Abstract
Background
As Nigeria prepares to introduce a rotavirus vaccine, the Gavi board has approved the extension of the transition period for the country until 2028. The current position of the country on Gavi’s funding profile calls for a pragmatic step in planning and implementation so that sustainability at the fully self-financing phase will be feasible.
Objective
This study aimed to inform the decisions of the country’s health policymakers on the costs, benefits, and implications of the introduction of rotavirus vaccine.
Methods
This study was an economic evaluation using a simulation-based Markov model. It compared four approaches: ‘no vaccination’ and vaccination with ROTARIX, ROTAVAC, or ROTASIIL. Ten cohorts from the year 2021 to 2030 were used in the analysis. Primary measures were the benefit-cost ratio (BCR) and the incremental cost-effectiveness ratio (ICER). Future costs and outcomes were discounted to 2019 values.
Results
The adjusted vaccine cost of ROTARIX was the highest, followed by ROTAVAC and ROTASIIL, whereas the immunization delivery cost was in the reverse order. All the vaccines were very cost effective, with ROTARIX being the optimal choice for the 10-year period, having a BCR of 27 and an ICER of $US100 (95% confidence interval [CI] 71–130)/disability-adjusted life-year averted. Adopting ROTARIX was the optimal choice from 2021 to 2027, whereas ROTAVAC was optimal from 2028 to 2030. The net budget impact of the programme was $US76.9 million for the 10-year period. The opportunity cost of a late introduction was about $US8 million per annum from 2021 to 2028.
Conclusions
The rotavirus vaccine ROTARIX should be implemented in Nigeria at the earliest opportunity. A switch to ROTAVAC should be considered from the year 2028. Cost-minimization measures are imperative to ensure the sustainability of the programme after the transition out of Gavi support.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
A rotavirus vaccine should be introduced in Nigeria as soon as possible to control the high rotavirus mortality. |
Early introduction of the vaccine will save the country about $US8 million per annum between 2021 and 2028 from Gavi support. |
To sustain the programme, a cost-minimization blueprint must be designed. |
1 Introduction
The 2019 Institute for Health Metrics and Evaluation (IHME) statistics ranked Nigeria as the country with the highest rotavirus mortality globally [1]. Nigeria is at the threshold of introducing the rotavirus vaccine to its national immunization programme, and the country’s application for support from Gavi, the Vaccine Alliance was approved in early 2020. However, introduction of the vaccine remains pending as at the last quarter of 2020 [2]. The country is currently in the accelerated transition phase 2 stage based on Gavi’s classification, which implies that Gavi’s subsidy will be phased out soon. However, the Gavi board has approved the extension of the transition period for Nigeria until 2028 [3].
The current position of Nigeria on Gavi’s funding profile calls for a pragmatic step in planning and implementation so that sustainability of the rotavirus vaccine and other co-financed vaccines at the fully self-financing phase in the national immunization program will be feasible for the country [3]. The competition for the allocation of healthcare budgets in Nigeria has increased over the years because of increasing healthcare demands [4]. The limited resources have led to a limited supply of healthcare, leading to market failure [4]. It is therefore pertinent to correct this anomaly, especially for a yet-to-be-introduced vaccine to ensure sustainability and efficiency in the healthcare supply.
Given the limited resources in Nigeria, this study aimed to inform the decisions of health policymakers of the country on the costs, benefits, and implications of introducing the rotavirus vaccine. The study informs the current and future implications of decisions that will be made on rotavirus vaccination and provide recommendations that could aid implementation and sustainability.
2 Methods
2.1 Study Setting and Sample Size
The study was carried for the population of Nigerian children aged < 5 years at risk of having diarrhoea. The population figure was estimated from the 2019 population report [5]. The starting population used was children aged < 1 year, since it is the vaccination age for rotavirus vaccine. Children aged < 1 year were followed for up to 5 years to capture the associated costs and benefits of rotavirus vaccination. Ten consecutive cohorts were used in the analysis from the year 2021 to 2030, which represent periods during and after the transition out of Gavi support.
2.2 Study Perspective
The study was carried out from two perspectives: the health sector (payer) and the societal. The payer perspective represents the government perspective, which captures the effect of Gavi support for Nigeria. In this perspective, the costs of the vaccines used were the co-financed costs that will be incurred by the Nigerian government and the associated cost of immunization delivery (cold chain cost, transportation cost, personnel cost, and other logistics-related costs). The societal perspective included the complete cost of the vaccines (assuming no Gavi subsidy).
2.3 Interventions
This study compared four scenarios. The first scenario was ‘no vaccination’. The second scenario was vaccination with ROTARIX, a live attenuated monovalent human liquid oral rotavirus vaccine (RV1) manufactured by GlaxoSmithKline Biologicals, Belgium. The third scenario was vaccination with ROTAVAC, a live attenuated monovalent human-bovine liquid frozen oral rotavirus vaccine (RV1), manufactured by Bharat Biotech, India. The fourth scenario was vaccination with ROTASIIL, a live attenuated pentavalent bovine-human reassortant lyophilized oral rotavirus vaccine (RV5), manufactured by the Serum Institute of India. The ‘no vaccination’ scenario was compared with the rotavirus vaccine scenarios. The three rotavirus vaccine scenarios were also compared to determine which scenario would optimize resource utilization.
2.4 Model and Assumptions
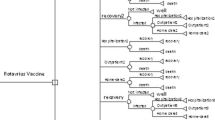
The study employed a simulation-based Markov model using retrospective data for Nigeria. The states in the model were well, moderate diarrhoea, severe diarrhoea, and death. The starting age in the model was 1 week. The population aged < 1 year was modelled from week 1 to week 260, which implies that the children were followed up for 5 years. The infants were modelled to start from the well state, and they could move to any or remain in a health state or die as a result of diarrhoea or all-cause mortality. The model states and transitions are illustrated in Fig. 1.
The transition probabilities of moving to the different health states (well, moderate, or severe diarrhoea), transition probabilities to diarrhoeal death for < 1 year, 1–4 years, and all-cause mortality, and the disability weights for moderate and severe diarrhoea were obtained from the 2019 IHME report for Nigeria [1]. The probabilities of recurrent moderate and severe diarrhoea were estimated from a systematic review [6], and the life table data were obtained from the World Health Organisation (WHO) report [7]. The yearly probabilities were modelled as weekly probabilities.
For each cycle, the model estimated the number of moderate and severe rotavirus gastroenteritis (RVGE) cases and the number of deaths and accumulated deaths over 5 years for each scenario. Based on the care-seeking characteristics of caregivers for children aged < 5 years in Nigeria [8,9,10], 75% of moderate RVGE and 50% of severe RVGE will be managed at home with oral rehydration salt (ORS) or other fluids purchased at drug stores, traditional approaches, or other means. Only 25% of moderate RVGE cases will seek outpatient healthcare [8,9,10], and 50% of severe RVGE cases will seek care and be managed in hospital [9, 10]. The duration of moderate RVGE was estimated to be 5 days, and severe RVGE was 8 days [6]. Children with moderate RVGE received ORS and zinc for 5 days; children with severe RVGE received ringer’s lactate intravenous fluid (IVF) (for 3 days), ORS (for the following 5 days), and zinc (for 10 days) [11]. From observed practices in Nigeria, for moderate RVGE, it was assumed that patients would visit the clinic once as outpatients except if the case worsened and led to severe diarrhoea, whereas patients with severe RVGE would be hospitalized for 3 days and visit the clinic on the last day (8th day). Details of the input parameters are shown in Table 1 and in the electronic supplementary material (ESM).
2.5 Time Horizon and Discount Rate
The model simulated costs and outcomes within the period of 260 weeks for the population at risk of having diarrhoea for the different scenarios and cohorts (the year 2021–2030). A recommended discount rate of 5% for low- and middle-income countries was used in the costs and outcomes analysis [20].
2.6 Vaccine Coverage, Effectiveness, and Wastage
The diphtheria-tetanus-pertussis (DTP3) coverage rate of 57% for Nigeria was used in estimating the costs and outcomes of the analysis [12]. The effectiveness of the first and last doses of ROTARIX was obtained from a systematic review for countries with high diarrhoeal mortality [13]. The effectiveness of the first, second, and last doses of ROTAVAC was estimated from a clinical trial in India [14], and the effectiveness of the first, second, and last doses of ROTASIIL was estimated from two clinical trials in India and Niger [15, 16]. The first dose of ROTARIX, ROTAVAC, and ROTASIIL was administered at week 6 after birth in the model. The second dose of ROTAVAC and ROTASIIL was administered at week 16, whereas the last dose of the three vaccines was administered at week 24 after birth. The effect of herd immunity with rotavirus vaccination was applied in the model by incorporating the post-vaccination effectiveness of the vaccine to the cohort in the model at 2, 4, 8, 12, 24, 36, 48, 72, 96, 144, and 192 weeks after vaccination [17].
Using the WHO vaccine wastage rates calculator, wastage rates were estimated at 4% for ROTARIX from the year 2021 to 2030; 50% for ROTAVAC (five-vial size) from the year 2021 to 2025 and 51% for the same pack size from the year 2026 to 2030; 21% for ROTASIIL (two-vial size) from the year 2021 to 2026 and 22% for the same pack size from the year 2027 to 2030 [18]. Details of the vaccines’ effectiveness and wastage are shown in Table 1.
2.7 Calculation of Cost
The WHO guidelines for estimating the costs of introducing new vaccines into the national immunization system were adopted to estimate the resource use and costs associated with the introduction of ROTARIX, ROTAVAC (five-vial size), or ROTASIIL (two-vial size) [25]. A bottom-up costing approach was used in the analysis. Costs were estimated from the payer and societal perspectives, respectively. The costs included vaccine cost (full dose), personnel cost (including salaries to healthcare professionals, health assistants, and administrative staff; advocacy and social mobilization costs; surveillance costs), and logistic costs (costs of vaccine storage in the cold chain, vaccine transportation, vehicles and vehicle maintenance, training/education of immunization staff, and wastage cost). The vaccine price used was the United Nations Children’s Fund (UNICEF) supply price for Gavi-eligible countries [2]. Based on vaccination coverage, population, and gross domestic product per capita (GDP), the immunization delivery costs were estimated from the immunization delivery cost catalogue of the Immunization Costing Action Network, Washington [23]. Costs captured all personnel and logistics costs related to immunization delivery, including buildings and utilities, safety boxes, syringes, and other recurrent costs. The UNICEF international handling cost of 3.5% for new vaccines for non-least developed countries and an international transportation cost of 7.5% of the vaccine price were included in the vaccine cost [22]. The waste-adjusted vaccine cost and the waste-adjusted cold chain volume and cost were factored in the final cost of each vial of vaccine and the immunization delivery cost. In calculating the annual vaccine cost based on the vaccination coverage and the WHO guideline [25], the cost of reserve stock was included in the first year. The reserve stock was estimated as 25% of the vaccine cost in the year 2021. The cost of rotavirus vaccination was calculated for each year from 2021 to 2030 using the formula c = p × n, where p is the price per dose of new vaccine (including wastage cost and international handling and freight) and n is the number of doses.
For the payer perspective, a starting fraction of 0.24 was used on the unadjusted vaccine price based on Nigeria’s co-financing contribution for all co-financed vaccines [3, 21]. Based on Gavi’s co-financing guideline for phase 2 transitioning countries, a 1.15 factor of price fraction was applied to the starting fraction in the year 2021 unadjusted vaccine costing [21]. From the year 2022 to 2028, the cost increased linearly to reach 100%. From the year 2029 to 2030, the cost was modelled such that Nigeria will fully self-finance the vaccine cost at the UNICEF’s price. For the societal perspective, Gavi subsidy was not considered. To estimate the cost of RVGE from the health sector or payer perspective, the cost included was the direct medical cost of treatment at the health facilities, whereas the societal perspective costs included direct medical, direct non-medical, and indirect costs of treatment at health facilities plus the costs of self-management at home (for cases that did not visit a health facility). The cost of self-care management at home was assumed to be the cost of ORS since the majority of caregivers use rehydration fluid [8]. An average cost of ORS for 3 days was assumed and used to estimate the costs of self-management at home [8]. The direct non-medical costs included the cost of diapers for 5 days (moderate RVGE) and 8 days (severe RVGE) and transportation costs. Indirect costs were estimated as productivity loss due to work absenteeism of caregiver (half a day for moderate RVGE and 1 day for severe RVGE) based on Nigeria’s 2019 GDP per capita. The direct costs of moderate and severe RVGE, including physician consultancy, nursing service, hospital bed, ORS, IVF, and zinc, were estimated from the Nigerian National Health Insurance Scheme drug price list [26]. The number of moderate and severe RVGE cases averted was multiplied by their respective mean cost of management to obtain the monetary savings. All costs were expressed in $US, year 2019 values. Gamma distribution was used to capture the uncertainty in the cost parameters. Details of the cost components are shown in Table 1 and the ESM.
The budget impact of the programme was also estimated. This was done by calculating the annual cost of RVGE healthcare at health facilities averted from 2021 to 2030. For each year, the net budget impact was estimated as the difference between the vaccination programme cost for that year and the healthcare cost averted for the same year. The total net budget impact was the sum of the net budget impact from 2021 to 2030.
2.8 Health Outcomes
The primary outcome was measured as disability-adjusted life-year (DALY) averted. The DALY was calculated as the sum of the years of life lived with disability (YLD) from morbidity and the years of life lost (YLL) from mortality. YLD = number of cases × duration till remission or death × disability weight [27, 28], and YLL = number of deaths due to diarrhoea × life expectancy at the age of death [28]. The DALYs across each cycle were summed and averaged to obtain the standard DALY. The DALY averted was calculated as the difference between the ‘no vaccination’ DALY and the DALY in the rotavirus vaccination scenarios. The primary outcome was used to evaluate the benefit-cost ratio (BCR) and the incremental cost-effectiveness ratio (ICER), which are the two approaches for assessment in this study. In calculating the monetary value of a DALY averted for the BCR evaluation, the Harvard-led guideline for conducting benefit-cost analysis projects was used [29]. The valuation was based on the value of statistical life-year with 1 DALY averted valued at 1.3 times the gross national income (GNI) per capita of a country in sub-Saharan Africa. For the ICER evaluation, a conservative cost-effectiveness threshold of 0.52 times the GDP per capita of Nigeria was used [30].
The secondary outcomes of the study included the total number of RVGE cases averted, the number of moderate RVGE cases averted, the number of severe RVGE hospitalizations averted, and the number of RVGE deaths averted. The averted cases were calculated as the difference between the ‘no vaccination’ scenario and the rotavirus vaccination scenarios.
2.9 Data Analyses
The appropriate distribution for each variable was used, as shown in Table 1. Half-cycle correction using the lifetable method was applied in the analysis [31]. Probabilistic sensitivity analysis (PSA) was used to assess simultaneous uncertainty in many variables [32]. To assess how simultaneous change of several variables affected the costs and outcomes, a Monte-Carlo simulation (1000 iterations per vaccine) was performed. This technique runs many simulations by repeatedly drawing samples from probability distributions of input variables. Univariate sensitivity analysis was also performed on the key parameters to observe their effect on the costs and outcomes. A ‘what-if’ scenario if vaccine wastage were reduced by 50% was tested to observe the effect of wastage on the optimal choice between the rotavirus vaccines. All analyses were performed using Microsoft Excel, 365.
3 Results
3.1 Costs of the Interventions
From the analysis, the adjusted vaccine cost of full immunization using ROTARIX was the highest, followed by ROTAVAC and ROTASIIL from start-up year to the year 2030. The immunization delivery cost for full immunization with ROTARIX was the lowest, with ROTASIIL having the highest delivery cost from start-up year to the year 2030. The total cost of full immunization per child was in favour of ROTARIX within the first 5 years, whereas the next 5 years favoured ROTAVAC. The median vaccine costs were $US2.66 (95% CI 2.61–2.71) for ROTARIX, $US2.03 (95% CI 1.99–2.07) for ROTAVAC, and $US1.89 (95% CI 1.86–1.92) for ROTASIIL. The median immunization delivery costs were $US2.14 (95% CI 2.10–2.18) for ROTARIX, $US2.82 (95% CI 2.77–2.87) for ROTAVAC, and $US3.14 (95% CI 3.08–3.20) for ROTASIIL. Thus, the overall cost was in favour of ROTARIX.
The total number of fully immunized children with ROTARIX was 42,439,371, which was higher than with ROTAVAC (42,418,928) and ROTASIIL (42,418,790). The total immunization cost over the 10 years was $US209,501,772 for ROTARIX, $US210,562,022 for ROTAVAC, and $US216,643,336 for ROTASIIL. The total immunization cost from the societal perspective shows a benefit of $US65 million from Gavi support during the transition phase. Details of the costs of the interventions are shown in Table 2 and the ESM.
3.2 Outcomes of the Interventions
The DALY averted per child was 0.0480 for ROTARIX, 0.0468 for ROTAVAC, and 0.0452 for ROTASIIL relative to ‘no vaccination’. The ICER of vaccination relative to ‘no vaccination’ was optimal with ROTARIX from the year 2021 to 2027 and optimal for ROTAVAC from the year 2028 to 2030. At no point within the time series was ROTASIIL the optimal choice; this was because of its higher delivery cost. The ICER of vaccination was $US100 (95% CI 71–130)/DALY averted for ROTARIX, $US104 (95% CI 78–126)/DALY averted for ROTAVAC, and $US111 (95% CI 87–134)/DALY averted for ROTASIIL. A cost-effectiveness threshold of 0.52 times the GDP per capita shows that the three vaccines were very cost effective. Valuation of a DALY averted at 1.3 times the GNI per capita yielded median BCRs of 27.00, 26.10, and 24.30 for ROTARIX, ROTAVAC, and ROTASIIL, respectively, per fully immunized child. Thus, the overall optimal choice within the 10 years was ROTARIX. The results also showed that the introduction of vaccination would avert 86 million cases of moderate RVGE, 337,000 cases of severe RVGE, and about 194,000 deaths within the 10 years. Table 2 provides details of the outcomes, and Fig. 2 shows the probability of ROTARIX cost effectiveness at the different willingness-to-pay thresholds.
3.3 Budget Impact
Given the low rate of care-seeking in Nigeria, only about 21.7 million cases of RVGE care-seeking at health facilities would be averted, which translates to an averted disease care cost of $US132.6 million using ROTARIX, which will offset 63% of the vaccination programme cost from the payer perspective. This presents a net rotavirus vaccination budget impact of $US76.9 million for the 10 years. The societal perspective, which included the cost of self-management, would lead to cost savings as it would offset the total vaccination programme cost. Details of the budget impact are shown in Table 2 and the ESM.
3.4 Decision to Switch Vaccine
Start-up of the vaccination programme with ROTARIX would be most beneficial to Nigeria in the early phase up to 7 years, after which ROTAVAC would become the optimal choice. Figure 3 shows the optimal vaccine each year and informs the decision of when to switch.
Wastage is a major dependent variable that can be adjusted during the programme. A what-if analysis of reducing the anticipated wastage of the three vaccines by 50% showed that ROTARIX would be the optimal choice from the year 2021 to 2023, whereas ROTAVAC would become the optimal choice from the year 2024 to 2030. Reduction in wastage would not make ROTASIIL an optimal choice between the three vaccines but would make it an optimal choice from the year 2027 if compared with ROTARIX alone. Figure 4 shows the effect of wastage reduction on the optimal choice.
3.5 Sensitivity Analyses
Figure 2 was developed from the PSA and shows that the results were insensitive to the parameters, which indicates the results are robust. The univariate sensitivity analysis shows that partial immunization would have a substantial impact on the result. Partial immunization has the potential to increase the ICER by 70%. Other parameters that can influence the outcome include the total cost of vaccination, transition probabilities of all-cause diarrhoea to death, and cost discount rate. Figure 5 shows the tornado diagram of the univariate sensitivity analysis.
4 Discussion
This study provides the costs, outcomes, and implications of decisions as Nigeria prepares to introduce a rotavirus vaccine and to transition out of Gavi support. It provides recommendations to support the decisions of health policymakers in the country. At this transition phase and beyond, every unit of cost saved and benefit gained becomes increasingly relevant to the country. Failure to make an optimal decision can affect healthcare supply, leading to market failure in the health system. The results show that the three vaccines would be very cost effective in Nigeria. ROTARIX would be the optimal choice for Nigeria in terms of costs and outcomes in the early phase. Implementing ROTARIX for the first 7 years and subsequently switching to ROTAVAC would save about $US3 million within the period of assessment.
These results indicate that ROTAVAC holds future promise as the optimal choice, but the feasibility of achieving this in Nigeria is narrow. Nigeria needs to improve its supply chain management practices to reduce wastage by about 50% to make ROTAVAC the optimal choice from the year 2024. An effort to reduce wastage will save costs for the country. Another challenge with ROTAVAC or ROTASIIL in Nigeria is the possibility of lower coverage because of low care-seeking [8,9,10]. It may be more feasible to administer two doses of ROTARIX than to administer three doses of ROTAVAC or ROTASIIL because of this low care-seeking. Thus, the use of ROTAVAC or ROTASIIL may result in lower coverage than use of ROTARIX. These results show that about 20,000 children will not be fully immunized with ROTAVAC or ROTASIIL compared with ROTARIX within the period of assessment, which could be higher if the possibility of lower care-seeking applies. Third, unlike for ROTARIX, no ROTAVAC or ROTASIIL manufacturers’ pricing commitments for transitioned countries exist [19]. Thus, it may not be possible for Nigeria to procure ROTAVAC or ROTASIIL at the UNICEF price from the year 2029, which could make ROTARIX the optimal choice after the transition.
Nigeria stands to lose about $US8 million every year (within the transition phase) from Gavi support if the introduction of rotavirus vaccination is delayed. It will be easier to introduce the vaccine since the country already has an existing national immunization program that can be leveraged. A major effort will be required to increase the cold chain capacity based on the estimated volume increase and to develop a sustainable system.
Long-term rotavirus vaccine pricing is a major determinant for the sustainability of the immunization programme. With the addition of two new rotavirus vaccines (ROTAVAC and ROTASIIL) to the UNICEF procurement list, price competition will promote sustainability [2]. Furthermore, the bovine–human reassortant rotavirus vaccine and the human neonatal rotavirus vaccine (RV3-BB) under development in Indonesia, India, China, and Brazil will help create a more competitive market and ensure price sustainability when the products are launched into the market after successful clinical trials [33]. The Global Vaccine Action Plan has emphasized the need to develop vaccine plants in Africa [34]. Africa represents about 14% of the world’s population but produces < 0.1% of the world’s vaccines [35]. By the year 2050, about 25% of the global population will live in Africa [35]. Currently, only five countries in Africa produce vaccines: Tunisia (limited Bacillus Calmette–Guerin and rabies vaccines), Senegal (yellow fever vaccine), Egypt (DTP), South Africa, and Ethiopia. Although the African Vaccine Manufacturers Initiative conducted a feasibility survey in 2015, implementing rotavirus vaccine manufacturing in Africa remains unlikely because of the challenges of economies of scale, financing and funding, the feasibility of sustaining manufacturing capacity, costs of development, and the cost effectiveness of vaccines relative to costs in India and China [35]. At the successful completion of the clinical trials of the vaccines under development, technology transfer to at least one vaccine plant in Africa could be beneficial to Nigeria and Africa at large in terms of vaccine cost, owing to the growing population in the continent.
The analysis has some limitations. Currently, no post-licensure vaccine effectiveness data are available for ROTAVAC and ROTASIIL, so we used efficacy data from phase III clinical trials for these vaccines. Use of post-licensure effectiveness data would have been more suitable. Second, the wastage rates used in the analysis were obtained using the WHO vaccine wastage rates calculator and do not represent actual wastage during implementation. Significant variations in wastage rates during implementation could affect the results of this study.
5 Conclusion
This study shows that introducing a rotavirus vaccine in Nigeria will be highly beneficial to the country. The programme should begin with ROTARIX and consider a switch to ROTAVAC after 7 years if cost-minimization measures are effective. The vaccine should be introduced as soon as possible to avert the opportunity costs of late introduction. Cost-minimization measures are imperative to ensure the sustainability of the programme in Nigeria after the transition.
Change history
09 February 2021
A Correction to this paper has been published: https://doi.org/10.1007/s41669-021-00258-7
References
Institute for Health Metrics and Evaluation. Global Burden of Disease Study 2019 (GBD 2019) Results. Seattle, United States: 2019. http://ghdx.healthdata.org/gbd-results-tool. Accessed 11 July 2020.
UNICEF Supply Division. Rotavirus vaccine: Supply and Demand update. 2020; February. https://www.unicef.org/supply/sites/unicef.org.supply/files/2020-03/rotavirus-vaccine-supply-and-demand-update_0.pdf. Accessed 11 July 2020.
GAVI. GAVI Co-financing Information: Nigeria. 2019. https://www.gavi.org/sites/default/files/document/co-financing-information-sheet-nigeriapdf.pdf. Accessed 6 Apr 2020.
Medic West Africa. 2019 Healthcare Market Insights: NIGERIA. 2019. https://www.medicwestafrica.com/content/dam/Informa/medic-west-africa/english/2019/HealthcareInsights.pdf. Accessed 15 Aug 2020.
United Nations. World Population Prospects 2019. 2019. https://population.un.org/wpp/Download/Standard/Population/. Accessed 5 May 2020.
Lamberti LM, Fischer Walker CL, Black RE. Systematic review of diarrhea duration and severity in children and adults in low- and middle-income countries. BMC Public Health. 2012;12:276. https://doi.org/10.1186/1471-2458-12-276.
WHO. Life Tables by Country. 2016. http://apps.who.int/gho/data/node.main.LIFECOUNTRY?lang=en. Accessed 15 Jun 2020.
Uchendu UO, Emodi IJ, Ikefuna AN. Pre-hospital management of diarrhoea among caregivers presenting at a tertiary health institution: implications for practice and health education. Afr Health Sci. 2011;11:41–7.
Ekpo O. Careseeking for childhood diarrhoea at the primary level of care in communities in Cross River State, Nigeria. J Epidemiol Glob Health. 2016;6:303–13. https://doi.org/10.1016/j.jegh.2016.08.002.
USAID/MCSP. Findings from the Enhancing Quality iCCM through Proprietary and Patent Medical Vendors (PPMV) and Partnerships (EQuiPP) Approach. 2019. https://www.childhealthtaskforce.org/sites/default/files/2019-08/MCSPEQuiPP%28PSESubgroupMeeting%29_07.30.2019.pdf. Accessed 10 Mar 2020.
World Health Organization. IMCI chart booklet. WHO. 2014. https://www.who.int/publications/i/item/9789241506823. Accessed 21 May 2020.
WHO/UNICEF. Vaccination Coverage Estimate: 1980–2018. 2018. https://apps.who.int/immunization_monitoring/globalsummary/timeseries/tswucoveragedtp3.html. Accessed 20 Aug 2020.
Jonesteller CL, Burnett E, Yen C, Tate JE, Parashar UD. Effectiveness of rotavirus vaccination: a systematic review of the first decade of global postlicensure data, 2006–2016. Clin Infect Dis. 2017;65:840–50.
Bhandari N, Rongsen-Chandola T, Bavdekar A, John J, Antony K, Taneja S, et al. Efficacy of a monovalent human-bovine (116E) rotavirus vaccine in Indian infants: a randomised, double-blind, placebo-controlled trial. Lancet. 2014;383:2136–43. https://doi.org/10.1016/S0140-6736(13)62630-6.
Kulkarni PS, Desai S, Tewari T, Kawade A, Goyal N, Garg BS, et al. A randomized Phase III clinical trial to assess the efficacy of a bovine-human reassortant pentavalent rotavirus vaccine in Indian infants. Vaccine. 2017;35:6228–37.
Isanaka S, Guindo O, Langendorf C, Matar Seck A, Plikaytis BD, Sayinzoga-Makombe N, et al. Efficacy of a low-cost, heat-stable oral rotavirus vaccine in Niger. N Engl J Med. 2017;376:1121–30.
Clark A, van Zandvoort K, Flasche S, Sanderson C, Bines J, Tate J, et al. Efficacy of live oral rotavirus vaccines by duration of follow-up: a meta-regression of randomised controlled trials. Lancet Infect Dis. 2019;19:717–27. https://doi.org/10.1016/S1473-3099(19)30126-4.
WHO | Vaccine management and logistics support. WHO. 2020. https://www.who.int/immunization/programmes_systems/supply_chain/resources/tools/en/. Accessed 28 June 2020.
GAVI. Gavi-supported rotavirus vaccines profiles to support country decision making. 2019. https://www.gavi.org/sites/default/files/document/rotavirus-vaccine-profilespdf.pdf. Accessed 10 June 2020.
Haacker M, Hallett TB, Atun R. On discount rates for economic evaluations in global health. Health Policy Plan. 2020;35:107–14. https://doi.org/10.1093/heapol/czz127.
GAVI. Co-financing policy. 2015;2015 12/06:1–6. https://www.gavi.org/programmes-impact/programmatic-policies/co-financing-policy. Accessed 19 June 2020.
UNICEF. Handling Fees. 2015. https://www.unicef.org/supply/handling-fees. Accessed 2 June 2020.
Immunization Costing Action Network (ICAN). Immunization Delivery Cost Catalogue. 2019. http://immunizationeconomics.org/ican-home. Accessed 5 June 2020.
WorldBank. Gross National Income per Capita. 2019. https://databank.worldbank.org/data/download/GNIPC.pdf. Accessed 10 Feb 2020.
World Health Organization Department of Vaccines and Biologicals. Guidelines for estimating costs of introducing new vaccines into the national immunization system. World Heal Organ. 2002;WHO/V&B/02.
National Assembly of the Federal Republic of Nigeria. National Health Insurance Scheme Drug Price List. 2013. http://www.nigeria-law.org/NationalHealthInsuranceSchemeDecree.htm. Accessed 23 Mar 2018.
WHO. Disability-Adjusted Life Year (DALY): Quantifying the Burden of Disease from mortality and morbidity. 2012. http://www.who.int/healthinfo/global_burden_disease/metrics_daly/en/. Accessed 1 Aug 2015.
WHO. Global Burden of Disease Concept. 2004. http://www.who.int/quantifying_ehimpacts/publications/en/9241546204chap3.pdf. Accessed 1 Aug 2015.
Robinson LA, Hammitt JK, O’Keeffe L. Valuing mortality risk reductions in global benefit-cost analysis. J Benefit Cost Anal. 2019;10:15–50. https://doi.org/10.1017/bca.2018.26.
Woods B, Revill P, Sculpher M, Claxton K. Country-Level Cost- Effectiveness Thresholds: Initial Estimates and the Need for Further Research. 2015. http://www.york.ac.uk/che/news/2015/che-research-paper-109/.
Barendregt JJ. The life table method of half cycle correction: getting it right. Med Decis Mak. 2014;34:283–5. https://doi.org/10.1177/0272989X13519863.
Briggs A, Claxton K, Sculpher M. Decision modelling for health economic evaluation. New York: Oxford University Press Inc; 2006. https://www.herc.ox.ac.uk/downloads/decision-modelling-for-health-economic-evaluation.
Kirkwood CD, Ma LF, Carey ME, Steele AD. The rotavirus vaccine development pipeline. Vaccine. 2019;37:7328–35. https://doi.org/10.1016/j.vaccine.2017.03.076.
WHO. Global Vaccine Action Plan. 2012. https://www.who.int/immunization/global_vaccine_action_plan/GVAP_doc_2011_2020/en/. Accessed 9 Apr 2020.
Ampofo W. Vaccine Manufacturing in Africa. Glob Vaccine Immun Res Forum. 2016. http://www.who.int/immunization/research/forums_and_initiatives/1_Wlliam_Ampofo_Vaccine_Manufacturing_Africa.pdf?ua=1.
Acknowledgements
The author is grateful to Bjørn Larsen, Nancy Dubosse, and Roland Mathiasson for their technical support during the project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Charles E. Okafor has no conflicts of interest that are directly relevant to the content of this article.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Code availability
The model is provided as a supplementary file.
Consent for publication
Not applicable.
Funding
Funding for this study was provided by the Copenhagen Consensus Center, USA Incorporated, in collaboration with the Bill & Melinda Gates Foundation. The results and conclusions contained are those of the author and do not necessarily reflect the position or policies of the Copenhagen Consensus Center or the Bill & Melinda Gates Foundation.
Availability of data and materials
The model used in this study was provided to the journal’s peer reviewers for their reference when reviewing the manuscript. The data used for the study are provided as a supplementary file.
Additional information
The original online version of this article was revised: In the Key Points for Decision Makers, the second point, should have read: “Early introduction of the vaccine will save the country about $US8 million per annum between 2021 and 2028 from Gavi support.”
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Okafor, C.E. Introducing Rotavirus Vaccination in Nigeria: Economic Evaluation and Implications. PharmacoEconomics Open 5, 545–557 (2021). https://doi.org/10.1007/s41669-020-00251-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41669-020-00251-6