Abstract
Background
Research on obstructive sleep apnea (OSA) is inadequate in Saudi Arabia, particularly among patients with comorbidities. This study investigates comorbidities in patients with different severity of apnea based on the Apnea–Hypopnea Index (AHI).
Methods
The retrospective charts review that included a cohort of 4391 patients who underwent polysomnography (PSG) between 2003 and 2019. The AHI is classified into four ordinal groups: normal, mild, moderate, and severe. Ordinal logistic regression was used to model proportional odds of a higher AHI category.
Results
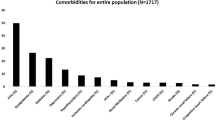
Gender was distributed equally in the study sample. The average age was 49.6 ± 14.8 years and the average AHI was 16.1 ± 22 per hour. Hypertension (43.2%) and diabetes mellitus (37.3%) were the most common comorbidities: Mild OSA 28.9%, Moderate OSA 15.6%, and severe 16.4%. The severity of apnea increased with age and BMI classes. The prevalence of hypertension increased with the severity of apnea: 42.9% in mild, 47.4% in moderate, and 54.6% in severe AHI. The prevalence of coronary artery disease (CAD), congestive heart failure (CHF), and diabetes mellitus (DM) increased with the severity of apnea. Comorbidities was more among OSA patients with excessive sleepiness. After adjustment for age and gender, greater proportional odds of severe AHI were observed in males (aOR = 1.8), 30–59 years (aOR = 2.064), 60 years or above (aOR = 2.873), obese class II (aOR = 2.016), obese class III (aOR = 2.527), and in patients with hypertension (aOR = 1.272).
Conclusion
Hypertension and obesity were highly prevalent in the study cohort and were associated with greater proportional odds of severe AHI.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Background
Obstructive sleep apnea (OSA) is a serious health problem affecting not only an individual’s sleep but also health in general including cardiopulmonary, endocrine, vascular, and central nervous systems [1,2,3,4,5,6,7]. Nevertheless, OSA is not given much public attention in terms of screening and diagnosis. OSA is diagnosed with the use of polysomnography (PSG) testing by recording repetitive episodes of upper airway closure during sleep [8, 9]. OSA is established in terms of the Apnea–Hypopnea Index (AHI), which is the number of apnea–hypopnea/per hour measured during a sleep study [8,9,10,11,12]. AHI ≥ 5 per hour is used to diagnose OSA [8,9,10, 13, 14]. OSA severity is classified as follows: AHI < 5 per hour (normal), 5–15 AHI per hour (mild sleep apnea), ≥ 15 and < 30 AHI per hour (moderate sleep apnea), and AHI ≥ 30 per hour (severe sleep apnea) [9]. Untreated sleep apnea is associated with major comorbidities and health problems [1, 2, 15,16,17]. The prevalence of sleep apnea is considerably higher in patients with comorbidities than within the general population [5].
Few local studies revealed a high prevalence of OSA among the Saudi adult population [18,19,20,21]. Most of the studies conducted in Saudi Arabia used the Berlin Questionnaire (BQ) to screen for symptoms of sleep apnea. Based on the BQ, high-risk patients for sleep apnea were present in three out of ten of the population [18,19,20]. A major limitation of these studies is the use of subjective sleep assessments such as the Berlin Questionnaire rather than a formal sleep study or polysomnography. Regardless, sleep apnea in patients with comorbidities in Saudi Arabia is not well studied. We hypothesized that comorbidities such as diabetes, hypertension, and obesity increase with severe sleep apnea. This study aimed to determine whether there was a trend between the severity of sleep apnea and comorbidities controlling for demographic characteristics.
2 Methods
A retrospective charts review was conducted at the Sleep Disorders Center (SDC) between 2003 and 2019. The SDC was established in 2003 at the King Abdulaziz Medical City in Riyadh (KAMC-R), Saudi Arabia. KAMC-R, with a capacity of 1800 beds, is considered as one of the biggest tertiary hospitals in Saudi Arabia and the Middle East. The study obtained ethical approval from the Ministry of National Guard—Health Affairs, Institution of Research Board (IRB) registered under number # RC15/058/R.
We have established a registry for all patients referred to our sleep disorders center since 2003. All patients filled out questionnaires concerning demographics, sleep symptoms, and comorbidities. All relevant data were collected, including anthropometric measurements (age, gender, body mass index (BMI), neck size, and Mallampati score), comorbidities, sleepiness scale as per the Epworth Sleepiness Scale (ESS) [22], sleep study results, and final diagnosis. All patients underwent standard in-lab type PSG type I (PSG). The PSG recording was performed using Alice® 5 and Alice® 6 diagnostic equipment (Respironics Inc., Murrysville, PA, USA). Manual scoring of the electronic raw data was performed by expert certified sleep technologists by following established criteria [23]. Hypopnea was defined as a reduction in airflow of ≥ 30% of the baseline that lasted for at least 10 s and resulted in either a ≥ 3% decrease in oxygen saturation from the pre-event baseline or arousal. Apnea was defined as a drop in the peak thermal sensor excursion greater than or equal to 90% of baseline for at least 10 s. The event was scored as obstructive apnea in the presence of continued respiratory effort. In this study, the study outcome, Apnea–Hypopnea Index (AHI), is categorized into four groups: normal (AHI < 5) per hour, mild (5 ≥ AHI < 15) per hour, moderate (15 ≥ AHI < 30) per hour, and severe (AHI ≥ 30) per hour [8, 9]. The Epworth Sleepiness Scale (ESS) was used to assess sleepiness in eight different situations on a scale of 0 (never doze) to 3 (high chance of dozing). The overall score is classified into four ordinal groups: normal 0–10, mild 11–14, moderate 15–17, and severe 18–24 [22, 24]. A cohort of 6030 patients who underwent PSGy during the study period was included in the analysis. We excluded all therapeutic sleep studies, narcolepsy, or less than 3 h of total sleeping time. We included 4391 patients in this analysis. All scored sleep studies were reviewed by one senior certified sleep technologist using AASM manual scoring of sleep and associated events [23].
The study population includes adults’ patients (18 years or above) who were suspected of sleep apnea and were referred to the in-lab polysomnographic sleep study. The study retrieved data on demographic characteristics such as age and gender. Data about patient clinical characteristics were collected (e.g., comorbidities such as hypertension, diabetes mellitus, coronary artery disease, obesity, congestive heart failure, and bronchial asthma).
2.1 Data Analysis
SAS v 9.4 (SAS Institute Inc., Cary, NC, USA) was used for data analysis. Overall comorbidities and sample descriptive analyses were reported by frequency distributions. The frequency distributions of demographic and comorbidities variables were classified by the severe sleep apnea index and severity of sleepiness as per ESS and were tested by chi-squared methods. Since severity of apnea—normal, mild, moderate, and severe AHI—is ordinal, we performed the ordinal logistic regression model to assess the association between demographic and comorbidities variables and the likelihood of the severe sleep apnea index. We estimated adjusted odds ratio (aOR) and 95% confidence interval (95% CI). The assumption of proportional odds was assessed using a chi-squared score test. A P value greater than 0.05 indicates that the ordered logit coefficients are equal across the severity of the sleep apnea index.
3 Results
We reported overall frequency distributions of demographic characteristics and comorbidities in Table 1. Gender was distributed equally in the study sample. The average age was 49.6 ± 14.8 years and the average AHI was 16.1 ± 22 per hour. Hypertension (43.2%) and diabetes mellitus (37.3%) were the most common comorbidities.
Table 2 compares demographics and comorbidities across different severity of apnea: normal, mild, moderate, and severe AHI. Of the sample, 1713 (39.1%) patients had normal AHI, 1269 (28.9%) patients had mild AHI, 687 (15.6%) patients had moderate AHI, and 722 (16.4) patients had severe AHI. There was an indicative difference in severity of apnea by gender (P = 0.001). Severe AHI (57.8%) was significantly more common among males, compared to 42% common among females (P = 0.001).
The severity of apnea increases with age (P = 0.001) and BMI (P = 0.001). The prevalence of hypertension (P = 0.001) increases with the severity of apnea: 36.9% in patients with normal AHI, 42.9% in mild, 47.4% in moderate, and 54.6% in patients with severe AHI. The prevalence of coronary artery disease (P = 0.001), congestive heart failure (P = 0.012), and diabetes mellitus (P = 0.001) increase with the severe apnea. Sleepiness (ESS ≥ 11) was common in severe AHI (P = 0.001).
Table 3 illustrates the prevalence of comorbidities and its relation to the severity of OSA with EDS (ESS > 11) and without sleepiness. OSA with significant EDS was more common in male, obesity and older patients. The prevalence of hypertension, congestive heart failure, and diabetes mellitus were more significant among patient with OSA and EDS. EDS without OSA was more among young patients.
When controlling for the demographic and comorbidities in Table 4, the ordinal logistic regression suggested that male gender, age, BMI, and hypertension were independent risk factors of severe AHI. Unlike unadjusted analysis, sleepiness as per the ESS was not associated with severe AHI.
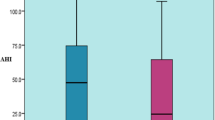
The proportional odds of severe AHI were greater in males compared to females (aOR = 1.8). The severity of AHI was greater in 30–59 years (aOR = 2.064) and in 60 years or above (aOR = 2.873) compared to 29 years or less, Fig. 1. The severity of AHI was greater in obese class II (aOR = 2.016) and in obese class III (aOR = 2.527) compared to normal BMI, Fig. 1.
Hypertension was associated with increased proportional odds of severe AHI (aOR = 1.272). Figures 2 and 3 illustrate BMI classes in relation to severe Apnea–Hypopnea Index by gender and age groups. The proportional odds assumption for this model was met, P value = 0.4540.
4 Discussion
There is a shortage of knowledge about the prevalence of comorbidities in Saudi Arabia in contrast to other populations. OSA is highly prevalent in the general population and occurs in all age groups and more in female than male [18,19,20, 25]. In this study, OSA was more in male compared to female. The higher prevalence of OSA among male in this study is consistent with reports from other countries [26]. Furthermore, compared to other local studies, we used objective diagnostic sleep study, PSG while other prevalence studies used questionaries [18,19,20]. This study is not about the prevalence of OSA among general population but among cases refereed to sleep center to role out OSA. Therefore, this result cannot be generalized. The main strength of the present study is the large sample size, and the description of the largest national experience. Furthermore, all the data were collected at the time of the sleep study, included an equal number of genders, and all the study scoring was reviewed by one senior certified technologist, avoiding any bias in scoring. It provides information about the association between OSA and comorbidities. Similar to other studies, we reported a higher prevalence of comorbidities among OSA [29, 30] and a higher prevalence of comorbidities in severe OSA and EDS [5, 27]. EDS is frequently reported by patients with OSA but is not invariably present. The overall prevalence of EDS in our population was 43%, and significantly high in severe OSA. The relation between OSA and EDS remains controversial [28,29,30]. EDS in OSA may indicate more arousals and is associated with a higher risk of comorbidities [7, 31, 32]. Nevertheless, recent studies reported the presence of EDS in sleep apnea patients associated with increases the risk of DM, obesity, hypertension, heart failure, and coronary heart disease [6, 7, 29,30,31, 33,34,35]. The present study EDS in OSA was associated with a higher risk of comorbidities. A higher prevalence of comorbidities including hypertension among our male patients is probably due to a higher prevalence of severe OSA among males compared to females (OR = 1.89). The Alharbi et al. study about the prevalence of hypertension among OSA patients reported a higher prevalence of hypertension among female patients [22]. However, this study includes a much larger number of participants, and this may reflect a true higher prevalence of hypertension among healthy male Saudis [33]. The prevalence of OSA is strongly associated with overweight and obesity [36, 37]. Obesity is quite prevalent among Saudi population. An epidemiological study about obesity among Saudis including 10,735 participants found 28.7% were obese (BMI ≥ 30 kg/m), which was higher among women (33.5% vs. 24.1%) [38]. The present study, obesity prevalence was higher among OSA patients (88%) and, using ordinal logistic regression obesity was an independent factor for OSA. The proportional odds of severe AHI were greater in obese classes II and III compared to normal BMI. Normal sleep study reported in almost more than third of our study sample, this is because we only recently used home sleep studies and in our center, we receive frequent referrals preoperative consultation to role out OSA before bariatric, ENT, and maxillofacial surgeries and to role sleep apnea as a cause of pulmonary hypertension and EDS.
The association of bronchial asthma and OSA is controversial. A systematic review by Davies et al. [44] found a weak association between asthma and OSA. The asthma prevalence in our study was 21.95% but we did not find any association between asthma and OSA or asthma.
Limitations include a retrospective design; therefore, no cause-and-effect relationship could be determined. It is a single-center experience, and it did not report the effect of the treatment on the severity or outcome. We encourage all sleep centers in the country to adopt registry information about OSA, to describe the prevalence, comorbidities, epidemiological patterns, and outcomes of OSA.
5 Conclusion
Our study revealed a high prevalence of associated comorbidities in patients with OSA and/or EDS.
Abbreviations
- OSA:
-
Obstructive sleep apnea
- AHI:
-
Apnea–Hypopnea Index
- CAD:
-
Coronary artery disease
- CHF:
-
Congestive heart failure
- BQ:
-
Berlin Questionnaire
- ESS:
-
Epworth Sleepiness Scale
- SDC:
-
Sleep Disorders Center
- PSG:
-
Polysomnography I
- 95% CI:
-
Confidence interval
- IRB:
-
Institution of Research Board
- DM:
-
Diabetes mellitus
References
Caples SM, Garcia-Touchard A, Somers VK. Sleep-disordered breathing and cardiovascular risk. Sleep. 2007;30(3):291–303.
Quan SF, Gersh BJ. Cardiovascular consequences of sleep-disordered breathing: past, present and future: report of a workshop from the National Center on Sleep Disorders Research and the National Heart, Lung, and Blood Institute. Circulation. 2004;109(8):951–7.
Shamsuzzaman AS, Gersh BJ, Somers VK. Obstructive sleep apnea: implications for cardiac and vascular disease. JAMA. 2003;290(14):1906–14.
Wolk R, Shamsuzzaman AS, Somers VK. Obesity, sleep apnea, and hypertension. Hypertension (Dallas, Tex: 1979). 2003;42(6):1067–74.
Appleton SL, Gill TK, Lang CJ, Taylor AW, McEvoy RD, Stocks NP, et al. Prevalence and comorbidity of sleep conditions in Australian adults: 2016 Sleep Health Foundation national survey. Sleep Health. 2018;4(1):13–9.
Javadi HR, Jalilolghadr S, Yazdi Z, Rezaie MZ. Correlation between obstructive sleep apnea syndrome and cardiac disease severity. Cardiovasc Psychiatry Neurol. 2014;2014: 631380.
Mazzotti DR, Keenan BT, Lim DC, Gottlieb DJ, Kim J, Pack AI. Symptom subtypes of obstructive sleep apnea predict incidence of cardiovascular outcomes. Am J Respir Crit Care Med. 2019;200(4):493–506.
Fleetham J, Ayas N, Bradley D, Ferguson K, Fitzpatrick M, George C, et al. Canadian Thoracic Society guidelines: diagnosis and treatment of sleep disordered breathing in adults. Can Respir J. 2006;13(7):387–92.
Force TRoaAAoSMT. Sleep-related breathing disorders in adults: recommendations for syndrome definition and measurement techniques in clinical research. Sleep. 1999;22(5):667–89.
Berry RB, Budhiraja R, Gottlieb DJ, Gozal D, Iber C, Kapur VK, et al. Rules for scoring respiratory events in sleep: update of the 2007 AASM Manual for the Scoring of Sleep and Associated Events. Deliberations of the Sleep Apnea Definitions Task Force of the American Academy of Sleep Medicine. J Clin Sleep Med (JCSM). 2012;8(5):597–619.
Johnson KG, Johnson DC. Frequency of sleep apnea in stroke and TIA patients: a meta-analysis. J Clin Sleep Med (JCSM). 2010;6(2):131–7.
Jean-Louis G, Zizi F, Clark LT, Brown CD, McFarlane SI. Obstructive sleep apnea and cardiovascular disease: role of the metabolic syndrome and its components. J Clin Sleep Med (JCSM). 2008;4(3):261–72.
Sleep-related breathing disorders in adults: recommendations for syndrome definition and measurement techniques in clinical research. The Report of an American Academy of Sleep Medicine Task Force. Sleep. 1999;22(5):667–89.
Mulgrew AT, Fox N, Ayas NT, Ryan CF. Diagnosis and initial management of obstructive sleep apnea without polysomnography: a randomized validation study. Ann Intern Med. 2007;146(3):157–66.
George CF. Sleep. 5: driving and automobile crashes in patients with obstructive sleep apnoea/hypopnoea syndrome. Thorax. 2004;59(9):804–7.
Ruel G, Martin SA, Lévesque JF, Wittert GA, Adams RJ, Appleton SL, et al. Association between multimorbidity and undiagnosed obstructive sleep apnea severity and their impact on quality of life in men over 40 years old. Glob Health Epidemiol Genomics. 2018;3: e10.
Mokhlesi B, Ham SA, Gozal D. The effect of sex and age on the comorbidity burden of OSA: an observational analysis from a large nationwide US health claims database. Eur Respir J. 2016;47(4):1162–9.
Bahammam AS, Al-Rajeh MS, Al-Ibrahim FS, Arafah MA, Sharif MM. Prevalence of symptoms and risk of sleep apnea in middle-aged Saudi women in primary care. Saudi Med J. 2009;30(12):1572–6.
BaHammam AS, Alrajeh MS, Al-Jahdali HH, BinSaeed AA. Prevalence of symptoms and risk of sleep apnea in middle-aged Saudi males in primary care. Saudi Med J. 2008;29(3):423–6.
Wali SO, Abalkhail B, Krayem A. Prevalence and risk factors of obstructive sleep apnea syndrome in a Saudi Arabian population. Ann Thor Med. 2017;12(2):88–94.
Alruwaili H, Ahmed A, Fatani A, Al-Otaibi K, Al-Jahdali S, Ali Y, et al. Symptoms and risk for obstructive sleep apnea among sample of Saudi Arabian adults. Sleep Biol Rhythms. 2015;13(4):332–41.
Johns MW. A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep. 1991;14(6):540–5.
Berry RBBR, Gottlieb DJ, Gozal D, Iber C, Kapur VK, Marcus CL, Mehra R, Parthasarathy S, Quan SF, Redline S, Strohl KP, Davidson Ward SL, Tangredi MM. American Academy of Sleep Medicine. Rules for scoring respiratory events in sleep. AASM Manual for the Scoring of Sleep and Associated Events. Deliberations of the Sleep Apnea Definitions Task Force of the American Academy of Sleep Medicine. J Clin Sleep Med. 2012;15(8):597–619.
Ahmed AE, Fatani A, Al-Harbi A, Al-Shimemeri A, Ali YZ, Baharoon S, et al. Validation of the Arabic version of the Epworth sleepiness scale. J Epidemiol Glob Health. 2014;4(4):297–302.
Lévy P, Kohler M, McNicholas WT, Barbé F, McEvoy RD, Somers VK, et al. Obstructive sleep apnoea syndrome. Nat Rev Dis Primers. 2015;1:15015.
Young T, Palta M, Dempsey J, Skatrud J, Weber S, Badr S. The occurrence of sleep-disordered breathing among middle-aged adults. N Engl J Med. 1993;328(17):1230–5.
Robichaud-Hallé L, Beaudry M, Fortin M. Obstructive sleep apnea and multimorbidity. BMC Pulm Med. 2012;12:60.
Corlateanu A, Pylchenko S, Sircu V, Botnaru V. Predictors of daytime sleepiness in patients with obstructive sleep apnea. Pneumologia (Bucharest, Romania). 2015;64(4):21–5.
Sauter C, Asenbaum S, Popovic R, Bauer H, Lamm C, Klösch G, et al. Excessive daytime sleepiness in patients suffering from different levels of obstructive sleep apnoea syndrome. J Sleep Res. 2000;9(3):293–301.
Garbarino S, Scoditti E, Lanteri P, Conte L, Magnavita N, Toraldo DM. Obstructive sleep apnea with or without excessive daytime sleepiness: clinical and experimental data-driven phenotyping. Front Neurol. 2018;9:505.
Feng J, He QY, Zhang XL, Chen BY. Epworth Sleepiness Scale may be an indicator for blood pressure profile and prevalence of coronary artery disease and cerebrovascular disease in patients with obstructive sleep apnea. Sleep Breathing Schlaf Atmung. 2012;16(1):31–40.
Allen AJH, Jen R, Mazzotti DR, Keenan BT, Goodfellow SD, Taylor CM, et al. Symptom subtypes and risk of incident cardiovascular and cerebrovascular disease in a clinic-based obstructive sleep apnea cohort. J Clin Sleep Med (JCSM). 2022;18(9):2093–102.
Šiarnik P, Klobučníková K, Šurda P, Putala M, Šutovský S, Kollár B, et al. Excessive daytime sleepiness in acute ischemic stroke: association with restless legs syndrome, diabetes mellitus, obesity, and sleep-disordered breathing. J Clin Sleep Med (JCSM). 2018;14(1):95–100.
Li X, Huang H, Xu H, Shi Y, Qian Y, Zou J, et al. Excessive daytime sleepiness, metabolic syndrome, and obstructive sleep apnea: two independent large cross-sectional studies and one interventional study. Respir Res. 2019;20(1):276.
Gabryelska A, Białasiewicz P. Association between excessive daytime sleepiness, REM phenotype and severity of obstructive sleep apnea. Sci Rep. 2020;10(1):34.
Schwartz AR, Patil SP, Laffan AM, Polotsky V, Schneider H, Smith PL. Obesity and obstructive sleep apnea: pathogenic mechanisms and therapeutic approaches. Proc Am Thorac Soc. 2008;5(2):185–92.
Peppard PE, Young T, Palta M, Dempsey J, Skatrud J. Longitudinal study of moderate weight change and sleep-disordered breathing. JAMA. 2000;284(23):3015–21.
Memish ZA, El Bcheraoui C, Tuffaha M, Robinson M, Daoud F, Jaber S, et al. Obesity and associated factors—Kingdom of Saudi Arabia, 2013. Prev Chronic Dis. 2014;11:E174.
Funding
This study has is funded by King Abdullah International Medical Research Center.
Author information
Authors and Affiliations
Contributions
HJ, AEA, HA, and YS participated in the study concepts, design of the study, and development of the questionnaire. A data acquisition and entry. AEA, JH, MG, and MA contributed in data analysis and statistical analysis of the data, participated in the intellectual content, reviewed and summarized the published literature and clinical studies. DB, AA, KA, and DH participated in outlining the resulting themes and manuscript preparation, editing, and review. Corresponding author JH takes responsibility for the integrity of the work as a whole. All authors have critically reviewed and approved the final draft and are responsible for the content and similarity index of the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
We declare that the authors declare they have no conflict of interest.
Consent for Publication
All methods were carried out in accordance with relevant guidelines and regulations.
Ethics Approval
The study obtained ethical approval from Ministry of National Guard—Health Affairs, IRB, registered under number # RC15/058/. This study was completed prior to Dr. Anwar Ahmed joining the Uniformed Services University of the Health Sciences and Henry M Jackson Foundation for the Advancement of Military Medicine.
Patient Consent
Not applicable. This a char review study with no specific consent needed other than ethics approval.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Al-Jahdali, H., Ahmed, A.E., Abdullah, AH. et al. Comorbidities in Clinical and Polysomnographic Features of Obstructive Sleep Apnea: A Single Tertiary Care Center Experience. J Epidemiol Glob Health 12, 486–495 (2022). https://doi.org/10.1007/s44197-022-00067-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s44197-022-00067-z