Abstract
We analyzed the influences of interspecific interactions and environmental factors on rodent community assembly in Mojave Desert wetlands that support the endangered Amargosa vole (Microtus californicus scirpensis). A total of 4927 rodents in six species were captured from 2010 to 2021 within 15 marshes, most frequently including Amargosa voles, harvest mice (Reithrodontomys megalotis), and house mice (Mus musculus), an invasive species. The structure of the community varied considerably over time and by marsh, driven mainly by reductions in harvest mice and increases in Amargosa voles and house mice between 2014 and 2017, and then reductions in Amargosa voles between 2018 and 2021. Northern marshes were dominated by voles, and southern marshes by house mice. Primary determinants of rodent capture rates were interspecific interactions, human proximity, and season. There was a significant inverse relationship between vole and house mouse captures per session, suggesting competitive exclusion. Distance to the nearest town was positively associated with the numbers of voles and negatively associated with the numbers of harvest mice, suggesting that voles may be more sensitive to human disturbance than other rodents. Amargosa voles and harvest mice had annual population cycles with peaks between July and September occurring 6–7 months after rainfall. Our results allow for valuable insight into the influences on community dynamics in desert rodents and highlight that conservation efforts that support Amargosa voles may simultaneously help control house mouse abundance and expansion.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Patterns of rodent community structure have important implications for human and animal health (Meerburg et al. 2009a), food security (Meerburg et al. 2009b; Singleton et al. 2010), and conservation (Hein and Jacob 2015). Interspecific interactions are of particular concern where endangered species and non-native, invasive species occur in sympatry. Temporal and spatial variation in populations and the structure of communities are influenced by abiotic and biotic factors, including seasonal weather patterns, resource availability, intra- and interspecific interactions, predation, habitat selection, life-history characteristics of the species, historical contingency, human disturbances, land use changes, and stochastic influences (Andreassen et al. 2021; Brown and Harney 1993; Brown and Heske 1990; Huitu et al. 2004). Many patterns of rodent community structure and population dynamics have been described, including erratic variations in numbers and regular cycles of fluctuations typically characterized by increase, peak, crash, and low phases (Andreassen et al. 2021). Regular cycles can lead to pronounced intra-annual (seasonal) or inter-annual changes in density (Andreassen et al. 2021; Brown and Heske 1990). Interspecific interactions and community structure may change over time when member populations are very small, or during the decline (crash) or extended low phase of a population (Andreassen et al. 2021). At times of low density particularly, native species can be subject to opportunistic competition by invasive species, such as the non-native house mouse (Mus musculus) (Crespo 1966). For example, the introduction of house mice in the Canary Islands was implicated as a driver of the extinction of the insular lava mouse, Malpaisomys insularis (Drake and Hunt 2009).
Desert rodents show erratic, seasonal, and inter-annual cyclic population dynamics (Brown and Heske 1990), primarily associated with rainfall and the consequent increase of plant production and food resources (Munger et al. 1983; Shenbrot 2014). Other community and population drivers include interspecific competition and habitat selection (Shenbrot 2014). Evidence for competition among desert rodents comes from studies where one population increases when a competitor is absent (Eccard and Ylönen 2003). Desert rodents may be impacted by the density-dependent habitat selection of competing species, with population dynamics potentially altered through changed micro- and macro-habitat use (Eccard and Ylönen 2003; Munger et al. 1983; Shenbrot 2014).
Management of vulnerable species is best accomplished when informed by documentation of patterns of community structure across space and time and an understanding of how invasive species affect native species. A particular example of this need relates to the critically endangered Amargosa vole (Microtus californicus scirpensis) in the Mojave Desert. Located in the southwestern United States, this desert is considered a conservation priority for North American biodiversity (Hereford et al. 2006), with numerous rare and endemic species, such as the vole, living in isolated wetland habitat patches in the region. However, ongoing habitat degradation, loss of water driven by anthropogenic hydrologic alterations (e.g. ground-water pumping and land clearing), and climate change (Castle et al. 2019; Iknayan and Beissinger 2018), alongside the introduction of invasive species such as house mice (Ott-Conn et al. 2014) may have considerable impacts on ecological interactions among these rodents and their community structure. In North America, most studies of populations of desert rodents have been conducted in the Chihuahuan Desert (Brown et al. 1986; Ernest et al. 2008; Lightfoot et al. 2012; Thibault et al. 2010), with only two short-term studies in the Mojave Desert (Chew and Butterworth 1964; Hafner 1977).
The aim of this present study was to analyze in time and space the influence of interspecific interactions between non-native house mice and native rodents, including the endangered Amargosa vole. Also, we studied the relationships between seasonal and landscape factors with population fluctuations and their influence on rodent community structure in this wetland landscape in the Mojave Desert, California. We hypothesized that annual fluctuations of granivorous and herbivorous rodents would be associated with rainfall, but rainfall would not be associated with the occurrence of omnivorous rodents which can facultatively switch diets based on the resource’s availability (Balčiauskas et al. 2023). We also anticipated that Amargosa vole capture numbers would be positively associated with marsh areas while invasive house mouse numbers would be higher in marshes nearer to human settlements. Finally, due to differences in body size among rodent species, we hypothesized that Amargosa voles would negatively impact smaller-size rodents leading either to spatial segregation or reduced abundance.
Materials and methods
Study area
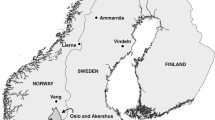
The study was conducted in Tecopa within the Amargosa River basin in the Mojave Desert in southeastern Inyo County, California, USA (35.8752 N, − 116.2343 E, Fig. 1) at elevations from 390 to 417 m. Local climate is arid and highly variable, with annual mean rainfall of 12.3 mm, and mean temperature ranges from 41.4 °C in summer to 3.2 °C in winter. Most of the Amargosa River in this region is subterranean, but ephemeral and spring-fed perennial surface flows in the region support marsh habitat. Marsh vegetation predominantly consists of bulrush (Schoenoplectus americanus (Pers.) Volkart) with a mix of desert salt grass (Distichlis spicata (L.) Greene), rushes (Juncus cooperi Engelm and J. balticus Willd.), and mixed herb communities including yerba mansa (Anemopsis californica (Nutt.) Hook. & Arn.) and common reed (Phragmites australis (Cav.) Trin. Ex Steud.).
Rodent trapping sites
A total of 15 marshes were surveyed from 2010 to 2021 for 3–5 consecutive days along transects using Sherman live traps (7.6 cm × 8.9 cm × 22.9 cm; HB Sherman, Tallahassee, FL, USA) (Fig. 1). Although sampling occurred when management and recovery actions dictated trapping and was not conducted at systematic intervals, each of the marshes was surveyed during at least three seasons of the year. Trap numbers varied by size of habitat fragment ranging from 20 to 140 traps per grid. Bait was peanut butter, horse feed (corn, barley, oats, and wheat with molasses), and apples, or peanut butter and oats. Baited traps were placed under vegetation, kept open overnight, and then checked at dawn. Each individual rodent was identified to species, sex, and age class. In addition, all animals were ear-tagged (model 1005–1; National Band and Tag Co., Newport, KY, USA) and weighed, while only Amargosa voles received a 12 mm passive integrated transponder (PIT) tag (Biomark, Boise, ID). After 5–15 min of handling, all animals other than two casualties (one vole in marsh 9 and one harvest mouse in marsh 7) were released at their site of capture.
Animal trapping and handling were performed in accordance with the guidelines of the American Society of Mammalogists (Sikes and Gannon 2011) and under an approved California Department of Fish & Wildlife (CDFW) Scientific Collecting Permit, US Fish and Wildlife Service Amargosa vole Recovery Permit #TE546414A-2, a permit from Bureau of Land Management (BLM), and approval by the UC Davis Institutional Animal Care and Use Committee.
Landscape features and climate variables
Marsh patches of possible relevance to voles were identified as those with continuous bulrush cover, identified from a vegetation layer provided by the United States Geological Survey (USGS) (Foley et al. 2017). For each marsh, we calculated the total area of the bulrush and the distance to the nearest human settlement using QGIS (www.qGIS.org) functions. Rainfall data were obtained from the Global Summary of the Month for 2010–2021 years (U.S. Department of Commerce, National Oceanic and Atmospheric Administration, Station: Death Valley National Park, CA US USC00042319, https://www.ncdc.noaa.gov).
Statistical analysis
Rodent community structure and species co-occurrence
Data were managed in Excel (Microsoft, Redmond, WA) and analyzed in the online freeware application iNEXT (Hsieh et al. 2016) and in R (R Core Team 2021). Statistical significance was inferred if P ≤ 0.05. For each marsh and trapping session, we calculated the capture rate per 100 trap nights by dividing the number of rodents captured by the sampling effort (number traps * number days) and multiplying by 100, to account for the effect of different survey durations and number of deployed traps. We developed rarefaction and extrapolation curves to compute sampling completeness and diversity of the rodent community using iNEXT (Hsieh et al. 2016). Rarefaction curves and species richness analysis were done with 100 bootstraps from our species capture rates per marsh.
To evaluate differences in rodent community structure among marshes, we performed a permutational multivariate ANOVA with 999 permutations (PERMANOVA), adding the site as a factor using the adonis2 function in the vegan package in R (Oksanen et al. 2020). In addition, we performed a 2-way crossed similarity percentage (SIMPER) analysis to identify the rodent species most responsible for the variation in the community structure among marshes, also using vegan.
A probabilistic model of species co-occurrence implemented in the COOCCUR package was applied to test for pair-wise patterns of species co-occurrence over three, four-year periods (2010–2013, 2014–2017, and 2018–2021) (Griffith et al. 2016; Veech 2013), since voles in the northern hemisphere tend to have regular population fluctuations manifesting as cycles with a peak every 3–5 years (Andreassen et al. 2021). Observed co-occurrence is compared to expected co-occurrence where the latter is the product of the probability of occurrence of the two species multiplied by the number of sampling sites. Pairs of species with expected co-occurrence < 1 were removed from the analysis, as recommended (Veech 2013).
Effects of biotic and abiotic factors on rodent populations
To evaluate the effects of environmental and biotic factors on the relative abundance of each rodent species, we performed generalized linear mixed models (GLMM) with a negative binomial distribution to account for overdispersion in response variables using the lme4 package (Bates et al. 2015). We estimated adjusted prediction values for the response variable using the ggeffects package (Lüdecke 2018). The total number of rodents per species was modeled as the number of captures per site within each trapping session. Predictor variables were season, mean monthly rainfall, total marsh area, minimum distance to the nearest human settlement, and capture rates of other species of rodents in the marsh. Sampling effort in the model included a number of trap nights as an offset. Each model also included a year as a random effect to account for inter-annual variation. Due to sample size limitations, we restricted the analysis to Amargosa voles, harvest mice (Reithrodontomys megalotis), and house mice. Seasonal patterns of captures were evaluated by combining data by species across all the marshes. We defined marsh area as the patch size dominated by bulrush using a vegetation cover layer provided by the United States Geological Survey (USGS).
Results
Rodent community structure and species co-occurrence
We captured 4927 rodents within 15 marshes over 195 trapping sessions in an 11-years period (Table 1). There were six small mammal species including 2114 Amargosa voles (42.9% of all captures), 1549 harvest mice (31.4%), 1079 house mice (21.9%), 161 cactus mice (Peromyscus eremicus, 3.3%), 20 desert woodrats (Neotoma lepida, 0.4%) and four kangaroo rats (Dipodomys merriami, 0.1%,).
From 2010 to 2013, the northern and central marshes were mainly dominated by harvest mice (marsh 5 = 100% of all captures in that marsh; marsh 6 = 81.2%; marsh 12 = 75%; marsh 17 = 75.8%; marsh 19 = 93.5%; marsh 54 = 78.4%). One marsh in the northern zone, marsh 1, was an exception to this pattern with 82.1% of 349 captures being voles and only 17.6% being harvest mice. Southern marshes were dominated by house mice (marsh 8 = 84.8%; marsh 9 = 82.7%; marsh 22 = 88%); in fact, excluding the three most southerly marshes, only two other marshes (marsh 54 to the north and marsh 12 in the central zone, Fig. 1) had any house mouse captures. Over the next four years (2014–2017), patterns of capture rates changed considerably. Besides Amargosa voles continued to dominate marsh 1 (82.1%), this species increased its numbers in other northern marshes (marsh 54 = 75.7%; M7 = 87.8%; marsh 10 = 64.8%) as well as in central marshes (marsh 12 = 41.8%; marsh 69 = 67.0%); their capture rates were roughly equal to those of harvest mice and house mice in central marsh 19 (31.6, 31.7 and 36.8% respectively) and marsh 21 (37.4, 37.4 and 24.1%, respectively), and harvest mice continued to outnumber voles in M5 (60.5%), M6 (56.8%), and M17 (54.1%). House mice continued to predominate to the south, comprising the majority of all captures in the four most southerly marshes (marsh 8 = 64.1%; marsh 9 = 61.7%; marsh 22 = 82.5%; marsh 67 = 54.5%), but importantly, expanding in range throughout central and northern marshes as well. In the last four years studied, 2018 to 2021, there was an alarming reduction of Amargosa voles except in the most northerly marshes where they were the most abundant (marsh 1 = 40.6%; marsh 7 = 70.4%; marsh 10 = 72.3%; and marsh 54 = 51.8%), while house mice became the most abundant species in all central (M12 = 100%; M19 = 50%, M69 = 70%) and southern marshes (M8 = 44%; M9 = 95.6%, Fig. 1). PERMANOVA analyses indicated that these differences in community structure among marshes were significant (F14 = 9.240, P = 0.001), while SIMPER analysis showed that the observed dissimilarities in rodent community assembly were driven mainly by the reductions in harvest mice, followed by increases in Amargosa voles and house mice.
Co-occurrence analysis of the 27 rodent species pair combinations did not detect any non-random negative relationships but found one positive association between cactus mice and desert wood rats.
Effects of biotic and abiotic factors on rodent populations
GLMM showed that capture rates of Amargosa voles and harvest mice were influenced by season (Tables 2 and 3). Capture rates of voles were significantly lower in spring (Z = − 2.749, P = 0.006) and marginally significant in winter (Z = − 1.874, P = 0.061) (Table 2, Fig. 2a). Harvest mice declined significantly in winter (P < 0.000), while in summer they underwent a significant increase (Z = 3.01, P = 0.003, Table 3, Fig. 2b). No statistically significant difference was observed between seasons in house mouse numbers (Table 4, Fig. 2c). Capture rates were not significantly associated with mean monthly precipitation for any rodent species, and therefore this predictor was excluded from further models. Overall, Amargosa voles, and harvest mice showed an annual pattern of population cycle with peaks between July and September, a 6–7 months time-lag after the rainfall peak in January–February (Fig. 3). Due to the lack of systematic intervals in time and locations of trapping sessions, we were not able to determine interannual patterns by time series analysis.
Seasonal variation in capture rates of a Amargosa vole (M. californicus scirpensis), b harvest mouse (Reithrodontomys megalotis), and c house mouse (Mus musculus) in a Mojave Desert wetland in southeastern California. Variable terms for a negative binomial model. The values are predicted values with upper and lower 95% confidence intervals
Models examining the influences of species on each other revealed a significant inverse relationship between the number of Amargosa voles and a number of house mice captured per session (P = 0.001, Table 2 and Fig. 4). There was no apparent influence of the number of harvest mice on other species. House mouse numbers were significantly and negatively related to vole numbers (Z = − 4.199, P < 0.001), and significantly and positively related to harvest mouse numbers (Z = 3.240, P = 0.001, Table 4).
Distance to the nearest town was positively correlated with the number of voles (Z = 3.2, P = 0.001, Table 2, Fig. 5a), and negatively correlated with the number of harvest mice (Z = − 4.386, P < 0.001, Table 3, Fig. 5b). Finally, area of marshes was a positive predictor of harvest mice (Z = 2.912, P = 0.004, Table 3), negatively correlated with the number of house mice (Z = − 4.716, P < 0.001, Table 4), and had no apparent influence on vole numbers (Z = 1.506, P = 0.132, Table 2).
Relationship between a Amargosa voles (M. californicus scirpiensis) and b harvest mouse (Reithrodontomys megalotis) capture rates and distance to the nearest human settlement in a Mojave Desert wetland in southeastern California. Variable terms for a negative binomial model. Lines reflect mean predicted values with upper and lower 95% confidence intervals
Discussion
Temporal and spatial variation in rodent community structure has important implications for human and animal health, and conservation programs (Hein and Jacob 2015; Meerburg et al. 2009a). In a wetland region of the Mojave Desert in California, we explored spatiotemporal dynamics in the community structure of native and invasive rodents. Patterns among rodent species were associated with interspecific interactions and environmental factors, with distance to human settlement, marsh area, and season, as the main predictors for the capture rates of several species of rodents. As we had anticipated, our results of co-ocurrence and interspecific interaction predictors showed no spatial segregation between rodent but effects on capture rates. Capture rates of Amargosa voles and house mice were negatively associated as was shown previously (McClenaghan and Montgomery 1998), while our recent work also confirmed that the detectability of both western harvest mouse and house mouse was negatively associated with vole presence (Haswell et al. 2023). Despite the northward spread of house mice, it is possible that Amargosa voles may impose some suppression of house mouse numbers, particularly in the northern marshes. This seems plausible as both experimental and field studies have shown high vole densities to reduce the reproductive success of both house mice and harvest mice (Blaustein 1980). Both harvest and house mice employ “fugitive” strategies against competitive interference from voles, and during phases of low vole population density, these smaller species leave their refugia, expand into more suitable habitat, and experience increased reproductive success (Blaustein 1980; Heske et al. 1984). Temporal partitioning has been also reported as one strategy for coexistence by limiting interspecific competition among mice and voles in the region (Haswell et al. 2023), and in other localities (Viviano et al. 2022).
Given they are sharing space in parts of our study area, the invasive house mice may still compete with Amargosa voles via exploitation of resources, apparent competition, or interference competition. The house mouse is a highly successful invader worldwide due to its high reproduction rate, breeding year-round if resources are available. They are omnivores with diets including grains, seeds, small (e.g. neonatal) mammals, and insects (Bronson 1979, 1984; Phifer-Rixey and Nachman 2015). For example in Australia, although the native New Holland mouse (Pseudomys novaehollandiae) is competitively superior than non-native house mice, the addition of house mice to treatment plots significantly reduced the abundance and habitat use of New Holland mouse (Fox and Gullick 1989). Introduced house mice contributed to the extinction of another rodent species in the Canary Islands (Drake and Hunt 2009). A mechanism of apparent competition could be a spillover of disease from house mice which can carry several potential pathogens of conservation concern such as the agents of plague and toxoplasmosis (Barbieri et al. 2020; Murphy et al. 2008).
The positive relationship we detected between the capture rates of harvest mice and house mice was consistent with Haswell et al. (2023) who also found a positive association between these species in the Mojave wetlands. This result could represent synchrony in population dynamics driven by resource availability or influences of competition in some marshes by the Amargosa vole (Blaustein 1980). To our knowledge, facilitation or positive interspecific interactions among rodents have not been reported in the literature, while competition appears to be relatively common among coexisting rodent species (Eccard and Ylönen 2003; Mori et al. 2019; Munger et al. 1983; Shenbrot 2014). Competition can be due to resource exploitation or interference by a competitor, reducing the target species’ fecundity, survivorship, or growth (Lang and Benbow 2013).
Contrary to what we predicted, voles were not more abundant in bigger marshes, which could be explained by regional population declines. Extrinsic factors that could contribute to vole decline and house mouse expansion include drought, other causes of reduced water, and habitat changes. In contrast with the wetland-specialized Amargosa vole, house mice are more tolerant of drier habitats, and population outbreaks have been reported after prolonged drought conditions (Haines et al. 1973; Saunders and Giles 1977). The differences in spatial distribution patterns found in this study are consistent with habitat preference, with southern marshes more subject to water insecurity occupied by house mice, cactus mice, and woodrats, whereas typically wetter northern marshes were occupied mainly by Amargosa voles, and harvest mice. In this Mojave region, both Amargosa voles and western harvest mice are highly marsh habitat-dependent (Klinger et al. 2015; López-Pérez et al. 2019), while cactus mice tend to live in drier areas, and desert woodrats in shrublands (Herndon 2004; Lewis 1972). Apparent habitat choice may encompass access to preferred foods and den areas, but also avoidance of potential competitors, as was shown among heteromyid species (Eccard and Ylönen 2003; Lemen and Rosenzweig 1978; Schroder and Rosenzweig 1975). Some evidence for competition among desert rodents comes from studies where one population increases when a competitor is absent, but also from desert rodent studies where one species may experience changed population dynamics in the face of density-dependent micro- and macro-habitat habitat use of a competing species (Eccard and Ylönen 2003; Munger et al. 1983; Shenbrot 2014). The extreme aridity of the Amargosa region only allows for resources to support small populations of one or two co-occurring granivorous rodent species (Brown and Ernest 2002; Chew and Butterworth 1964), which was consistent with our finding. In contrast, regions such as the Chihuahuan and Sonoran Deserts that receive higher annual precipitation may have up to ten coexisting species (Brown and Ernest 2002; Eccard and Ylönen 2003).
In addition to community structure, season associated with rainfall time-lag not surprisingly influenced the population dynamics of Mojave rodents as well. As predicted, only the populations of herbivorous (Amargosa voles) and granivorous (harvest mice), but not omnivorous (house mice), rodents showed seasonal dynamics likely related to rainfall. Results were consistent with annual fluctuations and a post-rain time lag which has been described in desert rodents, including harvest mice and other granivorous species, in the Chihuahuan Desert following rainfall and the consequent increase of plant production and food resources (Lightfoot et al. 2012; Munger et al. 1983; Thibault et al. 2010). In our study, lower numbers of Amargosa voles and harvest mice occurred in winter and spring during the rainfall peak whereas population peak occurred in summer and fall, six to seven months after the season with the highest precipitation. However, fluctuations of rodent populations of different species or even the same species in different locations can also be influenced by multiple factors such as top-down regulation (e.g. predation) and intraguild interactions (Andreassen et al. 2021; Brown and Ernest 2002). This complexity explains why our finding of annual periodicity in harvest mice is contrasting with a study conducted over 10 years in a Chihuahuan Desert rodent community found that Reithrodontomys megalotis showed little or no evidence of annual periodicity (Brown and Heske 1990). Our finding of no clear seasonal changes in house mouse fluctuations is consistent with the fact that they are opportunistically omnivorous in their foraging strategy (Le Roux et al. 2002) and litter density and breeding season can be almost year-round if resources are available (Le Roux et al. 2002; Singleton et al. 2005).
Regarding the effect of human activity on capture rates, we found a clear negative relationship between the vole population and proximity to human settlements, a positive relationship between harvest mice and humans, and no relationship between house mice and humans. This is likely explained by the fact that the highest quality habitat for voles in this area tends to be farther from the various houses, camping sites, and small businesses in the region because of less water diversion and direct destructive impact. We are not sure why harvest mice would have a different pattern except that it could be spurious, or perhaps reflects the avoidance of voles described above. Lastly, house mice numbers were not associated with proximity to humans. This is consistent with the fact that the numbers of house mice did not show a clear seasonal pattern suggesting that the species, which would have been introduced by humans, do not need refuge in human areas and is now well-established and spread across the entire region where habitat conditions are suitable.
As have other researchers (Brown and Ernest 2002; Lightfoot et al. 2012; Lima et al. 2008; Thibault et al. 2010), we demonstrated that population and community dynamics of the Mojave Desert wetland rodents are more complex than predicted based only on bottom-up regulation. The seasonal rainfall-plant production-rodent abundance relationship only applied to some native species but not house mice. Rather, community structure appeared to depend on interspecific interactions, human disturbance, and habitat selection.
Because our results support that Amargosa voles may be competitively superior to house mice, conservation efforts that support Amargosa voles, such as habitat improvement, ensuring water supplies, and reintroduction and translocation programs, may help control both house mouse abundance and expansion. We do emphasize the importance of long-term monitoring to better understand the mechanisms and effects of interspecific interactions, human disturbance, and environmental factors on community assembly and the fluctuation of desert rodent populations. Better understanding can prevent the spread and improve control of house mice and other invasive species and mitigate the decline of endangered species, such as Amargosa voles.
Data availability
Data are fully available upon written request.
References
Andreassen HP, Sundell J, Ecke F, Halle S, Haapakoski M, Henttonen H, Huitu O, Jacob J, Johnsen K, Koskela E (2021) Population cycles and outbreaks of small rodents: ten essential questions we still need to solve. Oecologia 195:601–622
Balčiauskas L, Stirkė V, Balčiauskienė L (2023) Abundance and population structure of small rodents in fruit and berry farms. Life 13:1–13
Barbieri R, Signoli M, Chevé D, Costedoat C, Tzortzis S, Aboudharam G, Raoult D, Drancourt M (2020) Yersinia pestis: the natural history of plague. Clin Microbiol Rev 34:e00044-e119
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Soft 67:1–48
Blaustein AR (1980) Behavioral aspects of competition in a three-species rodent guild of coastal Southern California. Behav Ecol Sociobiol 6:247–255
Bronson F (1979) The reproductive ecology of the house mouse. Quart Rev Biol 54:265–299
Bronson F (1984) The adaptability of the house mouse. Sci Am 250:116–127
Brown JH, Ernest SM (2002) Rain and rodents: complex dynamics of desert consumers: although water is the primary limiting resource in desert ecosystems, the relationship between rodent population dynamics and precipitation is complex and nonlinear. Bioscience 52:979–987
Brown JH, Harney B (1993) Population and community ecology of heteromyid rodents in temperate habitats. In: Genoways HH, Brown JH (eds) The biology of the Heteromyidae. Special Publication. The American Society of Mammalogy, Norman, Oklahoma, pp 618–651
Brown JH, Heske EJ (1990) Temporal changes in a Chihuahuan Desert rodent community. Oikos 59:290–302
Brown JH, Davidson D, Munger J, Inouye R (1986) Experimental community ecology: the desert granivore system. In: Diamond J, Case T (eds) Community ecology. Harper and Rown, New York, pp 41–61
Castle S, Rejmankova E, Foley J, Parmenter S (2019) Hydrologic alterations impact plant litter decay rate and ecosystem resilience in Mojave wetlands. Restor Ecol 27:1094–1104
Chew RM, Butterworth BB (1964) Ecology of rodents in Indian Cove (Mojave Desert), Joshua Tree National Monument, California. J Mammal 45:203–225
Crespo JA (1966) Ecología de una comunidad de roedores silvestres en el partido de Rojas, Pcia. de Buenos Aires. Rev Arg Cienc Nat Mus Bernardino Rivadavia 3:79–134
Drake DR, Hunt TL (2009) Invasive rodents on islands: integrating historical and contemporary ecology. Biol Invas 11:1483–1487
Eccard JA, Ylönen H (2003) Interspecific competition in small rodents: from populations to individuals. Evol Ecol 17:423–440
Ernest SM, Brown JH, Thibault KM, White EP, Goheen JR (2008) Zero sum, the niche, and metacommunities: long-term dynamics of community assembly. Am Nat 172:E257–E269
Foley J, Beechan A, Crews A, Lindauer A, Roy A, Vogel J, Klinger R (2017) Range-wide assessment of the endangered Amargosa vole and analysis of critical habitat stressors. University of California, Davis. Report to the California Department of Fish and Wildlife
Fox B, Gullick G (1989) Interspecific competition between mice: a reciprocal field manipulation experiment. Austral Ecol 14:357–366
Griffith DM, Veech JA, Marsh CJ (2016) Cooccur: probabilistic species co-occurrence analysis in R. J Stat Softw 69:1–17
Hafner MS (1977) Density and diversity in Mojave Desert rodent and shrub communities. J Anim Ecol 46:925–938
Haines H, Ciskowski C, Harms V (1973) Acclimation to chronic water restriction in the wild house mouse Mus musculus. Physiol Zool 46:110–128
Haswell PM, López-Pérez AM, Clifford DL, Foley JE (2023) Recovering an endangered vole and its habitat may help control invasive house mice. Food Webs 34:00267
Hein S, Jacob J (2015) Recovery of small rodent populations after population collapse. Wildl Res 42:108–118
Hereford R, Webb R, Longpré C (2006) Precipitation history and ecosystem response to multidecadal precipitation variability in the Mojave Desert region, 1893–2001. J Arid Env 67:13–34
Herndon C (2004) Vegetation characteristics associated with small mammal populations in the Las Vegas Wash. Master of Science Degree in Environmental Science. University of Nevada Las Vegas
Heske EJ, Ostfeld RS, Lidicker WZ Jr (1984) Competitive interactions between Microtus californicus and Reithrodontomys megalotis during two peaks of Microtus abundance. J Mammal 65:271–280
Hsieh T, Ma K, Chao A (2016) iNEXT: an R package for rarefaction and extrapolation of species diversity (H ill numbers). Methods Ecol Evol 7:1451–1456
Huitu O, Norrdahl K, Korpimäki E (2004) Competition, predation and interspecific synchrony in cyclic small mammal communities. Ecography 27:197–206
Iknayan KJ, Beissinger SR (2018) Collapse of a desert bird community over the past century driven by climate change. Proc Natl Acad Sci 115:8597–8602
Klinger R, Cleaver M, Anderson S, Maier P, Clark J (2015) Implications of scale-independent habitat specialization on persistence of a rare small mammal. Global Ecol Cons 3:100–114
Lang JM, Benbow ME (2013) Species interactions and competition. Nat Educ Knowl 4:1–8
Le Roux V, Chapuis JL, Frenot Y, Vernon P (2002) Diet of the house mouse (Mus musculus) on Guillou Island, Kerguelen archipelago, Subantarctic. Polar Biol 25:49–57
Lemen CA, Rosenzweig ML (1978) Microhabitat selection in two species of heteromyid rodents. Oecologia 33:127–135
Lewis A (1972) Seasonal population changes in the cactus mouse, Peromyscus eremicus. Southwest Natur 17:85–93
Lightfoot DC, Davidson AD, Parker DG, Hernández L, Laundré JW (2012) Bottom-up regulation of desert grassland and shrubland rodent communities: implications of species-specific reproductive potentials. J Mammal 93:1017–1028
Lima M, Ernest SM, Brown JH, Belgrano A, Stenseth NC (2008) Chihuahuan Desert kangaroo rats: nonlinear effects of population dynamics, competition, and rainfall. Ecology 89:2594–2603
López-Pérez AM, Foley J, Roy A, Pesapane R, Castle S, Poulsen A, Clifford DL (2019) Subpopulation augmentation among habitat patches as a tool to manage an endangered Mojave Desert wetlands-dependent rodent during anthropogenic restricted water climate regimes. PLoS One 14:e0224246
Lüdecke D (2018) ggeffects: tidy data frames of marginal effects from regression models. J Open Source Softw 3:1–5
McClenaghan LR, Montgomery SJ (1998) Draft Report. Distribution and abundance of the Amargosa vole (Microtus californicus scirpensis). California Department of Fish and Game, Sacramento, p 17
Meerburg BG, Singleton GR, Kijlstra A (2009a) Rodent-borne diseases and their risks for public health. Crit Rev Microbiol 35:221–270
Meerburg BG, Singleton GR, Leirs H (2009b) The year of the rat ends—time to fight hunger! Pest Manag Sci 65:351–352
Mori E, Ferretti F, Fattorini N (2019) Alien war: ectoparasite load, diet and temporal niche partitioning in a multi-species assembly of small rodents. Biol Invasions 21:3305–3318
Munger JC, Bowers MA, Jones WT (1983) Desert rodent populations: factors affecting abundance, distribution, and genetic structure. Gt Basin Nat Mem 7:91–116
Murphy RG, Williams RH, Hughes JM, Hide G, Ford NJ, Oldbury DJ (2008) The urban house mouse (Mus domesticus) as a reservoir of infection for the human parasite Toxoplasma gondii: an unrecognised public health issue? Int J Environ Health Res 18:177–185
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O’Hara R, Simpson G, Solymos P (2020) vegan: community ecology package. R Package Version 2(5–6):2019
Ott-Conn C, Clifford D, Branston T, Klinger R, Foley J (2014) Pathogen infection and exposure, and ectoparasites of the federally endangered Amargosa vole (Microtus californicus scirpensis), California, USA. J Wildl Dis 50:767–776
Phifer-Rixey M, Nachman MW (2015) The natural history of model organisms: insights into mammalian biology from the wild house mouse Mus musculus. Elife 4:e05959
R Core Team (2021) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Saunders G, Giles J (1977) A relationship between plagues of the house mouse, Mus musculus (Rodentia: Muridae) and prolonged periods of dry weather in south-eastern Australia. Wildl Res 4:241–247
Schroder GD, Rosenzweig ML (1975) Perturbation analysis of competition and overlap in habitat utilization between Dipodomys ordii and Dipodomys merriami. Oecologia 19:9–28
Shenbrot G (2014) Population and community dynamics and habitat selection of rodents in complex desert landscapes. Mammalia 78:1–10
Sikes RS, Gannon WL (2011) Guidelines of the American Society of Mammalogists for the use of wild mammals in research. J Mammal 92:235–253
Singleton GR, Brown PR, Pech RP, Jacob J, Mutze GJ, Krebs CJ (2005) One hundred years of eruptions of house mice in Australia—a natural biological curio. Biol J Linn Soc 84:617–627
Singleton GR, Belmain S, Brown PR, Aplin K, Htwe NM (2010) Impacts of rodent outbreaks on food security in Asia. Wildl Res 37:355–359
Thibault KM, Ernest SM, White EP, Brown JH, Goheen JR (2010) Long-term insights into the influence of precipitation on community dynamics in desert rodents. J Mammal 91:787–797
Veech JA (2013) A probabilistic model for analysing species co-occurrence. Glob Ecol Biogeogr 22:252–260
Viviano A, Scarfò M, Mori E (2022) Temporal partitioning between forest-dwelling small rodents in a Mediterranean deciduous woodland. Animals 12:1–9
Acknowledgements
We thank agency partners and landowners who facilitated the sampling and research. This project was funded by the Bureau of Land Management (BLM), US Fish and Wildlife Service, and California Department of Fish and Wildlife (CDFW).
Funding
This research was supported by California Department of Fish and Wildlife, U.S. Fish and Wildlife Service and U.S. Bureau of Land Management.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study’s conception and design. Material preparation and analysis were performed by AML-P. Data collection was performed by AML-P, PH, DC, and JF. The first draft of the manuscript was written by AML-P and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Handling editor: Sabine Begall.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
López-Pérez, A.M., Haswell, P., Clifford, D.L. et al. Interspecific interactions, human proximity, and season affect spatiotemporal structure of a Mojave Desert wetlands rodent community with a highly endangered species. Mamm Biol 103, 493–504 (2023). https://doi.org/10.1007/s42991-023-00367-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42991-023-00367-2