Abstract
Taurine is a conditionally essential amino acid that has been reported to have antioxidative and immunomodulatory effects either directly or through its interactions with gut microbiome. Although taurine has been widely used as a health supplement and also for treatment of congestive heart failure in Japan, its roles in the perioperative setting have not been well characterized. The purpose of this systematic review and meta-analysis was to assess whether oral or intravenous taurine, or its derivatives such as taurolidine, can offer benefits compared to placebo in the perioperative setting. Non-randomized studies, trials assessed topical taurine or taurolidine, or trials did not report clinical outcomes were excluded. Six randomized-controlled-trials (RCTs) involving a total of 596 adult surgical patients, in PubMed, MEDLINE and EMBASE databases on September 14, 2023 were identified and meta-analyzed using a fixed-effect model. Biases were assessed by reporting individual trial elements. Taurine or taurolidine (which is readily metabolized by hydrolysis to taurine) supplementation was associated with a reduction in all-cause infection (5 trials: pooled odds ratio (OR) 0.59; 95% confidence interval (CI) 0.38–0.92; p = 0.020) but not delirium (2 trials: OR 0.63, 95% CI 0.38–1.04; p = 0.071) or mortality (4 trials: OR 0.82, 95% CI 0.43–1.58; p = 0.557) compared to placebo. Using the ‘trim and fill’ technique to adjust for publication bias did not change the favorable effect of taurine on risk of infection (OR 0.61, 95% CI 0.40–0.95). Because the number of studies included was small, the promising benefits of taurine on risks of perioperative infection should be confirmed by adequately-powered RCTs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Taurine (2-aminoethane-sulfonic acid) is a unique amino acid in many ways. It is considered as a conditionally essential amino acid because it plays an important part in the development of the human infant brain when they are entirely dependent on dietary taurine intake with limited endogenous synthesis during that period of life [1]. Different from other amino acids, taurine is not used for protein synthesis or energy generation in humans.
A recent study showed that anesthesia and surgery can induce endogenous synthesis of taurine resulting in an increase in plasma concentrations within the first week after surgery [2]. Conversely, taurine is depleted in patients with septic shock [3]. Whether the pathophysiological changes in blood taurine concentration are a bystander phenomenon or an adaptive response to stress that can be overwhelmed, as in septic shock, are unknown. In an animal study, taurine pretreatment could partially prevent interleukin-6 production induced by acute pneumonia in vivo [4], suggesting that exogenous taurine supplementation could be useful as an anti-inflammatory or anti-infective agent in sepsis.
Mechanistically, taurine is believed to be able to regulate intracellular calcium homeostasis, work as an antioxidant that can optimize mitochondrial functions in particular the complex I reaction, and reduce endoplasmic reticulum stress [1]. Through these cellular level actions, taurine has been reported to improve myocardial, circulatory, metabolic and cerebral functions.
It has been argued that there are significant differences in the physiological and pharmacological actions of taurine, with the beneficial effects of taurine can only be realized and demonstrated when sufficiently high taurine concentrations are achieved beyond the levels that can be obtained through a standard Western diet (of < 400 mg/day) [5]. Because taurine is inexpensive and generally considered as safe when taken up to 6 g per day for adults [6], using taurine supplementation as a perioperative adjunct may have a huge potential in improving surgical outcomes.
We hypothesized that taurine supplementation could be a useful adjunct in reducing risk of postoperative infections, cardiac arrhythmias and delirium for surgical patients. In this meta-analysis, we summarized the current evidence on the potential benefits of supplementation of taurine or its derivatives for patents in the perioperative setting.
2 Methods
2.1 Study identification, and inclusion and exclusion criteria
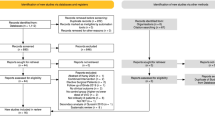
We used the PRISMA checklist when writing this report [7]. The following keywords or subject headings were used to search for RCTs using the PubMed, MEDLINE and EMBASE databases on September 14, 2023: taurine or taurolidine and surgery or perioperative and clinical trials or RCTs. There was no language restriction in study inclusion but non-human studies and studies involving children (< 18 years old) were excluded. The protocol for this systematic review and meta-analysis was registered on INPLASY (https://inplasy.com/) and is available in full on the https://inplasy.com/inplasy-2024-1-0071/. Only studies using oral or intravenous taurine or its derivatives such as taurolidine (which has a short half-life and is rapidly metabolized by hydrolysis into taurine, water and carbon dioxide) [8] supplementation were included. Studies asessing topical or intraperitoneal taurine or taurolidine (n = 2) or those comparing two interventions simultaneously (e.g., taurine with Coenzyme Q) against placebo (n = 1) were excluded. Of the 81 studies identified, only six RCTs met our inclusion criteria and were subject to meta-analysis [9,10,11,12,13,14]. Two reviewers independently extracted the data from the included studies and studies with missing data on all outcomes of interest were excluded.
2.2 Outcomes of the meta-analysis
The primary outcome of this meta-analysis was risk of all-cause infections after surgery. The other clinical outcomes assessed included delirium and hospital mortality. We used a fixed-effect model to pool the ORs of the outcomes between placebo and intervention groups because of the small number of studies included. Similarly, the I2 was not used to quantify heterogeneity in this meta-analysis due to limited statistical power of I2 when the number of included studies is small.
2.3 Sensitivity analysis and bias assessment
We conducted two sensitivity analyses to confirm whether publication bias and one aspect of the quality of the trial would affect the results of the primary outcome. First, we used the ‘trim and fill’ method to adjust for effect of any publication bias due to small studies with outlying results. Second, we excluded RCTs that were not prospectively registered in a clinical trial registry to assess whether this would affect the results of the primary outcome. The other aspects of study quality, such as blinding, allocation concealment, deviation from intended intervention, and missing data, were described individually without being summarized by an overall quality score. All analyses were conducted by the Comprehensive Meta-analysis software (version 2.2.034, 2006; Biostat, Englewood, USA) and a p-value < 0.05 was taken as significant.
3 Results
3.1 Study characteristics
A total of 596 adult surgical patients from six RCTs conducted in the perioperative setting were identified and meta-analyzed. Two RCTs were conducted on cardiac surgical patients [13, 14], one on colonic cancer surgery [11], one on liver transplantation [10], one on hip fractures requiring arthroplasty [9] and one on surgical patients who required at least five days of parenteral nutrition [12]. Five RCTs were double-blinded and four were registered prospectively in a clinical trial registry. No major adverse events from taurine or taurolidine supplementation was reported in these six RCTs. The details of each study were extracted by two investigators independently and are described in Table 1.
3.2 Outcomes after taurine or taurolidine supplementation compared to placebo
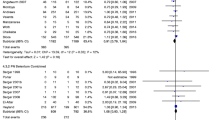
Taurine or taurolidine supplementation was associated with a reduction in all-cause infections (5 trials: OR 0.59; 95% CI 0.38–0.92; p = 0.020) (Fig. 1) but not delirium (2 trials: OR 0.63, 95% CI 0.38–1.04; p = 0.071) (Fig. 2) or mortality (4 trials: OR 0.82, 95% CI 0.43–1.58; p = 0.557) compared to placebo.
3.3 Sensitivity analyses
Using the ‘trim and fill’ technique to adjust for publication bias did not change the favorable effect of taurine on risk of infections (OR 0.61, 95% CI 0.40–0.95), suggesting that publication bias was not a major factor in explaining our results. Excluding an older study that was not prospectively registered in a clinical trial registry [12] also did not change the effect of taurine or taurolidine on risk of infections (OR 0.61, 95% CI 0.39–0.96; p = 0.031).
4 Discussion
This meta-analysis showed that oral or intravenous taurine or taurolidine supplementation in the surgical setting appeared to be well-tolerated, and was associated with a significant reduction in risk of all-cause infections compared to placebo. This result has some important research implications and requires further discussion.
First, to the best of our knowledge, this is the first meta-analysis assessing the effects of taurine or taurolidine supplementation in the perioperative setting with a special emphasis on testing the hypothesis that taurine could reduce infections in surgical patients. Taurine is present in high concentrations (up to 50 mM) in neutrophils and is needed to produce taurine chloramine (N-chlorotaurine) which is an important regulator of the immune system, especially in optimizing survival and antimicrobial activity of leuokocytes [15,16,17,18,19]. The effect of taurine or taurolidine on infections reported in this meta-analysis was consistent with the favorable immunomodulatory effects of taurine or taurolidine reported in a number of experimental studies [20, 21]. For instance, recent experimental studies showed that exogenous taurine supplementation alone is sufficient to induce alterations in gut microbiota, enhance resistance to infection, boost intestinal immunity in response to antibiotic exposure, resist colonization by Citrobacter rodentium, and increase the diversity of gut microflora during infection [21, 22]. It has also been shown that when taurine is released from bacterial deconjugation of taurine-conjugated bile acids, it can improve intestinal tight junction function [23], resulting in reduced gut permeability and risk of bacterial translocation. Because gut microbiota may play an important role in determining many important outcomes of surgical patients [24], the potential favorable effect of taurine on risk of surgical infections deserves confirmation by well-controlled RCTs.
It has been hypothesized that taurine may enhance survival of various advanced living organisms (that have no cell wall) including humans when they face adverse environmental challenges [25]. Major surgery induces substantial physiological stress to the human body; and for those who are already vulnerable with a limited physiological reserve before surgery, complications are common [26]. Based on the data from our current analysis, an 80%-powered RCT would need approximately 5800 subjects (in total) to reliably detect a 2% absolute risk reduction in surgical infections, if we assume the baseline infection risk of the control group is 9% [27]. Because taurine is inexpensive and safe as a supplement at a dose ≤ 6 g per day for an adult [6, 28], with the promising results of the current meta-analysis, a large RCT to confirm the potential benefits of perioperative taurine supplementation in reducing surgical infections would appear to be feasible and highly desirable. Apart from infection, other important patient-centered outcomes, such as delirium [29] and severe hyperglycemia, should also be carefully considered either as secondary outcomes in the same trial when infection is investigated or in separate trials targeting patients who are most vulnerable in developing these complications.
Second, taurine is licenced to be used for patients with congestive heart failure in Japan and current evidence suggests that taurine supplementation may offer long-term metabolic and cardiovascular benefits [28]. As such, it would be important to meta-analyze cardiovascular outcomes if we could get enough information from existing RCTs. Two trials included in this meta-analysis reported the effects of taurine or taurolidine on cardiac arrhythmias [13, 14]. At the end, we decided not to pool the outcomes of these two trials together because of heterogeneity in the formulations of study intervention and the duration of observation for arrhythmias after administering the study intervention. Previous animal and small human clinical studies showed that taurine could improve heart rate variability, reduce QT interval, and increase myocardial contractility [30,31,32,33,34]. The effects of taurine on cardaic arrhythmias in animal studies are, however, conflicting with at least one study showing that high-doses of taurine can induce cardiac arrhythmias [35, 36]. The underlying mechanism may be related to shortening of cardiac repolarizations and effective refractory periods, and this could induce re-entry arrhythmias [36]. On the other hand, one human study had demonstrated a substantial dose-related relationship between plasma taurine and hydrogen sulfide (H2S) levels [37]. Because H2S is an important signalling molecule and it has been suggested that having sufficient availability of H2S may confer cardiovascular protective effects especially against cardiac arrhythmias [38, 39], it is still possible for taurine — when taken at a non-toxic dose — to be anti-arrhythmogenic in humans. Nonetheless, given cases of sudden death after ingestion of large quantity of taurine-containing energy drinks have been reported [40], it would be prudent to assess cardiovascular events as a safety outcome in RCTs assessing perioperative benefits of taurine especially in cardiac surgical patients.
Finally, we need to acknowledge the limitations of this meta-analysis. This meta-analysis was limited by heterogeneity of the study interventions in their dosage, route of administration, and also timing and duration of taurine supplementation in relation to the surgical operation. The number of RCTs reporting clinical outcomes was also small. Furthermore, none of the included studies reported the duration and type of antibiotics used during the perioperative period. Infection was not the primary endpoint of these studies; it is probable that the use of antibiotics would not be significantly different between the two treatment groups within each RCT as both groups should have been treated similarly in terms of perioperative prophylactic antibiotics. That said, the favorable results of taurine or taurolidine on surgical infections should be treated as hypothesis-generating that requires further confirmation by an adequately-powered superiority RCT.
5 Conclusion
This meta-analysis showed that taurine or its derivative taurolidine supplementation was well-tolerated by surgical patients and their use was associated with a reduced risk of all-cause infections. More than one adequately-powered RCTs are likely needed to confirm (or refute) the promising benefits of perioperative taurine supplementation in surgical patients, as well as to identify the most appropriate dose and time to administer taurine in relation to surgery if it is proven to be effective.
Availability of data and materials
Data will be available upon receiving reasonable requests.
Abbreviations
- CI:
-
Confidence interval
- OR:
-
Odds ratio
- RCT:
-
Randomized controlled trials
References
Jong CJ, Sandal P, Schaffer SW. The role of taurine in mitochondria health: more than just an antioxidant. Molecules. 2021;26(16):4913. https://doi.org/10.3390/molecules26164913.
Mock-Ohnesorge J, Mock A, Hackert T, Fröhling S, Schenz J, Poschet G, et al. Perioperative changes in the plasma metabolome of patients receiving general anesthesia for pancreatic cancer surgery. Oncotarget. 2021;12(10):996–1010. https://doi.org/10.18632/oncotarget.27956.
Vermeulen MA, van Stijn MF, Visser M, Lemmens SM, Houdijk AP, van Leeuwen PA, et al. Taurine concentrations decrease in critically Ill patients with shock given enteral nutrition. JPEN J Parenter Enteral Nutr. 2016;40(2):264–72. https://doi.org/10.1177/0148607114567199.
Nguyen KH, Murakami S, Schaffer SW, Ito T. Examination of taurine chloramine and taurine on LPS-induced acute pulmonary inflammatory in mice. Adv Exp Med Biol. 2022;1370:23–9. https://doi.org/10.1007/978-3-030-93337-1_2.
Schaffer SW, Jong CJ, Ramila KC, Ito T, Kramer J. Differences between physiological and pharmacological actions of taurine. Adv Exp Med Biol. 2022;1370:311–21. https://doi.org/10.1007/978-3-030-93337-1_30.
EFSA Panel on Additives and Products or Substances used in Animal Feed (FEEDAP). Scientific Opinion on the safety and efficacy of taurine as a feed additive for all animal species. EFSA J. 2012;10(6):2736. https://doi.org/10.2903/j.efsa.2012.2736.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. J Clin Epidemiol. 2021;134:178–89. https://doi.org/10.1016/j.jclinepi.2021.03.001.
Schuller-Levis GB, Park E. Taurine: new implications for an old amino acid. FEMS Microbiol Lett. 2003;226(2):195–202. https://doi.org/10.1016/s0378-1097(03)00611-6.
Van Stijn MF, Bruins AA, Vermeulen MA, Witlox J, Teerlink T, Schoorl MG, et al. Effect of oral taurine on morbidity and mortality in elderly hip fracture patients: a randomized trial. Int J Mol Sci. 2015;16(6):12288–306. https://doi.org/10.3390/ijms160612288.
Mottaghi S, Nikoupour H, Firoozifar M, Jalali SS, Jamshidzadeh A, Vazin A, et al. The effect of taurine supplementation on delirium post liver transplantation: a randomized controlled trial. Clin Nutr. 2022;41(10):2211–8. https://doi.org/10.1016/j.clnu.2022.07.042.
Redmond HP, Neary PM, Jinih M, O’Connell E, Foley N, Pfirrmann RW, et al. RandomiSed clinical trial assessing use of an anti-inflammatoRy aGent in attenUating peri-operatiVe inflAmmatioN in non-meTastatic colon cancer – the S.U.R.G.U.V.A.N.T. trial. BMC Cancer. 2018;18(1):794. https://doi.org/10.1186/s12885-018-4641-x.
Arrieta F, Balsa JA, de la Puerta C, Botella JI, Zamarrón I, Elías E, et al. Phase IV prospective clinical study to evaluate the effect of taurine on liver function in postsurgical adult patients requiring parenteral nutrition. Nutr Clin Pract. 2014;29(5):672–80. https://doi.org/10.1177/0884533614533610.
Doddakula KK, Neary PM, Wang JH, Sookhai S, O’Donnell A, Aherne T, et al. The antiendotoxin agent taurolidine potentially reduces ischemia/reperfusion injury through its metabolite taurine. Surgery. 2010;148(3):567–72. https://doi.org/10.1016/j.surg.2010.01.006.
Milei J, Ferreira R, Llesuy S, Forcada P, Covarrubias J, Boveris A. Reduction of reperfusion injury with preoperative rapid intravenous infusion of taurine during myocardial revascularization. Am Heart J. 1992;123(2):339–45. https://doi.org/10.1016/0002-8703(92)90644-b.
Iwegbulem O, Wang J, Pfirrmann RW, Redmond HP. The role of taurine derivatives in the putative therapy of COVID-19-induced inflammation. Ir J Med Sci. 2022;191(1):485–6. https://doi.org/10.1007/s11845-021-02522-5.
Anich C, Orth-Höller D, Lackner M, Nagl M. N-chlorotaurine, a potent weapon against multiresistant bacteria. J Appl Microbiol. 2021;131(4):1742–8. https://doi.org/10.1111/jam.15052.
Cunningham C, Tipton KF, Dixon HB. Conversion of taurine into N-chlorotaurine (taurine chloramine) and sulphoacetaldehyde in response to oxidative stress. Biochem J. 1998;330(2):939–45. https://doi.org/10.1042/bj3300939.
Schuller-Levis GB, Park E. Taurine and its chloramine: modulators of immunity. Neurochem Res. 2004;29(1):117–26. https://doi.org/10.1023/b:nere.0000010440.37629.17.
Nagl M, Hess MW, Pfaller K, Hengster P, Gottardi W. Bactericidal activity of micromolar N-chlorotaurine: evidence for its antimicrobial function in the human defense system. Antimicrob Agents Chemother. 2000;44(9):2507–13. https://doi.org/10.1128/AAC.44.9.2507-2513.2000.
Huang J, Ita M, Zhou H, Zhao H, Hassan F, Bai Z, et al. Autophagy induced by taurolidine protects against polymicrobial sepsis by promoting both host resistance and disease tolerance. Proc Natl Acad Sci U S A. 2022;119(19):e2121244119. https://doi.org/10.1073/pnas.2121244119.
Stacy A, Andrade-Oliveira V, McCulloch JA, Hild B, Oh JH, Perez-Chaparro PJ, et al. Infection trains the host for microbiota-enhanced resistance to pathogens. Cell. 2021;184(3):615-27.e17. https://doi.org/10.1016/j.cell.2020.12.011.
Qian W, Li M, Yu L, Tian F, Zhao J, Zhai Q. Effects of taurine on gut microbiota homeostasis: an evaluation based on two models of gut dysbiosis. Biomedicines. 2023;11(4):1048. https://doi.org/10.3390/biomedicines11041048.
Ahmadi S, Wang S, Nagpal R, Wang B, Jain S, Razazan A, et al. A human-origin probiotic cocktail ameliorates aging-related leaky gut and inflammation via modulating the microbiota/taurine/tight junction axis. JCI Insight. 2020;5(9):e132055. https://doi.org/10.1172/jci.insight.132055.
Ho KM, Kalgudi S, Corbett JM, Litton E. Gut microbiota in surgical and critically ill patients. Anaesth Intensive Care. 2020;48(3):179–95. https://doi.org/10.1177/0310057X20903732.
Huxtable RJ. Physiological actions of taurine. Physiol Rev. 1992;72(1):101–63. https://doi.org/10.1152/physrev.1992.72.1.101.
Bolshinsky V, Ismail H, Li M, Basto J, Schier R, Hagemeier A, et al. Clinical covariates that improve surgical risk prediction and guide targeted prehabilitation: an exploratory, retrospective cohort study of major colorectal cancer surgery patients evaluated with preoperative cardiopulmonary exercise testing. Perioper Med (Lond). 2022;11(1):20. https://doi.org/10.1186/s13741-022-00246-3.
Corcoran TB, Myles PS, Forbes AB, Cheng AC, Bach LA, O’Loughlin E, et al. for PADDI Investigators; Australian and New Zealand College of Anaesthetists Clinical Trials Network; Australasian Society for Infectious Diseases Clinical Research Network. Dexamethasone and surgical-site infection. N Engl J Med. 2021;384(18):1731–41. https://doi.org/10.1056/NEJMoa2028982.
Ho KM, Lee A, Wu W, Chan MTV, Ling L, Lipman J, et al. Flattening the biological age curve by improving metabolic health: to taurine or not to taurine, that’s the question. J Geriatr Cardiol. 2023;20(11):813–23. https://doi.org/10.26599/1671-5411.2023.11.004.
Zhang Y, Li D, Li H, Hou D, Hou J. Taurine pretreatment prevents isoflurane-induced cognitive impairment by inhibiting er stress-mediated activation of apoptosis pathways in the hippocampus in aged rats. Neurochem Res. 2016;41(10):2517–25. https://doi.org/10.1007/s11064-016-1963-4.
Li C, Zhou Y, Niu Y, He W, Wang X, Zhang X, et al. Deficiency of Pdk1 drives heart failure by impairing taurine homeostasis through Slc6a6. FASEB J. 2023;37(9):e23134. https://doi.org/10.1096/fj.202300272R.
Belikova J, Lizogub V, Kuzminets A, Lavrenchuk I. Normalization of heart rate variability with taurine and meldonium complex in post-infarction patients with type 2 diabetes mellitus. J Med Life. 2019;12(3):290–5. https://doi.org/10.25122/jml-2019-0052.
Ahmadian M, Dabidi Roshan V, Ashourpore E. Taurine supplementation improves functional capacity, myocardial oxygen consumption, and electrical activity in heart failure. J Diet Suppl. 2017;14(4):422–32. https://doi.org/10.1080/19390211.2016.1267059.
Azuma J, Sawamura A, Awata N. Usefulness of taurine in chronic congestive heart failure. Jpn Circ J. 1992;56:95–9. https://doi.org/10.1253/jcj.56.95.
Qaradakhi T, Gadanec LK, McSweeney KR, Abraham JR, Apostolopoulos V, Zulli A. The anti-inflammatory effect of taurine on cardiovascular disease. Nutrients. 2020;12(9):2847. https://doi.org/10.3390/nu12092847.
Yin Y, Wen K, Wu Y, Kang Y, Lou J. Inhibition of sodium current by taurine magnesium coordination compound prevents cesium chloride-induced arrhythmias. Biol Trace Elem Res. 2012;146(2):192–8. https://doi.org/10.1007/s12011-011-9240-5.
Ellermann C, Hakenes T, Wolfes J, Wegner FK, Willy K, Leitz P, et al. Cardiovascular risk of energy drinks: Caffeine and taurine facilitate ventricular arrhythmias in a sensitive whole-heart model. J Cardiovasc Electrophysiol. 2022;33(6):1290–7. https://doi.org/10.1111/jce.15458.
Sun Q, Wang B, Li Y, Sun F, Li P, Xia W, et al. Taurine supplementation lowers blood pressure and improves vascular function in prehypertension: randomized, double-blind placebo-controlled study. Hypertension. 2016;67(3):541–9. https://doi.org/10.1161/HYPERTENSIONAHA.115.06624.
Kolluru GK, Shackelford RE, Shen X, Dominic P, Kevil CG. Sulfide regulation of cardiovascular function in health and disease. Nat Rev Cardiol. 2023;20(2):109–25. https://doi.org/10.1038/s41569-022-00741-6.
Song Y, Xu Z, Zhong Q, Zhang R, Sun X, Chen G. Sulfur signaling pathway in cardiovascular disease. Front Pharmacol. 2023;14:1303465. https://doi.org/10.3389/fphar.2023.1303465.
Lévy S, Santini L, Capucci A, Oto A, Santomauro M, Riganti C, et al. European cardiac arrhythmia society statement on the cardiovascular events associated with the use or abuse of energy drinks. J Interv Card Electrophysiol. 2019;56(1):99–115. https://doi.org/10.1007/s10840-019-00610-2.
Acknowledgements
None.
Funding
This study was solely funded internally by the Department of Intensive Care Medicine, Fiona Stanley Hospital, Perth, Western Australia.
Author information
Authors and Affiliations
Contributions
Dr. Ho had the idea for the article and performed the literature search, extracted the data and analyzed the data. Ms. Harahsheh conducted data extraction independently and both authors drafted and critically revised the work and approved the final version of the manuscript. Large language models were not used in any parts of this study and manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
All authors gave their consent for publication.
Competing interests
The authors have no conflict of finanical or non-financial interest to declare in relation to the subject matter or drugs discussed in this study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ho, K.M., Harahsheh, Y. Perioperative taurine or taurolidine supplementation on clinical outcomes: A systematic review with meta-analysis. APS 2, 12 (2024). https://doi.org/10.1007/s44254-024-00055-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s44254-024-00055-5