Abstract
Robertsonian translocations (ROBs) are the most common structural chromosomal abnormalities in the general population, with an estimated incidence rate of 1/1000 births. In this study, we retrospectively analyzed the cases of ROBs from September 2015 to August 2022 and totally identified ROB carriers from 84,569 specimens karyotyped in a single accredited laboratory in China, including 189 cases of balanced ROBs and 3 of mosaic ROBs. Microsoft Excel and descriptive statistics were used to record and analyze the collected data. The male/female ratio of ROBs is 1/1.29, with der(13;14) and der(14;21) being the main karyotypes. Among the 192 patients, 7 were lost to follow-up, 82 had given birth, and 103 were childless (such as miscarriage, fetal chromosomal abnormalities, in vitro fertilization (IVF) failure, or divorce). A total of 44 amniocenteses were performed in 42 couples; ROB cases with natural pregnancies showed that the normal karyotype and balanced ROBs of fetal accounted for 66.67% (16/24), while the results of assisted pregnancies showed 90.00% (18/20). This study represents the largest collections of ROBs in Jiangxi population and reminder that the ROB carriers can achieve the ideal outcome for pregnancy with the appropriate genetic guidance and assisted reproductive technologies (ART).
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Robertsonian translocations (ROBs) are special forms of translocation in which the entire long arm of proximal centromere chromosomes is fused, also known as centromere fusion [1]. Translocations can occur between homologous chromosomes, but are more common between non-homologous pairs. Acrocentric chromosomes (13, 14, 15, 21, 22) can fuse to form 5 homologous ROBs and 10 heterologous ROBs; involving translocations of chromosomes 13 and 14 [der(13;14)] is the most common rearrangement. The majority of heterologous ROBs are inherited from one partner carrier, the rest are produced in the meiosis I of oogenesis. Almost all homologous ROBs are formed during mitosis. Although ROBs may have a normal phenotype and almost no deletion of genetic material, their pregnancies are at an increased risk of infertility and miscarriage and may lead to a livebirth with intellectual disability secondary to unbalanced chromosomal arrangement [2, 3]. It was reported that the incidence of ROBs is 0.1% in the general population, 1.1% in patients with recurrent pregnancy loss, and 3% in infertile men, making ROB rearrangement one of the most common structural chromosomal abnormalities [4].
Nearly one million newborns are born in China each year with different types of birth defects. Chromosome abnormalities, including ROBs, are considered to be one of the most important causes [5]. Chromosomal abnormalities refer to genetic diseases caused by changes in chromosome structure and (or) abnormal number and are important causes of infertility, fetal abortion, neonatal malformation, mental retardation, developmental delay, and other diseases. Early and accurate detection is an important guarantee for the diagnosis of chromosomal abnormalities, which is of great significance for preventing the occurrence of abortion and improving the rate of good birth; hence, it is valuable to establish a birth defect surveillance system. However, only a few studies have reported data involving ROBs in Chinese populations [6, 7]. In order to know the occurrence of ROBs and pregnancy outcomes in Jiangxi province of China and to provide reference for genetic counseling and fertility guidance, 192 cases of ROBs were collected and tracked in this study.
Materials and Methods
A 7-year retrospective study from September 2015 to August 2022 was carried out in couples with a clinical diagnosis of infertility, miscarriage, recurrent abortion, and genetic counseling at Jiangxi Key Laboratory of Birth Defect Prevention and Control of Jiangxi Maternal and Child Health Hospital. The age of the patients ranged from 20 to 52 years old. The reproductive histories of each patient were recorded in detail, and peripheral blood was taken. Inform consent was signed before the study.
Peripheral blood karyotype: 2 mL of peripheral blood was taken with heparin anticoagulation. About 0.5 mL of blood was inoculated into lymphocyte culture medium (Dahui Biotechnology) at 37 ℃ for 72 h. Four hours before the end of culture, colchicine (20 μg/mL) was added and prepared by conventional method. Cytogenetic analysis was performed at approximately 400-band level. Leica Cytovision System was used to analyze 5 karyotypes and count 30 karyotypes and at least 50 karyotypes in chimeric cases. Karyotypes were named according to the International System for Human Cytogenetic Nomenclature (ISCN) 2020.
Fetal karyotype: Chromosome abnormality was one of the important indications of cytogenetic prenatal diagnosis [8]. Informed consent forms were signed by pregnant women willing to undergo amniocentesis. Amniotic fluid samples were collected by ultrasound-guided transabdominal amniocentesis. Cells were cultured and prepared for G-banding karyotyping using standard protocols. Leica karyotype analysis system was used to count 20 karyotypes and analyze 5 karyotypes. Karyotypes were named according to ISCN 2020.
Results
Clinical Features of ROB Carriers
Among the 84,569 specimens karyotyped in this study, 192 cases were identified as ROB carriers. According to the medical records, the main reasons for treatment of ROBs were infertility and miscarriage, accounting for 81.25% of 156 cases, including 6 cases of azoospermia. The rest cases of ROBs were confirmed with the situations like pregnancy examination or genetic counseling, had unhealthy children, such as chromosomal abnormalities and congenital malformations, abnormal fetal ultrasound or high risk of non-invasive prenatal testing (NIPT) during pregnancy, and menstrual disorder (Table 1). Based on the miscarriage history, most cases had at least two spontaneous abortions (Table 2).
Karyotype Distribution of ROBs
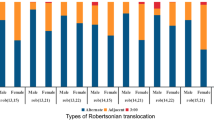
The 192 ROB cases can be classified into 189 balanced ROBs and 3 mosaic ROBs, 84 males and 108 females, producing male-to-female ratio of 1:1.29. There were 9 of homologous [der(13;13), der(21;21) and der(22;22)] and 183 of nonhomologous ROBs, while der(13;14) was the most common karyotype accounting for 60.94% (117/192). The detailed karyotype distribution of ROBs is shown in Table 3.
Pregnancy Outcomes of ROB Carriers
The 192 ROBs were followed up by telephone in March 2023, and the loss rate was 3.65% (7/192). Among the 185 tracked cases, 82 had given birth, 90 were childless (including infertile, miscarriage, abnormal fetal chromosome, and in vitro fertilization failed), 6 were divorced, 5 are ongoing pregnant, and 2 cases had adopted a child. There were 342 gravidities in total among the ROBs cases; although 170 pregnancies ended in miscarriage, at least 43.27% (148/342) were pregnant with normal phenotype fetus (Table 4).
Prenatal Diagnosis of ROB Carriers
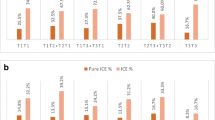
Forty-two pregnant ROB cases, including 24 natural pregnancies and 20 assisted pregnancies, have accepted 44 amniocenteses to test the fetal karyotypes. The results of prenatal diagnosis in ROBs cases with natural pregnancies showed that the normal karyotype and balanced ROBs of fetal accounted for 66.67% (16/24) and trisomy and other abnormities accounted for 33.33% (8/24), while the results of assisted pregnancies showed that 90.00% (18/20) were normal karyotype and balanced ROBs, and the other 2 cases were Trisomy-21 Syndrome (Table 5).
Discussion
Robertsonian translocation (ROB) is a common chromosomal abnormality, which is formed by the fusion of two acrocentric chromosomes. It is related to the oligospermia in male adults, miscarriage or infertility in female adults, and occurring at a rate of about 1/1000 newborns [1, 5]. Although the number of chromosomes was reduced, the main genetic material was not lost so that the ROB carriers were usually phenotypically normal except for reproductive disorders. In the process of meiosis of ROB carriers, apart from a normal gamete and a ROB, approximately two-thirds of their gametes feature disomy or nullisomy, that easily lead to early embryo abortion and stillbirth, means almost one-third of the probability to give birth to a phenotypically normal child [9, 10]. In our study, 81.25% of ROB carriers came to the clinic due to infertility or miscarriage, and 59 of them had at least two spontaneous abortions. A total of 342 pregnancies were recorded in the 185 tracked cases, of which 49.71% (170/342) ended in abortion and 43.27% (148/342) were born with normal phenotype, higher than the theoretical value. Therefore, history of multiple miscarriages is an obvious indication for genetic diagnosis.
Individuals with ROBs usually have only 45 chromosomes, because the short arms of two acrocentric chromosomes are lost during subsequent cell divisions [11, 12]. The most common type of ROB occurs between chromosomes 13 and 14, and the second common is between chromosomes 14 and 21; other possible combinations are infrequent [13]. Consistent with previous researches, among the 192 cases in our study, der(13;14) was the most common karyotype, accounting for 60.94%, followed by der(14; 21), with a total of 28 cases accounting for 14.58% (28/192). The gender of ROBs can obviously affect the proportion of meiotic segregation patterns. Studies of chromosomal segregation in ROBs have shown that the male carriers had a significantly higher proportion of normal/balanced gametes than the female carriers [14, 15]. In our study, we found 192 ROBs in the 84,569 patients, with a male to female ratio of 1:1.29. This proportion was contrary to previous studies, which may be attributed to the traditional belief that more miscarriages were caused by female factors than by male ones, so more women in this region coming for pregnancy counseling [16].
In recent decades, great progress in assisted reproductive technology (ART), encompass in vitro fertilization (IVF) with or without intracytoplasmic sperm injection (ICSI), have enabled previously untreatable infertility to be successfully treated [17, 18]. ART has become an important method to effectively improve the clinical pregnancy rate for the infertile couples. However, the patients undergoing ART still have the risk of genetic disorders, such as embryonic chromosomal abnormality. Researches have showed that, 20% of pregnant women receiving ART had spontaneous miscarriage in early stage of pregnancy [19, 20]. Therefore, it is necessary to have proper counseling and testing before preparing for pregnancy, especially for the embryo implantation in ART. Preimplantation genetic diagnosis (PGD) is a genetic analysis of an embryo or gamete, and embryos without chromosome or genetic abnormality are selected to transplanted into the mother’s uterus [21]. Therefore, for high-risk couples, the genetic material of embryos can be analyzed before embryo transfer to diagnose whether there are abnormalities and select healthy embryos for transplantation. For female carriers, PGD can be used to screen abnormal polar bodies, while male carriers need to use ICSI and PGD to select normal or balanced translocation embryos. It is proved that PGD can obviously reduce the abortion rate of ROBs carriers [22, 23]. Although guidelines recommend that couples with chromosome abnormalities could undergo amniocentesis to test fetal chromosome, amniocentesis was an invasive procedure; considering the risk of pregnancy loss or preterm birth, 42 women underwent a total of 44 amniocenteses during the second trimester of pregnancy. The proportion of normal karyotype and ROB in natural and ART pregnancies was 66.67% and 90%, respectively. The application of ART and PGD can remarkably improve the probability to have phenotypically normal children for ROB carriers.
This study provides data reference for pregnancy in Robertsonian translocation carriers. However, it has certain limitations, some carriers from diagnosis to follow-up only half a year; the next step will be to regularly follow up their pregnancy. Secondly, there is a lack of records of newborns after birth. We will follow up the growth and development of children with Robertson carriers. Thirdly, the portion of prenatal diagnosis is relatively low; concern about the pregnancy loss caused by amniocentesis is the main reason, especially for couples with IVF. Although prenatal diagnosis is based on voluntary principles, the probability of Robertson translocation carriers giving birth to trisomy and other abnormalities is significantly higher than that of other couples, and we will further strengthen the publicity and education of prenatal diagnosis.
To sum up, people with the normal type may carry balanced translocations or inversions that generally have no effect except for fertility problems. Parents with infertility and poor pregnancy history are recommended to undergo peripheral karyotyping to detect any balanced structural chromosomal abnormality. Couples with chromosomal abnormality should be given proper fertility guidance. Although PGD could reduce the pregnancy loss, the ability to achieve successful natural pregnancy should be carefully considered, especially for young fertile couples without a history of recurrent miscarriage [24]. Couples with chromosomal abnormalities, advanced age, and a history of adverse pregnancy could choose PGD. Preimplantation genetic diagnosis could be used before embryo implantation, and prenatal diagnosis (chorion villus sampling, amniocentesis, and percutaneous umbilical blood sampling) could be performed when necessary. More importantly, couples are advised to undergo genetic testing before pregnancy, which can prevent miscarriage or fetal abnormalities caused by chromosomal diseases or monogenic diseases.
Data Availability
Not applicable.
References
Wilch ES, Morton CC. Historical and clinical perspectives on chromosomal translocations. Adv Exp Med Biol. 2018;1044:1–14. https://doi.org/10.1007/978-981-13-0593-1_1.
Scriven PN, Flinter FA, Braude PR, Ogilvie CM. Robertsonian translocations–reproductive risks and indications for preimplantation genetic diagnosis. Hum Reprod. 2001;16(11):2267–73. https://doi.org/10.1093/humrep/16.11.2267.
Wang HL, Wu B, Guo KM, Tian RH. Psychological characteristics of and counseling for carriers of structural chromosome abnormalities. Genet Mol Res. 2016;15:2. https://doi.org/10.4238/gmr.15028159.
Fryns JP, Van Buggenhout G. Structural chromosome rearrangements in couples with recurrent fetal wastage. Eur J Obstet Gynecol Reprod Biol. 1998;81(2):171–6. https://doi.org/10.1016/s0301-2115(98)00185-7.
Morin SJ, Eccles J, Iturriaga A, Zimmerman RS. Translocations, inversions and other chromosome rearrangements. Fertil Steril. 2017;107(1):19–26. https://doi.org/10.1016/j.fertnstert.2016.10.013.
Lau TK, Fung HY, Rogers MS, Cheung KL. Racial variation in incidence of trisomy 21: survey of 57,742 Chinese deliveries. Am J Med Genet. 1998;75(4):386–8.
Zhao WW, Wu M, Chen F, Jiang S, Su H, Liang J, et al. Robertsonian translocations: an overview of 872 Robertsonian translocations identified in a diagnostic laboratory in China. PLoS ONE. 2015;10(5):e0122647. https://doi.org/10.1371/journal.pone.0122647.
Committee For The Prevention And Control Of Birth Defect Chinese Association Of Preventive Medicine, Zhang X, Qi Q, Hu T, Liu S. Guidelines for the interpretation of fetal chromosomal karyotyping analysis. Zhonghua Yi Xue Yi Chuan Xue Za Zhi. 2021;38(5):409–13. https://doi.org/10.3760/cma.j.cn511374-20201211-00867.
Sole M, Blanco J, Valero O, Verges L, Vidal F, Sarrate Z. Altered bivalent positioning in metaphase I human spermatocytes from Robertsonian translocation carriers. J Assist Reprod Genet. 2017;34(1):131–8. https://doi.org/10.1007/s10815-016-0809-y.
Godo A, Blanco J, Vidal F, Sandalinas M, Garcia-Guixe E, Anton E. Altered segregation pattern and numerical chromosome abnormalities interrelate in spermatozoa from Robertsonian translocation carriers. Reprod Biomed Online. 2015;31(1):79–88. https://doi.org/10.1016/j.rbmo.2015.04.003.
Bandyopadhyay R, Heller A, Knox-DuBois C, McCaskill C, Berend SA, Page SL, et al. Parental origin and timing of de novo Robertsonian translocation formation. Am J Hum Genet. 2002;71(6):1456–62. https://doi.org/10.1086/344662.
van Sluis M, Gailín MÓ, McCarter JGW, Mangan H, Grob A, McStay B. Human NORs, comprising rDNA arrays and functionally conserved distal elements, are located within dynamic chromosomal regions. Genes Dev. 2019;33(23–24):1688–701. https://doi.org/10.1101/gad.331892.119.
Poot M, Hochstenbach R. Prevalence and phenotypic impact of Robertsonian translocations. Mol Syndromol. 2021;12(1):1–11. https://doi.org/10.1159/000512676.
Zhang L, Jiang W, Zhu Y, Chen H, Yan J, Chen ZJ. Effects of a carrier’s sex and age on the segregation patterns of the trivalent of Robertsonian translocations. J Assist Reprod Genet. 2019;36(9):1963–9. https://doi.org/10.1007/s10815-019-01534-6.
Dang T, Xie P, Zhang Z, Hu L, Tang Y, Tan Y, et al. The effect of carrier characteristics and female age on preimplantation genetic testing results of blastocysts from Robertsonian translocation carriers. J Assist Reprod Genet. 2023;40(8):1995–2002. https://doi.org/10.1007/s10815-023-02853-5
Priya PK, Mishra VV, Roy P, Patel H. A study on balanced chromosomal translocations in couples with recurrent pregnancy loss. J Hum Reprod Sci. 2018;11(4):337–42. https://doi.org/10.4103/jhrs.JHRS_132_17.
Graham ME, Jelin A, Hoon AH Jr, Wilms Floet AM, Levey E, Graham EM. Assisted reproductive technology: short- and long-term outcomes. Dev Med Child Neurol. 2023;65(1):38–49. https://doi.org/10.1111/dmcn.15332.
De Geyter C. Assisted reproductive technology: impact on society and need for surveillance. Best Pract Res Clin Endocrinol Metab. 2019;33(1):3–8. https://doi.org/10.1016/j.beem.2019.01.004.
Magli MC, Crippa A, Benincasa M, Terzuoli G, Azzena S, Maresca L, et al. Sperm chromosome abnormalities in patients with normal karyotype and in translocation carriers: clinical relevance for assisted reproductive technology. Reprod Biomed Online. 2020;41(6):1055–69. https://doi.org/10.1016/j.rbmo.2020.08.005.
Hilbert SM, Gunderson S. Complications of assisted reproductive technology. Emerg Med Clin North Am. 2019;37(2):239–49. https://doi.org/10.1016/j.emc.2019.01.005.
Vitez SF, Forman EJ, Williams Z. Preimplantation genetic diagnosis in early pregnancy loss✰. Semin Perinatol. 2019;43(2):116–20. https://doi.org/10.1053/j.semperi.2018.12.009.
Beyer CE, Willats E. Natural selection between day 3 and day 5/6 PGD embryos in couples with reciprocal or Robertsonian translocations. J Assist Reprod Genet. 2017;34(11):1483–92. https://doi.org/10.1007/s10815-017-1009-0.
Zhang S, Lei C, Wu J, Zhou J, Sun H, Fu J, et al. The establishment and application of preimplantation genetic haplotyping in embryo diagnosis for reciprocal and Robertsonian translocation carriers. BMC Med Genomics. 2017;10(1):60. https://doi.org/10.1186/s12920-017-0294-x.
Chang EM, Han JE, Kwak IP, Lee WS, Yoon TK, Shim SH. Preimplantation genetic diagnosis for couples with a Robertsonian translocation: practical information for genetic counseling. J Assist Reprod Genet. 2012;29(1):67–75. https://doi.org/10.1007/s10815-011-9654-1.
Funding
This study was supported by the Youth Science Foundation of Jiangxi Province under Grant number 20192BAB215010 and Jiangxi Provincial Clinical Research Center for Birth Defects under Grant number 20223BCG74002.
Author information
Authors and Affiliations
Contributions
YL and BY contributed to the study conception and design. Material preparation, data collection, and analysis were performed by all authors. The first draft of the manuscript was written by WL, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration, and all participants have given a written informed consent.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lu, W., Zhou, J., Rao, H. et al. A Retrospective Analysis of Robertsonian Translocations from a Single Center in China. Reprod. Sci. 31, 851–856 (2024). https://doi.org/10.1007/s43032-023-01398-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-023-01398-3