Abstract
Purpose
The aim of this study was to confirm the impact of heat acclimation on aerobic performance in hot conditions and elucidate the transfer of heat adaptations to cool and hypoxic environments.
Methods
Ten males (VO2peak: 4.50 ± 0.50 L/min) completed two three-week interventions consisting of heat acclimation (HA: 36°C and 59% RH) and temperate training (TEMP: 18°C and 60% RH) in a counter-balanced crossover design. Training weeks consisted of four work-matched controlled heart rate sessions interspersed with one intermittent sprint session, and two rest days. Before and after the interventions VO2peak and 20-min time trial performance were evaluated in COOL (18°C), HOT (35°C) and hypoxic (HYP: 18°C and FiO2: 15.4%) conditions.
Results
Following HA, VO2peak increased significantly in HOT (0.24 L/min [0.01, 0.47], P = 0.040) but not COOL (P = 0.431) or HYP (P = 0.411), whereas TEMP had no influence on VO2peak (P ≥ 0.424). Mean time trial power output increased significantly in HOT (20 W [11, 28], P < 0.001) and COOL (12 W [4, 21], P = 0.004), but not HYP (7 W [−1, 16], P = 0.075) after HA, whereas TEMP had no influence on mean power output (P ≥ 0.110). Rectal (−0.13°C [−0.23, −0.03], P = 0.009) and skin (−0.7°C [−1.2, −0.3], P < 0.001) temperature were lower during the time trial in HOT after HA, whereas mean heart rate did not differ (P = 0.339).
Conclusions
HA improved aerobic performance in HOT in conjunction with lower thermal strain and enhanced cardiovascular stability (similar heart rate for higher workload), whereas the mechanistic pathways improving performance in COOL and HYP remain unclear.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Heat acclimation is known to improve peak oxygen uptake (VO2peak) and endurance performance in hot conditions [20, 25, 49]. These improvements stem from integrative adaptations that enhance heat dissipation, fluid balance, cardiovascular stability, and skeletal muscle metabolism under heat stress [38, 48]. However, whether these adaptations improve exercise performance in cool conditions remains a topic of debate [30, 34]. Similarly, while some have shown heat acclimation to benefit endurance performance in hypoxic conditions [11, 23], others have not [55].
Following ~ 1–3 weeks of heat acclimation, significant improvements in VO2peak [25, 49, 54, 57], power output at VO2peak (Wpeak) [31, 32, 44], lactate threshold [25, 31, 44] and endurance performance [25, 27, 44, 51] have been reported in cool conditions. The proposed pathways via which performance in cooler environments may be improved following heat acclimation include a lower metabolic rate during exercise at a given workload [48, 67, muscle glycogen sparing [5, 67], increased lactate threshold [25, 67], plasma volume (PV) expansion [52, 66], improved myocardial function (in rodent models) [15, 16], and increased skeletal muscle force generation [21, 42]. Despite the potential for these pathways to mediate performance improvements, some studies have reported that VO2peak and time trial performance in cool conditions remain unchanged in well-trained male cyclists following 10–14 days of heat acclimation/acclimatization [19, 20, 55]. As such, the transfer of benefits stemming from training in the heat to performance in cooler environments remains contentious. To elucidate the transferability of any training effect, heat acclimation studies with a control group (i.e. training in temperate conditions) and performance tests conducted in both hot and cool conditions are required.
The transfer of heat adaptations to performance in hypoxic conditions (i.e. cross-adaptation) has gained interest in recent years [4, 7, 62, 63]. Heled et al [11] were the first to examine the potential for heat acclimation to improve performance in hypoxia. The authors reported that VO2peak in hypoxia (FiO2: 0.15) was unaffected after 12 days of heat acclimation, but that the onset of blood lactate accumulation occurred at a higher heart rate and oxyhemoglobin saturation (SpO2) was elevated when walking at 7 km/h. Similarly, White et al [61] reported that VO2peak was unaffected in hypoxia (FiO2: 0.12) following 10 days of heat acclimation, but that training in the heat might confer an improvement (~ 1.6%; P = 0.07) in time trial performance. A separate study found that time trial performance in hypoxia (FiO2: 0.14) was improved by ~ 4.8% following 10 days of heat acclimation, whereas training in cool conditions provided no performance benefit (~ 0.8%) [23]. It has also been shown that 10 days of heat acclimation had no effect on VO2peak in hypoxia (FiO2: 0.13) [55], while others reported a lower heart rate and higher SpO2 during exercise at 65% VO2peak in hypoxic conditions (FiO2: 0.12) after heat acclimation, but not after training in cool conditions [8]. The purported mechanisms associated with performance improvements in hypoxia following heat acclimation include an expansion of PV that facilitates the maintenance of cardiac output and SpO2 (i.e. O2 delivery), along with reduced body temperature and improved blood flow distribution [7, 62]. Exposure to heat stress also activates the expression of heat shock proteins (HSP), which leads to acquired thermotolerance and cellular cross-tolerance [7, 22]. The expression of HSP72 and HSP90 increases the molecular stability of hypoxia-inducible factor-1α (HIF-1α), which stimulates erythropoiesis [53] and angiogenesis [6]. Heat acclimation has thus been suggested to provide a pathway for improving performance in hypoxia via enhanced cytoprotection and improved O2 transport and delivery [7, 23, 62]. Although heat acclimation has been shown to improve certain aspects of endurance performance (e.g. higher SpO2 and lower submaximal heart rate) in hypoxic conditions, the benefits to self-paced (i.e. time trial) exercise are unclear and those to VO2peak are absent.
Therefore, the aim of this study was to elucidate the transfer of heat adaptations to VO2peak and time trial performance in cool and hypoxic conditions, and confirm the impact of heat acclimation on these parameters in hot conditions following whole-body pre-heating. A cross-over design with participants also training in temperate conditions was employed to isolate the transferability of heat acclimation adaptations from those of training, to performance in cool and hypoxic conditions. It was hypothesized that heat acclimation would improve both VO2peak and time trial performance in the heat, but only time trial performance in cool and hypoxic conditions.
Methods
Participants
Ten endurance-trained (Tier 2–3) male cyclists and triathletes participated in and completed the study [28, 36]. Their characteristics on enrolment for age, height, body mass, VO2peak and Wpeak were: 34 ± 7 years, 177 ± 6 cm, 75.6 ± 7.5 kg, 4.50 ± 0.50 L/min, and 416 ± 39 W. All participants undertook personal training regimens and regularly competed in amateur endurance cycling races. Inclusion criteria for the study included cycling at least 250 km per week and undertaking cycling training for the previous 2 years at least. None of the participants had a prior history of exertional heat illness. The project was approved by the Anti-Doping Laboratory Qatar ethics committee (F2015000201) and conformed to the standards of the Declaration of Helsinki. Written informed consent was obtained from all participants before the beginning of testing.
Experimental Design
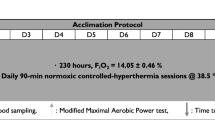
All participants completed two 3-week training interventions consisting of exercise heat acclimation (HA: 35.5 ± 1.8°C and 59.0% ± 7.7% relative humidity: RH) and work-matched exercise in temperate conditions (TEMP: 18.0 ± 0.5°C and 59.5% ± 9.9% RH) in a counter-balanced cross-over design (Fig. 1). The training interventions were separated by 21 ± 17 weeks and took place in Doha, Qatar. To minimise the influence of heat acclimatization, testing during the summer months was avoided. However, because of the high average daily peak temperatures of 23 to 42°C from the coldest to warmest months of the year in Qatar, respectively [56], participants were encouraged to minimise outdoor training to once per week for a minimum of 3 weeks before the first laboratory visit. Two participants had 4- and 5-week wash-outs due to time constraints, but these were following TEMP. Training consisted of five sessions per week including four controlled heart rate endurance sessions (days 1, 2, 4, 5, 8, 9, 11, 12, 15, 16, 18 and 19) and one intermittent sprint session (days 3, 10 and 17). Days 6, 7, 13, 14, 20 and 21 were rest days. Prior to and following HA and TEMP, participants completed three performance tests in hot (HOT: 35°C, 60% RH, 20.93% FiO2, with pre-heating to a rectal temperature (Tre) of 38.5°C), cool (COOL: 18°C, 60% RH, 20.93% FiO2), and hypoxic (HYP: 18°C, 60% RH, 15.4% FiO2; ~2500 m simulated altitude) conditions with a convective airflow of 3 m/s in each environment. Each testing session was separated by 48 h and included an incremental cycling test to exhaustion to determine VO2peak and 20-min time trial separated by 30 min of passive rest. Testing session order was randomized between participants but was maintained for each participant across interventions. The intervention was part of a larger project with physiological and hematological adaptations presented in a companion paper [40].
Schematic representation of the three-week cross-over heat acclimation and work-matched temperate training intervention. Training consisted of four controlled heart rate endurance sessions and one intermittent sprint session per week. Prior to and following each intervention, performance tests (VO2peak and 20-min time trial) were performed in hot (HOT: 35°C, 60% RH, 20.93% FiO2, with pre-heating to a rectal temperature of 38.5°C), cool (COOL: 18°C, 60% RH, 20.93% FiO2), and hypoxic (HYP: 18°C, 60% RH, 15.4% FiO2) conditions. Hemoglobin mass (Hbmass) measurements and venous blood samples were taken prior to, on days 3, 10 and 18 (Hbmass) or 4, 11 and 19 (venous blood), and following the intervention
Training Intervention
Participants attended the laboratory at the same time of day to conduct the training sessions. The controlled heart rate training sessions were designed to produce a workload that corresponded to the total work (kJ) completed in 60 min if the power output associated with 65% VO2peak was maintained. It was calculated as:
where, P65% is the power output corresponding to 65% VO2peak, 3600 is time in seconds, and 1000 is the conversion from joules to kilojoules. Total work was increased to 105% in week two and 110% in week three of the initial workload to sustain the thermal stimulus and week-to-week training duration. The same progression was applied during both training interventions. Each training session involved an initial period (20% of total work) of cycling at a constant workload equivalent to 65% VO2peak on a Lode ergometer (Corival, Groningen, Holland). Over the remainder of each session, computer software (Lode ergometry manager 9.0) adjusted resistance every 30 s so that an exercising heart rate associated with 65% VO2peak was maintained. Participants were instructed to maintain a steady cadence > 80 rev/min. The initial constant workload period was designed to increase heart rate and core temperature and promote the onset of sweating. Heart rate during HA was clamped 7 beats/min higher than in TEMP to maintain a similar relative training intensity between conditions [38, 64], in response to the direct effect of temperature (i.e. hyperthermia) on the sinoatrial node [17] and to vagal withdrawal and sympathetic activation [9].
The intermittent sprint session consisted of a 20-min warm-up followed by five all-out sprints of 30 s on an SRM cycle ergometer (Schoberer Rad Meßtechnik, Jülich, Germany) set to isokinetic mode at 105 rev/min. Participants were instructed to remain seated and were verbally encouraged throughout all sprints. The sprints were interspersed with 4 min 30 s of light resistance (~ 50 W) pedaling to reduce venous pooling in the lower extremities and minimize feelings of light-headedness or nausea. After the final sprint, participants rested for 5 min and then completed a 10-min cool-down at a light resistance (50–100 W) to ensure 60 min of heat exposure per day during HA and exercising duration was matched in TEMP training. During both the intermittent sprint and controlled heart rate training sessions participants consumed plain water ad libitum, participants were encouraged to drink an amount estimated from the previous days sweat rate to minimize post-training body mass losses to < 1%.
Performance Tests
Participants attended the laboratory at the same time of day and were instructed to ingest 7 mL/kg body mass of water 1 h prior to the start of the test. Upon arrival to the laboratory, participants were given a Tre probe to self-insert and asked to provide a urine sample for the measurement of urine specific gravity (USG: Pocket refractometer PAL-10S, Atago, Tokyo, Japan). A USG > 1.020 was considered a marker of hypohydration and participants were asked to consume additional water (300 mL) prior to commencing the trial. After instrumentation (heart rate and skin temperature) and baseline measures, participants rested in the environmental chamber for 15 min in the COOL and HYP trials prior to commencing the incremental cycling test. In the HOT trial, participants lay supine in a temperature-controlled bath (41.9 ± 0.9 °C) inside the environmental chamber set to HOT conditions until reaching a Tre of 38.5°C. Pre-heating was conducted to ensure participants undertook the performance tests in HOT in a hyperthermic state. Participants then towel dried, changed into cycling attire (cycling shorts, socks and shoes) and commenced the incremental cycling test 5 min after exiting the bath. The ramp protocol commenced with unloaded pedaling and increased by 1 W every 2 s (30 W/min) until volitional fatigue on the Lode ergometer. Oxygen uptake was measured continuously using an online breath-by-breath cardiopulmonary system (Oxycon Pro, CareFusion, Rolle, Switzerland). VO2peak and Wpeak were determined as the highest 30 s average of the second-by-second VO2 and power output data, respectively.
Following the incremental test, participants recovered in the environmental chamber for 30 min, which included 20 min of passive rest and a 10-min self-selected warm-up, before performing the 20-min time trial. In the HOT trial, ambient temperature (37.4 ± 2.7 °C) during the 30-min recovery period was adjusted to maintain Tre at ~ 38.5°C prior to commencing the time trial, which was performed on the SRM ergometer. Participants were asked to maintain the highest sustainable effort for 20 min, which simulates the demands of a 16.1-km (10-mile) time trial. Feedback during the time trial was limited to the time remaining, with no other information provided. Participants were permitted to drink water ad libitum. A similar protocol with trained cyclists in our laboratory reported a coefficient of variation of 1.1% in performance [2].
Physiological and Perceptual Measurements
Body core temperature was continuously monitored with a Tre probe (MRB, Ellab A/S, Hillerød, Denmark) inserted 15 cm beyond the anal sphincter and attached to a precision digital thermometer allowing measurement to the nearest 0.1°C (DM 852, Ellab A/S, Hillerød, Denmark). Skin temperature was monitored at four sites with iButton™ temperature sensors/data loggers (Maxim Integrated Products, Sunnyvale, CA) used to calculate mean skin temperature (Tsk) [43]. Temperature measurements were taken during the resting baseline period prior to the VO2peak test and at 2 min intervals during the test. SpO2 was measured via pulse oximetry (8000SL; Nonin Medical Inc, Plymouth, MN) on the right middle finger at the same intervals. During the 30-min recovery period and the time trial Tre and Tsk measures were taken at 5 min intervals and SpO2 upon completion. Heart rate was monitored continuously with a telemetric chest-strap transmitter (T-31 Polar Electro, Lake Success, NY) with resting heart rate determined as the average of 1 min after a 10-min seated period. Participants were asked to keep a 24-h food diary before testing and to replicate their diet before the second and third trials.
Hematological Measures
Hemoglobin mass (Hbmass) was determined via the modified optimized CO rebreathing technique [50]. Briefly, participants rebreathed a 1.2 mL/kg bolus of CO for 2 min after a period of seated rest. Arterialized capillary fingertip blood samples were analyzed in quintuplicate (ABL 90 FLEX, Radiometer, Brønshøj, Denmark) prior to and 7 min following the start of the rebreathing period to determine the percentage of hemoglobin saturated with CO. Expired CO was determined using a Draeger Pac 7000 (Lubeck, Germany). Absolute Hbmass (in g) was then calculated following corrections for remaining CO in the rebreathing apparatus, washout of CO following the procedure and estimates of residual lung volume. Between-day duplicate measurements were taken prior to (pre) and after (post) each intervention. In any case where Hbmass differed by > 2% between duplicate measurements, the test was repeated. During the intervention, single Hbmass measurements were taken on days 3, 10 and 18 (represented as days 4, 11 and 19 in Table 2). Hbmass was used to calculate blood volume (BV), red cell volume (RCV) and plasma volume (PV) using hemoglobin concentration (Hb) and hematocrit (Hct) values from venous blood samples (Beckman Coulter DxH 800, Beckman Coulter, Miami, FL) collected following 10 min of seated rest prior to training on days 4, 11 and 19 using the formulas:
Statistical Analyses
All statistical analyses were performed in R statistical software (v 4.2.1) [39] and data are presented as means and 95% confidence intervals [CI], unless otherwise indicated. Linear mixed effect models were estimated using the ‘lme4’ package [1], utilizing restricted maximum likelihood and ‘nloptwrap’ optimizer, with each model including participant ID as a random intercept. Q-Q plots and histograms were used to assess data normality. For the analysis of VO2peak, Wpeak, peak heart rate and Tsk during the incremental test to exhaustion, linear mixed effect models were implemented with testing environment (COOL, HYP, HOT), training condition (TEMP, HA) and training status (pre, post) used as fixed effects. Training status (pre, post) was implemented as an ordered factor. Initial and final Tre and SpO2 recorded during the incremental test were analyzed using the same random and fixed effects as the incremental exercise test outcome variables, except time was added as an ordered fixed effect with two levels (initial, final). For the analysis of power output, Tre, Tsk and heart rate during the time trial, the same random and fixed effects as the incremental exercise test outcome variables, except time was an ordered factor with four levels (4, 11, 19, Post). Hematological variables recorded at rest (i.e. PV and Hbmass) were analyzed using training condition and day of training (Pre, 4, 11, 19, Post) as fixed effects. Pairwise comparisons were conducted using the ‘emmeans’ package [24] and the Kenward-Roger method was used to approximate degrees of freedom. Significance was accepted at P < 0.05.
Results
Incremental Exercise Test
VO2peak was not significantly different between HA and TEMP (0.07 L/min [−0.03, 0.16], P = 0.171) and did not differ from pre- to post-intervention (0.06 L/min [−0.03, 0.16], P = 0.186; Fig. 2). However, VO2peak in COOL was significantly higher than in HYP (0.61 L/min [0.49, 0.72], P < 0.001) and HOT (0.28 L/min [0.16, 0.39], P < 0.001), while VO2peak in HYP was significantly lower than in HOT (−0.33 L/min [−0.44, −0.21], P < 0.001). Following HA, VO2peak increased significantly in HOT (0.24 L/min [0.01, 0.47], P = 0.040), but not in COOL (0.09 L/min [−0.14, 0.32], P = 0.431) or HYP (0.10 L/min [−0.13, 0.32], P = 0.411). Following TEMP, VO2peak did not significantly change in any condition (all P ≥ 0.424).
Peak power output, peak oxygen uptake (VO2peak) and peak heart rate during the incremental exercise test in cool (COOL), hypoxic (HYP) and hot (HOT) conditions, before and after temperate training (TEMP) and exercise heat acclimation (HA). n = 10 except for peak power output in HA (n = 9). Data presented as means (bars), 95% confidence intervals (error bars) and individual responses (black lines). * Significantly different from pre- to post-intervention (P ≤ 0.04)
Wpeak during the incremental exercise test was not significantly different between HA and TEMP (1 W [−4,7], P = 0.644), but significantly increased from pre- to post-intervention (11 W [6, 17], P < 0.001; Fig. 2). Wpeak in COOL was significantly higher than in HYP (40 W [33, 47], P < 0.001) and HOT (41 W [34, 48], P < 0.001), but there was no difference between HYP and HOT (P = 0.744). Following HA, Wpeak increased in HOT (25 W [12, 39], P < 0.001) but not in COOL (10 W [−4, 23], P = 0.159) or HYP (10 W [−4, 24], P = 0.147). Following TEMP, Wpeak was not significantly different in any condition (all P ≥ 0.170).
Peak heart rate during the incremental exercise test was not significantly different between HA and TEMP (1 beat/min [−1, 2], P = 0.477) and did not differ from pre- to post-intervention (0 beats/min [−1, 2], P = 0.667; Fig. 2). Peak heart rate in HOT was significantly higher than in COOL (3 beats/min [1, 5], P = 0.005) and HYP (7 beats/min [5, 9], P < 0.001), while peak heart rate in HYP was significantly lower than COOL (−4 beats/min [−6, −2], P < 0.001). Peak heart rate did not significantly change following either HA or TEMP in any condition (all P ≥ 0.16).
Initial Tre was similar between HA and TEMP (−0.00°C [−0.07, 0.07], P = 0.971), as well as from pre- to post-intervention (0.03°C [−0.04, 0.09], P = 0.429; Table 1). Final Tre was significantly higher in HOT compared to COOL (0.89°C [0.78, 1.01], P < 0.001) and HYP (1.02°C [0.90, 1.13], P < 0.001), while COOL was significantly higher than HYP (0.12°C [0.01, 0.24], P = 0.04). Final Tre was significantly higher than initial Tre during all incremental exercise tests (0.25°C [0.19, 0.32], P < 0.001). Tsk was not significantly different between HA and TEMP (0.1°C [−0.2, 0.3], P = 0.640), and did not differ from pre- to post-intervention (0.0°C [−0.2, 0.3], P = 0.693). Mean Tsk was significantly higher in HOT compared to COOL (4.7°C [4.4, 5.0], P < 0.001) and HYP (4.7°C [4.4, 5.0], P < 0.001), but there was no significant difference between COOL and HYP (P = 0.921).
SpO2 was significantly higher during HA than TEMP (1.2% [0.5, 1.9], P < 0.001), but did not differ from pre- to post-intervention (0.4% [−0.2, 1.1], P = 0.204; Table 1). SpO2 was significantly lower in HYP than HOT (−6.3% [−7.1, −5.5], P < 0.001) and COOL (−7.1% [−8.0, −6.3], P < 0.001), with a non-significant difference between COOL and HOT of 0.8% [0.2, 1.7] (P = 0.057). Final SpO2 was significantly lower than initial SpO2 during all incremental exercise tests (−4.8% [−5.6, −4.1], P < 0.001).
Time Trial Performance
Mean power output during the time trial was not significantly different between HA and TEMP (3 W [−0, 6], P = 0.089), but significantly increased from pre- to post-intervention (8 W [5, 12], P < 0.001); Fig. 3). Mean power output in COOL was significantly higher than in HYP (34 W [30, 38], P < 0.001) and HOT (42 W [38, 46], P < 0.001), while mean power output in HYP was significantly higher than HOT (8 W [4, 12], P < 0.001). Following HA mean power output significantly increased in COOL (12 W [4, 21], P = 0.004) and HOT (20 W [11, 28], P < 0.001), but not HYP (7 W [−1, 16], P = 0.075). Following TEMP mean power output did not significantly change in any condition (all P ≥ 0.110).
Mean power output and heart rate during the time trial in cool (COOL), hypoxic (HYP) and hot (HOT) conditions, before and after temperate training (TEMP) and exercise heat acclimation (HA). n = 10 except for mean power output in HOT following HA (n = 9). Data presented as means (bars), 95% confidence intervals (error bars) and individual responses (black lines). * Significantly different from pre- to post-intervention (P ≤ 0.004)
Mean heart rate during the time trial was not significantly different between HA and TEMP (−1 beat/min [−1, 0], P = 0.163; Fig. 3). Mean heart rate in HOT was significantly higher than in COOL (3 beats/min [2, 4], P < 0.001) and HYP (6 beats/min [5, 7], P < 0.001), while mean heart rate in HYP was significantly lower than COOL (−3 beats/min [−3, −2], P < 0.001). Mean heart rate did not significantly change in any condition following either intervention (all P ≥ 0.090).
Mean Tre during the time trial was significantly lower in HA than TEMP (−0.05°C [−0.09, −0.01], P = 0.015), but did not differ from pre- to post-intervention (−0.00°C [−0.04, 0.04], P = 0.951; Fig. 4). Mean Tre was significantly higher in HOT compared to COOL (0.44°C [0.39, 0.49], P < 0.001) and HYP (0.55°C [0.50, 0.60], P < 0.001), while mean Tre in HYP was significantly lower than in COOL (−0.11°C [−0.16, −0.06], P < 0.001). Following HA, mean Tre was significantly lower in HYP (−0.10°C [−0.20, −0.00], P = 0.044) and HOT (−0.13°C [−0.23, −0.03], P = 0.009), but not COOL (P = 0.618). Following TEMP, mean Tre was significantly higher in HYP (0.11°C [0.02, 0.21], P = 0.024) and HOT (0.16°C [0.06, 0.25], P = 0.002), but not COOL (P = 0.184).
Mean rectal temperature and skin temperature during the time trial in cool (COOL), hypoxic (HYP) and hot (HOT) conditions, before and after temperate training (TEMP) and exercise heat acclimation (HA). Data presented as means (bars), 95% confidence intervals (error bars) and individual responses (black lines). * Significantly different from pre- to post-intervention (P ≤ 0.044)
Mean Tsk during the time trial was not significantly different between HA and TEMP (0.1°C [−0.1, 0.3], P = 0.362), but did significantly decrease from pre- to post-intervention (−0.3°C [−0.4, −0.1], P = 0.003; Fig. 4). Mean Tsk was significantly higher in HOT compared to COOL (4.9°C [4.7, 5.1], P < 0.001) and HYP (5.0°C [4.8, 5.2], P < 0.001), but there was no significant difference between COOL and HYP (P = 0.339). Following HA, mean Tsk was significantly decreased in HOT (−0.7°C [−1.2, −0.3], P < 0.001), but not COOL (P = 0.085) or HYP (P = 0.145). Following TEMP, mean Tsk significantly increased in HYP (0.5°C [0.1, 0.9], P = 0.023), but not COOL (P = 0.112) or HOT (P = 0.127).
Hematological Responses
PV was not significantly different between HA and TEMP (37 mL [−30, 103], P = 0.277; Table 2). During HA, PV increased significantly from pre-intervention to day 11 (168 mL [23, 314], P = 0.024) and 19 (150 mL [0, 300], P = 0.050), as well as during TEMP from pre-intervention to day 11 (166 mL [20, 311], P = 0.027) and 19 (157 mL [7, 307], P = 0.041). There was a decrease in PV following TEMP from day 11 (161 mL [−16, −307], P = 0.030) and day 19 (153 mL [−2, −303], P = 0.046) to post-intervention. Hbmass was significantly lower in HA than TEMP (−18 g [−24, −11], P < 0.001; Table 2). During HA, Hbmass decreased significantly from pre-intervention to day 4 (−22 g [−8, −37], P = 0.003), day 11 (−17 g [−2, −32], P = 0.022) and day 19 (−21 g [−6, −36], P = 0.007); whereas there was no change in TEMP (P ≥ 0.445).
Heat Adaptations and Training
Resting heart rate decreased post-intervention in HA (−5 beats/min [−9, −2], P = 0.007)), but not TEMP (P = 0.749; Table 3). Resting Tre was significantly lower post-intervention in HA than TEMP (−0.26°C [−0.48, −0.04], P = 0.200). Mean Tre was similar during HA and TEMP (−0.02°C [−0.15, 0.10], P = 0.683) and remained unchanged from pre- to post-intervention (P ≥ 0.529). However, mean power output was significantly higher in TEMP than HA (27 W [19, 36], P < 0.001) and increased significantly from pre- to post-intervention in both TEMP (14 W [1, 27], P = 0.04) and HA (24 W [12, 36], P < 0.001). Training time was similar from pre- to post-intervention (P = 0.529), but took longer in HA (10.1 min [6.8, 13.6], P < 0.001), whereas work completed was similar between TEMP and HA (0 kJ [−3, 4], P = 0.784), but increased from pre- to post-intervention (69 kJ [66, 73], P < 0.001). Whole-body sweat rate was higher during HA than TEMP (0.86 L/h [0.71, 1.01], P < 0.001) and increased significantly from pre- to post-intervention in HA (0.38 L/h [0.17, 0.59], P = 0.001).
Discussion
To our knowledge, this is the first study to compare the impact of HA against TEMP training on VO2peak and time trial performance in HOT, COOL and HYP conditions in a group of trained cyclists. Our data demonstrate that VO2peak increased by 6% (0.24 L/min) in HOT following HA, but not in COOL or HYP. During the 20-min time trial, mean power output increased by 8% (20 W) in HOT and 4% (12 W) in COOL following HA, with a non-significant increase of 3% (7 W [−1, 16]) in HYP. The increased power output during the time trial in HOT was associated with a similar heart rate to pre-HA, lower mean Tre (0.13°C) and Tsk (0.7°C), and a higher whole-body sweat rate (during HA). Tre was also lower in HYP (0.10°C) after HA. The improvements in VO2peak in HOT and time trial performance in HOT, COOL and HYP occurred in conjunction with an increase in PV (4.2%) and reduction Hbmass (2.2%) on day 19 of HA, but a return towards baseline post-intervention (PV: 1.0%; Hbmass: −0.7%). These data confirm the benefits of HA on aerobic capacity and time trial performance in HOT and provide evidence of improvements in time trial performance in COOL and HYP. These improvements are associated with greater cardiovascular stability (i.e. higher workload for similar heart rate) and lower thermal strain during exercise in HOT, whereas the mechanistic pathways improving performance in COOL and HYP remain less clear.
Exercise Performance in a Hot Environment
The increase in VO2peak (6%) and Wpeak (7%) during the incremental test (Fig. 2) and the improvement in time trial power output (8%; Fig. 3) in HOT when initiated in a hyperthermic state after HA support previous findings. Indeed, HA has been shown to increase VO2peak by 4%–10% and Wpeak by 2%–8% following whole-body pre-heating [20, 25, 49]. Time trial performance has also been shown to increase by 8%–10% [20, 25], with a meta-analysis indicating that HA generally enhances performance under heat stress by ~ 7% [59]. Of note, the increase in VO2peak and Wpeak in HOT following HA almost fully restored the decrement in these parameters relative to COOL prior to HA, as evidenced by VO2peak increasing from 92% to 98% and Wpeak from 89% to 95% of COOL. Concurrently, time trial performance increased from 85% to 92% of that achieved in COOL prior to HA. Had the time trial in HOT not been initiated in a hyperthermic state (Tre: ~ 38.5°C, Tsk: ~ 35°C) and a warm-up routine commensurate with outdoor cycling been conducted, perhaps the initial decrement in performance would not have been as pronounced. Notwithstanding, the restoration of performance in HOT relative to COOL conditions has previously been observed in laboratory [20] and field settings [41]. In a study by Keiser et al. [20], a ~ 10% increase in VO2peak under heat stress (35ºC) following HA fully compensated for the initial reduction when compared to values obtained in 18°C, whereas Wpeak and 30-min time trial power output were almost completely restored (both ~ 98%). Racinais et al [41] reported that following two weeks of heat acclimatization, trained cyclists could produce a similar time (~ 66 min) over 43 km in hot (36°C) as in cold (~ 8°C) conditions. The 15% improvement in time was associated with a 15% improvement in mean power output (256 to 294 W) relative to an initial time trial conducted in the heat at the start of acclimatization, although still 3% lower than mean power output (304 W) in cold conditions. Our data therefore reaffirm the impact of HA on aerobic capacity and performance under heat stress.
The pathway via which improvements in aerobic capacity and performance under heat stress occur involves integrative adaptations that include a lowered resting body temperature, improved thermoregulatory responses, increased total body water and expanded PV, improved skeletal muscle metabolism, enhanced cardiovascular stability, and increased thermal tolerance [14, 38]. In the current study, Tre and Tsk, heart rate and SpO2 during the incremental test were similar prior to and following HA (Table 1), but Wpeak increased by 25 W and VO2peak by 0.24 L/min. Similarly, time trial performance in HOT was improved by 20 W after HA, concomitant with a lower mean Tre (0.13°C) and Tsk (0.7°C), along with a similar heart rate to pre-HA (Figs. 2, 3). These improvements in VO2peak and performance reflect an enhanced capacity to both thermoregulate and maintain O2 delivery during exercise at a high workload. However, while we noted a 150 mL expansion of PV on the final day of HA (i.e. day 19), which has been suggested to increase vascular filling pressure to support cardiovascular stability (i.e. increase stroke volume and maintain cardiac output and arterial blood pressure) [33, 52, 65], post-HA measurements indicate that PV was only ~ 37 mL higher than pre-HA.
Previous studies have highlighted the potential for PV expansion following HA to increase VO2peak in the heat by increasing cardiac output [25], but also that no association was found between changes in PV and time trial performance [20]. In cool conditions, acute (e.g. dextran, albumin infusion) PV expansion (200–400 mL) has been shown to improve cardiovascular stability and increase VO2peak in untrained and moderately active individuals [3, 10, 12]. In contrast, cardiovascular stability was not affected in endurance trained individuals following an acute 400 mL expansion [13]. During exercise in the heat, a 13%–15% acute expansion of PV (500–600 mL) has been shown not to enhance VO2peak and self-paced exercise performance, despite maximal cardiac output increasing [20, 60]. The lack of improvement in performance has been attributed to the concomitant hemodilution that accompanies a large acute PV expansion, which may offset any perfusion and O2 delivery improvements to exercising muscles, especially in endurance trained individuals [3, 18]. In the current study, the ~ 4% expansion of PV on day 19 occurred in conjunction with a ~ 2% decrease in Hbmass (i.e. ~ 3% decrease in Hb concentration) and a return towards baseline values post-HA (Table 2). As such, the combination of a lower thermal strain experienced during exercise in HOT following HA, in response to an improved whole-body sweat rate (Table 3), along with attenuated sinoatrial node and sympathetic activity [9, 17], appears to have contributed to improve VO2peak and time trial performance.
Exercise Performance in a Cool Environment
This study used a counter-balanced cross-over design in which absolute workload and relative exercise intensity where matched between HA and COOL. The unique approach allowed for isolating the effects of training in the heat from those of training in cool conditions on the adaptive process and transferability of adaptations to performance in hot, cool and hypoxic conditions. Our data indicate that VO2peak in COOL was not enhanced following HA (Fig. 2). This finding is in line with three previous reports of a lack of increase in VO2peak in well-trained (VO2peak > 60 mL/min/kg) individuals following 8–14 days of HA, with two studies noting a PV expansion of 6% and 15% [19, 20, 54]. The expansion of PV in these studies was larger than in the current study on day 19 of HA (4.2%) and following the intervention (1%). In contrast, others have demonstrated a 4% to 13% improvement in VO2peak in less trained participants (VO2peak 48–55 mL/min/kg), with two studies reporting a 3% and 8% PV expansion [49, 54, 57, 58]. However, in similarly trained participants (VO2peak: 41–61 mL/min/kg), a PV expansion of 3.7% did not improve VO2peak [55]. Of note, Lorenzo et al [25] observed a 5% increase in VO2peak in trained athletes (VO2peak: 67 mL/min/kg) following a 10-day HA regimen, which the authors attributed in part to a moderate (6.5%) expansion of PV and increase in maximal cardiac output (9%), concomitant to a small (3.5%) hemodilution. Across individuals of different aerobic fitness, it appears that the potential for PV expansion to increase VO2peak may relate to the balance between an increase in stroke volume and cardiac output, and a decrease in hemoglobin concentration (i.e. hemodilution), and thus arterial O2 content [3]. Despite the lack of change in VO2peak in the current study, 20-min time trial performance was improved by 4% (12 W) in COOL following HA (Fig. 3), a finding supported by previous work with HA regimens of 10 to 21 days [25, 27, 44, 51].
Recently, Lundby et al [26] reported that VO2peak in cool conditions increased by 6% and 4% in elite female (VO2peak 59 mL/min/kg) and male (VO2peak 75 mL/min/kg) cyclists after longer-term HA (i.e. 5 × 50 min session per week for 5 week), respectively. The authors also reported a 10% and 7% improvement in 15-min time trial power output in females and males, along with a 6% and 4% increase in power output at lactate threshold (i.e. 3 mmol/L), respectively. These improvements correlated with a 4% increase in Hbmass in both sexes, whereas skeletal muscle properties (i.e. citrate synthase activity, fiber type distribution, and capillary density) were unaffected [26]. Although these data suggest that prolonged HA-induced increases in Hbmass contribute to improve aerobic capacity and performance in cool conditions, similar long-term HA studies with elite male cyclists (VO2peak: 77–78 mL/min/kg) from the same research group failed to show greater improvements in VO2peak, 15-min time trial performance and power output at 4 mmol/L following HA compared to cool training, despite a 2%–5% increase in Hbmass after HA and either a 5% increase or 3% decrease in PV [45, 46]. A lack of change in VO2peak and Wpeak in cool conditions following 5.5 weeks of HA was also reported in well-trained sub-elite male cyclists (VO2peak: 60 mL/min/kg), along with a similar improvement (6%) in 15-km time trial performance to that of training in cool conditions [29]. In a companion paper, it was reported that Hbmass and PV increased in response to HA and cool training, with larger but non-significant increases following HA than training in cool conditions (3% vs. 0.2%; 8% vs. 5%, respectively) [35]. The lack of consistent benefit from HA across studies may stem from low statistical power and the potential for marginal performance differences to be observed in elite athletes [26]. In the current 3-week HA study, Hbmass decreased (~ 2%) during HA and returned to pre-intervention values after HA, whereas PV experienced the opposite pattern (Table 2). As such, the 4% improvement in time trial performance noted in COOL following HA may relate to the integrated adaptations highlighted to enhance performance in HOT, along with adaptations not measured in the current study and requiring further investigation, including improved efficiency, lactate threshold and cardiovascular function.
Exercise Performance in a Hypoxic Environment
VO2peak in HYP (FiO2: 15.4%) was not significantly improved following HA in the current study (Fig. 2). The lack of increase in VO2peak at altitude (FiO2: 0.12 to 0.15) following HA corroborates previous findings [11, 55, 61]. Similarly, 20-min time trial performance in HYP did not increase significantly following HA (Fig. 3). However, the 7 W ([−1, 16], P = 0.075) difference in self-paced exercise performance aligns with data from White et al [61] who reported a ‘possible’ HA benefit (~ 2%, P = 0.07) to 16-km time trial performance at altitude (FiO2: 0.12), along with the significant improvement (~ 5%) noted by Lee et al [23] at an FiO2 of 0.14 over the same distance.
Based on previous findings, improvements in time trial performance at altitude may stem from an elevated SpO2 and/or lower heart rate during exercise at a given submaximal workload [8, 11]. Although SpO2 was not measured during the time trials in the current study, mean heart rate was similar from pre- to post-intervention, including in HYP, which suggests greater cardiovascular stability following HA for a slightly, albeit not significantly, higher mean power output (7 W [−1, 16]). Moreover, while mean Tre was 0.10ºC lower during the time trial in HYP following HA, it is unlikely to have meaningfully influenced performance (Fig. 4). As such, the mechanistic pathways mediating the benefit of HA on performance in HYP remain to be elucidated. Potential pathways include acquired thermotolerance and cellular cross-tolerance [7, 22], as well as erythropoiesis [53] and angiogenesis [6] stemming from the expression of HSP increasing the molecular stability of HIF-1α. However, as we noted a decrease in Hbmass after HA, enhanced O2 transport and delivery are unlikely to have mediated any improvement in endurance performance in HYP. As such, the benefits of HA on endurance performance in HYP remain unclear and require further investigation, with longer (e.g. 5-week) HA regimens potentially providing insight into how increases in Hbmass might enhance performance.
Conclusion
Heat acclimation significantly improved VO2peak in HOT conditions and time trial performance in HOT and COOL, with a non-significant change in HYP of 7 W [−1, 16]. In contrast, training in temperate conditions, which was matched for both absolute and relative training workload with HA, did not affect aerobic capacity and time trial performance. While there were links between performance and greater cardiovascular stability and lower thermal strain in HOT following HA, the benefits of heat adaptations on endurance performance in COOL and HYP are less clear. This lack of clarity raises further questions regarding the mechanisms that support endurance performance improvements in cool and hypoxic conditions after heat acclimation.
Data availability
Data from the current study are available from the corresponding author on reasonable request.
References
Bates D, Maechler M, Bolker B, Walker S, Christensen RHB, Singmann H, Dai B, Scheipl F, Grothendieck G, Green P. Package ‘lme4’. 2009. http://www.lme4r-forger-projectorg. Accessed 16 Jan 2024.
Cocking S, Wilson MG, Nichols D, Cable NT, Green DJ, Thijssen DHJ, Jones H. Is there an optimal ischemic-preconditioning dose to improve cycling performance? Int J Sport Physiol Perform. 2018;13(3):274–82.
Coyle EF, Hopper MK, Coggan AR. Maximal oxygen uptake relative to plasma volume expansion. Int J Sports Med. 1990;11(2):16–119.
Ely BR, Lovering AT, Horowitz M, Minson CT. Heat acclimation and cross tolerance to hypoxia: bridging the gap between cellular and systemic responses. Temperature (Austin). 2014;1(2):107–14.
Febbraio MA, Snow RJ, Hargreaves M, Stathis CG, Martin IK, Carey MF. Muscle metabolism during exercise and heat stress in trained men: effect of acclimation. J Appl Physiol. 1994;76(2):589–97.
Fong G-H. Mechanisms of adaptive angiogenesis to tissue hypoxia. Angiogenesis. 2008;11(2):121–40.
Gibson OR, Taylor L, Watt PW, Maxwell NS. Cross-adaptation: heat and cold adaptation to improve physiological and cellular responses to hypoxia. Sports Med. 2017;47(9):1751–68.
Gibson OR, Turner G, Tuttle JA, Taylor L, Watt PW, Maxwell NS. Heat acclimation attenuates physiological strain and the HSP72, but not HSP90alpha, mRNA response to acute normobaric hypoxia. J Appl Physiol. 1985;119(8):889–99
Gorman AJ, Proppe DW. Mechanisms producing tachycardia in conscious baboons during environmental heat stress. J Appl Physiol Respir Environ Exerc Physiol. 1984;56(2):441–6.
Grant SM, Green HJ, Phillips SM, Sutton JR. Effects of acute expansion of plasma volume on cardiovascular and thermal function during prolonged exercise. Eur J Appl Physiol Occup Physiol. 1997;76(4):356–62.
Heled Y, Peled A, Yanovich R, Shargal E, Pilz-Burstein R, Epstein Y, Moran DS. Heat acclimation and performance in hypoxic conditions. Aviat Space Environ Med. 2012;83(7):649–53.
Helyar R, Green H, Zappe D, Sutton J. Comparative effects of acute volume expansion and short-term training on thermal and cardiovascular responses to prolonged exercise. Can J Physiol Pharmacol. 1996;74(9):1087–94.
Hopper MK, Coggan AR, Coyle EF. Exercise stroke volume relative to plasma-volume expansion. J Appl Physiol (1985). 1988;64(1):404–8.
Horowitz M. Heat acclimation, epigenetics, and cytoprotection memory. Compr Physiol. 2014;4(1):199–230.
Horowitz M, Parnes S, Hasin Y. Mechanical and metabolic performance of the rat heart: effects of combined stress of heat acclimation and swimming training. J Basic Clin Physiol Pharmacol. 1993;4(1–2):139–56.
Horowitz M, Shimoni Y, Parnes S, Gotsman MS, Hasin Y. Heat acclimation: cardiac performance of isolated rat heart. J Appl Physiol. 1985;60(1):9–13
Jose AD, Stitt F, Collison D. The effects of exercise and changes in body temperature on the intrinsic heart rate in man. Am Heart J. 1970;79(4):488–98.
Kanstrup IL, Ekblom B. Blood volume and hemoglobin concentration as determinants of maximal aerobic power. Med Sci Sports Exerc. 1984;16(3):256–62.
Karlsen A, Racinais S, Jensen MV, Nørgaard SJ, Bonne T, Nybo L. Heat acclimatization does not improve VO2max or cycling performance in a cool climate in trained cyclists. Scand J Med Sci Sports. 2015;25:269–76.
Keiser S, Fluck D, Huppin F, Stravs A, Hilty MP, Lundby C. Heat training increases exercise capacity in hot but not in temperate conditions: a mechanistic counter-balanced cross-over study. Am J Physiol Heart Circ Physiol. 2015;309(5):H750–61.
Kodesh E, Horowitz M. Soleus adaptation to combined exercise and heat acclimation: physiogenomic aspects. Med Sci Sports Exerc. 2010;42(5):943–52.
Kregel KC. Heat shock proteins: modifying factors in physiological stress responses and acquired thermotolerance. J Appl Physiol. 1985;92(5):2177–86
Lee BL, Miller A, James RS, Thake CD. Cross acclimation between heat and hypoxia: heat acclimation improves cellular tolerance and exercise performance in acute normobaric hypoxia. Front Physiol. 2016;7:1–15.
Lenth R. Emmeans: estimated marginal means, aka least-squares means. R package version 1.9.0. 2023. https://CRAN.R-project.org/package=emmeans. Accessed 16 Jan 2024.
Lorenzo S, Halliwill JR, Sawka MN, Minson CT. Heat acclimation improves exercise performance. J Appl Physiol. 1985;109(4):1140–47
Lundby C, Hamarsland H, Hansen J, Bjorndal H, Berge SN, Hammarstom D, Ronnestad BR. Hematological, skeletal muscle fiber, and exercise performance adaptations to heat training in elite female and male cyclists. J Appl Physiol (1985). 2023;135(1):217–26.
McCleave EL, Slattery KM, Duffield R, Saunders PU, Sharma AP, Crowcroft SJ, Coutts AJ. Temperate performance benefits after heat, but not combined heat and hypoxic training. Med Sci Sports Exerc. 2017;49(3):509–17.
McKay AKA, Stellingwerff T, Smith ES, Martin DT, Mujika I, Goosey-Tolfrey VL, Sheppard J, Burke LM. Defining training and performance caliber: a participant classification framework. Int J Sports Physiol Perform. 2022;17(2):317–31.
Mikkelsen CJ, Junge N, Piil JF, Morris NB, Oberholzer L, Siebenmann C, Lundby C, Nybo L. Prolonged heat acclimation and aerobic performance in endurance trained athletes. Front Physiol. 2019;10:1372.
Minson CT, Cotter JD. CrossTalk proposal: heat acclimatization does improve performance in a cool condition. J Physiol. 2016;594(2):241–3.
Neal RA, Corbett J, Massey HC, Tipton MJ. Effect of short-term heat acclimation with permissive dehydration on thermoregulation and temperate exercise performance. Scand J Med Sci Sports. 2016;26(8):875–84.
Neal RA, Massey HC, Tipton MJ, Young JS, Corbett J. Effect of permissive dehydration on induction and decay of heat acclimation, and temperate exercise performance. Front Physiol. 2016;7:564.
Nielsen B, Hales JR, Strange S, Christensen NJ, Warberg J, Saltin B. Human circulatory and thermoregulatory adaptations with heat acclimation and exercise in a hot, dry environment. J Physiol. 1993;460:467–85.
Nybo L, Lundby C. CrossTalk opposing view: Heat acclimatization does not improve exercise performance in a cool condition. J Physiol. 2016;594(2):245–7.
Oberholzer L, Siebenmann C, Mikkelsen CJ, Junge N, Piil JF, Morris NB, Goetze JP, Meinild Lundby AK, Nybo L, Lundby C. Hematological adaptations to prolonged heat acclimation in endurance-trained males. Front Physiol. 2019;10:1379.
De Pauw K, Roelands B, Cheung SS, de Geus B, Rietjens G, Meeusen R. Guidelines to classify subject groups in sport-science research. Int J Sport Physiol Perfor. 2013;8(2):111–22.
Périard JD, Eijsvogels TMH, Daanen HAM. Exercise under heat stress: thermoregulation, hydration, performance implications and mitigation strategies. Physiol Rev. 2021;101:1873–979.
Périard JD, Racinais S, Sawka MN. Adaptations and mechanisms of human heat acclimation: applications for competitive athletes and sports. Scand J Med Sci Sports. 2015;25:20–38.
R Core Team. R: A language and environment for statistical computing. Vienna: R Foundation for Statistical Computing; 2022.
Racinais S, Nichols D, Townsend N, Travers G, Cocking S, Brown HA, Rubio J, Périard JD. Hematological adaptations to training with and without heat. J Sci Sport Exerc. 2024. https://doi.org/10.1007/s42978-024-00302-y.
Racinais S, Périard JD, Karlsen A, Nybo L. Effect of heat and heat-acclimatization on cycling time-trial performance and pacing. Med Sci Sports Exerc. 2015;47(3):601–6.
Racinais S, Wilson MG, Périard JD. Passive heat acclimation improves skeletal muscle contractility in humans. Am J Physiol Regul Integr Comp Physiol. 2017;312(1):R101–7.
Ramanathan NL. A new weighting system for mean surface temperature of the human body. J Appl Physiol. 1964;19:531–3.
Rendell RA, Prout J, Costello JT, Massey HC, Tipton MJ, Young JS, Corbett J. Effects of 10 days of separate heat and hypoxic exposure on heat acclimation and temperate exercise performance. Am J Physiol Regul Integr Comp Physiol. 2017;313(3):R191–201.
Rønnestad BR, Hamarsland H, Hansen J, Holen E, Montero D, Whist JE, Lundby C. Five weeks of heat training increases hemoglobin mass in elite cyclists. Exp Physiol. 2021;160(1):316–27.
Rønnestad BR, Urianstad T, Hamarsland H, Hansen J, Nygaard H, Ellefsen S, HammarstrÖM D, Lundby C. Heat training efficiently increases and maintains hemoglobin mass and temperate endurance performance in elite cyclists. Med Sci Sport Exerc. 2022;54(9):1515–26.
Sawka MN, Leon LR, Montain SJ, Sonna LA. Integrated physiological mechanisms of exercise performance, adaptation, and maladaptation to heat stress. Compr Physiol. 2011;1(4):1883–928.
Sawka MN, Pandolf KB, Avellini BA, Shapiro Y. Does heat acclimation lower the rate of metabolism elicited by muscular exercise? Aviat Space Environ Med. 1983;54(1):27–31.
Sawka MN, Young AJ, Cadarette BS, Levine L, Pandolf KB. Influence of heat stress and acclimation on maximal aerobic power. Eur J Appl Physiol Occup Physiol. 1985;53(4):294–8.
Schmidt W, Prommer N. The optimised CO-rebreathing method: a new tool to determine total haemoglobin mass routinely. Eur J Appl Physiol. 2005;95(5–6):486–95.
Scoon GS, Hopkins WG, Mayhew S, Cotter JD. Effect of post-exercise sauna bathing on the endurance performance of competitive male runners. J Sci Med Sport. 2007;10:259–62.
Senay LC, Mitchell D, Wyndham CH. Acclimatization in a hot, humid environment: body fluid adjustments. J Appl Physiol. 1976;40(5):786–96.
Shein NA, Horowitz M, Alexandrovich AG, Tsenter J, Shohami E. Heat acclimation increases hypoxia-inducible factor 1alpha and erythropoietin receptor expression: implication for neuroprotection after closed head injury in mice. J Cereb Blood Flow Metab. 2005;25(11):1456–65.
Shvartz E, Shapiro Y, Magazanik A, Meroz A, Birnfeld H, Mechtinger A, Shibolet S. Heat acclimation, physical fitness, and responses to exercise in temperate and hot environments. J Appl Physiol Respir Environ Exerc Physiol. 1977;43(4):678–83.
Sotiridis A, Debevec T, Ciuha U, Eiken O, Mekjavic IB. Heat acclimation does not affect maximal aerobic power in thermoneutral normoxic or hypoxic conditions. Exp Physiol. 2019;104(3):345–58.
AS TaD. Climate and weather averages in Doha, Qatar. 2024. https://www.timeanddate.com/weather/qatar/doha/climate. Accessed 16 Jan 2024.
Takeno Y, Kamijo YI, Nose H. Thermoregulatory and aerobic changes after endurance training in a hypobaric hypoxic and warm environment. J Appl Physiol (1985). 2001;91(4):1520–8.
Travers G, Nichols D, Riding N, González-Alonso J, Périard JD. Heat acclimation with controlled heart rate: influence of hydration status. Med Sci Sports Exerc. 2020;52(8):1815–24.
Tyler CJ, Reeve T, Hodges GJ, Cheung SS. The effects of heat adaptation on physiology, perception and exercise performance in the heat: a meta-analysis. Sports Med. 2016;46(11):1699–724.
Watt MJ, Garnham AP, Febbraio MA, Hargreaves M. Effect of acute plasma volume expansion on thermoregulation and exercise performance in the heat. Med Sci Sports Exerc. 2000;32(5):958–62.
White AC, Salgado RM, Astorino TA, Loeppky JA, Schneider SM, McCormick JJ, McLain TA, Kravitz L, Mermier CM. The effect of ten days of heat acclimation on exercise performance in acute hypobaric hypoxia (4350 m). Temperature. 2016;3(1):176–85.
White A, Salgado R, Schneider S, Loeppky J, Astorino T, Mermier C. Does heat acclimation improve exercise capacity at altitude? A cross-tolerance model. Int J Sports Med. 2014;35(12):975–81.
Willmott AGB, Diment AG, Chung HC, James CA, Maxwell NS, Roberts JD, Gibson OR. Cross-adaptation from heat stress to hypoxia: a systematic review and exploratory meta-analysis. J Therm Biol. 2024;120:103793.
Wingo JE. Exercise intensity prescription during heat stress: a brief review. Scand J Med Sci Sports. 2015;25:90–5.
Wyndham CH, Benade AJ, Williams CG, Strydom NB, Goldin A, Heyns AJ. Changes in central circulation and body fluid spaces during acclimatization to heat. J Appl Physiol. 1968;25(5):586–93.
Wyndham CH, Rogers GG, Senay LC, Mitchell D. Acclimatization in a hot, humid environment: cardiovascular adjustments. J Appl Physiol. 1976;40(5):779–85.
Young AJ, Sawka MN, Levin L, Cadarette BS, Pandolf KB. Skeletal muscle metabolism during exercise is influenced by heat acclimation. J Appl Physiol. 1985;59(6):1929–35.
Acknowledgements
The authors thank the participants for their time and effort.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions. The authors declare that no funds or grants were received to conduct the study.
Author information
Authors and Affiliations
Contributions
JDP, DN and SR conceived and designed the study. Material preparation and data collection were performed by DN, GT, SC and NT. Data analysis was performed by DN and HB. The first draft of the manuscript was written by DN and JDP and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interests
The authors have no relevant financial or non-financial interests to disclose.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Périard, J., Nichols, D., Travers, G. et al. Impact of Exercise Heat Acclimation on Performance in Hot, Cool and Hypoxic Conditions. J. of SCI. IN SPORT AND EXERCISE 6, 275–287 (2024). https://doi.org/10.1007/s42978-024-00300-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42978-024-00300-0