Abstract
Abiotic stresses are the most major limitation to crop production, causing yield losses up to 50%, and consider the major challenges for production of crops. Thus, an experiment was conducted to determine how sugar beet would respond to soil application of K-humate rates and foliar application of biostimulants under salt stress. Split-plot design in three replicates was used in both seasons, where K-humate rates fertigation (control, 12, and 24 kg ha−1) allocated in the main plots, and different foliar application of BS arranged within the subplot (i.e., (1) water spray, (2) salicylic acid (SA) at the rate of 100 mg L−1, (3) fulvic acid (FA) at the rate of 1.2 kg ha−1, (4) hydroxyproline (HP) 1000 mg L−1, (5) SA at 100 mg L−1 + FA at 1.2 kg ha−1, (6) SA at 100 mg/L + HP at 1000 mg L−1, (7) FA at 1.2 kg ha−1 + HP at 1000 mg L−1, (8) SA 100 mg L−1 + FA 1.2 kg ha−1 + HP 1000 mg L−1) in the two seasons. The finding results revealed that soil application of K-humate, foliar application of BS, and their interaction significantly affected yield, growth, and quality characteristics of sugar beet under soil salinity, furthermore with soil application of K-humate at the rate of 24 kg ha−1 with foliar spray of FA + HP recorded the highest of growth traits, top, root, sucrose %, sugar yields/ha−1 and the lowest values of Na content in the juice and quality of sugar beet under the study conditions, Alexandria, Egypt.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Sugar beet is a member of the Amaranthaceae family and scientifically known as Beta vulgaris L. with a chromosome number (2n = 18). Sugar beet is regarded as a potential lucrative crop, second only to sugarcane (Mukherjee and Gantait, 2023), due to its various industrial uses. It is an essential crop for people since it provides them with high-quality energy and serves as a source of feed for household animals. In Egypt, sugar beet production accounted for 67.7% of total sugar production. Totally, 1.5 million ha of sugar beet is cultivated in Egypt, reaching an average tonnage of 48.0 t/ha−1 (FAOStat, 2021). Sugar beet can play a vital role in reducing production costs, decreasing crop periods, mitigating factor productivity reduction, and maintaining crop productivity at a higher level under abiotic stresses such as water and salt stress (Alkharabsheh et al. 2021).
Sugar beet is considered a good model for examining salt acclimation in crops (Lv et al. 2019). In other studies, sugar beet is a good crop model for studying salt tolerance mechanisms as it can withstand up to 500 mM of sodium chloride (NaCl) for a week without losing viability (Yang et al. 2012). However, sugar beet is relatively sensitive to salt during the early stages of germination and seedling growth, and also under prolonged exposure to high salt concentrations (Skorupa et al. 2019).
Humic substances (HS) such as potassium humate (K-humate) have been extensively researched for their potential use in agriculture and environmental remediation (de Melo et al. 2016). HS have been used to improve plant growth and nutrition, serve as soil bactericidal and plant fungicidal agents, and eliminate contaminants from water and soil (Rupiasih and Vidyasagar, 2005; Zavarzina et al. 2021). HA improves soil structure, enhances fertilizer efficiency, increases water-holding and cation-exchange capacity, and stimulates plant growth and root development (Hartwigsen and Evans, 2000). K-humate enhanced yield and quality. It accelerates germination by stimulating the membrane of seeds as well as metabolic activity. HS have a direct and indirect effect on plants, where they are widely utilized in agriculture as a soil addition and used to reduce slaty stress and improve growth and productivity of sugar beet as soil amendments (Mosaad et al. 2022). The use of organic compounds such as HA is one method for regulating various biochemical effects such as cell membrane permeability, photosynthetic rate, and cell elongation, as well as improving growth, yield, and water usage efficiency under stress (Khodadadi et al. 2020). According to Pulkrábek et al. (2001), the chlorophyll content is significantly positively correlated to the biomass and yield of sugar beet. The SPAD index (Soil and Plant Analysis Development) evaluated on sugar beet leaves and showed strongly positively linked with the sugar production, according to Ghasemi et al. (2017). Also, application of K improved growth, yield, and quality of sugar beet with/without stresses. The benefits of K on physiological and biochemical processes lead to a rise in the sugar beet plant’s production of storage roots and leaves (Neseim et al. 2014).
Biostimulants (BS) are products of biological origin that are designed to enhance plant productivity (Yakhin et al. 2017). These products are characterized chemically and non-chemically in order to be utilized in agriculture (Aremu et al. 2015). The use of biostimulants is now widely accepted as an economically and ecologically sustainable method of cultivating field crops (Castiglione et al. 2021; Rašovský et al. 2022). It is derived from natural sources, including FA, SA, and HP, which are a modern approach to plant-growing technologies and smart cultivation.
Fulvic acid (FA) is particularly effective due to its low molecular weight and ability to rapidly link minerals and elements into its molecular structure, leading them to resolve and create mobilized fulvic complexes (Boguta and Sokołowska, 2020). According to Elrys et al. (2020), under salt stress conditions, FA might increase the growth of the wheat by reducing the concentration of arousable oxygen species and enhancing the antioxidant protective mechanism. FA could significantly increase the availability of soil nutrients, decrease soil salinity, and stimulate plant development by interacting with oxides, hydroxides, metal ions, organic matter, and minerals in the environment (Braziene et al. 2021).
Salicylic acid (SA) is a phytohormone created through a series of chemical reactions (Maruri-López et al. 2019). SA is a defense mechanism against abiotic stress in tobacco plants as safe plant protector and growth regulator (Koo et al. 2020). It is vital for several physiological processes as a benzoic acid derivative, such as photosynthesis, nutrient intake, membrane permeability, and survival under diverse biotic and abiotic challenges. SA has been well shown a wide range of reactions in plants, including enhanced yield, increased plant tolerance capacity, and stem elongation under abiotic stress. Foliar application of SA protects plant from oxidative stress by enhancing antioxidant enzyme activity, resulting in higher fresh root and shoot masses of sugar beet plants (Tahjib-Ul-Arif et al. 2018). Also, SA improved growth, production, and quality of sugar beet under salt stress (Merwad, 2016). The top fresh mass and root biomass of sugar beet plants were significantly impacted by the foliar application of SA, while noticeable increase with high SA concentration, in the fresh mass, sugar yield, sucrose percentage, and purity percentage of sugar beet (El-Safy and Abo-Marzoka, 2021). In addition to reducing stress, SA enhanced sugar beetroot leaf biomass, plant height, photosynthetic pigment content, net photosynthetic rate, and PSII’s photochemical efficiency. Exogenous of SA preserved the integrity of the cell membrane by lowering the levels of malondialdehyde and electrolyte permeability and controlling the activity of antioxidant enzymes such as superoxide dismutase, peroxidase, catalase, and polyphenol (Miao et al. 2020).
Nevertheless, the supply of HA and SA significantly raised the antioxidant capacity and proline concentrations in sugar beet and enhanced the beet plant ability to withstand drought stress. Exogenously administered HA and SA improved sugar content, chlorophyll index, LAI, and RWC (Khodadadi et al. 2020). Applications of HA and SA significantly improved the photosynthesis, transpiration, and stomatal conductance characteristics and antioxidant activity (catalase, guaiacol peroxidase, and superoxide dismutase) of water starved maize plants. Foliar SA spray, followed by HA + SA treatment, significantly enhanced the yield and net benefit cost ratio of maize under water scarce circumstances. Foliar SA spray also raised the activity of enzymatic antioxidants and the effectiveness of photosynthetic antioxidants (Altaf et al. 2023).
Proline has a positive influence on sugar beets and increases the membrane integrity to endure osmotic stressors by decreasing reactive oxygen species (ROS) production and reactive oxygen species scavenging under abiotic stresses (Meena et al. 2019). Proline is considered a significant amino acid regarded as an osmoregulator, playing a key function in osmoregulatory mechanisms and improving growth and physiological characteristics such as chlorophyll index, relative water content, and yield, in addition, improving the water relations and sugar beet production under water stress (Ghaffari et al. 2021). The superior effect of proline may be attributed to its involvement in enhancing sugar beet as a storage sink for nutritious components such as carbon and nitrogen and as a scavenger for free radicals and reducing the detrimental effects of drought stress (AlKahtani et al. 2021).
The objective of the current study is to investigate how sugar beet root plants react to soil applications of K-humate and foliar applications of various biostimulants, as well as how they interact with one another, in salt affected soil conditions.
2 Material and Methods
Two field experiments were conducted out at the Experimental Farm, Faculty of Agriculture (Saba Basha), Alexandria University, Alexandria, Egypt, to study the response of sugar beet variety (Karam) to soil application of potassium humate (K-humate fertigation) and foliar application of some BS under salt affected soil during 2021/2022 and 2022/2023 seasons.
2.1 Type of Soil
Before planting, soil samples were collected at random from the experimental site at depths ranging from 0 to 60 cm below the soil surface and prepared for chemical analysis using the method described by Chapman and Pratt (1961). The results of the average of physical and chemical values of the experimental soil were clay loam texture, PH (1:1) = 8.23, EC (1:1) = 4.74 dS/m, organic matter = 1.45%, P = 3.55 mg/kg, total N = 1.21%, calcium carbonate = 7.15%, soluble cations (1:2) such as Na+ = 14.38; K+ = 1.59, soluble anions (1:2) such as CO−3 + HCO−3 = 2.58; CL− = 20.94; SO−4 = 16.75 cmol/kg soil.
2.2 Meteorological Data
The monthly average of the meteorological data noted during the cropping period (August to May) such as minimum temperature (16.29 °C), maximum temperature (24.2 °C), rainfall (18.7 mm), relative humidity (RH = 18.7%), wind (237.7 km/day), and sun (8.17 H) at Alexandria location (altitude: 32 m, latitude: 31.36, °N longitude: 29.95 °E, Egypt) according to the FAO database using CLIMWAT (Muñoz and Grieser, 2006). According to FAO, climate conditions and water management may hasten salinization. Evapotranspiration is critical in the pedogenesis of saline and sodic soils in arid lands. According to Wanjogu et al. (2001), most arid land gets less than 200 mm of rainfall/year, which results in salinization.
2.3 Experimental Design
The experimental design was a split-plot design with three replicates. The main plot was allocated by the soil application of three K-humate rates (0, 12, and 24 kg ha−1), while subplot occupied by foliar application of biostimulant treatments: (1) water spray (control), (2) SA 100 mg L−1, (3) FA 1.2 kg ha−1, (4) HP 1000 mg L−1, (5) SA 100 mg L−1 + FA 1.2 kg ha−1, (6) SA 100 mg L−1 + HP 1000 mg L−1, (7) FA 1.2 kg ha−1 + HP 1000 mg L−1, and (8) SA 100 mg/L + FA 1.2 kg ha−1 + HP 1000 mg L−1 in both seasons. The selection criteria for the biostimulants used in the study were based on their ability to enhance plant growth and development, improve plant tolerance to abiotic stress such as salinity, and increase crop yield and quality. The biostimulants were also chosen based on their composition, mode of action, and compatibility with the other soil amendments used in the study. The area of each subplot was 10.5 m2 as 3.5 m long and 3.0 m width (6 ridges width 50 cm); plant spacing was 20 cm.
2.4 Soil Preparation and Planting
The soil of the field experiments was well prepared through two plows, and leveling sugar beet cultivar poly germ (cv. Karam) obtained from Sugar Factory El Nile Company (Brand KWS – Type N – Resistance C). Seed balls were mechanized sown as usual on one side of the ridge in hills 20 cm apart at the rate of 3–4 seed balls per hill, sown in the following two growing seasons of 2021/2022 and 2022/2023. The plants were thinned once to one plant at 35 days from sowing.
2.5 Treatments
K-humate (KH) powder (C9H8K2O4, MW = 258.35 g mol−1, pH = 9–11) bought from Setra Company, Tanta, Egypt, under the marketing name HABICAR HUMICO WSP. It was applied after 50 DAS at the rate of 0, 12, and 24 kg KH ha−1 for plot (0, 12.6, and 25.2 g plot−1) as a soil application with irrigation water (fertigation). Commercial SA was purchased from Oasis Company–Egypt, Alexandria Desert Road, Egypt, whose commercial name is Anti-free (2-hydroxybenzoic acid, MW = 138.121 g mol−1, pH = 5.8), was prepared at a the rate of 100 mg L−1, and sprayed on the leaves twice after 65 and 90 days after sowing (DAS); at the same time, the control was sprayed with water. The commercial FA (MW = 308.24 g mol−1, pH = 6–7) was obtained from Setra Company, Egypt, whose trading name is Free Fulvic acid (70%), which sprayed twice at a rate of 1.2 kg ha−1 at 65 and 90 DAS; at the same time, control was sprayed with water. Finally, the industrial HP (C5H9NO3, MW = 131.131 g mol−1, pH=7) was bought from SAMA Company-Egypt, Alexandria Desert Road, Egypt, prepared at the rate of 1000 mg L−1, and sprayed two times after 65 and 90 DAS. At the same time, the control was sprayed with tap water. The other agricultural practices for cultivating sugar beet were followed, as recommended by the Ministry of Agriculture and Land Reclamation, Egypt. The application of FA, SA, and HP and their interaction was conducted in a liquid form using a backpack sprayer (foliar application) on sugar beet plants. This was done two times, with foliar spraying done at sunset to avoid damage from strong sunlight and high temperatures. The application of the treatments was done at the rate of 750 L ha−1, with each plot receiving 1.2 L time−1.
2.6 Studied Characteristics
2.6.1 Yield and its Components
After 200 days from sowing (maturity stage), the sugar beet plants were harvested from the three middle ridges of each subplot, and the yield parameters and its components were estimated as follows: root yield (t ha−1); top yield (t ha−1); biological yield (t ha−1); sucrose percentage (%), and sugar yield (SY) t ha−1 was calculated according to the following equation. SY = root yield t ha−1 × sucrose/100.
2.6.2 Growth Characters
Root length and diameter (cm) were measured on roots from 10 plants picked at random from inside the second ridge of each subplot avoiding the border ridge. To determine the leaf area index (LAI) of sugar beet leaves, leaf blades were collected and arranged on a sheet of paper with a black rectangle, photographed them using a digital camera and processed the photos with ImageJ software. The average distance between the camera and the leaf was 50 cm. To calculate LA, unnecessary objects were removed, set the scale, adjusted contrast, and computed the leaf area using the “wand” tool to select the leaf and calculate LAI = LA (cm2)/land area (cm2) (Ereqat et al. 2018; Martin et al. 2020). Total chlorophyll index: green color degree (SPAD unit), determined by chlorophyll meter (SPAD-502, Minolta Co., Japan) and represented by SPAD value as an average of ten random leaves from each subplot at 90 DAS, according to the method described by Uddling et al. (2007).
2.6.3 Quality Parameters
Samples of ten sugar beet roots were taken randomly from each subplot, washed, and dried to calculate the following parameters: sucrose percentage according to the method of McGinnis (1971). Recoverable sugar percentage (RS %) was determined as R.S.% = Sucrose % − D% (Hoffmann, 2010); extracted sugar (%); total soluble solids (TSS %) was recorded in the juice of fresh roots using a hand refractometer; sucrose% was measured polarimetrically on a lead acetate extract of fresh macerated root (Carruthers and Oldfield, 1961); juice purity percentage (QZ) was calculated using QZ = (extracted sugar %/sucrose %) × 100; potassium (K+), sodium (Na+), and α-amino N content in the roots were assessed as meq/100 g sugar beet root, were determined by an automated analyzer as described by Carruthers and Oldfield (1961) and Hoffmann (2010) and K+/Na+ ratio were measured.
2.6.4 Economic Analysis
To alleviate the negative impacts of salt stress, an economic analysis was performed to compare the outcomes and advantages of soil application of K-humate (as a source of HA) and foliar application of SA, FA, and HP. KH rates were 0, 12, and 24 kg ha−1, while SA, FA, and HP foliar applications were 100 mg L−1, 1.2 kg ha−1, and 1000 mg L−1, respectively. In the first and second seasons, respectively, the cost of K-humate as a source of HA was 0, 1440, and 2880 LE (Egyptian pounds per hectare) and 0, 1680, and 3360 LE (Egyptian pounds per hectare), while the cost of twice foliar application of SA, FA, and HP was 370, 576, and 672 LE and 420, 691, and 902 LE, respectively. The fixed cost includes land preparation, seed sowing, fertilizer application, insecticide, herbicide application, and harvesting. The gross income was calculated using the average marketing price of sugar beetroot in Egypt in the first season, which was 800 LE (43.24 $), and 1500 LE (49.18 $) in the second season.
Total production costs (TPC) was calculated in Egyptian pounds at the local market price.
Gross income (GI) = total from selling sugar beet crop.
Net return (NR) = gross income (GI) − total production costs (TPC).
Benefit cost ratio (BCR) = NR/TPC.
2.7 Statistical Analysis
All recorded data were statistically analyzed according to the technique of analysis of variance (ANOVA) for the split-plot design as published by Gomez and Gomez (1984). The least significant difference (LSD) method was used to evaluate the differences between treatment means at 5% level of probability. All the statistical analyses were performed application of CoStat (2005) for Windows.
3 Results
In general, the findings indicated that the combination of KH rates and foliar application of BS had a significant impact on various parameters of sugar beet, including root yield, top yield, biological yield, sugar yield, sucrose percentage, root length, LAI, chlorophyll index, RS%, extracted sugar, TSS%, purity, K%, Na%, K/N ratio, and α-amino N% in salinity-affected soil during the 2021/2022 and 2022/2023 growing seasons.
3.1 Effect of K-Humate Rates and BS Application on Sugar Beet
3.1.1 Effect of K-Humate
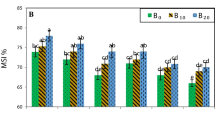
The results in Table 1 showed a significant effect of K-humate as a source of HA on the yield characteristics, where increasing K-humate rates from 0 up to 24 kg ha−1 caused an increase and recorded the highest mean values of root yield (66.64 and 67.85 t ha−1), top yield (30.10 and 30.83 t ha−1), biological yield (96.74 and 98.69 t ha−1), sugar yield (12.30 and 12.70 t ha−1), and sucrose percentage (18.87 and 19.49%), while the lowest mean values of the previous mentioned traits were obtained by control treatment (untreated) in the cropping seasons, respectively. Also, results in Table 2 revealed that the highest value of root length (cm) was given with application of 12 or 24 kg KH. On the other hand, the maximum values of root diameter (cm), leaf area index (LAI), and chlorophyll index (SPAD unit) were achieved with soil application of KH at the rate of 24 kg ha−1 followed by a rate of 12 kg ha−1, while the control treatment (0 kg KH ha−1) gave the minimum value of these characters in the two growing seasons. Data in Table 3 revealed that application of 12 or 24 kg KH ha−1 attained the highest values of recoverable sugar (RS%), extracted sugar (%), and total soluble solids (TSS %), while purity (%) did not differ under the various rates of KH application; on the other side, control (untreated) recorded the lowest values of these traits in both seasons. Application of KH at 24 kg ha−1 reduced the impurity characters such as potassium (K %), α-amino N (%), sodium (Na %), and K+/N+ ratio, followed by soil application of KH at the rate of 12 kg KH ha−1 in comparison with the control treatment as shown in Table 4.
3.1.2 Effect of BS
Concerning the effect of different BS, the results showed that foliar treatments of FA at the rate of 1.2 kg ha−1 + HP at 1000 mg L−1 attained the maximum values of root yield (64.76 and 66.37 t ha−1) and sugar yield (11.84 and 94.29 t ha−1), while using FA application increased top yield (32.32 and 31.15 t ha−1) and biological yield (95.95 and 96.77 t ha−1), while the highest percent of sucrose (18.58 and 19.51%, 18.57 and 19.30%) were indicated with foliar application of HP + SA and HP + FA, respectively. On the other hand, the minimum values of these traits were recorded with the application of water spray (control treatment) under soil affected by salinity in both seasons (Table 1). The presented results in Table 2 revealed that the maximum values of root length (cm) and leaf chlorophyll index (SPAD) were achieved with foliar application of FA + HP; on the other hand, the highest value of root diameter was given with SA + FA + HP, while the highest leaf area index (LAI) was given with HP, as compared with the other treatments, while application of water gave the minimum value of the previous mentioned characters. Application of SA + HP or FA + HP recorded the highest values of recoverable sugar (RS%), extracted sugar (B%), and TSS %, beside purity (%), comparing with the other treatments; on the other hand, water spray recorded the lowest value of these traits (Table 3). Foliar treatment of FA + HP increased potassium (K %) which decreased α-amino N (%), sodium (Na %), and K+/N+ ratio in comparison with the others treatment, while the highest values recorded with water spray as shown in Table 4.
3.2 Interaction Effect Between K-Humate and Application of BS on Sugar Beet Characters
Application of 24 kg K-humate/ha−1 with foliar application of FA + HP recorded the maximum value of root yield (70.92 t ha−1) in the first season with LSD at 5% being 1.54, but the highest yield in the second season with LSD value was 3.45, the highest value (72.96 t ha−1) was attained with 12 kg KH + SA + FA, while the maximum sucrose percent (20.08 and 21.03%) was achieved with application of 24 kg ha−1 from soil application of KH with foliar application of FA + HP, respectively, at LSD5% = 0.64 and 0.71 in the first and second seasons, respectively. On the other hand, the application of 24 kg K-humate with FA achieved the highest top yield (36.59 t ha−1) in the first season and 12 kg KH + SA was 35.56 t ha−1 in the second season and 24 kg K-humate + FA gave the highest biological yield (105.69 and 105.46 t ha−1), while the highest sugar yield (13.59 and 14.13 t ha−1) was recorded with using 24 kg K-humate with FA + SA or HP + FA, respectively, as compared to the other treatments, while the lowest values of these traits were given with control (untreated + water spray) as presented in Table 1. Results in Table 2 revealed that soil application of 24 kg KH ha−1 with foliar application of FA recorded the maximum values of root length in the first season with LSD = 3.23, while in the second season the longest roots were obtained with 0 kg K-humate + FA + HP with LSD = 3.38; on the other hand, 12 kg KH ha−1 with SA + FA + HP gave the largest values of root diameter, while 24 kg KH ha−1 + HP gave the maximum LAI; in addition, the maximum value of chlorophyll index was with application of 24 kg K-humate as source of HA with FA in comparison with the other treatments, while the minimum ones were given with control (untreated + water spray) in both seasons. From Table 3, the results indicated that foliar application of FA + HP or their combination among its caused a significant increase under 24 kg of KH ha−1 in sugar beet quality, recoverable sugar (RS%), and extracted sugar (%) as well as TSS, while the highest juice purity (%) recorded with 0 K-humate + HP application and using a higher rate of 24 kg KH ha−1 + SA + FA increased purity (%), while the lowest ones were given with untreated + water spray in the two seasons. Also, foliar treatments of BS such as + FA + HP + SA under soil application of 12 or 24 kg KH ha−1 decreased potassium (K%), sodium (Na %), K/N ratio, and α-amino N (%) of sugar beet as grown in soil affected by salinity (Table 4).
3.3 Relationship Between Treatments and the Parameters Studied and Correlation Analysis
A correlation analysis was conducted to investigate the interrelationship between physiological and biochemical parameters of sugar beet sown in soil affected by salts. The Data in Fig. 1 suggests that there is a complex interrelationship between physiological and biochemical parameters of sugar beet plants grown under salinity conditions. Pearson’s correlation analysis shows that several parameters have a significant positive correlation with each other. These include root yield, top yield, biological yield, root length, sugar yield, sucrose %, leaf area index, chlorophyll index, recoverable sugar percentage, alkalinity percentage, juice purity, and TSS %. This shows that as these parameters increase or decrease, they are likely to influence each other in a positive way. For example, an increase in leaf area index, which is a measure of the total area of leaves per unit ground area, is likely to lead to an increase in photosynthesis, which can result in higher sugar yields and sucrose content. On the other hand, root diameter shows a negative correlation with all the studied parameters. This suggests that as root diameter increases, there may be a decrease in the other parameters. This may be due to a trade-off between root growth and other plant processes, such as photosynthesis and sugar accumulation. The positive correlation between sodium and α-amino nitrogen is also notable. This may be since salinity stress can lead to an increase in sodium uptake, which can affect the metabolism of amino acids and other nitrogen-containing compounds. Overall, the results suggest that there are complex interrelationships among the physiological and biochemical parameters of sugar beet plants under salinity conditions, and further research is needed to fully understand these relationships and their implications for plant growth and productivity.
Pearson’s correlation coefficients between the traits. White color indicates non-significant correlations and dark color indicates significant correlation by t-test the 5% of probability. Where biological yield (BY), sugar yield (SY), root length (RL), root diameter (RD), leaf area index (LAI), total chlorophyll index (Chl), recoverable sugar (Rs), extracted sugar (Es %), total soluble solids (TSS %), juice purity (JP%), potassium (K %), sodium (Na %), α-amino (N%), alkalinity (Alk%)
The heat map presented in the current study supplies a visual representation of the impact of different applications of treatments on the morpho-physiological and biochemical parameters of sugar beet plants. The hierarchical clustering analysis used in conjunction with the heat map allows for easy identification of treatment groups that have similar effects on the measured parameters. The results of the hierarchical clustering analysis show that the control and soil application of 12 kg K-humate are clearly separated from the other treatments, showing that these two treatments have the lowest impact on the measured parameters. This suggests that the application of K-humate treatments is necessary to improve the growth and productivity of sugar beet plants. Among the treatments, 24 kg K-humate as soil application + foliar application of FA + HP and soil application of 24 kg K-humate + foliar spray of SA + HP gave the highest values for the measured parameters, except sodium and α-amino nitrogen. This shows that the application of these combinations can significantly improve the morpho-physiological and biochemical parameters of sugar beet plants. Spray of HP, soil application of 12 kg K-humate + foliar spray of SA + HP, and FA + HP were clustered together, showing that these treatments have similar effects on the measured parameters. The main differences between these three treatments were due to the increase in root diameter and juice purity. Similarly, soil application of 24 kg K-humate + foliar application of SA + FA and soil application of 24 kg K-humate + spray of FA were clustered together, showing that these two treatments have similar effects on the measured parameters. The primary differences between these two treatments were increased biological yield, root yield, sugar yield, and juice purity. Overall, the heat map and hierarchical clustering analysis results give useful insights into the impacts of various treatments on the development and production of sugar beet plants and may be used to influence future agricultural practices (Fig. 2).
3.4 Regression Analysis
Figure 3 shows the regression coefficients for all characteristics as well as the regression coefficient of determination (R2). Figure 3 depicts the visual dependency of root yield (RY) on major yield-related factors. Sugar yield (SY) had the greatest coefficient of determination (0.95), followed by biological yield (BY) (0.94) and root diameter (RD) (0.89), while root length percent had the lowest (0.12), followed by juice purity (0.23). The regression coefficient of RY for chlorophyll index and LAI revealed that one unit change in chlorophyll index and LAI induced a 69% and 65% change in root yield (the dependent variable), respectively (increase or decrease). The application of various treatments, such as K-humate, biostimulants, and their combinations, improved the net benefit-cost ratio (BCR) as shown in Table 5; however, the control (untreated treatment or untreated + water spray) resulted in less net income, whereas HA + SA + FA application was found to be more economical for improving sugar beet yield under salt stress conditions (Table 5).
4 Discussion
In general, the current study showed that KH rate, foliar application of BS, and their interactions had a substantial impact on sugar beetroot characteristics, growth, yield, and quality under salt conditions. These beneficial effects are due to the vital role of these materials in increasing plant growth, yield, and quality, where we discovered that increasing KH as a potassium (K) source from 12 to 24 kg ha−1 increased all yield attributes and quality characters of sugar beetroot, which may be due to the role of K, where K is an essential element for plant growth with respect to its physiological and biochemical functions. Many studies in sugar beet reported that its required for the starch synthetase enzyme to be activated, protein synthesis, photosynthesis, osmoregulation, stomatal conductance, energy transfer, phloem transport, cation-anion balance, and stress tolerance (Milford et al. 2000; Mubarak et al. 2016; El-Mageed et al. 2022).
In the current data, there is higher top and root yield due to improved vegetative development, such as LAI and chlorophyll index, which resulted in increased top and root production/ha. Sugar yield may have increased because of an increase in root yield and quality; also, the increase in root length and diameter produced by higher KH rates may be associated with enhanced cell elongation and cell division, resulting in increased root length (Badawi et al. 2013). Increasing KH and BS increases root yield and sugar yield due to their favorable impacts on chlorophyll index, LAI, top yield, TSS%, and sucrose%. This generated the highest sugar yield/ha−1 since sugar yield/ha is a function of root yield multiplied by sucrose percent; these findings are consistent with those of Moradzadeh et al. (2021) and Hemati et al. (2022). This improvement in sugar beet growth characteristics by increasing K-humate rate can be attributed to its influence on providing plants and soil with a concentrated dose of vital minerals, vitamins, and trace elements, which improves sugar beet growth and leaf canopy. K treatment increased root production, top yield, sugar yield, sugar content, and other qualitative characteristics of sugar beetroot crops, as shown by Ibrahim et al. (2020).
Improvement in sugar beet growth characteristics by raising K-humate rate can be due to its influence on giving plant and soil with a concentrated dosage of vital minerals, vitamins, and trace elements (Ayuso et al. 1996), which improves sugar beet growth and leaf canopy. Like our observations, K treatment boosted root production, top yield, sugar yield, sugar content, and other qualitative features of sugar beet crops (Zare Abyaneh et al. 2017; Ibrahim et al. 2020). Furthermore, using of K-humate in the current study has been shown to significantly improve plant growth and the uptake of moisture and nutrient via valuable impacts at the cell membrane and cytoplasm, such as increasing photosynthesis and respiration rates in plants, and hormones such as activity involved in plant growth stimulation and yield These results are in a line with Zavarzina et al. (2021), Hemati et al. (2022), Rašovský et al. (2022), and Kamran et al. (2023).
In the current results, using application of KH decreased impurity characters (K, Na, K/Na) and increased quality of sugar beet, where potassium humate enhanced sucrose%, extractable sugar, purity, and top and roots yields; these finding are in agreement with Hartwigsen and Evans (2000), Rassam et al. (2015), de Melo et al. (2016), El-Hassanin et al. (2016), and Hemati et al. (2022), Also, KH and biostimulant application rates improve plant tolerance to stress such as salinity, heat, drought, and cold, and make it more resistant to diseases, insects, and other environmental and agronomical pressures (Hemati et al. 2022; Kamran et al. 2023).
In this study, K-humate administration boosted root production by 22.80 and 28.38%, sugar production by 26.56 and 32.44%, and sucrose percentage by 0.41 and 0.61%, respectively, when compared to untreated plant; it also increased root length, diameter, and fresh weight of root, as well as sucrose percent, purity percent, root and sugar yields of sugar beets when compared to controls which followed the same trend as El-Galad et al. (2013), Shaban et al. (2014), and de Melo et al. (2016).
The current study found that applying some BS such as FA, HP, and SA to sugar beet increased top, root, sugar yield, sucrose percent, and quality characteristics, where it is used to increase plant resistance to the negative effects of biotic and abiotic stresses and participates in regulating their physiological processes; these finding agreeing with those of Mosaad et al. (2022), Rašovský et al. (2022), and Kamran et al. (2023), based on the chemical structure which includes macro and micronutrients, as well as amino acids. These FA shoot development and improve plant yield quality characteristics making them a potentially useful factor and ecologically friendly agent (Kandil et al. 2020; Kamran et al. 2023).
Foliar application of HP + FA (as a foliar treatment) and KH (as a soil amendment) at 24 kg ha−1 enhanced growth parameters, chlorophyll index (SPAD), LAI, top yield, root yield, sugar yield, and quality traits of sugar beetroot in salinity-affected soil. Even though salt stress promoted proline accumulation, fresh weight remained constant at the start of the treatment and grew as treatment progressed. Under abiotic stress, proline administration produces structural and ultrastructural changes in plants under different stress condition to improve the plant root surface with water and nutrient shortages. That agreed with our results for decreasing impurity and increasing quality characters of sugar beet due to the application of HP (AlKahtani et al. 2021; Godoy et al. 2021). SA is recognized as a plant hormone; it plays diverse physiological roles in plants including plant growth, photosynthesis, improved sugar content, quality, and nutrient uptake as reviewed by Tahjib-Ul-Arif et al. (2018), El-Shazoly et al. (2019), and Maruri-López et al. (2019).
5 Conclusion
As a result of the findings described above, this investigation can conclude that use of K-humate and biostimulants (BS) can have a substantial influence on the development and production of sugar beetroot in saline soils. Increasing KH rates increased root yield, top yield, biological yield, sugar yield, sucrose percentage, root length, LAI, and chlorophyll index while reducing impurity attributes such as potassium %, α-amino nitrogen %, and sodium %. The application of biostimulants, particularly foliar spray of FA + HP, enhanced root production, sugar yield, and sucrose percentage. The soil application of K-humate and foliar application of BS treatments had a major influence on sugar beet productivity and quality. It may be recommended to use the combination of K-humate at a rate of 24 kg ha−1 as a soil amendment with the foliar application of some BS to enhance the maximum sucrose percentage, root, and sugar yield, as well as quality characteristics, and to reduce the sodium content in the juice and impurity characteristics.
Data Availability
All data generated or analyzed during this study are included in this article.
References
AlKahtani MD, Hafez YM, Attia K, Rashwan E, Husnain LA, AlGwaiz HI, Abdelaal KA (2021) Evaluation of silicon and proline application on the oxidative machinery in drought-stressed sugar beet. Antioxidants 10(3):398. https://doi.org/10.3390/antiox10030398
Alkharabsheh HM, Seleiman MF, Hewedy OA, Battaglia ML, Jalal RS, Alhammad BA, Schillaci C, Ali N, Al-Doss A (2021) Field crop responses and management strategies to mitigate soil salinity in modern agriculture: a review. Agronomy 11(11):2299. https://doi.org/10.3390/agronomy11112299
Altaf A, Nawaz F, Majeed S, Ahsan M, Ahmad KS, Akhtar G, Shehzad MA, Javeed HMR, Farman M (2023) Foliar humic acid and salicylic acid application stimulates physiological responses and antioxidant systems to improve maize yield under water limitations. JSFA Rep:119–128. https://doi.org/10.1002/jsf2.106
Aremu AO, Stirk WA, Kulkarni MG, Tarkowská D, Turečková V, Gruz J, Šubrtová M, Pěnčík A, Novák O, Doležal K, Strnad M, Van Staden J (2015) Evidence of phytohormones and phenolic acids variability in garden-waste-derived vermicompost leachate, a well-known plant growth stimulant. Plant Growth Regul 75(2):483–492. https://doi.org/10.1007/s10725-014-0011-0
Ayuso M, Hernández T, Garcia C, Pascual JA (1996) Stimulation of barley growth and nutrient absorption by humic substances originating from various organic materials. Bioresource Technol 57(3):251–257. https://doi.org/10.1016/S0960-8524(96)00064-8
Badawi MA, Attia AN, El- Moursy SA, Seadh SE, Hamada AMA (2013) Effect of compost, humic acid and nitrogen fertilizer rates on: 1- growth of sugar beet crop. J Plant Prod 4(5):705–719. https://doi.org/10.21608/jpp.2013.73043
Boguta P, Sokołowska Z (2020) Zinc binding to fulvic acids: assessing the impact of pH, metal concentrations and chemical properties of fulvic acids on the mechanism and stability of formed soluble complexes. Molecules 25(6):1297. https://doi.org/10.3390/molecules25061297
Braziene Z, Paltanavicius V, Avizienytė D (2021) The influence of fulvic acid on spring cereals and sugar beets seed germination and plant productivity. Environ Res 195:110824. https://doi.org/10.1016/j.envres.2021.110824
Carruthers A, Oldfield J (1961) Methods for the assessment of beet quality. Int Sugar J 63:72–74. https://doi.org/10.1016/B978-1-4832-2907-2.50024-
Castiglione AM, Mannino G, Contartese V, Bertea CM, Ertani A (2021) Microbial biostimulants as response to modern agriculture needs: composition, role and application of these innovative products. Plants 10(8):1533. https://doi.org/10.3390/plants10081533
Chapman HD, Pratt F (1961) Methods of analysis for soils, plants and waters. Univ California Div Agr Sci 309. https://doi.org/10.2136/sssaj1963.03615995002700010004x
CoStat, V. (2005) Cohort software798 light house Ave. PMB320, Monterey, CA93940, and USA. email: info@ cohort. com and Website: http://www.cohort.com.DownloadCoStatPart2.html
de Melo BAG, Motta FL, Santana MHA (2016) Humic acids: structural properties and multiple functionalities for novel technological developments. Mater Sci Eng: C 62:967–974. https://doi.org/10.1016/j.msec.2015.12.001
El-Galad MA, Sayed DA, El-Shal RM (2013) Effect of humic acid and compost applied alone or in combination with sulphur on soil fertility and faba bean productivtiy under saline soil conditions. J Soil Sci Agric Eng 4(10):1139–1157. https://doi.org/10.21608/jssae.2013.52501
El-Hassanin AS, Samak MR, Moustafa N, Khalifa AM, Inas MI (2016) Effect of foliar application with humic acid substances under nitrogen fertilization levels on quality and yields of sugar beet plant. Int J Curr Microbiol Appl Sci 5:668–680. https://doi.org/10.20546/ijcmas.2016.511.078
El-Mageed TAA, Mekdad AAA, Rady MOA, Abdelbaky AS, Saudy HS, Shaaban A (2022) Physio-biochemical and agronomic changes of two sugar beet cultivars grown in saline soil as influenced by potassium fertilizer. J Soil Sci Plant Nutr 22(3):3636–3654. https://doi.org/10.1007/s42729-022-00916-7
Elrys AS, Abdo AI, Abdel-Hamed EM, Desoky E-SM (2020) Integrative application of licorice root extract or lipoic acid with fulvic acid improves wheat production and defenses under salt stress conditions. Ecotoxicol Environ Saf 190:110144. https://doi.org/10.1016/j.ecoenv.2019.110144
El-Safy NK, Abo-Marzoka E (2021) Growth of some sugar beet varieties under different locations as affected by foliar application with salicylic acid on yield and quality. Egypt Acad J Biol Sci Bot 12(1):161–173. https://doi.org/10.21608/EAJBSH.2021.171439
El-Shazoly RM, Metwally AA, Hamada AM (2019) Salicylic acid or thiamin increases tolerance to boron toxicity stress in wheat. J Plant Nutr 42(7):702–722. https://doi.org/10.1080/01904167.2018.1549670
Ereqat SI, Abdelkader AA, Nasereddin AF, Al-Jawabreh AO, Zaid TM, Letnik I, Abdeen ZA (2018) Isolation and characterization of phenol degrading bacterium strain Bacillus thuringiensis J20 from olive waste in Palestine. J Environ Sci Health Part A 53(1):39–45. https://doi.org/10.1080/10934529.2017.1368300
FAOStat (2021): http://www. fao. org/faostat/en/# data. QC (accessed 11 January 2021)
Ghaffari H, Tadayon MR, Bahador M, Razmjoo J (2021) Investigation of the proline role in controlling traits related to sugar and root yield of sugar beet under water deficit conditions. Agric Water Manag 243:106448. https://doi.org/10.1016/j.agwat.2020.106448
Ghasemi H, Esmaeili MA, Mohammadian R (2017) Effects of nitrogen on chlorophyll fluorescence and the relationship between chlorophyll content and SPAD values in sugar beet (Beta vulgaris L.) under drip-tape system. J Agricult Biol Sci 12(3):117–122. https://doi.org/10.18380/SZIE.COLUM.2022.9.2.23
Godoy F, Olivos-Hernández K, Stange C, Handford M (2021) Abiotic stress in crop species: improving tolerance by applying plant metabolites. Plants 10(2). https://doi.org/10.3390/plants10020186
Gomez KA, Gomez AA (1984) Statistical procedures for agricultural research. John wiley & sons
Hartwigsen JA, Evans MR (2000) Humic acid seed and substrate treatments promote seedling root development. HortScience 35(7):1231–1233
Hemati A, Alikhani HA, Babaei M, Ajdanian L, Asgari Lajayer B, van Hullebusch ED (2022) Effects of foliar application of humic acid extracts and indole acetic acid on important growth indices of canola (Brassica napus L.). Sci Rep 12(1):20033. https://doi.org/10.1038/s41598-022-21997-5
Hoffmann CM (2010) Root quality of sugarbeet. Sugar Tech 12(3):276–287. https://doi.org/10.1007/s12355-010-0040-6
Ibrahim MEM, Faiyad RMN, El-Gamal ISH (2020) Impact of foliar spraying of some potassium sources and boron levels on sugar beet quantity and quality. J Soil Sci Agric Eng 11(12):835–844. https://doi.org/10.21608/JSSAE.2020.160928
Kamran A, Mushtaq M, Arif M, Rashid S (2023) Role of biostimulants (ascorbic acid and fulvic acid) to synergize rhizobium activity in pea (Pisum sativum L. var. Meteor). Plant Physiol Biochem 196:668–682. https://doi.org/10.1016/j.plaphy.2023.02.018
Kandil EE, Abdelsalam NR, El Aziz AAA, Ali HM, Siddiqui MH (2020) Efficacy of nano-fertilizer, fulvic acid and boron fertilizer on sugar beet (Beta vulgaris L.) Yield and Quality. Sugar Tech 22(5):782–791. https://doi.org/10.1007/s12355-020-00837-8
Khodadadi S, Chegini MA, Soltani A, Ajam Norouzi H, Sadeghzadeh Hemayati S (2020) Influence of foliar-applied humic acid and some key growth regulators on sugar beet (Beta vulgaris L.) under drought stress: antioxidant defense system, photosynthetic characteristics and sugar yield. Sugar Tech 22(5):765–772. https://doi.org/10.1007/s12355-020-00839-6
Koo YM, Heo AY, Choi HW (2020) Salicylic acid as a safe plant protector and growth regulator. Plant Pathol J 36(1):1. https://doi.org/10.5423/PPJ.RW.12.2019.0295
Lv X, Chen S, Wang Y (2019) Advances in understanding the physiological and molecular responses of sugar beet to salt stress. Front Plant Sci 10. https://doi.org/10.3389/fpls.2019.01431
Martin TN, Fipke GM, Minussi Winck JE, Márchese JA (2020) ImageJ software as an alternative method for estimating leaf area in oats. Acta Agron 69(3):162–169
Maruri-López I, Aviles-Baltazar NY, Buchala A, Serrano M (2019) Intra and extracellular journey of the phytohormone salicylic acid. Front Plant Sci 10. https://doi.org/10.3389/fpls.2019.00423
McGinnis R (1971) Beet-sugar technology, 2nd edn. Reinhold Publishing Corporation, New York
Meena M, Divyanshu K, Kumar S, Swapnil P, Zehra A, Shukla V, Upadhyay RS (2019) Regulation of L-proline biosynthesis, signal transduction, transport, accumulation and its vital role in plants during variable environmental conditions. Heliyon 5(12):e02952. https://doi.org/10.1016/j.heliyon.2019.e02952
Merwad A-RM (2016) Efficiency of potassium fertilization and salicylic acid on yield and nutrient accumulation of sugar beet grown on saline soil. Commun Soil Sci Plant Anal 47(9):1184–1192. https://doi.org/10.1080/00103624.2016.1166242
Miao Y, Luo X, Gao X, Wang W, Li B, Hou L (2020) Exogenous salicylic acid alleviates salt stress by improving leaf photosynthesis and root system architecture in cucumber seedlings. Sci Hortic 272:109577. https://doi.org/10.1016/j.scienta.2020.109577
Milford GFJ, Armstrong MJ, Jarvis PJ, Houghton BJ, Bellett-Travers DM, Jones J, Leigh RA (2000) Effect of potassium fertilizer on the yield, quality and potassium offtake of sugar beet crops grown on soils of different potassium status. J Agric Sci 135(1):1–10. https://doi.org/10.1017/S0021859699007881
Moradzadeh S, Siavash Moghaddam S, Rahimi A, Pourakbar L, Sayyed RZ (2021) Combined bio-chemical fertilizers ameliorate agro-biochemical attributes of black cumin (Nigella sativa L.). Sci Rep 11(1):11399. https://doi.org/10.1038/s41598-021-90731-4
Mosaad IS, Serag AH, Sheta MH (2022) Promote sugar beet cultivation in saline soil by applying humic substances in-soil and mineral nitrogen fertilization. JPlant Nutr 45(16):2447–2464
Mubarak MU, Zahir M, Ahmad S, Wakeel A (2016) Sugar beet yield and industrial sugar contents improved by potassium fertilization under scarce and adequate moisture conditions. J Integr Agric 15(11):2620–2626. https://doi.org/10.1016/S2095-3119(15)61252-7
Mukherjee E, Gantait S (2023) Genetic transformation in sugar beet (Beta vulgaris L.): technologies and applications. Sugar Tech 25(2):269–281. https://doi.org/10.1007/s12355-022-01176-6
Muñoz, G., Grieser, J. (2006) Water resources-development and management service, environment and natural resources service, in CLIMWAT 2.0 for CROPWATt. Food and Agriculture Organization of the UN
Neseim M, Amin A, El-Mohammady M (2014) Effect of potassium applied with foliar spray of yeast on sugar beet growth and yield under drought stress. Glob Adv Res J Agric Sci 3(8):211–222
Pulkrábek J, Jozefyová L, Famera O, Sroller J, Stepanek P (2001) Differences in chlorophyll content in leaves of sugar beet. In: Rostlinna Vyroba-UZPI. (Czech Republic)
Rašovský M, Pačuta V, Ducsay L, Lenická D (2022) Quantity and quality changes in sugar beet (Beta vulgaris Provar. Altissima Doel) induced by different sources of biostimulants. Plants 11(17):2222
Rassam G, Dadkhah A, Yazdi AK, Dashti M (2015) Impact of humic acid on yield and quality of sugar beet (Beta vulgaris L.) grown on calcareous soil. Not Sci Biol 7(3):367–371. https://doi.org/10.15835/nsb739568
Rupiasih NN, Vidyasagar P (2005) A review: compositions, structures, properties and applications of humic substances. J Adv Sci Technol 8:16–25
Shaban KAH, Abdel Fatah EM, Syed DA (2014) Impact of humic acid and mineral nitrogen fertilization on soil chemical properties and yield and quality of sugar beet under saline soil. J Soil Sci Agric Eng 5(10):1335–1353. https://doi.org/10.21608/jssae.2014.49752
Skorupa M, Gołębiewski M, Kurnik K, Niedojadło J, Kęsy J, Klamkowski K, Wójcik K, Treder W, Tretyn A, Tyburski J (2019) Salt stress vs. salt shock - the case of sugar beet and its halophytic ancestor. BMC Plant Biol 19(1):57. https://doi.org/10.1186/s12870-019-1661-x
Tahjib-Ul-Arif M, Siddiqui MN, Sohag AAM, Sakil MA, Rahman MM, Polash MAS, Mostofa MG, Tran L-SP (2018) Salicylic acid-mediated enhancement of photosynthesis attributes and antioxidant capacity contributes to yield improvement of maize plants under salt stress. J Plant Growth Regul 37(4):1318–1330. https://doi.org/10.1007/s00344-018-9867-y
Uddling J, Gelang-Alfredsson J, Piikki K, Pleijel H (2007) Evaluating the relationship between leaf chlorophyll concentration and SPAD-502 chlorophyll meter readings. Photosynth Res 91(1):37–46. https://doi.org/10.1007/s11120-006-9077-5
Wanjogu, S., Muya, E., Gicheru, P., Waruru, B. (2001) Soil degradation: management and rehabilitation in Kenya. Paper presented at the Proceedings of the FAO/ISCW expert consultation on Management of Degraded Soil in Southern and Eastern Africa (MADS-SEA) 2nd Networking meeting, Pretoria, South Africa, PR102-113
Yakhin OI, Lubyanov AA, Yakhin IA, Brown PH (2017) Biostimulants in plant science: a global perspective. Front Plant Sci 7. https://doi.org/10.3389/fpls.2016.02049
Yang L, Ma C, Wang L, Chen S, Li H (2012) Salt stress induced proteome and transcriptome changes in sugar beet monosomic addition line M14. J Plant Physiol 169(9):839–850. https://doi.org/10.1016/j.jplph.2012.01.023
Zare Abyaneh H, Jovzi M, Albaji M (2017) Effect of regulated deficit irrigation, partial root drying and N-fertilizer levels on sugar beet crop (Beta vulgaris L.). Agric Water Manag 194:13–23. https://doi.org/10.1016/j.agwat.2017.08.016
Zavarzina AG, Danchenko NN, Demin VV, Artemyeva ZS, Kogut BM (2021) Humic substances: hypotheses and reality (a review). Eur Soil Sci 54(12):1826–1854. https://doi.org/10.1134/S1064229321120164
Acknowledgements
Authors would like to acknowledge their universities for supporting the research.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB).
Author information
Authors and Affiliations
Contributions
N.M.A.A., S.S.E.E., N.S.I., and E.E.K. design idea, methodology, data analysis and wrote the draft manuscript text and N.R.A. and E.E.K. editing and reviewing. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
This article does not contain any studies with human or animal subjects. The current experimental research and field study including the collection of plant material is complying with relevant institutional, national, and international guidelines and legislation, and used for research and development.
Consent for Publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nassar, M.A.A., El-Magharby, S.S., Ibrahim, N.S. et al. Productivity and Quality Variations in Sugar Beet Induced by Soil Application of K-Humate and Foliar Application of Biostimulants Under Salinity Condition. J Soil Sci Plant Nutr 23, 3872–3887 (2023). https://doi.org/10.1007/s42729-023-01307-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42729-023-01307-2