Abstract
Objectives
The aim of this study was to evaluate the cost effectiveness of ambulatory blood pressure monitoring (ABPM) compared with home blood pressure monitoring (HBPM) and clinic blood pressure monitoring (CBPM) in diagnosing hypertension in Australia.
Methods
A cohort-based Markov model was built from the Payer’s perspective (Australian government) comparing lifetime costs and effectiveness of ABPM, HBPM and CBPM in people aged ≥ 35 years with suspected hypertension who have a CBPM between ≥ 140/90 mmHg and ≤ 180/110 mmHg using a sphygmomanometer and have not yet commenced antihypertensive treatment. The main outcome measures were incremental cost-effectiveness ratio (ICER) assessing cost per quality-adjusted life-year (QALY) and life-years (LYs) gained by ABPM versus HBPM and CBPM. Cost was measured in Australian dollars (A$).
Results
Over a lifetime model, ABPM had lower total costs (A$8,491) compared with HBPM (A$9,648) and CBPM (A$10,206) per person. ABPM was associated with a small but significant improvement in the quality and quantity of life for people with suspected hypertension with 12.872 QALYs and 17.449 LYs compared with 12.857 QALYs and 17.433 LYs with HBPM, and 12.850 QALYs and 17.425 LYs with CBPM. In the base-case analysis, ABPM dominated HBPM and CBPM. In scenario analyses, at 100% specificity of HBPM, ABPM no longer remained cost effective at a A$50,000/QALY threshold. However, in probabilistic sensitivity analysis, over 10,000 iterations, ABPM remained dominant.
Conclusion
ABPM was the dominant strategy for confirming the diagnosis of hypertension among Australian adults aged ≥ 35 years old with suspected hypertension. The findings of this study are important for reimbursement decision makers to support policy change and for clinicians to make practice changes consistent with ABPM recommendations in primary care.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Lifetime analyses of cost and quality and quantity of life estimates (QALYs and LYs) indicated ABPM was the most cost-effective strategy for confirming the diagnosis of hypertension among adults aged ≥ 35 years old with suspected hypertension in Australia. |
The results support practice change in primary care for diagnosing hypertension. |
1 Introduction
The National Health Survey, published by the Australian Bureau of Statistics, reports that one in three (34%) adults had high blood pressure in 2017–2018 [1]. Hypertension is a major risk factor for chronic conditions such as stroke, coronary heart disease, heart failure and chronic kidney disease [1]. In 2015, 5.8% of the total disease burden and 38% of cardiovascular disease burden were attributed to high blood pressure. It was ranked in Australia's top five risk factors causing the most burden [2].
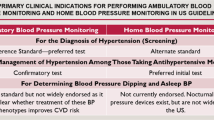
High blood pressure is defined by the National Heart Foundation of Australia guidelines as a clinic blood pressure monitoring (CBPM) results of ≥ 140/90 mmHg [3]. However, patients with suspected ‘white-coat’ hypertension with a reading of between ≥ 140/90 mmHg and ≤ 180/110 mmHg are known to give misleading results on CBPM. ‘White-coat’ hypertension relates to an elevated blood pressure reading above the normal range when measured with CBPM in the clinic but which is normal when measured using a portable device (ambulatory blood pressure monitoring [ABPM] or home blood pressure monitoring [HBPM]) [4]. ABPM is a portable monitor connected to a standard cuff on the upper arm and measures blood pressure at intervals of 20–30 minutes over a period of ≥ 24 hours, while HBPM involves a patient self-measuring blood pressure around the same time in the morning and evening at home for over 1 week using a validated and automated device [4].
In Australia, the current guidelines for diagnosing hypertension recommend ABPM and/or HBPM if CBPM is ≥ 140/90 mmHg [3]. Blood pressure measurements with ABPM and HBPM have a stronger association with cardiovascular outcomes than CBPM [3]. Furthermore, the guidelines from the United Kingdom (UK) and Canada recommend ABPM for patients with a CBPM of ≥ 140/90 mmHg for diagnosing hypertension in the case of suspected hypertension [5, 6]. ABPM and HBPM have better diagnostic accuracy than CBPM but may have higher costs to implement due to the cost of equipment or the clinician consultation service. Hence, cost-effectiveness analyses are essential before recommendations on device usage can be made in the Australian setting [7].
Internationally, studies have suggested that ABPM is more cost effective than HBPM or CBPM for adults with initially suspected hypertension [8, 9]. However, data are sparse for the long-term impact on costs and the impact of these diagnostic strategies in the Australian primary care setting to guide reimbursement decisions. A local economic evaluation is essential as the results of international studies do not apply directly to the Australian context for decision makers. This study aimed to model the long-term costs and health benefits of ABPM, HBPM and CBPM among adults suspected of hypertension from an Australian health system perspective.
2 Methods
The cost-effectiveness study was undertaken and reported according to best practice guidelines, using the Consolidated Health Economic Evaluation Reporting Standards (CHEERS) checklist (electronic supplementary material [ESM]) [10].
2.1 Literature Search
A literature search was performed on October 17, 2019, using OVID MEDLINE, EMBASE, NHS Economic Evaluation Database and the Health Technology Assessment (HTA) agency websites for studies published from January 1, 2012, to October 17, 2019. The National Institute for Health and Care Excellence (NICE) clinical guideline on Hypertension in Adults: Diagnosis and Management undertook a systematic review up to 2011 and an economic analysis to study the diagnostic accuracy and cost effectiveness of blood pressure monitoring methods for confirming hypertension [5]. The economic analysis as part of the guideline was published in 2011 [8]. The NICE systematic review was used as the foundation review; therefore, the timeframe for a review starting in 2012 was considered appropriate. A total of 486 citations were retrieved, and one relevant cost-utility analysis of ABPM versus CBPM and versus HBPM in the United States (US) population was found relevant [9]. On searching HTA agency websites, one Canadian HTA report was found relevant [11].
The US model had the same structure as the UK model, however it also included people who were not suspected of hypertension at initial clinic BP reading [9]. The Canadian model did not have HBPM as the comparator, and the structure did not represent all the cardiovascular health states [11]. Therefore, the UK model was appropriate in the Australian setting.
2.2 Model Overview
A cost-effectiveness analysis was undertaken comparing ABPM with (i) repeat CBPM using a validated and regularly maintained non-mercury sphygmomanometer taken during a consultation with a medical practitioner and (ii) multiple HBPM measurements (over a 7-day period) taken using a validated and automated device by patients in their home. The target population consisted of a hypothetical cohort of adults aged ≥ 35 years with suspected hypertension, with a clinic blood pressure measurement between ≥ 140/90 mmHg (Grade 1, mild hypertension) and ≤ 180/110 mmHg (Grade 2, moderate hypertension) using a sphygmomanometer, who had not commenced anti-hypertensive therapy. Adults with Grade 3 severe hypertension and blood pressure of ≥ 180/110 mmHg were excluded.
A standard cohort-based Markov model was developed comparing lifetime costs, life-years (LYs) and quality-adjusted life-years (QALYs) associated with different blood pressure monitoring approaches in the proposed population [12, 13]. The model was constructed in TreeAge Pro software (version 2021).
Figure 1 illustrates the main health states in the model and the possible transitions at regular intervals. Similar to the UK model, it was assumed in the Australian setting that it would take 3 months to confirm the diagnosis of hypertension with CBPM. Hence 3-monthly transitions (cycle length) were chosen [8]. The proposed population entering the model were adults with suspected hypertension (starting age 35 years), that is, they may be truly hypertensive or truly normotensive based on the first CBPM. The starting age of 35 years was chosen in the model as the proportion of people with hypertension increases with age, especially from the age of 35 years [8, 14].

Copyright © 2011, Elsevier Ltd. ABPM ambulatory blood pressure monitoring, CBPM clinic blood pressure monitoring, CHD coronary heart disease, HBPM home-based blood pressure monitoring, MI myocardial infarction, SA stable angina, TIA transient ischaemic attack, UA unstable angina
State-transition Markov model with all the health states. Individuals suspected of hypertension based on the initial CBPM reading undergo further test with either ABPM, HBPM or repeat CBPM. Based on the test results, their progression to different health states begins. Model structure adapted from Lovibond et al. with permission.
Upon entering the model, an individual received a blood pressure measurement using ABPM, HBPM or repeat CBPM. The individual then transitioned to a diagnosed health state (true positive, true negative, false positive, false negative for hypertension), non-fatal coronary heart diseases (i.e., stable angina, unstable angina, myocardial infarction), stroke, transient-ischaemic attack health states, or death based on the underlying model probabilities.
The model was run in repeated 3-monthly cycles for the individual’s lifetime, a total of 192 cycles (equivalent to 48 years), to calculate lifetime costs and QALYs. All the health states were attributed a specific cost and quality-of-life weight. Hence, the time spent in each health state was used to calculate the total costs and QALYs of ABPM, HBPM and CBPM. The model took the perspective of the Australian health care payer, with future costs and QALYs discounted at the standard rate of 5%, and does not include any indirect costs (e.g., productivity losses due to unemployment) [15]. The authors set a willingness-to-pay threshold of A$50,000 per QALY or LY to assess the incremental cost-effectiveness ratio (ICER). Although there is no single threshold per QALY in Australia, Lowe and Dyson (2013) suggested that previous decisions by the Pharmaceutical Benefits Advisory Committee (PBAC) indicate that the willingness to pay is around A$50,000 per QALY gained [16]. Furthermore, we conducted a sensitivity analysis in line with a recent paper recommending an empirical threshold of A$28,003 per QALY in Australia [17]. All the model inputs are listed in Table 1.
2.3 Model Mechanism and Assumptions
The transition probability to a true-positive, true-negative, false-positive or false-negative health state depended on the sensitivity and specificity of the blood pressure measurement test and the prevalence of hypertension (i.e., Grade 1 and Grade 2 hypertension at 35 years). The diagnostic accuracy of ABPM, HBPM and CBPM was based on a meta-analysis conducted which used ABPM as the reference standard (100% sensitive; 100% specific) [18]. We assumed ABPM may not always work correctly, therefore, in the base case, a 5% failure rate was considered, requiring repeat testing [8, 9]. It was assumed that HBPM would provide a successful diagnosis, and no failure rate was accounted for with HBPM [8, 9]. After diagnosis, an individual continued to remain in either a true-positive, true-negative, false-positive, or false-negative health state if they didn’t have a non-fatal coronary heart disease, stroke, transient ischaemic attack, or death.
For normotensive individuals (true negative and false positive), the transition probability to the hypertension health state depended on the age-specific probability of becoming hypertensive over subsequent cycles. Moreover, these individuals were assumed to receive only an accurate diagnosis (i.e., true positive) if they became hypertensive irrespective of the test used. This was a conservative assumption as these individuals may have received an incorrect diagnosis, especially if the test used was CBPM or HBPM.
For individuals diagnosed as false-negative, there was a lack of evidence on their next screening period for hypertension and the probability of receiving a correct diagnosis. Hence, in the model, 10% of individuals diagnosed as false-negative with HBPM and CBPM were assumed to undergo another test at each cycle (3-monthly) and received a confirmed true-positive diagnosis, transitioning to the hypertension health state. The remaining individuals continued to be false-negative until they transitioned to the hypertension health state in the subsequent cycles. The individuals continuing in the false-negative health state had a higher risk of cardiovascular disease because their true status of hypertension was not detected. The assumption of 10% of false-negative individuals receiving a correct diagnosis at each cycle was deemed appropriate as these individuals were expected to receive an accurate diagnosis at some point in their lifetime; this also avoided the complexity of re-introducing subsequent false-positive and false-negative test results. This was a conservative assumption (against ABPM) as HBPM and CBPM, if used again for screening, would have inferior diagnostic accuracy compared with ABPM.
The risk of non-fatal coronary heart diseases, stroke and the transient ischaemic attack were dependent on the age group of the individual and their diagnosed state (i.e., hypertensive or normotensive). It was assumed that anti-hypertensive treatment would affect the transition to these health states. To this end, age-specific relative risk reductions related to anti-hypertensive medications were taken from the literature and applied to the transition probabilities to the relevant health states [5, 8, 9, 19, 20].
It was assumed that all individuals diagnosed as true positive and false positive would commence anti-hypertensive medications and remain adherent to these medications for the time horizon. However, in the real world, it is likely that individuals may have moderate medication adherence, and their risk of cardiovascular events may be higher. It was also assumed that false-positive individuals taking anti-hypertensive medications would receive no health benefits, and their future risk of hypertension and cardiovascular disease events would be similar to that of the general population.
2.4 Death
The probability of death due to other causes was estimated using 2018–2020 Australian life tables [21]. Deaths related to coronary heart disease, stroke and transient ischaemic attack were calculated using the standardised mortality ratios obtained from best evidence sources [5, 8, 9, 22,23,24,25].
2.5 Quality of Life
Following best-practice methods, the quality of life or QALY weights for each health state reported in UK and US studies were multiplied by the age-specific Australian general population short-form six domains (SF-6D) index (0 = death and 1 = full health) to produce Australian utility values required to calculate QALYs [8, 9, 26,27,28,29,30,31]. The methodology of deriving utility weights used in these studies is described elsewhere [26, 28,29,30,31]. The quality-of-life weight for the health states in the model based on the estimates derived from the economic analysis performed in other studies with reference to the primary source is summarised in Table 1.
2.6 Costs
Healthcare use associated with modelled health states was obtained from studies published in Australia and internationally (NICE HTA) [32,33,34,35,36]. The costs related to healthcare resources were estimated from the Australian Government—subsidised, the Medicare Benefits Schedule (MBS, 2020), published economic evaluations and costing studies [32,33,34,35,36,37]. International studies’ costs were converted to 2020 Australian dollars (A$) and adjusted for purchasing power parity [38]. The studies reporting costs in Australian dollars were inflated to the 2019–2020 reference year using the appropriate health price index [39].
2.7 Modelling Uncertainty
A thorough and systematic approach was used to identify key drivers of the model. Deterministic one-way sensitivity analyses accounting for parameter uncertainty of key variables was conducted. Each variable was deviated by a constant proportion of ±10% to mitigate any issues relating to upper bound or lower bound values (such as proportions deviating above 100% or below 0%). We conducted a probabilistic sensitivity analysis using a second-order Monte-Carlo simulation with appropriate distribution for the parameters, which generated uncertainty in the deterministic analysis (Gamma distribution for costs, Beta distribution for utilities and diagnostic accuracy and lognormal distribution for standardised mortality ratio of the cardiovascular events). Precision estimates reported were used to propagate uncertainty. The uncertainty was propagated for cost variables by changing the base-case value by a fixed proportion of ± 20% as the precision estimates were unavailable. The input values in the model were randomly propagated across 10,000 simulations.
The impact of utility values was further explored using alternative values that may be appropriate in this patient population [40]. Furthermore, the effect of the starting age in the model, the model’s time horizon, the discount rate, failure rate of ABPM and the cost of HBPM and CBPM monitoring were also tested. Lastly, the assumption of 10% of false-negative patients receiving a confirmed true-positive diagnosis (on repeat testing) at each cycle was varied over the range of 0% to 20%.
3 Results
3.1 Base Case
Over a lifetime time horizon, the total direct costs associated with ABPM were A$8,491, and the costs associated with HBPM and CBPM were A$9,648 and A$10,206 per person, respectively. In terms of QALYs and LYs gained, ABPM showed superior effectiveness of 12.872 QALYs and 17.449 LYs gained compared with 12.857 QALYs and 17.433 LYs gained with HBPM; and 12.850 QALYs and 17.426 LYs gained with CBPM (Table 2).
ABPM was dominant with lower costs and higher effectiveness (higher quality and quantity of life measured in QALYs and LYs) compared with HBPM and CBPM in confirming the diagnosis of hypertension among adults aged ≥ 35 years old with suspected hypertension (Table 2). The cost effectiveness and incremental cost-effectiveness planes of ABPM versus HBPM and CBPM with base-case assumptions are shown in Figs. 2 and 3, respectively.
Cost-effectiveness plane. ABPM has the lowest costs and highest effectiveness and is the dominant strategy compared with HBPM and CBPM. ABPM ambulatory blood pressure monitoring, AU Australian dollars, CBPM clinic blood pressure monitoring, HBPM home blood pressure monitoring, QALYs quality-adjusted life-years
3.2 Sensitivity Analysis
Tables 3 and 4 show the results of deterministic one-way sensitivity analyses of the key variables. ABPM remained dominant with lower costs and higher QALY gains when any model parameter was changed by ± 10%. Furthermore, as shown in Table 5, in most of the scenarios tested, ABPM remained a cost-effective strategy. The exceptions to this were when the specificity of ABPM was assumed to be 80%. At this specificity, ABPM was dominant over CBPM, however, it was dominated by HBPM, and the most optimal strategy would be to use HBPM. Furthermore, on increasing the specificity of HBPM and CBPM to 100%, the ICER for ABPM was A$72,052 and A$48,013 per QALY, respectively. At a willingness to pay threshold of A$50,000 per QALY, ABPM no longer remained cost effective when the specificity of HBPM was similar to ABPM.
In probabilistic analysis, on changing parameters across 10,000 simulations, ABPM still had higher QALY gains than HBPM and CBPM. The total mean cost per patient with ABPM was A$8,966 (95% CI A$7,087–A$11,093) and QALYs of 13.087 (95% CI 12.946–13.226). The total mean costs per person were A$10,138 (95% CI A$7,667–A$13,328) and A$10,721 (95% CI A$7,767–A$15,271) with QALYs of 13.071 (95% CI 12.935–13.205) and 13.063 (95% CI 12.922–13.200) with HBPM and CBPM, respectively. At a willingness-to-pay threshold of A$28,003 and A$50,000, ABPM was an optimal strategy in 99% iterations, respectively (Fig. 4).
4 Discussion
This study assessed the cost effectiveness of ABPM, HBPM and CBPM over a lifetime model for individuals with initial raised blood pressure measured in the clinic from an Australian healthcare system’s perspective. Our study findings show that ABPM is the dominant strategy with lower costs and higher QALY gains than HBPM and CBPM. The conclusion is consistent and robust across a wide range of deterministic one-way sensitivity analyses and probabilistic sensitivity analyses of the key variables suggesting ABPM to be both cost-saving and generating more QALYs (0.002–0.03 QALYs) and LYs compared with HBPM and CBPM.
The assumption of the superior diagnostic accuracy of ABPM (100% sensitivity and 100% specificity) was the key driver as it resulted in zero misdiagnoses. In comparison, the inferior diagnostic accuracy of HBPM and CBPM led to the misdiagnosis of truly hypertensive individuals (false-negative), who thereby did not receive any benefit from the treatment as they were not prescribed anti-hypertension medications, putting them at increased risk of non-fatal cardiac events and death. Overall, it resulted in higher costs and lower QALYs with HBPM and CBPM. Furthermore, individuals who were truly normotensive but diagnosed as hypertensive (false-positive) with HBPM and CBPM received an unnecessary prescription of anti-hypertension medications, adding more costs. We understand the assumption of the perfect diagnostic accuracy of ABPM may be flawed; therefore, similar to the previous models, a 5% failure rate was allowed with ABPM to address any device failure requiring retesting and tested this assumption in the sensitivity analysis [8, 9]. Furthermore, a lower diagnostic accuracy was permitted in the sensitivity analysis. Reducing the diagnostic accuracy of the ABPM by 10% still resulted in cost savings and QALY gains, and it remained a dominant strategy. However, the conclusions in the model were sensitive to the variations in the specificity of the diagnostic tests, with HBPM becoming an optimal strategy at a specificity of 100% and when the specificity of ABPM was assumed to be 80%. Although the variation in the diagnostic accuracy is essential to capture, the NICE 2019 hypertension guideline agreed that ABPM remains the gold standard for the accurate measurement of blood pressure in primary care and retains it as the preferred method of diagnosing hypertension [18]. Therefore, using ABPM as the model's reference standard with 100% sensitivity and specificity was appropriate.
Unlike the previous economic models, we assumed that 10% of individuals diagnosed by CBPM and HBPM as false-negative would receive an accurate diagnosis (i.e., hypertensive) upon repeat testing within the same cycle. This was a conservative assumption (against ABPM) as these individuals may remain falsely diagnosed in the model for several years, thereby increasing the risk of cardiovascular disease events, subsequent healthcare costs and lowering their QALYs. However, this assumption was deemed appropriate to make the model simple and avoid the complexity of screening these individuals again. We conducted a series of sensitivity analyses to test this assumption, and ABPM remained a cost-effective strategy.
The cost-effectiveness model presented in this study is helpful for clinicians and policymakers to understand the incremental costs and benefits and whether any changes in parameters might apply to their patients and practices and further ABPM recommendations in their clinics. Similar to the results from our study, previous studies in the UK and US also suggested ABPM be the most cost-effective strategy for individuals suspected of hypertension and recommended using it before commencing anti-hypertension medications [8, 9]. In our study, we found modest gains in QALYs and LYs per individual undergoing ABPM versus HBPM and CBPM. However, these gains will likely become significant when the prevalent population suspected of hypertension is considered. Furthermore, a significant amount of cost savings is expected at a population level.
The strengths of our study are that we adopted a comprehensive and validated model published in other healthcare settings and included all relevant health states reflecting the patient progression over a lifetime. Furthermore, this is a unique Australian study that modelled longer-term costs and effects of all the diagnostic strategies relevant in the current clinical setting in the population suspected of hypertension who have a CBPM between ≥ 140/90 mmHg and ≤ 180/110 mmHg.
The results of our study should be interpreted in light of a few limitations. First, unlike the study in the US, where the starting age was 21 years, the population in our model was > 35 years of age. We did not model the younger population who might be at risk of white-coat hypertension. However, as per the Australian Bureau of Statistics, the proportion of people with hypertension increases with age, especially from the age of 35 years; hence, the model's starting age was 35 years [14]. Second, unlike the US study, we only included individuals who tested positive with initial CBPM (suspected of hypertension) and did not include those who tested negative. These individuals might have masked hypertension. The individuals with masked hypertension were out of the scope of the current study. In the US study, ABPM remained a cost-effective strategy despite including both white-coat hypertension and masked hypertension populations [9]. Third, we assumed all true-positive patients remained compliant to their anti-hypertension medications treatment regimen throughout the model; however, in the real world, patients may not remain adherent to the treatment. This may lead to uncontrolled blood pressure levels, increasing the risk of cardiovascular disease events. However, to keep the model simple and to reduce further uncertainty without better evidence on adherence rates, all true-positive patients receiving anti-hypertension treatment were modelled with a lower probability of cardiovascular disease events. Fourth, we assumed all false-positive patients will commence on the anti-hypertension medications but will not receive any benefit, and their risk of cardiovascular disease events will be similar to that of the general population. The blood pressure-lowering drugs may reduce the risk of cardiovascular disease events at a constant proportion irrespective of pre-treatment blood pressure [19]; however, it was not well established, and the UK and the US models used the same assumption. Fifth, similar to the model by Lovibond et al. (2011) [8], once a non-fatal cardiac event (e.g., stroke) occurred, an individual remained in that health state (post-stroke) until death. Our model did not account for repeat non-fatal cardiovascular events. While this approach is typical for pragmatic reasons, any additional healthcare resource utilisation and associated quality of life would not be accounted for in the model. Lastly, there are some applicability issues in our model that used utility weights and costs of health states from other countries. This included using utilities specific to cardiac events and quality-of-life multipliers from international studies, and health state costs (for initial stable angina and post-cardiovascular events) from the UK where costs were transformed to the Australian setting using the currency converter. These substitutes were needed as no Australian-specific data were available for these abovementioned measures.
Future health economic analysis would benefit from including a younger cohort, and individuals who failed to test positive on initial CBPM (masked hypertension). Furthermore, the availability of the Australian data on population-level risk factor prevalence (hypertension, diabetes mellitus, smoking and cholesterol) to estimate the risk of coronary heart disease and cerebrovascular disease (stroke and transient ischaemic attack), and although minor, Australian-specific post-coronary heart disease, stroke and transient ischaemic disease would improve the future analysis.
5 Conclusion
This study suggests that ABPM is the most cost-effective strategy for confirming the diagnosis of hypertension among adults suspected of hypertension, compared with HBPM and CBPM in Australia. The superior diagnostic accuracy of ABPM resulted in identifying truly hypertensive and normotensive individuals, thereby substantially saving costs due to avoidance of unnecessary hypertension treatment and future cardiovascular disease events. The findings of this study are important for reimbursement decision makers to support policy change and clinicians to make practice change consistent with ABPM recommendations in primary care.
Change history
31 October 2022
A Correction to this paper has been published: https://doi.org/10.1007/s41669-022-00379-7
References
Australian Institute of Health Welfare. High blood pressure. Canberra: AIHW; 2019.
Australian Institute of Health Welfare. Burden of disease. Canberra: AIHW; 2020.
Gabb GM, Mangoni AA, Anderson CS, Cowley D, Dowden JS, Golledge J, et al. Guideline for the diagnosis and management of hypertension in adults—2016. Med J Aust. 2016;205(2):85–9.
Sharman JE, Howes FS, Head GA, McGrath BP, Stowasser M, Schlaich M, et al. Home blood pressure monitoring: Australian expert consensus statement. J Hypertens. 2015;33(9):1721–8.
National Clinical Guideline Centre. Hypertension: the clinical managment of primary hypertension in adults: update of clinical guidelines 18 and 34. NICE clinical guideline 127. London: National Clinical Guideline Centre; 2011.
Nerenberg KA, Zarnke KB, Leung AA, Dasgupta K, Butalia S, McBrien K, et al. Hypertension Canada’s 2018 guidelines for diagnosis, risk assessment, prevention, and treatment of hypertension in adults and children. Can J Cardiol. 2018;34(5):506–25.
Hodgkinson J, Mant J, Martin U, Guo B, Hobbs FD, Deeks JJ, et al. Relative effectiveness of clinic and home blood pressure monitoring compared with ambulatory blood pressure monitoring in diagnosis of hypertension: systematic review. BMJ. 2011;342: d3621.
Lovibond K, Jowett S, Barton P, Caulfield M, Heneghan C, Hobbs FR, et al. Cost-effectiveness of options for the diagnosis of high blood pressure in primary care: a modelling study. The lancet. 2011;378(9798):1219–30.
Beyhaghi H, Viera AJ. Comparative cost-effectiveness of clinic, home, or ambulatory blood pressure measurement for hypertension diagnosis in US adults: a modeling study. Hypertension. 2019;73(1):121–31.
Husereau D, Drummond M, Petrou S, Carswell C, Moher D, Greenberg D, et al. Consolidated health economic evaluation reporting standards (CHEERS)—explanation and elaboration: a report of the ISPOR health economic evaluation publication guidelines good reporting practices task force. Value Health. 2013;16(2):231–50.
Health Quality Ontario. Twenty-four-hour ambulatory blood pressure monitoring in hypertension: an evidence-based analysis. Ontario health technology assessment series; 2012.
Whitehead SJ, Ali S. Health outcomes in economic evaluation: the QALY and utilities. Br Med Bull. 2010;96(1):5–21.
Briggs A, Sculpher M. An introduction to Markov modelling for economic evaluation. Pharmacoeconomics. 1998;13(4):397–409.
Australian Bureau of Statistics. National Health Survey: first results, 2017–18. ABS cat. no. 4364.0.55.001 Canberra; 2018.
Australian Department of Health. Guidelines for preparing submissions to the Pharmaceutical Benefits Advisory Committee. 2015.
Lowe A, Dyson S. New therapies for advanced cancers: can our society afford them? Is it ethical to deny patients access to them? 2013.
Edney LJP, News O. Empirical WTP $28 000 per QALY gained in Australia. 2018;794(1):14.
NICE. Hypertension in adults: diagnosis and management [A] Evidence review for diagnosis. NICE guideline NG136 Diagnostic evidence review underpinning recommendations 1.2.1 to 1.2.5 and 1.2.8 in the guideline. In: Excellence NIfHaC, editor. 2019.
Law M, Morris J, Wald N. Use of blood pressure lowering drugs in the prevention of cardiovascular disease: meta-analysis of 147 randomised trials in the context of expectations from prospective epidemiological studies. BMJ. 2009;338: b1665.
Craig R MJ. Joint Health Surveys Unit: health survey for England. 2006.
Australian Bureau of Statistics. 3302055001DO001_20182020 life tables, 2018–2020. 2021.
Rosengren A, Wilhelmsen L, Hagman M, Wedel H. Natural history of myocardial infarction and angina pectoris in a general population sample of middle-aged men: a 16-year follow-up of the Primary Prevention Study, Göteborg, Sweden. J Internal Med. 1998;244(6):495–505.
Brønnum-Hansen H, Davidsen M, Thorvaldsen P. Long-term survival and causes of death after stroke. Stroke. 2001;32(9):2131–6.
Dennis M, Bamford J, Sandercock P, Warlow C. Prognosis of transient ischemic attacks in the Oxfordshire Community Stroke Project. Stroke. 1990;21(6):848–53.
Brønnum-Hansen H, Jørgensen T, Davidsen M, Madsen M, Osler M, Gerdes LU, et al. Survival and cause of death after myocardial infarction: the Danish MONICA study. 2001;54(12):1244–50.
Ward S, Jones ML, Pandor A, Holmes M, Ara R, Ryan A, et al. A systematic review and economic evaluation of statins for the prevention of coronary events. NIHR health technology assessment programme: executive summaries: NIHR Journals Library; 2007.
Norman R, Church J, van den Berg B, Goodall S. Australian health-related quality of life population norms derived from the SF-6D. Aust N Z J Public Health. 2013;37(1):17–23.
Hutchins R, Pignone MP, Sheridan SL, Viera AJJBo. Quantifying the utility of taking pills for preventing adverse health outcomes: a cross-sectional survey. 2015;5(5):e006505.
Melsop KA, Boothroyd DB, Hlatky MAJAhj. Quality of life and time trade-off utility measures in patients with coronary artery disease. 2003;145(1):36–41.
Tengs TO, Lin THJP. A meta-analysis of quality-of-life estimates for stroke. 2003;21(3):191–200.
Goodacre S, Nicholl J, Dixon S, Cross E, Angelini K, Arnold J, et al. Randomised controlled trial and economic evaluation of a chest pain observation unit compared with routine care. 2004;328(7434):254.
Kaambwa B, Ratcliffe J, Horsfall M, Astley C, Karnon J, Coates P, et al. Cost effectiveness of high-sensitivity troponin compared to conventional troponin among patients presenting with undifferentiated chest pain: a trial based analysis. Int J Cardiol. 2017;238:144–50.
Ioannides-Demos LL, Makarounas-Kirchmann K, Ashton E, Stoelwinder J, McNeil JJ. Cost of myocardial infarction to the Australian community. Clin Drug Investig. 2010;30(8):533–43.
Deloitte Access Economics. Off beat: atrial fibrillation and the cost of preventable strokes. Australia; 2011.
Chowdhury EK, Reid CM, Zomer E, Kelly DJ, Liew D. Cost-effectiveness of renal denervation therapy for treatment-resistant hypertension: a best case scenario. Am J Hypertens. 2018;31(10):1156–63.
National Institute for Health and Care Excellence. Hypertension in adults: diagnosis and management. Cost-effectiveness analysis: Treatment initiation threshold for people with stage 1 hypertension. NICE clinical guideline 136. Economic analysis report. London: National Clinical Guideline Centre; 2019.
MBS Online: medicare benefits schedule [Internet]. 2020. http://www.mbsonline.gov.au/internet/mbsonline/publishing.nsf/Content/Home. Cited Jan 2020.
CCEMG – EPPI-Centre Cost Converter (version 1.6) [Internet]. 2020. https://eppi.ioe.ac.uk/costconversion/. Cited Dec 2019.
Australian Institute of Health and Welfare. Health expenditure Australia 2019–20. 2019.
McCaffrey N, Kaambwa B, Currow DC, Ratcliffe J. Health-related quality of life measured using the EQ-5D–5L: South Australian population norms. Health Qual Life Outcomes. 2016;14(1):1–12.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclaimer
This manuscript is based on a health technology assessment report commissioned by the Australian Government Department of Health: Diagnosis of hypertension using ambulatory blood pressure monitoring (ABPM) in individuals with clinic blood pressure ≥ 140/90 mmHg and ≤ 180/110 mmHg, MSAC Application 1572, Assessment Report (Commonwealth of Australia, Canberra, ACT). This manuscript was an independent assessment conducted by the authors, and the Australian Department of Health had no role in the study design, analysis and interpretation. All the opinions expressed solely belong to the authors or cited writers alone.
Source of funding
This manuscript is based on a health technology assessment report commissioned by the Australian Government Department of Health. The institution fully funds the lead author. The remaining authors declare that they have no other relevant financial interests.
Conflict of interest
None.
Author contributions
All authors contributed to the study’s conception and design. Karan K. Shah and Blaise Agresta performed material preparation, data collection and analysis. Karan K. Shah wrote the first draft of the manuscript and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Ethics approval
Not applicable.
Informed consent
Not applicable.
Consent for publication
Not applicable.
Data availability
The datasets generated during and/or analysed during the current study are not publicly available due to a confidentiality agreement with the Department of Health, but are available from the corresponding author on reasonable request.
Additional information
The original online version of this article was revised: There were errors in the figures
Supplementary Information
Below is the link to the electronic supplementary material.
41669_2022_364_MOESM1_ESM.pdf
CHEERS Checklist: Consolidated Health Economic Evaluation Reporting Standards for reporting economic evaluations (PDF 785 KB)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Shah, K.K., Willson, M., Agresta, B. et al. Cost Effectiveness of Ambulatory Blood Pressure Monitoring Compared with Home or Clinic Blood Pressure Monitoring for Diagnosing Hypertension in Australia. PharmacoEconomics Open 7, 49–62 (2023). https://doi.org/10.1007/s41669-022-00364-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41669-022-00364-0