Abstract
Soil contamination by uranium presents a burgeoning global environmental concern, exerting detrimental effects on both agricultural production and soil health. Biochar, a carbonaceous material derived from biomass pyrolysis, exhibits considerable potential for remediating uranium-contaminated soils. However, a comprehensive review of the effects of biochar on the fate and accumulation of uranium in soil–plant systems remains conspicuously absent. In this paper, uranium sources and contamination are reviewed, and the impact of biochar on uranium immobilization and detoxification in soil–plant systems is analyzed. We reviewed the status of uranium contamination in soils globally and found that mining activities are currently the main sources. Further meta-analysis revealed that biochar addition significantly reduced the soil uranium bioavailability and shoot uranium accumulation, and their effect value is 58.9% (40.8–76.8%) and 39.7% (15.7–63.8%), respectively. Additionally, biochar enhances the soil microenvironment, providing favourable conditions for promoting plant growth and reducing uranium mobility. We focused on the mechanisms governing the interaction between biochar and uranium, emphasising the considerable roles played by surface complexation, reduction, ion exchange, and physical adsorption. The modification of biochar by intensifying these mechanisms can promote uranium immobilisation in soils. Finally, biochar alleviates oxidative stress and reduces uranium accumulation in plant tissues, thereby mitigating the adverse effects of uranium on plant growth and development. Overall, our review highlights the capacity of biochar to remediate uranium contamination in soil–plant systems through diverse mechanisms, providing valuable insights for sustainable environmental remediation.
Highlights
-
Biochar reduces uranium mobility through a variety of mechanisms, including surface complexation, reduction, ion exchange, and physical adsorption.
-
Biochar significantly reduces uranium bioavailability in soil and limits its accumulation in plants.
-
Modified biochar has been shown to enhance its effectiveness in immobilising uranium.
-
Biochar application to soil not only promotes uranium remediation but also improves soil quality.
Graphical Abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Uranium, an element with an atomic number of 92 and an atomic weight of 238.03, was first discovered in 1789 by German chemist Martin Heinrich Klaproth as a component of pitchblende (Monreal and Diaconescu 2010; Yang 2023). This naturally occurring radioactive element is crucial for electricity generation in nuclear reactors. It is primarily found in trace amounts in the Earth's crust, at approximately 2.5 parts per million (Malaviya and Singh 2012; Akash et al. 2022). However, anthropogenic activities such as mining, nuclear power generation, nuclear accidents (e.g., Chernobyl and Fukushima), and phosphate fertiliser application have markedly elevated uranium levels in soil, resulting in radiological and chemical toxicity risks (Rani et al. 2023). The widespread accumulation of uranium, extending beyond localized regions, can be attributed to a multitude of factors. These include geochemical processes, anthropogenic activities, and natural weathering, all of which significantly impact soil–plant systems over considerable distances (Pérez-Sánchez and Thorne 2014). Given its chemical and radiological toxicity, uranium poses significant risks to plant growth, agricultural productivity, and food safety (Srivastava et al. 2020). Furthermore, uranium accumulation by plants leads to its transfer along the food chain, ultimately affecting human health, with potential implications for cancer development at levels exceeding 0.05 mg kg−1 body mass d−1 (Gao et al. 2019). Thus, there is an urgent need to develop effective and sustainable strategies to mitigate the adverse effects of uranium contamination.
Biochar, a carbonaceous material derived from biomass pyrolysis, has garnered considerable attention for its potential role in techniques aimed at remediating uranium-contaminated soil (Fahad et al. 2023; Li et al. 2024). The unique properties of biochar, including its high surface area, high porosity, and ability to retain water and nutrients, strongly mitigate uranium contamination in soil (Azeem et al. 2022). For instance, Liu et al. (2022c) demonstrated that the addition of biochar to uranium-containing tailings resulted in uranium adsorption onto the porous surface of the biochar through chemical reactions involving oxygen-rich organic groups. This led to a 40% reduction in uranium concentration in the pore water of the tailing soil. The immobilisation of uranium by biochar is a complex process that involves various mechanisms such as surface complexation, reduction, ion exchange, and physical adsorption (Qiu et al. 2021). The high porosity and extensive surface area of biochar, as explained by Guilhen et al. (2021), make it an efficient platform for the physical adsorption of uranium, significantly limiting its mobility in soil. Physical adsorption not only contributes to uranium immobilisation but also plays a pivotal role in preventing further dispersion (Ahmed et al. 2021d). As uranium associates with biochar, various mechanisms are activated, and surface complexation is particularly prominent (Liao et al. 2022b; Lingamdinne et al. 2022). This process involves a sequence of chemical reactions with the oxygen-rich organic groups present in biochar. Furthermore, ion exchange and physical adsorption ensure the immobilisation of uranium (Liao et al. 2023). Simultaneously, the interaction between biochar and microorganisms in soil promotes the microbial reduction of uranium(VI) to uranium(IV), the uranium shifts from a more soluble and mobile form to a less soluble and more stable form (Ouyang et al. 2023). This reduction leads to the precipitation or adsorption of uranium onto soil particles or biochar, reducing its mobility in the soil (Hilpmann et al. 2023). These multifaceted mechanisms significantly expand the interactions between uranium and biochar, providing a potent strategy for the remediation of uranium-contaminated soils. Furthermore, researchers have investigated the potential of enhancing the efficacy of biochar in uranium remediation through modification (Chen et al. 2023b; Guo et al. 2023). This modification aims to alter the inherent characteristics of biochar, including its microstructure and surface functional group composition, thereby enhancing its potential as an effective tool for uranium immobilisation.
Plants acquire uranium primarily through passive diffusion, facilitated transport, or active uptake mechanisms, which predominantly occur through the root system (Chen et al. 2021; Lai et al. 2021). Once absorbed, uranium can be transported to the aerial plant parts, potentially inducing toxicity and adverse effects on growth and development (Cui et al. 2023). Biochar plays a crucial role in preventing the migration of uranium in soil–plant systems, thereby detoxifying uranium and mitigating its toxicity to plants (Qi et al. 2021; Yin et al. 2022). Detoxification protects plants from uranium-induced damage and mitigates the potential transfer of uranium along the food chain. Recent evidence has demonstrated the effectiveness of biochar in preventing uranium migration and detoxifying soil–plant systems (Qi et al. 2021; Liu et al. 2022c). For instance, biochar application to uranium-contaminated land has led to a remarkable increase in plant production, highlighting the ability of biochar to alleviate uranium toxicity and enhance plant growth (Qi et al. 2022). The application of biochar provides the additional advantage of improving soil quality and fertility by improving its physical, chemical, and biological properties, thereby facilitating healthy plant growth (Khan et al. 2022; Mousavi et al. 2023). Biochar is produced from sustainable biomass sources, which enhances its environmental benefits. These combined strengths confirm that biochar is a promising and versatile solution for the remediation of uranium-contaminated soils.
A large number of studies have demonstrated that biochar can effectively immobilise uranium in soil, reduce plant uranium uptake, and alleviate its toxic effects on plants. Recent reviews have highlighted the role of biochar in uranium removal from aqueous medium, but they have overlook the effects of biochar on uranium immobilisation in soils (Fahad et al. 2023; Shen et al. 2023). Moreover, meta-analysis, a powerful analytical method in environmental studies, can provide a comprehensive analysis of the effects of biochar on uranium-contaminated soil and plant uranium accumulation (Chen et al. 2023a; Huang et al. 2023, 2024). Despite its potential, this specific area of research remains unexplored. This review, therefore, combines meta-analysis to provide significant insights into the impact of biochar on uranium immobilisation, uptake, and detoxification in soil–plant systems. In this paper, we analyze sources of uranium pollution and uranium contamination status globally. We illustrate effects of biochar application on uranium immobilisation in soil. We also review the inner mechanisms for uranium immobilisation by biochar including surface complexation, reduction, ion exchange, and physical adsorption. Lastly, we summarize the effects of biochar application on uranium accumulation and toxicity in plants. This comprehensive analysis highlights the crucial role of biochar in mitigating uranium contamination, and provides a promising pathway for sustainable environmental management and agricultural practices.
2 Global status of uranium contaminations
2.1 Occurrence, existing forms, and sources
Uranium, a vital element for electricity generation in nuclear reactors, has garnered increasing attention globally due to its pivotal role in nuclear power production (Cui et al. 2023). It is naturally and widely distributed in the Earth's crust, surpassing the abundance of silver, mercury, and cadmium (Peng and Cao 2021). The reserves of uranium in oceans alone exceed the quantity found in the Earth's crust by over a thousand times, amounting to approximately 4.5 billion tons (Endrizzi et al. 2016). Naturally occurring uranium comprises three isotopes: uranium-234, uranium-235, and uranium-238. Uranium-238 is the most abundant, accounting for approximately 99.3% of natural uranium, while uranium-235, although less abundant, has the ability to sustain a nuclear chain reaction (Adebiyi et al. 2021). All these isotopes have half-lives exceeding 105 years, posing long-term environmental and health risks (Sharma et al. 2022).
Uranium exists in diverse chemical and physical forms in the environment, and its occurrence and behaviour are influenced by various factors. These factors include the geological composition of the soil and rocks, pH, redox conditions, organic matter content, and interactions with other elements and minerals (Ganguly and Bhan 2023; Kumar et al. 2023). In terms of chemical form, uranium can exist in different oxidation states, with uranium(VI) and uranium(IV) being the most common forms (John et al. 2022). Uranium(VI) is typically present in oxidised environments and can form soluble complexes such as uranyl ions (UO22+) and various carbonate complexes. Uranium (IV), on the other hand, tends to be found in reduced environments and may be associated with minerals or solid phases (Ganguly and Bhan 2023). The oxidation state of uranium in the environment is not fixed but can change in response to shifts in environmental factors, leading to a dynamic equilibrium between the two forms. Such factors include the presence of certain bacteria, the composition of the surrounding material (soil, rock, etc.), and the availability of oxygen or other electron acceptors/donors (Ganguly and Bhan 2023). Physically, uranium can be present in soluble, sorbed, or precipitated forms in soil (Satpathy et al. 2023). Therefore, a BCR extraction method was developed to assess the bioavailability and mobility of uranium in the environment (Thorgersen et al. 2023). The BCR extraction method typically includes four fractions: exchangeable, reducible, oxidisable, and residual (HongE et al. 2022; Liu et al. 2023a). The exchangeable phase of uranium is commonly used to assess its bioavailability in soil–plant systems. The exchangeable fraction refers to the portion of uranium that is loosely bound and readily available for uptake by plants and other organisms (Zhang et al. 2014). In contrast, the residual fraction represents a relatively stable form of uranium that is tightly bound and is less accessible to plants or other organisms (Huang et al. 2022; Wang et al. 2023). The residual fraction is typically composed of insoluble compounds, such as metal oxides or minerals, where uranium ions are strongly bound and less mobile. The reducible fraction of uranium, associated with iron and manganese oxides, typically remains stable. However, it can become mobile if these oxides dissolve in reducing environments such as waterlogged or anaerobic areas (Gao et al. 2021). Similarly, the oxidizable fraction of uranium, bound to organic matter and sulfide minerals, is ordinarily immobile. Nevertheless, its availability may increase due to the decomposition of organic matter or the oxidation of sulfide minerals (Peña et al. 2020). The uranium forms in the soil near a uranium mine in southeastern China are primarily in the residual fraction, whereas the soils in Oak Ridge are dominated by carbonate-bound fractions, and the soils surrounding abandoned uranium mines in Brazil are primarily in the oxidised fraction (Zhou and Gu 2005; Ouyang et al. 2019; Galhardi et al. 2020). The complex interactions between uranium and the surrounding matrix result in the mutual transformation of different uranium fractions through various reactions, including adsorption, desorption, precipitation, redox reaction and complex formation (Florez-Vargas et al. 2023).
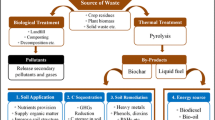
Uranium originates from both natural and anthropogenic sources (Fig. 1). Natural sources include the geological distribution of uranium in rocks, soils, and minerals as well as atmospheric deposition and volcanic eruptions (Chen et al. 2021). Anthropogenic sources, on the other hand, arise from human activities, including mining and milling operations, fertilizer use, nuclear fuel cycle activities, industrial processes, and accidents related to nuclear power plants (Ma et al. 2020). Notably, uranium mining and milling, which account for 41.14% of contamination, are significant contributors to uranium contamination and represent the primary sources (Ramadan et al. 2022). These activities involve the extraction and processing of uranium ore, resulting in the release of uranium and its associated contaminants into the soil, water, and air (Morereau et al. 2022). Groundwater accounts for 39.67% of contamination, primarily due to the leaching of uranium from soils and deposits, which poses a significant risk to the integrity of drinking water (Ma et al. 2020). Furthermore, uranium contamination is intensified by various other anthropogenic activities, including the use of phosphate fertilisers, nuclear facilities, and military operations, which can accelerate the release of uranium into the environment (Ma et al. 2020). Nuclear accidents, such as the Chernobyl and Fukushima nuclear accidents, can also result in local pollution and environmental issues (Schilz et al. 2022).
2.2 Degree of pollution and the phenomenon of compound pollution
Because the sources of uranium contamination can vary significantly, the concentration of uranium in soil also exhibits considerable variation in background levels (Table 1). For example, the average concentrations of uranium in soils in Chile and Germany are 0.79 mg kg−1 and 1.9 mg kg−1, respectively (Table 1) (Utermann and Fuchs 2008; Cabral Pinto et al. 2014). The soil around uranium mining areas typically has a high uranium content. In abandoned uranium mining areas in central Portugal, the uranium concentration in sampled soil ranged from 17.3 to 271 mg kg−1 (Antunes et al. 2011). Surface sediments from sampling sites downstream of uranium mining and milling areas in Guangdong province, China, exhibited a high uranium content ranging from 17.4 to 3935.0 mg kg−1 (Liu et al. 2015).
Moreover, the phenomenon of compound pollution should be taken into account. Uranium often coexists with other heavy metals. For example, Skipperud et al. (2013) reported that large quantities of tailings containing Cd, Pb, and As are generated from U mining. Furthermore, a global meta-analysis indicated that most of the soil near uranium mines is heavily polluted by uranium and Cd (Chen et al. 2022a). A global meta-analysis reported that the average contents of U, Cd, Cr, Pb, Cu, Zn, As, Mn, and Ni in soils from uranium mining areas were 39.88-, 55.33-, 0.88-, 3.81-, 3.12-, 3.07-, 9.26-, 1.83-, and 1.17-fold greater, respectively, than those in the upper continental crust (Chen et al. 2022a). The combined pollution of uranium with other metals in soils can induce synergistic or co-cumulative effects, increasing environmental and health risks.
3 Effects of biochar application on uranium immobilisation in soils
The presence of exchangeable uranium in soils is a considerable concern because it can be readily absorbed by plants, posing risks to human health (Nduka et al. 2022). The application of biochar to uranium-contaminated soils is a promising strategy. The sorptive capacity of biochar acts as a barrier that both physically and chemically interacts with uranium, thereby effectively inhibiting its migration in the soil (Lyu et al. 2021a; Li et al. 2024). This review collected the existing literature on the remediation of uranium-contaminated soil using biochar and conducted a meta-analysis. This comprehensive analysis encompasses the impact of biochar on exchangeable uranium in the soil and the subsequent uptake of uranium by plants (Fig. 2). We observed that biochar addition had a significant negative effect on the available soil uranium concentration, with a mean decrease of 58.9% (ranging from 40.8% to 76.8%) (Fig. 2a). Furthermore, biochar significantly inhibited shoot uranium accumulation, with a mean decrease of 39.7% (ranging from 15.7% to 63.8%) (Fig. 2b). These results provide solid evidence for the considerable impact of biochar on immobilising uranium in soil, thereby reducing its uptake by the aerial parts of plants. For instance, a recent study observed that the application of 10% biochar to uranium-contaminated soils notably decreased uranium leaching, from 26.37% to 3.18%, highlighting the potential of biochar in mitigating uranium migration and protecting the surrounding environment (Fu et al. 2022).
Additionally, our meta-analysis revealed that the modified biochar possesses an enhanced potential for uranium remediation. The modified biochar exhibited a greater capacity to reduce exchangeable uranium in soil (67.2%) compared to the original biochar (51.6%). This effect arises from the surface modification of the biochar, which enhances its adsorption capacity and interaction with uranium in the soil (Xia et al. 2022). The modification of biochar involves physical, chemical, and biological methods to improve its structure and properties (Liu et al. 2022d). Specifically, physical methods include thermal treatment and grinding to alter the surface area and porosity (Sun et al. 2022; Stasi et al. 2022). These processes significantly increase the surface area of the biochar, providing more sites for uranium adsorption and interaction. Similarly, chemical methods involve acid/base treatment or impregnation with metals or other compounds to enhance adsorption and catalytic properties (Jin et al. 2018; Hu et al. 2020; Lingamdinne et al. 2022). This not only introduces additional functional groups onto the biochar surface, such as hydroxyl, carboxyl, and amine groups, but also optimizes its overall charge distribution, enhancing its ability to interact with uranium. Moreover, biological methods may include the use of microorganisms to modify the biochar’s surface properties, further increasing its functionality and specificity towards uranium (Qi et al. 2022). These modifications expand the biochar's adsorption capacity, ion exchange ability, and interaction with uranium, thus contributing to the observed improvement in remediation (Yin et al. 2022). It was discovered that the addition of phosphorus-modified bamboo biochar (PBC) cross-linked with magnesium–aluminium layered double-hydroxide (LDH) composite ("PBC@LDH") resulted in a 54% reduction in the uranium leaching efficiency (Lyu et al. 2021a). The PBC@LDH composite exhibited strong affinity for uranium, effectively minimising uranium leaching and safeguarding groundwater quality. The scanning electron microscopy images before and after biochar modification are shown in Fig. 3. We have observed that most of the stomates on the surface of the unmodified (Fig. 3a) biochar were blocked by its flaky structure, whereas the surface of the modified (Fig. 3b, c) biochar appeared rough and uneven, with increased pores and rougher pore channels, indicating the successful modification of biochar with phosphorous and LDH (Lyu et al. 2021a). These modifications increased the functional groups and contributed to the increased effectiveness of biochar in repairing uranium-contaminated soils (Chen et al. 2023b).
Scanning electron microscope images of (a) bamboo biochar, (b) phosphorus-modified bamboo biochar, (c) phosphorus-modified bamboo biochar cross-linked with magnesium–aluminium layered double-hydroxide composite (Lyu et al. 2021a). The surface of the modified biochar appeared rough and uneven, with increased pores and rougher pore channels, providing more adsorption sites for uranyl ions
Besides its direct uranium immobilisation capabilities, biochar has been shown to enhance soil health and fertility by improving soil pH, organic matter content, and cation exchange capacity (Singh et al. 2022; Li et al. 2023b; Yang et al. 2023c). Moreover, biochar positively influences soil microbial communities, creating conditions conducive to the growth of beneficial microorganisms (Yang et al. 2023b). This, in turn, enhances soil nutrient cycling and plays a crucial role in uranium immobilisation (Dai et al. 2021b). Biochar improves soil pH because of its alkaline nature, which reduces uranium availability in soils (Yang et al. 2023a). Organic matter in the soil has a high capacity to adsorb uranium owing to the presence of functional groups. Therefore, an increased organic matter content in soil is beneficial for uranium immobilisation. In a recent study, it was discovered that the mixed bacteria-loaded biochar application to soil altered the microenvironment by increasing the soil organic matter, cation exchange capacity, fluorescein diacetate hydrolysis activity, and dehydrogenase activity (Qi et al. 2022). The mixed bacteria-loaded biochar significantly decreased the availability of uranium and Cd in soil by 67.4% and 54.2%, respectively, reducing their accumulation in vegetables and promoting celery growth. In addition, the mixed bacteria-loaded biochar alters the structure and function of rhizosphere soil microbial communities by increasing the abundance of beneficial bacteria, while reducing plant pathogenic fungi, which may play an indirect role in inhibiting uranium uptake by celery (Qi et al. 2022). Similarly, Li et al. (2024) found that the pH, cation exchange capacity, enzyme activities, nutritional level of the rhizosphere soil, and abundance and diversity of the bacterial community significantly improved after biochar application. The improvement in soil quality was mainly due to increased microbial activity and organic matter content in the soil after biochar application (Li et al. 2024).
Although biochar has significant potential as a remediation technique for uranium-contaminated soils, several limitations and challenges must be considered. Firstly, the effectiveness of biochar in immobilising uranium may vary depending on its properties such as the type of feedstock, pyrolysis conditions, and post-treatment processes (Qiu et al. 2022). A previous study investigated the impact of biochar produced from five different materials—corn stover, herbal residue, cow manure, distiller's grains, and sugarcane bagasse—on uranium availability in soil. It was found that biochar derived from corn stover significantly reduced uranium availability, with a 36.2% decrease compared to the control. In contrast, soil amended with biochar derived from herbal residues demonstrated the least reduction in uranium availability, with a decrease of only 10% compared to the control (Qi et al. 2021). Furthermore, it has been observed that the preparation of biochar at different pyrolysis temperatures influenced its uranium adsorption capacity, with biochar produced at 500 °C exhibiting the highest efficiency, attributable to the abundance of active sites (Liao et al. 2022c). Therefore, the selection of the feedstock type and production method is crucial for optimising uranium immobilisation. Secondly, the efficacy of biochar application for uranium remediation may depend on site-specific conditions, including soil properties, uranium speciation, and local environmental factors (Kanwar et al. 2023). Taskin et al. (2019) demonstrated that biochar amendments had no effect on uranium concentration in maize grown in calcareous soil. Thus, a comprehensive understanding of site-specific characteristics is essential to determine the appropriate biochar type, application rate, and method for optimal uranium immobilisation. Finally, there is limited research available regarding the long-term effects of biochar on soil–plant systems and the potential accumulation of uranium in biochar-amended soils. Indeed, this is a critical area of study that warrants further exploration to fully understand and responsibly harness the benefits of biochar.
4 Inner mechanisms of uranium immobilisation by biochar
A comprehensive understanding of the complex mechanisms that govern uranium immobilisation through biochar is essential for the development of effective soil remediation strategies. In this section, we present a thorough description of the mechanisms that enable biochar to effectively immobilise uranium and elucidate the interactions involving surface complexation, reduction reactions, ion exchange, and physical adsorption (Fig. 4). Table 2 provides a comprehensive summary of the primary mechanisms governing uranium immobilisation for various types of biochar.
Schematics of immobilisation mechanisms of uranium onto biochar. Surface complexation occurs between uranium and functional groups; Fe-modified and microbial modified biochar indirectly reduce uranium(VI) to uranium(IV); Physical adsorption includes electrostatic and van der Waals forces; UO22+ takes the place of Ca2+, Mg2+, Na+, and K+ on the biochar surface
4.1 Surface complexation
Surface complexation, a crucial mechanism for uranium immobilisation by biochar, involves the creation of coordination bonds between the surface functional groups of biochar (primarily oxygen-, sulphur-, nitrogen-, and phosphorus-containing groups such as carboxyl, hydroxyl, and phosphate) and uranium ions in soil (Leng et al. 2020; Dai et al. 2021a; Tan et al. 2021). Inner- and outer-sphere complexation are the two ways in which surface complexation occurs (Xue et al. 2023). Inner-sphere complexation involves direct covalent bonding between uranium ions and the functional groups on the biochar surface. Inner-sphere complexation typically leads to strong bonds, often resulting in permanent attachment of ions to the biochar surface. In contrast, outer-sphere complexation is characterised by electrostatic interactions between the adsorbate and mineral surface (Yi et al. 2023). This adsorption mechanism, also known as physisorption, involves the hydration shells of water molecules forming a barrier between the adsorbate and adsorbent. Outer-sphere complexes are generally weak and reversible. Therefore, surface complexation typically refers to inner-sphere complexation (Alam et al. 2018; Li et al. 2018). In theoretical research, the effect of ionic strength on the adsorption efficiency is often used to distinguish between inner- and outer-sphere complexation (Liu et al. 2022a). Outer-sphere complexes are generally formed by non-specific, often Coulombic or Van der Waals forces, which can be largely influenced by the ionic strength of the solution (Lepore et al. 2022). However, inner-sphere complexes, which involve direct coordination between the adsorbate and the surface, are less influenced by changes in ionic strength, which explains the minor variations in the adsorption efficiency (Philippou et al. 2022).
The surface functional groups on biochar, specifically the carboxylic and hydroxyl groups, play a critical role in the formation of strong and stabilising complexes with uranyl ions (Dai et al. 2021a; Wu et al. 2022a; Gan et al. 2023; Li et al. 2023a). Ahmed et al (2021a) demonstrated the importance of –COOH and –OH groups in the sorption of uranyl ions onto biochar surface, indicating the presence of these groups significantly influences the sorption capacity. Similarly, Fu et al (2022) confirmed the important role of –COOH and –OH groups in the formation of complex surface bonds, effectively which effectively reduces the mobility and bioavailability of uranium in the soil. Additionally, though the literature is limited, some reports indicated the involvement of aromatic groups in the complexation process through the formation of π–π bonds (Liu et al. 2021a). It is common for biochar to possess aromatic functional groups, containing π-electrons within their aromatic rings. Uranium ions interact with these π-electrons because of their empty d orbitals, forming stable complexes that significantly immobilise uranium ions in soil.
However, the abundance and reactivity of the surface functional groups in biochar are determined by several factors, including the pyrolysis temperature, type of feedstock, and modification (Janu et al. 2021). Research showed that biochar produced at low temperatures (500 °C) tended to have a high concentration of oxygen-rich functional groups, which enhanced its capacity to adsorb uranium. Conversely, biochar produced at high temperatures (750 °C) tended to have reduced adsorption capabilities due to high-temperature metamorphism and obscured pores (Guilhen et al. 2019). Furthermore, the type of feedstock has significant effects on the quantity of surface oxygen-carrying functional groups, thereby profoundly impacting uranium adsorption performance. Jin et al. (2018) indicated this variation in a study, demonstrating that uranium adsorption capacities of wheat-straw-derived biochar and cow-manure-derived biochar were found to be 8.7 mg g−1 and 64.0 mg g−1, respectively (Table 2). This significant variation in adsorption capacity has led to increased research on innovative biochar modification techniques aimed at enhancing the abundance of functional groups. An example of such success is demonstrated in the work of Liao et al. (2022b), where the functionalization of biochar with KMnO4 and H2O2 led to a drama tic increase in the quantity of –OH groups on the surface of the biochar, thereby considerably improving the uranium immobilisation efficiency.
The details of surface complexation and uranium adsorption on biochar were elucidated through the application of advanced analytical techniques. Spectroscopic methods such as Fourier Transform Infrared spectroscopy (FT-IR), X-ray absorption spectroscopy (XAS), and X-ray photoelectron spectroscopy (XPS), have provided crucial insights into the structural and chemical intricacies at the biochar–uranyl interface (Liu et al. 2023b). A comprehensive investigation using these methods determined that uranium mainly adhered to the biochar surface via carboxyl and phenolic hydroxyl groups that were reactive to protons, suggesting the presence of both inner- and outer-sphere complexation (Alam et al. 2018). These methodologies provide important evidence that highlights the crucial role of surface functional groups in uranium immobilisation (Hu et al. 2018b).
4.2 Reduction reactions
Uranium, which is found in various valence states, significantly influences its behaviour and mobility in soil. Hexavalent uranium(VI), primarily in the form of uranyl ions (UO22+), is highly soluble and mobile in soil solutions, whereas tetravalent uranium(IV) is relatively insoluble and less mobile (John et al. 2022). To mitigate the risks posed by highly soluble uranium(VI), researchers have focused on modifying biochar properties to facilitate the reduction of uranium(VI) into its more stable and less soluble form, uranium(IV) (Chen et al. 2021). Previous research has indicated that biochar lacks the capability to directly reduce uranium(VI) to uranium(IV), and this process can only be facilitated indirectly through specific modifications (Yuan et al. 2017). Two primary methods for modifying biochar have been found to effectively reduce uranium (VI) to uranium (IV): Fe modification and microbial modification (Fig. 4) (Ouyang et al. 2023; Ren et al. 2023).
Investigations showed that biochar functionalized with Fe possesses potent electron donor properties (Pang et al. 2022). Through their interaction with UO22+ in soil, these active metals contribute electrons from the biochar surface, consequently promoting the reduction of uranium(VI) to a more stable state, uranium(IV). This approach has great potential for enhancing the remediation efficiency of uranium-contaminated land. For instance, Yin et al. (2022) conducted a study on phosphorus-modified bamboo biochar cross-linked with a magnesium–aluminium-layered double-hydroxide composite. Their investigation revealed the compound's effectiveness in catalysing the reduction of uranium(VI) to uranium(IV) in uranium-contaminated soil, accompanied by a significant reduction in available uranium by up to 55.97% compared with the control (Yin et al. 2022).
Microbe-modified biochar is another important method used for uranium(VI) reduction. Metal-reducing bacteria in the environment, such as Bacillus sp. dwc-2, Shewanella, and Geobacter, which have unique metabolic capabilities, act as electron acceptors and actively participate in reduction reactions, ultimately reducing uranium (VI) to uranium (IV) (Li et al. 2017; Lovley et al. 1991). Numerous studies have demonstrated that biochar application can enhance microbial reduction ability (Rushimisha et al. 2022; Wu et al. 2022b; Dong et al. 2023). The interaction between microorganisms and biochar for uranium reduction is influenced by various factors: (i) biochar as a microbial shelter: biochar plays a crucial role in complex soil, functioning as a shelter for microbes, thus considerably contributing to uranium reduction (Zheng et al. 2022). The porous structure of biochar facilitates both microbial colonisation and growth by providing an extensive surface area and an interconnected pore network, creating an optimal environment for microbial habitation (Li et al. 2022). Frankel et al. (2016) demonstrated that biochar application substantially increased microbial biomass and activity, providing evidence for the role of biochar as a microbial shelter. This shelter not only provides a suitable habitat for microbes but also catalyses their remediation activities, ultimately leading to the reduction and immobilisation of uranium (Arshad et al. 2017); (ii) Biochar-mediated soil improvement: biochar application enhances the inherent soil properties, building a favourable environment for microbial activity (Wei et al. 2023). This was accomplished by enriching the soil with essential nutrients, such as potassium and phosphorus, thus stimulating the growth of microorganisms (Limwikran et al. 2018). Furthermore, biochar improved soil cation exchange capacity and water retention, creating a favourable environment for microbes, thereby considerably contributing to uranium reduction and immobilisation (Tan et al. 2022). (iii) Biochar as an Electron Shuttle: Enriched with carbon, biochar exhibits characteristics similar to those of electron shuttles. A previous study suggested two unique electron shuttle pathways related to biochar, namely “geo-battery” and “geo-conductor” (Sun et al. 2017). The former relies on the surface functionality of the biochar, which can store and release electrons, thereby facilitating redox reactions in the soil (Hou et al. 2022). One gram of biochar can store and utilise as much as 2 mM of electrons for redox reactions in soil (Klüpfel et al. 2014). The geo-conductor pathway is attributed to the high conductivity of biochar due to its carbon structure, which supports direct electron transfer (Sun et al. 2017). These unique characteristics contribute to the role of biochar as an electron shuttle during a wide range of biogeochemical processes. The interaction of biochar with reducing microorganisms establishes an ongoing electron exchange between microorganisms and metal ions, encouraging redox reactions, and ultimately resulting in uranium immobilisation (Yu et al. 2023). Ren et al. (2023) revealed that the oxido-reductively active segment of biochar, predominantly the component containing oxygen, serves as a potent electron shuttle agent in microbial reduction processes, effectively facilitating chromium (Cr) reduction in soil. Notably, chromium is similar to the polyvalent metal ions in uranium, which suggests potential analogous processes. Ding et al. (2018) supported this perspective by showing that biochar-supported microorganisms accelerate the reduction of uranium from U(VI) to U(IV), accompanied by mineral coprecipitation. Overall, microbial modification of biochar provides an effective approach for uranium(VI) reduction, with promising prospects for remediating U-contaminated environments.
4.3 Ion exchange
The ion exchange properties of biochar also contribute to the immobilisation of uranium (Fig. 4) (Liao et al. 2022d). Biochar, which contains exchangeable cations such as Ca2+ and K+, actively participates in these ion exchange reactions, thereby immobilising positively charged uranium ions on its surface (Lingamdinne et al. 2022). For example, cattle-bone biochar had a large number of phosphate groups, and the results showed that UO22+ takes the place of Ca2+ and is incorporated into phosphate groups, effectively immobilising uranium(VI) on the biochar surface (Ashry et al. 2016). Similarly, metal ions (such as Ca2+, Mg2+, Na + , and K+) in anaerobic granular sludge biochar act as effective adsorption sites and actively engage in ion exchange processes which contribute to the removal of uranium(VI) (Zeng et al. 2020).
The efficiency of uranium immobilisation through ion exchange depends on the cation content of biochar and the environment conditions (Kumar et al. 2023). The adsorption and immobilisation capacity of biochar for uranium generally increases with its cation content (Thakur et al. 2022). The impact of different treatment methods on the cationic content of biochar is quite significant (Lingamdinne et al. 2022). For instance, biochar produced from municipal waste typically exhibits higher levels of cationic content compared to those derived from plant materials (Zhao et al. 2015). The content of exchangeable cations in the biochar decreases with increasing pyrolysis temperature (Li et al. 2019c). Additionally, the immobilisation process of uranium is further affected by the presence of competing ions in the soil. High concentrations of common cations, such as Ca2+ and Al3+, can reduce the uranium binding capacity of biochar (Liao et al. 2022d). This is attributed to these ions occupying the binding sites on the biochar, thereby limiting the availability of sites for uranium immobilisation (Mishra et al. 2017).
4.4 Physical adsorption
Physical adsorption is governed by nonchemical interactions between molecules, including electrostatic and van der Waals forces (Fig. 4) (Wen et al. 2018; Ji et al. 2020). Although the van der Waals forces are relatively weak, they form a common adsorption mechanism (Yang et al. 2019). The strength of van der Waals forces depends on the specific surface area, pore volume, and pore radius of the material (An et al. 2022). Biochar, which is notable for its extensive specific surface area and proliferous pore structure, enables the initial adsorption of uranium ions through van der Waals forces (Zhang et al. 2023). The pore structure of biochar is substantially influenced by the feedstock, pyrolysis temperature, and modification processes. For example, biochar derived from crop residues and wood typically has a larger specific surface area than that derived from animal manure and solid waste (Tomczyk et al. 2020).
The adsorption of uranium on biochar is also influenced by electrostatic interactions, which are determined by the surface potential of the biochar, known as the point of zero charge (Pzc), in combination with the environmental pH (Mukherjee et al. 2011). When the environmental pH decreased below the Pzc of biochar, the biochar surface became positively charged. This positive charge weakens the electrostatic attraction between the biochar and positively charged uranium, leading to a reduced adsorption efficiency (Kumar et al. 2011). Conversely, when the environmental pH exceeds the Pzc of the biochar, the biochar surface becomes negatively charged, enhancing electrostatic attraction and facilitating the interaction between the biochar and positively charged uranium ions (Kasera et al. 2022). Understanding the surface potential of biochar and its interaction with the environmental pH is crucial for optimising the application of biochar in uranium pollution remediation.
However, physical adsorption is not the dominant mechanism of uranium immobilisation by biochar. Li et al. (2019b) examined the effects of biochar modified with potassium permanganate on its performance in uranium immobilisation. They observed a significant decrease in the specific surface area of the biochar, reducing it to one-third of its original value. Despite that the surface area decreased, the introduction of functional groups significantly increased the uranium adsorption capacity. These results highlight that the interaction between the functional groups and uranyl ions through complexation is more significant than that through physical interactions. The modification of biochar with potassium permanganate increased the presence of O–H groups and introduced Mn–O groups, improving its ability to form complexes with uranium.
4.5 Other mechanisms
It is noteworthy that biochar enriched with phosphorus groups not only aids in uranium immobilisation through surface complexation but also engages in a distinct precipitation reaction (Liao et al. 2022d). In these biochar, phosphate ions can chemically react with uranyl ions adsorbed on their surfaces (Ruan et al. 2022). Such reactions lead to the formation of uranium phosphate minerals, notably autunite (Ca[(UO2)(PO4)]2·11H2O), characterized by their extremely low solubility (Lyu et al. 2021b). Unlike surface complexation, this precipitation process results in the emergence of new mineral phases, transforming uranium ions into forms that are not bioavailable (Lyu et al. 2021b). Consequently, the formation of these insoluble minerals substantially improves the stability of uranium in the environment, thereby reducing its migration in soil.
Overall, uranium immobilisation by biochar in soil is a complex process involving four primary mechanisms: surface complexation, reduction, ion exchange, and physical adsorption. Although their relative contributions vary, these mechanisms often function simultaneously. These mechanisms vary based on factors such as the biochar properties, soil conditions, and concentrations and chemical forms of uranium ions. It is crucial to comprehensively understand the complex mechanisms of uranium interaction with biochar for practical applications.
5 Effect of biochar application on uranium accumulation and toxicity in plants
5.1 Biochar reduced uranium uptake by plants
The accumulation of uranium in plants poses significant environmental and public health risks, particularly in uranium-contaminated areas (Liu et al. 2022c). Thus, investigating the transfer of uranium from soil to plants has become a priority for soil remediation (Chen et al. 2021; Ao et al. 2022). Plants generally absorb UO22+ through epidermal root cells, utilising carriers and ion channels similar to essential elements such as calcium, iron, and magnesium. Anionic uranium species such as UO2(CO3)22− and UO2PO42− may traverse cell membranes via anionic channels resembling bicarbonate or phosphate (Duquène et al. 2010; Chen et al. 2021; Lai et al. 2021). As reviewed above, biochar application has shown significant potential for decreasing uranium accumulation in plants. Biochar effectively immobilises uranium, reducing its bioavailability and consequently limiting uranium uptake by plant roots, thereby preventing uranium translocation to the aboveground parts of plants (Fig. 5). Indeed, the rhizosphere—the area of soil influenced by root secretions and associated soil microorganisms—plays a vital role in uranium remediation (Wu et al. 2022a, b). The Red Flinders grass has been found to exude acetic acid, citric acid, oxalic acid, formic acid, and lactic acid in roots (Liu et al. 2022c). These low-molar-weight organic acids can facilitate the dissolution of Fe (oxyhydr)oxides, including goethite and magnetite, in Fe-rich matrices, promoting the transformation of Fe-bearing minerals from magnetite to ferrihydrite (Liu et al. 2022c). This process leads to the formation of amorphous ferrihydrite-like minerals, which typically exhibits extensive surface area, facilitating uranium immobilization via co-precipitation or inner-sphere surface complexation. Biochar could facilitate the association between uranium and iron, thereby contributing to the effective immobilization of uranium (Liu et al. 2022c).
However, it is important to note that the effectiveness of biochar in reducing uranium accumulation in plants varies depending on various factors. As previously stated, key determinants of biochar efficacy are its type and properties, including the feedstock used in its production, pyrolysis temperature, and modification method. The inherent properties of the soil in which biochar is applied also influence its functionality (Boghi et al. 2018). Taskin et al. (2019) found that the addition of biochar to calcareous soil did not reduce the uranium content in soybeans. This observation may be attributed to the formation of highly mobile anionic complexes between UO22+ and carbonate in calcareous soil, leading to increased absorption of uranium by plants compared to that in natural soil (Shahandeh and Hossner 2002). Another factor affecting the effectiveness of biochar is the plant species to which it is applied (Gao et al. 2019; Cui et al. 2023). Different plant species have unique physiological and metabolic responses to uranium and biochar. Tomatoes and kohlrabi may exclude uranium, whereas cucumbers and radishes may accumulate uranium in their tissues, thereby posing a risk to the food chain (Hou et al. 2018). Subsequent research explored the effect of biochar on reducing uranium accumulation in vegetables and found that its effectiveness was species-dependent (Qi et al. 2022). The finding indicated that the order of enrichment in the above-ground parts was Brassica chinensis L. > Apium graveolens L. > Lycopersicon esculentum Mill in biochar-treated contaminated soil. Overall, the application of biochar can be regarded as a promising and environmentally friendly approach for mitigating uranium accumulation in plants and promoting safe agricultural production on uranium-contaminated lands. However, a more comprehensive assessment of their potential and limitations is necessary to improve our understanding of their efficacy in environmental remediation.
5.2 Biochar inhibited root-shoot transport of uranium
The translocation of uranium from plant roots to shoots forms a critical connection in the relationship between plants and uranium, substantially influencing the accumulation and distribution of uranium in plant tissues, and is usually measured using transfer factors (TFs) (Dhir 2021). Upon entry into the roots, uranium ions complex with endogenous phosphate residues, resulting in precipitation and fixation within plant organs, thereby preventing their translocation from the roots to the leaves (Laurette et al. 2012). This regulation can significantly minimise the potential phytotoxic effects associated with uranium accumulation, thereby promoting the survival and well-being of plants in uranium-contaminated environments (Wang et al. 2019b). This transportation is modulated by various parameters, including soluble uranium concentrations in the soil (Vandenhove et al. 2007), expression levels of transporter genes in plants (John et al. 2022), and external environmental conditions (Mertens et al. 2022). In the presence of uranium, transporter genes (IRT1, FRO2, and FIT1) and calcium transporter CAX7 in Arabidopsis thaliana, for instance, have been observed to be affected, and external calcium inhibits uranium accumulation in roots (Doustaly et al. 2014; Mertens et al. 2022; Sarthou et al. 2022). The addition of biochar to the soil has a significant influence on this transport process. Yin et al. (2022), for instance, discovered that the TF of uranium for phosphorus-modified bamboo biochar treatments decreased by 11.9% compared with the control treatment, suggesting a positive effect in limiting the migration of uranium from roots to shoots in Indian mustard. A recent study found that biochar application to rice plants under heavy metal stress significantly upregulated the expression of OsMTP11 and CATa, with OsMTP11 showing a 3.2-fold increase and CATa exhibiting a 5.1-fold increase (Pehlivan et al. 2023). These results indicated that the presence of biochar induced a transcriptional response, aiding plant growth under the stress imposed by heavy metals.
5.3 Biochar alleviated uranium phytotoxicity
Biochar alleviates uranium phytotoxicity by reducing uranium availability in soil and improving soil quality (Qi et al. 2022). Physiological indicators, such as chlorophyll levels, nutrition, and photosynthetic performance are essential metrics for quantifying crop health and productivity under stressful conditions (Sapre et al. 2022). The application of biochar significantly enhanced nutrient uptake and synthesis of photosynthetic pigments, leading to a significant improvement in plant performance under stress (Fig. 5) (Majeed et al. 2022). Moreover, uranium induces the overproduction of reactive oxygen species (ROS) in plant cells, which damages membrane integrity and increases the accumulation of oxidative markers, including malondialdehyde (MDA), hydrogen peroxide, and electrolyte leakage (Khan et al. 2022). Biochar has significant potential for mitigating uranium-induced oxidative damage in plants. The application of biochar has been shown to effectively decrease MDA concentrations in ryegrass experiencing uranium stress, thereby indicating its potential in alleviating such stress-induced damage (Zhang et al. 2019b). Plants have a defence system to cope with ROS, which includes various antioxidant enzymes that mitigate the deleterious effects of oxidative stress (Han et al. 2023). Biochar application improves plant tolerance to abiotic stress by increasing antioxidant activity (Irshad et al. 2020). Biochar application enhances antioxidant activities in plants by increasing the content of antioxidant enzymes such as superoxide dismutase (SOD), peroxidase (POD), catalase (CAT), and ascorbate peroxidase (APX) (Rizwan et al. 2018). SOD mitigates superoxide radicals, whereas POD and CAT catalyse the breakdown of hydrogen peroxide, thereby reducing potential oxidative damage. APX utilises ascorbate to detoxify peroxides (Zhu et al. 2023a). By significantly increasing antioxidant activity, biochar improves plant tolerance to abiotic stress and promotes overall plant health and productivity.
6 Concluding remarks and future perspectives
This review comprehensively describes the potential of biochar as a remediation solution for uranium-contaminated soils and its beneficial effects on plant productivity. The global challenge of uranium contamination, primarily from industrial activities, results in soil toxicity, which has detrimental effects on plant growth. Biochar is a promising remediation agent owing to its unique properties. Biochar not only reduces the mobility and bioavailability of uranium in soil through surface complexation, reduction, ion exchange, and physical adsorption processes but also enhances soil nutrition. Notably, the modification of biochar to enhance its surface complexation effect as well as the synergistic reduction mechanism in interactions with microorganisms is recognised as a promising direction for further research. Additionally, biochar substantially decreases uranium uptake by plants, transport and related toxicity, and its effectiveness depends on multiple factors, including the biochar type, soil characteristics, and plant species.
Based on published papers and our review, future research should explore the following four points. Firstly, an examination of the relationships among various factors influencing the remediation efficacy of biochar in the soil–plant system, including soil properties, levels of uranium contamination, plant species, and biochar characteristics, is imperative because of the existing limitations in our understanding of this complex system. Secondly, exploring the potential of biochar to improve soil quality and crop growth is critical. This involves assessing the impact of biochar on soil nutrient cycling, water retention, and crop growth, providing insights for sustainable agricultural practices. However, related investigations have been limited to date. Thirdly, the effects of biochar application on plant responses to uranium exposure should be investigated using molecular techniques such as transcriptomics or proteomics. In particular, the mechanisms and expression of transporter genes in plant cells are poorly understood. Last but not least, long-term field trials and sustained investigations are necessary to track the performance and effect of biochar on different soil compositions and environmental conditions. Emphasis should be placed on the intricate interactions between biochar, soil microbiota, and plant physiology (Additional file 1).
Availability of data and materials
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
References
Adebiyi FM, Ore OT, Adeola AO, Durodola SS, Akeremale OF, Olubodun KO, Akeremale OK (2021) Occurrence and remediation of naturally occurring radioactive materials in Nigeria: a review. Environ Chem Lett 19:3243–3262
Ahmed W, Mehmood S, Núñez-Delgado A, Ali S, Qaswar M, Khan ZH, Ying H, Chen D-Y (2021a) Utilization of Citrullus lanatus L. seeds to synthesize a novel MnFe2O4-biochar adsorbent for the removal of U(VI) from wastewater: Insights and comparison between modified and raw biochar. Sci Total Environ 771:144955
Ahmed W, Mehmood S, Núñez-Delgado A, Qaswar M, Ali S, Ying H, Liu Z, Mahmood M, Chen D-Y (2021b) Fabrication, characterization and U(VI) sorption properties of a novel biochar derived from Tribulus terrestris via two different approaches. Sci Total Environ 780:146617
Ahmed W, Mehmood S, Qaswar M, Ali S, Khan ZH, Ying H, Chen D-Y, Núñez-Delgado A (2021c) Oxidized biochar obtained from rice straw as adsorbent to remove uranium (VI) from aqueous solutions. J Environ Chem Eng 9:105104
Ahmed W, Núñez-Delgado A, Mehmood S, Ali S, Qaswar M, Shakoor A, Chen D-Y (2021d) Highly efficient uranium (VI) capture from aqueous solution by means of a hydroxyapatite-biochar nanocomposite: adsorption behavior and mechanism. Environ Res 201:111518
Akash S, Sivaprakash B, Raja VCV, Rajamohan N, Muthusamy G (2022) Remediation techniques for uranium removal from polluted environment—review on methods, mechanism and toxicology. Environ Pollut 302:119068
Alam MS, Gorman-Lewis D, Chen N, Safari S, Baek K, Konhauser KO, Alessi DS (2018) Mechanisms of the removal of U(VI) from aqueous solution using biochar: a combined spectroscopic and modeling approach. Environ Sci Technol 52:13057–13067
Albayari M, Nazal MK, Khalili FI, Nordin N, Adnan R (2021) Biochar derived from Salvadora persica branches biomass as low-cost adsorbent for removal of uranium(VI) and thorium(IV) from water. J Radioanal Nucl Chem 328:669–678
Alsabbagh AH, Abuqudaira TM (2017) Phytoremediation of Jordanian uranium-rich soil using sunflower. Water Air Soil Pollut 228:219
An K, Wang Z, Yang X, Qu Z, Sun F, Zhou W, Zhao H (2022) Reasons of low formaldehyde adsorption capacity on activated carbon: multi-scale simulation of dynamic interaction between pore size and functional groups. J Environ Chem Eng 10:108723
Antunes SC, Pereira R, Marques SM, Castro BB, Gonçalves F (2011) Impaired microbial activity caused by metal pollution: a field study in a deactivated uranium mining area. Sci Total Environ 410–411:87–95
Ao M, Chen X, Deng T, Sun S, Tang Y, Morel JL, Qiu R, Wang S (2022) Chromium biogeochemical behaviour in soil-plant systems and remediation strategies: a critical review. J Hazard Mater 424:127233
Arshad M, Khan AHA, Hussain I, Badar uz Z, Anees M, Iqbal M, Soja G, Linde C, Yousaf S (2017) The reduction of chromium (VI) phytotoxicity and phytoavailability to wheat (Triticum aestivum L.) using biochar and bacteria. Appl Soil Ecol 114:90–98
Ashry A, Bailey EH, Chenery SRN, Young SD (2016) Kinetic study of time-dependent fixation of U(VI) on biochar. J Hazard Mater 320:55–66
Azeem M, Shaheen SM, Ali A, Jeyasundar PGSA, Latif A, Abdelrahman H, Li R, Almazroui M, Niazi NK, Sarmah AK, Li G, Rinklebe J, Zhu Y-G, Zhang Z (2022) Removal of potentially toxic elements from contaminated soil and water using bone char compared to plant- and bone-derived biochars: a review. J Hazard Mater 427:128131
Bern CR, Walton-Day K, Naftz DL (2019) Improved enrichment factor calculations through principal component analysis: examples from soils near breccia pipe uranium mines, Arizona, USA. Environ Pollut 248:90–100
Bigalke M, Ulrich A, Rehmus A, Keller A (2017) Accumulation of cadmium and uranium in arable soils in Switzerland. Environ Pollut 221:85–93
Boghi A, Roose T, Kirk GJD (2018) A model of uranium uptake by plant roots allowing for root-induced changes in the soil. Environ Sci Technol 52:3536–3545
Boteva S, Radeva G, Traykov I, Kenarova A (2016) Effects of long-term radionuclide and heavy metal contamination on the activity of microbial communities, inhabiting uranium mining impacted soils. Environ Sci Pollut Res 23:5644–5653
Cabral Pinto MMS, Ferreira da Silva EA, Silva MMVG, Dinis PA (2014) Estimated background values maps of uranium in Santiago island topsoil and stream sediments. Procedia Earth Planet Sci 8:23–27
Chen L, Liu J, Zhang W, Zhou J, Luo D, Li Z (2021) Uranium (U) source, speciation, uptake, toxicity and bioremediation strategies in soil-plant system: a review. J Hazard Mater 413:125319
Chen L, Wang J, Beiyuan J, Guo X, Wu H, Fang L (2022a) Environmental and health risk assessment of potentially toxic trace elements in soils near uranium (U) mines: a global meta-analysis. Sci Total Environ 816:151556
Chen W, Feng J, Liu S, Zhang J, Cai Y, Lv Z, Fang M, Tan X (2022b) A green and economical MgO/biochar composite for the removal of U(VI) from aqueous solutions. Chem Eng Res Des 180:391–401
Chen L, Wang F, Zhang Z, Chao H, He H, Hu W, Zeng Y, Duan C, Liu J, Fang L (2023a) Influences of arbuscular mycorrhizal fungi on crop growth and potentially toxic element accumulation in contaminated soils: a meta-analysis. Crit Rev Environ Sci Technol 53:1795–1816
Chen X, Wang Y, Xia H, Ren Q, Li Y, Xu L, Xie C, Wang Y (2023b) “One-can” strategy for the synthesis of hydrothermal biochar modified with phosphate groups and efficient removal of uranium(VI). J Environ Radioact 263:107182
Costelloe BGLPMAMT. 2005. Contaminant dispersion at the rehabilitated Mary Kathleen uranium mine, Australia.
Cui Q, Zhang Z, Beiyuan J, Cui Y, Chen L, Chen H, Fang L (2023) A critical review of uranium in the soil-plant system: Distribution, bioavailability, toxicity, and bioremediation strategies. Crit Rev Environ Sci Technol 53:340–365
Dai L, Li L, Zhu W, Ma H, Huang H, Lu Q, Yang M, Ran Y (2020) Post-engineering of biochar via thermal air treatment for highly efficient promotion of uranium(VI) adsorption. Biores Technol 298:122576
Dai L, Lu Q, Zhou H, Shen F, Liu Z, Zhu W, Huang H (2021a) Tuning oxygenated functional groups on biochar for water pollution control: a critical review. J Hazard Mater 420:126547
Dai Z, Xiong X, Zhu H, Xu H, Leng P, Li J, Tang C, Xu J (2021b) Association of biochar properties with changes in soil bacterial, fungal and fauna communities and nutrient cycling processes. Biochar 3:239–254
Dhir B (2021) Effective removal of radioactive waste from environment using plants. In: Prasad R (ed) Phytoremediation for environmental sustainability. Springer Nature Singapore, Singapore, pp 71–82
Ding L, Tan W-f, Xie S-b, Mumford K, Lv J-w, Wang H-q, Fang Q, Zhang X-w, Wu X-y, Li M (2018) Uranium adsorption and subsequent re-oxidation under aerobic conditions by Leifsonia sp.-coated biochar as green trapping agent. Environ Pollut 242:778–787
Dong L, Yang J, Mou Y, Sheng G, Wang L, Linghu W, Asiri AM, Alamry KA (2017) Effect of various environmental factors on the adsorption of U(VI) onto biochar derived from rice straw. J Radioanal Nucl Chem 314:377–386
Dong H, Coffin ES, Sheng Y, Duley ML, Khalifa YM (2023) Microbial reduction of Fe(III) in nontronite: role of biochar as a redox mediator. Geochim Cosmochim Acta 345:102–116
Doustaly F, Combes F, Fiévet JB, Berthet S, Hugouvieux V, Bastien O, Aranjuelo I, Leonhardt N, Rivasseau C, Carrière M, Vavasseur A, Renou J-P, Vandenbrouck Y, Bourguignon J (2014) Uranium perturbs signaling and iron uptake response in Arabidopsis thaliana roots. Metallomics 6:809–821
Duquène L, Vandenhove H, Tack F, Van Hees M, Wannijn J (2010) Diffusive gradient in thin FILMS (DGT) compared with soil solution and labile uranium fraction for predicting uranium bioavailability to ryegrass. J Environ Radioact 101:140–147
Endrizzi F, Leggett CJ, Rao L (2016) Scientific basis for efficient extraction of uranium from seawater. I: understanding the chemical speciation of uranium under seawater conditions. Ind Eng Chem Res 55:4249–4256
Fahad SA, Nawab MS, Shaida MA, Verma S, Khan MU, Siddiqui V, Naushad M, Saleem L, Farooqi IH (2023) Carbon based adsorbents for the removal of U(VI) from aqueous medium: a state of the art review. J Water Process Eng 52:103458
Feng Y, Qiu X, Tao Z, Zhengyang E, Song J, Dong Y, Liang J, Li P, Fan Q (2022) Oxygen-containing groups in cellulose and lignin biochar: their roles in U(VI) adsorption. Environ Sci Pollut Res 29:76728–76738
Florez-Vargas O, Vilanova E, Alcaide C, Henao JA, Villarreal-Jaimes CA, Medina-Pérez OM, Rodriguez-Villamizar LA, Idrovo AJ, Sánchez-Rodríguez LH (2023) Geological context and human exposures to element mixtures in mining and agricultural settings in Colombia. Sci Total Environ 898:165632
Frankel ML, Bhuiyan TI, Veksha A, Demeter MA, Layzell DB, Helleur RJ, Hill JM, Turner RJ (2016) Removal and biodegradation of naphthenic acids by biochar and attached environmental biofilms in the presence of co-contaminating metals. Biores Technol 216:352–361
Fu H, Wang S, Zhang H, Dai Z, He G, Li G, Ding D (2022) Remediation of uranium-contaminated acidic red soil by rice husk biochar. Environ Sci Pollut Res 29:77839–77850
Galhardi JA, de Mello JWV, Wilkinson KJ (2020) Bioaccumulation of potentially toxic elements from the soils surrounding a legacy uranium mine in Brazil. Chemosphere 261:127679
Gan J, Zhang L, Wang Q, Xin Q, Hu E, Lei Z, Wang H, Wang H (2023) Synergistic action of multiple functional groups enhanced uranium extraction from seawater of porous phosphorylated chitosan/coal-based activated carbon composite sponge. Desalination 545:116154
Ganguly S, Bhan U (2023) Occurrences and mobility of uranium in soil profile due to groundwater–soil interaction. Hydrogeochem Aqua Ecosyst. https://doi.org/10.1002/9781119870562.ch8
Gao N, Huang Z, Liu H, Hou J, Liu X (2019) Advances on the toxicity of uranium to different organisms. Chemosphere 237:124548
Gao L, Li R, Liang Z, Wu Q, Yang Z, Li M, Chen J, Hou L (2021) Mobilization mechanisms and toxicity risk of sediment trace metals (Cu, Zn, Ni, and Pb) based on diffusive gradients in thin films: a case study in the Xizhi River basin, South China. J Hazard Mater 410:124590
Gramss G, Voigt K-D (2016) Gradual accumulation of heavy metals in an industrial wheat crop from uranium mine soil and the potential use of the herbage. Agriculture 6:51
Guilhen SN, Mašek O, Ortiz N, Izidoro JC, Fungaro DA (2019) Pyrolytic temperature evaluation of macauba biochar for uranium adsorption from aqueous solutions. Biomass Bioenerg 122:381–390
Guilhen SN, Rovani S, Araujo LGd, Tenório JAS, Mašek O (2021) Uranium removal from aqueous solution using macauba endocarp-derived biochar: effect of physical activation. Environ Pollut 272:116022
Guo L, Peng L, Li J, Zhang W, Shi B (2023) Superefficient separation of Th(IV) and U(VI) by lignin-derived magnetic biochar via competitive adsorption mechanism. Sep Purif Technol 315:123635
Han L, Gu H, Lu W, Li H, Peng W-x, Ling Ma N, Lam SS, Sonne C (2023) Progress in phytoremediation of chromium from the environment. Chemosphere 344:140307
Hilpmann S, Rossberg A, Steudtner R, Drobot B, Hübner R, Bok F, Prieur D, Bauters S, Kvashnina KO, Stumpf T, Cherkouk A (2023) Presence of uranium(V) during uranium(VI) reduction by Desulfosporosinus hippei DSM 8344T. Sci Total Environ 875:162593
Hong EY, Wan Z, Kim Y, Yu J (2022) Submerged zone and vegetation drive distribution of heavy metal fractions and microbial community structure: Insights into stormwater biofiltration system. Sci Total Environ 853:158367
Hou J, Wang C, Zhou Y, Li S, Hayat T, Alsaedi A, Wang X (2018) Effects of uranium stress on physiological and biochemical characteristics in seedlings of six common edible vegetables. J Radioanal Nucl Chem 316:1001–1010
Hou N, Li X, Jiang X, Zhang N, Wang R, Li D (2022) The role of biochar in the photocatalytic treatment of a mixture of Cr(VI) and phenol pollutants: biochar as a carrier for transferring and storing electrons. Sci Total Environ 844:157145
Hu H, Zhang X, Wang T, Sun L, Wu H, Chen X (2018a) Bamboo (Acidosasa longiligula) shoot shell biochar: its potential application to isolation of uranium(VI) from aqueous solution. J Radioanal Nucl Chem 316:349–362
Hu Q, Zhu Y, Hu B, Lu S, Sheng G (2018b) Mechanistic insights into sequestration of U(VI) toward magnetic biochar: Batch, XPS and EXAFS techniques. J Environ Sci 70:217–225
Hu R, Xiao J, Wang T, Chen G, Chen L, Tian X (2020) Engineering of phosphate-functionalized biochars with highly developed surface area and porosity for efficient and selective extraction of uranium. Chem Eng J 379:122388
Huang M, Zhou M, Li Z, Ding X, Wen J, Jin C, Wang L, Xiao L, Chen J (2022) How do drying-wetting cycles influence availability of heavy metals in sediment? A perspective from DOM molecular composition. Water Res 220:118671
Huang F, Hu J, Chen L, Wang Z, Sun S, Zhang W, Jiang H, Luo Y, Wang L, Zeng Y, Fang L (2023) Microplastics may increase the environmental risks of Cd via promoting Cd uptake by plants: a meta-analysis. J Hazard Mater 448:130887
Huang F, Li Z, Yang X, Liu H, Chen L, Chang N, He H, Zeng Y, Qiu T, Fang L (2024) Silicon reduces toxicity and accumulation of arsenic and cadmium in cereal crops: a meta-analysis, mechanism, and perspective study. Sci Total Environ 918:170663
Irshad MK, Noman A, Alhaithloul HAS, Adeel M, Rui Y, Shah T, Zhu S, Shang J (2020) Goethite-modified biochar ameliorates the growth of rice (Oryza sativa L.) plants by suppressing Cd and As-induced oxidative stress in Cd and As co-contaminated paddy soil. Sci Total Environ 717:137086
Janu R, Mrlik V, Ribitsch D, Hofman J, Sedláček P, Bielská L, Soja G (2021) Biochar surface functional groups as affected by biomass feedstock, biochar composition and pyrolysis temperature. Carbon Resour Convers 4:36–46
Ji W, Parameswarappa Jayalakshmamma M, Abou Khalil C, Zhao L, Boufadel M (2020) Removal of hydrocarbon from soils possessing macro-heterogeneities using electrokinetics and surfactants. Chem Eng J Adv 4:100030
Jin J, Li S, Peng X, Liu W, Zhang C, Yang Y, Han L, Du Z, Sun K, Wang X (2018) HNO3 modified biochars for uranium(VI) removal from aqueous solution. Biores Technol 256:247–253
John WA, Lückel B, Matschiavelli N, Hübner R, Matschi S, Hoehenwarter W, Sachs S (2022) Endocytosis is a significant contributor to uranium(VI) uptake in tobacco (Nicotiana tabacum) BY-2 cells in phosphate-deficient culture. Sci Total Environ 823:153700
Kanwar P, Mina U, Thakur IS, Srivastava S (2023) Heavy metal phytoremediation by the novel prospect of microbes, nanotechnology, and genetic engineering for recovery and rehabilitation of landfill site. Bioresour Technol Rep 23:101518
Kasera N, Kolar P, Hall SG (2022) Nitrogen-doped biochars as adsorbents for mitigation of heavy metals and organics from water: a review. Biochar 4:17
Khan Z, Fan X, Khan MN, Khan MA, Zhang K, Fu Y, Shen H (2022) The toxicity of heavy metals and plant signaling facilitated by biochar application: implications for stress mitigation and crop production. Chemosphere 308:136466
Klüpfel L, Keiluweit M, Kleber M, Sander M (2014) Redox properties of plant biomass-derived black carbon (biochar). Environ Sci Technol 48:5601–5611
Kumar S, Loganathan VA, Gupta RB, Barnett MO (2011) An Assessment of U(VI) removal from groundwater using biochar produced from hydrothermal carbonization. J Environ Manage 92:2504–2512
Kumar A, Kumar V, Saroop S, Arsenov D, Bali S, Radziemska M, Bhardwaj R (2023) A comprehensive review of Uranium in the terrestrial and aquatic environment: bioavailability, immobilization, tolerance and remediation approaches. Plant Soil 490:31–65
Lai J-l, Liu Z-w, Li C, Luo X-g (2021) Analysis of accumulation and phytotoxicity mechanism of uranium and cadmium in two sweet potato cultivars. J Hazard Mater 409:124997
Laurette J, Larue C, Llorens I, Jaillard D, Jouneau P-H, Bourguignon J, Carrière M (2012) Speciation of uranium in plants upon root accumulation and root-to-shoot translocation: A XAS and TEM study. Environ Exp Bot 77:87–95
Leng L, Xu S, Liu R, Yu T, Zhuo X, Leng S, Xiong Q, Huang H (2020) Nitrogen containing functional groups of biochar: an overview. Biores Technol 298:122286
Lepore GO, Schingaro E, Mesto E, Lacalamita M, Cristiani C, Stampino PG, Dotelli G, Finocchio E, d’Acapito F, Giuli G (2022) Lanthanum captured in montmorillonite: evidence of inner-sphere complexes from X-ray absorption spectroscopy investigations. Appl Clay Sci 230:106676
Li X, Ding C, Liao J, Du L, Sun Q, Yang J, Yang Y, Zhang D, Tang J, Liu N (2017) Microbial reduction of uranium (VI) by Bacillus sp. dwc-2: a macroscopic and spectroscopic study. J Environ Sci 53:9–15
Li X, Pan H, Yu M, Wakeel M, Luo J, Alharbi NS, Liao Q, Liu J (2018) Macroscopic and molecular investigations of immobilization mechanism of uranium on biochar: EXAFS spectroscopy and static batch. J Mol Liq 269:64–71
Li M, Liu H, Chen T, Dong C, Sun Y (2019a) Synthesis of magnetic biochar composites for enhanced uranium(VI) adsorption. Sci Total Environ 651:1020–1028
Li N, Yin M, Tsang DCW, Yang S, Liu J, Li X, Song G, Wang J (2019b) Mechanisms of U(VI) removal by biochar derived from Ficus microcarpa aerial root: a comparison between raw and modified biochar. Sci Total Environ 697:134115
Li S, Harris S, Anandhi A, Chen G (2019c) Predicting biochar properties and functions based on feedstock and pyrolysis temperature: a review and data syntheses. J Clean Prod 215:890–902
Li R, Wang B, Niu A, Cheng N, Chen M, Zhang X, Yu Z, Wang S (2022) Application of biochar immobilized microorganisms for pollutants removal from wastewater: a review. Sci Total Environ 837:155563
Li A, Ye C, Jiang Y, Deng H (2023a) Enhanced removal performance of magnesium-modified biochar for cadmium in wastewaters: role of active functional groups, processes, and mechanisms. Biores Technol 386:129515
Li Y, Abdo AI, Shi Z, Merwad A-RMA, Zhang J (2023b) Biochar derived from invasive plants improved the pH, macronutrient availability and biological properties better than liming for acid rain-affected soil. Biochar 5:59
Li N, Wang Y, Zhou L, Fu D, Chen T, Chen X, Wang Q, Zhu W (2024) The joint action of biochar and plant roots on U-stressed soil remediation: insights from bacteriomics and metabolomics. J Hazard Mater 461:132635
Liao J, Chen H, Zhang Y, Zhu W (2022a) Pyrolysis of animal manure under nitrogen atmosphere: an environment protection way to obtain animal manure biochar for high-efficient adsorption of uranium(VI). J Anal Appl Pyrol 163:105493
Liao J, Ding L, Zhang Y, Zhu W (2022b) Efficient removal of uranium from wastewater using pig manure biochar: understanding adsorption and binding mechanisms. J Hazard Mater 423:127190
Liao J, He X, Zhang Y, Zhang L, He Z (2022c) Constructing a novel carbon material for efficient separation of uranium(VI) from solution. Biomass Convers Bioref. https://doi.org/10.1007/s13399-022-02856-9
Liao J, He X, Zhang Y, Zhu W, Zhang L, He Z (2022d) Bismuth impregnated biochar for efficient uranium removal from solution: adsorption behavior and interfacial mechanism. Sci Total Environ 819:153145
Liao J, Xiong T, Ding L, Zhang Y, Zhu W (2022e) Effective separation of uranium(VI) from wastewater using a magnetic carbon as a recyclable adsorbent. Sep Purif Technol 282:120140
Liao J, He X, Zhang Y, Zhang L, He Z (2023) The construction of magnetic hydroxyapatite-functionalized pig manure-derived biochar for the efficient uranium separation. Chem Eng J 457:141367
Liatsou I, Michail G, Demetriou M, Pashalidis I (2017) Uranium binding by biochar fibres derived from Luffa cylindrica after controlled surface oxidation. J Radioanal Nucl Chem 311:871–875
Limwikran T, Kheoruenromne I, Suddhiprakarn A, Prakongkep N, Gilkes RJ (2018) Dissolution of K, Ca, and P from biochar grains in tropical soils. Geoderma 312:139–150
Lingamdinne LP, Choi J-S, Angaru GKR, Karri RR, Yang J-K, Chang Y-Y, Koduru JR (2022) Magnetic-watermelon rinds biochar for uranium-contaminated water treatment using an electromagnetic semi-batch column with removal mechanistic investigations. Chemosphere 286:131776
Liu J, Wang J, Li H, Shen C-C, Chen Y, Wang C, Ye H, Long J, Song G, Wu Y (2015) Surface sediment contamination by uranium mining/milling activities in South China. Clean: Soil, Air, Water 43:414–420
Liu J, Ge Y, Wang G, Liu Y, Xu X (2021a) Highly efficient removal of U(VI) in aqueous solutions by tea waste-derived biochar-supported iron-manganese oxide composite. J Radioanal Nucl Chem 330:871–882
Liu R, Wang H, Han L, Hu B, Qiu M (2021b) Reductive and adsorptive elimination of U(VI) ions in aqueous solution by SFeS@Biochar composites. Environ Sci Pollut Res 28:55176–55185
Liu X, Tournassat C, Grangeon S, Kalinichev AG, Takahashi Y, Marques Fernandes M (2022a) Molecular-level understanding of metal ion retention in clay-rich materials. Nat Rev Earth Environ 3:461–476
Liu Y, Wang Y, Xia H, Wang Q, Chen X, Lv J, Li Y, Zhao J, Liu Y, Yuan D (2022b) Low-cost reed straw-derived biochar prepared by hydrothermal carbonization for the removal of uranium(VI) from aqueous solution. J Radioanal Nucl Chem 331:3915–3925
Liu Y, Wu S, Nguyen TAH, Chan T-S, Lu Y-R, Huang L (2022c) Biochar mediated uranium immobilization in magnetite rich Cu tailings subject to organic matter amendment and native plant colonization. J Hazard Mater 427:127860
Liu Z, Xu Z, Xu L, Buyong F, Chay TC, Li Z, Cai Y, Hu B, Zhu Y, Wang X (2022d) Modified biochar: synthesis and mechanism for removal of environmental heavy metals. Carbon Res 1:8
Liu B, Jiang S, Guan D-X, Song X, Li Y, Zhou S, Wang B, Gao B (2023a) Geochemical fractionation, bioaccessibility and ecological risk of metallic elements in the weathering profiles of typical skarn-type copper tailings from Tongling, China. Sci Total Environ 894:164859
Liu Y, Yuan W, Lin W, Yu S, Zhou L, Zeng Q, Wang J, Tao L, Dai Q, Liu J (2023b) Efficacy and mechanisms of δ-MnO2 modified biochar with enhanced porous structure for uranium(VI) separation from wastewater. Environ Pollut 335:122262
Lovley DR, Phillips EJP, Gorby YA, Landa ER (1991) Microbial reduction of uranium. Nature 350:413–416
Lyu P, Wang G, Cao Y, Wang B, Deng N (2021a) Phosphorus-modified biochar cross-linked Mg–Al layered double-hydroxide composite for immobilizing uranium in mining contaminated soil. Chemosphere 276:130116
Lyu P, Wang G, Wang B, Yin Q, Li Y, Deng N (2021b) Adsorption and interaction mechanism of uranium (VI) from aqueous solutions on phosphate-impregnation biochar cross-linked MgAl layered double-hydroxide composite. Appl Clay Sci 209:106146
Ma M, Wang R, Xu L, Xu M, Liu S (2020) Emerging health risks and underlying toxicological mechanisms of uranium contamination: lessons from the past two decades. Environ Int 145:106107
Majeed A, Muhmood A, Niaz A, Ditta A, Rajpar MN (2022) Comparative efficacy of different biochars and traditional manures in the attenuation of cadmium toxicity in rice (Oryza sativa L.). Arab J Geosci 15:209
Malaviya P, Singh A (2012) Phytoremediation strategies for remediation of uranium-contaminated environments: a review. Crit Rev Environ Sci Technol 42:2575–2647
Mertens A, Horemans N, Saenen E, Nauts R, Cuypers A (2022) Calcium affects uranium responses in Arabidopsis thaliana: from distribution to toxicity. Plant Physiol Biochem 185:101–111
Mishra V, Sureshkumar MK, Gupta N, Kaushik CP (2017) Study on sorption characteristics of uranium onto biochar derived from eucalyptus wood. Water Air Soil Pollut 228:309
Mo G, Hu Q, Wang G, Xie S, Nong H, Zhang X, Zeng T (2021) Fe3O4-modified sewage sludge biochar for U(VI) removal from aqueous solution: performance and mechanism. J Radioanal Nucl Chem 329:225–237
Monreal MJ, Diaconescu PL (2010) The riches of uranium. Nat Chem 2:424–424
Morereau A, Jaegler H, Hain K, Steier P, Golser R, Beaumais A, Lepage H, Eyrolle F, Grosbois C, Cazala C, Gourgiotis A (2022) Deciphering sources of U contamination using isotope ratio signatures in the Loire River sediments: exploring the relevance of 233U/236U and stable Pb isotope ratios. Chemosphere 307:135658
Mousavi SM, Srivastava AK, Cheraghi M (2023) Soil health and crop response of biochar: an updated analysis. Arch Agron Soil Sci 69:1085–1110
Mukherjee A, Zimmerman AR, Harris W (2011) Surface chemistry variations among a series of laboratory-produced biochars. Geoderma 163:247–255
Narasimharao K, Angaru GKR, Momin ZH, Al-Thabaiti S, Mokhtar M, Alsheshri A, Alfaifi SY, Koduru JR, Chang Y-Y (2023) Orange waste Biochar-Magnesium Silicate (OBMS) composite for enhanced removal of U(VI) ions from aqueous solutions. J Water Process Eng 51:103359
Nduka JK, Umeh TC, Kelle HI, Ozoagu PC, Okafor PC (2022) Health risk assessment of radiation dose of background radionuclides in quarry soil and uptake by plants in Ezillo-Ishiagu in Ebonyi South-Eastern Nigeria. Case Stud Chem Environ Eng 6:100269
Neiva AMR, Albuquerque MTD, Antunes IMHR, Carvalho PCS, Santos ACT, Boente C, Cunha PP, Henriques SBA, Pato RL (2019) Assessment of metal and metalloid contamination in soils trough compositional data: the old Mortórios uranium mine area, central Portugal. Environ Geochem Health 41:2875–2892
Ouyang J, Liu Z, Ye T, Zhang L (2019) Uranium pollution status and speciation analysis in the farmland-rice system around a uranium tailings mine in southeastern China. J Radioanal Nucl Chem 322:1011–1022
Ouyang P, Narayanan M, Shi X, Chen X, Li Z, Luo Y, Ma Y (2023) Integrating biochar and bacteria for sustainable remediation of metal-contaminated soils. Biochar 5:63
Pang H, Zhang E, Zhang D, Wang X, Zhao B, Liu L, Ma X, Song G, Yu S (2022) Precursor impact and mechanism analysis of uranium elimination by biochar supported sulfurized nanoscale zero-valent iron. J Environ Chem Eng 10:107288
Papp Z, Dezső Z, Daróczy S (2002) Significant radioactive contamination of soil around a coal-fired thermal power plant. J Environ Radioact 59:191–205
Pehlivan N, Gedik K, Wang JJ (2023) Tea-based biochar-mediated changes in cation diffusion homeostasis in rice grown in heavy metal (loid) contaminated mining soil. Plant Physiol Biochem 201:107889
Pehoiu G, Radulescu C, Murarescu O, Dulama ID, Bucurica IA, Teodorescu S, Stirbescu RM (2019) Health risk assessment associated with abandoned copper and uranium mine tailings. Bull Environ Contam Toxicol 102:504–510
Peña J, Straub M, Flury V, Loup E, Corcho J, Steinmann P, Bochud F, Froidevaux P (2020) Origin and stability of uranium accumulation-layers in an Alpine histosol. Sci Total Environ 727:138368
Peng Z, Cao J (2021) Natural uranium-bearing nanoparticles in surface media. Environ Chem Lett 19:2713–2721
Pérez-Sánchez D, Thorne MC (2014) An investigation into the upward transport of uranium-series radionuclides in soils and uptake by plants. J Radiol Prot 34:545
Philippou M, Pashalidis I, Theocharis CR (2022) Uranium Isotope (U-232) removal from waters by biochar fibers: an adsorption study in the sub-picomolar concentration range. Molecules 27:6765
Plant J, Reeder S, Salminen R, Smith D, Tarvainen T, De Vivo B, Petterson M (2003) The distribution of uranium over Europe: geological and environmental significance. Appl Earth Sci (transactions of the Institute of Mining and Metallurgy B) 112:B1–B17
Qi X, Gou J, Chen X, Xiao S, Ali I, Shang R, Wang D, Wu Y, Han M, Luo X (2021) Application of mixed bacteria-loaded biochar to enhance uranium and cadmium immobilization in a co-contaminated soil. J Hazard Mater 401:123823
Qi X, Xiao S, Chen X, Ali I, Gou J, Wang D, Zhu B, Zhu W, Shang R, Han M (2022) Biochar-based microbial agent reduces U and Cd accumulation in vegetables and improves rhizosphere microecology. J Hazard Mater 436:129147
Qiu B, Tao X, Wang H, Li W, Ding X, Chu H (2021) Biochar as a low-cost adsorbent for aqueous heavy metal removal: a review. J Anal Appl Pyrol 155:105081
Qiu M, Liu L, Ling Q, Cai Y, Yu S, Wang S, Fu D, Hu B, Wang X (2022) Biochar for the removal of contaminants from soil and water: a review. Biochar 4:19
Ramadan RS, Dawood YH, Yehia MM, Gad A (2022) Environmental and health impact of current uranium mining activities in southwestern Sinai, Egypt. Environ Earth Sci 81:213
Rani N, Singh P, Kumar S, Kumar P, Bhankar V, Kamra N, Kumar K (2023) Recent advancement in nanomaterials for the detection and removal of uranium: a review. Environ Res 234:116536
Ren J, Huang H, Zhang Z, Xu X, Zhao L, Qiu H, Cao X (2023) Enhanced microbial reduction of Cr(VI) in soil with biochar acting as an electron shuttle: crucial role of redox-active moieties. Chemosphere 328:138601
Rizwan M, Ali S, Abbas T, Rehman MZu, Al-Wabel MI (2018) Residual impact of biochar on cadmium uptake by rice (Oryza sativa L.) grown in Cd-contaminated soil. Arab J Geosci 11:630
Ruan Y, Zhang H, Yu Z, Diao Z, Song G, Su M, La H, Chen D, Wang S, Kong L (2022) Phosphate enhanced uranium stable immobilization on biochar supported nano zero valent iron. J Hazard Mater 424:127119
Rushimisha IE, Li X, Han T, Chen X, Abdoul Magid ASI, Sun Y, Li Y (2022) Application of biochar on soil bioelectrochemical remediation: behind roles, progress, and potential. Crit Rev Biotechnol 44(1):120–138
Sahoo SK, Hosoda M, Kamagata S, Sorimachi A, Ishikawa T, Tokonami S, Uchida S (2011) Thorium, uranium and rare earth elements concentration in weathered Japanese soil samples. Prog Nucl Sci Technol 1:416–419
Sapre S, Gontia-Mishra I, Tiwari S (2022) Plant growth-promoting Rhizobacteria ameliorates salinity stress in pea (Pisum sativum). J Plant Growth Regul 41:647–656
Sarthou MCM, Devime F, Baggio C, Figuet S, Alban C, Bourguignon J, Ravanel S (2022) Calcium-permeable cation channels are involved in uranium uptake in Arabidopsis thaliana. J Hazard Mater 424:127436
Satpathy A, Sharma N, Pan W, Catalano JG, Giammar DE (2023) Exchange between dissolved U(VI) and adsorbed and precipitated forms of solid-associated U. ACS Earth Space Chem 7:1528–1535
Schilz JR, Dashner-Titus EJ, Simmons KA, Erdei E, Bolt AM, MacKenzie DA, Hudson LG (2022) The immunotoxicity of natural and depleted uranium: from cells to people. Toxicol Appl Pharmacol 454:116252
Schipper LA, Sparling GP, Fisk LM, Dodd MB, Power IL, Littler RA (2011) Rates of accumulation of cadmium and uranium in a New Zealand hill farm soil as a result of long-term use of phosphate fertilizer. Agicr Ecosyst Environ 144:95–101
Shahandeh H, Hossner L (2002) Role of soil properties in phytoaccumulation of uranium. Water Air Soil Pollut 141:165–180
Sharma DA, Keesari T, Pant D, Rishi MS, Sangwan P, Thakur N, Sinha UK (2022) Appraising the factors favouring uranium mobilization and associated health risk assessment in groundwaters of north-western India. Ecotoxicol Environ Saf 229:113086
Shen C, Pan J, Chen M, Su M, Chen D, Song G (2023) Statistically and visually analyzing the latest advancements and future trends of uranium removal. Environ Res 239:117280
Singh H, Northup BK, Rice CW, Prasad PVV (2022) Biochar applications influence soil physical and chemical properties, microbial diversity, and crop productivity: a meta-analysis. Biochar 4:8
Skipperud L, Strømman G, Yunusov M, Stegnar P, Uralbekov B, Tilloboev H, Zjazjev G, Heier LS, Rosseland BO, Salbu B (2013) Environmental impact assessment of radionuclide and metal contamination at the former U sites Taboshar and Digmai, Tajikistan. J Environ Radioact 123:50–62
Srivastava RR, Pathak P, Perween M (2020) Environmental and health impact due to uranium mining. In: Gupta DK, Walther C (eds) Uranium in plants and the environment. Springer International Publishing, Cham, pp 69–89
Stasi C, Georgiou E, Ioannidis I, Pashalidis I (2022) Uranium removal from laboratory and environmental waters by oxidised biochar prepared from palm tree fibres. J Radioanal Nucl Chem 331:375–381
Sun T, Levin BD, Guzman JJ, Enders A, Muller DA, Angenent LT, Lehmann J (2017) Rapid electron transfer by the carbon matrix in natural pyrogenic carbon. Nat Commun 8:14873
Sun Y, Zeng B, Dai Y, Liang X, Zhang L, Ahmad R, Su X (2022) Modification of sludge-based biochar using air roasting-oxidation and its performance in adsorption of uranium(VI) from aqueous solutions. J Colloid Interface Sci 614:547–555
Takeda A, Tsukada H, Takaku Y, Si H, Nanzyo M (2006) Accumulation of uranium derived from long-term fertilizer applications in a cultivated Andisol. Sci Total Environ 367:924–931
Tan X-F, Zhu S-S, Wang R-P, Chen Y-D, Show P-L, Zhang F-F, Ho S-H (2021) Role of biochar surface characteristics in the adsorption of aromatic compounds: pore structure and functional groups. Chin Chem Lett 32:2939–2946
Tan S, Narayanan M, Thu Huong DT, Ito N, Unpaprom Y, Pugazhendhi A, Lan Chi NT, Liu J (2022) A perspective on the interaction between biochar and soil microbes: a way to regain soil eminence. Environ Res 214:113832
Taskin MB, Kadioglu YK, Sahin O, Inal A, Gunes A (2019) Effect of acid modified biochar on the growth and essential and non- essential element content of bean, chickpea, soybean, and maize grown in calcareous soil. Commun Soil Sci Plant Anal 50:1604–1613
Thakur A, Kumar R, Sahoo PK (2022) Uranium and fluoride removal from aqueous solution using biochar: a critical review for understanding the role of feedstock types, mechanisms, and modification methods. Water 14:413
Thorgersen MP, Goff JL, Poole FL, Walker KF, Putt AD, Lui LM, Hazen TC, Arkin AP, Adams MWW (2023) Mixed nitrate and metal contamination influences operational speciation of toxic and essential elements. Environ Pollut. https://doi.org/10.1016/j.envpol.2023.122674
Tomczyk A, Sokołowska Z, Boguta P (2020) Biochar physicochemical properties: pyrolysis temperature and feedstock kind effects. Rev Environ Sci Bio/Technol 19:191–215
Tsai T-L, Lin C-C, Wang T-W, Chu T-C (2008) Radioactivity concentrations and dose assessment for soil samples around nuclear power plant IV in Taiwan. J Radiol Prot 28:347
Tunney H, Stojanović M, MrdakovićPopić J, McGrath D, Zhang C (2009) Relationship of soil phosphorus with uranium in grassland mineral soils in Ireland using soils from a long-term phosphorus experiment and a National Soil Database. J Plant Nutr Soil Sci 172:346–352
Um W, Icenhower JP, Brown CF, Serne RJ, Wang Z, Dodge CJ, Francis AJ (2010) Characterization of uranium-contaminated sediments from beneath a nuclear waste storage tank from Hanford, Washington: implications for contaminant transport and fate. Geochim Cosmochim Acta 74:1363–1380
Utermann J, Fuchs M (2008) Uranium in German soils. Loads and fate of fertilizer-derived uranium. Backhuys Publishers, Leiden, pp 978–990
Vandenhove H, Van Hees M, Wannijn J, Wouters K, Wang L (2007) Can we predict uranium bioavailability based on soil parameters? Part 2: Soil solution uranium concentration is not a good bioavailability index. Environ Pollut 145:577–586
Vodyanitskii YN (2011) Chemical aspects of uranium behavior in soils: a review. Eurasian Soil Sci 44:862–873
Wang Z, Qin H, Liu X (2019a) Health risk assessment of heavy metals in the soil-water-rice system around the Xiazhuang uranium mine, China. Environ Sci Pollut Res 26:5904–5912
Wang Z, Qin H, Wang J (2019b) Accumulation of uranium and heavy metals in the soil–plant system in Xiazhuang uranium ore field, Guangdong Province, China. Environ Geochem Health 41:2413–2423
Wang B, Li Y, Zheng J, Hu Y, Wang X, Hu B (2020) Efficient removal of U(VI) from aqueous solutions using the magnetic biochar derived from the biomass of a bloom-forming cyanobacterium (Microcystis aeruginosa). Chemosphere 254:126898
Wang C, Wang G, Xie S, Wang J, Guo Y (2022) Removal behavior and mechanisms of U(VI) in aqueous solution using aloe vera biochar with highly developed porous structure. J Radioanal Nucl Chem 331:2273–2283
Wang C, Ma R, Wang J, Zhong C, Zhao Y, Browne P (2023) Fractionations of heavy metals and their correlations with magnetic susceptibility in soil from a typical alluvial island in the lower Yangtze river, China. J Clean Prod 418:138060
Wei B, Peng Y, Lin L, Zhang D, Ma L, Jiang L, Li Y, He T, Wang Z (2023) Drivers of biochar-mediated improvement of soil water retention capacity based on soil texture: a meta-analysis. Geoderma 437:116591
Wen J, Fang Y, Zeng G (2018) Progress and prospect of adsorptive removal of heavy metal ions from aqueous solution using metal–organic frameworks: a review of studies from the last decade. Chemosphere 201:627–643
Wu C, Zhi D, Yao B, Zhou Y, Yang Y, Zhou Y (2022a) Immobilization of microbes on biochar for water and soil remediation: a review. Environ Res 212:113226
Wu Z, Firmin KA, Cheng M, Wu H, Si Y (2022b) Biochar enhanced Cd and Pb immobilization by sulfate-reducing bacterium isolated from acid mine drainage environment. J Clean Prod 366:132823
Xia D, Liu Y, Cheng X, Gu P, Chen Q, Zhang Z (2022) Temperature-tuned fish-scale biochar with two-dimensional homogeneous porous structure: a promising uranium extractant. Appl Surf Sci 591:153136
Xu N, Wei FS, Ten EJ, Chen LQ (1993) Evaluation of indigenous concentrations of uranium and thorium in soils of China. Commun Soil Sci Plant Anal 24:1795–1803
Xu Z, Xing Y, Ren A, Ma D, Li Y, Hu S (2020) Study on adsorption properties of water hyacinth-derived biochar for uranium (VI). J Radioanal Nucl Chem 324:1317–1327
Xue C, Yi X, Yang Y, Jiang F, Yin H, Dang Z (2023) Chemical speciation and distribution of adsorbed Cd(II) on goethite: influence of pH and sulfate. Environ Sci Nano 10:2500–2513
Yang G (2023) Sorption and reduction of hexavalent uranium by natural and modified silicate minerals: a review. Environ Chem Lett 21:2441–2470
Yang X, Wan Y, Zheng Y, He F, Yu Z, Huang J, Wang H, Ok YS, Jiang Y, Gao B (2019) Surface functional groups of carbon-based adsorbents and their roles in the removal of heavy metals from aqueous solutions: a critical review. Chem Eng J 366:608–621
Yang X, Dai Z, Ge C, Yu H, Bolan N, Tsang DCW, Song H, Hou D, Shaheen SM, Wang H, Rinklebe J (2023a) Multiple-functionalized biochar affects rice yield and quality via regulating arsenic and lead redistribution and bacterial community structure in soils under different hydrological conditions. J Hazard Mater 443:130308
Yang X, Wen E, Ge C, El-Naggar A, Yu H, Wang S, Kwon EE, Song H, Shaheen SM, Wang H, Rinklebe J (2023b) Iron-modified phosphorus- and silicon-based biochars exhibited various influences on arsenic, cadmium, and lead accumulation in rice and enzyme activities in a paddy soil. J Hazard Mater 443:130203
Yang Y, Zhong M, Bian X, You Y, Li F (2023c) Preparation of carbon-based material with high water absorption capacity and its effect on the water retention characteristics of sandy soil. Biochar 5:61
Ye T, Huang B, Wang Y, Zhou L, Liu Z (2020) Rapid removal of uranium(VI) using functionalized luffa rattan biochar from aqueous solution. Colloids Surf, A 606:125480
Yi P, Yan Y, Kong Y, Chen Q, Wu M, Liang N, Zhang L, Pan B (2023) The opposite influences of Cu and Cd cation bridges on sulfamethoxazole sorption on humic acids in wetting-drying cycles. Sci Total Environ 898:165547
Yin Q, Lyu P, Wang G, Wang B, Li Y, Zhou Z, Guo Y, Li L, Deng N (2022) Phosphorus-modified biochar cross-linked Mg-Al layered double-hydroxide stabilizer reduced U and Pb uptake by Indian mustard (Brassica juncea L.) in uranium contaminated soil. Ecotoxicol Environ Saf 234:113363
Ying D, Hong P, Jiali F, Qinqin T, Yuhui L, Youqun W, Zhibin Z, Xiaohong C, Yunhai L (2020) Removal of uranium using MnO2/orange peel biochar composite prepared by activation and in-situ deposit in a single step. Biomass Bioenerg 142:105772
Yu S, Wu X, Ye J, Li M, Zhang Q, Zhang X, Lv C, Xie W, Shi K, Liu Y (2022) Dual effect of acetic acid efficiently enhances sludge-based biochar to recover uranium from aqueous solution. Front Chem 10:835959
Yu C, Zhu X, Mohamed A, Dai K, Cai P, Liu S, Huang Q, Xing B (2023) Enhanced Cr(VI) bioreduction by biochar: Insight into the persistent free radicals mediated extracellular electron transfer. J Hazard Mater 442:129927
Yuan Y, Bolan N, Prévoteau A, Vithanage M, Biswas JK, Ok YS, Wang H (2017) Applications of biochar in redox-mediated reactions. Biores Technol 246:271–281
Zeng T, Mo G, Zhang X, Liu J, Liu H, Xie S (2020) U(VI) removal efficiency and mechanism of biochars derived from sewage sludge at two pyrolysis temperatures. J Radioanal Nucl Chem 326:1413–1425
Zhang C, Yu Z-g, Zeng G-m, Jiang M, Yang Z-z, Cui F, Zhu M-y, Shen L-q, Hu L (2014) Effects of sediment geochemical properties on heavy metal bioavailability. Environ Int 73:270–281
Zhang H, Ruan Y, Liang A, Shih K, Diao Z, Su M, La H, Chen D, Lu H, Kong L (2019a) Carbothermal reduction for preparing nZVI/BC to extract uranium: insight into the iron species dependent uranium adsorption behavior. J Clean Prod 239:117873
Zhang P, Qi X, Wen J, Liu J (2019b) Effect and mechanism of mixed biochar with rice straw and pig manure on remediation of uranium contaminated soil by Lolium Perenne. J Univ S China 33:90–96
Zhang S, Yang F, Cao X, Tang Y, Yin T, Bo T, Liu Y, Lisak G, Kano N, Na B, Chang M, Liu Y (2023) Enhanced uranium separation by charge enabling γ-MnO2 with oxygen vacancies. J Hazard Mater 459:132112
Zhao L, Cao X, Zheng W, Wang Q, Yang F (2015) Endogenous minerals have influences on surface electrochemistry and ion exchange properties of biochar. Chemosphere 136:133–139
Zhao Q, Xu Z, Yu Z (2021) Straw-derived biochar as the potential adsorbent for U(VI) and Th(IV) removal in aqueous solutions. Biomass Convers Biorefinery 13:15707–15718
Zheng X, Xu W, Dong J, Yang T, Shangguan Z, Qu J, Li X, Tan X (2022) The effects of biochar and its applications in the microbial remediation of contaminated soil: a review. J Hazard Mater 438:129557
Zhou P, Gu B (2005) Extraction of oxidized and reduced forms of uranium from contaminated soils: effects of carbonate concentration and pH. Environ Sci Technol 39:4435–4440
Zhou Y, Xiao J, Hu R, Wang T, Shao X, Chen G, Chen L, Tian X (2020) Engineered phosphorous-functionalized biochar with enhanced porosity using phytic acid-assisted ball milling for efficient and selective uptake of aquatic uranium. J Mol Liq 303:112659
Zhu J, Wei R, Wang X, He D, Jiang X, Wang M, Yang Y, Yang L (2023a) Polyphosphate promotes oxidation resistance of ppk-expressing transgenic rice in low phosphorus culture. Plant Physiol Biochem 203:108006
Zhu K, Xian H, Peng L, Wang S, Chen C, Liu J (2023b) Adsorption performance and mechanism of MoS2/BC composite for U(VI) from aqueous solution. J Radioanal Nucl Chem 332:1743–1754
Zou Z, Yang L, Liu Y, Zhang Y, Cao D, Du Z, Jin J (2023) Removal and recovery of uranium(VI) from aqueous solutions by residual sludge and its biochars. Environ Sci Pollut Res 30:19907–19917
Acknowledgements
We would like to thank the College of Environment and Resource, Xichang University, China.
Funding
This study was supported by the National Natural Science Foundation of China (U21A20237, 41977031, 41402248); the Key R&D Projects of Liangshan Prefecture Science and Technology Plan (22ZDYF0162, 22ZDYF0171); the Ministry of Science and Technology of the People’s Republic of China (2019YFC1803500, 2019YFC1803504); the Key R&D Projects of Sichuan Provincial Science and Technology Department (2018SZ0298, 2023YFS0390); the Bureau of Science and Technology Aba Qiang Tibetan Autonomous Prefecture (R22YYJSYJ0004, R23YYJSYJ0010).
Author information
Authors and Affiliations
Contributions
FH: data curation, investigation, visualization, writing- original draft. FD: writing-review and editing. LC: conceptualization, writing—review and editing. YZ: writing-review and editing. LZ: writing—review and editing. SS: writing-review and editing. ZW: writing-review and editing. JL: writing-review and editing. LF: conceptualization, supervision, writing-review and editing. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Handling editor: Xing Yang
Supplementary Information
Additional file 1: Text S1.
Literature search. Text S2. Methods of meta-analysis. Table S1. Basic information of the publications extracted for this meta-analysis.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Huang, F., Dong, F., Chen, L. et al. Biochar-mediated remediation of uranium-contaminated soils: evidence, mechanisms, and perspectives. Biochar 6, 16 (2024). https://doi.org/10.1007/s42773-024-00308-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42773-024-00308-3