Abstract
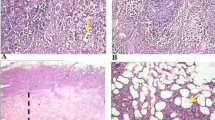
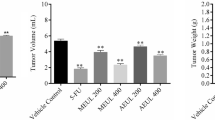
At present, there is a rapidly growing interest in studying the cytotoxic effects of Artemisia herba alba Asso, Asteraceae, in various cancer cell lines. However, its antitumor effectiveness has not been investigated. Therefore, the current study was conducted to study the effect of A. herba alba extract on the proliferation and growth of solid tumor cells in Ehrlich Solid Carcinoma bearing mice. Oral administration of A. herba alba extract resulted in significant reductions in tumor size, tumor weight and mice body weight, as well as caused concurrent significant increases in the DNA breakages and apoptotic DNA damage induction in a time-dependent manner. A. herba alba extract also raised the expression level of p53 gene and reduced of K-ras expression in a time-dependent manner. Minor histological lesions were observed in the liver and kidney tissues sections of mice administered A. herba alba extract compared with the high histological lesions observed in the liver and kidney tissues of artesunate and cisplatin treated groups. Thus, we concluded that A. herba alba extract exhibited promising potential antitumor efficacy with greater safety than artesunate and the commercially used anticancer drug cisplatin in mice.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Abdallah, H.M.I., Abdel-Rahman, R.F., El Awdan, SA, Allam, R.M., El-Mosallamy, A.E.M.K., Selim, M.S., Mohamed, S.S., Arbid, M.S., Farrag, A.H., 2019. Protective effect of some natural products against chemotherapy-induced toxicity in rats. Heliyon 5, e01590.
Abou El-Hamd, H.M., El-Sayed, M.A., Hegazy, M.E., Helaly, S.E., Esmail, A.M., Mohamed, N.S., 2010. Chemical constituents and biological activities of Artemisia herba-alba. Rec. Nat. Prod. 4, 1–25.
Almasad, M.M., Qazan, W.S., Daradka, H., 2007. Reproductive toxic effects of Artemisia herba-alba ingestion in female Spague-Dawley rats. Pak. J. Biol. Sci. 10, 3158–3161.
Bartkova, J., Rezaei, N., Liontos, M., Karakaidos, P., Kletsas, D., Issaeva, N., Vassiliou, L.V., Kolettas, E., Niforou, K., Zoumpourlis, V.C., 2006. Oncogene induced senescence is part of the tumorigenesis barrier imposed by DNA damage checkpoints. Nature 444, 633–637.
Bjelland, S., Seeberg, E., 2003. Mutagenicity, toxicity and repair of DNA base damage induced by oxidation. Mutat. Res. 531, 37–80.
Boudjelal, A.C., Henchiri, M., Sari, D., Sarri, N., Hendel, A., Benkhaled, A., Ruberto, G., 2013. Herbalists and wild medicinal plants in M’Sila (North Algeria): an ethnopharmacology survey. J. Ethnopharmacol. 148, 395–402.
Brydøy, M., Fosså, S.D., Dahl, O., Bjøro, T., 2007. Gonadal dysfunction and fertility problems in cancer survivors. Acta Oncol. 46, 480–489.
Cadet, J., Douki, T., Gasparutto, D., Ravanat, J.L., 2003. Oxidative damage to DNA: formation, measurement and biochemical features. Mutat. Res. 531, 5–23.
Can, G., Demir, M., Erol, O., Aydiner, A., 2013. A comparison of men and women’s experiences of chemotherapy-induced alopecia. Eur. J. Oncol. Nurs. 17, 255–260.
Chen, T., Li, M., Zhang, R., Wang, H., 2009. Dihydroartemisinin induces apoptosis and sensitizes human ovarian cancer cells to carboplatin therapy. J. Cell. Mol. Med. 13, 1358–1370.
Cramp, F., Byron-Daniel, J., 2012. Exercise for the management of cancer-related fatigue in adults. Cochrane Database Syst. Rev. 11, CD006145.
de Jonge, M.J., Verweij, J., 2006. Renal toxicities of chemotherapy. Semin. Oncol. 33, 68–73.
Di Micco, R., Fumagalli, M., Cicalese, A., Piccinin, S., Gasparini, P., Luise, C., Schurra, C., Garre, M., Nuciforo, P.G., Bensimon, A., 2006. Oncogene-induced senescence is a DNA damage response triggered by DNA hyper replication. Nature 444, 638–642.
Dizdaroglu, M., 2003. Substrate specificities and excision kinetics of DNA glycosy- lases involved in base-excision repairof oxidative DNA damage. Mutat. Res. 531, 109–126.
Dob, T., Benabdelkader, T., 2006. Chemical composition of the essential oil of Artemisia herba-alba Asso grown in Algeria. J. Essent. Oil Res. 6, 685–686.
Du, J.H., Zhang, H.D., Ma, Z.-J., Ji, K.M., 2010. Artesunate induces oncosis-like cell death in vitro and has antitumor activity against pancreatic cancer xenograftsin vivo. Cancer Chemother. Pharmacol. 65, 895–902.
Efferth, T., Dunstan, H., Sauerbrey, A., 2001. The anti-malarial artesunate is also active against cancer. Int. J. Oncol. 18, 767–773.
Efferth, T., Romero, M.R., Wolf, D.G., Stamminger, T., Marin, J.J., Marschall, M., 2008. The antiviral activities of artemisinin and artesunate. Clin. Infect. Dis. 47, 804–811.
Elad, S., Zadik, Y., Hewson, I., Hovan, A., Correa, M.E., Logan, R., Elting, L.S., Spijkervet, F.K., Brennan, M.T., 2010. A systematic review of viral infections associated with oral involvement in cancer patients: a spotlight on Herpes viridea. Support. Care Cancer 18, 993–1006.
Eweka, A.O., Adjene, J.O., 2008. Histological studies of the effects of oral administration of artesunate on the stomach of adult wistar rats. Internet J. Health 7, 1–7.
Fink, S.L., Cookson, B.T., 2005. Minireview: apoptosis, pyroptosis, and necrosis: mechanistic description of dead and dying eukaryotic cells. Infect. Immun. 73, 1907–1916.
Genovese, R.F., Newman, B.D., Brewer, T.G., 2000. Behavioral and neural toxicity of the artemisinin antimalaria arteether, but not artesunate and artelinate in rats. Pharmacol. Biochem. Behav. 67, 37–44.
Gibson, R.J., Keefe, D.M., 2006. Cancer chemotherapy-induced diarrhoea and constipation: mechanisms of damage and prevention strategies. Support. Care Cancer 14, 890–900.
Greenwell, M., Rahman, P.K.S.M., 2015. Medicinal plants: their use in anticancer treatment. Int. J. Pharm. Sci. Res. 6, 4103–4112.
Groopman, J.E., Itri, L.M., 1999. Chemotherapy-induced anemia in adults: incidence and treatment. J. Natl. Cancer Inst. Monographs 91, 1616–1634.
Gutierrez, M.I., Bhatia, K., Siwarski, D., Wolff, L, Magrath, I.T., Mushinski, J.F., Huppi, K., 1992. Infrequent p53 mutation in mouse tumors with deregulated myc. Cancer Res. 52, 1032–1035.
Ho, W.E., Peh, H.Y., Chan, T.K., Wong, W.S., 2014. Artemisinins: pharmacological actions beyond anti-malarial. Pharmacol. Ther. 142, 126–139.
Hou, J., Wang, D., Zhang, R., 2008. Experimental therapy of hepatoma with artemisinin and its derivatives: in vitro and in vivo activity, chemosensitization, and mechanisms of action. Clin. Cancer Res. 14, 5519–5530.
Iriadam, M., Musa, D., Gümühan, H., Baba, F., 2006. Effects of two Turkish medicinal plants Artemisia herba-alba and Teucrium polium on blood glucose levels and other biochemical parameters in rabbits. J. Cell Mol. Biol. 5, 19–24.
Ittarat, W., Udomsangpeth, R., Chotivanich, K.T., Looareesuwan, S., 1999. The effects of quinine and artesunate treatment on plasma tumor necrosis factor Levels in malaria infected patients. Southeast Asian J. Trop. Med. Public Health 30, 7–10.
Izunya, A.M., Nwaopara, A.O., Aigbiremolen, A., Oaikhena, G.A., 2010. Body and testicular weight changes in adult wistar rats following oral administration of artesunate. Res. J. Appl. Sci. Eng. Technol 2, 302–306.
Jackson, S.P., Bartek, J., 2009. The DNA-damage response in human biology and disease. Nature 461, 1071–1078.
Khlifi, D., Sghaier, R.M., Amouri, S., Laouini, D., Hamdi, M., Bouajila, J., 2013. Composition and antioxidant, anti-cancer and antiinflammatory activities of Artemisia herba-alba, Ruta chalpensis L. and Peganum harmala L. Food Chem. Toxicol. 55, 202–208.
Kim, T.H., Ito, H., Hatano, T., Taniguchi, S., Khennouf, S., Yoshida, T., 2004. Chemical constituents of Artemisia herba-alba Asso. Nat. Med. 58, 165.
King, P.D., Perry, M.C., 2001. Hepatotoxicity of chemotherapy. Oncologist 6, 162–176.
Lips, J., Kaina, B., 2001. DNA double-stranded breaks triggered apoptosis in p53 deficient fibroblast. Carcinogen 22, 579e585.
Liu, L., Zuo, L.F., Zuo, J., Wang, J., 2015. Artesunate induces apoptosis and inhibits growth of Eca109 and Ec9706 human esophageal cancer cell lines in vitro and invivo. Mol. Med. Rep. 12, 1465–1472.
Lupidi, G., Bramucci, M., Quassinti, L, Fornari, E., Avenali, L, Khalife, H., Gali-Muhtasib, H., 2011. Antiproliferative activities of Artemisia herba-alba ethanolic extract in human colon cancer cell line (HCT116). Alt. Med. Stud. 1, 55–59.
Moding, E.J., Min, H.D., Castle, K.D., Ali, M., Woodlief, L., Williams, N., Ma, Y., Kim, Y., Lee, CL, Kirsch, D.G., 2016. An extra copy of p53 suppresses development of spontaneous Kras-driven but not radiation-induced cancer. JCI Insight 1, e86698.
Nontprasert, A., Pukrittayakamee, S., Dondorp, A.M., Clemens, R., Looareesuwan, S., White, N.J., 2002. Neuropathologic toxicity of artemisinin derivatives in a mouse model. Am. J. Trop. Med. Hyg. 67, 423–429.
Ozaslan, M., Karagoz, I.D., Kılıç, I.H., Guldur, M.E., 2011. Ehrlich ascites carcinoma. African J. Biotech. 10, 2375–2378.
Perry, M.J., 2008. The ChemotherapySource Book. Wolters KluwerHealth/Lippincott Williams and Wilkins, Philadelphia.
Romero, M.R., Efferth, T., Serrano, M.A., Castano, B., Macias, R.I., Briz, O., Marin, J.J., 2005. Effect of artemisinin/artesunate as inhibitors of hepatitis B virus production in an in vitro replicative system. Antiviral Res. 68, 75–83.
Saleh, M.A., Belal, M.H., el-Baroty, G., 2006. Fungicidal activity of Artemisia herba-alba Asso(Asteraceae). J. Environ. Sci. Health B41, 237–244.
Schwartz, S.M., Bennett, M.R., 1995. Death by any other name. Am. J. Pathol. 147, 229–234.
Shaikh, A.Y., Shih, J.A., 2012. Chemotherapy-induced cardiotoxicity. Curr. Heart Failure Rep. 9, 117–127.
Skiker, M., Mekhfi, H., Aziz, M., Haloui, B., Lahlou, S., Legssyer, A., Bnouham, M., Ziyyat, A., 2010. Artemisia herba-alba Asso relaxes the rat aorta through activation of NO/cGMP pathway and K(ATP) channels. J. Smooth Mus. Res. 46, 165–174.
Sriram, M.I., Kanth, S.B., Kalishwaralal, K., Gurunathan, S., 2010. Antitumor activity of silver nanoparticles in Dalton’s lymphoma ascites tumor model. Int. J. Nanomedicine. 5, 753–762.
Tahraoui, A., El-Hilaly, J., Israili, Z.H., Lyoussi, B., 2007. Ethnopharmacological survey of plants used in the traditional treatment of hypertension and diabetes in South-Eastern Morocco (Errachidia province). J. Ethanopharmacol. 110, 105–117.
Tice, R.R., Agurell, E., Anderson, V., Burlinson, B., Hartmann, A., Kobayashi, H., Miyamae, Y., Rojas, E., Ryu, J.C., Sasaki, Y.F., 2000. Single cell gel/comet assay: guidelines for in vitro and in vivo genetic toxicology testing. Environ. Mol. Mutagen. 35, 206e221.
Tilaoui, M., Mouse, A.H., Jaafari, A., Zyad, A., 2015. Comparative phytochemical analysis of essential oils from different biological parts of Artemisia herba alba and their cytotoxic effect on cancer cells. PLoS One 10, e0131799, https://doi.org/10.1371/journal.pone.0131799.
Tounekti, O., Kenani, A., Foray, N., Orlowski, S., Mir, L.M., 2001. The ratio of single- to double-strand DNA breaks and their absolute values determine cell death pathway. Br. J. Canc. 84, 1272–1279.
Van Agtmael, M.A., Cheng-Qi, S., Qing, J.X., Mull, R., Van Boxtel, C.J., 1999. Multiple dose pharmacokinetics of artemether in Chinese patients with uncomplicated falciparum malaria. Int.J. Antimicrob. Agent 12, 151–158.
Vernin, G., Merad, O., Vernin, G.M.F., Zamkotsian, R.M., Párkányi, C., 1995. GC-MS analysis of Artemisia herba alba Asso essential oils from Algeria. Develop. Food Sci. 37, 147–205.
Watanabe, H., Shimokado, K., Asahara, T., Dohi, K., Niwa, O., 1999. Analysis of the c-myc, K-ras and p53 genes in methylcholanthrene-induced mouse sarcomas. Jpn. J. Canc. Res. 90, 40–47.
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors’ contributions
HM and A-EF designed, executed experimental work, performed data analyses and wrote the manuscript. HM revised the final manuscript.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mohamed, H.R.H., Amer, M. & El Faky, A.S.A. Growth retardation and apoptotic death of tumor cells by Artemisia herba-alba oral administration in Ehrlich solid carcinoma bearing mice. Rev. Bras. Farmacogn. 29, 763–772 (2019). https://doi.org/10.1016/j.bjp.2019.06.007
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2019.06.007