Abstract
Preterm birth (PTB), defined as the birth of a child before 37 completed weeks gestation, affects approximately 11% of live births and is the leading cause of death in children under 5 years. PTB is a complex disease with multiple risk factors including genetic variation. Much research has aimed to establish the biological mechanisms underlying PTB often through identification of genetic markers for PTB risk. The objective of this review is to present a comprehensive and updated summary of the published data relating to the field of PTB genetics. A literature search in PubMed was conducted and English studies related to PTB genetics were included. Genetic studies have identified genes within inflammatory, immunological, tissue remodeling, endocrine, metabolic, and vascular pathways that may be involved in PTB. However, a substantial proportion of published data have been largely inconclusive and multiple studies had limited power to detect associations. On the contrary, a few large hypothesis-free approaches have identified and replicated multiple novel variants associated with PTB in different cohorts. Overall, attempts to predict PTB using single “-omics” datasets including genomic, transcriptomic, and epigenomic biomarkers have been mostly unsuccessful and have failed to translate to the clinical setting. Integration of data from multiple “-omics” datasets has yielded the most promising results.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Preterm birth (PTB) (defined as the birth of a child before 37 completed weeks gestation) is the leading cause of death and disability in children under 5 years of age worldwide [1, 2]. Whilst PTB is becoming a preventable condition for a very small subset of women [3,4,5,6,7,8,9], global rates of PTB continue to rise [10, 11]. Recent estimates report that PTB affects approximately 11% of all livebirths, or approximately 15 million PTB per year [11]. PTB has substantial short- and long-term sequelae related to physical and neurocognitive development [12, 13]. Short-term, PTB is associated with early mortality [14,15,16]. Long-term complications of PTB include early development of adult-onset diseases like certain cancers, cardiovascular disease, insulin resistance, chronic kidney disease, obesity, and neurodevelopmental and social disability [17, 18]. Multiple risk factors for PTB have been identified including environmental factors such as demographic and lifestyle risk factors, maternal medical disorders, and antenatal risk factors (Table 1). One of the strongest risk factors, and thus greatest predictors, for PTB is a prior history of preterm delivery [19, 20], indicating that there may be genetic predispositions to PTB. Since the first draft of the human genome sequence was completed in 2001 [21], there has been an increase in information regarding the genetics of diseases, including PTB. Significant advances in technology have occurred within the last two decades that have increased the ability to obtain genetic data, providing the opportunity to address challenges in preventing and managing diseases. Studies have been conducted to improve the understanding of biological mechanisms underpinning PTB and translate research findings into a clinical setting. The development of prediction tools that stratify care to provide targeted and personalized prevention of PTB has been a major focus in many PTB-related genetic studies.
Preterm Birth Phenotype
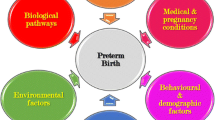
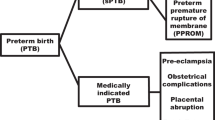
Investigating the genetics of PTB is challenging because the PTB population is highly heterogenous. Multiple mechanisms have been implicated in PTB development including infection and inflammation, excess activation of the hypothalamic-pituitary-adrenal axis, uterine defects or over-distention, stress, cervical disease, vascular disease, maternal-fetal tolerance disruption, and decreased progesterone action (Fig. 1) [30]. Independent of these various risk factors, PTB can be grouped into three broad clinical phenotypes: medically indicated PTB, idiopathic PTB (iPTB), and preterm prelabor rupture of membranes (PPROM). The latter two are often grouped as “spontaneous PTB (sPTB)” because labor occurs spontaneously in both cases (Fig. 2) [31]. PTB can also be subcategorized based on gestational age at delivery: for example, the World Health Organization categorizes PTB as extreme PTB (<28 weeks’ gestation), very preterm (28 to <32 weeks’ gestation), and moderate or late PTB (32 to <37 weeks’ gestation) [32]. One approach to study the genetics of PTB is to aggregate all sub-phenotypes into a single broadly phenotyped cohort. If the genetic mechanisms underpinning medically indicated PTB and sPTB are shared, combining sub-phenotypes may increase statistical power [33]. An alternative approach is to stratify by the diverse clinical conditions of PTB. Given the complex and heterogenous nature of PTB, testing in more homogenous cohorts may increase the sensitivity to detect genetic associations, even if they are only limited to specific risk groups [34].
Mechanisms proposed to underpin spontaneous preterm birth. Figure adapted from [30]
Preterm birth (PTB) phenotypes. PTB accounts for approximately 11% of all livebirths and can be subcategorized into medically indicated PTB and spontaneous PTB (sPTB). sPTB includes both preterm prelabor rupture of membranes (PPROM) and idiopathic PTB. PTB can be medically indicated for several reasons involving both mother and baby. Estimates are based off data published by Chawanpaiboon et al. and Morken et al. [11, 31]
Heritability of Preterm Birth
The heritability of PTB has been assessed through large intergenerational studies, as well as in studies of monozygotic twins and siblings. There is a 20% increased risk of PTB if a mother was born preterm herself; this risk is inversely correlated to the mother’s gestational age at birth [19, 35]. Furthermore, women with sisters who delivered preterm babies are at an 80% increased risk of delivering preterm themselves [20, 36]. Twin studies have also suggested that genetics account for 17–36% of PTB risk [37, 38]. In attempting to understand the heritability of PTB, both maternal and fetal genetics have been assessed. It has been suggested that maternal genetics are responsible for 22.8% of the variations in gestational age in spontaneous births, with fetal genetics responsible for 12.7% of the variation [39]. The rates of PTB have also been consistently higher among African Americans, even after adjustment for socioeconomic confounders [35, 40,41,42,43,44]. The ethnic disparities, in conjunction with findings from twin and heritability studies, have driven researchers to further explore the genetics underpinning this disease.
Approaches to Investigate the Genetic Basis of Preterm Birth
There are three distinct observational study designs that are commonly used to investigate the genetic basis of diseases including PTB: cohort studies, case-control studies, and family-based studies [34]. Cohort studies define a population either retrospectively or prospectively based on exposure [45]. An advantage of cohort studies is that they allow gestational age to be considered as a continuous outcome. In contrast, case-control studies retrospectively define a population based on outcome and dichotomize the cohort to assess for, in this case, genetic variants present in cases versus controls [45]. Family-based studies, such as case-parent trio analyses, utilize relatives as controls to overcome population stratification. In PTB genetic studies, family-based approaches allow for the genetic contributions of both the mother and baby to be assessed [34, 46].
Employing these broad study designs, many approaches to investigate the genetics of PTB have been established including large intergenerational studies, twin studies, family-based linkage studies, candidate gene studies, genome-wide association studies (GWAS), whole-exome sequencing (WES) and whole genome sequencing (WGS) analyses, copy number variant (CNV) analyses, and mitochondrial genetic studies [47, 48]. Research has also been conducted to explore gene-gene interactions, gene-environment interactions, epigenetics modifications, and transcriptomic changes associated with PTB [49]. Some studies have investigated the biological consequences of genetic polymorphisms by assessing the proteome and metabolome of PTB cases [50, 51]. Recently, studies have also conducted multiple “-omics” analyses to investigate the complex disease of PTB [52].
This review will focus on candidate gene studies, GWAS, WES and WGS analyses, CNV analyses, and de novo mutation analyses, as well as PTB-related transcriptomic and epigenomic studies. Refer to Table 2 and Fig. 3 for definitions and descriptions of the genetic variants and study designs mentioned in this review. We aim to present a comprehensive and updated summary of the published data relating to this field of research reviewing the literature arising from PubMed searches.
Preterm Birth Genetics
Candidate Gene Studies
The candidate gene approach has been applied in many studies that aim to understand the mechanisms underlying PTB and identify potential biomarkers of prematurity. Candidate gene studies have identified over 750 SNPs within more than 240 genes in both mothers and infants of varying ethnicities that may be associated with PTB or gestational duration at birth (Online resource 2). This review aims to broadly discuss and summarize the findings from the large number of candidate gene studies published. These studies have implicated genes involved in tissue remodeling, vascular, endothelial, metabolic, inflammatory, and immune processes.
Inflammatory and Immunological Pathway-Related Polymorphisms
Inflammation is a biological response of the immune as well as nonimmune system [63], and inflammatory responses are tightly regulated during pregnancy [64, 65]. It is thought that labor is partly initiated by a switch from a balanced inflammatory state to a pro-inflammatory state [66]. Genetic polymorphisms that play a role in immunological responses during pregnancy could potentially result in overwhelming inflammatory responses and subsequently impact gestational duration. Candidate gene studies have extensively analyzed genomic loci involved in these processes, with a prominent focus on the innate immune system.
Polymorphisms within genes involved in pathogen recognition have been associated with both early and late PTB. Specifically, toll-like receptor 1 (TLR 1), toll-like receptor 2 (TLR 2), and toll-like receptor 7 (TLR 7) gene polymorphisms have exhibited associations with PTB when analyzing maternal samples [67,68,69]. Studies analyzing infant samples have highlighted similar results [68, 70].
Inflammation can be activated by both pathogen recognition and the recognition of tissue damage. It is an integral component of the innate immune system and is produced by eicosanoids and cytokines that are released from affected cells [63]. Genes encoding both pro- and anti-inflammatory cytokines, particularly interleukins, have been investigated in several candidate gene studies. Polymorphisms within genes encoding pro-inflammatory interleukins have been associated with PTB in mothers and infants [68, 71,72,73,74,75]. Several studies have also identified SNPs associated with PTB in genes encoding anti-inflammatory cytokines like interleukin 10 (IL10) and interleukin 13 (IL13) genes [67, 76,77,78]. Furthermore, genes encoding interleukin receptors and co-receptors involved in immunomodulation have been highlighted as potential genes involved in PTB [69, 79,80,81]. In addition to interleukins, cytokines such as tumor necrosis factor-α (TNFα), a pro-inflammatory cytokine, have been implicated in candidate gene studies that have investigated polymorphisms within encoding genes [71, 82,83,84,85].
Polymorphisms within genes involved in complement activation and regulation, as well as complement-opsonized pathogen clearance, have been associated with PTB [86, 87]. Furthermore, genes involved in innate immune cell functions such as cell migration and receptor function have been highlighted as potential risk genes [88, 89].
Genetic polymorphisms within genes involved in B and T cell activation, migration, and proliferation were implicated in those with preterm delivery [76, 80]. Furthermore, maternal polymorphisms within genes encoding proteins involved in immunoglobulin synthesis and function have been identified as potential risks for PTB [90,91,92].
Tissue Remodeling Pathway-Related Polymorphisms
Extensive tissue remodeling occurs throughout pregnancy and labor; the uterus gains smooth muscle and connective tissue, and prior to labor, the cervix softens [93]. Genes involved in tissue remodeling and hormones involved in both maintaining pregnancy and parturition have been analyzed. SNPs within genes involved in collagen biosynthesis and the synthesis and inhibition of matrix metalloproteases have exhibited associations with PTB [68, 94,95,96]. Polymorphisms within genes encoding fibroblast growth factors 1 and 4, both involved in embryonic development, morphogenesis, cell growth, and angiogenesis, have demonstrated associations with sPTB in maternal and infant samples [68, 87, 97]. Genes encoding parturition-related hormones including progesterone, follicle-stimulating hormone, corticotrophin-releasing hormone, and relaxin have also been implicated in candidate genes studies [98,99,100,101,102,103,104]. Kim et al. (2013) analyzed approximately 650 case-parents triads; maternal genetic effect analyses identified SNPs within leucyl and cysteinyl aminopeptidase gene (LNPEP) associated with prematurity. LNPEP encodes an enzyme responsible for the degradation of hormones vital for maintaining pregnancy [105].
Metabolic and Biosynthetic Pathway-Related Polymorphisms
Sustaining metabolic homeostasis is vital for embryonic development and survival. Polymorphisms within genes responsible for the biosynthesis and metabolism of fatty acids, lipoproteins, triglycerides, and cholesterol have been associated with PTB in women and infants of various ethnicities [90, 106, 107]. Several polymorphisms within insulin growth factor 1 gene (IGF1), insulin growth factor 2 gene (IGF2) and the genes encoding their receptors have demonstrated a higher risk of PTB [68, 108, 109]. Both IGF1 and IGF2 encode growth factors similar in function to insulin, with roles in mediating growth, including fetoplacental development [110]. SNPs associated with sPTB within genes involved in glucose and protein homeostasis folate and methionine synthesis as well as the metabolism and storage of vitamin D and other minerals have been identified in PTB cases [109, 111,112,113,114]. A recent study of 254 Korean women also established the role of transcobalamin transport in PTB [115]. Genes involved in the detoxification and metabolism of organophosphates and xenobiotics have also shown an association with PTB [81, 116].
Hematological, Vascular, and Endothelial Pathway-Related Polymorphisms
Genes involved in producing coagulation factors have been investigated and are associated with PTB in some candidate gene studies [95, 117]. Genes that reduce thrombin formation, as well as both clot formation and degradation, have also been implicated in studies using infant samples [118, 119]. Prostaglandins have a well-established role in inducing myometrial contraction and are involved in inflammatory, angiogenic, and platelet aggregation pathways [120, 121]. A 2012 study assessed genes involved in prostaglandin synthesis and function including prostaglandin-endoperoxide synthase 1 (PTGS1), prostaglandin-endoperoxide synthase 2 (PTGS2), prostaglandin E synthase (PTGES), and prostaglandin E synthase 2 (PTGES2) genes [122]. The study included 542 preterm and 568 term African American mothers, adjusted for multiple lifestyle-related confounders, and identified several SNPs significantly associated with PTB. Similar findings have been established within PTGS1, PTGS2, prostaglandin E receptor 2 (PTGER2), and prostaglandin E receptor 3 (PTGER3) genes in maternal samples and infant samples [49, 69, 81, 123]. Polymorphisms within genes encoding growth factors involved in vasculogenesis, angiogenesis, endothelin, and endothelial growth have all been implicated in PTB [97, 124, 125]. Potential vascular smooth muscle relaxation via polymorphisms in nitric oxide synthase 2 gene (NOS2) and nitric oxide synthase 3 gene (NOS3) have also been associated with this phenotype [118, 126], supporting the role of the vascular and endothelial system in preterm delivery.
Limitations in Candidate Gene Studies
Candidate gene studies present a large body of research in PTB genetics and are relatively cost-effective with less sample size demands compared to other large-scale hypothesis-free studies such as GWAS. However, the findings are mostly inconclusive; studies commonly lacked adequate sample sizes and often did not conduct replication/validation analyses. Likewise, SNPs identified in candidate gene studies have not been replicated in large GWAS. Confounders were often not accounted for in these studies, despite knowledge that PTB is influenced by several environmental factors. Overall, published findings have been of minimal clinical utility thus far. Candidate gene studies are limited in that they are influenced by prior research and, due to study design, cannot identify novel variants involved in PTB. Online Resource 1 provides an overview of the genes identified in PTB-related candidate gene studies, grouped by biological pathway. A comprehensive catalogue of summary findings from the candidate gene studies is provided in Online Resource 2. Few mitochondrial SNPs have been studied; those associated with PTB are included in Online Resource 2.
Genome-Wide Association Studies
A substantial improvement in understanding the genetics of complex diseases has occurred within recent decades with the implementation of GWAS. To our knowledge, 13 GWAS published in the last 10 years have assessed PTB phenotypes including sPTB, idiopathic PTB, PPROM, PTB at varying gestational ages, and gestational duration. Whilst there is a clinical overlap between PTB and decreased gestational duration, note that gestational duration also includes normal variations in birth timing and therefore the genetic variants identified in analyses of gestational duration may not play the same role in PTB.
A large maternal genome-wide meta-analysis has recently been conducted with sample sizes fivefold larger than previously published [127]. GWAS data from 18 cohorts was utilized, providing almost 200,000 European maternal samples for gestational duration analyses and over 270,000 samples for sPTB analyses (18,797 preterm and 260,245 term births). The study identified genetic variants at 22 loci that were associated with gestational duration and 6 loci that were associated with sPTB, at a genome-wide significance (defined as P <5×10−08 [128]). Whilst most identified loci were distributed between the two phenotypes, the study calculated the observed estimate of genetic correlation to be moderate (rg = −0.62, 95% confidence interval (CI) −0.72 to −0.51), indicating that there may be variations in the genetic effects on gestational duration versus sPTB. The authors also demonstrated that maternal genetics had a larger impact on PTB when compared to fetal genetics, although overall effect sizes were observed to be small. Subsequent analyses to evaluate the use of the variants identified to predict PTB phenotypes including GA and sPTB, expectedly, suggested poor predictive performances (area under the receiver operator characteristic (AUROC) = 0.61, OR = 0.69, 95% CI 0.56–0.85) [127].
Zhang et al. (2017) published a large GWAS that utilized DNA samples from over 40,000 European women (3331 preterm and 40,236 term births) from 23andMe and identified several genetic variants associated with sPTB or gestational duration in both discovery and replication analyses at a genome-wide significance. Polymorphisms located in EBF transcription factor 1 (EBF1), angiotensin II receptor type 2 (AGTR2), adenylate cyclase 5 (ADCY5), and ras-related protein rap-2c (RAP2C) genes were strongly associated with decreased gestational duration. SNPs within both EBF1 and AGTR2 were associated with an increased risk of sPTB (<37 weeks). Variants located within eukaryote elongation factor selenocysteine-tRNA-specific gene (EEFSEC) and Wnt family member 4 gene (WNT4) were associated with increased gestational duration, and deletion variations within SNPs in EEFSEC were also associated with a decreased risk of sPTB [129].
Recently, Gupta et al. (2022) published a GWAS that examined the relationship between the maternal genome and early PTB of various sub-phenotypes. The GWAS analyses of 310 Caucasian women compared sPTB, iPTB, and PPROM against both low-risk and high-risk term births, as determined by the absence or presence of a previous history of PTB, respectively. The study identified several polymorphisms associated with all three sub-phenotypes at a genome-wide significance [130].
To assess the fetal effects on timing of parturition, Lui et al. (2019) conducted a large fetal GWAS meta-analysis of gestational duration, early preterm (<34 weeks), preterm (<37 weeks), and post-term births. The study identified a polymorphism within cytoskeleton associated protein 2 like gene (CKAP2L), a gene involved in the pro-inflammatory pathway [131], that was protective against decreased gestational duration in combined discovery and replication analyses (β = 0.034, 95% CI 0.025–0.043, P=3.96×10−14). The GWAS also identified a SNP in LIM domain containing preferred translocation partner in lipoma gene (LPP) and a SNP in spermatogenesis associated 6 gene (SPATA6); both were associated with a 1.64-fold increased odds of early sPTB. However, the SNPs could not be replicated in an independent cohort due to insufficient power [131].
Infant data has also been analyzed in a GWAS that aimed to identify genetic polymorphisms associated with PTB <30 weeks’ gestation [132]. Within an African sub-cohort of 190 cases and 1684 controls, this study identified an intergenic SNP located between YWHAQ pseudogene 9 (YWHAQP9) and RP11-136B18 that was associated with a 2.81-fold increased odds of PTB (P=4.5×10−09). The study also identified a second intergenic SNP located between family with sequence similarity 87 member A gene (FAM87A) and F-box protein 25 gene (FBXO25) that was associated with a 0.57-fold decreased odds of PTB within an American sub-cohort (n=1847) (P=3.71×10−08) [132].
An overview of the GWAS discussed in this review is provided in Table 3. Online Resource 3 includes further details of the GWAS published between 2011 and 2023 with genome-wide significant results. Several other PTB-related GWAS have been published that have not identified genetic variants associated with PTB at a genome-wide level, although some have published data that almost reach this P-value threshold [133,134,135,136,137,138,139]. It is likely that a subset of these studies did not identify genome-wide significant results due to the use of broadly and heterogeneously phenotyped cohorts that reflect different genetic pathways and relatively small sample sizes. Most GWAS discussed above analyzed iPTBs and pPROMs as a single group of sPTBs. Whilst these conditions share several etiological and clinical similarities, the events leading to both conditions are often distinct. Grouping the clinical conditions could be a limitation for GWAS and future studies should consider analyzing the sub-phenotypes separately. Another common limitation across published GWAS is the lack of successful replication with independent cohorts and among studies. Future studies with large sample sizes that utilize carefully phenotyped cohorts may increase the power needed to successfully identify significant loci with larger effect size. However, the economic and feasibility concerns associated with this pose challenges to conducting such studies; hence, few large-scale PTB-related GWAS have been conducted. The results from GWAS published to date have identified novel variants and biological pathways, advancing our understanding of the role of genetics in PTB. The challenge is translating these findings for clinical use to identify women at increased genetic risk of delivering preterm.
Whole-Exome Sequencing and Whole-Genome Sequencing Analyses
Recent advances in DNA sequencing technologies have allowed genetic association studies to conduct WES and WGS. However, to date, few WES or WGS studies have been performed to investigate the role of genetics in PTB, likely due to the increased costs of genetic sequencing [140].
Modi et al. (2017) performed whole-exome sequencing using neonatal DNA derived from PPROM cases (n=49) and term births (n=20) to identify candidate genes involved in extracellular matrix synthesis that may be associated with preterm labor. The subsequent candidate gene analyses conducted on 188 PPROM cases and 175 controls identified several risk variants in fibrillar collagen genes associated with PPROM [141]. Whilst the small sample size of the discovery phase is a limitation, the study presents an alternate approach to identify rare variants associated with PTB. Similar case-control studies have been published implicating rare variants in genes involved in the KEGG complement and coagulation cascade [86], as well as innate and host defense pathways [142]. Huusko et al. in 2018 conducted WES on families with predispositions to sPTB. Discovery analyses including samples from 17 Finnish women identified rare variants that significantly affected the glucocorticoid receptor signaling pathway. These findings were replicated in a validation cohort of 93 Danish sister pairs and two triads [143].
The small number of published WES/WGS studies using PTB samples has provided promising results, although most have been limited in sample size. As the cost of sequencing continues to decrease, the ability to conduct sequencing analyses using larger sample sizes may increase, potentially enabling the identification of multiple variants associated with this disease. Furthermore, sequencing data may allow better identification of functional variants rather than index variants that are only in linkage disequilibrium with the functional variants. The capability to detect rare and potentially functional variants in WES and WGS analyses may also enable future studies to identify polymorphisms with relatively large effect sizes that are associated with PTB.
Copy Number Variant Analyses
CNVs involving genes that are sensitive to changes in copy numbers have the potential to cause genetic disorders, as previously established in diseases such as neuropsychiatric and neurodegenerative disorders, autism, and immune deficiency [144,145,146,147]. However, few studies have analyzed CNVs associated with PTB, with fewer obtaining significant results.
A 2017 study conducted WES and assessed transcript levels in 160 mothers with PTB (<35 weeks’ gestation) and identified several ubiquitin-proteasome-collagen (CUP) pathway-related mRNAs that were differentially expressed in cases. Whilst the study then identified four CNVs within CUP-related genes within sPTB samples, the CNV results were inconclusive, likely due to sample size constraints [148]. Uzun et al. (2016) conducted a genome-wide approach to analyze CNVs in 454 women who delivered preterm (<34 weeks’ gestation) and 1018 women who delivered at term. However, the study found no evidence of an increased burden of CNVs in preterm cases compared to those born at term [149]. In contrast, Zheng et al. (2013) analyzed 898 PTB cases and 978 term deliveries and identified CNVs associated with a significantly increased risk of PTB (<35 weeks’ gestation). The CNVs were located within glutathione s-transferase theta 2 (GSTT2a) and glutathione s-transferase theta 2b (GSTT2b) genes [150].
CNV analyses represent a small proportion of published PTB related genetic studies and further investigations should be conducted to establish the role of CNVs in PTB risk. Improvements in sequencing methods have enhanced precise detection of CNVs. However, clinical interpretation and determining the functional effects of CNVs poses an ongoing challenge for future studies [151].
De Novo Mutation Analyses
There have been a limited number of studies that have investigated the role of fetal de novo mutations on PTB risk. A study published in 2017 conducted WGS analyses to assess fetal de novo mutations from 816 trio families. Data suggested that preterm infants (<37 weeks’ gestation) had a higher prevalence of de novo mutations than those born at term (P=6.9×10−03). The mutations were predominantly located in genes essential to embryonic development, especially fetal brain development [152]. A recent study conducted genome-wide genotyping and CNV calling on 488 infant cases and 3208 parent controls and identified 14 de novo CNV significantly associated with PTB (<32 weeks’ gestation). These CNV had a mutation rate of >2.9% compared to 2.1% in the control cohort (P=0.002) indicating that the mutations were significantly more common in those that experienced preterm delivery [153]. Both studies were underpowered and did not stratify analyses based on PTB sub-phenotypes. Nevertheless, they highlight new methods of extending PTB genetic risk and future analyses should consider examining rare fetal de novo mutations in larger, carefully phenotyped cohorts.
Preterm Birth Transcriptomics
PTB-related transcriptomic studies have conducted analyses on a wide variety of tissues including the placenta, basal plate, myometrium, decidua, fetal membranes (chorion and amnion), cervix, umbilical cord, fetal blood, and maternal blood, as identified in a systematic review published in 2015 [154].
A study recently published by Gupta et al. (2022) performed genetic association and differential gene expression analyses using blood samples from pregnant women at 16- and 20-weeks gestation; the results from the GWAS are discussed in the PTB genetics section above. The study included 114 RNA samples for analyses and identified 147 differentially expression genes associated with PPROM in the samples taken at 20-week gestation, implicating genes involved in local inflammatory responses [130]. Studies have also identified associations between PTB and differentially expressed genes involved in both anti- and pro-inflammatory cytokine pathways [77, 155,156,157,158], the complement pathway [159, 160], and white blood cell function [161, 162]. An upregulation of cyclooxygenase 2 gene (COX2) has been identified in those with PTB [163, 164]; this results in increased prostaglandin production including prostaglandin F2α and prostaglandin E2, which have both been shown to induce cervical ripening and myometrial contractions [165]. Whilst the data supports the role of immune function and tissue remodeling in PTB, most studies have sampled women during preterm labor. Therefore, the data cannot be used to identify women earlier in pregnancy that are at risk of PTB.
Maternal peripheral blood sampled during pregnancy has been frequently utilized to conduct transcriptome profiling and develop risk prediction models. Zhou et al. in 2020 conducted a nested case-control study of 51 sPTB cases and 106 term births and utilized four EBF1-based microRNA (miRNA) in maternal blood sampled in the third-trimester to create a risk prediction score for sPTB with an AUROC of 0.82, sensitivity of 81%, and specificity of 72% [166]. EBF1 gene expression and its association with PTB were further examined via transcript analyses that identified two EBF1-correlated lncRNA transcripts that were differentially expressed in sPTB [167]. A study published in 2017 utilized 30 miRNA from first-trimester peripheral blood mononuclear cells in a cohort of 39 pregnant women to create a risk score for developing sPTB. The study developed risk scores for early sPTB (<34 weeks’ gestation) and late sPTB (34 to 38 weeks’ gestation); the early sPTB risk score conferred an AUROC of 0.98 (P<0.0001) and AUROC for the late sPTB group of 0.92 (P<0.0001) [168]. Ngo et al. (2019) performed a similar study with 38 participants and highlighted seven RNA transcripts in maternal blood associated with PTB risk with an AUROC of 0.86 [169]. Studies have also developed similar risk scores using biological samples from multiple time points during pregnancy [170, 171] . These proof-of-concept studies provide promising results; however, the scores need to be validated in larger independent cohorts to confirm the predictive performance.
The performance of published transcriptomic predictive models using maternal blood samples is promising. Other studies have also successfully created predictive models utilizing RNA transcripts within tissues such as placenta, myometrium, and fetal membranes [172,173,174]. However, most studies have been limited by relatively small sample sizes, potentially resulting in inflated effect size estimations or false discovery rates. Nevertheless, there is a role in utilizing transcriptomic profiling in predicting PTB, and it is likely that risk prediction models may improve through the incorporation of clinical data.
Preterm Birth Epigenetics
A limited number of epigenetic studies have been conducted utilizing maternal samples. In a cohort of 300 African American women, DNA methylation changes in maternal blood were significantly associated with early sPTB within genes involved in the immune response and cell differentiation processes [175]. Ross et al. (2020) examined the relationship between advanced biological age (measured using epigenetic aging indices) and gestational duration. The study utilized whole blood samples from 77 women and found that higher levels of the epigenetic indices in pregnancy were associated with shorter gestational length [176].
The epigenetic changes associated with PTB development have also been examined utilizing newborn blood samples. Merid et al. (2020) conducted epigenome-wide meta-analyses utilizing cord blood from 3648 newborns in 17 cohorts. The study identified 8999 CpG sites, annotated to 4966 genes, associated with gestational age. The annotated genes were broadly involved in processes critical for embryonic development [177]. A recent study conducted epigenome-wide analyses in two independent cohorts (combined n=502) to assess DNA methylation in cord blood that was significantly associated with gestational age of birth. Differentially methylated regions were identified and mapped to genes involved in the inflammatory and innate immune systems [178]. Other studies have also identified differential methylation regions and CpG sites in genes involved in the inflammatory and immune systems [179, 180], as well as embryonic development, metabolic processes, and parturition [181,182,183,184].
Whilst the infant studies have identified several epigenetic modifications that may be associated with PTB, there is a significant lack of epigenetic studies utilizing maternal samples. Of those published, the findings are somewhat inconclusive and provide little use in clinical settings. Studies of larger sample size may be more successful in identifying epigenetic biomarkers associated with PTB.
Multiple “-Omics” Analyses
The majority of PTB-related “-omics” studies have analyzed a single set of “-omics” data such as genomics, transcriptomics, and epigenomics, as discussed in this review. Other “-omics” studies have also been performed in PTB-related studies but have not been reviewed in this paper. Systematic reviews of PTB biomarkers using multiplex, proteomic, metabolomic, and microbiomic approaches have been published [51, 185,186,187]. Common limitations across the single “-omics” studies are the lack of replication and that currently the findings have not been transferrable to clinical settings. Recently, some studies have integrated multiple “-omics” datasets to analyze the complex phenotype of PTB.
Huusko et al. in 2021 analyzed large genetic and transcriptomic datasets from mothers, infants, and placentas to examine the role of heat shock proteins and nuclear hormone receptors in sPTB. The study investigated 146 genes related to these proteins and receptors due to their potential involvement in stress responses such as immune activation. Several genes were associated with sPTB at a genome-wide significance. Transcriptomic results identified that the expression of these genes change in PTB and suggested that the activation of the specific heat shock proteins may be implicated with increased risk of prematurity [139].
A 2019 study utilized whole blood samples from 270 PTB case and 521 controls to integrate findings from WGS, RNA sequencing, and DNA methylation data [188]. The data were integrated and analyzed, and the results identified 160 significant genomic variants associated with various PTB-related phenotypes at a genome-wide significance. The identified genes were involved in biological pathways related to inflammation and immune function, and chemokine, interferon gamma, notch-1, endothelial growth factor receptor, and prolactin signaling [188].
Jehan et al. in 2020 conducted transcriptomic and proteomic profiling in plasma and metabolic profiling from urine samples in 81 pregnant women. Transcriptomic, proteomic, and metabolomic analyses predicted PTB with similar predictive performance (AUROC = 0.73, 0.75, and 0.59, respectively) [189]. However, an integrated model of all three datasets improved the results with an AUROC of 0.83. This potentially suggests that PTB is influenced by processes and biological systems at several levels that probably operate somewhat independently, and that combining datasets increases risk prediction performance [189]. However, Tarca et al. (2021) developed similar predictive models using transcriptomic and proteomic sampling and found that combining the separate predictive models did not change predictive performance [190].
The current body of multiple “-omics” research in the field of PTB is limited and the data from published studies were often not replicated in independent cohorts. This is potentially due to the computational challenges of conducting integrated analyses as well as the logistical concerns in obtaining data from multiple biological samples of the same pregnancies. Heterogeneity in the definition of the phenotype, biological samples assayed and time of collection, assay approaches and instrumentation, and analytical strategies can complicate integrated data analysis when studies do not follow a standard criterion or follow specific guidelines. Continuing advances in artificial intelligence coupled with well-planned study designs, model development, appropriate feature selection and extraction, and increasing sample size certainly has the potential to allow the development of improved predictive models for PTB. However, limitations exist with the lack of a suitable study cohort or a suitable database, and the ability to afford the computational demands needed to perform the analyses [191]. Overall, the results from the multiple “-omics” studies are promising and show potential to overcome current challenges in identifying clinical tools to predict PTB provided the limitations of these analyses are addressed in future studies.
The Future of PTB Genetics Research
The existing body of research supports a possible role of inflammatory, immunological, metabolic, endocrine, tissue remodeling, vascular, and endothelial pathways in PTB, providing some insight into the pathophysiological processes underpinning this disease. Transcriptomic and epigenetic studies have been moderately successful in identifying PTB biomarkers of interest. Large GWAS have identified several replicated polymorphisms associated with PTB, and the biological significance of these findings via laboratory tissue analyses should be further investigated. However, the clinical translational value of published findings so far has been minimal. Most published studies have been limited in power and have often lacked replication in independent cohorts. Multiple studies have not adjusted for confounders or multiple testing, decreasing the validity of results. Heterogenous ethnic groups were also commonly used despite knowledge that ethnicity influences PTB risk. Moreover, studies that utilized mixed ethnic groups often did not adjust appropriately. Numerous studies also grouped PTB phenotypes into one large cohort, rather than disaggregate based on sub-phenotypes. Given the complexity of PTB, this may have heterogenized study cohorts, leading to decreased ability to detect true associations.
Current approaches are yet to work due to phenotyping, ethnic, and analytic issues, as well as limitations in sample size. Combining study cohorts, likely through the use of public databases or international collaborations, may mitigate some limitations with sample size requirements. However, this poses a feasibility challenge as currently, aside from GWAS, there is minimal use of catalogues and public databases. Many studies, particularly candidate gene studies, have also not provided the necessary information to utilize their results, such as direction of effect and risk alleles. Regardless of the feasibility challenges, the research community should consider sample size and phenotyping requirements when designing future studies. Our review has also highlighted the promising performance of multiple “-omics” analyses in creating effective prediction tools for PTB. It is likely that integrating multiple “-omics” datasets is a suitable approach to overcome the limitations in analyzing the complex disease of PTB. Integrating genetic data with clinical data, such as ultrasound findings and other clinical biomarkers, and environmental risk factors, may also yield tools with higher predictive performance given the multifactorial nature of PTB. Overall, future research needs to be conducted using large cohorts that are carefully phenotyped and should consider integrating multiple biological datasets.
Conclusion
PTB is a global obstetric concern, affecting almost 15 million babies globally every year. The development of PTB is multifactorial with influence from both environmental and genetic factors. There is a large body of research exploring PTB genetics, with a particular focus on identifying associated genetic polymorphisms. A smaller subset of studies has investigated transcriptomic and epigenetic involvement in PTB. This review has summarized the body of literature regarding the role of genetics in PTB and has highlighted the need for conducting future research with large sample sizes, detailed phenotyping, and integrated multi “-omics” analyses with clinical biomarkers. Adopting these approaches may enable the successful development of prediction tools for PTB. This is critical to ensure that clinicians can determine individualized PTB risk and stratify clinical care to prevent PTB.
References
Liu L, Oza S, Hogan D, Chu Y, Perin J, Zhu J, et al. Global, regional, and national causes of under-5 mortality in 2000–15: an updated systematic analysis with implications for the Sustainable Development Goals. Lancet. 2016;388(10063):3027–35. https://doi.org/10.1016/s0140-6736(16)31593-8.
Perin J, Mulick A, Yeung D, Villavicencio F, Lopez G, Strong KL, et al. Global, regional, and national causes of under-5 mortality in 2000–19: an updated systematic analysis with implications for the Sustainable Development Goals. Lancet Child Adolesc Health. 2022;6(2):106–15. https://doi.org/10.1016/s2352-4642(21)00311-4.
Conde-Agudelo A, Romero R, Da Fonseca E, O’Brien JM, Cetingoz E, Creasy GW, et al. Vaginal progesterone is as effective as cervical cerclage to prevent preterm birth in women with a singleton gestation, previous spontaneous preterm birth, and a short cervix: updated indirect comparison meta-analysis. Am J Obstet Gynecol. 2018;219(1):10–25. https://doi.org/10.1016/j.ajog.2018.03.028.
Dawes L, Groom K, Jordan V, Waugh J. The use of specialised preterm birth clinics for women at high risk of spontaneous preterm birth: a systematic review. BMC Pregnancy Childbirth. 2020;20(1) https://doi.org/10.1186/s12884-020-2731-7.
Medley N, Vogel JP, Care A, Alfirevic Z. Interventions during pregnancy to prevent preterm birth: an overview of Cochrane systematic reviews. Cochrane Database Syst Rev. 2018. https://doi.org/10.1002/14651858.cd012505.pub2.
Mol BW, Jacobsson B, Grobman WA, Moley K, Jacobsson B, Simpson JL, et al. FIGO good practice recommendations on reduction of preterm birth in pregnancies conceived by assisted reproductive technologies. Int J Gynaecol Obstet. 2021;155(1):13–5. https://doi.org/10.1002/ijgo.13834.
Shennan A, Story L, Jacobsson B, Grobman WA, Simpson JL, Norman J, et al. FIGO good practice recommendations on cervical cerclage for prevention of preterm birth. Int J Gynaecol Obstet. 2021;155(1):19–22. https://doi.org/10.1002/ijgo.13835.
Shennan A, Suff N, Leigh Simpson J, Jacobsson B, Mol BW, Grobman WA, et al. FIGO good practice recommendations on progestogens for prevention of preterm delivery. Int J Gynaecol Obstet. 2021;155(1):16–8. https://doi.org/10.1002/ijgo.13852.
Valencia CM, Mol BW, Jacobsson B, Simpson JL, Norman J, Grobman W, et al. FIGO good practice recommendations on modifiable causes of iatrogenic preterm birth. Int J Gynaecol Obstet. 2021;155(1):8–12. https://doi.org/10.1002/ijgo.13857.
Beck S, Wojdyla D, Say L, Pilar Bertran A, Meraldi M, Harris Requejo J, et al. The worldwide incidence of preterm birth: a systematic review of maternal mortality and morbidity. Bull World Health Organ. 2010;88(1):31–8. https://doi.org/10.2471/blt.08.062554.
Chawanpaiboon S, Vogel JP, Moller A-B, Lumbiganon P, Petzold M, Hogan D, et al. Global, regional, and national estimates of levels of preterm birth in 2014: a systematic review and modelling analysis. Lancet Global Health. 2019;7(1):e37–46. https://doi.org/10.1016/s2214-109x(18)30451-0.
Saigal S, Doyle LW. An overview of mortality and sequelae of preterm birth from infancy to adulthood. Lancet. 2008;371(9608):261–9. https://doi.org/10.1016/s0140-6736(08)60136-1.
Platt MJ. Outcomes in preterm infants. Public Health. 2014;128(5):399–403. https://doi.org/10.1016/j.puhe.2014.03.010.
Crump C. Preterm birth and mortality in adulthood: a systematic review. J Perinatol. 2020;40(6):833–43. https://doi.org/10.1038/s41372-019-0563-y.
Crump C, Sundquist J, Winkleby MA, Sundquist K. Gestational age at birth and mortality from infancy into mid-adulthood: a national cohort study. Lancet Child Adolesc Health. 2019;3(6):408–17. https://doi.org/10.1016/s2352-4642(19)30108-7.
Risnes K, Bilsteen JF, Brown P, Pulakka A, Andersen A-MN, Opdahl S, et al. Mortality among young adults born preterm and early term in 4 Nordic nations. JAMA Network Open. 2021;4(1):e2032779. https://doi.org/10.1001/jamanetworkopen.2020.32779.
Crump C, Groves A, Sundquist J, Sundquist K. Association of preterm birth with long-term risk of heart failure into adulthood. JAMA Pediatr. 2021. https://doi.org/10.1001/jamapediatrics.2021.0131.
Raju TNK, Pemberton VL, Saigal S, Blaisdell CJ, Moxey-Mims M, Buist S. Long-term healthcare outcomes of preterm birth: an executive summary of a conference sponsored by the National Institutes of Health. J Pediatr. 2017;181:309–18.e1. https://doi.org/10.1016/j.jpeds.2016.10.015.
Porter TF, Fraser AM, Hunter CY, Ward RH, Varner MW. The risk of preterm birth across generations. Obstetr Gynecol. 1997;90(1):63–7. https://doi.org/10.1016/S0029-7844(97)00215-9.
Winkvist A. Familial patterns in birth characteristics: impact on individual and population risks. Int J Epidemiol. 1998;27(2):248–54. https://doi.org/10.1093/ije/27.2.248.
Lander ES, Linton LM, Birren B, Nusbaum C, Zody MC, Baldwin J, et al. Initial sequencing and analysis of the human genome. Nature. 2001;409(6822):860–921. https://doi.org/10.1038/35057062.
Crider KS, Whitehead N, Buus RM. Genetic variation associated with preterm birth: a HuGE review. Genet Med. 2005;7(9):593–604. https://doi.org/10.1097/01.gim.0000187223.69947.db.
Goldenberg RL, Culhane JF, Iams JD, Romero R. Epidemiology and causes of preterm birth. Lancet. 2008;371(9606):75–84. https://doi.org/10.1016/s0140-6736(08)60074-4.
Goldenberg RL, Hauth JC, Andrews WW. Intrauterine infection and preterm delivery. New Engl J Med. 2000;342(20):1500–7. https://doi.org/10.1056/nejm200005183422007.
Jackson RA, Gibson KA, Wu YW, Croughan MS. Perinatal outcomes in singletons following in vitro fertilization: a meta-analysis. Obstetr Gynecol. 2004;103(3)
Menon R. Spontaneous preterm birth, a clinical dilemma: etiologic, pathophysiologic and genetic heterogeneities and racial disparity. Acta Obstet Gynecol Scand. 2008;87(6):590–600. https://doi.org/10.1080/00016340802005126.
Romero R, Gomez R, Chaiworapongsa T, Conoscenti G, Cheol Kim J, Mee KY. The role of infection in preterm labour and delivery. Paediatr Perinat Epidemiol. 2001;15(s2):41–56. https://doi.org/10.1046/j.1365-3016.2001.00007.x.
Steer P. The epidemiology of preterm labour. BJOG. 2005;112:1–3. https://doi.org/10.1111/j.1471-0528.2005.00575.x.
Cobo T, Kacerovsky M, Jacobsson B. Risk factors for spontaneous preterm delivery. Int J Gynaecol Obstet. 2020;150(1):17–23. https://doi.org/10.1002/ijgo.13184.
Romero R, Dey SK, Fisher SJ. Preterm labor: one syndrome, many causes. Science. 2014;345(6198):760–5. https://doi.org/10.1126/science.1251816.
Morken N-H, Källen K, Hagberg H, Jacobsson B. Preterm birth in Sweden 1973-2001: rate, subgroups, and effect of changing patterns in multiple births, maternal age, and smoking. Acta Obstet Gynecol Scand. 2005;84(6):558–65. https://doi.org/10.1111/j.0001-6349.2005.00765.x.
Howson CP, Kinney MV, Lawn JE (eds) Born Too Soon: The Global Action Report on Preterm Birth. 2012;Geneva: World Health Organization
Savitz DA, Dole N, Herring AH, Kaczor D, Murphy J, Siega-Riz AM, et al. Should spontaneous and medically indicated preterm births be separated for studying aetiology? Paediat Perinat Epidemiol. 2005;19(2):97–105. https://doi.org/10.1111/j.1365-3016.2005.00637.x.
Pennell CE, Jacobsson B, Williams SM, Buus RM, Muglia LJ, Dolan SM, et al. Genetic epidemiologic studies of preterm birth: guidelines for research. Am J Obstetr Gynecol. 2007;196(2):107–18. https://doi.org/10.1016/j.ajog.2006.03.109.
Kistka ZAF, Palomar L, Lee KA, Boslaugh SE, Wangler MF, Cole FS, et al. Racial disparity in the frequency of recurrence of preterm birth. Am J Obstet Gynecol. 2007;196(2):131-e1. https://doi.org/10.1016/j.ajog.2006.06.093.
Svensson AC, Sandin S, Cnattingius S, Reilly M, Pawitan Y, Hultman CM, et al. Maternal effects for preterm birth: a genetic epidemiologic study of 630,000 families. Am J Epidemiol. 2009;170(11):1365–72. https://doi.org/10.1093/aje/kwp328.
Treloar SA, Macones GA, Mitchell LE, Martin NG. Genetic influences on premature parturition in an Australian twin sample. Twin Res. 2000;3(2):80–2.
Clausson B, Lichtenstein P, Cnattingius S. Genetic influence on birthweight and gestational length determined by studies in offspring of twins. BJOG. 2000;107(3):375–81. https://doi.org/10.1111/j.1471-0528.2000.tb13234.x.
York TP, Eaves LJ, Lichtenstein P, Neale MC, Svensson A, Latendresse S, et al. Fetal and maternal genes’ influence on gestational age in a quantitative genetic analysis of 244,000 Swedish births. Am J Epidemiol. 2013;178(4):543–50. https://doi.org/10.1093/aje/kwt005.
Schaaf JM, Liem SM, Mol BW, Abu-Hanna A, Ravelli AC. Ethnic and racial disparities in the risk of preterm birth: a systematic review and meta-analysis. Am J Perinatol. 2013;30(6):433–50. https://doi.org/10.1055/s-0032-1326988.
Bryant AS, Worjoloh A, Caughey AB, Washington AE. Racial/ethnic disparities in obstetric outcomes and care: prevalence and determinants. Am J Obstet Gynecol. 2010;202(4):335–43. https://doi.org/10.1016/j.ajog.2009.10.864.
Kildea S, Gao Y, Hickey S, Kruske S, Nelson C, Blackman R, et al. Reducing preterm birth amongst Aboriginal and Torres Strait Islander babies: a prospective cohort study, Brisbane, Australia. EClinicalMedicine. 2019;12:43–51. https://doi.org/10.1016/j.eclinm.2019.06.001.
Kramer MS, Goulet L, Lydon J, Seguin L, McNamara H, Dassa C, et al. Socio-economic disparities in preterm birth: causal pathways and mechanisms. Paediatr Perinat Epidemiol. 2001;15(s2):104–23. https://doi.org/10.1046/j.1365-3016.2001.00012.x.
Burris HH, Wright CJ, Kirpalani H, Collins JW Jr, Lorch SA, Elovitz MA, et al. The promise and pitfalls of precision medicine to resolve black–white racial disparities in preterm birth. Pediatr Res. 2020;87(2):221–6. https://doi.org/10.1038/s41390-019-0528-z.
Song JW, Chung KC. Observational studies: cohort and case-control studies. Plast Reconst Surg. 2010;126(6):2234–42. https://doi.org/10.1097/prs.0b013e3181f44abc.
Wijsman EM. Family-based approaches: design, imputation, analysis, and beyond. BMC Genet. 2016;17(S2) https://doi.org/10.1186/s12863-015-0318-5.
Bezold KY, Karjalainen MK, Hallman M, Teramo K, Muglia LJ. The genomics of preterm birth: from animal models to human studies. Genome Med. 2013;5(4):34. https://doi.org/10.1186/gm438.
Wadon M, Modi N, Wong HS, Thapar A, O’Donovan MC. Recent advances in the genetics of preterm birth. Ann Hum Genet. 2020;84(3):205–13. https://doi.org/10.1111/ahg.12373.
Brummaier T, Kabeer BSA, Chaussabel D, Utzinger J, McGready R, Paris DH. Blood gene transcript signature profiling in pregnancies resulting in preterm birth: a systematic review. Eur J Obstet Gynecol Reprod Biol. 2020;8:100118. https://doi.org/10.1016/j.eurox.2020.100118.
Law KP, Han TL, Tong C, Baker PN. Mass spectrometry-based proteomics for pre-eclampsia and preterm birth. Int J Mol Sci. 2015;16(5):10952–85. https://doi.org/10.3390/ijms160510952.
Carter RA, Pan K, Harville EW, McRitchie S, Sumner S. Metabolomics to reveal biomarkers and pathways of preterm birth: a systematic review and epidemiologic perspective. Metabolomics. 2019;15(9):124. https://doi.org/10.1007/s11306-019-1587-1.
Gracie S, Pennell C, Ekman-Ordeberg G, Lye S, McManaman J, Williams S, et al. An integrated systems biology approach to the study of preterm birth using “-omic” technology - a guideline for research. BMC Pregnancy Childbirth. 2011;11(1):71. https://doi.org/10.1186/1471-2393-11-71.
Giarratano G. Genetic influences on preterm birth. MCN The American Journal of Maternal Child. Nursing. 2006;31(3):169–75. https://doi.org/10.1097/00005721-200605000-00008.
Sherry ST, Ward MH, Kholodov M, Baker J, Phan L, Smigielski EM, et al. dbSNP: the NCBI database of genetic variation. Nucleic Acids Res. 2001;29(1):308–11. https://doi.org/10.1093/nar/29.1.308.
Redon R, Ishikawa S, Fitch KR, Feuk L, Perry GH, Andrews TD, et al. Global variation in copy number in the human genome. Nature. 2006;444(7118):444–54. https://doi.org/10.1038/nature05329.
Feuk L, Carson AR, Scherer SW. Structural variation in the human genome. Nat Rev Genet. 2006;7(2):85–97. https://doi.org/10.1038/nrg1767.
Veltman JA, Brunner HG. De novo mutations in human genetic disease. Nat Rev Genet. 2012;13(8):565–75. https://doi.org/10.1038/nrg3241.
Tabor HK, Risch NJ, Myers RM. Candidate-gene approaches for studying complex genetic traits: practical considerations. Nat Rev Genet. 2002;3(5):391–7. https://doi.org/10.1038/nrg796.
Hardy J, Singleton A. Genomewide association studies and human disease. New Engl J Med. 2009;360(17):1759–68. https://doi.org/10.1056/nejmra0808700.
Milward EA, Shahandeh A, Heidari M, Johnstone DM, Daneshi N, Hondermarck H. Transcriptomics. In: Bradshaw RA, Stahl PD, editors. Encycl Cell Biol. Waltham: Academic Press; 2016. p. 160–5.
Gil N, Ulitsky I. Regulation of gene expression by cis-acting long non-coding RNAs. Nat Rev Genet. 2020;21(2):102–17. https://doi.org/10.1038/s41576-019-0184-5.
Mitsuya K, Singh N, Sooranna SR, Johnson MR, Myatt L. Epigenetics of human myometrium: DNA methylation of genes encoding contraction-associated proteins in term and preterm labor1. Biol Reprod. 2014;90(5):98. https://doi.org/10.1095/biolreprod.113.113209.
Medzhitov R. Inflammation 2010: new adventures of an old flame. Cell. 2010;140(6):771–6. https://doi.org/10.1016/j.cell.2010.03.006.
Christian LM, Porter K. Longitudinal changes in serum proinflammatory markers across pregnancy and postpartum: effects of maternal body mass index. Cytokine. 2014;70(2):134–40. https://doi.org/10.1016/j.cyto.2014.06.018.
Gillespie SL, Porter K, Christian LM. Adaptation of the inflammatory immune response across pregnancy and postpartum in Black and White women. J Reprod Immunol. 2016;114:27–31. https://doi.org/10.1016/j.jri.2016.02.001.
Di Renzo GC, Tosto V, Giardina I. The biological basis and prevention of preterm birth. Best Pract Res: Clin Obstet Gynaecol. 2018;52:13–22. https://doi.org/10.1016/j.bpobgyn.2018.01.022.
Ramos BRDA, Mendes ND, Tanikawa AA, Amador MAT, dos Santos NPC, dos Santos SEB, et al. Ancestry informative markers and selected single nucleotide polymorphisms in immunoregulatory genes on preterm labor and preterm premature rupture of membranes: a case control study. BMC Pregnancy Childbirth. 2016;16(1) https://doi.org/10.1186/s12884-016-0823-1.
Romero R, Velez Edwards DR, Kusanovic JP, Hassan SS, Mazaki-Tovi S, Vaisbuch E, et al. Identification of fetal and maternal single nucleotide polymorphisms in candidate genes that predispose to spontaneous preterm labor with intact membranes. Am J Obstet Gynecol. 2010;202(5):431.e1. https://doi.org/10.1016/j.ajog.2010.03.026.
Velez DR, Fortunato SJ, Thorsen P, Lombardi SJ, Williams SM, Menon R. Preterm birth in Caucasians is associated with coagulation and inflammation pathway gene variants. PLoS ONE. 2008;3(9):e3283. https://doi.org/10.1371/journal.pone.0003283.
Bitner A, Sobala W, Kalinka J. Association between maternal and fetal TLR4 (896A>G, 1196C>T) gene polymorphisms and the risk of pre-term birth in the Polish population. Am J Reprod Immunol. 2013;69(3):272–80. https://doi.org/10.1111/aji.12068.
Harmon QE, Engel SM, Olshan AF, Moran T, Stuebe AM, Luo J, et al. Association of polymorphisms in natural killer cell-related genes with preterm birth. Am J Epidemiol. 2013;178(8):1208–18. https://doi.org/10.1093/aje/kwt108.
Karakaş NM, Ecevit AN, Yalçın Y, Özdemir B, Verdi H, Tekindal MA, et al. Effect of maternal and neonatal interleukin-6 − 174 G/C polymorphism on preterm birth and neonatal morbidity. J Matern-Fetal Neonatal Med. 2018;31(8):1009–15. https://doi.org/10.1080/14767058.2017.1304911.
Velez DR, Fortunato S, Thorsen P, Lombardi SJ, Williams SM, Menon R. Spontaneous preterm birth in African Americans is associated with infection and inflammatory response gene variants. Am J Obstet Gynecol. 2009;200(2):209.e1. https://doi.org/10.1016/j.ajog.2008.08.051.
Gómez LM, Sammel MD, Appleby DH, Elovitz MA, Baldwin DA, Jeffcoat MK, et al. Evidence of a gene-environment interaction that predisposes to spontaneous preterm birth: a role for asymptomatic bacterial vaginosis and DNA variants in genes that control the inflammatory response. Am J Obstet Gynecol. 2010;202(4):386.e1–6. https://doi.org/10.1016/j.ajog.2010.01.042.
Frey HA, Stout MJ, Pearson LN, Tuuli MG, Cahill AG, Strauss JF, et al. Genetic variation associated with preterm birth in African-American women. Am J Obstet Gynecol. 2016;215(2):235.e1. https://doi.org/10.1016/j.ajog.2016.03.008.
Han SH, Lee NR, Kim HJ, Kang YD, Kim JS, Park JW, et al. Association between the IL-6, IL-10, and TNFα gene polymorphisms and preterm-birth in Korean women. Genes Genom. 2020;42(7):743–50. https://doi.org/10.1007/s13258-020-00946-4.
Pandey M, Awasthi S, Singh U, Mahdi AA. Association of IL-10 gene polymorphism (−819C > T, −592C > A and −1082G > A) with preterm birth. Indian J Pediatr. 2018;85(2):93–101. https://doi.org/10.1007/s12098-017-2496-9.
Gillespie SL, Neal JL, Christian LM, Szalacha LA, McCarthy DO, Salsberry PJ. Interleukin-1 receptor antagonist polymorphism and birth timing. Nurs Res. 2017;66(2):95–104. https://doi.org/10.1097/nnr.0000000000000200.
Fortunato SJ, Menon R, Velez DR, Thorsen P, Williams SM. Racial disparity in maternal-fetal genetic epistasis in spontaneous preterm birth. Am J Obstetr Gynecol. 2008;198(6):666.e1. https://doi.org/10.1016/j.ajog.2008.02.003.
Falah N, McElroy J, Snegovskikh V, Lockwood CJ, Norwitz E, Murray JC, et al. Investigation of genetic risk factors for chronic adult diseases for association with preterm birth. Hum Genet. 2013;132(1):57–67. https://doi.org/10.1007/s00439-012-1223-x.
Ryckman KK, Morken N-H, White MJ, Velez DR, Menon R, Fortunato SJ, et al. Maternal and fetal genetic associations of PTGER3 and PON1 with preterm birth. PLoS ONE. 2010;5(2):e9040. https://doi.org/10.1371/journal.pone.0009040.
As Sayaril N, Bhat RS, Warsy AS, Babay ZA, Addar MH, Shaik J, et al. Polymorphisms in proinflammatory cytokine genes, effect on gene expression and association with preterm delivery in Saudi females. Cell Mol Biol. 2018;64(10):55–60.
Drews-Piasecka E, Seremak-Mrozikiewicz A, Barlik M, Kurzawińska G, Wolski H, Woyciechowska A, et al. The significance of TNF-alpha gene polymorphisms in preterm delivery. Ginekol Polska. 2014;85(6):428–34. https://doi.org/10.17772/gp/1748.
Harper M, Zheng SL, Thom E, Klebanoff MA, Thorp J, Sorokin Y, et al. Cytokine gene polymorphisms and length of gestation. Obstet Gynecol. 2011;117(1):125–30. https://doi.org/10.1097/aog.0b013e318202b2ef.
Yılmaz Y, Verdi H, Taneri A, Yazıcı AC, Ecevit AN, Karakaş NM, et al. Maternal–fetal proinflammatory cytokine gene polymorphism and preterm birth. DNA Cell Biol. 2012;31(1):92–7. https://doi.org/10.1089/dna.2010.1169.
McElroy JJ, Gutman CE, Shaffer CM, Busch TD, Puttonen H, Teramo K, et al. Maternal coding variants in complement receptor 1 and spontaneous idiopathic preterm birth. Hum Genet. 2013;132(8):935–42. https://doi.org/10.1007/s00439-013-1304-5.
Romero R, Friel LA, Velez Edwards DR, Kusanovic JP, Hassan SS, Mazaki-Tovi S, et al. A genetic association study of maternal and fetal candidate genes that predispose to preterm prelabor rupture of membranes (PROM). Am J Obstet Gynecol. 2010;203(4):361.e1. https://doi.org/10.1016/j.ajog.2010.05.026.
Kwon H-S, Sohn I-S, Lee J-Y, Lee S-J, Kim S-N, Kim B-J. Intercellular adhesion molecule-1 K469E polymorphism in Korean patients with spontaneous preterm delivery. Int J Gynaecol Obstet. 2009;104(1):37–9. https://doi.org/10.1016/j.ijgo.2008.08.020.
Song J, Li J, Liu H, Gan Y, Sun Y, Yu M, et al. A genetic variant in the placenta-derived MHC class I chain-related gene A increases the risk of preterm birth in a Chinese population. Hum Genet. 2017;136(10):1375–84. https://doi.org/10.1007/s00439-017-1834-3.
Bream ENA, Leppellere CR, Cooper ME, Dagle JM, Merrill DC, Christensen K, et al. Candidate gene linkage approach to identify DNA variants that predispose to preterm birth. Pediatr Res. 2013;73(2):135–41. https://doi.org/10.1038/pr.2012.166.
Sugita N, Kobayashi T, Kikuchi A, Shimada Y, Hirano E, Sasahara J, et al. Immunoregulatory gene polymorphisms in Japanese women with preterm births and periodontitis. J Reprod Immunol. 2012;93(2):94–101. https://doi.org/10.1016/j.jri.2012.01.005.
Speer EM, Gentile DA, Zeevi A, Pillage G, Huo D, Skoner DP. Role of single nucleotide polymorphisms of cytokine genes in spontaneous preterm delivery. Hum Immunol. 2006;67(11):915–23. https://doi.org/10.1016/j.humimm.2006.08.291.
Malmström E, Sennström M, Holmberg A, Frielingsdorf H, Eklund E, Malmström L, et al. The importance of fibroblasts in remodelling of the human uterine cervix during pregnancy and parturition. Mol Hum Reprod. 2007;13(5):333–41. https://doi.org/10.1093/molehr/gal117.
Mann PC, Cooper ME, Ryckman KK, Comas B, Gili J, Crumley S, et al. Polymorphisms in the fetal progesterone receptor and a calcium-activated potassium channel isoform are associated with preterm birth in an Argentinian population. J Perinatol. 2013;33(5):336–40. https://doi.org/10.1038/jp.2012.118.
Gimenez LG, Momany AM, Poletta FA, Krupitzki HB, Gili JA, Busch TD, et al. Association of candidate gene polymorphisms with clinical subtypes of preterm birth in a Latin American population. Pediatr Res. 2017;82(3):554–9. https://doi.org/10.1038/pr.2017.109.
Pandey M, Awasthi S. Role of MMP-1, MMP-8 and MMP-9 gene polymorphisms in preterm birth. J Genet. 2020;99
Preda A, Caracostea G, Ona D, Zaharie G, Stamatian F. Association between maternal/newborn genetic variants, placental pathology and spontaneous preterm birth risk: a Romanian population-based study. J Matern-Fetal Neonatal Med. 2020;33(7):1171–7. https://doi.org/10.1080/14767058.2018.1517311.
Chun S, Plunkett J, Teramo K, Muglia LJ, Fay JC. Fine-mapping an association of FSHR with preterm birth in a Finnish population. PLoS ONE. 2013;8(10):e78032. https://doi.org/10.1371/journal.pone.0078032.
Manuck TA, Major HD, Varner MW, Chettier R, Nelson L, Esplin MS. Progesterone receptor genotype, family history, and spontaneous preterm birth. Obstet Gynecol. 2010;115(4)
Plunkett J, Doniger S, Orabona G, Morgan T, Haataja R, Hallman M, et al. An evolutionary genomic approach to identify genes involved in human birth timing. PLoS Genet. 2011;7(4):e1001365. https://doi.org/10.1371/journal.pgen.1001365.
Schmid M, Grimm C, Leipold H, Knöfler M, Haslinger P, Egarter C. A polymorphism of the corticotropin-releasing hormone receptor 2 (CRHR2) and preterm birth. Dis Markers. 2010;28(1):37–42. https://doi.org/10.3233/dma-2010-0681.
Vogel I, Hollegaard MV, Hougaard DM, Thorsen P, Grove J. Polymorphisms in the promoter region of relaxin-2 and preterm birth: involvement of relaxin in the etiology of preterm birth. In Vivo. 2009;23(6):1005–9.
Dominguez-Lopez P, Diaz-Cueto L, Arechavaleta-Velasco M, Caldiño-Soto F, Ulloa-Aguirre A, Arechavaleta-Velasco F. The follicle-stimulating hormone receptor Asn680Ser polymorphism is associated with preterm birth in Hispanic women. J Matern-Fetal Neonatal Med. 2018;31(5):580–5. https://doi.org/10.1080/14767058.2017.1292245.
Kadivnik M, Kralik K, Muller-Vranješ A, Vučemilović-Jurić V, Šijanović S, Wagner J. Progesterone receptor genetic variants in pregnant women and fetuses as possible predictors of spontaneous premature birth: a preliminary case–control study. J Obstetr Gynaecol Res. 2022;48(5):1099–109. https://doi.org/10.1111/jog.15194.
Kim J, Stirling KJ, Cooper ME, Ascoli M, Momany AM, McDonald EL, et al. Sequence variants in oxytocin pathway genes and preterm birth: a candidate gene association study. BMC Med Genet. 2013;14(1):77. https://doi.org/10.1186/1471-2350-14-77.
Liu G-J, He J-R, Kuang Y-S, Fan X-J, Li W-D, Lu J-H, et al. Associations of maternal PLA2G4C and PLA2G4D polymorphisms with the risk of spontaneous preterm birth in a Chinese population. Mol Med Rep. 2017;15(6):3607–14. https://doi.org/10.3892/mmr.2017.6475.
Salem H, Rosenfeld T, Altarescu G, Grisaru-Granovsky S, Birk R. Maternal and neonatal leptin and leptin receptor polymorphisms associated with preterm birth. Gene. 2016;591(1):209–13. https://doi.org/10.1016/j.gene.2016.07.014.
Haataja R, Karjalainen MK, Luukkonen A, Teramo K, Puttonen H, Ojaniemi M, et al. Mapping a new spontaneous preterm birth susceptibility gene, IGF1R, using linkage, haplotype sharing, and association analysis. PLoS Genet. 2011;7(2):e1001293. https://doi.org/10.1371/journal.pgen.1001293.
He J-R, Lai Y-M, Liu H-H, Liu G-J, Li W-D, Fan X-J, et al. Maternal IGF1 and IGF1R polymorphisms and the risk of spontaneous preterm birth. J Clin Lab Anal. 2017;31(6):e22125. https://doi.org/10.1002/jcla.22125.
Stelzer G, Rosen N, Plaschkes I, Zimmerman S, Twik M, Fishilevich S, et al. The GeneCards suite: from gene data mining to disease genome sequence analyses. Curr Protocols Bioinform. 2016;54(1) https://doi.org/10.1002/cpbi.5.
Hwang IW, Kang YD, Kwon BN, Hong JH, Han SH, Kim JS, et al. Genetic variations of MTHFR gene and their association with preterm birth in Korean women. Medicina. 2017;53(6):380–5. https://doi.org/10.1016/j.medici.2018.01.001.
Engel SM, Olshan AF, Siega-Riz AM, Savitz DA, Chanock SJ. Polymorphisms in folate metabolizing genes and risk for spontaneous preterm and small-for-gestational age birth. Am J Obstet Gynecol. 2006;195(5):1231.e1. https://doi.org/10.1016/j.ajog.2006.07.024.
Barchitta M, Maugeri A, La Rosa M, Magnano San Lio R, Favara G, Panella M, et al. Single nucleotide polymorphisms in vitamin D receptor gene affect birth weight and the risk of preterm birth: results from the “Mamma & Bambino” cohort and a meta-analysis. Nutrients. 2018;10(9):1172. https://doi.org/10.3390/nu10091172.
Wang S, Xin X, Luo W, Mo M, Si S, Shao B, et al. Association of vitamin D and gene variants in the vitamin D metabolic pathway with preterm birth. Nutrition. 2021;89:111349. https://doi.org/10.1016/j.nut.2021.111349.
Kwon BN, Lee NR, Kim HJ, Kang YD, Kim JS, Park JW, et al. Folate metabolizing gene polymorphisms and genetic vulnerability to preterm birth in Korean women. Genes Genom. 2021;43:937–45. https://doi.org/10.1007/s13258-021-01082-3.
Lee BE, Park H, Park EA, Gwak H, Ha EH, Pang M-G, et al. Paraoxonase 1 gene and glutathione S-transferase μ 1 gene interaction with preterm delivery in Korean women. Am J Obstet Gynecol. 2010;203(6):569.e1. https://doi.org/10.1016/j.ajog.2010.07.029.
Yu Y, Tsai H-J, Liu X, Mestan K, Zhang S, Pearson C, et al. The joint association between F5 gene polymorphisms and maternal smoking during pregnancy on preterm delivery. Hum Genet. 2009;124(6):659–68. https://doi.org/10.1007/s00439-008-0589-2.
Gibson CS, MacLennan AH, Dekker GA, Goldwater PN, Dambrosia JM, Munroe DJ, et al. Genetic polymorphisms and spontaneous preterm birth. Obstet Gynecol. 2007;109(2 Pt 1):384–91. https://doi.org/10.1097/01.AOG.0000252712.62241.1a.
Chen BH, Carmichael SL, Shaw GM, Iovannisci DM, Lammer EJ. Association between 49 infant gene polymorphisms and preterm delivery. Am J Med Genet Part A. 2007;143A(17):1990–6. https://doi.org/10.1002/ajmg.a.31868.
Olson DM, Ammann C. Role of the prostaglandins in labour and prostaglandin receptor inhibitors in the prevention of preterm labour. Front Biosci. 2007;12:1329–43. https://doi.org/10.2741/2151.
Romero R, Espinoza J, Kusanovic J, Gotsch F, Hassan S, Erez O, et al. The preterm parturition syndrome. BJOG. 2006;113:17–42. https://doi.org/10.1111/j.1471-0528.2006.01120.x.
Liu X, Wang G, Hong X, Tsai H-J, Liu R, Zhang S, et al. Associations between gene polymorphisms in fatty acid metabolism pathway and preterm delivery in a US urban black population. Hum Genet. 2012;131(3):341–51. https://doi.org/10.1007/s00439-011-1079-5.
Jeffcoat MK, Jeffcoat RL, Tanna N, Parry SH. Association of a common genetic factor, PTGER3, with outcome of periodontal therapy and preterm birth. J Periodontol. 2014;85(3):446–54. https://doi.org/10.1902/jop.2013.130006.
Andraweera PH, Dekker GA, Thompson SD, North RA, McCowan LME, Roberts CT. The interaction between the maternal BMI and angiogenic gene polymorphisms associates with the risk of spontaneous preterm birth. Mol Hum Reprod. 2012;18(9):459–65. https://doi.org/10.1093/molehr/gas016.
Langmia IM, Apalasamy YD, Omar SZ, Mohamed Z. Association of VEGFA gene polymorphisms and VEGFA plasma levels with spontaneous preterm birth. Pharmacogenet Genom. 2015;25(4):199–204. https://doi.org/10.1097/fpc.0000000000000125.
Silva LVCD, Javorski N, André Cavalcanti Brandão L, Lima MDC, Crovella S, Eickmann SH. Influence of MBL2 and NOS3 polymorphisms on spontaneous preterm birth in North East Brazil: genetics and preterm birth. J Matern-Fetal Neonatal Med. 2020;33(1):127–35. https://doi.org/10.1080/14767058.2018.1487938.
Solé-Navais P, Flatley C, Steinthorsdottir V, Vaudel M, Juodakis J, Chen J, et al. Genetic effects on the timing of parturition and links to fetal birth weight. Nat Genet. 2023;55(4):559–67. https://doi.org/10.1038/s41588-023-01343-9.
Panagiotou OA, Ioannidis JPA. What should the genome-wide significance threshold be? Empirical replication of borderline genetic associations. Int J Epidemiol. 2012;41(1):273–86. https://doi.org/10.1093/ije/dyr178.
Zhang G, Feenstra B, Bacelis J, Liu X, Muglia LM, Juodakis J, et al. Genetic associations with gestational duration and spontaneous preterm birth. New Engl J Med. 2017;377(12):1156–67. https://doi.org/10.1056/nejmoa1612665.
Gupta JK, Care A, Goodfellow L, Alfirevic Z, Müller-Myhsok B, Alfirevic A. Genome and transcriptome profiling of spontaneous preterm birth phenotypes. Sci Rep. 2022;12(1) https://doi.org/10.1038/s41598-022-04881-0.
Liu X, Helenius D, Skotte L, Beaumont RN, Wielscher M, Geller F, et al. Variants in the fetal genome near pro-inflammatory cytokine genes on 2q13 associate with gestational duration. Nature Communications. 2019;10(1) https://doi.org/10.1038/s41467-019-11881-8.
Rappoport N, Toung J, Hadley D, Wong RJ, Fujioka K, Reuter J, et al. A genome-wide association study identifies only two ancestry specific variants associated with spontaneous preterm birth. Sci Rep. 2018;8(1) https://doi.org/10.1038/s41598-017-18246-5.
Alleman BW, Myking S, Ryckman KK, Myhre R, Feingold E, Feenstra B, et al. No observed association for mitochondrial SNPs with preterm delivery and related outcomes. Pediatr Res. 2012;72(5):539–44. https://doi.org/10.1038/pr.2012.112.
Bacelis J, Juodakis J, Sengpiel V, Zhang G, Myhre R, Muglia LJ, et al. Literature-informed analysis of a genome-wide association study of gestational age in Norwegian women and children suggests involvement of inflammatory pathways. PLoS ONE. 2016;11(8):e0160335. https://doi.org/10.1371/journal.pone.0160335.
Li J, Hong X, Mesiano S, Muglia LJ, Wang X, Snyder M, et al. Natural selection has differentiated the progesterone receptor among human populations. Am J Hum Genet. 2018;103(1):45–57. https://doi.org/10.1016/j.ajhg.2018.05.009.
Myking S, Boyd HA, Myhre R, Feenstra B, Jugessur A, Devold Pay AS, et al. X-chromosomal maternal and fetal SNPs and the risk of spontaneous preterm delivery in a Danish/Norwegian genome-wide association study. PLoS ONE. 2013;8(4):e61781. https://doi.org/10.1371/journal.pone.0061781.
Tiensuu H, Haapalainen AM, Karjalainen MK, Pasanen A, Huusko JM, Marttila R, et al. Risk of spontaneous preterm birth and fetal growth associates with fetal SLIT2. PLoS Genet. 2019;15(6):e1008107. https://doi.org/10.1371/journal.pgen.1008107.
Zhang H, Baldwin DA, Bukowski RK, Parry S, Xu Y, Song C, et al. A genome-wide association study of early spontaneous preterm delivery. Genet Epidemiol. 2015;39(3):217–26. https://doi.org/10.1002/gepi.21887.
Huusko JM, Tiensuu H, Haapalainen AM, Pasanen A, Tissarinen P, Karjalainen MK, et al. Integrative genetic, genomic and transcriptomic analysis of heat shock protein and nuclear hormone receptor gene associations with spontaneous preterm birth. Sci Rep. 2021;11(1) https://doi.org/10.1038/s41598-021-96374-9.
Christensen K, Dukhovny D, Siebert U, Green R. Assessing the costs and cost-effectiveness of genomic sequencing. J Personal Med. 2015;5(4):470–86. https://doi.org/10.3390/jpm5040470.
Modi BP, Teves ME, Pearson LN, Parikh HI, Chaemsaithong P, Sheth NU, et al. Rare mutations and potentially damaging missense variants in genes encoding fibrillar collagens and proteins involved in their production are candidates for risk for preterm premature rupture of membranes. PLoS ONE. 2017;12(3):e0174356. https://doi.org/10.1371/journal.pone.0174356.
Modi BP, Teves ME, Pearson LN, Parikh HI, Haymond-Thornburg H, Tucker JL, et al. Mutations in fetal genes involved in innate immunity and host defense against microbes increase risk of preterm premature rupture of membranes (PPROM). Mol Genet Genom Med. 2017;5(6):720–9. https://doi.org/10.1002/mgg3.330.
Huusko JM, Karjalainen MK, Graham BE, Zhang G, Farrow EG, Miller NA, et al. Whole exome sequencing reveals HSPA1L as a genetic risk factor for spontaneous preterm birth. PLoS Genet. 2018;14(7):e1007394. https://doi.org/10.1371/journal.pgen.1007394.
Rovelet-Lecrux A, Hannequin D, Raux G, Meur NL, Laquerrière A, Vital A, et al. APP locus duplication causes autosomal dominant early-onset Alzheimer disease with cerebral amyloid angiopathy. Nat Genet. 2006;38(1):24–6. https://doi.org/10.1038/ng1718.
Cook EH Jr, Scherer SW. Copy-number variations associated with neuropsychiatric conditions. Nature. 2008;455(7215):919–23. https://doi.org/10.1038/nature07458.
Sebat J, Lakshmi B, Malhotra D, Troge J, Lese-Martin C, Walsh T, et al. Strong association of de novo copy number mutations with autism. Science. 2007;316(5823):445–9. https://doi.org/10.1126/science.1138659.
Gonzalez E, Kulkarni H, Bolivar H, Mangano A, Sanchez R, Catano G, et al. The influence of CCL3L1 gene-containing segmental duplications on HIV-1/AIDS susceptibility. Science. 2005;307(5714):1434–40. https://doi.org/10.1126/science.1101160.
Zhao X, Dong X, Luo X, Pan J, Ju W, Zhang M, et al. Ubiquitin-proteasome-collagen (CUP) pathway in preterm premature rupture of fetal membranes. Front Pharmacol. 2017:8. https://doi.org/10.3389/fphar.2017.00310.
Uzun A, Sahin Y, Schuster JS, Zheng X, Ryckman K, Feingold E, et al. Structural and genomic variation in preterm birth. Pediatr Res. 2016;80(6):829–36. https://doi.org/10.1038/pr.2016.152.
Zheng X, Feingold E, Ryckman KK, Shaffer JR, Boyd HA, Feenstra B, et al. Association of maternal CNVs in GSTT1/GSTT2 with smoking, preterm delivery, and low birth weight. Front Genet. 2013:4. https://doi.org/10.3389/fgene.2013.00196.
Pös O, Radvanszky J, Styk J, Pös Z, Buglyó G, Kajsik M, et al. Copy number variation: methods and clinical applications. Appl Sci. 2021;11(2):819.
Li J, Oehlert J, Snyder M, Stevenson DK, Shaw GM. Fetal de novo mutations and preterm birth. PLoS Genet. 2017;13(4):e1006689. https://doi.org/10.1371/journal.pgen.1006689.
Wong HS, Wadon M, Evans A, Kirov G, Modi N, O’Donovan MC, et al. Contribution of de novo and inherited rare CNVs to very preterm birth. J Med Genet. 2020;57(8):552–7. https://doi.org/10.1136/jmedgenet-2019-106619.
Eidem HR, Ackerman WE, McGary KL, Abbot P, Rokas A. Gestational tissue transcriptomics in term and preterm human pregnancies: a systematic review and meta-analysis. BMC Med Genom. 2015;8(1) https://doi.org/10.1186/s12920-015-0099-8.
Bukowski R, Sadovsky Y, Goodarzi H, Zhang H, Biggio JR, Varner M, et al. Onset of human preterm and term birth is related to unique inflammatory transcriptome profiles at the maternal fetal interface. PeerJ. 2017;5:e3685. https://doi.org/10.7717/peerj.3685.
Wang P, Pan J, Tian X, Dong X, Ju W, Wang Y, et al. Transcriptomics-determined chemokine-cytokine pathway presents a common pathogenic mechanism in pregnancy loss and spontaneous preterm birth. Am J Reprod Immunol. 2021;86(1) https://doi.org/10.1111/aji.13398.
Enquobahrie DA, Williams MA, Qiu C, Muhie SY, Slentz-Kesler K, Ge Z, et al. Early pregnancy peripheral blood gene expression and risk of preterm delivery: a nested case control study. BMC Pregnancy Childbirth. 2009;9(1):56. https://doi.org/10.1186/1471-2393-9-56.
Chim SSC, Lee WS, Ting YH, Chan OK, Lee SWY, Leung TY. Systematic identification of spontaneous preterm birth-associated RNA transcripts in maternal plasma. PLoS ONE. 2012;7(4):e34328. https://doi.org/10.1371/journal.pone.0034328.
Nowicki S, Izban MG, Pawelczyk E, Agboto VK, Pratap S, Olson G, et al. Preterm labor: CD55 in maternal blood leukocytes. Am J Reprod Immunol. 2009;61(5):360–7. https://doi.org/10.1111/j.1600-0897.2009.00702.x.
Pacheco LD, Hankins GD, Costantine MM, Anderson GD, Pawelczyk E, Nowicki S, et al. The role of human decay-accelerating factor in the pathogenesis of preterm labor. Am J Perinatol. 2011;28(7):565–70. https://doi.org/10.1055/s-0031-1274510.
Prearo Moço N, Camargo Batista RA, Fernandes Martin L, de Oliveira LG, de Lima G, Parada CM, Alarcão Dias-Melicio L, et al. Toll-like receptor-2 and -4 expression by maternal neutrophils in preterm labor. Gynecol Obstet Invest. 2018;83(1):1–8. https://doi.org/10.1159/000468930.
Pawelczyk E, Nowicki BJ, Izban MG, Pratap S, Sashti NA, Sanderson M, et al. Spontaneous preterm labor is associated with an increase in the proinflammatory signal transducer TLR4 receptor on maternal blood monocytes. BMC Pregnancy Childbirth. 2010;10(1):66. https://doi.org/10.1186/1471-2393-10-66.
Mustafa M, Garg N, Banerjee BD, Sharma T, Tyagi V, Dar SA, et al. Inflammatory-mediated pathway in association with organochlorine pesticides levels in the etiology of idiopathic preterm birth. Reprod Toxicol. 2015;57:111–20. https://doi.org/10.1016/j.reprotox.2015.05.018.
Tyagi V, Mustafa MD, Sharma T, Banerjee BD, Ahmed RS, Tripathi AK, et al. Association of organochlorine pesticides with the mRNA expression of tumour necrosis factor-alpha (TNF-α) & cyclooxygenase-2 (COX-2) genes in idiopathic preterm birth. Indian J Med Res. 2016;143(6):731–8. https://doi.org/10.4103/0971-5916.191986.
Sloboda D, Alfaidy N, Lye S, Gibb W, Patel F, Whittle W, et al. Prostaglandins and mechanisms of preterm birth. Reproduction. 2002;124(1):1–17. https://doi.org/10.1530/rep.0.1240001.
Zhou G, Holzman C, Heng YJ, Kibschull M, Lye SJ. Maternal blood EBF1-based microRNA transcripts as biomarkers for detecting risk of spontaneous preterm birth: a nested case-control study. J Matern-Fetal Neonatal Med. 2020;35(7):1239–47. https://doi.org/10.1080/14767058.2020.1745178.
Zhou G, Holzman C, Chen B, Wang P, Heng YJ, Kibschull M, et al. EBF1-correlated long non-coding RNA transcript levels in 3rd trimester maternal blood and risk of spontaneous preterm birth. Reprod Sci. 2021;28(2):541–9. https://doi.org/10.1007/s43032-020-00320-5.
Winger EE, Reed JL, Ji X. Early first trimester peripheral blood cell microRNA predicts risk of preterm delivery in pregnant women: proof of concept. PLoS ONE. 2017;12(7):e0180124. https://doi.org/10.1371/journal.pone.0180124.
Ngo TTM, Moufarrej MN, Rasmussen M-LH, Camunas-Soler J, Pan W, Okamoto J, et al. Noninvasive blood tests for fetal development predict gestational age and preterm delivery. Science. 2018;360(6393):1133–6. https://doi.org/10.1126/science.aar3819.
Weiner C, Dong Y, Zhou H, Cuckle H, Ramsey R, Egerman R, et al. Early pregnancy prediction of spontaneous preterm birth before 32 completed weeks of pregnancy using plasma RNA: transcriptome discovery and initial validation of an RNA panel of markers. BJOG. 2021;128(11):1870–80. https://doi.org/10.1111/1471-0528.16736.
Heng YJ, Pennell CE, McDonald SW, Vinturache AE, Xu J, Lee MWF, et al. Maternal whole blood gene expression at 18 and 28 weeks of gestation associated with spontaneous preterm birth in asymptomatic women. PLoS ONE. 2016;11(6):e0155191. https://doi.org/10.1371/journal.pone.0155191.
Montenegro D, Romero R, Kim S, Tarca A, Draghici S, Kusanovic J, et al. Expression patterns of microRNAs in the chorioamniotic membranes: a role for microRNAs in human pregnancy and parturition. J Pathol. 2009;217(1):113–21. https://doi.org/10.1002/path.2463.
Tiwari D, Bose PD, Das S, Das CR, Datta R, Bose S. MTHFR (C677T) polymorphism and PR (PROGINS) mutation as genetic factors for preterm delivery, fetal death and low birth weight: a Northeast Indian population based study. Meta Gene. 2015;3:31–42. https://doi.org/10.1016/j.mgene.2014.12.002.
Truong G, Guanzon D, Kinhal V, Elfeky O, Lai A, Longo S, et al. Oxygen tension regulates the miRNA profile and bioactivity of exosomes released from extravillous trophoblast cells – liquid biopsies for monitoring complications of pregnancy. PLoS ONE. 2017;12(3):e0174514. https://doi.org/10.1371/journal.pone.0174514.
Hong X, Sherwood B, Ladd-Acosta C, Peng S, Ji H, Hao K, et al. Genome-wide DNA methylation associations with spontaneous preterm birth in US blacks: findings in maternal and cord blood samples. Epigenetics. 2018;13(2):163–72. https://doi.org/10.1080/15592294.2017.1287654.
Ross KM, Carroll JE, Horvath S, Hobel CJ, Coussons-Read ME, Dunkel Schetter C. Epigenetic age and pregnancy outcomes: GrimAge acceleration is associated with shorter gestational length and lower birthweight. Clinical Epigenetics. 2020;12(1) https://doi.org/10.1186/s13148-020-00909-2.
Merid SK, Novoloaca A, Sharp GC, Küpers LK, Kho AT, Roy R, et al. Epigenome-wide meta-analysis of blood DNA methylation in newborns and children identifies numerous loci related to gestational age. Genome Med. 2020;12(1) https://doi.org/10.1186/s13073-020-0716-9.
York TP, Latendresse SJ, Jackson-Cook C, Lapato DM, Moyer S, Wolen AR, et al. Replicated umbilical cord blood DNA methylation loci associated with gestational age at birth. Epigenetics. 2020;15(11):1243–58. https://doi.org/10.1080/15592294.2020.1767277.
Lin VW, Baccarelli AA, Burris HH. Epigenetics—a potential mediator between air pollution and preterm birth. Environ. Epigenetics. 2016;2(1):dvv008. https://doi.org/10.1093/eep/dvv008.
Burris HH, Baccarelli AA, Wright RO, Wright RJ. Epigenetics: linking social and environmental exposures to preterm birth. Pediatr Res. 2016;79(1-2):136–40. https://doi.org/10.1038/pr.2015.191.
Parets SE, Conneely KN, Kilaru V, Fortunato SJ, Syed TA, Saade G, et al. Fetal DNA methylation associates with early spontaneous preterm birth and gestational age. PLoS ONE. 2013;8(6):e67489. https://doi.org/10.1371/journal.pone.0067489.
Lee H, Jaffe AE, Feinberg JI, Tryggvadottir R, Brown S, Montano C, et al. DNA methylation shows genome-wide association ofNFIX,RAPGEF2andMSRB3with gestational age at birth. Int J Epidemiol. 2012;41(1):188–99. https://doi.org/10.1093/ije/dyr237.
Wang X-M, Tian F-Y, Fan L-J, Xie C-B, Niu Z-Z, Chen W-Q. Comparison of DNA methylation profiles associated with spontaneous preterm birth in placenta and cord blood. BMC Med Genom. 2019;12(1) https://doi.org/10.1186/s12920-018-0466-3.
Fernando F, Keijser R, Henneman P, Van Der Kevie-Kersemaekers A-MF, Mannens MM, van der Post JA, et al. The idiopathic preterm delivery methylation profile in umbilical cord blood DNA. BMC Genom. 2015;16(1) https://doi.org/10.1186/s12864-015-1915-4.
Kacerovsky M, Lenco J, Musilova I, Tambor V, Lamont R, Torloni MR, et al. Proteomic biomarkers for spontaneous preterm birth: a systematic review of the literature. Reprod Sci. 2014;21(3):283–95. https://doi.org/10.1177/1933719113503415.
Polettini J, Cobo T, Kacerovsky M, Vinturache AE, Laudanski P, Peelen MJCS, et al. Biomarkers of spontaneous preterm birth: a systematic review of studies using multiplex analysis. J Perin Med. 2017;45(1):71–84. https://doi.org/10.1515/jpm-2016-0097.
Peelen MJ, Luef BM, Lamont RF, De Milliano I, Jensen JS, Limpens J, et al. The influence of the vaginal microbiota on preterm birth: a systematic review and recommendations for a minimum dataset for future research. Placenta. 2019;79:30–9. https://doi.org/10.1016/j.placenta.2019.03.011.
Knijnenburg TA, Vockley JG, Chambwe N, Gibbs DL, Humphries C, Huddleston KC, et al. Genomic and molecular characterization of preterm birth. Proc Natl Acad Sci. 2019;116(12):5819–27. https://doi.org/10.1073/pnas.1716314116.
Jehan F, Sazawal S, Baqui AH, Nisar MI, Dhingra U, Khanam R, et al. Multiomics characterization of preterm birth in low- and middle-income countries. JAMA Network Open. 2020;3(12):e2029655. https://doi.org/10.1001/jamanetworkopen.2020.29655.
Tarca AL, Pataki BÁ, Romero R, Sirota M, Guan Y, Kutum R, et al. Crowdsourcing assessment of maternal blood multi-omics for predicting gestational age and preterm birth. Cell Rep Med. 2021;2(6):100323. https://doi.org/10.1016/j.xcrm.2021.100323.
Feldner-Busztin D, Firbas Nisantzis P, Edmunds SJ, Boza G, Racimo F, Gopalakrishnan S, et al. Dealing with dimensionality: the application of machine learning to multi-omics data. Bioinformatics. 2023;39(2) https://doi.org/10.1093/bioinformatics/btad021.
Acknowledgements
We would like to acknowledge Alexander Johns, Sanjna Gangakhedkar, and Liam McAuliffe for their assistance conducting the literature search.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions
Author information
Authors and Affiliations
Contributions
Elyse Mead, Jason Phung, Carol Wang, and Craig Pennell were involved in the design and evaluation in this review and drafting the manuscript. Elyse Mead and Joanna Fu were involved in the execution of the literature search. All authors were involved in editing and critical discussion. All authors approved of the final version of this manuscript.
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mead, E.C., Wang, C.A., Phung, J. et al. The Role of Genetics in Preterm Birth. Reprod. Sci. 30, 3410–3427 (2023). https://doi.org/10.1007/s43032-023-01287-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-023-01287-9