Abstract
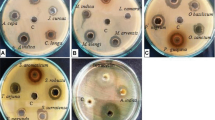
In Sudan, some medicinal plants, such as Acacia seyal, Calotropisprocera and Balanites aegyptiaca have been used to prevent or treat oral health problems. The stem and stem bark of Terminalia laxiflora Engl., Combretaceae, are used as antiseptics for mouthwash to prevent gingivitis and thrush in Africa. Methanol and 50% hydroethanolic extracts of 25 plants that are used in traditional Sudanese medicine for several diseases and cavity disorders were screened for anti-cavity activities. T. laxiflora methanolic wood extracts, which exhibited such activity, were investigated. The crude extracts were assayed for their antimicrobial activities against Streptococcus sobrinus in terms of minimum inhibitory concentration and glucosyltransferase inhibition. The active extract of T. laxiflora wood was subsequently fractionated by different chromatographic techniques. Isolated compounds were identified by spectroscopic methods and assessed for S. sobrinus and glucosyltransferase inhibitory effects. Methanolic extracts of Terminalia brownii (bark), T. laxiflora (wood), A. seyal (bark), Persicaria glabra (leaves) and Tamarix nilotica (stem) showed good activities against both S. sobrinus and glucosyltransferase (MIC ≤ 1 mg/ml, IC50 values <50 μg/ml). Over all plant extracts, T. laxiflora demonstrated the good combined activities (MIC 0.5 mg/ml, glucosyltransferase, IC50 10.3 μg/ml); therefore, its methanolic wood extracts were selected for further phytochemical studies. Four constituents were isolated by chromatographic techniques and identified by spectroscopic techniques. Pharmacological evaluation of the obtained compounds showed that flavogallonic acid dilactone had comparatively good antibacterial activity. In the glucosyltransferase inhibitory test, terchebulin displayed potent activity with an IC50 of 7.5 μM. The screening presented in this study showed that methanol extracts of T. laxiflora wood possessed promising anti-cavity effects.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ahmad, A., Alkarkhi, A.A., Hena, S., Lim, H.K., 2009. Extraction, separation and identification of chemical ingredients of Elephantopus scaber (L) using factorial design of experiment. Int. J. Chem. 1, 36–49.
Bowen, W.H., Koo, H., 2011. Biology of Streptococcus mutans-derived glucosyltrans-ferases: role in extracellular matrix formation of cariogenic biofilms. Caries Res. 45, 69–86.
Cabral, C.T., Fernandes, M.H., 2007. In vitro comparison of Chlorhexidine and povidone-iodine on the long-term proliferation and functionalactivity of human alveolar bone cells. Clin. Oral Invest. 11, 155–164.
Caunii, A., Pribac, G., Grozea, I., Gaitin, D., Samfira, I., 2012. Design of optimal solvent for extraction of bioactive ingredients from six varieties of Medicago sativa. Chem. Cent. J. 6, 123. El Ghazali, G.B., Abdalla, W.E., Khalid, H.E., Khalafalla, M.M., Hamad, A.A., 2003. Medicinal Plants of Sudan. Part V. Medicinal Plants of Ingassana Area. National Council for Research, Khartoum Press, Khartoum, Sudan.
Eloff, J.N., 2001. Antibacterialactivity of Marula (Sclerocarya birrea (A. rich) Hochst. subsp. caffra (Sond.) Kokwaro) (Anacardiaceae) bark and leaves. J. Ethnopharmacol. 76, 305–308.
Faria, G., Cardoso, C.R.B., Larson, R.E., Silva, J.S., Rossi, M.A., 2009. Chlorhexidine-induced apoptosis or necrosis in L929 fibroblasts: a role for endoplasmic reticulum stress. Toxicol. Appl. Pharmacol. 234, 256–265.
Fasola, T.R., Oluwole, M.E., Olaniyi, I.F., Adeboye, I.E., 2013. The phytochemical and antimicrobialactivities of Terminalia laxiflora Engl. & Diels root bark extract. Nat. Sci. 11, 122–127.
Gauniyal, P., Teotia, U.V.S., 2014. Phytochemical screening and antimicrobialactivity of some medicinal plants against oral flora. Asian Pac. J. Health Sci. 1, 236–255.
Giannelli, M., Chellini, F., Margheri, M., Tonelli, P., Tani, A., 2008. Effect of Chlorhexidine digluconate on different cell types: a molecular and ultrastructural investigation. Toxicol. In Vitro 22, 308–317.
Gibbons, S., 2005. Plants as a source of bacterial resistance modulators and anti-infective agents. Phytochem. Rev. 4, 63–78.
Grimshaw, J., Haworth, R.D., 1956. Flavogallol. J. Chem. Soc, 4225–4232.
Gulati, V., Harding, LH., Palombo, E.A., 2012. Enzyme inhibitory and antioxidantactivities of traditional medicinal plants: potential application in the management of hyperglycemia. BMC Complement. Altern. Med., https://doi.org/10.1186/1472-6882-12-77.
Hashizume-Takizawa, T., Shinozaki-Kuwahara, N., Tomita, N., Kurita-Ochiai, T., 2014. Establishment of a convenient sandwich-ELISA for direct quantification of glucosyltransferase-I: application for dual diagnosis of dental caries. Monoclon. Antib. Immunodiagn. Immunother. 33, 89–93.
Hirano, Y., Kondo, R., Sakai, K., 2003. 5α-Reductase inhibitory tannin-related compound isolated from Shorea laevifoiia. J. Wood Sci. 49, 339–342.
Ibrahim, M.A., Mohammed, A., Isah, M.B., Aliyu, A.B., 2014. Antitrypanosomalactivity of African medicinal plants: a review update. J. Ethnopharmacol. 154, 26–54.
Iwaki, K., Koya-Miyata, S., Kohno, K., Ushio, S., Fukuda, S., 2006. Antimicrobialactivity of Polygonum tinctorium Lour: extract against oral pathogenic bacteria. J. Nat. Med. 60, 121–125.
Kang, M.S., Oh, J.S., Kang, I.C., Hong, S.J., Choi, C.H., 2008. Inhibitory effect of methyl gallate and gallicacid on oral bacteria. J. Microbiol. (Seoul, Korea) 46, 744–750.
Karuppiah, P., Rajaram, S., 2012. Antibacterial effect of Allium sativum cloves and Zingiber officinale rhizomes against multiple drug resistant clinical pathogens. Asian Pac. J. Trop. Biomed. 2, 597–601.
Khalid, H.E., Abdalla, W.E., Haider, A., Till, O., Thomas, E., 2012. Gems from traditional north-African medicine: medicinal and aromatic plants from Sudan. Nat. Prod. Bioprospect. 2, 92–103.
Kinjo, J., Nagao, T., Tanaka, T., Nonaka, G.I., Okabe, H., 2001. Antiproliferative constituents in the plant 8. Seeds of Rhynchosia volubilis. Biol. Pharm. Bull. 24, 1443–1445.
Kolenbrander, P.E., Palmer, R.J., Rickard, A.H., Jakubovics, N.S., Chalmers, N.I., Diaz, P.I., 2006. Bacterial interactions and successions during plaque development. Periodontology 42, 47–79.
Koo, H., Xiao, J., Klein, M.I., 2009. Extracellular polysaccharides matrix - an often forgotten virulence factor in oral biofilm research. Int. J. Oral Sci. 1, 229–234.
Li, Y.C., Kuan, Y.H., Lee, T.H., Huang, F.M., Chang, Y.C., 2014. Assessment of the cytotoxicity of Chlorhexidine by employing an in vitro mammalian test system. J. Dent. Sci. 9, 130–135.
Lin, T.C., Nonaka, G.I., Nishioka, I., Ho, F.C., 1990. Tannins and related compounds, CIL Structures of terchebulin, an ellagitannin having a novel tetraphenylcar-boxylicacid (terchebulicacid) moiety, and biogenetically related tannins from Terminalia chebula Retz. L. Chem. Pharm. Bull. 38, 3004–3008.
Livia, S., Silvia, F., Katarina, R., Jan, K., Pavel, M., 2016. Antibiofilmactivity of plant polyphenols. Molecules 21, 1717.
Matsumoto, M., Hamada, S., Ooshima, T., 2003. Molecular analysis of the inhibitory effects of oolong tea polyphenols on glucan-binding domain of recombinant glucosyltransferases from Streptococcus mutans MT8148. FEMS Microbiol. Lett. 228, 73–80.
Mitsunaga, T., Abe, I., Kontani, M., Ono, H., Tanaka, T., 1997. Inhibitory effects of bark proanthocyanidins on theactivities of glucosyltransferases of Streptococcus sobrinus. J. Wood Chem. Technol. 17, 327–340.
Muddathir, A.M., Mitsunaga, T., 2013. Evaluation of anti-acneactivity of selected Sudanese medicinal plants. J. Wood Sci. 59, 73–79.
Muddathir, A.M., Mitsunaga, T., Yamauchi, K., 2013. Anti-acneactivity of tannin-related compounds isolated from Terminalia iaxiflora. J. Wood Sci. 59, 426–431.
Ncube, B., Finnie, J.F., Van Staden, J., 2012. in vitro antimicrobial synergism within plant extract combinations from three South African medicinal bulbs. J. Ethnopharmacol. 139, 81–89.
Nishimura, J., Saito, T., Yoneyama, H., Bai, LL., Okumura, K., Isogai, E., 2012. Biofilm formation by Streptococcus mutans and related bacteria. Adv. Microbiol. 2, 208–215.
Orabi, M.A.A., Yoshimura, M., Amakura, Y., Hatano, T., 2015. Ellagitannins, gallotan-nins, and gallo-ellagitannins from the galls of Tamarix aphylla. Fitoterapia 104, 55–63.
Paes Leme, A.F., Koo, H., Bellato, C.M., Bedi, G., Cury, J.A., 2006. The role of sucrose in cariogenic dental biofilm formation - new insight. J. Dent. Res. 85, 878–887.
Rajendran, R., Sivapathasundharam, B., Shafer, Hine, Levy, 2009. Shafer’s Textbook of Oral Pathology, 6th ed. Elsevier, India, pp. 409.
Samuelsen, A.B., 2000. The traditional uses, chemical constituents and biologicalactivities of Plantago major L. A review. J. Ethnopharmacol. 71, 1–21.
Sarabhai, S., Sharma, P., Capalash, N., 2013. Ellagicacid derivatives from Terminalia chebula Retz. downregulate the expression of quorum sensinggenes to attenuate Pseudomonas aeruginosa PAO1 virulence. PLoS One 8, 1–12.
Sawamura, S., Tonosaki, Y., Hamada, S., 1992. Inhibitory effect of ellagicacid on glucosyltransferase from mutans streptococci. Biosci. Biotechnol. Biochem. 56, 766–768.
Shuaibu, M.N., Wuyep, P.A., Yanagi, T., Hirayama, K., Tanaka, T., Kouno, I., 2008. The use of microfluorometric method foractivity-guided isolation of antiplasmodial compound from plant extracts. J. Parasitol. Res. 102, 1119–1127.
Silva, O., Gomes, E.T., Wolfender, J.L., Marston, A., Hostettmann, K, 2000. Application of high performance liquid chromatography coupled with ultraviolet spectroscopy and electrospray mass spectrometry to the characterisation of ellagitannins from Terminalia macroptera roots. Pharm. Res. 17, 1396–1401.
Siqueira, J.F., Rocas, I.N., 2005. Exploiting molecular methods to explore endodontic infection: Part 2 - redefining the endodontic microbiota. J. Endod. 3, 488–498.
Tanaka, T., Nonaka, G., Nishioka, I., 1986. Tannins and related compounds. XLII. Isolation and characterization of four new hydrolysable tannins, terflavins A and B, tegallagin and tercatain from the leaves of Terminalia catappa L. Chem. Pharm. Bull. 34, 1039–1049.
Tanaka, T., Ueda, N., Shinohara, H., Nonaka, G., Fujioka, T., Mihashi, K., Kouno, I., 1996. C-glycosidic ellagitannin metabolites inthe heartwood of Japanese chestnuttree (Castanea crenata Sieb. et Zucc). Chem. Pharm. Bull. 44, 2236–2242.
Thanh, V.N., Christopher, J.S., Michael, C.B., Phuong, D.N., Quan, V., 2016. Impact of different extraction solvents on bioactive compounds and antioxidant capacity from the root of Saiacia chinensis L. J. Food Qual. 2017, 8.
Yamauchi, K., Mitsunaga, T., Muddathir, A.M., 2016. Screening for melanogenesis-controlled agents using Sudanese medicinal plants and identification ofactive compounds in the methanol extract oiTerminalia brownii bark. J. Wood Sci. 62, 285–293.
Yanagida, A., Kanda, T., Tanabe, M., Matsudaira, F., Oliveira Cordeiro, J.G., 2000. Inhibitory effects of apple polyphenols and related compounds on cariogenic factors of mutans streptococci. J. Agric. Food Chem. 48, 5666–5671.
Zhi, R., Lulu, C., Jiyao, L., Yuqing, L., 2016. Inhibition of Streptococcus mutans polysaccharide synthesis by molecules targeting glycosyltransferaseactivity. J. Oral Microbiol. 8, 31095.
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors’ contributions
EAMM, AMM, KY and TM all contributed to the writing of this article. EAMM and AMM obtained samples. TM designed the study and supervised the laboratory work. EAMM and AMM performed the different assays and statistical analysis. KM contributed to compound identification.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mohieldin, E.A.M., Muddathir, A.M., Yamauchi, K. et al. Anti-caries activity of selected Sudanese medicinal plants with emphasis on Terminalia laxiflora. Rev. Bras. Farmacogn. 27, 611–618 (2017). https://doi.org/10.1016/j.bjp.2017.04.002
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2017.04.002