Abstract
Background
Compared with other types of acute pancreatitis (AP), hypertriglyceridemic acute pancreatitis (HTG-AP) is younger, recurrent and more prone to exacerbation. Severe HTG-AP has a high fatality rate. Early and accurate prediction of the severity is crucial. However, there is currently a lack of a specific scoring system for the severity of HTG-AP.
Aim/Purpose
To construct a risk prediction model that can accurately predict severe HTG-AP in the early stage and evaluate its clinical value.
Methods
The clinical data of 1768 patients with AP admitted to the Second Affiliated Hospital of Anhui Medical University from January 2020 to May 2023 were analyzed retrospectively, and 136 HTG-AP patients were finally selected. Univariate and multivariate analysis were performed for the early onset indicators to identify the independent risk factors for developing SAP in the patients of HTG-AP. Logistic regression was then utilized to establish a risk prediction model for the severity of HTG-AP, which was subsequently evaluated for its performance through discrimination and calibration analysis.
Results
Of the 136 patients with HTG-AP, 39 patients (28.7%) progressed to severe acute pancreatitis (SAP). Multivariate analysis revealed that CRP, RDW/SC, and D-dimer were independent risk factors for developing SAP in the patients of HTG-AP. The logistic regression analysis to establish prediction model was: Logit P = − 8.101 + 0.008 × CRP + 0.425 × D-dimer + 0.743 × RDW/SC. The receiver-operating characteristics (ROC) curve showed that area under curve (AUC) value of CRP, RDW/SC, D-dimer, and the prediction model were 0.831, 0.843, 0.874, and 0.915, respectively. Moreover, the AUC value of the prediction model and commonly used scoring systems of AP were compared: prediction model (AUC = 0.915) > Ranson (AUC = 0.900) > SOFA (AUC = 0.899) > CTSI (AUC = 0.889) > BISAP (AUC = 0.887).
Conclusion
CRP, RDW/SC and D-dimer were independent risk factors for SAP in the patients of HTG-AP. Compared with commonly used scoring systems of AP, the prediction model had good clinical prediction ability, providing reference for early identification of the patients developing severe HTG-AP and active intervention.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
1 Introduction
Acute pancreatitis (AP) is a common acute and severe diseases in clinical practice. With changing of dietary and lifestyle habits, hypertriglyceridemic acute pancreatitis (HTG-AP) has emerged as the second leading incidence type of AP with an increasing incidence rate. Compared to other types of AP, HTG-AP is characterized by being younger, greater tendency for recurrence, faster progression, and susceptibility to exacerbation [1, 2]. Accurately assessment of the severity and prognosis of HTG-AP in the early stages is crucial for developing treatment measures and reducing mortality rates. The commonly used scoring systems of AP including bedside index for severity of AP (BISAP), Ranson score, CT severity index (CTSI), sequential organ failure assessment (SOFA), and acute physiology and chronic health evaluation II (APACHE II), have limited in clinical use due to various constraints [3,4,5,6]. Furthermore, there is a deficiency of specific scoring systems for the severity of HTG-AP. Therefore, it is urgent to find effictive indicators that are simple to operate, easy to obtain, better alternative to various scoring systems, and can accurately predict the severity of HTG-AP. The aims of this study are:(1) to construct a prediction model by screening risk factors for the severity of HTG-AP in the early stage, and evaluate the model performance by discrimination and calibration analysis, (2) to assess prediction model ability in predicting the severity of HTG-AP, and (3) to compare the predictive ability of prediction model with other serum markers and commonly used scoring systems of AP.
2 Materials and Methods
2.1 Study Patients
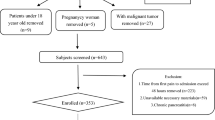
This study included 1768 patients with AP admitted to the Second Affiliated Hospital of Anhui Medical University from January 2020 to May 2023. Among them, 169 patients with HTG-AP were screened out. Inclusion criteria: ① Meeting the diagnostic criteria for AP [7], ② Having a serum TG level of 11.3 mmol/L, or a TG level ranging from 5.65 to 11.3 mmol/L but with chylous serum, ③ Age over 18 years old. Exclusion criteria: ① Pancreatitis caused by other types, ② Chronic pancreatitis attack, ③ Minors (those under the age of 18), ④ Malignant tumors, autoimmune diseases, and chronic organ failure. 136 patients with HTG-AP were ultimately included as the study subjects. The severity of HTG-AP was stratified according to the 2012 revision of the Atlanta classification and definitions by the international consensus [7]. They were divided into severe acute pancreatitis (SAP) group and non-severe acute pancreatitis (NSAP) group. The inclusion and exclusion process diagram are shown in Fig. 1.
2.2 Clinical Data
The clinical data were collected at admission, including gender, age, height, weight, basic diseases (diabetes, hypertension, fatty liver, prior history of pancreatitis, ect.), vital signs [heart rate (HR), systolic blood pressure (SBP), diastolic blood pressure (DBP), mean arterial pressure (MAP), respiration (RR), temperature], scoring systems (SOFA, BISAP, Ranson, CTSI, ect.), Laboratory indicators[white blood cell (WBC), neutrophil (N), hemoglobin (Hb), platelet (PLT), red cell distribution width (RDW), hematocrit (HCT), prothrombin time (PT), activated partial thromboplastin time (APTT), D-dimer, albumin (ALB), alanine aminotransferase (ALT), aspartate aminotransferase (AST), lactate dehydrogenase (LDH), creatinine (Scr), urea nitrogen (BUN), uric acid (BUA), Cystatin C, fasting blood glucose (GLu), triglycerides (TG), total cholesterol (TC), high-density lipoprotein cholesterol (HDL), low-density lipoprotein cholesterol (LDL), serum amylase (AMY), lipase (LIP), serum calcium (Ca), C-reactive protein (CRP), etc. Calculation indicator: RDW/SC is the ratio of RDW to Ca; RDW/PLT is the ratio of RDW to PLT.
2.3 Statistical Analysis
The statistical software SPSS25.0 were used for data analysis. The continuous variables are represented by a normal distribution using mean ± standard deviation, and independent sample t-tests are used for intergroup comparisons. The skewed distributions are represented by the median (quartile), and the M-U test is used for intergroup comparisons. The categorical variables are expressed as frequency and percentage, and intergroup comparisons are conducted using chi-square test. Determine independent risk factors for SAP through multivariate logistic regression,and construct a risk prediction model for the severity of HTG-AP based on the risk composition ratio. The calibration of the prediction model was tested for the Hosmer Limeshow goodness of fit test. Analysis of the receiver operating characteristic (ROC) curves and the area under the ROC curves (AUC) were performed to compare the prediction model and other single indicators for predicting the severity of patients with HTG-AP. In addition, we compared the ROC curves between the prediction model and commonly used scoring systems. P-value < 0.05 indicates statistically significant differences.
3 Results
3.1 Clinical Features
A total of 136 patients with HTG-AP were enrolled finally in this study, including 96 males (70.6%) and 40 females (29.4%). The average age of onset for the patients was 36 years. Among them, 39 patients (28.7%) were classified as SAP and 97 (71.3%) as NSAP. There was no significant difference in gender, age, height, weight, hypertension, diabetes and fatty liver between SAP group and NSAP group (P > 0.05). In comparison on the NSAP group, the patients in SAP group had higher levels of HR, RR, GLu, BUN, Scr, LDH, CRP, D-dimer, RDW/SC. Conversely, the patients in SAP group had lower levels of DBP, MAP, HCT, Ca, ALB, and HDL than in NSAP group (P < 0.05) (Table 1). There were no statistically significant difference in any other factors between the two groups (P > 0.05) (Table 1).
3.2 Constructing of Prediction Model
After conducting univariate analysis, meaningful indicators underwent multicollinearity tests. The indicators with VIFs < 10 were included as independent variables for the multivariate logistic regression analysis. Multivariate analysis showed that CRP, RDW/SC, and D-dimer were independent risk factors for predicting SAP in patients with HTG-AP (Table 2). Using a logistic regression model to ultimately incorporate CRP, RDW/SC, and D-dimer to construct an early risk prediction model for the severity of HTG-AP. The resulting regression equation is: Logit P = -8.101 + 0.008 × CRP + 0.425 × D-dimer + 0.743 × RDW/SC. The likelihood ratio of model coefficient indicates that the model construction is statistically significant (χ2 = 68.675, P < 0.05).
3.3 Performance Evaluation of Predictive Model
ROC curves were constructed to evaluate the ability of each markers to predict the severity of HTG-AP, the AUC value of CRP, RDW/SC, D-dimer, and the prediction model were 0.831, 0.843, 0.874, and 0.915, respectively (Fig. 2). The results indicate that the prediction model has better predictive performance and higher accuracy compared to single indicators, with the AUC value highest for the prediction model, followed by D-dimer, RDW/SC, and CRP. The sensitivity and specificity of prediction model were 91.9% and 79.1%, respectively. The maximum Jordan index calculated was 0.71 and the optimal cut-off value was 0.20 to distinguish between severe and non- severe patients with HTG-AP. When Logit P > 0.20, patients with HTG-AP had a higher risk of developing SAP. Hosmer Limeshow goodness of fit test showed that the fit and calibration of model are good (χ2 = 4.760, P = 0.783 > 0.05) (Fig. 3). The above results demonstrate that the prediction model has good discrimination and calibration in accurately predicting the severity of HTG-AP.
3.4 Predictive Ability of Predictive Model
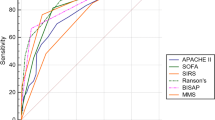
The predictive ability was evaluated by comparing the prediction model with the commonly usely scoring systems of AP by using ROC curve. The AUC value of the prediction model was 0.915 (95% CI: 0.865 ~ 0.965), whereas the other scoring systems (SOFA, Ranson, CTSI, and BISAP) had AUC value of 0.899, 0.900, 0.889, and 0.887, respectively (P < 0.05)(Fig. 4). The prediction model had the highest accuracy for predicting the severity of HTG-AP. The sensitivity and specificity were 91.9% and 79.1% for prediction model, 83.8% and 82.6% for SOFA, 89.2% and 82.6% for CTSI, 94.6% and 75.6% for BISAP, and 83.8% and 82.6% for Ranson (Table 3). The above results demonstrate that the predictive model had the better predictive ability compared with other scoring systems.
4 Discussion
HTG-AP has become the second common type of AP after biliary pancreatitis, and is now recognized as the primary etiologic factor in young and pregnant patients [8,9,10,11]. Compared to other types of AP, HTG-AP is characterized by rapid progression, high recurrence rate, and susceptibility to SAP. The mortality rate of patients who progress to SAP is as high as 30–40% [12]. SAP may cause prolonged hospital stays, extended ICU stays, and high hospitalization costs for patients. Finally, it has imposed a tremendous burden on patients' physical, mental, and economy. Therefore, as an extremely dangerous AP, how to stratify the severity of the disease has been a popular area of HTG-AP. Early identification of severe patients with HTG-AP, precise evaluation of risks and timely implementation of intervention measures are very importance.
Currently, there is no specific scoring system to evaluate the severity of HTG-AP, physicians still use the AP scoring system for stratification. Common scoring systems of AP include BISAP, Ranson, CTSI, SOFA, and APACHE II, ect. Numerous studies indicate that no single scoring system can accurately predict organ damage, severity and mortality of AP, it is recommended to use multiple scoring systems for comprehensive evaluation [3,4,5,6]. Furthermore, the above scoring systems have several shortcomings, such as lack of specificity, multiple parameters, difficulty in quickly obtaining part parameters, strong subjectivity, poor timeliness, limited value in dynamic assessment and poor clinical operability, which have not been widely applied in clinical practice. Therefore, there is an urgent need to investigate a fast, operable, uncomplicated, pragmatic, sensitive, and specific assessment method to predict the severity of HTG-AP in the early stage. This study collected clinical data from 136 patients with HTG-AP. Multivariate analysis indicated that CRP, D-dimer, and RDW/SC were independent risk factors for SAP in patients with HTG-AP. These indicators have been used to construct a risk prediction model to identify early exacerbation of HTG-AP. Clinical applicability of the predictive model was validated by comparing with commonly used scoring systems of AP.
The relationship between various influencing factors and SAP can be further elaborated as follows:
-
1.
Serum CRP level: The level of CRP, which is a common non-specific inflammatory marker, reflects the degree of inflammatory response in the body. Previous studies have indicated that CRP is a useful indicator in evaluating the prognosis and severity of AP and HTG-AP [13, 14]. The level of CRP is positively correlated with the severity of the disease. The higher peak of CRP or the slower decline rate always indicate that patients have a severe condition and worse prognosis. Dynamic monitoring of CRP in the early stages can assist in assessing the severity of the disease. Our study confirms the same results.
-
2.
Serum D-dimer level: Patients with AP may develop coagulation dysfunction. The reason is that the systemic inflammatory response causes an imbalance in the anticoagulant and fibrinolytic systems, leading to thrombosis. This coagulation dysfunction is particularly evident in SAP patients. The value of D-dimer in evaluating coagulation dysfunction has been widely confirmed. High level of D-dimer is associated with the severity and mortality of AP [15]. A study found that the D-dimer level can predict whether patients with HTG-AP will have acute renal [16]. Patients with high level of D-dimer have an increased risk of developing SAP in the early stage, which has a poorer prognosis [17]. Our study aligns with these results.
-
3.
RDW/SC: RDW is a common parameter used in blood test to reflect the variation of red blood cell volume. In recent years, RDW has gained attention due to its easy acquisition, objectivity, and simplicity. Numerous studies have elucidated the relationship between RDW and the prognosis of various diseases, such as sepsis, cerebrovascular diseases, solid malignant tumors, hematological disorders, venous thrombosis, ect [18,19,20,21,22,23,24,25,26]. This indicates that RDW can serve as an effective predictive indicator for diseases. RDW has been confirmed as a risk factor for the severity of AP [27,28,29]. According to the research by YAO et al. [25], comparing RDW of AP patients with different severity levels, it was found that RDW of SAP patients was significantly higher than that of NSAP patients. The ROC curve showed that the AUC value of RDW in predicting mortality was 0.846, with the sensitivity and specificity of 75.0% and 89.8%, respectively. Gravito et al. [26] also found that RDW had a strong reliability in predicting the severity and mortality in patients with AP, superior to traditional scoring systems such as Ranson, BISAP, and Marshall. The cut-off values of RDW for predicting severity were 13. If RDW is greater than 13, the progression to SAP became more likely. The reduction of serum Ca is one of the classic indicators for clinical evaluation of SAP. Combing RDW and SC for predictive analysis, that study revealed that RDW/SC can also be used to evaluate severity and mortality of AP. The AUC value of RDW/SC is higher than that of individual indicators and traditional scoring systems, indicating that RDW/SC had greater advantages in predicting the severity of SAP in the early stage. Domestic scholars have also reached similar conclusions. However, there is no research on RDW/SC predicting the severity of HTG-AP both domestically and internationally. Our study found that the AUC value of RDW/SC for predicting the severity of HTG-AP was 0.843, with the sensitivity of 91.9%, and specificity of 79.1%. RDW/SC may serve as a reliable indicator for evaluating the severity of HTG-AP.
To construct a risk prediction model for the severity of HTG-AP in the early stage, three independent risk factors were selected. Differentiation and calibration analysis confirmed that the prediction model has good predictive performance and high consistency with real data. The prediction model exhibits great clinical applicability. Meanwhile, to evaluate the prediction efficacy of the model, the AUC value of prediction model were compared to commonly used scoring systems of AP by ROC curve. Among them, APACHE II, a commonly used scoring system, was limited by various factors. Specifically, APACHE II requires numerous indicators, many of which are challenging to obtain and have strong subjectivity. All of these factors prevent APACHE II from being widely applied, so it was not included in this study. The AUC value of various scoring systems were compared: the prediction model > Ranson > SOFA > CTSI > BISAP. The prediction model has better predictive ability compared to other scoring systems. Finally, the prediction model has a higher specificity in assessing the severity of HTG-AP, and indicators of predictive model are easily to obtain and objective. Based on these factors, this study concludes that predictive model has greater advantages in clinical applications. However, it is worth noting that this study also has many limitations. It remains a single center retrospective study. Additionally, the sample size used in the study was relatively small, and external validation data were lacking. In the future, we plan to collect larger sample sizes from multiple centers to validate the prediction model. The ultimate goal is to implement it in clinical practice.
5 Conclusion
In summary, CRP, D-dimer and RDW/SC are independent risk factors for SAP in patients with HTG-AP. The establishment of prediction model can help identify patients at a high risk of exacerbation of HTG-AP in the early stages. The prediction model exhibits good discrimination, calibration and clinical applicability. Overall, this prediction model will greatly assist clinical physicians in conducting accurate assessment and ensuring prompt and effective intervention measures for patients with HTG-AP.
Data availability
All data generated or analysed during this study are included in this published article.
References
Vipperla K, Somerville C, Furlan A, et al. Clinical profile and natural course in a large cohort of patients with hypertriglyceridemia and pancreatitis [J]. J Clin Gastroenterol. 2017;51(1):77–85.
Incidence and clinical features of hyperlipidemic acute pancreatitis from Guangdong, China: a retrospective multicenter study.
Mounzer R, Langmead CJ, Wu BU, et al. Comparison of existing clinical scoring systems to predict persistent organ failure in patients with acute pancreatitis [J ]. Gastroenterology. 2012;142(7):1476–82.
Qing W, Jie W, Mengbin Q, et al. Accuracy of conventional and novel scoring systems in predicting severity and outcomes of acute pancreatitis: a retrospective study. Lipids Health Dis. 2021;20:41.
Piero A, Elizabeth P, Rodrigo M, et al. Evaluation of the modified computed tomography severity index (MCTSI) and computed tomography severity index (CTSI) in predicting severity and clinical outcomes in acute pancreatitis. J Dig Dis. 2021;22:41–8.
Papachristou GI, Muddana V, Yadav D et al. Comparison of BISAP, Ranson's, APACHE-II, and CTSI scores in predicting organ failure, complications, and mortality in acute pancreatitis.[J] .Am J Gastroenterol, 2010, 105: 435–41; quiz 442.
Banks PA, Bollen TL, Dervenis C et al. Classification of acute pancreatitis--2012: revision of the Atlanta classification and definitions by international consensus[J] .Gut. 2013;62 (1):102–111.
Petrov MS, Yadav D. Global epidemiology and holistic prevention of pancreatitis [J]. Nat Rev Gastroenterol Hepatol. 2019;16(3):175–84.
Perrone S, Brunelli R, Perrone G, et al. A successful term pregnancy with severe hypertriglyceridaemia and acute pancreatitis. Clinical management and review of the literature [J]. Atheroscler Suppl. 2019;40:117–21.
Russi G. Severe dyslipidemia in pregnancy: The role of therapeutic apheresis [J]. Transfus Apher Sci. 2015;53(3):283–7.
Valdivielso P, Alba R-B, Ewald N. Current knowledge of hypertriglyceridemic pancreatitis[J]. Eur J Int Med. 2014. https://doi.org/10.1016/j.ejim.2014.08.008
Párniczky A, Kui B, Szentesi A, et al. Prospective, Multicentre, Nationwide Clinical Data from 600 Cases of Acute Pancreatitis[J]. PLoS ONE. 2016;11(10): e0165309.
Rau B, Steinbach G, Baumgart K, et al. Serum amyloid A versus C-reactive protein in acute pancreatitis: clinical value of an alternative acute-phase reactant[J]. Crit Care Med. 2000;28(3):736–42.
Staubli SM, Oertli D, Nebiker CA. Laboratory markers predicting severity of acute pancreatitis[J]. Crit Rev Clin Lab Sci. 2015;52(6):273–83.
Jianhua W, Xiaoyu Y, Wenhua H, et al. Serum D-dimer levels at admission for prediction of outcomes in acute pancreatitis [J]. BMC Gastroenterol. 2019;19:67.
Lin Y, Yu S, Wu X et al. Clinical analysis of the therapeutic effect of plasma exchange on hypertriglyceridemic acute pancreatitis: a retrospective study[J]. Transfusion, 2021.
Wan J, Yang X, He W, et al. Serum D-dimer levels at admission for prediction of outcomes in acute pancreatitis[J]. BMC Gastroenterol. 2019;19(1):67.
Ku NS, Kim HW, Oh HJ, et al. Red blood cell distribution width is an independent predictor of mortality in patients with gram-negative bacteremia[J]. Shock. 2012;38(2):123–7.
Kim CH, Park JT, Kim EJ, et al. An increase in red blood cell distribution width from baseline predicts mortality in patients with severe sepsis or septic shock[J]. Crit Care. 2013;17(6):R282.
Söderholm M, Borné Y, Hedblad B, et al. Red cell distribution width in relation to incidence of stroke and carotid atherosclerosis: a population-based cohort study[J]. PLoS ONE. 2015;10(5): e0124957.
ParizadehSeyed M, Jafarzadeh-Esfehani R, Bahreyni A, et al. The diagnostic and prognostic value of red cell distribution width in cardiovascular disease; current status and prospective [J]. BioFactors. 2019;45:507–16.
Dan L, Li Bing X, Zefeng, et al. RBC distribution width predicts thrombosis risk in polycythemia vera [J]. Leukemia. 2022;36:566–8.
Shuai L, Liuyuan Z, Guohua Z. Peripheral blood markers predict immunotherapeutic efficacy in patients with advanced non-small cell lung cancer: a multicenter study [J]. Front Genet. 2022;13:1016085.
Herraez I, Bento L, Del Campo R et al. Prognostic Role of the Red Blood Cell Distribution Width (RDW) in Hodgkin Lymphoma. Cancers (Basel), 2020, 12: undefined.
Yalun L, Chengzhong X, Minjie W, et al. Combining Red Blood Cell Distribution Width (RDW-CV) and CEA Predict Poor Prognosis for Survival Outcomes in Colorectal Cancer. J Cancer. 2019;10:1162–70.
Rezende SM, Lijfering WM, Rosendaal FR et al. Hematologic variables and venous thrombosis: red cell distribution width and blood monocyte count are associated with an increased risk.[J] .Haematologica 2014, 99: 194–200.
Yao J, Lv G. Association between red cell distribution width and acute pancreatitis: a cross-sectional study[J]. BMJ Open. 2014;4(8):0047 21.
Gravito-Soares M, Gravito-Soares E, Gomes D, et al. Red cell distribution width and red cell distribution width to total serum calcium ratio as major predictors of severity and mortality in acute pancreatitis[J]. BMC Gastroenterol. 2018;18(1):108.
Zhang F-X, Li Z-L, Zhang Z-D et al. Prognostic value of red blood cell distribution width for severe acute pancreatitis.[J] .World J Gastroenterol, 2019, 25: 4739–4748
Acknowledgements
The authors did not receive support from anyone who contributed to the article who does not meet the criteria for author ship including anyone who provided professional writing services or materials. We thank all researchers, patients, and surrogates involved in the individual trials.
Funding
This work is supported by the National Natural Science Foundation of China (NO.82072134) and Anhui Medical University Science Fund Project (2022xkj042).
Author information
Authors and Affiliations
Contributions
Juanjuan Hu, Yuansong Sun and Min Yang contributed to the study conception and design. Juanjuan Hu gathered the data and drafted the manuscript. Wenyan Xiao and Tianfeng Hua revised the manuscript for important intellectual content. The authors critically reviewed the fnal version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare that they have no conficts of interest.
Ethical Approval
This study was approved by the Ethics Committee of the Second Affiliated Hospital of Anhui Medical University (Approval No. YX2023-062).
Consent to Participate
Consent to participate was waived because of the retrospective design of the study.
Consent for Publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hu, J., Sun, Y., Hua, T. et al. Establishment of Prediction Model for Severe Hypertriglyceridemic Acute Pancreatitis Based on Early Laboratory Indicators. Intensive Care Res 4, 91–98 (2024). https://doi.org/10.1007/s44231-024-00061-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s44231-024-00061-y