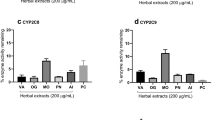
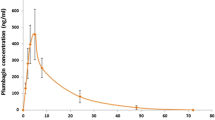
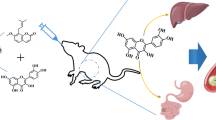
Abstract
The chronic and comorbid nature of HIV infection necessitate the use of multiple drugs including herbs to relieve symptoms with a possible increase in herb-drug interaction cases. This study was designed to evaluate the effect of Millettia aboensis (Hook. f.) Baker, Fabaceae, on cytochrome P450 3A isoenzyme and the influence of this effect on the bioavailability of two antiretroviral agents. In vitro effect of ethanol extract of M. aboensis on intestinal and liver microsomes extracted from female rats was assessed using erythromycin-N-demethylation assay method while in vivo effects were determined by estimating simvastatin plasma concentrations in rats. The effect of the extract on pharmacokinetic parameters of orally administered efavirenz (25 mg/kg) and nevirapine (20 mg/kg) was determined in rats divided into groups (n = 5). Plasma drug concentrations were assayed using HPLC and pharmacokinetic parameters determined through a non-compartmental analysis as implemented in WinNonlin pharmacokinetic program. The extract inhibited both intestinal and liver microsomal cytochrome P450 3A isoenzyme activities in vitro and enhanced simvastatin absorption in vivo with possible inhibition of metabolizing enzymes as indicated by significant (p<0.05) increase in maximal concentration, area under curve and mean resident time of the drug. However, further in vivo interaction studies in animal model did not produce significant (p>0.05) changes in the pharmacokinetic parameters of efavirenz and nevirapine. HPLC fingerprinting indicated the presence of quercetin and kaempferol in the extract. These findings revealed M. aboensis as an inhibitor of cytochrome P450 3A enzyme but, with no significant effect on the bioavailability of orally administered nevirapine and efavirenz.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ajaghaku, D.L., Ilodigwe, E.E., Obi, H.I., Uzodimma, S.U., 2012. Toxicological evaluation of ethanol leaf extract ofMilletia aboensis (Hook. f) Baker. IJPI J. Pharmacol. Toxicol. 2, 1–8.
Banzouzi, J.T., Prost, A., Rajemiarimiraho, M., Ongoka, P., 2008. Traditional uses of the African Millettia species (Fabaceae). Int. J. Bot. 4, 406–420.
Borokini, T.I., Omotayo, F.O., 2012. Phytochemical and ethnobotanical study of some selected medicinal plants from Nigeria. J. Med. Plants Res. 6, 1106–1118.
Ciesela, L., 2012. Biological fingerprinting ofherbal samples by means of liquid chromatography. Chromatogr. Res. Int., http://dx.doi.org/10.1155/2012/532418.
Cotreau, M.M., von Moltke, L.L., Beinfeld, M.C., Greenblatt, D.J., 2003. Methodologies to study the induction of rat hepatic and intestinal cytochrome P450 3A at the mRNA, protein, and catalytic activity level. J. Pharmacol. Toxicol. Methods 33, 45–55.
Dudhatra, G.B., Mody, S.K., Awale, M.M., Patel, H.B., Modi, C.M., Kumar, A., Kamani, D.R., Chauhan, N.B., 2012. Comprehensive review on pharmacotherapeutics of herbal bioenhancers. Sci. World J. 2012, 1–33.
Dumond, J.B., Kashuba, A.D.M., 2009. Pharmacotherapy of human immunodeficiency virus infection. In: Koda-Kimble, M.A., Young, L.Y., Alldredge, B.K., Corelli, R.L., Guglielino, B.J., Kradjan, W.A., Williams, B.R. (Eds.), Applied Therapeutics. The Clinical Use of Drugs., 9th ed. Lippincott Williams and Wilkins, New York, pp. 61-1-24.
Eggadi, V., Ponna, S.K., Kankanala, S.R., Sheshagiri, S.B.B., Gaddam, S.R., 2013. Determination of simvastatin and diltiazem in rat plasma by HPLC and pharmacokinetic studies. Int. J. Pharm. Sci. Res. 21, 53–56.
Fagot, J.P., Mockenhaupt, M., Bouwes-Bavinck, J.N., 2001. Nevirapine and the risk of Stevens-Johnson syndrome ortoxic epidermal necrolysis. AIDS 15, 1843–1848.
Foti, R.S., Rock, D.A., Wienkers, L.C., Wahlstrom, J.L., 2010. Selection of alternative CYP3A4 probe substrates for clinical drug interaction studies using in vitro data and in vivo simulation. Am. Soc. Pharmacol. Exp. Ther. 38, 981–987.
Greenblatt, D.J., Zhao, Y., Venkatakrishnan, K., Duan, S.X., Harmatz, J.S., Parent, S.J., Court, M.H., von Moltke, L.L., 2011. Mechanism of cytochrome P4503A inhibition by ketoconazole. J. Pharm. Pharmacol. 63, 214–221.
Harrison, J.J.E.K., Dankyi, E., Kingsford-Adaboh, R., Ishida, H., 2011. In search of new leads: a closer look at the therapeutic potential of the constituents of Millettia thonningii, Millettia pachycarpa and their structural analogues. Int. J. Pharm. Pharm. Sci. 3, 71–81.
Hu, O.Y., Hsiong, C.H., Kuo, B.P., Pao, L., 2007. Cytochrome P450 3A inhibitors and enhancers. United States Patent, US 7, 16976. B2.
Jambhekar, S.S., Breen, P.J., 2009. Basic Pharmaceutics: Extravascular Routes of Drugs Administration. Pharmaceutical Press, London.
Kanazu, T., Yamaguchi, Y., Okamura, N., Baba, T., Koike, M., 2004. Model for the drug-drug interaction responsible for CYP3A enzyme inhibition. II: establishment and evaluation of dexamethasone-pretreated female rats. Xenobiotica 34, 403–413.
Kang, M.J., Cho, J.Y., Shim, B.H., Kim, D.K., Lee, J., 2009. Bioavailability enhancing activities of natural compounds from medicinal plants. J. Med. Plants Res. 3, 1204–1211.
Kasibhatta, R., Naidu, M.U.R., 2007. Influence of piperine on the pharmacokinetics of nevirapine under fasting conditions: a randomized crossover placebo controlled study. Drug Dev. Res. 8, 383–391.
Kumar, C.H., Kumar, D.A., Rao, J.V.L.N.S., 2010. Anew validated RP-HPLC method for the determination of nevirapine in human plasma. Eur. J. Chem. 7, 821–826.
Labaune, J.P., 1989. Handbook of Pharmacokinetics. Ellis Harwood Ltd., Chichester.
Lin, J.H., Chiba, M., Baillie, T.A., 1999. Is the role of the small intestine in first pass metabolism overemphasized? Am. Soc. Pharmacol. Exp. Ther. 51, 135–157.
Machtinger, E.L., Bangsberg, D.R., 2013. Adherence to HIV Antiretroviral Therapy. HIV InSite Knowledge Base Chapter. UCSF Center for HIV Information.
Mazzari, A.L.D.A., Prieto, J.M., 2014. Herbal medicines in Brazil: pharmacokinetic profile and potential herb-drug interactions. Front. Pharmacol. 5, 1–12.
Michaud, V., Bar-Magen, T., Turgeon, J., Flockhart, D., Desta, Z., Wainberg, M.A., 2012. The dual role of pharmacogenetics in HIV treatment: mutations and polymorphisms regulating antiretroviral drug resistance and disposition. Pharm. Rev. 64, 803–833.
Mitschke, D., Reichel, A., Fricker, G., Moenning, U., 2008. Characterisation of cytochrome P450 protein expression along the entire length of the intestine of male and female rats. Drug Metab. Dispos. 36, 1039–1045.
Nduka, S.O., Okonta, J.M., Esimone, C.O., 2013. Effects of Zingiber officinale on the plasma pharmacokinetics and lung penetrations of ciprofloxacin and isoniazid. Am. J. Ther. 20, 501–513.
Onyegeme-Okerenta, B.M., Okafor, U.A., 2014. Antimicrobial properties of ethanol leaf extract of Millettia aboensis on some selected clinical isolates. Univ. J. Plant Sci. 2, 97–101.
Paine, M.F., Khalighi, M., Fisher, J.M., 1997. Characterisation of interintestinal and intraintestinal variations in human CYP3A-dependent metabolism. J. Pharmacol. Exp. Ther. 283, 1552–1562.
Proudfoot, S.G., 1999. Factors influencing bioavailability: factors influencing absorption from the gastrointestinal tract. In: Aulton, M.E. (Ed.), Pharmaceutics: The Science of Dosage Form Designs. Churchill Livingston, London, pp. 135–137.
Raffanti, S., Haas, D.W., 2001. Antimicrobial agents: antiretroviral agents. In: Hardman, J.C., Limbird, L.E. (Eds.), The Pharmacological Basis of Therapeutics., 10th ed. McGraw-Hill, US, pp. 3–30.
Rahman, S., Singhal, K., 2002. Problems in Pharmacovigilance of Medicinal Products of Herbal Origin and Means to Minimize Them. Uppsala Reports January Supplement, Uppsala.
Randhawa, G.K., Kullar, J.S., Kumar, R., 2011. Bioenhancer from mother nature and their applicability in modern medicine. Int. J. Appl. Basic Med. Res. 1, 5–10.
Sailaja, A.L., Kumar, K.K., Kumar, D.V.R., Yugandhar, N.M., Srinubabu, G., 2007. Development and validation of a liquid chromatographic method for determination of efavirenz in human plasma. Chromatographia 65, 359–361.
Schenkman, J.B., Cinti, D.L., 1978. Preparation of microsomes with calcium. Methods Enzymol. 52, 83–89.
Takemoto, K., Yamazaki, H., Tanaka, Y., Nakajima, M., Yokoi, T., 2003. Catalytic activities of cytochrome P450 enzymes and UDP-glucuronosyl transferases involved in drug metabolism in rat everted sacs and intestinal microsomes. Xenobiotica 33, 43–55.
Umathe, S.N., Dixit, P.V., Kumar, V., Bansod, K.U., Wanjari, M.M., 2008. Quercetin pretreatment increases the bioavailability of pioglitazone in rats: involvement of CYP3A inhibition. Biochem. Pharmacol. 75, 1670–1676.
van Waterschoot, R.A.B., Schinkel, A.H., 2011. A critical analysis of the interplay between cytochrome P4503Aand P-glycoprotein: recent insight from knockout and transgenic mice. Am. Soc. Pharmacol. Exp. Ther. 63, 390–408.
Veldkamp, A.I., Harris, M., Montaner, J.S.G., 2001. The steady state pharmacokinetics of efavirenz and nevirapine when used in combination in human immunodeficiency virus type 1 infected persons. J. Infect. Dis. 184, 37–42.
Ward, B.A., Gorski, J.C., Jones, D.R., Hall, S.D., Flockhart, D.A., Desta, Z., 2003. The cytochrome P450 2B6 (CYP2B6) is the main catalyst of efavirenz metabolism: implication for HIV/AIDS therapy and utility of efavirenz as substrate marker of CYP 2B6 catalytic activity. J. Pharmacol. Exp. Ther. 306, 287–300.
WHO, 2003. Traditional Medicine Facts Sheet. World Health Organisation, http://www.who.int/mediacentre/factsheets/2003/fs134/en/ (accessed August 2015).
Wrighton, S.A., Schuetz, E.G., Watkins, P.B., Maurel, P., Barwick, J., Bailey, B.S., 1985. Demonstrations in multiple species of inducible hepatic cytochrome P-450 and their mRNAs related to the glucocorticoid-inducible cytochrome P-450 of the rat. Mol. Pharmacol. 28, 312–321.
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors contribution
SON and MJO designed the study. SON and DA were responsible for execution of the project and conducted data analysis. SON, KCA and CVU drafted the manuscript. All the authors reviewed and approved the manuscript.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nduka, S.O., Okonta, M.J., Ajaghaku, D.L. et al. Inhibition of cytochrome P450 3A enzyme by Millettia aboensis: its effect on the pharmacokinetic properties of efavirenz and nevirapine. Rev. Bras. Farmacogn. 27, 228–235 (2017). https://doi.org/10.1016/j.bjp.2016.10.008
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2016.10.008