymptomatic (66%) or mild (27%) (Forrest et al., 2022). Milder symptoms, when present, can be similar to those seen in adults and include loss of smell and/or taste, fever, headache, cough, rhinorrhea, sore throat, shortness of breath, body aches, and gastrointestinal symptoms including abdominal pain, nausea, vomiting, and diarrhea. Severe symptoms may include acute kidney injury, cardiovascular abnormalities, and respiratory compromise with the need for mechanical ventilation and vasopressor support though this occurs much less commonly in children than in adults. Neurological symptoms and complications may also occur, which will be reviewed in detail below. Symptoms appear to vary with the age of the child and the SARS-CoV-2 variant causing the infection.
SARS-CoV-2 infection in children has also been associated with some delayed complications including multisystem inflammatory syndrome in children (MIS-C) and post-COVID condition (PCC). MIS-C is a rare but serious condition that occurs in < 1% of children and adolescents approximately 4–6 weeks after they have been infected with SARS-CoV-2 (Payne et al., 2021; Zhu & Ang, 2022). It most often occurs in school age children over 4 years of age and is felt to be due to an abnormal immune response to the virus. Symptoms can occasionally be confused with other inflammatory diseases in children including Kawasaki disease and macrophage activation syndrome as well as with severe COVID-19. The symptoms of MIS-C can vary but typically include a persistent fever, abdominal pain, vomiting, diarrhea, rash, conjunctivitis, mucous membrane changes (red swollen lips), swollen hands and feet, and cervical lymphadenopathy. Cardiac involvement is a key finding and can include depressed cardiac function as well as coronary artery abnormalities. Neurologic involvement may also be present in children with MIS-C with symptoms including headache, lethargy, and confusion. In one US study of children < 21 years hospitalized with MIS-C between March and December 2020, 20% of 616 children with MIS-C had neurologic involvement and 20 patients had life-threatening symptoms including severe encephalopathy, central nervous system demyelination, stroke, cerebral edema, and Guillain–Barre syndrome (LaRovere et al., 2021).
PCC is a term used to describe all of the longer term health consequences that may result ≥ 4 weeks from a SARS-CoV-2 infection including what is known as “long COVID.” The WHO definition of PCC requires symptoms lasting for at least 2 months which may include fatigue, shortness of breath, cognitive dysfunction, and symptoms must have an impact on everyday functioning (A Clinical Case Definition of Post COVID-19 Condition by a Delphi Consensus, 6 October 2021, n.d.). Other symptoms may include headache, sleep disturbance, dizziness, and dysautonomia. Data is still unclear as to how many children are affected by long COVID as it can be difficult to distinguish symptoms related to a prior SARS-CoV-2 infection from symptoms that may have existed prior to infection that may be due to other factors including the stressors of the pandemic itself. The presence of PCC does seem to be associated with history of more severe COVID-19 disease (Maddux et al., 2022).
Neurological Manifestations of SARS-CoV-2
One of the most concerning aspects of COVID-19 is its potential to cause acute neurologic symptoms. In one study of over 15,000 children 2 months to 18 years of age hospitalized with COVID-19, 7% had a neurologic complication. The most frequent of these included febrile seizures (3.9%), nonfebrile seizures (2.3%), and encephalopathy (2.2%) (Antoon et al., 2022a).
Both central and peripheral nervous system manifestations of SARS-CoV-2 have been reported in the literature. Likewise, neuroinvasion may occur by various mechanisms. These include transmucosal entry via regional nervous structures, such as the olfactory tract, as well as viral binding to angiotensin-converting enzyme (ACE) 2 receptors which are abundant in the adult and fetal brain (Bourgonje et al., 2020; Meinhardt et al., 2021; Schober et al., 2021). Heterogenous mechanisms of neuroinvasion and neurotropism, as well as patients’ individual history and risk factors, may explain the varied clinical neurological manifestations associated with this virus.
Seizures, one of the most common neuropathologies in the pediatric age group at large, are also prevalent in children with SARS-CoV-2 infection. In a cross-sectional multicenter study of 52 children’s hospitals from March 2020 to March 2022, febrile seizures (3.8%) and nonfebrile seizures (2.3%) were the most frequent neurologic complications for children with COVID-19 infection (Antoon et al., 2022b). Frequency of seizures was approximately 5 times greater in children than that in adults with COVID-19 infection (Cho et al., 2022). Certain SARS-CoV-2 variants may also be associated with a greater risk of seizures. In a single-center Japanese study, there was a higher incidence of seizures in febrile children in the Omicron era than in pre-Omicron era (Iijima et al., 2022). The Omicron variant also was found to be associated with more complex provoked seizures and febrile status epilepticus (Thongsing et al., 2022). While patients with SARS-CoV-2 infection may have febrile seizures, there does not appear to be an increased risk of febrile seizures with COVID-19 infection compared to other infectious causes (Cadet et al., 2022; Hanlon et al., 2023).
Acute encephalitis has also been associated with COVID-19, including with active infection, during the post-infectious period, and in setting of MIS-C. Symptoms of this condition may range from headache, fever, confusion, and behavioral changes, to neuropsychiatric symptoms such as visual hallucinations, ritualistic behaviors, akinetic mutism, and seizures (Gaughan et al., 2021; Olivotto et al., 2021; Urso et al., 2022). Electroencephalogram often shows abnormal background slowing. Methylprednisolone is the mainstay of treatment; however, its use is extrapolated from other etiologies of encephalitis and presumed pathophysiology rather than from direct evidence in COVID-19.
Cases of neuroinflammatory and demyelinating disease have been reported both with active COVID-19 infection and as post-infectious sequelae. One of the first reported pediatric cases of acute COVID-19 infection-related transverse myelitis occurred in an 11-year-old previously healthy female presenting with acute lower limb paresis and urinary and fecal retention (Nejad Biglari et al., 2021). Spinal cord magnetic resonance imaging (MRI) showed high signal intensity and swelling at multiple thoracic segments on T2-weighted sequences, with infectious and inflammatory workup mostly unrevealing except for a positive nasopharyngeal COVID-19 PCR test. Longitudinally extensive transverse myelitis involving the medulla, cervical, and thoracic spine has also been reported with SARS-CoV-2 infection (Kaur et al., 2020). Symptoms occurred 3 weeks after exposure and the patient presented with extremity weakness progressing to flaccid quadriparesis and neurogenic respiratory failure requiring intubation within 12 h from onset. Other cases have reported transverse myelitis with SARS-Cov-2 antibody detected in the CSF and serum with negative nasopharyngeal PCR (Akçay et al., 2023; Poyrazoğlu et al., 2022). Treatment has involved the use of IVIG and high-dose steroids, as well as therapeutic plasma exchange. Similarly, acute disseminated encephalomyelitis (ADEM), a disorder characterized by widespread inflammation and demyelination in the brain and spinal cord, has also been documented after SARS-CoV-2 infection (de Miranda Henriques-Souza et al., 2021; Manzo et al., 2021). ADEM manifestations in setting of SARS-CoV-2 may include vision changes, incoordination, weakness, decreased alertness, or seizures. MRI typically shows areas of diffusion restriction in the subcortical and deep white matter and T2/FLAIR hyperintense lesions in the corpus callosum, brainstem, and spinal cord (Tenembaum et al., 2002).
Guillain–Barre syndrome (GBS), a post-infectious immune-mediated demyelinating polyneuropathy affecting the peripheral nervous system, was reported early in the pandemic (Khalifa et al., 2020). An 11-year-old boy presented with acute onset of unsteady gait, inability to walk or climb stairs, and paresthesias of the lower extremities for 1 day. He had an upper respiratory infection and low-grade fever approximately 22 days prior to onset of gait instability. Spinal cord imaging showed cauda equina nerve root enhancement, with albuminocytologic dissociation on CSF studies, and nasopharyngeal swab PCR positive for SARS-CoV-2. Similarly, a 14-year-old girl presented with a history of progressive ascending quadriparesthesia and mild lower limb weakness. Her father had presented to the same hospital days prior, also for ascending paresthesia and bilateral facial droop, and was also diagnosed with GBS. Labs revealed albuminocytologic cytologic dissociation with throat swab PCR positive for COVID-19 in both in the father and the daughter (Paybast et al., 2020). These patients were treated with intravenous immunoglobulin.
Isolated or mixed cranial nerve pathologies have also been described with COVID-19 infection in the pediatric population. In a descriptive study of 280 Spanish patients from 5 to 18 years of age, 45 patients (16.1%) had anosmia and/or hypogeusia as a symptom of SARS-CoV-2 infection (Púa Torrejón et al., 2022). The incidence of anosmia/hypogeusia in children with SARS-CoV-2 was lower than reported in adults, although with a longer duration with a mean symptom duration of 36.4 days and 27.6 days, for anosmia and hypogeusia, respectively. In addition, facial nerve palsy has also been an associated finding, reported in a 15-month-old girl who presented with right peripheral facial nerve palsy without any other neurological signs (Decio et al., 2021). In this case, serological tests for common infectious etiologies were negative, as well as SARS-CoV-2 nasopharyngeal PCR testing; however, serological testing yielded positive SARS-CoV-2 IgG antibodies. Treatment with prednisone achieved clinical resolution.
Cerebrovascular events have also been associated with COVID-19 infection. In a Peruvian study of pediatric intensive care patients, cerebrovascular events occurred in 6 patients: 5 with hemorrhagic stroke and 1 with ischemic stroke (Coronado Munoz et al., 2022). Cerebral venous sinus thrombosis and acute ischemic strokes have also been reported for patients with MIS-C as well (Trapani et al., 2022; Whitworth et al., 2021). Reversible cerebral vasoconstriction syndrome (RCVS), a condition of vascular spasms in the brain, has also been reported in patients with MIS-C (Sadeghizadeh et al., 2022). These patients presented with severe thunderclap headaches during their treatment course and cerebrovascular imaging was consistent with RCVS, demonstrating multifocal areas of vasoconstriction in multiple vascular territories with resolution on follow-up imaging. Focal cerebral arteriopathy has also been reported with COVID-19 infection. One such patient had cerebrovascular imaging that showed unilateral focal irregular narrowing and banding of the proximal left middle cerebral artery with subsequent left basal ganglia and insula infarction (Mirzaee et al., 2020). Possible mechanisms of stroke with COVID-19 infection include hypercoagulability caused by an increase in inflammatory and procoagulant markers that lead to thromboses and endothelial dysfunction resulting from the virus binding to ACE2 receptors on endothelial cells that then triggers an inflammatory response and upregulation of the coagulation cascade leading to formation of thrombin and fibrin clots, increasing the risk of thromboembolism (Hess et al., 2020; Stamm et al., 2022; Varga et al., 2020).
As with adults, long COVID-19/PCC has also affected the pediatric population. In an Italian study of 689 COVID-19-positive children, long COVID-19 symptoms were found in 188 children (24.3% primary care patients, 58.3% hospitalized patients), with most frequently reported symptoms including abnormal fatigue, neurological and respiratory disorders, as well as muscle/joint pain, gastrointestinal symptoms, and psychological symptoms (Trapani et al., 2022). According to a 2022 meta-analysis, the prevalence of long COVID in the pediatric population was 25% with the most common clinical manifestations being mood symptoms, fatigue, and sleep disorders (Lopez-Leon et al., 2022).
Unique Neuroimaging Findings Associated with SARS-CoV-2 in the Pediatric Population
Like the clinical neurological manifestations of COVID-19 infection, neuroimaging abnormalities associated with COVID-19 are also variable. The neuroimaging manifestations of COVID-19 infection in previously healthy children, often with no comorbidities, can range from mild to severe. Since clinicoradiological peripheral and central nervous system syndromes in children with COVID-19 are predominantly of neuroinflammatory origin, neuroimaging findings most often resemble immune-mediated para-infectious patterns of disease involving the brain, spine, and nerves. The common findings include multifocal T2 bright lesions in brain white matter (ADEM)-like), vasculitic patterns with ischemia, enhancing neuritis or polyradiculitis, venous thrombosis, splenial lesions of the corpus callosum, longitudinally extensive myelitis, and myositis (Lindan et al., 2021). Cerebrovascular complications appear to be less common in children than in adults. Lindan et al. observed that cranial nerve enhancement did not always correlate with cranial nerve deficits. COVID-19 antibody-positive acquired demyelinating syndromes (MOG-IgG-associated disorders (MOG-AD), neuromyelitis optica spectrum disorder (NMOSD), and autoimmune encephalitis (anti-N-methyl-d-aspartate receptor (NMDA), MOG) have presentations similar to the established clinical phenotypes with these antibodies. Optic neuritis and multifocal ADEM-like lesions represent the most common imaging patterns related to anti-AQP4 and anti-MOG antibodies respectively (Aubart et al., 2022).
Pediatric COVID-19-related neuroinflammatory disease can also occur in the setting of MIS-C, but it is less common than other system involvement (12–20%), and MRI abnormalities are infrequent. However, when present, central splenial T2-hyperintensity with restricted diffusion has been described (11–64%) (Abdel-Mannan et al., 2020; Aubart et al., 2022; LaRovere et al., 2021; Lindan et al., 2021; Sa et al., 2021). Signal changes may extend to involve the rest of the corpus callosum and adjacent white matter with interval complete resolution. In most cohorts of MIS-C, this is an isolated finding occurring independently of seizures, except in a single study where co-existent ADEM-like lesions were present in a similar prevalence (Lindan et al., 2021). A low percentage of vascular phenomena has also been reported in MIS-C- including infarcts, parenchymal hemorrhage, cerebral venous thrombosis, and microhemorrhages from thrombotic microangiopathy (LaRovere et al., 2021; Lindan et al., 2021; Sa et al., 2021).
Conclusions
The neurological manifestations of SARS-CoV-2 are heterogeneous in timing, severity, and clinical presentation. While our present understanding of these manifestations is limited given much of the literature on specific neurological complications consists of case reports or small case series, based on the evidence to date, the neurological complications and associated imaging findings seen in SARS-CoV-2 are largely similar to other post-infectious neurological conditions. That being said, the wide array of central, peripheral, and combined neurological disorders associated with SARS-CoV-2, as a single virus, is somewhat unique. In addition, given the relative recency of this disease, its long-term consequences have yet to be clearly defined. This is particularly relevant for pediatric patients with SARS-CoV-2 who have many years to develop and be impacted by delayed complications. Thus, it is important for clinicians to be familiar with the various neurological signs, symptoms, and sequelae seen in children and adolescents with COVID-19 infection and consider their downstream neuropsychological impact, as discussed in more detail in the Koturba et al. article in this issue. Further research needs to explore the long-term impact of COVID-19-related neurological complications in children and adolescents.
References
A clinical case definition of post COVID-19 condition by a Delphi consensus, 6 October 2021. (n.d.). Retrieved May 24, 2023, from https://www.who.int/publications-detail-redirect/WHO-2019-nCoV-Post_COVID-19_condition-Clinical_case_definition-2021.1
Abdel-Mannan, O., Eyre, M., Löbel, U., Bamford, A., Eltze, C., Hameed, B., Hemingway, C., & Hacohen, Y. (2020). Neurologic and radiographic findings associated with COVID-19 Infection in Children. JAMA Neurology, 77(11), 1440–1445. https://doi.org/10.1001/jamaneurol.2020.2687
Akçay, N., Menentoğlu, M. E., Oğur, M., Tosun, D., Palabıyık, F. B., & Şevketoğlu, E. (2023). COVID-19-associated transverse myelitis treated by therapeutic plasma exchange: A case report. Journal of Clinical Apheresis, 38(1), 65–68. https://doi.org/10.1002/jca.22024
Antoon, J. W., Hall, M., Howard, L. M., Herndon, A., Freundlich, K. L., Grijalva, C. G., & Williams, D. J. (2022a). COVID-19 and acute neurologic complications in children. Pediatrics, 150(5), 5. https://doi.org/10.1542/peds.2022-058167
Antoon, J. W., Hall, M., Howard, L. M., Herndon, A., Freundlich, K. L., Grijalva, C. G., & Williams, D. J. (2022b). COVID-19 and acute neurologic complications in children. Pediatrics, 150(5), e2022058167. https://doi.org/10.1542/peds.2022-058167
Aubart, M., Roux, C.-J., Durrleman, C., Gins, C., Hully, M., Kossorotoff, M., Gitiaux, C., Levy, R., Moulin, F., Debray, A., Belhadjer, Z., Georget, E., Kom, T., Blanc, P., Wehbi, S., Mazeghrane, M., Tencer, J., Gajdos, V., Rouget, S., & Desguerre, I. (2022). Neuroinflammatory disease following severe acute respiratory syndrome coronavirus 2 infection in children. The Journal of Pediatrics, 247, 22-28.e2. https://doi.org/10.1016/j.jpeds.2022.05.018
Bourgonje, A. R., Abdulle, A. E., Timens, W., Hillebrands, J., Navis, G. J., Gordijn, S. J., Bolling, M. C., Dijkstra, G., Voors, A. A., Osterhaus, A. D., van der Voort, P. H., Mulder, D. J., & van Goor, H. (2020). Angiotensin-converting enzyme 2 (ACE2), SARS-CoV-2 and the pathophysiology of coronavirus disease 2019 (COVID-19). The Journal of Pathology, 251(3), 3. https://doi.org/10.1002/path.5471
Cadet, K., Boegner, J., Ceneviva, G. D., Thomas, N. J., & Krawiec, C. (2022). Evaluation of febrile seizure diagnoses associated with COVID-19. Journal of Child Neurology, 37(5), 410–415. https://doi.org/10.1177/08830738221086863
CDC. (2020, March 28). COVID Data Tracker. Centers for Disease Control and Prevention. https://covid.cdc.gov/covid-data-tracker. Accessed 16 Feb 2024.
Cho, S.-M., White, N., Premraj, L., Battaglini, D., Fanning, J., Suen, J., Bassi, G. L., Fraser, J., Robba, C., Griffee, M., Singh, B., Citarella, B. W., Merson, L., Solomon, T., & Thomson, D. (2022). Neurological manifestations of COVID-19 in adults and children. Brain, 146(4), 1648–1661. https://doi.org/10.1093/brain/awac332
Coronado Munoz, A., Tasayco, J., Morales, W., Moreno, L., Zorrilla, D., Stapleton, A., Pajuelo, P., Reyes, G., Estupiñan, M., Seminario, R., Ortiz, M., & Domínguez, J. (2022). High incidence of stroke and mortality in pediatric critical care patients with COVID-19 in Peru. Pediatric Research, 91(7), 1730–1734. https://doi.org/10.1038/s41390-021-01547-x
da Costa, B. K., & Sato, D. K. (2020). Viral encephalitis: A practical review on diagnostic approach and treatment. Jornal De Pediatria, 96(Suppl 1), 12–19. https://doi.org/10.1016/j.jped.2019.07.006
de Miranda Henriques-Souza, A. M., de Melo, A. C. M. G., de Aguiar Coelho Silva Madeiro, B., Freitas, L. F., Sampaio Rocha-Filho, P. A., & Gonçalves, F. G. (2021). Acute disseminated encephalomyelitis in a COVID-19 pediatric patient. Neuroradiology, 63(1), 141–145. https://doi.org/10.1007/s00234-020-02571-0
Decio, A., Mazza, A., Quadri, V., Ronconi, M. S., Brusadelli, C., Ruggeri, M., & D’Antiga, L. (2021). Neurological manifestations of COVID-19 in Children: A Case of Facial Nerve Palsy. Pediatric Neurology, 116, 59. https://doi.org/10.1016/j.pediatrneurol.2020.12.006
Flaxman, S., Whittaker, C., Semenova, E., Rashid, T., Parks, R. M., Blenkinsop, A., Unwin, H. J. T., Mishra, S., Bhatt, S., Gurdasani, D., & Ratmann, O. (2023). Assessment of COVID-19 as the underlying cause of death among children and young people aged 0 to 19 years in the US. JAMA Network Open, 6(1), e2253590. https://doi.org/10.1001/jamanetworkopen.2022.53590
Forrest, C. B., Burrows, E. K., Mejias, A., Razzaghi, H., Christakis, D., Jhaveri, R., Lee, G. M., Pajor, N. M., Rao, S., Thacker, D., & Bailey, L. C. (2022). Severity of acute COVID-19 in children <18 years old March 2020 to December 2021. Pediatrics, 149(4), e2021055765. https://doi.org/10.1542/peds.2021-055765
Gaughan, M., Connolly, S., O’Riordan, S., Tubridy, N., McGuigan, C., & Kinsella, J. A. (2021). Pediatric parainfectious encephalitis associated with COVID-19. Neurology, 96(11), 541–544. https://doi.org/10.1212/WNL.0000000000011476
Global COVID-19 Tracker | KFF. (n.d.). Retrieved May 24, 2023, from https://www.kff.org/coronavirus-covid-19/issue-brief/global-covid-19-tracker/
Grose, C., Shaban, A., & Fullerton, H. J. (2023). Common features between stroke following varicella in children and stroke following herpes zoster in adults: Varicella-zoster virus in trigeminal ganglion. Current Topics in Microbiology and Immunology, 438, 247–272. https://doi.org/10.1007/82_2021_236
Hanlon, S. M., Sim, D., Schneider, J. G., Yang, Z., & Thompson, S. M. (2023). The association between COVID-19 and febrile seizure: A retrospective case-control study. Pediatric Emergency Care. https://doi.org/10.1097/PEC.0000000000002935
Hess, D. C., Eldahshan, W., & Rutkowski, E. (2020). COVID-19-related stroke. Translational Stroke Research, 11(3), 322–325. https://doi.org/10.1007/s12975-020-00818-9
Iijima, H., Kubota, M., & Ogimi, C. (2022). Change in seizure incidence in febrile children with COVID-19 in the era of omicron variant of concern. Journal of the Pediatric Infectious Diseases Society, 11(11), 514–517. https://doi.org/10.1093/jpids/piac085
Kaur, H., Mason, J. A., Bajracharya, M., McGee, J., Gunderson, M. D., Hart, B. L., Dehority, W., Link, N., Moore, B., Phillips, J. P., & Rogers, D. (2020). Transverse myelitis in a child with COVID-19. Pediatric Neurology, 112, 5–6. https://doi.org/10.1016/j.pediatrneurol.2020.07.017
Khalifa, M., Zakaria, F., Ragab, Y., Saad, A., Bamaga, A., Emad, Y., & Rasker, J. J. (2020). Guillain-Barré syndrome associated with severe acute respiratory syndrome coronavirus 2 detection and coronavirus disease 2019 in a Child. Journal of the Pediatric Infectious Diseases Society, 9(4), 510–513. https://doi.org/10.1093/jpids/piaa086
LaRovere, K. L., Riggs, B. J., Poussaint, T. Y., Young, C. C., Newhams, M. M., Maamari, M., Walker, T. C., Singh, A. R., Dapul, H., Hobbs, C. V., McLaughlin, G. E., Son, M. B. F., Maddux, A. B., Clouser, K. N., Rowan, C. M., McGuire, J. K., Fitzgerald, J. C., Gertz, S. J., Shein, S. L., Overcoming COVID-19 Investigators. (2021). Neurologic involvement in children and adolescents hospitalized in the United States for COVID-19 or multisystem inflammatory syndrome. JAMA Neurology, 78(5), 536–547. https://doi.org/10.1001/jamaneurol.2021.0504
Lindan, C. E., Mankad, K., Ram, D., Kociolek, L. K., Silvera, V. M., Boddaert, N., Stivaros, S. M., Palasis, S., ASPNR PECOBIG Collaborator Group. (2021). Neuroimaging manifestations in children with SARS-CoV-2 infection: A multinational, multicentre collaborative study. The Lancet. Child & Adolescent Health, 5(3), 167–177. https://doi.org/10.1016/S2352-4642(20)30362-X
Lopez-Leon, S., Wegman-Ostrosky, T., Valle, N. C. A. del, Perelman, C., Sepulveda, R., Rebolledo, P. A., Cuapio, A., & Villapol, S. (2022). Long-COVID in children and adolescents: A systematic review and meta-analyses (p. 2022.03.10.22272237). medRxiv. https://doi.org/10.1101/2022.03.10.22272237
Maddux, A. B., Berbert, L., Young, C. C., Feldstein, L. R., Zambrano, L. D., Kucukak, S., Newhams, M. M., Miller, K., FitzGerald, M. M., He, J., Halasa, N. B., Cvijanovich, N. Z., Loftis, L. L., Walker, T. C., Schwartz, S. P., Gertz, S. J., Tarquinio, K. M., Fitzgerald, J. C., Kong, M., Overcoming COVID-19 Investigators. (2022). Health impairments in children and adolescents after hospitalization for acute COVID-19 or MIS-C. Pediatrics, 150(3), e2022057798. https://doi.org/10.1542/peds.2022-057798
Manzo, M. L., Galati, C., Gallo, C., Santangelo, G., Marino, A., Guccione, F., Pitino, R., & Raieli, V. (2021). ADEM post-Sars-CoV-2 infection in a pediatric patient with Fisher-Evans syndrome. Neurological Sciences, 42(10), 4293–4296. https://doi.org/10.1007/s10072-021-05311-1
McGill, F., Griffiths, M. J., & Solomon, T. (2017). Viral meningitis: Current issues in diagnosis and treatment. Current Opinion in Infectious Diseases, 30(2), 248–256. https://doi.org/10.1097/QCO.0000000000000355
Meinhardt, J., Radke, J., Dittmayer, C., Franz, J., Thomas, C., Mothes, R., Laue, M., Schneider, J., Brünink, S., Greuel, S., Lehmann, M., Hassan, O., Aschman, T., Schumann, E., Chua, R. L., Conrad, C., Eils, R., Stenzel, W., Windgassen, M., & Heppner, F. L. (2021). Olfactory transmucosal SARS-CoV-2 invasion as a port of central nervous system entry in individuals with COVID-19. Nature Neuroscience, 24(2), 2. https://doi.org/10.1038/s41593-020-00758-5
Mirzaee, S. M. M., Gonçalves, F. G., Mohammadifard, M., Tavakoli, S. M., & Vossough, A. (2020). Focal cerebral arteriopathy in a pediatric patient with COVID-19. Radiology, 297(2), E274–E275. https://doi.org/10.1148/radiol.2020202197
Murphy, O. C., Messacar, K., Benson, L., Bove, R., Carpenter, J. L., Crawford, T., Dean, J., DeBiasi, R., Desai, J., Elrick, M. J., Farias-Moeller, R., Gombolay, G. Y., Greenberg, B., Harmelink, M., Hong, S., Hopkins, S. E., Oleszek, J., Otten, C., Sadowsky, C. L., & Pardo, C. A. (2021). Acute flaccid myelitis: Cause, diagnosis, and management. Lancet (London, England), 397(10271), 334–346. https://doi.org/10.1016/S0140-6736(20)32723-9
Nejad Biglari, H., Sinaei, R., Pezeshki, S., & Khajeh Hasani, F. (2021). Acute transverse myelitis of childhood due to novel coronavirus disease 2019: The first pediatric case report and review of literature. Iranian Journal of Child Neurology, 15(1), 107–112. https://doi.org/10.22037/ijcn.v15i1.31579
Olivotto, S., Basso, E., Lavatelli, R., Previtali, R., Parenti, L., Fiori, L., Dilillo, D., Zuccotti, G. V., Veggiotti, P., & Bova, S. M. (2021). Acute encephalitis in pediatric multisystem inflammatory syndrome associated with COVID-19. European Journal of Paediatric Neurology, 34, 84–90. https://doi.org/10.1016/j.ejpn.2021.07.010
Parvez, M. S. A., & Ohtsuki, G. (2022). Acute cerebellar inflammation and related ataxia: Mechanisms and pathophysiology. Brain Sciences, 12(3), 367. https://doi.org/10.3390/brainsci12030367
Paybast, S., Gorji, R., & Mavandadi, S. (2020). Guillain-Barré syndrome as a neurological complication of novel COVID-19 infection. The Neurologist, 25. https://doi.org/10.1097/NRL.0000000000000291
Payne, A. B., Gilani, Z., Godfred-Cato, S., Belay, E. D., Feldstein, L. R., Patel, M. M., Randolph, A. G., Newhams, M., Thomas, D., Magleby, R., Hsu, K., Burns, M., Dufort, E., Maxted, A., Pietrowski, M., Longenberger, A., Bidol, S., Henderson, J., Sosa, L., & Cholette, J. M. (2021). Incidence of multisystem inflammatory syndrome in children among US persons infected with SARS-CoV-2. JAMA Network Open, 4(6), e2116420. https://doi.org/10.1001/jamanetworkopen.2021.16420
Poyrazoğlu, H. G., Kırık, S., Sarı, M. Y., Esen, İ, Toraman, Z. A., & Eroğlu, Y. (2022). Acute demyelinating encephalomyelitis and transverse myelitis in a child with COVID-19. The Turkish Journal of Pediatrics, 64(1), 133. https://doi.org/10.24953/turkjped.2020.3385
Púa Torrejón, R. C., Ordoño Saiz, M. V., González Alguacil, E., Furones García, M., Cantarín Extremera, V., Ruiz Falcó, M. L., & Soto Insuga, V. (2022). Smell and taste dysfunction in pediatric patients with SARS-CoV-2 infection. Pediatric Neurology, 136, 28–33. https://doi.org/10.1016/j.pediatrneurol.2022.07.006
Sa, M., Mirza, L., Carter, M., Carlton Jones, L., Gowda, V., Handforth, J., Hedderly, T., Kenny, J., Lascelles, K., Lin, J.-P., Lumsden, D., McDougall, M., Miller, O., Rossor, T., Shivamurthy, V., Siddiqui, A., Singh, R., Tang, S., White, M., … Evelina PIMS-TS Study Group. (2021). Systemic inflammation is associated with neurologic involvement in pediatric inflammatory multisystem syndrome associated with SARS-CoV-2. Neurology(R) Neuroimmunology & Neuroinflammation, 8(4), e999. https://doi.org/10.1212/NXI.0000000000000999
Sadeghizadeh, A., Pourmoghaddas, Z., Zandifar, A., Tara, S. Z., Rahimi, H., Saleh, R., Ramezani, S., Ghazavi, M., Yaghini, O., Hosseini, N., Aslani, N., Saadatnia, M., & Vossough, A. (2022). Reversible cerebral vasoconstriction syndrome and multisystem inflammatory syndrome in children with COVID-19. Pediatric Neurology, 129, 1–6. https://doi.org/10.1016/j.pediatrneurol.2021.12.013
Schober, M. E., Pavia, A. T., & Bohnsack, J. F. (2021). Neurologic manifestations of COVID-19 in Children: Emerging pathophysiologic insights. Pediatric Critical Care Medicine, 22(7), 7. https://doi.org/10.1097/PCC.0000000000002774
Stamm, B., Huang, D., Royan, R., Lee, J., Marquez, J., & Desai, M. (2022). Pathomechanisms and treatment implications for stroke in COVID-19: A review of the literature. Life, 12(2), 2. https://doi.org/10.3390/life12020207
Tenembaum, S., Chamoles, N., & Fejerman, N. (2002). Acute disseminated encephalomyelitis: A long-term follow-up study of 84 pediatric patients. Neurology, 59(8), 1224–1231. https://doi.org/10.1212/WNL.59.8.1224
Thongsing, A., Eizadkhah, D., Fields, C., & Ballaban-Gil, K. (2022). Provoked seizures and status epilepticus in a pediatric population with COVID-19 disease. Epilepsia. https://doi.org/10.1111/epi.17293.10.1111/epi.17293
Trapani, S., Rubino, C., Lasagni, D., Pegoraro, F., Resti, M., Simonini, G., & Indolfi, G. (2022). Thromboembolic complications in children with COVID-19 and MIS-C: A narrative review. Frontiers in Pediatrics, 10, 944743. https://doi.org/10.3389/fped.2022.944743
Urso, L., Distefano, M. G., Cambula, G., Colomba, A. I., Nuzzo, D., Picone, P., Giacomazza, D., & Sicurella, L. (2022). The case of encephalitis in a COVID-19 pediatric patient. Neurological Sciences, 43(1), 105–112. https://doi.org/10.1007/s10072-021-05670-9
Varga, Z., Flammer, A. J., Steiger, P., Haberecker, M., Andermatt, R., Zinkernagel, A. S., Mehra, M. R., Schuepbach, R. A., Ruschitzka, F., & Moch, H. (2020). Endothelial cell infection and endotheliitis in COVID-19. The Lancet, 395(10234), 1417–1418. https://doi.org/10.1016/S0140-6736(20)30937-5
Whitworth, H., Sartain, S. E., Kumar, R., Armstrong, K., Ballester, L., Betensky, M., Cohen, C. T., Diaz, R., Diorio, C., Goldenberg, N. A., Jaffray, J., Keegan, J., Malone, K., Randolph, A. G., Rifkin-Zenenberg, S., Leung, W. S., Sochet, A., Srivaths, L., Zia, A., & Raffini, L. (2021). Rate of thrombosis in children and adolescents hospitalized with COVID-19 or MIS-C. Blood, 138(2), 190–198. https://doi.org/10.1182/blood.2020010218
Woodruff, R. C., Campbell, A. P., Taylor, C. A., Chai, S. J., Kawasaki, B., Meek, J., Anderson, E. J., Weigel, A., Monroe, M. L., Reeg, L., Bye, E., Sosin, D. M., Muse, A., Bennett, N. M., Billing, L. M., Sutton, M., Talbot, H. K., McCaffrey, K., Pham, H., & Havers, P. F. (2022). Risk factors for severe COVID-19 in children. Pediatrics, 149(1), e2021053418. https://doi.org/10.1542/peds.2021-053418
Zhu, F., & Ang, J. Y. (2022). COVID-19 infection in children: Diagnosis and management. Current Infectious Disease Reports, 24(4), 51–62. https://doi.org/10.1007/s11908-022-00779-0
Acknowledgements
The authors would like to acknowledge Dr. Grace Mucci for her assistance in reviewing this paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kung, M., Adler-Shohet, F., Lai, H. et al. Neurological and Neuroimaging Implications of COVID-19 in the Pediatric Population. J Pediatr Neuropsychol 10, 5–19 (2024). https://doi.org/10.1007/s40817-024-00157-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40817-024-00157-9