Abstract
Background
Due to its antioxidant properties, vitamin C might be a promising and effective strategy for preventing postoperative atrial fibrillation (POAF) after cardiac surgery. This study was aimed at evaluating whether vitamin C supplementation is effective in reducing the cost of in-hospital postoperative medication used for patients undergoing coronary artery bypass (CABG) or valve surgery (±CABG).
Objective
The primary goal of this study was to evaluate the impact of perioperative vitamin C supplementation in patients undergoing cardiac surgery on in-hospital postoperative medication costs, while secondary endpoints were the effects on length of stay (LOS) in both the intensive care unit (ICU) and the hospital, and the incidence of POAF.
Material and Methods
From November 2018 to January 2021, 253 patients planned for CABG or valve surgery (±CABG) in AZ Maria Middelares, Ghent, Belgium, and who met the inclusion criteria (≥18 years of age, all having cardiac sinus rhythm, and who provided written informed consent) were randomised into a placebo group or vitamin C group. The medication was administered orally (1 g twice daily), starting from 5 days preoperatively until 10 days postoperatively. The medication used, LOS in the hospital/ICU, and development of clinically relevant POAF in the ICU were registered.
Results
Mean medication costs were €264.6 ± 98.1 for patients in the vitamin C group and €294.9 ± 267.3 for patients in the placebo group. When stratifying according to the type of surgery (CABG or valve surgery [±CABG]), these costs did not significantly differ. There was no significant difference in LOS or the incidence of clinically relevant POAF.
Conclusion
Our data did not identify any short-term financial impact on postoperative medication costs after oral perioperative vitamin C supplementation (1 g twice daily) for patients undergoing a CABG procedure or valve surgery (±CABG). No effect was found on the LOS or the incidence of clinically relevant POAF. Potential effects in the longer term, after intravenous administration of vitamin C or in other types of (cardiac) surgery, are still to be investigated.
Clinical Trials registration number
NCT03592680.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The economic impact of vitamin C supplementation on in-hospital medical costs after cardiac surgery is unclear. |
Oral vitamin C supplementation (1 g twice daily), 5 days before surgery until 10 days after surgery, had no effect on the postoperative medication costs for patients undergoing coronary artery bypass (CABG) or valve surgery (± CABG). No effect on the duration of postoperative stay in the intensive care unit or hospital, or on the incidence of clinically relevant postoperative atrial fibrillation, could be observed. |
The results do not indicate vitamin C supplementation as a short-term, effective, cost-saving strategy for patients undergoing CABG or valve surgery (± CABG). |
1 Introduction
Postoperative atrial fibrillation (POAF) is the most common arrhythmia after cardiac surgery [1]. The incidence of POAF depends on the type of cardiac surgery and is reported to be 20–50% for patients undergoing coronary artery bypass grafting (CABG) and about 60% for those undergoing combined CABG and valve surgery [2, 3].
POAF is considered a major problem in cardiac surgery due to its strong association with increased stroke rates, haemodynamic instability, increased thromboembolic events, uncontrolled heart rate and mortality [4, 5]. Consequently, as increased postoperative costs are associated with the occurrence of POAF, the financial burden of POAF in the overall costs of cardiac surgery is substantial [6,7,8]. Besides a prolonged length of stay (LOS) in the intensive care unit (ICU), the cost of postoperative medication used has also been reported as an important determinant of the higher postoperative costs found after POAF [4, 8, 9].
The search for novel therapeutic approaches to prevent or interfere with the development of POAF is ongoing. The pathophysiology of POAF is not yet totally elucidated but increasing evidence suggests a pivotal role for oxidative stress [10,11,12]. Several studies have already reported that the well-known antioxidant vitamin C is a promising and effective therapy for preventing POAF, however results remain inconsistent [13,14,15,16,17,18,19]. These previous reports did not evaluate the economic impact of vitamin C supplementation on in-hospital medical costs after cardiac surgery. Therefore, the primary aim of this study was to determine whether perioperative oral vitamin C intake could be a promising effective and cost-saving strategy for patients undergoing CABG or valve surgery (± CABG). The effect of perioperative oral vitamin C intake on the costs of in-hospital medication used, LOS in the ICU and the hospital, as well as the incidence of symptomatic POAF, was evaluated.
2 Material and Methods
2.1 Design
Patients scheduled to undergo elective CABG or valve surgery with or without CABG from November 2018 to January 2021 in AZ Maria Middelares, Ghent, Belgium, were screened for participation in this randomised, prospective, double-blind, placebo-controlled trial. The study protocol was in accordance with the Helsinki Declaration of the World Medical Association (2000) and was approved by the local Ethics Committee AZ Maria Middelares, Ghent, Belgium, and the central Ethics Committee UZA/UA, Antwerp, Belgium. The impact of perioperative vitamin C supplementation on costs of postoperative medication and clinical outcomes was studied over a short-term time horizon, i.e in-hospital stay.
2.2 Setting and Participants
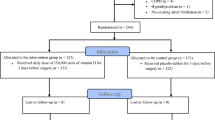
A total of 600 patients scheduled to undergo elective CABG or valve surgery (± CABG) were screened, of whom 253 entered the study. Included patients were ≥ 18 years of age, all having cardiac sinus rhythm, and who provided written informed consent. Patients on renal dialysis or taking supplementary vitamin C were excluded from the study. Before surgery, demographic and clinical characteristics such as age, sex, history of atrial fibrillation, type of surgery and risk factors were registered in the REDCap electronic data capture tool (Vanderbilt University, Nashville, TN, USA).
2.3 Randomisation and Intervention
Patients were randomly allocated to the placebo or vitamin C groups. Randomisation was performed by computer-generated random number sequence; the allocation was concealed in an envelope. Overall, 117 patients were randomly assigned to receive vitamin C (intervention group) and the remaining 136 patients were assigned to receive placebo (control group). Patients in the intervention group received 1 g of vitamin C twice daily starting from 5 days preoperatively until 10 days postoperatively. This dosing regimen was similar to dosing regimens used in prior studies [15,16,17, 19,20,21]. Patients in the placebo group received identical tablets containing lactose. Medication was administered orally, and both patient and surgeon were blinded to the medication administered. For all patients, premedication was on demand and was thus only taken when the patients asked for it. The anaesthesia protocol as well as the surgical procedure were performed according to the standard of care.
2.4 Outcomes and Follow-Up
In the postoperative period, patients were closely clinically evaluated and received standard of care. POAF was diagnosed with an electrocardiogram (ECG), which was taken continuously in the ICU. On the cardiac surgical ward, an ECG was prompted in the case of symptoms possibly related to POAF. Symptomatic POAF was defined as the presence of one or more of the following symptoms: palpitations, syncope, dyspnoea, chest pain, dizziness, and fatigue. All patients diagnosed with POAF were treated. As rhythm control is the favourable approach for POAF in this population, all patients diagnosed with POAF were treated with initial correction of electrolytes, if necessary, followed by amiodarone and anticoagulant therapy unless contraindicated. In the case of persistent and symptomatic POAF after 24 h of amiodarone administration, electrical cardioversion was performed.
2.5 Data Collection
To evaluate the impact of oral vitamin C supplementation on costs of in-hospital medication used, the postoperative medication costs were extracted from the pharmaceutical database of the hospital and expressed in Euros (valuation year, 2021). These data did not include the costs of vitamin C that was supplemented in the study. Costs of the used surgical materials were also not included in the analysed data as these are specific to the chosen type of surgical technique, which was determined by the surgeon. ICU and hospitalisation LOS were registered in the REDCap electronic data capture tool (Vanderbilt University).
2.6 Power Analysis
Due to the lack of a priori data, no reliable sample size calculation could be performed for the primary outcome (mean medication costs). Therefore, sample size was first calculated based on the incidence of POAF after cardiac surgery reported in the literature [16, 19, 21]: 30–50% in the placebo group, and a reduction of this risk of up to 30% after antioxidant supplementation. Considering the numerous confounders affecting the postoperative medication costs, a convenience sample size of 600 patients was set. To evaluate the study’s progress, an interim analysis was conducted after 50 patients were enrolled. Using the mean ± standard deviation (SD) of postoperative medication costs showed that 74 patients were required in each group to obtain a statistically significant alpha score of 0.05 and power of 85%.
2.7 Statistical Analysis
Continuous variables are reported as mean ± SD. Postoperative medication costs were analysed using a generalised linear model with a logarithmic link function and a gamma distribution, with treatment (placebo vs. vitamin C) and surgery type (CABG vs. valve surgery [± CABG]) as predictors. Confidence intervals (CIs) and p values were based on a permutation test. Additionally, a Mann–Whitney test was performed, stratified on surgery type. Time-to-event outcomes were analysed using a log-rank test stratified on surgery type. The binary outcome POAF was analysed using logistic regression, with treatment and surgery type as predictors. Analyses were performed in R 4.0 by Stat-Gent Crescendo, Ghent University, Ghent, Belgium. p values <0.05 were considered statistically significant.
3 Results
The study population consisted of 253 patients—136 in the placebo group and 117 in the vitamin C group. Both groups had comparable baseline and surgery-related characteristics, which are reported in Table 1.
3.1 Primary Endpoint
Mean postoperative medication costs for patients in the placebo group were €294.9 ± 267.3 and €264.6 ± 98.1 for patients in the vitamin C group (Table 2). Table 2 shows a breakdown of these costs for the placebo and vitamin C groups according to the Anatomical Therapeutic Chemical (ATC) class of the medication used. The main reason for the lower mean postoperative medication costs in the vitamin C group seems to be related to the costs of medication for the ‘Anti-infectives for systemic use’ and ‘Nervous system’ classifications. When stratifying patients according to the type of surgery, e.g. CABG or valve surgery (± CABG), no significant differences between the placebo and vitamin C groups could be observed for postoperative medication costs (cost ratio = 0.90; 95% CI 0.77–1.06; p = 0.31) [Fig. 1; p > 0.05].
Boxplot displaying the mean postoperative medication costs (€) for placebo- versus vitamin C-supplemented patients following CABG or valve surgery (± CABG). Vitamin C administration did not significantly affect mean postoperative medication costs compared with placebo administration for CABG patients or for patients undergoing valve surgery. Generalised linear model with a logarithmic link function and a gamma distribution, with treatment and surgery type as predictors (p > 0.05). CABG coronary artery bypass
3.2 Secondary Endpoints
The mean ICU LOS after cardiac surgery was 54.2 ± 44.8 h in the placebo group and 48.4 ± 35.2 h in the vitamin C group (Table 3). The log-rank test, stratified by surgery type, did not demonstrate a significant difference between the placebo and vitamin C groups (hazard ratio [HR] 1.24, 95% CI 0.96–1.59; p = 0.09). The Kaplan–Meier survival curves illustrate the LOS in the ICU (Fig. 2) for patients undergoing CABG or valve surgery (± CABG) in the placebo and vitamin C groups.
Kaplan–Meier curve showing LOS in the ICU (hours) for placebo- versus vitamin C-supplemented patients following CABG or valve surgery (± CABG). Vitamin C administration did not significantly decrease the mean LOS in the ICU. Log-rank test stratified by surgery type (p > 0.05). LOS length of stay, ICU intensive care unit, CABG coronary artery bypass
The mean hospital LOS after cardiac surgery was 8.1 ± 5.4 days in the placebo group and 8.0 ± 5.6 days in the vitamin C group (Table 3). The log-rank test, stratified by surgery type, did not demonstrate a significant difference in hospital LOS between the placebo and vitamin C groups (HR 1.09, 95% CI 0.85–1.4; p = 0.37). The Kaplan–Meier survival curves illustrate the LOS in hospital (Fig. 3) for patients undergoing CABG or valve surgery (± CABG) in the placebo and vitamin C groups.
Kaplan–Meier curve showing the LOS in hospital (days) for placebo- versus vitamin C-supplemented patients following CABG or valve surgery (±CABG). Vitamin C administration did not significantly decrease the mean LOS in the hospital. Log-rank test stratified by surgery type (p > 0.05). LOS length of stay, CABG coronary artery bypass
The incidence of clinically relevant POAF was 29%. In patients undergoing CABG surgery, POAF was diagnosed in 14 patients receiving placebo (20%) versus 12 patients receiving vitamin C (19%). In patients undergoing valve surgery (±CABG), POAF occurred in 26 patients receiving placebo (40%) versus 22 patients receiving vitamin C (41%). The logistic regression model did not show any benefit of vitamin C supplementation versus placebo on the incidence of POAF for patients with the same type of surgery (odds ratio 1.00, 95% CI 0.57–1.75; p > 0.05).
4 Discussion
POAF is one of the most common arrhythmias after cardiac surgery, with a substantial financial burden on the overall postoperative costs [4, 6, 7, 9]. It has been shown that one of the main sources of these higher costs associated with the occurrence of POAF is the cost of postoperatively used medication [8]. Although the underlying mechanism of POAF is multifactorial, growing evidence suggests a role of oxidative stress in the initiation and maintenance of atrial fibrillation, hence the large number of studies exploring antioxidant vitamins as both preventive and therapeutic agents. Indeed, many studies already investigated the antioxidant capacity of vitamin C and its potential in preventing POAF after cardiac surgery [15,16,17, 19,20,21,22,23]. Perioperative vitamin C supplementation is suggested to be a promising and well-tolerated option for patients undergoing cardiac surgery [16, 17, 19,20,21]; however, as previous studies did not focus on the impact of vitamin C intake on the short-term medical costs after cardiac surgery, it remains unclear whether prophylaxis with vitamin C may be an effective and cost-saving strategy in the prevention of clinically relevant POAF [24]. The present study is the first to assess whether perioperative oral vitamin C administration is effective in reducing in-hospital postoperative medication costs of patients undergoing CABG or valve surgery with or without CABG.
Patients were divided into two groups—one group received 1 g of vitamin C orally twice daily from day 5 before operation until day 10 after operation, while the other group received placebo during the same period. The mean cost of postoperative medication used was higher in the placebo group compared with the vitamin C group. This difference seemed to be caused by the higher need for medicines for the ‘Anti-infectives for systemic use’ and ‘Nervous system’ classifications in the placebo group. However, when stratifying according to type of surgery (CABG or valve surgery [±CABG]), no statistical difference in costs of the postoperatively used medication for patients in the intervention or placebo groups for both types of surgery could be observed. This result suggests no immediate financial impact of oral vitamin C supplementation on the cost of postoperatively used medication after cardiac surgery.
In this study, we primarily focused on the costs of postoperatively used medication and excluded any costs related to surgical materials. The latter costs are specific to the type of surgery and could be a 100-fold higher than the costs of medication. Consequently, including the costs of surgical materials could mask a potential financial benefit of vitamin C supplementation. In addition, surgical materials vary widely in price, without any documented influence on the incidence of POAF.
The effect of vitamin C supplementation on the ICU/hospital LOS has been extensively investigated previously, however contradictory results have been reported [15,16,17, 19, 22, 23]. In line with some of these studies, we were unable to demonstrate that oral vitamin C shortens ICU/hospital LOS and/or decreases the incidence of symptomatic POAF.
4.1 Limitations
This study has some limitations that should be acknowledged. First, the study only focused on the postoperative medication costs, therefore no firm conclusions could be drawn regarding the overall cost-effective potential of vitamin C intake for cardiac patients. Whether vitamin C should be recommended in daily therapeutic practice for these patients should be addressed in future research. Nonetheless, in this study, vitamin C intake did not alter costs or other outcomes such as LOS or the incidence of symptomatic POAF, therefore our results are a first indication that vitamin C supplementation has no short-term financial or clinical benefit for patients undergoing CABG or valve surgery (± CABG).
Second, it should be noted that this study only investigated the potential effect of oral vitamin C supplementation on postoperative medication costs during a short perioperative period (from 5 days before surgery until 10 days after surgery). POAF can be recurrent during the first weeks of the postoperative period [25], which implies that patients may have consulted medical services for postoperative complications later than the studied period. Therefore, although in this studied period no statistical differences were observed, potential significant differences of oral vitamin C administration in costs and outcomes over a longer time, for instance ranging from weeks to several months post operation, cannot be ruled out and should be investigated in future research.
Third, this study investigated the effect of oral (1 g twice daily) vitamin C administration. Plasma concentrations are limited after oral administration of vitamin C due to limited uptake capacity in the human bowel [15, 17, 19, 20, 26, 27], therefore the dose of vitamin C that was used in this study might have produced plasma concentrations that were too low to affect the incidence of POAF, costs or LOS. Nonetheless, similar oral doses have been reported to be effective in decreasing the incidence of POAF and/or LOS in the ICU [16, 17, 20], indicating that the dose of vitamin C that was used in this study could have been sufficient to observe the potential effects on medication costs and/or outcomes. Supplementing patients with higher oral doses would not have resulted in higher plasma concentrations as it was shown previously that peak plasma vitamin C concentrations are reached when 1.25 g of vitamin C was administered orally [27, 28]. Since intravenous administration can produce plasma concentrations 30- to 70-fold higher compared with what may be achieved by oral doses, possibly different results might have been seen when vitamin C was administered intravenously during the observed perioperative period. However, in this study, it was not an option to administer vitamin C intravenously to patients as this can only be performed in a hospital setting. Consequently, patients participating in this study should have been admitted to the hospital 5 days before the surgery to receive vitamin C or placebo. Similarly, after surgery, patients receiving intravenous treatment would be kept in the hospital longer, until 10 days post operation, solely for the administration of vitamin C or placebo. However, administering vitamin C or placebo intravenously during the time of hospital admission could have been an option but unfortunately this was not explored in the present study. As this study only investigated oral vitamin C administration, no conclusions on the effect of intravenous vitamin C on costs and clinical outcomes after cardiac surgery could be drawn and should be further examined.
Fourth, in contrast to previous trials, patients in this study were deliberately not monitored continuously, after being dismissed from the ICU, to detect cardiac arrhythmias. Since asymptomatic POAF patients might have been missed, the real incidence of POAF might be higher than the reported 29%. However, the aim of this study was to evaluate the impact of perioperative vitamin C supplementation on in-hospital medication costs after cardiac surgery rather than exploring the effect on POAF itself. Therefore, the option to treat only clinically relevant POAFs was chosen, as, in a non-trial setting, asymptomatic patients would not have been detected and hence not treated. Consequently, postoperative medication costs of asymptomatic POAF patients would not have been affected by whether or not POAF was present.
5 Conclusion
From our data, we were not able to identify any short-term financial impact of oral perioperative vitamin C supplementation (1 g twice daily) on postoperative medication costs for patients undergoing a CABG procedure or valve surgery (± CABG). No clinical effect on LOS in the hospital or ICU, or on the incidence of symptomatic POAF, could be observed. Since the potential effects of vitamin C supplementation (1) in the longer term, (2) after intravenous administration of vitamin C, and (3) in other types of (cardiac) surgery are still to be investigated, it is yet unclear whether vitamin C supplementation should be implemented in daily therapeutic routine for these patients.
References
Fuster V, Rydén LE, Cannom DS, Crijns HJ, Curtis AB, Ellenbogen KA, et al. ACC/AHA/ESC 2006 Guidelines for the Management of Patients with Atrial Fibrillation: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the European Society of Cardiology Committee for Practice Guidelines (Writing Committee to Revise the 2001 Guidelines for the Management of Patients With Atrial Fibrillation): developed in collaboration with the European Heart Rhythm Association and the Heart Rhythm Society. Circulation. 2006;114(7):e257-354.
Hakala T, Hedman A. Predicting the risk of atrial fibrillation after coronary artery bypass surgery. Scand Cardiovasc J. 2003;37(6):309–15.
Maisel WH, Rawn JD, Stevenson WG. Atrial fibrillation after cardiac surgery. Ann Intern Med. 2001;135(12):1061–73.
Omae T, Kanmura Y. Management of postoperative atrial fibrillation. J Anesth. 2012;26(3):429–37.
Shrivastava R, Smith B, Caskey D, Reddy P. Atrial fibrillation after cardiac surgery: does prophylactic therapy decrease adverse outcomes associated with atrial fibrillation. J Intensive Care Med. 2009;24(1):18–25.
Mauldin PD, Weintraub WS, Becker ER. Predicting hospital costs for first-time coronary artery bypass grafting from preoperative and postoperative variables. Am J Cardiol. 1994;74(8):772–5.
Taylor GJ, Mikell FL, Moses HW, Dove JT, Katholi RE, Malik SA, et al. Determinants of hospital charges for coronary artery bypass surgery: the economic consequences of postoperative complications. Am J Cardiol. 1990;65(5):309–13.
Hernández-Leiva E, Alvarado P, Dennis RJ. Postoperative atrial fibrillation: evaluation of its economic impact on the costs of cardiac surgery. Braz J Cardiovasc Surg. 2019;34(2):179–86.
LaPar DJ, Speir AM, Crosby IK, Fonner E Jr, Brown M, Rich JB, et al. Postoperative atrial fibrillation significantly increases mortality, hospital readmission, and hospital costs. Ann Thorac Surg. 2014;98(2):527–33 (discussion 33).
Boos CJ, Anderson RA, Lip GY. Is atrial fibrillation an inflammatory disorder? Eur Heart J. 2006;27(2):136–49.
Maesen B, Nijs J, Maessen J, Allessie M, Schotten U. Post-operative atrial fibrillation: a maze of mechanisms. Europace. 2012;14(2):159–74.
Zakkar M, Ascione R, James AF, Angelini GD, Suleiman MS. Inflammation, oxidative stress and postoperative atrial fibrillation in cardiac surgery. Pharmacol Ther. 2015;154:13–20.
Ali-Hassan-Sayegh S, Mirhosseini SJ, Rezaeisadrabadi M, Dehghan HR, Sedaghat-Hamedani F, Kayvanpour E, et al. Antioxidant supplementations for prevention of atrial fibrillation after cardiac surgery: an updated comprehensive systematic review and meta-analysis of 23 randomized controlled trials. Interact Cardiovasc Thorac Surg. 2014;18(5):646–54.
Baker WL, Coleman CI. Meta-analysis of ascorbic acid for prevention of postoperative atrial fibrillation after cardiac surgery. Am J Health Syst Pharm. 2016;73(24):2056–66.
Bjordahl PM, Helmer SD, Gosnell DJ, Wemmer GE, O’Hara WW, Milfeld DJ. Perioperative supplementation with ascorbic acid does not prevent atrial fibrillation in coronary artery bypass graft patients. Am J Surg. 2012;204(6):862–7 (discussion 7).
Dehghani MR, Majidi N, Rahmani A, Asgari B, Rezaei Y. Effect of oral vitamin C on atrial fibrillation development after isolated coronary artery bypass grafting surgery: a prospective randomized clinical trial. Cardiol J. 2014;21(5):492–9.
Eslami M, Badkoubeh RS, Mousavi M, Radmehr H, Salehi M, Tavakoli N, et al. Oral ascorbic acid in combination with beta-blockers is more effective than beta-blockers alone in the prevention of atrial fibrillation after coronary artery bypass grafting. Tex Heart Inst J. 2007;34(3):268–74.
Hemilä H, Suonsyrjä T. Vitamin C for preventing atrial fibrillation in high risk patients: a systematic review and meta-analysis. BMC Cardiovasc Disord. 2017;17(1):49.
Papoulidis P, Ananiadou O, Chalvatzoulis E, Ampatzidou F, Koutsogiannidis C, Karaiskos T, et al. The role of ascorbic acid in the prevention of atrial fibrillation after elective on-pump myocardial revascularization surgery: a single-center experience–a pilot study. Interact Cardiovasc Thorac Surg. 2011;12(2):121–4.
Carnes CA, Chung MK, Nakayama T, Nakayama H, Baliga RS, Piao S, et al. Ascorbate attenuates atrial pacing-induced peroxynitrite formation and electrical remodeling and decreases the incidence of postoperative atrial fibrillation. Circ Res. 2001;89(6):E32–8.
Rodrigo R, Korantzopoulos P, Cereceda M, Asenjo R, Zamorano J, Villalabeitia E, et al. A randomized controlled trial to prevent post-operative atrial fibrillation by antioxidant reinforcement. J Am Coll Cardiol. 2013;62(16):1457–65.
Mirmohammadsadeghi M, Mirmohammadsadeghi A, Mahmoudian M. Preventive use of ascorbic acid for atrial fibrillation after coronary artery bypass graft surgery. Heart Surg Forum. 2018;21(5):E415–7.
Sadeghpour A, Alizadehasl A, Kyavar M, Sadeghi T, Moludi J, Gholizadeh F, et al. Impact of vitamin C supplementation on post-cardiac surgery ICU and hospital length of stay. Anesth Pain Med. 2015;5(1): e25337.
Davis EM, Packard KA, Hilleman DE. Pharmacologic prophylaxis of postoperative atrial fibrillation in patients undergoing cardiac surgery: beyond beta-blockers. Pharmacotherapy. 2010;30(7):749 (274e–318e).
Ambrosetti M, Tramarin R, Griffo R, De Feo S, Fattirolli F, Vestri A, et al. Late postoperative atrial fibrillation after cardiac surgery: a national survey within the cardiac rehabilitation setting. J Cardiovasc Med (Hagerstown). 2011;12(6):390–5.
Orenes-Piñero E, Valdés M, Lip GY, Marín F. A comprehensive insight of novel antioxidant therapies for atrial fibrillation management. Drug Metab Rev. 2015;47(3):388–400.
Padayatty SJ, Sun H, Wang Y, Riordan HD, Hewitt SM, Katz A, et al. Vitamin C pharmacokinetics: implications for oral and intravenous use. Ann Intern Med. 2004;140(7):533–7.
Padayatty SJ, Levine M. New insights into the physiology and pharmacology of vitamin C. CMAJ. 2001;164(3):353–5.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
No funding was received for conducting this study.
Conflict of interest/competing interests
Jan Heerman, Charlotte Boydens, Silvie Allaert, Koen Cathenis, Koen Deryckere and Henk Vanoverschelde certify that they have no affiliations with or involvement in any organisation or entity with any financial or non-financial interest in the subject matter discussed in this manuscript.
Ethics approval
The study protocol was in accordance with the Helsinki Declaration of the World Medical Association (2000) and was approved by the local Ethics Committee AZ Maria Middelares, Ghent, Belgium, and the central Ethics Committee UZ Antwerpen/Universiteit Antwerpen, Antwerp, Belgium.
Consent to participate
Written informed consent was obtained from all included patients.
Consent to publish
Patients signed informed consent regarding publication of their data and figures.
Code availability
Not applicable.
Data availability
The corresponding author has full access to all the data in the study. Data sharing is available upon request.
Author contributions
JH and SA contributed to the study conception, design and data collection. JH and CB interpreted the results of the data analysis. CB drafted the manuscript, and JH assisted with writing and revising the manuscript. SA, KC, KD and HV assisted with revision of the manuscript. All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Heerman, J., Boydens, C., Allaert, S. et al. Effect of Perioperative Oral Vitamin C Supplementation on In-Hospital Postoperative Medication Costs for Cardiac Surgery Patients: A Prospective, Single-Centre, Randomised Clinical Trial. PharmacoEconomics Open 7, 655–663 (2023). https://doi.org/10.1007/s41669-023-00412-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41669-023-00412-3