Abstract
Ever since the concept of “plant cell totipotency” was first proposed in the early twentieth century, plant regeneration has been a major focus of study. Regeneration-mediated organogenesis and genetic transformation are important topics in both basic research and modern agriculture. Recent studies in the model plant Arabidopsis thaliana and other species have expanded our understanding of the molecular regulation of plant regeneration. The hierarchy of transcriptional regulation driven by phytohormone signaling during regeneration is associated with changes in chromatin dynamics and DNA methylation. Here, we summarize how various aspects of epigenetic regulation, including histone modifications and variants, chromatin accessibility dynamics, DNA methylation, and microRNAs, modulate plant regeneration. As the mechanisms of epigenetic regulation are conserved in many plants, research in this field has potential applications in boosting crop breeding, especially if coupled with emerging single-cell omics technologies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
From unicellular green algae to angiosperms, plants are highly regenerative, meaning that new organs or whole bodies can be rebuilt following injury (Ikeuchi et al. 2016, 2019). Research on regeneration can be traced back to Gottlieb Haberlandt’s description of totipotency proposed in the early twentieth century (Haberlandt 2003; Krikorian and Berquam 2003; Thorpe 2007). In 1957, Skoog and Miller demonstrated that the ratio of exogenous auxin to cytokinin (CK) affects the fate of callus differentiation in tobacco (Nicotiana tabacum) (Skoog and Miller 1957), providing experimental tools and a conceptual framework for exploring the functions of phytohormones and their interactions during regeneration (Birnbaum and Alvarado 2008). Plant tissue culture has been widely used in both basic research and agriculture and provides an excellent system for studying plant organogenesis and somatic embryogenesis. Strategies that combine tissue culture and genome editing technologies provide opportunities to genetically improve crops (Loyola-Vargas and Ochoa-Alejo 2018).
Plant regeneration can be divided into two categories: injury-induced regeneration and tissue culture-induced regeneration (Mathew and Prasad 2021). In the former, different regeneration processes occur depending on the type of injury (Ikeuchi et al. 2016). When the meristem is partially damaged, the plant will reconstruct the meristem, whereas when the meristem is completely absent, the plant will grow axillary shoots or lateral roots. Some plant species, such as those in the Crassulaceae and Gesneriaceae families, can undergo de novo organogenesis to form new shoots or roots from cut sites (Ikeuchi et al. 2016). Other plants, however, require tissue culture to regenerate the entire plant. Injury activates a range of genes, including cell cycle genes, genes involved in CK synthesis and responses, and genes encoding transcription factors (TFs) of the AP2/ERF family (Ikeuchi et al. 2017). WOUND INDUCED DEDIFFERENTIATION 1 (WIND1) is rapidly induced at the wounding site and promotes cell dedifferentiation to form callus via type-B ARR (Iwase et al. 2011). Another gene in the AP2/ERF family, ETHYLENE RESPONSE FACTOR 115 (ERF115), promotes reconstitution of the stem cell niche after root tip excision (Heyman et al. 2016).
Tissue culture-induced regeneration can be divided into three types based on the culture system and regeneration process used: de novo root regeneration, de novo shoot regeneration, and somatic embryogenesis (Mathew and Prasad 2021). Both de novo root regeneration and shoot regeneration are two-step processes. The explant forms callus on an auxin-rich callus induction medium (CIM). The callus then differentiates into roots on root induction medium (RIM), which contains little or no auxin, or shoots on CK-rich shoot induction medium (SIM) (Lardon and Geelen 2020). Regardless of the origin of the explant, the process of callus formation induced on CIM follows the root developmental pathway (Sugimoto et al. 2010), and the identity of the callus is similar to that of root primordia (Zhai and Xu 2021). During this process, auxin signaling in Arabidopsis (Arabidopsis thaliana) first activates WUSCHEL RELATED HOMEOBOX 11 (WOX11) and WOX12, which transforms the regenerative pericycle or pericycle-like cells of the explant into root founder cells (Atta et al. 2009; Liu et al. 2014; Sang et al. 2018; Xu 2018). Subsequently, in the continuous presence of auxin, WOX11/WOX12 activates the expression of WOX5, WOX7, LATERAL ORGAN BOUNDARIES-DOMAIN 16 (LBD16), and LBD29, which in turn transform root founder cells into root primordium cells (Hu and Xu 2016; Xu 2018; Liu et al. 2018b). Thus, callus is formed on CIM.
Subsequently, the ratio of auxin to CK determines the direction of callus re-differentiation. On RIM, callus resembling root primordia continues to undergo cell division and differentiates into a well-organized root apical meristem (RAM). During this process, LBD16 expression in the root meristem gradually decreases and the expression of WOX5 and WOX7 is restricted to the stem cell niche (Hu and Xu 2016; Xu 2018; Jing et al. 2020). In addition, PLETHORA 1 (PLT1), PLT2, SCARECROW (SCR), and SHORT ROOT (SHR) are essential for quiescent center specification and stem cell activity in the RAM (Della Rovere et al. 2015; Shimotohno et al. 2018). However, under the induction of CK in SIM, the callus differentiates into shoots. First, the expression of CUP-SHAPED COTYLEDON1 (CUC1) and CUC2 in callus is spatially reorganized to mark promeristem regions, and PIN-FORMED 1 (PIN1) is induced by CUCs to determine the future locations of shoot progenitors (Hibara et al. 2003; Daimon et al. 2003; Gordon et al. 2007; Shin et al. 2020). Along with the up-regulation of PIN1, SHOOT MERISTEMLESS (STM) is expressed in the promeristem to maintain shoot meristem activity (Gordon et al. 2007; Shin et al. 2020). In addition, type-B ARRs in the CK signaling pathway, including ARABIDOPSIS RESPONSE REGULATOR 1 (ARR1), ARR2, ARR10, and ARR12, directly bind to and activate WUSCHEL (WUS), which directs the shoot apical meristem (SAM) formation program (Negin et al. 2017; Zhang et al. 2017b; Shin et al. 2020).
Unlike de novo root or shoot regeneration, somatic embryogenesis leads to the formation of a bipolar structure with an apical and basal pole. In Arabidopsis, somatic embryogenesis is often induced from 2,4-dichlorophenoxyacetic acid (2,4-D)-treated immature zygotic embryos at the bent cotyledon stage of development (Horstman et al. 2017). Ectopic expression of the embryo identity genes LEAFY COTYLEDON 1 (LEC1) (Lotan et al. 1998) and LEC2 (Stone et al. 2001), the meristem identity genes BABY BOOM (BBM) (Boutilier et al. 2002) and WUS (Gaj 2004; Chatfield et al. 2013), and wound-induced WIND1 (Ikeuchi et al. 2013) induces somatic embryogenesis.
Plant cells undergo multiple rapid cell fate transitions during regeneration, which is accompanied by the reprogramming of the transcriptome and chromatin landscape. Cell identity genes, especially TF genes, are induced by phytohormones to participate in plant regeneration (Sang et al. 2018; Ikeuchi et al. 2019; Sugimoto et al. 2019; Mathew and Prasad 2021). The expression of these key TF genes is partially regulated by various epigenetic regulators. In 2007, Crane and Gelvin reported that RNAi lines in which 24 genes encoding epigenetic regulators, including chromatin remodeling complexes, DNA methyltransferases, and various histone modification-related enzymes, had been silenced showed altered genetic transformation efficiencies (Crane and Gelvin 2007). Further studies have uncovered epigenetic dynamics during plant regeneration and highlighted the importance of the epigenetic regulation of key TFs that drive the cell fate transition during regeneration (Wang et al. 2020; Xu et al. 2021; Wu et al. 2022). Here, we summarize recent advances in understanding the epigenetic regulation of the plant regeneration process (Fig. 1, Table 1), with a focus on tissue culture-induced regeneration, and propose future directions for better understanding the different layers of epigenetic regulation of plant regeneration and their applications in crop breeding.
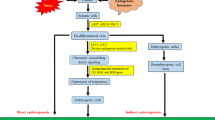
Roles of epigenetic regulators in plant regeneration. Mechanisms of histone methylation (A), histone acetylation (B), DNA methylation (C), and miRNA (D) in regulating plant regeneration. The font color indicates the transcriptional status of the gene, with red representing transcriptional activation and blue representing transcriptional repression. The scissors represent the injury-induced regeneration. The arrows represent activation and the T-ended arrows represent inhibition
Layers of epigenetic regulation of transcription
Gene transcription is regulated at multiple levels. In general, trans-acting TFs bind to cis-elements in their target promoters to promote or inhibit gene transcription. However, in the cellular environment, DNA is wrapped around histones and packaged into nucleosomes, which limits the access of TFs. Transcription factors compete with histones and other chromatin-binding proteins to bind to DNA (Klemm et al. 2019). Chromatin remodeling complexes can directly alter nucleosome composition and interactions, thereby affecting chromatin accessibility (Ojolo et al. 2018). Methylation or acetylation of histone H3 and H4 can affect the interactions between histones and DNA, resulting in loose or dense chromatin (Pfluger and Wagner 2007). DNA methylation is also involved in transcriptional regulation. DNA methylation in promoters inhibits gene transcription, whereas DNA methylation in gene bodies is mostly associated with constitutive expression (Zhang et al. 2018a). Post-transcriptional regulation, such as processes mediated by microRNA (miRNA), can also affect gene expression (Gibney and Nolan 2010).
Regulation of plant regeneration via histone modifications and histone variants
Histone methylation-regulated plant regeneration
The lysine or arginine residues of histone tails can be modified by mono-, di- and tri-methylation, which affects transcription by altering the local chromatin state. Methylation of residues at different positions of histone has various effects on the transcriptional regulation of genes. In general, trimethylation at lysine 27 of histone H3 (H3K27me3) and trimethylation at lysine 9 of histone H3 (H3K9me3) negatively regulate transcription, whereas trimethylation at lysine 4 of histone H3 (H3K4me3) and trimethylation at lysine 36 of histone H3 (H3K36me3) are associated with transcriptional activation (Xiao et al. 2016). Histone methylation dynamics are regulated by “writers” that add methyl groups, such as histone lysine methyltransferase (HKMTs) and protein arginine methyltransferases (PRMTs), and “erasers” that remove methyl group, such as histone demethylases (HDMs) (Liu et al. 2010).
H3K27me3 dynamics regulate plant regeneration
In Arabidopsis, H3K27me3 is catalyzed by Polycomb repressive complex 2 (PRC2), which consists of four subunits. The catalytic subunit Enhancer of zeste homolog2 (EZH2) is encoded by three functionally independent genes: CURLY LEAF (CLF), SWINGER (SWN), and MEDEA (MEA). The spatial–temporal-specific expression of these genes leads to the functional diversification of PRC2 (Bieluszewski et al. 2021). Instead of exhibiting normal plant architecture, the clf swn double mutant spontaneously forms a callus-like tissue that accumulates neutral lipids and occasionally somatic embryo-like structures (Chanvivattana et al. 2004; Ikeuchi et al. 2015). Although auxin failed to induce somatic embryogenesis from wild-type shoots, this treatment induced somatic embryo formation from clf swn shoots (Mozgová et al. 2017). This observation suggests that the loss of function of PRC2 promotes somatic embryogenesis from vegetative tissues in Arabidopsis. In rice (Oryza sativa), the H3K27me3 levels of OsWOX6, OsWOX9, OsWOX11, OsPLT3, OsPLT6, and OsPLT8 are significantly lower in callus than in seedlings (Zhao et al. 2020). In peach (Prunus persica), during the leaf-to-callus transition, the up-regulation of auxin-related genes PpPIN6 and AUXIN-INDUCED PROTEIN 13 (PpIAA13) and the lateral root development-related genes PpLBD1, LATERAL ORGAN BOUNDARIES (PpLOB), SHI RELATED SEQUENCE 1 (PpSRS1), and LATERAL ROOT PRIMORDIUM 1 (PpLRP1) is accompanied by a decrease in H3K27me3 levels (Zheng et al. 2022). Furthermore, treatment with the H3K27me3 demethylase inhibitor GSK-J4 significantly reduced the rate of callus induction in peach (Zheng et al. 2022). In hexaploid wheat (Triticum aestivum), H3K27me3 deceases at auxin signaling genes and root meristem formation-related genes such as TaPIN1 and TaLBD17 during the late stage of callus formation from immature embryos (Liu et al. 2022b). Thus, the attenuation of H3K27me3 of auxin signaling and meristematic-related genes could facilitate their activation and promote callus formation. By contrast, leaf tissue of the Arabidopsis clf swn mutant failed to form callus on CIM, because leaf identity genes, such as SAWTOOTH 1 (SAW1), SAW2, ARABIDOPSIS THALIANA HOMEOBOX GENE 1 (ATH1), and TCP DOMAIN PROTEIN 10 (TCP10), cannot be repressed without PRC2-mediated H3K27me3 deposition during dedifferentiation and callus induction (Fig. 1A) (He et al. 2012). In summary, PRC2-mediated H3K27me3 marks meristematic genes in differentiated tissues to prevent callus formation, while it is also required to turn off tissue identity genes for the acquisition of pluripotency and to facilitate callus formation.
Altering H3K4me3 and H3K4me2 affects plant regeneration
During the leaf-to-callus transition in Arabidopsis, the down-regulation of leaf identity genes is also regulated by the decrease in H3K4me3 levels. ARABIDOPSIS TRITHORAX 4 (ATX4) (Foroozani et al. 2021) catalyzes the trimethylation of H3K4 and participates in de novo shoot regeneration from leaf explants (Lee et al. 2019). ATX4 is highly expressed in leaves and deposits H3K4me3 on the leaf identity genes ATH1, KNOTTED1-LIKE HOMEOBOX GENE 4 (KNAT4), SAW1, SAW2, TCP10, and YABBY 5 (YAB5) to maintain leaf cell identify. Upon induction on CIM, the expression level of ATX4 decreases rapidly and remains low throughout callus induction. As a result, the H3K4me3 and expression levels of leaf identity genes decrease, which results in the loss of leaf cell identity (Lee et al. 2019). Compared to the wild type, atx4 more readily generates callus from leaf tissue (Lee et al. 2019). ATX4 also affects re-differentiation from callus to shoot tissue. When callus is transferred to SIM, ATX4 is temporarily up-regulated, and ATX4 re-deposits H3K4me3 on ATH1, SAW1, SAW2, TCP10, and YAB5 to regain shoot identity (Fig. 1A) (Lee et al. 2019). Thus, ATX4-mediated H3K4me3 has dual functions in callus induction and the re-differentiation of callus in Arabidopsis. By contrast, in crops such as wheat (Liu et al. 2022b) and rice (Zhao et al. 2020), H3K4me3 deposition increases during immature embryo- or seed-induced callus formation, pointing to an opposite role for H3K4me3 in promoting callus formation in these crops compared to Arabidopsis. This discrepancy might be due to the different types of explants used for each species.
In general, dimethylation at lysine 4 of histone H3 (H3K4me2) positively regulates gene expression in animals (Barski et al. 2007) but is associated with gene repression in plants (Liu et al. 2019). LYSINE-SPECIFIC DEMETHYLASE 1-LIKE 3 (LDL3) accumulates and specifically erases H3K4me2 marks on genes required for the acquisition of shoot traits during callus formation from root cells (Ishihara et al. 2019). Interestingly, LDL3-mediated removal of H3K4me2 does not immediately activate target genes, but rather primes genes for subsequent activation during shoot induction on SIM (Ishihara et al. 2019). Accordingly, the competency of shoot regeneration is severely impaired in the ldl3 mutant due to its failure to eliminate H3K4me2 on shoot regeneration genes, such as CBL-INTERACTING PROTEIN KINASE 23 (CIPK23) and UBIQUITIN-PROTEIN LIGASE 4 (UPL4) (Ishihara et al. 2019).
The status of H3K36me3 and H3K9me3 influences plant regeneration
Signaling factors in the auxin and CK pathways interact with histone modifiers to regulate gene expression during plant regeneration. The H3K36me3 methyltransferase gene ARABIDOPSIS TRITHORAX-RELATED 2 (ATXR2) (Lee et al. 2017) and the H3K9me3 demethylase gene JUMONJI C DOMAIN-CONTAINING PROTEIN 30 (JMJ30) (Lee et al. 2018a) are constitutively up-regulated upon callus induction on CIM. The auxin signaling factors AUXIN RESPONSE FACTOR 7 (ARF7) and ARF19 interact with and recruit ATXR2 and JMJ30 to the promoters of LBD16 and LBD29 and activate their expression to promote the dedifferentiation of leaf explants and callus formation (Fig. 1A) (Lee et al. 2017, 2018a). Notably, both JMJ30-mediated decreases in H3K9me3 and ATXR2-mediated increases in H3K36me3 are required for the activation of LBDs during the leaf-to-callus transition. ATXR2 also promotes root organogenesis on a phytohormone-free medium (Lee et al. 2018b) and inhibits shoot regeneration on SIM (Lee et al. 2021). The atxr2 mutant shows enhanced shoot regeneration from the callus regardless of the origin of the explant (Lee et al. 2021). ATXR2 interacts with type-B ARR1 in the CK signaling pathway to deposit H3K36me3 and activate ARR5 and ARR7 on SIM. ARR5 and ARR7 are type-A ARRs that inhibit WUS expression and shoot formation (Lee et al. 2021). Therefore, auxin-inducible ATXR2 regulates CK signaling and precisely controls WUS expression to prevent premature shoot induction. Another H3K36me3 methyltransferase, ASH1-RELATED 3 (ASHR3), promotes wound-induced callus formation. Following wounding, ASHR3 is rapidly activated and deposits H3K36me3 on ARR1, PLT3, and WIND3 to promote callus formation (Fig. 1A) (Lee et al. 2020).
Role of H4R3sme2 in plant regeneration
In addition to lysine methylation, histone arginine methylation is also involved in plant regeneration. Arabidopsis PROTEIN ARGININE METHYLTRANSFERASE 5 (PRMT5), which catalyzes the symmetric dimethylation at arginine 3 of histone H4 (H4R3sme2) and RNA splicing factors, affects the transcription and protein levels of the cyclin-dependent kinase inhibitor KIP-RELATED PROTEIN 1 (KRP1) to participate in shoot regeneration (Liu et al. 2016a). PRMT5 deposits H4R3sme2 on KRP1 and KRP2 to inhibit their transcription (Liu et al. 2016a). Furthermore, AtPRMT5 affects the alternative splicing of the E3 ubiquitin ligase gene RELATED TO KPC1 (RKP), which produces an abnormal RKP protein that cannot degrade KRP1 (Liu et al. 2016a). KRP1 is up-regulated and KRP1 protein is stabilized in the atprmt5 mutant. Since KRP1 negatively regulates cell division, cell division and shoot regeneration are inhibited in this mutant (Liu et al. 2016a).
Histone acetylation-regulated plant regeneration
The level of histone acetylation is regulated by histone acetyltransferases (HATs) and histone deacetylases (HDACs) (Kumar et al. 2021). HATs activate gene expression by catalyzing the acetylation of histone lysine tails, while HDACs remove acetyl groups to repress gene expression. HATs and HDACs can add or remove acetylation at multiple lysine sites, including lysine 9 of histone H3 (H3K9), lysine 14 of histone H3 (H3K14), lysine 36 of histone H3 (H3K36), lysine 5 of histone H4 (H4K5), lysine 8 of histone H4 (H4K8), lysine 12 of histone H4 (H4K12), and lysine 16 of histone H4 (H4K16) (Kumar et al. 2021).
Histone acetylation levels change dynamically during various regeneration processes. During wound-induced callus formation, acetylation at lysine 9/14 of histone H3 (H3K9/14ac) and acetylation at lysine 27 of histone H3 (H3K27ac) accumulate on genes that are up-regulated by wounding, such as WIND1, ERF113/RAP2.6L, and LBD (Rymen et al. 2019). The histone acetyltransferases HISTONE ACETYLTRANSFERASE OF THE GNAT FAMILY 1 (HAG1) and HAG3 promote callus formation during wounding, as the callus formation rate is significantly reduced in hag1 and hag3 mutants (Rymen et al. 2019). HAG1 also promotes the transition of callus to shoots. During de novo shoot regeneration, HAG1 catalyzes the acetylation of WOX5, SCR, PLT1, and PLT2 and promotes their expression, allowing the callus to acquire competence for shoot regeneration (Fig. 1B) (Kim et al. 2018). In addition to HATs, HDACs also affect plant regeneration. Inhibiting HDAC activity in Arabidopsis using the chemical inhibitor Trichostatin A (TSA) induced the transformation of hypocotyls into callus (Furuta et al. 2011) and induced somatic embryogenesis in the absence of auxin (Wójcikowska et al. 2018). Consistent with this observation, explants with a knocked-down expression of HISTONE DEACETYLASE 19 (had19) showed enhanced embryogenic responses. Specifically, HDA19 inhibits somatic embryogenesis by negatively regulating LEC1, LEC2, and BBM expression by reducing their acetylation levels (Fig. 1B) (Morończyk et al. 2022). However, when leaves were used as the explant, TSA inhibited callus formation (Lee et al. 2016). Consistently, both hda9 and hd-tuins protein1 (hdt1) mutants show reduced callus induction from leaves (Lee et al. 2016).
In rice, mature embryos are used as explants for callus formation. TSA treatment inhibited the formation of rice callus (Zhang et al. 2020). OsHDA710 decreases the acetylation levels of the transcriptional repressor genes OsARF18 and OsARF22, thereby activating OsPLT1 and OsPLT2 to promote callus formation (Fig. 1B) (Zhang et al. 2020). However, low concentrations of TSA promoted callus and shoot formation from mature wheat embryos, whereas high concentrations of TSA inhibited these processes (Bie et al. 2020). In addition, treatment with the histone deacetylase inhibitor sodium butyrate enhanced regeneration in wheat (Bie et al. 2020). Therefore, HDACs play various roles in the regeneration of different explants in different species. This variability is likely due to the promiscuous nature of histone acetylation modifiers, which modify multiple lysine residues of different histones.
The roles of histone variants in plant regeneration
In addition to histone modification, histone variants affect chromatin status and gene transcription (Foroozani et al. 2021). For instance, the histone variant H2A.Z has dual functions in transcriptional activation and repression (Kumar 2018). In rice callus, H2A.Z is enriched at the 5′ ends of highly expressed genes, while inactive gene bodies are covered by H2A.Z (Zhang et al. 2017a). The atypical H3 variant HISTONE THREE RELATED 15 (H3.15) is involved in cell fate reprogramming during plant regeneration in Arabidopsis (Yan et al. 2020). H3.15 lacks the K27 residue that is trimethylated, so its replacement would dilute H3K27me3 levels (Yan et al. 2020). The H3.15-encoding gene HISTONE THREE RELATED 15 (HTR15) is gradually up-regulated by auxin signaling during callus formation induced by wounding or culture on CIM (Yan et al. 2020). During callus formation, H3.15 is deposited on WOX11 and helps remove H3K27me3, thus promoting WOX11 expression and callus formation (Yan et al. 2020). Consistently, the htr15 mutant has reduced callus formation ability (Yan et al. 2020).
Chromatin accessibility dynamics are associated with the cell fate transition during plant regeneration
Chromatin accessibility dynamics are important for the regulation of gene expression and are in turn generally regulated by ATP-dependent chromatin remodeling complexes (CRCs). Remodelers can alter the accessibility of a specific genomic region to regulate DNA–histone interactions by changing the position, occupancy, and composition of nucleosomes using energy from ATP hydrolysis. Remodelers are highly conserved. Four subfamilies of remodeler complexes have been characterized in plants: CHROMODOMAIN HELICASE DNA BINDING (CHD), SWITCH DEFECTIVE/SUCROSE NON-FERMENTING (SWI/SNF), IMITATION SWITCH (ISWI), and INOSITOL REQUIRING 80/SWI2/SNF2-RELATED 1 (INO80/SWR1) (Han et al. 2015; Ojolo et al. 2018).
Arabidopsis INO80 and the histone chaperones NAP1-RELATED PROTEIN1 (NRP1) and NRP2 synergistically regulate inflorescence meristem (IM) size and RAM activity by affecting the expression of auxin-related genes and preventing DNA damage to maintain chromatin stability (Kang et al. 2019). PICKLE (PKL) is a CHD3 homolog in Arabidopsis that facilitates root meristem activity (Aichinger et al. 2011) and maintains root cell identity to limit embryogenesis by regulating the expression of the PRC2-encoding genes CLF and SWN (Aichinger et al. 2009). BRAHMA (BRM) is an SWI/SNF chromatin remodeling ATPase that maintains root stem cell activity by directly targeting PIN genes (Yang et al. 2015). SPLAYED (SYD) is a SWI2/SNF2-like protein in the SNF2 subclass whose eviction, combined with the deposition of H3K27me3 at the WUS promoter, contributes to terminate floral meristem development in Arabidopsis (Sun et al. 2019). Thus, chromatin remodelers generally participate in the regulation of meristem identity in plant tissues. However, few reports have documented how manipulating chromatin remodelers alters chromatin accessibility to influence plant regeneration. In monocot wheat, the regeneration efficiency is generally low (Wang et al. 2017; Zhang et al. 2018b). Co-expressing GROWTH REGULATING FACTOR and 4-GRF INTERACTING FACTOR 1 (TaGRF4-TaGIF1) greatly promoted regeneration in different wheat varieties (Debernardi et al. 2020). GIF recruits SWI/SNF chromatin remodeling complexes to its target genes to open the chromatin structure, thus allowing GRF4 to regulate downstream gene expression (Kim 2019; Luo and Palmgren 2021). Moreover, GIF1 functions together with GRFs to recruit SWI/SNF chromatin remodeling complexes to shape inflorescence architecture in maize (Zea mays) (Li et al. 2022).
In Arabidopsis, auxin treatment altered the chromatin accessibility of genes related to meristems and the cell cycle, such as CYCLIN DEPENDENT KINASE B2;1 (CDKB2;1) and PLT7, to rewire the cell totipotency network and drive somatic embryogenesis (Wang et al. 2020). A comparison of immature embryos and seedling explants revealed that open chromatin and the activated expression of embryonic genes such as ABA INSENSITIVE 3 (ABI3), BBM, FUSCA 3 (FUS3), LEC1, and LEC2 are required for somatic embryogenesis in Arabidopsis (Wang et al. 2020). Similarly, in wheat, the gain of chromatin accessibility, along with the activation of key genes (such as TaBBM and TaWOX5) that mediate the cell fate transition, occurs during callus induction from immature embryos (Liu et al. 2022b). During shoot regeneration from pluripotent callus in Arabidopsis, root identity genes such as WOX5 gradually lose their chromatin accessibility, while shoot identity genes such as PHYTOCHROME INTERACTING FACTOR 1 (PIF1) gain chromatin accessibility. Furthermore, the chromatin states of genes related to epidermal cell differentiation, CK responses, and secondary metabolism gradually become more open (Wu et al. 2022). In rice, chromatin is generally more open in the callus than in seedlings, with 58% more DNase I hypersensitive sites in the callus that are positively correlated with transcription (Zhang et al. 2012). During the process of protoplast generation from leaf mesophyll cells in Arabidopsis, more accessible chromatin regions are created, leading to the random activation of WUS, which ultimately promotes regeneration (Xu et al. 2021).
Therefore, an accessible chromatin environment leads to higher totipotency, which is required for regeneration. Chromatin accessibility dynamics are associated with changes in the expression of key genes that drive the cell fate transition during different steps of plant regeneration. However, the general or specific roles of individual chromatin remodelers in plant regeneration remain unclear.
DNA methylation status affects plant regeneration
In plants, DNA methylation occurs on cytosine, including symmetrical CG methylation, CHG methylation, and asymmetric CHH methylation (Law and Jacobsen 2010). The establishment, maintenance, and removal of DNA methylation marks are catalyzed by different enzymes (Law and Jacobsen 2010). DOMAINS REARRANGED METHYLTRANSFERASE 2 (DRM2) catalyzes de novo methylation, whereas the maintenance of DNA methylation requires different enzymes: CG methylation is maintained by DNA METHYLATRANSFERASE 1 (MET1, also known as DMT1), CHG methylation is maintained by CHROMOMETHYLASE 3 (CMT3), and CHH methylation is maintained by DRM2 and CMT2 (Zhong et al. 2014). DNA demethylation is initially mediated by DNA glycosidases, including DEMETER (DME), REPRESSOR OF SILENCING 1 (ROS1), DEMETER-LIKE 2 (DML2), and DML3 (Law and Jacobsen 2010).
Significant changes in DNA methylation levels both globally and at local key genes occur during multiple plant regeneration processes. Compared to leaves, global CHG methylation levels are higher and CHH methylation levels are lower in callus, which is consistent with the up-regulation of CMT3 and down-regulation of CMT2 in callus (Shim et al. 2022). Cell proliferation-related genes, including PLT1, PLT2, ORIGIN RECOGNITION COMPLEX 1 (ORC1), REPLICATION FACTORC 2 (RFC2), MITOTIC ARREST DEFICIENT 1 (MAD1), and DISRUPTION OF MEIOTIC CONTROL 1 (DMC1), are hypomethylated in callus (Shim et al. 2022). The binding motifs of the circadian rhythm regulator genes CIRCADIAN CLOCK-ASSOCIATED 1 (CCA1) and LATE ELONGATED HYPOCOTYL (LHY) are enriched in these CHH-hypomethylated regions (Shim et al. 2022). Indeed, CCA1 directly binds to the promoter of the cell division-related gene ORC1 to inhibit its expression, which may be related to the high CHH methylation levels of this promoter in leaves (Shim et al. 2022). During callus formation, CCA1 is inhibited and the CHH methylation level of the ORC1 promoter decreases, thus releasing the expression of ORC1 and enhancing cell proliferation (Shim et al. 2022).
DNA methyltransferases affect shoot regeneration. Compared to wild-type Arabidopsis, both the met1 single mutant and drm1 drm2 cmt3 (ddc) triple mutant show enhanced competence for shoot regeneration (Shemer et al. 2015; Liu et al. 2018a; Shim et al. 2021). Furthermore, the ddc mutant regenerated shoots directly from roots on SIM without inducing callus formation (Shemer et al. 2015). During the two-step shoot regeneration process, MET1 is highly expressed in the callus under the activation of ATE2FA (E2FA), and its expression is down-regulated on SIM (Liu et al. 2018a). MET1 maintains the DNA methylation of WUS and inhibits WUS expression in the callus (Fig. 1C) (Liu et al. 2018a). When MET1 was mutated, WUS, the CK signaling genes ARR1 and ARR10, and the blue light receptor gene CRYPTOCHROME 1 (CRY1) were activated to promote shoot regeneration (Liu et al. 2018a; Shim et al. 2021). Similarly, the up-regulation of WUS in the ddc mutant resulted in the direct conversion of roots into shoots on SIM (Fig. 1C) (Shemer et al. 2015). However, treatment with 5-azacytidine, which inhibits DNA methylation, has different effects on regeneration in different species. 5-azacytidine promoted the transformation of peach leaves to callus (Zheng et al. 2022) but inhibited callus formation in strawberry (Fragaria ananassa) (Liu et al. 2022a). In addition, treatment with 5-azacytidine enhanced somatic embryogenesis in Arabidopsis (Grzybkowska et al. 2018) but inhibited this process in rice (Hsu et al. 2018). These findings suggest that DNA methylation plays diverse roles in the regeneration of different plant species.
Plant regeneration competence is affected by the explant’s age and variety, which are also related to DNA methylation. The regeneration capacity of Boea hygrometrica leaves decreases during aging, which may be related to the high CHH methylation levels in mature leaves (Sun et al. 2020). There are significant differences in somatic embryogenesis competence between the cotton (Gossypium hirsutum) cultivars Yuzao1 and Lumian1, which may be related to the level of CHH methylation (Guo et al. 2020). Yuzao1 has a high somatic embryo induction rate and CHH hypomethylation, whereas Lumian1 has a low somatic embryo induction rate and CHH hypermethylation (Guo et al. 2020). Therefore, high DNA methylation levels reduce the regeneration ability of plants.
MicroRNA levels are associated with plant regeneration capacity
miRNAs are a class of 21-nt non-coding small RNAs that reduce gene transcription by targeting mature mRNAs (Axtell 2013). miRNAs such as miR156, miR160, miR167, miR319, and miR393 are involved in plant regeneration via the direct or indirect regulation of auxin and CK signaling genes.
miR156 is involved in several age-related developmental processes (Xu et al. 2016; Guo et al. 2017). The shoot regeneration capacity of Arabidopsis and tobacco decreases with plant age, which can be compensated for by overexpressing MiR156 (Zhang et al. 2015). SQUAMOSA PROMOTER BINDING PROTEIN-LIKE 9 (SPL9), encoded by a gene targeted by miR156, directly binds to type-B ARR genes, including ARR1, ARR2, ARR10, and ARR12, to impair CK responses (Fig. 1D) (Zhang et al. 2015). High levels of miR156 inhibited SPL9 expression at the juvenile stage of Arabidopsis seedlings (Zhang et al. 2015). After juvenile-to-adult transition, decreased miR156 levels led to the up-regulation of SPL9 and the inhibition of CK responses, thus weakening the capacity for shoot regeneration (Zhang et al. 2015). The miR156-SPL regulatory circuit plays a similar role in somatic embryogenesis in citrus. For the majority of citrus cultivars, the callus gradually loses its embryogenesis capacity and fails to differentiate into shoots after long-term culture (Long et al. 2018). miR156 levels are significantly lower in non-embryonic than in embryonic callus, while its target genes CsSPL3 and CsSPL14 show the opposite trend (Long et al. 2018). The expression levels of CsSPL3 and CsSPL14 are highly negatively correlated with somatic embryogenesis capacity in different citrus varieties (Long et al. 2018). In the orange varieties ‘Anliu’, ‘Newhall’, ‘Valencia’, and ‘American’ sour orange, which can undergo somatic embryogenesis, the expression levels of CsSPL3 and CsSPL14 are relatively low, while in varieties with weak competence for somatic embryogenesis, the expression levels of CsSPL3 and CsSPL14 are high (Long et al. 2018). These observations suggest that miR156-SPLs are involved in regulating age-dependent and variety-specific somatic embryogenesis in citrus.
Similar to miR156, miR319 also promotes shoot regeneration by affecting CK responses. The target genes of miR319 are TCP3 and TCP4, encoding proteins that directly activate ARR16, which encodes a negative regulator of shoot regeneration (Fig. 1D) (Yang et al. 2020). Loss-of-function of HUA ENHANCER 1 (HEN1), a small RNA methyltransferase that stabilizes miR319, decreased miR319 levels, leading to the up-regulation of TCP3 and TCP4, which in turn activated ARR16 and inhibited shoot regeneration (Yang et al. 2020).
miRNAs also affect auxin signaling during plant regeneration. miR160 targets ARF10 and inhibits auxin signaling, which in turn inhibits callus and shoot regeneration (Fig. 1D) (Qiao et al. 2012; Liu et al. 2016b). Furthermore, the transcriptional repressor ARF10 binds directly to AuxRE in the promoter region of ARR15, which encodes a negative regulator of callus formation (Fig. 1D) (Liu et al. 2016b). Therefore, miR160 inhibits regeneration by affecting both auxin and CK signaling pathways. In cotton, miR167 negatively regulates somatic embryogenesis by targeting ARF6 and ARF8 (Fig. 1D) (Arora et al. 2020). In plants overexpressing the miR167 target mimic (35S::MIM167), ARF6, ARF8, the auxin-responsive gene GRETCHEN HAGEN 3 (GH3), and the auxin transporter genes AUXIN RESISTANT 1 (AUX1), LIKE AUX1 3 (LAX3), PIN1, and PIN2 were significantly up-regulated, suggesting that miR167 promotes somatic embryogenesis by enhancing auxin signaling (Arora et al. 2020). In addition to affecting ARF expression, miRNAs also affect the expression of auxin receptor-encoding genes, such as TRANSPORT INHIBITOR RESPONSE 1 (TIR1) and AUXIN SIGNALING F-BOX 3 (AFB3). TIR1 and AFB3 were up-regulated in a miR393 mutant, and the capacity for shoot regeneration and somatic embryogenesis was higher in the mutant than in the wild type (Fig. 1D) (Wójcik and Gaj 2016; Wang et al. 2018).
Conclusions and future prospects
Cell totipotency and cell fate determination are fundamental research topics in biology. Plant regeneration provides an excellent system for studying these topics. Multi-step cell fate transitions occur during plant regeneration, which are accompanied by chromatin landscape remodeling and transcriptome reprogramming, particularly for cell identity genes such as WOX11, WOX5, and WUS. Recent studies have improved our understanding of the functions of various epigenetic regulators, such as histone modification ‘writers’ and ‘erasers’, chromatin remodelers, DNA methyltransferases, and miRNAs, in shaping plant regeneration by altering the expression of cell identity genes. However, many open questions remain.
Cell identity is associated with the accessibility of specific portions of the genome, which is controlled by interactions between genomic DNA and nucleosomes containing various histones (Chen and Dent 2014). Altering DNA–histone interactions via chromatin modifiers would affect the transcriptional competency of genes associated with specific regions of the genome (Klemm et al. 2019). Since cell identity frequently switches during plant regeneration, multiple chromatin modifiers are required to broadly alter the accessibility of certain portions of the genome and specifically fine-tune the expression of key genes in coordination with the activity of specific TFs. One challenging question is how different chromatin modifiers function cooperatively to control regeneration. The specific expression or induction patterns of chromatin modifiers and their recruiters might differ for different targets or for the same targets but at different stages of regeneration. For example, the methyltransferase ATXR2 of H3K36me3 and demethylase JMJ30 of H3K9me3 both regulate LBD16 and LBD19, but their regulation is interdependent (Lee et al. 2018a). However, different chromatin modifiers might function together at the same loci. For instance, the removal of H3K27me3 and gain of chromatin accessibility as well as increases in H3K4me3 at specific gene clusters were detected during the early callus induction step of wheat shoot regeneration from immature embryos (Liu et al. 2022b). The detailed mechanism that coordinates the activities of different chromatin modifiers remains to be elucidated.
Phytohormone signals, especially auxin and CK signals, are essential during plant regeneration. Auxin and CK signals are transmitted to downstream target genes via ARF (Powers and Strader 2020) and type-B ARR (Li et al. 2021) TFs, respectively. On the one hand, epigenetic regulators can directly affect the expression of ARF and ARR (Zhang et al. 2020) or targets of ARF and ARR by “hijacking” ARF and ARR (Lee et al. 2017, 2021) to deposit specific histone modifications that alter their transcriptional activity. On the other hand, certain epigenetic regulators are induced by auxin or CK signaling, showing specific expression patterns during regeneration (Lee et al. 2018b, 2021). Therefore, additional studies are needed to explore the relationship between plant hormonal signals and epigenetic regulators, particularly to establish how auxin and CK influence epigenetic regulators for global chromatin remodeling and thus the cell fate transition.
The mechanisms of epigenetic regulation of plant regeneration, such as chromatin accessibility, H3K27me3, and H3K4me3, are generally conserved among different species, indicating that knowledge obtained studying model plants can be transferred to less-studied species, such as crops with large and complex genomes [e.g., wheat, maize, and barley (Hordeum vulgare)]. However, epigenetic regulators show diverse, pleiotropic effects; the same factor may behave differently or even in an opposite manner during different stages of the regeneration process. Moreover, orthologous factors might exhibit various functions during the same stage of regeneration in different species or even in different explants of the same species. For these pleiotropic effects, in addition to epigenetic regulators per se, more attention needs to be paid to stage-specific recruiters that set the ‘specificity’ of epigenetically modified targets. The diverse effects of orthologous factors are likely related to the different pre-existing cell identities in various explants or similar explants of different species. Special attention should be paid to characterizing the explant- or species-specific reprogramming of epigenomics during regeneration.
Regeneration is widely used during the production of genetically manipulated plants for agriculture. Whereas in Arabidopsis, transgenic or genome-edited plants can be directly generated using the floral dip method (Clough and Bent 1998), major crops, including rice, wheat, and maize, require long-term tissue culture (Hiei et al. 2014). The efficiency of genetic transformation methods of crops has been improved by optimizing their regeneration systems (Hayta et al. 2019) and by the ectopic expression of genes encoding key regeneration factors such as WUS, BBM, and WOX5 (Lowe et al. 2016; Wang et al. 2022). However, due to the diversity among species and explants, not all factors that function in Arabidopsis regeneration can improve the efficiency of the genetic transformation of crops. Therefore, it is important to systematically study the regeneration processes of crops and to identify ‘novel’ factors that can enhance the efficiency of crop regeneration. Several recent studies have systematically analyzed gene expression and chromatin dynamics during the regeneration process of rice (Zhao et al. 2020; Shim et al. 2020), wheat (Liu et al. 2022b), and barley (Suo et al. 2021), providing valuable resources for mining key factors that enhance regeneration, such as TaDOF3.4 and TaDOF5.6 in wheat (Liu et al. 2022b). However, more in-depth analysis is still urgently needed to better understand the regeneration process and improve the genetic transformation efficiency of crops.
Finally, the development of single-cell and spatial omics technologies (Xia et al. 2022) provides additional tools for tracing cells with regenerative origins in various explants and exploring the heterogeneity of callus in the same generation or during transmission to the next generation (Mironova and Xu 2019; Xu et al. 2021; Zhai and Xu 2021). Such analyses will further enhance our mechanistic understanding of plant regeneration, thereby facilitating the development of advanced crop breeding tools.
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Aichinger E, Villar CBR, Farrona S, Reyes JC, Hennig L, Köhler C (2009) CHD3 proteins and polycomb group proteins antagonistically determine cell identity in Arabidopsis. PLOS Genet 5:e1000605. https://doi.org/10.1371/journal.pgen.1000605
Aichinger E, Villar CBR, Di Mambro R, Sabatini S, Köhler C (2011) The CHD3 chromatin remodeler PICKLE and polycomb group proteins antagonistically regulate meristem activity in the Arabidopsis root. Plant Cell 23:1047–1060. https://doi.org/10.1105/tpc.111.083352
Arora S, Singh AK, Chaudhary B (2020) Target-mimicry based miRNA167-diminution ameliorates cotton somatic embryogenesis via transcriptional biases of auxin signaling associated miRNAs and genes. Plant Cell Tiss Organ Cult 141:511–531. https://doi.org/10.1007/s11240-020-01810-9
Atta R, Laurens L, Boucheron-Dubuisson E, Guivarch A, Carnero E, Giraudat-Pautot V, Rech P, Chriqui D (2009) Pluripotency of Arabidopsis xylem pericycle underlies shoot regeneration from root and hypocotyl explants grown in vitro. Plant J 57:626–644. https://doi.org/10.1111/j.1365-313X.2008.03715.x
Axtell MJ (2013) Classification and comparison of small RNAs from plants. Annu Rev Plant Biol 64:137–159. https://doi.org/10.1146/annurev-arplant-050312-120043
Barski A, Cuddapah S, Cui K, Roh T-Y, Schones DE, Wang Z, Wei G, Chepelev I, Zhao K (2007) High-resolution profiling of histone methylations in the human genome. Cell 129:823–837. https://doi.org/10.1016/j.cell.2007.05.009
Bie XM, Dong L, Li XH, Wang H, Gao X-Q, Li XG (2020) Trichostatin A and sodium butyrate promotes plant regeneration in common wheat. Plant Signal Behav 15:1820681. https://doi.org/10.1080/15592324.2020.1820681
Bieluszewski T, Xiao J, Yang Y, Wagner D (2021) PRC2 activity, recruitment, and silencing: a comparative perspective. Trends Plant Sci 26:1186–1198. https://doi.org/10.1016/j.tplants.2021.06.006
Birnbaum KD, Alvarado AS (2008) Slicing across kingdoms: regeneration in plants and animals. Cell 132:697–710. https://doi.org/10.1016/j.cell.2008.01.040
Boutilier K, Offringa R, Sharma VK, Kieft H, Ouellet T, Zhang L, Hattori J, Liu C-M, van Lammeren AAM, Miki BLA, Custers JBM, van LookerenCampagne MM (2002) Ectopic expression of BABY BOOM triggers a conversion from vegetative to embryonic growth. Plant Cell 14:1737–1749. https://doi.org/10.1105/tpc.001941
Chanvivattana Y, Bishopp A, Schubert D, Stock C, Moon Y-H, Sung ZR, Goodrich J (2004) Interaction of Polycomb-group proteins controlling flowering in Arabidopsis. Development 131:5263–5276. https://doi.org/10.1242/dev.01400
Chatfield SP, Capron R, Severino A, Penttila P-A, Alfred S, Nahal H, Provart NJ (2013) Incipient stem cell niche conversion in tissue culture: using a systems approach to probe early events in WUSCHEL-dependent conversion of lateral root primordia into shoot meristems. Plant J 73:798–813. https://doi.org/10.1111/tpj.12085
Chen T, Dent SYR (2014) Chromatin modifiers and remodellers: regulators of cellular differentiation. Nat Rev Genet 15:93–106. https://doi.org/10.1038/nrg3607
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16:735–743. https://doi.org/10.1046/j.1365-313x.1998.00343.x
Crane YM, Gelvin SB (2007) RNAi-mediated gene silencing reveals involvement of Arabidopsis chromatin-related genes in Agrobacterium-mediated root transformation. Proc Natl Acad Sci 104:15156–15161. https://doi.org/10.1073/pnas.0706986104
Daimon Y, Takabe K, Tasaka M (2003) The CUP-SHAPED COTYLEDON genes promote adventitious shoot formation on calli. Plant Cell Physiol 44:113–121. https://doi.org/10.1093/pcp/pcg038
Debernardi JM, Tricoli DM, Ercoli MF, Hayta S, Ronald P, Palatnik JF, Dubcovsky J (2020) A GRF–GIF chimeric protein improves the regeneration efficiency of transgenic plants. Nat Biotechnol 38:1274–1279. https://doi.org/10.1038/s41587-020-0703-0
Della Rovere F, Fattorini L, D’Angeli S, Veloccia A, Del Duca S, Cai G, Falasca G, Altamura MM (2015) Arabidopsis SHR and SCR transcription factors and AUX1 auxin influx carrier control the switch between adventitious rooting and xylogenesis in planta and in in vitro cultured thin cell layers. Ann Bot 115:617–628. https://doi.org/10.1093/aob/mcu258
Foroozani M, Vandal MP, Smith AP (2021) H3K4 trimethylation dynamics impact diverse developmental and environmental responses in plants. Planta 253:4. https://doi.org/10.1007/s00425-020-03520-0
Furuta K, Kubo M, Sano K, Demura T, Fukuda H, Liu Y-G, Shibata D, Kakimoto T (2011) The CKH2/PKL chromatin remodeling factor negatively regulates cytokinin responses in Arabidopsis calli. Plant Cell Physiol 52:618–628. https://doi.org/10.1093/pcp/pcr022
Gaj MD (2004) Factors influencing somatic embryogenesis induction and plant regeneration with particular reference to Arabidopsis thaliana (L.) Heynh. Plant Growth Regul 43:27–47. https://doi.org/10.1023/B:GROW.0000038275.29262.fb
Gibney ER, Nolan CM (2010) Epigenetics and gene expression. Heredity 105:4–13. https://doi.org/10.1038/hdy.2010.54
Gordon SP, Heisler MG, Reddy GV, Ohno C, Das P, Meyerowitz EM (2007) Pattern formation during de novo assembly of the Arabidopsis shoot meristem. Development 134:3539–3548. https://doi.org/10.1242/dev.010298
Grzybkowska D, Morończyk J, Wójcikowska B, Gaj MD (2018) Azacitidine (5-AzaC)-treatment and mutations in DNA methylase genes affect embryogenic response and expression of the genes that are involved in somatic embryogenesis in Arabidopsis. Plant Growth Regul 85:243–256. https://doi.org/10.1007/s10725-018-0389-1
Guo C, Xu Y, Shi M, Lai Y, Wu X, Wang H, Zhu Z, Poethig RS, Wu G (2017) Repression of miR156 by miR159 regulates the timing of the juvenile-to-adult transition in Arabidopsis. Plant Cell 29:1293–1304. https://doi.org/10.1105/tpc.16.00975
Guo H, Fan Y, Guo H, Wu J, Yu X, Wei J, Lian X, Zhang L, Gou Z, Fan Y, Zeng F (2020) Somatic embryogenesis critical initiation stage-specific mCHH hypomethylation reveals epigenetic basis underlying embryogenic redifferentiation in cotton. Plant Biotechnol J 18:1648–1650. https://doi.org/10.1111/pbi.13336
Haberlandt G (2003) Culturversuche mit isolierten Pflanzenzellen. In: Laimer M, Rücker W (eds) Plant tissue culture: 100 years since Gottlieb Haberlandt. Springer, Vienna, pp 1–24
Han S-K, Wu M-F, Cui S, Wagner D (2015) Roles and activities of chromatin remodeling ATPases in plants. Plant J 83:62–77. https://doi.org/10.1111/tpj.12877
Hayta S, Smedley MA, Demir SU, Blundell R, Hinchliffe A, Atkinson N, Harwood WA (2019) An efficient and reproducible Agrobacterium-mediated transformation method for hexaploid wheat (Triticum aestivum L.). Plant Methods 15:121. https://doi.org/10.1186/s13007-019-0503-z
He C, Chen X, Huang H, Xu L (2012) Reprogramming of H3K27me3 is critical for acquisition of pluripotency from cultured Arabidopsis tissues. PLOS Genet 8:e1002911. https://doi.org/10.1371/journal.pgen.1002911
Heyman J, Cools T, Canher B, Shavialenka S, Traas J, Vercauteren I, Van den Daele H, Persiau G, De Jaeger G, Sugimoto K, De Veylder L (2016) The heterodimeric transcription factor complex ERF115–PAT1 grants regeneration competence. Nature Plants 2:1–7. https://doi.org/10.1038/nplants.2016.165
Hibara K, Takada S, Tasaka M (2003) CUC1 gene activates the expression of SAM-related genes to induce adventitious shoot formation. Plant J 36:687–696. https://doi.org/10.1046/j.1365-313X.2003.01911.x
Hiei Y, Ishida Y, Komari T (2014) Progress of cereal transformation technology mediated by Agrobacterium tumefaciens. Front Plant Sci 5:2
Horstman A, Bemer M, Boutilier K (2017) A transcriptional view on somatic embryogenesis. Regeneration 4:201–216. https://doi.org/10.1002/reg2.91
Hsu F-M, Gohain M, Allishe A, Huang Y-J, Liao J-L, Kuang L-Y, Chen P-Y (2018) Dynamics of the methylome and transcriptome during the regeneration of rice. Epigenomes 2:14. https://doi.org/10.3390/epigenomes2030014
Hu X, Xu L (2016) Transcription factors WOX11/12 directly activate WOX5/7 to promote root primordia initiation and organogenesis. Plant Physiol 172:2363–2373. https://doi.org/10.1104/pp.16.01067
Ikeuchi M, Sugimoto K, Iwase A (2013) Plant callus: mechanisms of induction and repression. Plant Cell 25:3159–3173. https://doi.org/10.1105/tpc.113.116053
Ikeuchi M, Iwase A, Rymen B, Harashima H, Shibata M, Ohnuma M, Breuer C, Morao AK, de Lucas M, De Veylder L, Goodrich J, Brady SM, Roudier F, Sugimoto K (2015) PRC2 represses dedifferentiation of mature somatic cells in Arabidopsis. Nature Plants 1:1–7. https://doi.org/10.1038/nplants.2015.89
Ikeuchi M, Ogawa Y, Iwase A, Sugimoto K (2016) Plant regeneration: cellular origins and molecular mechanisms. Development 143:1442–1451. https://doi.org/10.1242/dev.134668
Ikeuchi M, Iwase A, Rymen B, Lambolez A, Kojima M, Takebayashi Y, Heyman J, Watanabe S, Seo M, De Veylder L, Sakakibara H, Sugimoto K (2017) Wounding triggers callus formation via dynamic hormonal and transcriptional changes. Plant Physiol 175:1158–1174. https://doi.org/10.1104/pp.17.01035
Ikeuchi M, Favero DS, Sakamoto Y, Iwase A, Coleman D, Rymen B, Sugimoto K (2019) Molecular mechanisms of plant regeneration. Annu Rev Plant Biol 70:377–406. https://doi.org/10.1146/annurev-arplant-050718-100434
Ishihara H, Sugimoto K, Tarr PT, Temman H, Kadokura S, Inui Y, Sakamoto T, Sasaki T, Aida M, Suzuki T, Inagaki S, Morohashi K, Seki M, Kakutani T, Meyerowitz EM, Matsunaga S (2019) Primed histone demethylation regulates shoot regenerative competency. Nat Commun 10:1786. https://doi.org/10.1038/s41467-019-09386-5
Iwase A, Mitsuda N, Koyama T, Hiratsu K, Kojima M, Arai T, Inoue Y, Seki M, Sakakibara H, Sugimoto K, Ohme-Takagi M (2011) The AP2/ERF transcription factor WIND1 controls cell dedifferentiation in Arabidopsis. Curr Biol 21:508–514. https://doi.org/10.1016/j.cub.2011.02.020
Jing T, Ardiansyah R, Xu Q, Xing Q, Müller-Xing R (2020) Reprogramming of cell fate during root regeneration by transcriptional and epigenetic networks. Front Plant Sci 11:2
Kang H, Ma J, Wu D, Shen W-H, Zhu Y (2019) Functional coordination of the chromatin-remodeling factor AtINO80 and the histone chaperones NRP1/2 in inflorescence meristem and root apical meristem. Front Plant Sci 10:2
Kim JH (2019) Biological roles and an evolutionary sketch of the GRF-GIF transcriptional complex in plants. BMB Rep 52:227–238. https://doi.org/10.5483/BMBRep.2019.52.4.051
Kim J-Y, Yang W, Forner J, Lohmann JU, Noh B, Noh Y-S (2018) Epigenetic reprogramming by histone acetyltransferase HAG1/AtGCN5 is required for pluripotency acquisition in Arabidopsis. EMBO J 37:e98726. https://doi.org/10.15252/embj.201798726
Klemm SL, Shipony Z, Greenleaf WJ (2019) Chromatin accessibility and the regulatory epigenome. Nat Rev Genet 20:207–220. https://doi.org/10.1038/s41576-018-0089-8
Krikorian AD, Berquam DL (2003) Plant cell and tissue cultures: the role of haberlandt. In: Laimer M, Rücker W (eds) Plant tissue culture: 100 years since gottlieb haberlandt. Springer, Vienna, pp 25–53
Kumar SV (2018) H2A.Z at the core of transcriptional regulation in plants. Mol Plant 11:1112–1114. https://doi.org/10.1016/j.molp.2018.07.002
Kumar V, Thakur JK, Prasad M (2021) Histone acetylation dynamics regulating plant development and stress responses. Cell Mol Life Sci 78:4467–4486. https://doi.org/10.1007/s00018-021-03794-x
Lardon R, Geelen D (2020) Natural variation in plant pluripotency and regeneration. Plants 9:1261. https://doi.org/10.3390/plants9101261
Law JA, Jacobsen SE (2010) Establishing, maintaining and modifying DNA methylation patterns in plants and animals. Nat Rev Genet 11:204–220. https://doi.org/10.1038/nrg2719
Lee K, Park O-S, Jung S-J, Seo PJ (2016) Histone deacetylation-mediated cellular dedifferentiation in Arabidopsis. J Plant Physiol 191:95–100. https://doi.org/10.1016/j.jplph.2015.12.006
Lee K, Park O-S, Seo PJ (2017) Arabidopsis ATXR2 deposits H3K36me3 at the promoters of LBD genes to facilitate cellular dedifferentiation. Sci Signal 10:316. https://doi.org/10.1126/scisignal.aan0316
Lee K, Park O-S, Seo PJ (2018a) JMJ30-mediated demethylation of H3K9me3 drives tissue identity changes to promote callus formation in Arabidopsis. Plant J 95:961–975. https://doi.org/10.1111/tpj.14002
Lee K, Park O-S, Seo PJ (2018b) ATXR2 as a core regulator of de novo root organogenesis. Plant Signal Behav 13:e1449543. https://doi.org/10.1080/15592324.2018b.1449543
Lee K, Park O-S, Choi CY, Seo PJ (2019) ARABIDOPSIS TRITHORAX 4 facilitates shoot identity establishment during the plant regeneration process. Plant Cell Physiol 60:826–834. https://doi.org/10.1093/pcp/pcy248
Lee K, Park O-S, Lee HG, Seo PJ (2020) The ASHR3 SET-domain protein is a pivotal upstream coordinator for wound-induced callus formation in Arabidopsis. J Plant Biol 63:361–368. https://doi.org/10.1007/s12374-020-09259-1
Lee K, Park O-S, Go JY, Yu J, Han JH, Kim J, Bae S, Jung YJ, Seo PJ (2021) Arabidopsis ATXR2 represses de novo shoot organogenesis in the transition from callus to shoot formation. Cell Rep 37:109980. https://doi.org/10.1016/j.celrep.2021.109980
Li S-M, Zheng H-X, Zhang X-S, Sui N (2021) Cytokinins as central regulators during plant growth and stress response. Plant Cell Rep 40:271–282. https://doi.org/10.1007/s00299-020-02612-1
Li M, Zheng Y, Cui D, Du Y, Zhang D, Sun W, Du H, Zhang Z (2022) GIF1 controls ear inflorescence architecture and floral development by regulating key genes in hormone biosynthesis and meristem determinacy in maize. BMC Plant Biol 22:127. https://doi.org/10.1186/s12870-022-03517-9
Liu C, Lu F, Cui X, Cao X (2010) Histone methylation in higher plants. Annu Rev Plant Biol 61:395–420. https://doi.org/10.1146/annurev.arplant.043008.091939
Liu J, Sheng L, Xu Y, Li J, Yang Z, Huang H, Xu L (2014) WOX11 and 12 are involved in the first-step cell fate transition during de novo root organogenesis in Arabidopsis. Plant Cell 26:1081–1093. https://doi.org/10.1105/tpc.114.122887
Liu H, Ma X, Han HN, Hao YJ, Zhang XS (2016a) AtPRMT5 regulates shoot regeneration through mediating histone H4R3 dimethylation on KRPs and Pre-mRNA splicing of RKP in Arabidopsis. Mol Plant 9:1634–1646. https://doi.org/10.1016/j.molp.2016.10.010
Liu Z, Li J, Wang L, Li Q, Lu Q, Yu Y, Li S, Bai M, Hu Y, Xiang F (2016b) Repression of callus initiation by the miRNA-directed interaction of auxin–cytokinin in Arabidopsis thaliana. Plant J 87:391–402. https://doi.org/10.1111/tpj.13211
Liu H, Zhang H, Dong YX, Hao YJ, Zhang XS (2018a) DNA METHYLTRANSFERASE1-mediated shoot regeneration is regulated by cytokinin-induced cell cycle in Arabidopsis. New Phytol 217:219–232. https://doi.org/10.1111/nph.14814
Liu J, Hu X, Qin P, Prasad K, Hu Y, Xu L (2018b) The WOX11–LBD16 pathway promotes pluripotency acquisition in callus cells during de novo shoot regeneration in tissue culture. Plant Cell Physiol 59:739–748. https://doi.org/10.1093/pcp/pcy010
Liu Y, Liu K, Yin L, Yu Y, Qi J, Shen W-H, Zhu J, Zhang Y, Dong A (2019) H3K4me2 functions as a repressive epigenetic mark in plants. Epigenet Chromatin 12:40. https://doi.org/10.1186/s13072-019-0285-6
Liu D, Mu Q, Li X, Xu S, Li Y, Gu T (2022a) The callus formation capacity of strawberry leaf explants is modulated by DNA methylation. Horticult Res 9:73. https://doi.org/10.1093/hr/uhab073
Liu X, Bie X, Lin X, Li M, Wang H, Zhang X, Yang Y, Zhang C, Zhang X, Xiao J (2022b) Uncovering transcriptional regulatory network during regeneration for boosting wheat transformation. 2022b.10.21.513305
Long J-M, Liu C-Y, Feng M-Q, Liu Y, Wu X-M, Guo W-W (2018) miR156-SPL modules regulate induction of somatic embryogenesis in citrus callus. J Exp Bot 69:2979–2993. https://doi.org/10.1093/jxb/ery132
Lotan T, Ohto M, Yee KM, West MAL, Lo R, Kwong RW, Yamagishi K, Fischer RL, Goldberg RB, Harada JJ (1998) Arabidopsis LEAFY COTYLEDON1 is sufficient to induce embryo development in vegetative cells. Cell 93:1195–1205. https://doi.org/10.1016/S0092-8674(00)81463-4
Lowe K, Wu E, Wang N, Hoerster G, Hastings C, Cho M-J, Scelonge C, Lenderts B, Chamberlin M, Cushatt J, Wang L, Ryan L, Khan T, Chow-Yiu J, Hua W, Yu M, Banh J, Bao Z, Brink K, Igo E, Rudrappa B, Shamseer P, Bruce W, Newman L, Shen B, Zheng P, Bidney D, Falco C, Register J, Zhao Z-Y, Xu D, Jones T, Gordon-Kamm W (2016) Morphogenic regulators baby boom and wuschel improve monocot transformation. Plant Cell 28:1998–2015. https://doi.org/10.1105/tpc.16.00124
Loyola-Vargas VM, Ochoa-Alejo N (2018) An introduction to plant tissue culture: advances and perspectives. In: Loyola-Vargas VM, Ochoa-Alejo N (eds) Plant cell culture protocols. Springer, New York, pp 3–13
Luo G, Palmgren M (2021) GRF-GIF chimeras boost plant regeneration. Trends Plant Sci 26:201–204. https://doi.org/10.1016/j.tplants.2020.12.001
Mathew MM, Prasad K (2021) Model systems for regeneration: Arabidopsis. Development 148:195347. https://doi.org/10.1242/dev.195347
Mironova V, Xu J (2019) A single-cell view of tissue regeneration in plants. Curr Opin Plant Biol 52:149–154. https://doi.org/10.1016/j.pbi.2019.09.003
Morończyk J, Brąszewska A, Wójcikowska B, Chwiałkowska K, Nowak K, Wójcik AM, Kwaśniewski M, Gaj MD (2022) Insights into the histone acetylation-mediated regulation of the transcription factor genes that control the embryogenic transition in the somatic cells of Arabidopsis. Cells 11:863. https://doi.org/10.3390/cells11050863
Mozgová I, Muñoz-Viana R, Hennig L (2017) PRC2 represses hormone-induced somatic embryogenesis in vegetative tissue of Arabidopsis thaliana. PLoS Genet 13:e1006562. https://doi.org/10.1371/journal.pgen.1006562
Negin B, Shemer O, Sorek Y, Williams LE (2017) Shoot stem cell specification in roots by the WUSCHEL transcription factor. PLoS ONE 12:e0176093. https://doi.org/10.1371/journal.pone.0176093
Ojolo SP, Cao S, Priyadarshani SVGN, Li W, Yan M, Aslam M, Zhao H, Qin Y (2018) Regulation of plant growth and development: a review from a chromatin remodeling perspective. Front Plant Sci 9:2
Pfluger J, Wagner D (2007) Histone modifications and dynamic regulation of genome accessibility in plants. Curr Opin Plant Biol 10:645–652. https://doi.org/10.1016/j.pbi.2007.07.013
Powers SK, Strader LC (2020) Regulation of auxin transcriptional responses. Dev Dyn 249:483–495. https://doi.org/10.1002/dvdy.139
Qiao M, Zhao Z, Song Y, Liu Z, Cao L, Yu Y, Li S, Xiang F (2012) Proper regeneration from in vitro cultured Arabidopsis thaliana requires the microRNA-directed action of an auxin response factor. Plant J 71:14–22. https://doi.org/10.1111/j.1365-313X.2012.04944.x
Rymen B, Kawamura A, Lambolez A, Inagaki S, Takebayashi A, Iwase A, Sakamoto Y, Sako K, Favero DS, Ikeuchi M, Suzuki T, Seki M, Kakutani T, Roudier F, Sugimoto K (2019) Histone acetylation orchestrates wound-induced transcriptional activation and cellular reprogramming in Arabidopsis. Commun Biol 2:1–15. https://doi.org/10.1038/s42003-019-0646-5
Sang YL, Cheng ZJ, Zhang XS (2018) Plant stem cells and de novo organogenesis. New Phytol 218:1334–1339. https://doi.org/10.1111/nph.15106
Shemer O, Landau U, Candela H, Zemach A, Eshed Williams L (2015) Competency for shoot regeneration from Arabidopsis root explants is regulated by DNA methylation. Plant Sci 238:251–261. https://doi.org/10.1016/j.plantsci.2015.06.015
Shim S, Kim HK, Bae SH, Lee H, Lee HJ, Jung YJ, Seo PJ (2020) Transcriptome comparison between pluripotent and non-pluripotent calli derived from mature rice seeds. Sci Rep 10:21257. https://doi.org/10.1038/s41598-020-78324-z
Shim S, Lee HG, Seo PJ (2021) MET1-dependent DNA methylation represses light signaling and influences plant regeneration in Arabidopsis. Mol Cells 44:746–757. https://doi.org/10.14348/molcells.2021.0160
Shim S, Lee HG, Park O-S, Shin H, Lee K, Lee H, Huh JH, Seo PJ (2022) Dynamic changes in DNA methylation occur in TE regions and affect cell proliferation during leaf-to-callus transition in Arabidopsis. Epigenetics 17:41–58. https://doi.org/10.1080/15592294.2021.1872927
Shimotohno A, Heidstra R, Blilou I, Scheres B (2018) Root stem cell niche organizer specification by molecular convergence of PLETHORA and SCARECROW transcription factor modules. Genes Dev. https://doi.org/10.1101/gad.314096.118
Shin J, Bae S, Seo PJ (2020) De novo shoot organogenesis during plant regeneration. J Exp Bot 71:63–72. https://doi.org/10.1093/jxb/erz395
Skoog F, Miller CO (1957) Chemical regulation of growth and organ formation in plant tissues cultured in vitro. Symp Soc Exp Biol 11:118–130
Stone SL, Kwong LW, Yee KM, Pelletier J, Lepiniec L, Fischer RL, Goldberg RB, Harada JJ (2001) LEAFY COTYLEDON2 encodes a B3 domain transcription factor that induces embryo development. Proc Natl Acad Sci 98:11806–11811. https://doi.org/10.1073/pnas.201413498
Sugimoto K, Jiao Y, Meyerowitz EM (2010) Arabidopsis regeneration from multiple tissues occurs via a root development pathway. Dev Cell 18:463–471. https://doi.org/10.1016/j.devcel.2010.02.004
Sugimoto K, Temman H, Kadokura S, Matsunaga S (2019) To regenerate or not to regenerate: factors that drive plant regeneration. Curr Opin Plant Biol 47:138–150. https://doi.org/10.1016/j.pbi.2018.12.002
Sun B, Zhou Y, Cai J, Shang E, Yamaguchi N, Xiao J, Looi L-S, Wee W-Y, Gao X, Wagner D, Ito T (2019) Integration of transcriptional repression and polycomb-mediated silencing of WUSCHEL in floral meristems. Plant Cell 31:1488–1505. https://doi.org/10.1105/tpc.18.00450
Sun R-Z, Zuo E-H, Qi J-F, Liu Y, Lin C-T, Deng X (2020) A role of age-dependent DNA methylation reprogramming in regulating the regeneration capacity of Boea hygrometrica leaves. Funct Integr Genom 20:133–149. https://doi.org/10.1007/s10142-019-00701-3
Suo J, Zhou C, Zeng Z, Li X, Bian H, Wang J, Zhu M, Han N (2021) Identification of regulatory factors promoting embryogenic callus formation in barley through transcriptome analysis. BMC Plant Biol 21:145. https://doi.org/10.1186/s12870-021-02922-w
Thorpe TA (2007) History of plant tissue culture. Mol Biotechnol 37:169–180. https://doi.org/10.1007/s12033-007-0031-3
Wang K, Liu H, Du L, Ye X (2017) Generation of marker-free transgenic hexaploid wheat via an Agrobacterium-mediated co-transformation strategy in commercial Chinese wheat varieties. Plant Biotechnol J 15:614–623. https://doi.org/10.1111/pbi.12660
Wang L, Liu Z, Qiao M, Xiang F (2018) miR393 inhibits in vitro shoot regeneration in Arabidopsis thaliana via repressing TIR1. Plant Sci 266:1–8. https://doi.org/10.1016/j.plantsci.2017.10.009
Wang F-X, Shang G-D, Wu L-Y, Xu Z-G, Zhao X-Y, Wang J-W (2020) Chromatin accessibility dynamics and a hierarchical transcriptional regulatory network structure for plant somatic embryogenesis. Dev Cell 54:742-757.e8. https://doi.org/10.1016/j.devcel.2020.07.003
Wang K, Shi L, Liang X, Zhao P, Wang W, Liu J, Chang Y, Hiei Y, Yanagihara C, Du L, Ishida Y, Ye X (2022) The gene TaWOX5 overcomes genotype dependency in wheat genetic transformation. Nat Plants 8:110–117. https://doi.org/10.1038/s41477-021-01085-8
Wójcik AM, Gaj MD (2016) miR393 contributes to the embryogenic transition induced in vitro in Arabidopsis via the modification of the tissue sensitivity to auxin treatment. Planta 244:231–243. https://doi.org/10.1007/s00425-016-2505-7
Wójcikowska B, Botor M, Morończyk J, Wójcik AM, Nodzyński T, Karcz J, Gaj MD (2018) Trichostatin A triggers an embryogenic transition in Arabidopsis explants via an auxin-related pathway. Front Plant Sci 9:2
Wu L-Y, Shang G-D, Wang F-X, Gao J, Wan M-C, Xu Z-G, Wang J-W (2022) Dynamic chromatin state profiling reveals regulatory roles of auxin and cytokinin in shoot regeneration. Dev Cell 57:526-542.e7. https://doi.org/10.1016/j.devcel.2021.12.019
Xia K, Sun H-X, Li J, Li J, Zhao Y, Chen L, Qin C, Chen R, Chen Z, Liu G, Yin R, Mu B, Wang X, Xu M, Li X, Yuan P, Qiao Y, Hao S, Wang J, Xie Q, Xu J, Liu S, Li Y, Chen A, Liu L, Yin Y, Yang H, Wang J, Gu Y, Xu X (2022) The single-cell stereo-seq reveals region-specific cell subtypes and transcriptome profiling in Arabidopsis leaves. Dev Cell 57:1299-1310.e4. https://doi.org/10.1016/j.devcel.2022.04.011
Xiao J, Lee U-S, Wagner D (2016) Tug of war: adding and removing histone lysine methylation in Arabidopsis. Curr Opin Plant Biol 34:41–53. https://doi.org/10.1016/j.pbi.2016.08.002
Xu L (2018) De novo root regeneration from leaf explants: wounding, auxin, and cell fate transition. Curr Opin Plant Biol 41:39–45. https://doi.org/10.1016/j.pbi.2017.08.004
Xu M, Hu T, Zhao J, Park M-Y, Earley KW, Wu G, Yang L, Poethig RS (2016) Developmental functions of miR156-regulated SQUAMOSA PROMOTER BINDING PROTEIN-LIKE (SPL) genes in Arabidopsis thaliana. PLOS Genet 12:e1006263. https://doi.org/10.1371/journal.pgen.1006263
Xu M, Du Q, Tian C, Wang Y, Jiao Y (2021) Stochastic gene expression drives mesophyll protoplast regeneration. Sci Adv 7:8466. https://doi.org/10.1126/sciadv.abg8466
Yan A, Borg M, Berger F, Chen Z (2020) The atypical histone variant H3.15 promotes callus formation in Arabidopsis thaliana. Development 147:184895. https://doi.org/10.1242/dev.184895
Yang S, Li C, Zhao L, Gao S, Lu J, Zhao M, Chen C-Y, Liu X, Luo M, Cui Y, Yang C, Wu K (2015) The arabidopsis SWI2/SNF2 chromatin remodeling ATPase BRAHMA targets directly to PINs and is required for root stem cell niche maintenance. Plant Cell 27:1670–1680. https://doi.org/10.1105/tpc.15.00091
Yang W, Choi M-H, Noh B, Noh Y-S (2020) De novo shoot regeneration controlled by HEN1 and TCP3/4 in arabidopsis. Plant Cell Physiol 61:1600–1613. https://doi.org/10.1093/pcp/pcaa083
Zhai N, Xu L (2021) Pluripotency acquisition in the middle cell layer of callus is required for organ regeneration. Nat Plants 7:1453–1460. https://doi.org/10.1038/s41477-021-01015-8
Zhang W, Wu Y, Schnable JC, Zeng Z, Freeling M, Crawford GE, Jiang J (2012) High-resolution mapping of open chromatin in the rice genome. Genome Res 22:151–162. https://doi.org/10.1101/gr.131342.111
Zhang T-Q, Lian H, Tang H, Dolezal K, Zhou C-M, Yu S, Chen J-H, Chen Q, Liu H, Ljung K, Wang J-W (2015) An intrinsic microRNA timer regulates progressive decline in shoot regenerative capacity in plants. Plant Cell 27:349–360. https://doi.org/10.1105/tpc.114.135186
Zhang K, Xu W, Wang C, Yi X, Zhang W, Su Z (2017a) Differential deposition of H2A.Z in combination with histone modifications within related genes in Oryza sativa callus and seedling. Plant J 89:264–277. https://doi.org/10.1111/tpj.13381
Zhang T-Q, Lian H, Zhou C-M, Xu L, Jiao Y, Wang J-W (2017b) A two-step model for de novo activation of WUSCHEL during plant shoot regeneration. Plant Cell 29:1073–1087. https://doi.org/10.1105/tpc.16.00863
Zhang H, Lang Z, Zhu J-K (2018a) Dynamics and function of DNA methylation in plants. Nat Rev Mol Cell Biol 19:489–506. https://doi.org/10.1038/s41580-018-0016-z
Zhang W, Yin M, Zhao P, Wang K, Lipu D, Ye X (2018b) Regeneration capacity evaluation of some largely popularized wheat varieties in China. Acta Agron Sin 2:2
Zhang H, Guo F, Qi P, Huang Y, Xie Y, Xu L, Han N, Xu L, Bian H (2020) OsHDA710-mediated histone deacetylation regulates callus formation of rice mature embryo. Plant Cell Physiol 61:1646–1660. https://doi.org/10.1093/pcp/pcaa086
Zhao N, Zhang K, Wang C, Yan H, Liu Y, Xu W, Su Z (2020) Systematic analysis of differential H3K27me3 and H3K4me3 deposition in callus and seedling reveals the epigenetic regulatory mechanisms involved in callus formation in rice. Front Genet 11:2
Zheng B, Liu J, Gao A, Chen X, Gao L, Liao L, Luo B, Ogutu CO, Han Y (2022) Epigenetic reprogramming of H3K27me3 and DNA methylation during leaf-to-callus transition in peach. Horticult Res 9:132. https://doi.org/10.1093/hr/uhac132
Zhong X, Du J, Hale CJ, Gallego-Bartolome J, Feng S, Vashisht AA, Chory J, Wohlschlegel JA, Patel DJ, Jacobsen SE (2014) Molecular mechanism of action of plant DRM de novo DNA methyltransferases. Cell 157:1050–1060. https://doi.org/10.1016/j.cell.2014.03.056
Acknowledgements
We apologize to colleagues whose works were not cited due to space limitations. This research was supported by the Strategic Priority Research Program of the Chinese Academy of Sciences (XDA24010204), the National Key Research and Development to Program of China (2021YFD1201500), and the National Natural Sciences Foundation of China (31970529) to J.X.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, X., Zhu, K. & Xiao, J. Recent advances in understanding of the epigenetic regulation of plant regeneration. aBIOTECH 4, 31–46 (2023). https://doi.org/10.1007/s42994-022-00093-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42994-022-00093-2