Abstract
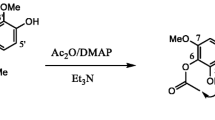
Eleven compounds, 12-oxo-phytodienoic acid (1), persicogenin (2), eriodictyol 3′, 4′, 7-trimethyl ether (3), phytol (4), spathulenol (5), 4-hydroxycinnamic acid (6), onopordin (7), 5, 8, 4′-trihydroxy-7, 3′-dimethoxyflavone (8), quercetin (9), jaceosidin (10), and 8-hydroxyluteolin (11), were isolated from an ethanol extract of Lantana balansae Briq., Verbenaceae, that was found to possess antileishmanial activity. The structures of the compounds were determined by NMR spectroscopy and HR mass spectrometry, and 1, 2, 3, 7, 8 and 9 were investigated for antiprotozoal activity toward promastigotes of Leishmania amazonensis and Leishmania braziliensis. Compound 1 was shown to be the most potent, with the IC50 values 2.0 μM toward L. amazonensis and 0.68 μM toward L. braziliensis, although less potent than the positive control Amphotericin B. All compounds have been reported previously, but this is the first report of the isolation of a cyclopentenone fatty acid (1) and flavanones (2 and 3) from a Lantana species.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Altiok, N., Mezzadra, H., Patel, P., Koyuturk, M., Altiok, S., 2008. A plant oxylipin, 12-oxo-phytodienoic acid, inhibits proliferation of human breast cancer cells by targeting cyclin Dl. Breast Cancer Res. Treat. 109, 315–323.
Blee, E., 1998. Phytooxylipins and plant defense reactions. Prog. Lipid Res. 37, 33–72.
Bohlmann, F., Jakupovic, J., Ahmed, M., Schuster, A., 1983. Sesquiterpene lactones and other constituents from Schistostephium species. Phytochemistry 22, 1623–1636.
Böttcher, C., Pollmann, S., 2009. Plant oxylipins: plant responses to 12-oxophytodienoic acid are governed by its specific structural and functional properties. FEBS J. 276, 4693–4704.
da Silva, E.R., Maquiaveli, C.G., Magalhães, P.P., 2012. The leishmanicidal flavonols quercetin and quercitrin target Leishmania (Leishmania) amazonensis arginase. Exp. Parasitol. 130, 183–188.
De Viana, M.E.L, Talenti, E.C.J., Retamar, J.A., 1973. Essential oils of Lontana balansae. Essenze Deriv. Agrum. 43, 299–306.
Fernandez, C., Fraga, B.M., Hernandez, M.G., Arteaga, J.M., 1988. Flavonoid aglycones from some Canary Islands species of Sideritis. J. Nat. Prod. 51, 591–593.
Gfeller, A., Dubugnon, L., Liechti, R., Farmer, E.E., 2010. Jasmonate biochemical path-way. Sci. Signal. 3, 1–7, https://doi.org/10.1126/scisignal.3109cm3.
Ghantous, A., Gali-Muhtasib, H., Vuorela, H., Saliba, N.A., Darwiche, N., 2010. What made sesquiterpene lactones reach cancer clinical trials? Drug. Discov. Today 15, 668–678.
Ghisalberti, E.X., 2000. Lontana camara L. (Verbenaceae). Fitoterapia 71, 467–486.
Grace-Lynn, C., Darah, I., Chen, Y., Latha, L.Y., Jothy, S.L., Sasidharan, S., 2012. In vitro antioxidant activity potential of lantadene A, a pentacyclic triterpenoid of Lontana plants. Molecules 17, 11185–11198.
Grecco, S.S., Reimao, J.Q., Tempone, A.G., Salterelli, P., Romoff, P., Ferreira, M.J.P., Favero, O.A., Lago, J.H.G., 2010. Isolation of an antileishmanial and antitrypanosomal flavanone from the leaves of Baccharis refusa DC. (Asteraceae). Parasitol. Res. 106, 1245–1248.
Hasan, M., Burdi, D.K., Ahmad, V.U., 1991. Diterpene fatty acid ester from Leucas nutans. J. Nat. Prod. 54, 1444–1446.
Mithöfer, A., Maitrejean, M., Boland, W., 2004. Structural and biological diversity of cyclic octadecanoids, jasmonates, and mimetics. J. Plant Growth Regul. 23, 170–178.
Mittra, B., Saha, A., Chowdhury, A.R., Pal, C., Mandai, S., Mukhopadhyay, S., Bandyopadhyay, S., Majumder, H.K., 2000. Luteolin, an abundant dietary component is a potent antileishmanial agent that acts by inducing topoisomerase ll-mediated kinetoplast DNA cleavage leading to apoptosis. Mol. Med. 6, 527–541.
Pan, H., Lundgren, L.N., 1995. Phenolic extractives from root bark of Picea abies. Phytochemistry 39, 1423–1428.
Pisutthanan, N., Liawruangrath, B., Liawruangrath, S., Bremner, J.B., 2006. A new flavonoid from Chromolaena odorata. Nat. Prod. Res. 20, 1192–1198.
Pollmann, S., 2009. Plant oxylipins: the versatile functions of cyclic octadecanoids and jasmonates. FEBS J. 276, 4665.
Reynaud. J., Raynaud. J., 1984. Presence of onopordin in Doronicum grandiflorum Lam. (Compositae). Pharmazie 39, 126.
Salamanca, E.C., Ruiz, G., Ticona, J.C., Giménez, A., 2008. Método colorimétrico-XXT: como evaluación de alto rendimiento de sustancias con actividad leishmanicida. Biofarbo 16, 21–27.
Salvat, A., Antonacci, L., Fortunato, R.H., Suarez, E.Y., Godoy, H.M., 2004. Antimicrobial activity in methanolic extracts of several plant species from northern Argentina. Phytomedicine 11, 230–234.
Samuelsson, G., Bohlin, L., 2010. Drugs of Natural Origin: A Treatise of Pharmacognosy. Swedish Pharmaceutical Press, Stockholm, Sweden.
Sena Filho, J.G., Rabbani, A.R.C., dos, S.S.T.R., Cruz, D.S.A.V., Souza, I.A., Santos, M.J.B.A., Romariode, J.J., Nogueira, P.C.D.L., Duringer, J.M., 2012. Chemical and molecular characterization of fifteen species from the Lontana (Verbenaceae) genus. Biochem. Syst. Ecol. 45, 130–137.
Slowing, K., Sollhuber, M., Carretero, E., Villar, A., 1994. Flavonoid glycosides from Eugenia jambos. Phytochemistry 37, 255–258.
Sousa, E.O., Costa, J.G.M., 2012. Genus Lontana: chemical aspects and biological activities. Rev. Bras. Farmacogn. 22, 1115–1180.
Taleb-Contini, S.H., Salvador, M.J., Balanco, J.M.F., Albuquerque, S., de Oliveira, D.C.R., 2004. Antiprotozoal effect of crude extracts and flavonoids isolated from Chromolaena hirsuta (asteraceae). Phytother. Res. 18, 250–254.
Tasdemir, D., Kaiser, M., Brun, R., Yardley, V., Schmidt, T.J., Tosun, F., Ruedi, P., 2006. Antitrypanosomal and antileishmanial activities of flavonoids and their analogues: in vitro, in vivo, structure-activity relationship, and quantitative structure-activity relationship studies. Antimicrob. Agents Chemother. 50, 1352–1364.
Taskova, R.M., Kokubun, T., Grayer, R.J., Ryan, K.G., Garnock-Jones, P.J., 2008. Flavonoid profiles in the Heliohebe group of New Zealand Veronica (Plantaginaceae). Biochem. Syst. Ecol. 36, 110–116.
Ulubelen, A., Miski, M., Neuman, P., Mabry, T.J., 1979. Flavonoids of Salvia tomentosa (Labiatae). J. Nat. Prod. 42, 261–263.
Vieira, I.J.C., Azevedo, O.D.A., Jorgeanede, S.J., Braz-Filho, R., Goncalves, M.D.S., Francisco, D.A.M., 2013. Hirtinone, a novel cycloartane-type triterpene and other compounds from Trichilia hirta L. (Meliaceae). Molecules 18, 2589–2597.
Whalen, M.D., Mabry, T.J., 1979. New 8-hydroxyflavonoids from Solanum section Androceras. Phytochemistry 18, 263–265.
Acknowledgments
The financial support of the Swedish International Development Agency (SIDA) in a frame of collaboration agreement between Lund University (Sweden) and San Simón University (Bolivia) is gratefully acknowledged. We thank to Lic. Modesto Zárate for identifiying the plant.
Author information
Authors and Affiliations
Contributions
EMM have contributed to plant collection, carried out the laboratory work, analysis of the NMR data and wrote the manuscript. ES and AG performed the biological studies. OS supervised the laboratory work, interpreted the NMR data and wrote the manuscript. All the authors have read the final manuscript and approved the submission.
Corresponding author
Ethics declarations
The authors declare no conflicts of interest.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Maldonado, E.M., Salamanca, E., Giménez, A. et al. Antileishmanial metabolites from Lantana balansae. Revista Brasileira de Farmacognosia 26, 180–183 (2016). https://doi.org/10.1016/j.bjp.2015.11.007
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2015.11.007