Abstract
Introduction
Bronchial thermoplasty (BT) is a bronchoscopic procedure that involves the delivery of thermal radiofrequency energy to the bronchial wall for treating severe asthma. It has been suggested that too many radiofrequency activations could induce serious adverse events (SAEs) at an early stage. We aimed to examine the number of radiofrequency activations at each session and early lung function changes from baseline to determine whether these are related to SAEs.
Methods
We retrospectively investigated 13 consecutive patients who underwent three sessions each of BT for severe asthma from February 2015 to January 2016. Lung function tests were performed on the day before and after each BT procedure. Since we compared the number of activations and lung function changes from baseline after each session, a total of 39 sessions were reviewed. The relationship between the number of radiofrequency activations and each lung function change from baseline was also examined by linear regression analysis.
Results
A total of 10 SAEs (4 of pneumonia, 3 of atelectasis, 2 of bronchial asthma exacerbation and 1 of hemoptysis) were observed following the 39 BT sessions. When we compared sessions with and without SAEs, there were no differences in the number of activations (mean ± SD, 71.5 ± 28.6 times in sessions with SAEs; 66.5 ± 25.1 times in sessions without SAEs; p = 0.772) and lung function changes (mean changes in FVC/%FVC/FEV1/%FEV1/%PEF from baseline; − 0.49 l/− 14.2%/− 0.36 l/− 11.7%/− 9.6% in sessions with SAEs; − 0.43 l/− 13.3%/− 0.34 l/− 12.1%/− 9.4% in sessions without SAEs; p > 0.05 for all the above). Increase in the number of activations correlated with decreased FEV1 (R2 = 0.17, p = 0.0088) and %FEV1 (R2 = 0.11, p = 0.0357).
Conclusions
Increase in the number of radiofrequency activations during BT is related to a decrease in FEV1 and %FEV1 from baseline. The number of radiofrequency activations, however, is not associated with SAEs after BT.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Plain Language Summary
Bronchial thermoplasty (BT) is a bronchoscopic procedure that involves the delivery of thermal radiofrequency energy to the bronchial wall for treating severe asthma. BT is performed as three sessions at 3-week intervals. A previous in vitro study and a case report demonstrated that too much thermal energy caused irreversible epithelial cell necrosis. It might induce early serious adverse events (SAEs). This study evaluated the number of radiofrequency activations during successive BT sessions and early lung function changes from baseline to determine whether these are related to SAEs. We retrospectively investigated 13 consecutive patients (mean age, 55.2 years; 7 males) who underwent the BT procedure for severe asthma from February 2015 to January 2016 at the National Center for Global Health and Medicine (Tokyo, Japan). Lung function tests were performed on the day before and after each BT procedure. A total of 10 SAEs (4 of pneumonia, 3 of atelectasis, 2 of bronchial asthma exacerbation and 1 of hemoptysis) were observed following 39 BT sessions. Comparison of sessions with and without SAE indicated no difference in the number of activations (mean ± SD, 71.5 ± 28.6 times during sessions with SAEs; 66.5 ± 25.1 times during sessions without SAEs; p = 0.31) and lung function changes from baseline. Increase in the number of radiofrequency activations correlated with reduction in FEV1 and %FEV1.
Key Summary Points
Why carry out this study? |
Bronchial thermoplasty (BT) is a novel bronchoscopic procedure that involves the delivery of thermal radiofrequency energy to the bronchial wall for treating severe asthma |
It is hypothesized that too many radiofrequency activations could induce early serious adverse events (SAEs) |
What was learned from the study? |
When we compared BT sessions with and without SAEs, there was no difference in the number of radiofrequency activations (mean ± SD, 71.5 ± 28.6 times in sessions with SAEs; 66.5 ± 25.1 times in sessions without SAEs; p = 0.77) |
Increase in the number of activations correlated with a decrease in FEV1 (R2 = 0.17, p = 0.0088) and %FEV1 (R2 = 0.11, p = 0.0357) |
This study revealed that the number of radiofrequency activations is not associated with SAEs; further studies elucidating the predictive factors for SAEs are required to enhance patient safety |
Introduction
Asthma remains an important health problem with significant morbidity, mortality and economic burden. Airway smooth muscle contraction and the resultant bronchoconstriction induce the many variable symptoms of asthma. Increased airway smooth muscle mass is a characteristic feature of asthma, especially among patients with severe asthma. Bronchial thermoplasty (BT) is a novel intervention that involves the delivery of thermal radiofrequency energy to the bronchial wall during a series of bronchoscopies, resulting in prolonged reduction of airway smooth muscle mass. In previous clinical studies, treatment of patients with airways between 3 and 10 mm in diameter led to clinically meaningful reduction in muscle-mediated narrowing of the airway and to improvement of asthma symptoms [1,2,3].
The most common complications of BT are transient adverse respiratory events that occur a median of 1 day after the procedure and rarely require hospitalization [4]. Recently, however, serious adverse events (SAEs) involving the airways, which required prolonged hospitalization or threatened the patient’s life, have been reported after the BT procedure [5,6,7,8]. Hence, it is necessary to determine early predictors of SAEs after BT to enhance patient safety.
Even a single radiofrequency activation can affect the bronchial mucosa and cause epithelial sloughing [9]. A previous in vitro study and a case report demonstrated that too much thermal energy caused irreversible epithelial cell necrosis [10, 11]. Several studies suggested that intense thermal stimulation of the bronchial mucosa could represent a strong boost for inflammation, with microvascular alteration being directly induced by the heat or through the release of chemical mediators causing mucosal exudation and edema [5, 12]. Since the number of activations is directly related to the amount of thermal energy, we hypothesized that the number of activations was related to SAEs in the early post-procedure stage. Generally, lung function tests are used to evaluate airway obstruction in persons with asthma. Hence, we hypothesized that early changes in lung function following BT might be useful to predict SAEs.
In this study, we aimed to examine the number of radiofrequency activations during each BT session and early lung function changes from baseline to determine whether these are related to SAEs following BT.
Methods
Patients
We retrospectively investigated adult asthma patients aged 18 years or older who underwent three sequential BT procedures at the National Center for Global Health and Medicine, Tokyo, Japan, from February 2015 to January 2016. All of them performed lung function tests on the day before and after each BT procedure. As previously described [3], BT is indicated in patients aged > 18 years and with severe uncontrolled asthma despite high-dose inhalational corticosteroid therapy. The contraindications to BT include [3]: (1) presence of a pacemaker, internal defibrillator or other implantable electronic devices; (2) known sensitivity to the medications used during bronchoscopy; (3) previous treatment with BT at the same area; (4) active respiratory infection; (5) pulmonary emphysema or any cystic disease; (6) asthma exacerbation or change in the dose of systemic corticosteroids for asthma in the past 14 days; (7) known coagulopathy; (8) therapy with anticoagulants, antiplatelet agents and non-steroidal anti-inflammatory drugs before the procedure.
Study Design
We conducted a retrospective case-control study. Patient factors (age, sex, height, body weight, smoking status, nasal comorbidities, laboratory data, Global Initiative for Asthma treatment step, quality of life, medications and lung function tests) and BT procedure factors (number of radiofrequency activations, procedure time and time/activation ratio) were investigated from the patients’ charts. SAEs were defined as undesirable adverse experiences occurring within 30 days after the BT procedure, as previously described [13]. They included death, life-threatening conditions, prolonged hospitalization, disability and conditions requiring intervention (e.g., antibiotics, systemic steroid, oxygen therapy and artificial ventilator). The primary aim of this study was to examine the number of radiofrequency activations during each session and early lung function changes from baseline following BT and to determine whether these are related to SAEs. The secondary aim was to evaluate the relationship between the number of activations and each lung function change from baseline. Each treatment session was divided into two groups: the SAE group, which represented sessions with SAEs, and non-SAE group, which represented sessions without SAEs. This study followed the principles of the Declaration of Helsinki and was approved by the institutional review board of the National Center for Global Health and Medicine, Tokyo, Japan (approval no. NCGM-G-001801-00) on June 15, 2015. We verified that written informed consent was obtained before each patient’s participation in this study.
Bronchial Thermoplasty
Using the ALAIR® device (Boston Scientific Japan, Tokyo, Japan), BT was performed in three sessions at 3-week intervals. The lower lobe bronchi were treated in turn during the first two sessions, beginning with the right bronchus in the first session and the left bronchus in the second session, and both upper lobe bronchi in the third BT procedure. The right middle lobe bronchus was not treated in order to avoid the theoretical risk of obstruction and right middle lobe syndrome. The system was adjusted to deliver a constant temperature of 65°C for 10 s at each treatment site from the small 3-mm bronchi to the lobar bronchi. The procedure was performed under intravenous sedation with midazolam. The patients underwent close clinical monitoring immediately after the BT procedure. If there were no SAEs, they were discharged a few days after the BT procedure. Prednisone (50 mg/day) was systematically administered orally from 3 days before the BT procedure to 1 day after each procedure.
Lung Function Tests
BT was performed as three sessions at 3-week intervals. We examined lung function tests by spirometry (Superior Spiro Discom-21 FXIII; Chest, Tokyo, Japan) on the day before (as baseline) and the day after each BT procedure. Differences between lung function on the day before and after BT were calculated as the change from baseline. A single experienced clinical technologist performed all the lung function tests with the patients in the seated position, the tests being performed at least twice to verify the stability of lung function values. For quantitative evaluation of airway status, the parameters of forced vital capacity (FVC), percent forced vital capacity (%FVC), forced expiratory volume in 1 s (FEV1), percent forced expiratory volume in 1 s (%FEV1) and percent peak expiratory flow (%PEF) were used. We used each prediction formula to predict the FVC value by Baldwin, FEV1 value by Berglund and PEF value by Cherniack.
Statistical Analysis
Qualitative variables are shown as numbers and percentages. Quantitative variables are reported as mean values and SDs, unless otherwise indicated. We used Mann-Whitney U test to compare the treated lobes, mean number of activations, procedure time, time/activation ratio and lung function changes from baseline (FVC, %FVC, FEV1, %FEV1, %PEF) between sessions with and without SAEs. The transition of lung function among all bronchial thermoplasty sessions was compared using the paired t-test. The potential associations between the number of activations and lung function changes from baseline were assessed using Pearson’s correlation coefficient (R2). All tests were two-tailed. P values < 0.05 were considered significant. Data were analyzed statistically with JMP® 14.0.0 software (SAS Institute Inc., Cary, NC, USA).
Results
Study Population
Patient demographics and baseline characteristics are shown in Table 1. A total of 13 patients were investigated in this study. The patients were on several medications, including high-dose inhaled corticosteroids (mean beclomethasone-equivalent dose of 1700 μg/day), systemic steroids (39% of patients) and molecular targeted drugs (46% of patients). All patients used high-dose inhaled corticosteroids and long-acting beta-agonists. As previously described, systemic steroids were given in the perioperative period (from 3 days before to the day after procedure; total 5 days)[14]. This is consistent with the standard preparation before the BT procedure. Judging according to the Global Initiative for Asthma (GINA) treatment steps [15], all patients in this study were categorized as severe asthma patients (31% of patients: GINA step 4; 69%: GINA step 5). Quality of life associated with asthma remained relatively stable (mean baseline Asthma Control Test/Asthma Quality of Life Questionnaire scores, 19.0/4.8, respectively). This was considered the result of high-intensity pharmacologic treatment. We performed chest CT scans in all patients before the procedure, which did not demonstrate the notable changes in the patient’s lungs (e.g., no emphysema, no cystic disease, no interstitial opacity, no bronchiectasis and no thickness of bronchi).
Early Lung Function Changes after BT
Table 2 demonstrates early lung function changes after the BT procedure. Evaluation of all BT procedures showed that all lung function parameters decreased after BT (mean change in FVC/%FVC/FEV1/%FEV1/%PEF from baseline, − 0.44 l/− 13.3%/− 0.35 l/− 12.2%/− 9.5%, respectively). Figure 1 shows the transition of lung function. Comparison of the first, second and third sessions indicated the most significant lung function change was at the third session. Although lung function decreased with each BT procedure, the change was temporary. Furthermore, lung function on the day before BT progressively improved with each BT procedure.
Transition of lung function among all bronchial thermoplasty sessions. Mean value of lung function test at six examination points (day before and after 1st, 2nd and 3rd BT procedure) is demonstrated. a Mean percent forced vital capacity. b Mean percent forced expiratory volume in 1 s. c Mean percent peak expiratory flow. In all the lung function tests, most values significantly decreased after the bronchial thermoplasty (BT) procedure (p < 0.05 in all cases except c). There were no significant differences between values on the day before the first BT procedure and the day before the third BT procedure (p > 0.05 in all cases). The mean value of each session was compared using the paired t-test
Comparison of SAE and Non-SAE Groups
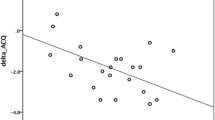
A total of 10 SAEs (4 of pneumonia, 3 of atelectasis, 2 of bronchial asthma exacerbation and 1 of hemoptysis) were observed over the 39 BT sessions. Comparison of the SAE and non-SAE groups (Table 3) showed that hospital stay in the SAE group tended to be longer than that in the non-SAE group (p = 0.064). Two SAEs occurred within 3 weeks of the first BT session, and four each occurred within 3 weeks of the second and third BT sessions. Asthma exacerbation was only seen within 3 weeks of the third BT session. In two cases, exacerbations of asthma were treated with additional bronchodilators and systemic steroids, and these patients needed longer hospitalization than those with the other adverse events. Three patients had complete atelectasis although the lung segment re-inflated without treatment, while four cases who developed pneumonia needed standard antibiotic therapy. One of the cases had a serious course with hemoptysis. During hospitalization, the subject had a small amount of bloody phlegm that was treated with a hemostatic agent, and the patient was subsequently discharged 8 days after the BT procedure. A few weeks after discharge, the subject was referred to the emergency department because of hemoptysis, and unconsciousness and was diagnosed with cerebral infarction, although the causal relationship with BT was unknown. There were no significant differences in the number of radiofrequency activations, procedure time or time-activation ratios between the two groups (mean number of activations/procedure time/time-activation ratio, 71.5 times/78.4 min/1.27 vs. 66.5 times/78.4 min/1.29, respectively, in the SAE and non-SAE groups, p > 0.05 for all values). There was also no significant difference in lung function change from baseline between the two groups (mean change in FVC/%FVC/FEV1/%FEV1/%PEF from baseline, − 0.49 l/− 14.2%/− 0.36 l/− 11.7%/− 9.6% vs. − 0.43 l/− 13.3%/− 0.34 l/− 12.1%/− 9.4%, respectively, in the SAE and non-SAE groups, p > 0.05 for all values). Linear regression analysis of the changes in lung function from baseline, performed to evaluate the relationship between the number of activations and lung function (Fig. 2), showed that FEV1 and %FEV1 had a negative correlation with the number of activations. On the other hand, FVC, %FVC and %PEF did not have any correlation.
Correlation between the number of radiofrequency activations and lung function changes from baseline. Linear regression analysis was performed to evaluate the relationship between the number of activations and changes in lung function from baseline, using the following parameters: a forced vital capacity (FVC), b percent forced vital capacity (%FVC), c forced expiratory volume in 1 s (FEV1), d percent forced expiratory volume in 1 s (%FEV1) and e percent peak expiratory flow (%PEF). Increase in the number of activations correlated with a decrease in FEV1 (R2 = 0.17, p = 0.0088) and %FEV1 (R2 = 0.11, p = 0.0357). There were no significant correlations between the number of activations and FVC/%FVC/%PEF
Discussion
To the best of our knowledge, this is the first report assessing the relationship between the number of radiofrequency activations and early SAEs after BT. We performed lung function tests to quantitatively evaluate morphologic airway changes to enable objective evaluation of the changes. Generally speaking, bronchial asthma exacerbation is involved with worsening lung function (FEV1 and PEF) [16], so lung function tests are suitable for assessment of asthmatic patients. Although Langton et al. focused on the number of activations as a predictor of treatment response [17], we hypothesized that this parameter could be a predictor of SAEs in the early stages after BT. Actually, in the present study, the number of activations was significantly related to the reduction in FEV1 and %FEV1 after the BT procedure (change in FEV1, R2 = 0.171, p = 0.009; change in %FEV1, R2 = 0.114, p = 0.036). The amount of FEV1 reduction on the day after BT and the correlation between the number of activations and FEV1 followed the same trend as in Langton’s report [18]. In addition, both Langton’s and our reports demonstrated that upper lobe treatment causes a greater decrease in FEV1 than lower lobe treatment. Increase in the number of activations might affect edematous changes in the bronchial mucosa and around the bronchus [4, 9, 19]. According to previous in vitro research, administration of thermal energy at a temperature of 37–70 °C to cultured airway epithelial cells and airway smooth muscle cells can cause irreversible epithelial cell necrosis [10]. In this research, we concluded that not only the number of activations, but also lung function changes over a short period were not enough to predict SAE occurrence.
Our research group has previously reported on airway morphologic and functional changes after BT therapy. We studied asthma-related quality of life, lung function tests and the number of asthma exacerbations both 1 and 12 months after the third BT session in 12 Japanese patients. We found that %FEV1 significantly improved a month after the third BT session (mean %FEV1 before first BT session: 70.5%, mean FEV1 a month after third BT session: 82.2%; p < 0.05) [20]. The report also confirmed that patients maintain the lung function improvement for a year (mean %FEV1 within a year of the third BT session: 82.3%). The reason why the BT procedure was capable of maintaining a sustained effect over a year in our study is that the total number of activations throughout BT in our institute was more than 1.3 times higher than in the AIR2 trial. Due to the larger number of activations, more peripheral airways could be treated. It was also previously reported that the number of activations could play a role in determining clinical response to treatment [17]. In the present study, %FEV1 followed the same trend as in our previous report. Moreover, %FEV1 tended to improve the day before the second BT procedure (mean %FEV1 a day before first BT session: 74.5%, mean %FEV1 a day before second BT session: 78.2%). After the first BT procedure, %FEV1 decreased temporarily, although %FEV1 on the day before the third BT procedure was better than that on the day before the first BT procedure (mean %FEV1 on the day before third BT: 85.2%) (Fig. 1). Our findings were supported by the recent study by Ishii et al. conducted in a 68-year-old Japanese woman with severe asthma who underwent BT, which confirmed dilation of the bronchial lumen and decreased bronchial wall thickness by comparing the changes in the bronchial lumen and walls before BT and after the first and third sessions by three-dimensional (3D) airway analysis [21]. Previous studies demonstrated the occurrence of morphologic changes in airways right after the BT procedure. However, there is no proof that the changes are related to functional improvement. Our reports suggest that the BT procedure leads to an initial decrease in lung function, although repeated sessions lead to gradual dilatation of the bronchial lumen and a decrease in bronchial wall thickness.
Previous major clinical trials have proved the safety and efficacy of BT procedures [1,2,3]. Compared with the incidence of SAEs in previous clinical trials, including the AIR trial, AIR2 trial and RISA study (7.3%, 8.4% and 26.7%, respectively), adverse events occurred relatively frequently among elderly and adult patients with severe asthma in this study (25.6%). Apart from the number of activations, the differences between the above trials and the present study are asthma severity, age and study design. In terms of asthma severity, the AIR and AIR2 trials targeted persons with moderate-to-severe persistent asthma. On the other hand, the present study and the RISA study targeted persons with severe asthma who were symptomatic despite maximal pharmacologic treatment. Asthma severity might affect SAE occurrence, although we could not assess the relationship between asthma severity and SAE occurrence because of the small number of subjects analyzed. Using two independent data sources, Burn et al. showed that there were more reports of re-admissions in the group who underwent the BT procedure in clinical practice (re-admission rate, 95% CI; 19.1%, 13.2–26.2) compared with the previous clinical trials. Further, they considered asthma severity and age as factors affecting the complication rate of BT. The average age of patients in the AIR trial, AIR2 trial, RISA study, Burn et al’s report and this study was 39.4 years, 40.7 years, 39.1 years, 42.6 years and 55.2 years, respectively [12]. Actually, the incidence of adverse events in elderly patients in the present study (25.6%) is slightly high compared with the other studies. The difference could be because this study reflects real-world data that were generated during routine clinical practice and enrolled many elderly and/or adult patients with severe asthma. Among the SAE group, additional needs for antibiotics or corticosteroid therapy were observed but significant prolongations of hospital stays were not observed (SAE group, 10.1 days; non-SAE group 6.1 days; p = 0.064) (Table 3). The previously reported treatments for adverse events after BT included fibrin plug removal by bronchoscopy for atelectasis secondary to occlusion by the plug [5], chest tube insertion for pneumothorax after partial atelectasis [7] and bronchial artery thrombosis and intubation for bronchial artery aneurysm rupture [6]. Such adverse events are the leading causes of death without proper treatment. Since a significant therapeutic effect of BT has been reported, the number of eligible patients for BT are expected to increase. However, the determinant factors related to SAEs remain unclear. This study is the first step to proving whether the number of activations and changes in lung function after the BT procedure are related to SAEs. Future studies should consider asthma severity and include an age-matched population.
There are some limitations to our study. The retrospective study conducted at a single center with a small number of patients limits the weight with which we can interpret the results. The limited number of patients and practitioners could lead to bias in terms of observation of SAEs and treatment, so a larger multicenter study is warranted. In this study, we used lung function tests to assess the occurrence of SAEs. Many previous studies have used chest CT scans to evaluate the lung damage resulting from radiofrequency activation [4, 19]. While CT scans provide high spatial resolution that is especially beneficial in evaluation of fibrin plugs in the airway and airway thickness, diagnostic x-rays are problematic in terms of radiation exposure. Japan has the highest annual frequency of diagnostic x-rays among developed countries. Reportedly, the cumulative cancer risk is related to diagnostic x-ray exposure [22]. Since women of childbearing age might need to undergo the BT procedure, we recommend that frequent diagnostic x-rays should be avoided.
Conclusions
Increase in the number of radiofrequency activations is involved in a decrease in FEV1 and %FEV1 from baseline after BT. The number of activations, however, is not associated with SAEs after BT. Therefore, further studies elucidating the factors predictive of SAEs are required to enhance patient safety after the procedure.
References
Pavord ID, Cox G, Thomson NC, et al. Safety and efficacy of bronchial thermoplasty in symptomatic, severe asthma. Am J Respir Crit Care Med. 2007;176:1185–91.
Cox G, Thomson NC, Rubin AS, et al. Asthma control during the year after bronchial thermoplasty. N Engl J Med. 2007;356:1327–37.
Castro M, Rubin AS, Laviolette M, et al. Effectiveness and safety of bronchial thermoplasty in the treatment of severe asthma: a multicenter, randomized, double-blind, sham-controlled clinical trial. Am J Respir Crit Care Med. 2010;181:116–24.
Debray MP, Dombret MC, Pretolani M, et al. Early computed tomography modifications following bronchial thermoplasty in patients with severe asthma. Eur Respir J. 2017;49:1601565.
Facciolongo N, Menzella F, Lusuardi M, et al. Recurrent lung atelectasis from fibrin plugs as a very early complication of bronchial thermoplasty: a case report. Multidiscip Respir Med. 2015;10:9.
Nguyen DV, Murin S. Bronchial artery pseudoaneurysm with major hemorrhage after bronchial thermoplasty. Chest. 2016;149:e95–7.
Funatsu A, Kobayashi K, Iikura M, et al. A case of pulmonary cyst and pneumothorax after bronchial thermoplasty. Respirol Case Rep. 2018;6:e00286.
Balu A, Ryan D, Niven R. Lung abscess as a complication of bronchial thermoplasty. J Asthma. 2015;52:740–2.
Goorsenberg AWM, d’Hooghe JNS, de Bruin DM, et al. Bronchial thermoplasty-induced acute airway effects assessed with optical coherence tomography in severe asthma. Respiration. 2018;96:564–70.
Chernyavsky IL, Russell RJ, Saunders RM, et al. In vitro, in silico and in vivo study challenges the impact of bronchial thermoplasty on acute airway smooth muscle mass loss. Eur Respir J. 2018;51:1701680.
Menzella F, Lusuardi M, Galeone C, et al. Heat-induced necrosis after bronchial thermoplasty: a new concern? Allergy Asthma Clin Immunol. 2018;14:25.
Burn J, Sims AJ, Keltie K, et al. Procedural and short-term safety of bronchial thermoplasty in clinical practice: evidence from a national registry and Hospital Episode Statistics. J Asthma. 2017;54:872–9.
Ioannidis JP, Lau J. Completeness of safety reporting in randomized trials: an evaluation of 7 medical areas. JAMA. 2001;285:437–43.
Cox G, Miller JD, McWilliams A, Fitzgerald JM, Lam S. Bronchial thermoplasty for asthma. Am J Respir Crit Care Med. 2006;173:965–9.
Walsh LJ, Wong CA, Oborne J, et al. Adverse effects of oral corticosteroids in relation to dose in patients with lung disease. Thorax. 2001;56:279–84.
Global Initiative for Asthma., National Heart Lung and Blood Institute. Global initiative for asthma: global strategy for asthma management and prevention. 2014 revision. ed. Bethsda: U.S. Dept. of Health and Human Services, Public Health Service; 2014. viii.
Langton D, Sha J, Ing A, et al. Bronchial thermoplasty: activations predict response. Respir Res. 2017;18:134.
Langton D, Wang W, Thien F, Plummer V. The acute effects of bronchial thermoplasty on FEV1. Respir Med. 2018;137:147–51.
d’Hooghe JNS, van den Berk IAH, Annema JT, Bonta PI. Acute radiological abnormalities after bronchial thermoplasty: a prospective cohort trial. Respiration. 2017;94:258–62.
Iikura M, Hojo M, Nagano N, et al. Bronchial thermoplasty for severe uncontrolled asthma in Japan. Allergol Int. 2018;67:273–5.
Ishii S, Iikura M, Hojo M, Sugiyama H. Use of 3D-CT airway analysis software to assess a patient with severe persistent bronchial asthma treated with bronchial thermoplasty. Allergol Int. 2017;66:501–3.
Berrington de Gonzalez A, Darby S. Risk of cancer from diagnostic X-rays: estimates for the UK and 14 other countries. Lancet. 2004;363:345–51.
Acknowledgements
The authors thank the participants of this study.
Funding
Boston Scientific Japan loaned the authors’ department the Alair thermoplasty system for 6 months and also donated six catheters. This work was supported by a grant from the National Center for Global Health and Medicine (NCGM-27-6001). The Rapid Service Fee was funded by the authors.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this manuscript, take responsibility for the integrity of the work as a whole, and have given final approval for the version to be published.
Medical Writing and/or Editorial Assistance
Additional statistical support was provided by Osamu Narumoto, MD, PhD, of National Hospital Organization Tokyo Hospital.
Disclosures
Motoyasu Iikura received lecture fees from Astra Zeneca K.K. Masayuki Hojo received lecture fees from Astra Zeneca K.K., Novartis Pharma K.K., KYORIN Holdings, Inc., and Boehringer Ingelheim GmbH. The other authors have no conflict of interest. Shota Yamamoto, Tamaki Kakuwa, Yoshie Tsujimoto, Sachi Matsubayashi, Naoko Nagano, Tomoyuki Suzuki, Keita Sakamoto, Konomi Kobayashi, Ayako Shiozawa, Masao Hashimoto, Satoru Ishii, Manabu Suzuki, Shinyu Izumi, Terumitsu Hasebe and Haruhito Sugiyama have nothing to disclose.
Compliance with Ethics Guidelines
This study followed the principles of the Declaration of Helsinki and was approved by the institutional review board of the National Center for Global Health and Medicine, Tokyo, Japan (approval no. NCGM-G-001801-00) on June 15, 2015. We verified that written informed consent was obtained before each patient’s participation in this study.
Data Availability
The datasets generated and/or analyzed during the present study are available from the corresponding author on reasonable request.
Author information
Authors and Affiliations
Corresponding author
Additional information
Enhanced digital features
To view enhanced digital features for this article go to https://doi.org/10.6084/m9.figshare.10043030.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any non-commercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Yamamoto, S., Iikura, M., Kakuwa, T. et al. Can the Number of Radiofrequency Activations Predict Serious Adverse Events after Bronchial Thermoplasty? A Retrospective Case-Control Study. Pulm Ther 5, 221–233 (2019). https://doi.org/10.1007/s41030-019-00103-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41030-019-00103-7