Abstract
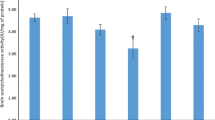
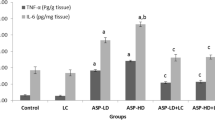
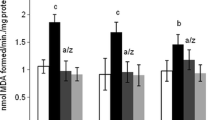
This study investigated the possible antioxidant and neuroprotective effects of alcesefoliside, isolated from Astragalus monspessulanus L., Fabaceae, against carbon tetrachloride (CCl4)-induced brain injury in Wistar rats. Iron sulphate/ascorbic acid lipid peroxidation was induced in rat brain microsomes and pre-incubated with alcesefoliside and silybin. Male rats were treated in vivo with alcesefoliside and with silymarin alone; animals challenged with CCl4; and pre-treated with alcesefoliside or silymarin in respective doses for 7 days, challenged with CCl4, followed by curative treatment (additional 14 days). The activity of acetylcholine esterase and the antioxidant enzymes: superoxide-dismutase, catalase, glutathione-peroxidase, glutathione reductase and glutathione-S-transferase as well as the biomarkers of oxidative stress malondialdehyde and reduced glutathione were measured. The alcesefoliside pre-treatment and consecutive curative treatment normalizes the activity of the antioxidant enzymes as well as levels of malondialdehyde and reduced glutathione. The observed effects on tissue level correlate with the histopathological observations of the brain. They were comparable to the effects of silymarin, used as a positive control. The results showed that alcesefoliside has a neuroprotective effect against CCl4-induced brain toxicity in rats.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Aebi, H., 1974. Catalase. Methods of enzymatic analysis, vol. 2., 2nd ed. Elsevier, pp. 673–684.
Ahn, T.H., Yang, Y.S., Lee, J.-C., Moon, C.J., Kim, S.-H., Jun, W., Park, S.-C., Kim, J.-C., 2007. Ameliorative effects of pycnogenol® on carbon tetrachloride-induced hepatic oxidative damage in rats. Phyther. Res. 21, 1015–1019.
Bancroft, J.D., Gamble, M., 2002. Theory and practice of histological techniques, 5th ed. Churchill Livingstone Publications, Edinburgh.
Borah, A., Paul, R., Choudhury, S., Choudhury, A., Bhuyan, B., Das Talukdar, A., Dutta Choudhury, M., Mohanakumar, K.P., 2013. Neuroprotective potential of silymarin against CNS disorders: insight into the pathways and molecular mechanisms of action. CNS Neurosci. Ther. 19, 847–853.
Borsari, M., Gabbi, C., Ghelfi, F., Grandi, R., Saladini, M., Severi, S., Borella, F., 2001. Silybin, a new iron-chelating agent. J. Inorg. Biochem. 85, 123–129.
Bourezzane, S., Haba, H., Long, C., Benkhaled, M., 2018. Chemical composition and antioxidant activity of Astragalus monspessulanus L. growing in semiarid areas of Algeria. J. Serb. Chem. Soc. 83, 31–38.
Bump, E.A., Taylor, Y.C., Brown, J.M., 1983. Role of glutathione in the hypoxic cell cytotoxicity of misonidazole. Cancer Res. 43, 997–1002.
Dashti, H., Jeppsson, B., Hägerstrand, I., Hultberg, B., Srinivas, U., Abdulla, M., Beng-mark, S., 1989. Thioacetamide- and carbon tetrachloride-induced liver cirrhosis. Eur. Surg. Res. Eur. Chir. Forschung. Rech. Chir. Eur. 21, 83–91.
Deby, C., Goutier, R., 1990. New perspectives on the biochemistry of superoxide anion and the efficiency of superoxide dismutases. Biochem. Pharmacol. 39, 399–405.
Ellman, G.L., 1959. Tissue sulfhydryl groups. Arch. Biochem. Biophys. 82, 70–77.
Habbu, P.V., Shastry, RA, Mahadevan, K.M., Joshi, H., Das, S.K., 2008. Hepatoprotec-tive and antioxidant effects ofArgyreiaspeciosa in rats. Afr.J. Tradit. Complement. Altern. Med. 5, 158–164.
Habig, W.H., Pabst, M.J., Jakoby, W.B., 1974. Glutathione S transferases. The first enzymatic step in mercapturic acid formation. J. Biol. Chem. 249, 7130–7139.
Jiménez, W., Clària, J., Arroyo, V., Rodés, J., 1992. Carbon tetrachloride induced cirrhosis in rats: a useful tool for investigating the pathogenesis of ascites in chronic liverdisease. J. Gastroenterol. Hepatol. 7, 90–97.
Kondeva-Burdina, M., Krasteva, I., Mitcheva, M., 2014. Effects of rhamnocitrin 4-ß-D-galactopyranoside, isolated from Astragalus hamosus on toxicity models in vitro. Pharmacogn. Mag. 10, S487–S493.
Kondeva-Burdina, M., Shkondrov, A., Simeonova, R., Vitcheva, V., Krasteva, I., Ionkova, I., 2018. In vitro/in vivo antioxidant and hepatoprotective potential of defatted extract and flavonoids isolated from Astragalus spruneri Boiss (Fabaceae). Food Chem. Toxicol. 111, 631–640.
Krasteva, I., Bratkov, V., Bucar, F., Kunert, O., Kollroser, M., Kondeva-Burdina, M., Ionkova, I., 2015. Flavoalkaloidsand flavonoids from Astragalus monspessulanus. J. Nat. Prod. 78, 2565–2571.
Lima Gonçalves, CF., de Souza dos Santos, M.C., Ginabreda, M.G., Soares Fortunato, R., Pires de Carvalho, D., Freitas Ferreira, A.C., 2013. Flavonoid rutin increases thyroid iodide uptake in rats. PLoS ONE 8, https://doi.org/10.1371/journal.pone.0073908.
Lowry, O.H., Rosebrough, N.J., Farr, A.L., Randall, R.J., 1951. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193, 265–275.
Makni, M., Chtourou, Y., Barkallah, M., Fetoui, H., 2012. Protective effect of vanillin against carbon tetrachloride (CCl4)-induced oxidative brain injury in rats. Toxicol. Ind. Health 28, 655–662.
Mansuy, D., Sassi, A., Dansette, P.M., Plat, M., 1986. A new potent inhibitor of lipid peroxidation in vitro and in vivo, the hepatoprotective drug anisyldithiolthione. Biochem. Biophys. Res. Commun. 135, 1015–1021.
Mishchenko, V.A., Goriukhina, O.A., Iliuk, R.D., 1993. Changes in the hemato-encephalic barrier in experimental livercirrhosis. BiulletenEksp. Biol. Med. 116, 638–641.
Misra, H.P., Fridovich, I., 1972. The role of superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase. J. Biol. Chem. 247, 3170–3175, doi: 4623845.
Nencini, C., Giorgi, G., Micheli, L., 2007. Protective effect of silymarin on oxidative stress in rat brain. Phytomedicine 14, 129–135.
Pinto, M.C., Mata, A.M., Lopez-Barea, J., 1984. Reversible inactivation of Saccha-romyces cerevisiae glutathione reductase under reducing conditions. Arch. Biochem. Biophys. 228, 1–12.
Polizio, A.H., Pena, C., 2005. Effects of angiotensin II type 1 receptor blockade on the oxidative stress in spontaneously hypertensive rat tissues. Regul.Pept. 128, 1–5.
Rice-Evans, C., 2001. Flavonoid antioxidants. Curr. Med. Chem. 8, 797–807.
Simeonova, R., Bratkov, V.M., Kondeva-Burdina, M., Vitcheva, V., Manov, V., Krasteva, I., 2015. Experimental liver protection of n-butanolic extract of Astragalus monspessulanus L. on carbon tetrachloride model of toxicity in rat. Redox Rep. Commun. Free Radic. Res. 20, 145–153.
Simeonova, R.L., Vitcheva, V.B., Kondeva-Burdina, M.S., Krasteva, I.N., Nikolov, S.D., Mitcheva, M.K., 2010. Effect of purified saponin mixture from Astragalus cor-niculatus on enzyme- and non-enzyme-induced lipid peroxidation in liver microsomes from spontaneously hypertensive rats and normotensive rats. Phytomedicine 17, 346–349.
Tappel, A.L., 1978. Glutathione peroxidase and hydroperoxides. Methods Enzymol. 52, 506–513.
Tirkey, N., Pilkhwal, S., Kuhad, A., Chopra, K., 2005. Hesperidin, a citrus bioflavonoid, decreases the oxidative stress produced by carbon tetrachloride in rat liver and kidney. BMC Pharmacol. 5, 2.
Vitcheva, V., Simeonova, R., Krasteva, I., Nikolov, S., Mitcheva, M., 2013. Protective effects of a purified saponin mixture from Astragalus corniculatus Bieb., in vivo hepatotoxicity models. Phyther. Res. 27, 731–736.
Voirol, P., Jonzier-Perey, M., Porchet, F., Reymond, M.J., Janzer, R.C., Bouras, C., Strobel, H.W., Kosel, M., Eap, C.B., Baumann, P., 2000. Cytochrome P-450 activities in human and rat brain microsomes. Brain Res. 855, 235–243.
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors’ contributions
IK collected the plant material and designed the study. AS carried out the extraction and purification of AF. RS and VV performed the in vivo experiment and wrote the first draft. MKB performed in vitro experiment. GP and VM performed histological examination of the tissues. All authors contributed to the critical revision of the manuscript.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Simeonova, R., Vitcheva, V., Kondeva-Burdina, M. et al. Alcesefoliside protects against oxidative brain injury in rats. Rev. Bras. Farmacogn. 29, 221–227 (2019). https://doi.org/10.1016/j.bjp.2018.10.002
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2018.10.002