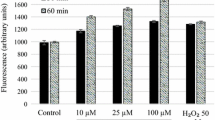
Abstract
Pentavalent antimonials and amphotericin B remain as the main drugs to treat human leishmaniasis. However, the high toxicity and variable efficacy of treatment have stimulated the search for novel drug candidates. Naturally occurring alkaloids have a long history of antileishmanial activity. Here, we investigate the effects of the β-carboline-1-propionic acid alkaloid isolated from Quassia amara L., Simaroubaceae, against Leishmania amazonensis and Leishmania infatum. The alkaloid was isolated after liquid-liquid fractionation followed by chromatographic purification of the Q. amara methanol extract. The antileishmanial activity was evaluated by the microdilution method, using resazurin as the viability indicator. In addition, annexin and propidium iodide were used to detect parasites undergoing apoptosis. The anti-amastigote activity of the β-carboline-1-propionic acid alkaloid was determined by the infection of RAW 264.7 macrophages. The alkaloid displayed leishmanicidal activity against Leishmania amazonensis and L. infantum promastigotes and intracellular amastigotes with 50% inhibitory concentration ranging from 2.7 ± 0.82 to 9.4 ± 0.5 μg/ml and selectivity indexes >10. Moreover, apoptotic Leishmania amazonensis (19.5%) and L. infantum (40.4%) promastigotes were detected after 5 h incubation with the alkaloid. Finally, the β-carboline-1-propionic acid alkaloid inhibited the production of NO of infected macrophages, suggesting that the intracellular amastigote elimination occurs in a nitrosative stress-independent way. The results shown here suggest that the β-carboline-1-propionic acid alkaloid has potential as an antileishmanial agent.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ashok, P., Ganguly, S., Murugesan, S., 2013. Review on in-vitro anti-malarial activity of natural ß-carboline alkaloids. Mini Rev. Med. Chem. 13, 1778–1791.
Ashok, P., Lathiya, H., Murugesan, S., 2015. Manzamine alkaloids as antileishmanial agents: a review. Eur. J. Med. Chem. 97, 928–936.
Ashok, P., Chander, S., Tejeria, A., Garcia-Calvo, L., Balana-Fouce, R., Murugesan, S., 2016. Synthesis and anti-leishmanial evaluation of 1-phenyl-2, 3, 4, 9-tetrahydro-1H-ß-carboline derivatives against Leishmania infantum. Eur. J. Med. Chem. 123, 814–821.
Brasil, P.F., de Freitas, J.A., Barreto, A.L.S., Adade, C.M., Reis de Sá, L.F., Constantino-Teles, P., Toledo, F.T., de Sousa, B.A., Gonçalves, A.C., Romanos, M.T.V., Comasseto, J.V., Dos Santos, A.A., Tessis, A.C., Souto-Padrón, T., Soares, R.M.A., Ferreira-Pereira, A., 2017. Antiproliferative and ultrastructural effects of phenethylamine derivatives on promastigotes and amastigotes of Leishmania (Leishmania) infantum chagasi. Parasitol. Int. 66, 47–55.
Cao, R., Peng, W., Chen, H., Ma, Y., Liu, X., Hou, X., Guan, H., Xu, A., 2005. DNA binding properties of 9-substituted harmine derivatives. Biochem. Biophys. Res. Commun. 338, 1557–1563.
Chen, H., Bai, J., Fang, Z.F., Yu, S.S., Ma, S.G., Xu, S., Li, Y., Qu, J., Ren, J.H., Li, L, Si, Y.K., Chen, X.G., 2011. Indole alkaloids and quassinoids from the stems of Brucea mollis. J. Nat. Prod. 74, 2438–2445.
Chowdhury, S.R., Kumar, A., Godinho, J.L.P., De Macedo Silva, S.T., Zuma, A.A., Saha, S., Kumari, N., Rodrigues, J.C.F., Sundar, S., Dujardin, J.C., Roy, S., De Souza, W., Mukhopadhyay, S., Majumder, H.K., 2017. Voacamine alters Leishmania ultra-structure and kills parasite by poisoning unusual bi-subunit topoisomerase IB. Biochem. Pharmacol. 138, 19–30.
Chua, L.S., Amin, N.A.M., Neo, J.C.H., Lee, T.H., Lee, C.T., Sarmidi, M.R., Aziz, R.A, 2011. LC-MS/MS-based metabolites of Eurycoma longifolia (Tongkat Ali) in Malaysia (Perak and Pahang). J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 879, 3909–3919.
Costa, E.V., Pinheiro, M.L., de Souza, A.D., Barison, A., Campos, F.R., Valdez, R.H., Ueda-Nakamura, T., Filho, B.P., Nakamura, C.V., 2011. Trypanocidal activity of oxoaporphine and pyrimidine-ß-carboline alkaloids from the branches of Annona foetida Mart. (Annonaceae). Molecules 16, 9714–9720.
de Freitas, C.S., Kato, L., de Oliveira, C.M., Queiroz Jr, L.H., Santana, M.J., Schuquel, I.T., Delprete, P.G., da Silva, R.A., Quintino, G.O., da Silva Neto, B.R., Soares, C.M., Pereira, M., 2014. ß-Carboline alkaloids from Galianthe ramosa inhibit malate synthase from Paracoccidioides spp. Planta Med. 80, 1746–1752.
Denton, H., Mcgregor, J.C., Coombs, G.H., 2004. Reduction of antileishmanial pentavalent antimonial drugs by a parasite-specific thiol-dependent reductase, TDR1. Biochem. J. 381, 405–412.
Di Giorgio, C., Delmas, F., Ollivier, E., Elias, R., Balansard, G., Timon-David, P., 2004. In vitro activity of the beta-carboline alkaloids harmane, harmine, and harmaline toward parasites of the species Leishmania infantum. Exp. Parasitol. 106, 67–74.
Gabriel, R.S., Amaral, A.C.F., Corte-Real, S., Lopes, R.C., Alviano, C.S., Vermelho, A.B., Alviano, D.S., Rodrigues, I.A., 2016. Antileishmanial effects of the alkaloid-rich fraction of Quassia amara L. J. Med. Plants Res. 10, 775–782.
Garcia, A.R., Amaral, A.C.F., Azevedo, M.M.B., Corte-Real, S., Lopes, R.C., Alviano, C.S., Pinheiro, A.S., Vermelho, A.B., Rodrigues, I.A., 2017. Cytotoxicity and anti-Leishmania amazonensis activity of Citrus sinensis leaf extracts. Pharm. Biol. 55, 1780–1786.
Green, S.J., Meltzer, M.S., Hibbs Jr, J.B., Nacy, CA, 1990. Activated macrophages destroy intracellular Leishmania major amastigotes by an L-arginine-dependent killing mechanism. J. Immunol. 144, 278–283.
Kanchanapoom, T., Kasai, R., Chumsri, P., Hiraga, Y., Yamasaki, K., 2001. Canthin-6-one and ß-carboline alkaloids from Eurycoma harmandiana. Phytochemistry 56, 383–386.
Kardono, L.B., Angerhofer, C.K., Tsauri, S., Padmawinata, K., Pezzuto, J.M., Kinghorn, A.D., 1991. Cytotoxic and antimalarial constituents of the roots of Eurycoma longifolia. J. Nat. Prod. 54, 1360–1367.
Kumar, A., Chowdhury, S.R., Jatte, K.K., Chakrabarti, T., Majumder, H.K., Jha, T., Mukhopadhyay, S., 2015. Anthocephaline, a new indole alkaloid and cadambine, a potent inhibitor of DNA topoisomerase IB of Leishmania donovani(LdTOP1LS), isolated from Anthocephalus cadamba. Nat. Prod. Commun. 10, 297–299.
Kuo, P.C., Shi, L.S., Damu, A.G., Su, C.R., Huang, C.H., Ke, C.H., Wu, J.B., Lin, A.J., Bastow, K.F., Lee, K.H., Wu, T.S., 2003. Cytotoxic and antimalarial beta-carboline alkaloids from the roots of Eurycoma longifolia. J. Nat. Prod. 66, 1324–1327.
Lala, S., Pramanick, S., Mukhopadhyay, S., Bandyopadhyay, S., Basu, M.K., 2004. Harmine: evaluation of its antileishmanial properties in various vesicular delivery systems. J. Drug Target. 12, 165–175.
Liu, P., Li, H., Luan, R., Huang, G., Liu, Y., Wang, M., Chao, Q., Wang, L., Li, D., Fan, H., Chen, D., Li, L, Matsuzaki, K., Li, W., Koike, K., Zhao, F., 2019. Identification of ß-carboline and canthinone alkaloids as antiinflammatory agents but with different inhibitory profile on the expression of iNOS and COX-2 in lipopolysaccharide-activated RAW 264.7 macrophages. J. Nat. Med. 73, 124–130.
Lumonadio, L., Vanhaelen, M., 1985. Two quassinoid glycosides and a ß-carboline-1-propionic acid from Hannoa klaineana. Phytochemistry 24, 2387–2389.
Machado, M., Dinis, A.M., Santos-Rosa, M., Alves, V., Salgueiro, L., Cavaleiro, C., Sousa, M.C., 2014. Activity of Thymus capitellatus volatile extract, 1, 8-cineole and bor-neol against Leishmania species. Vet. Parasitol. 200, 39–49.
Mishra, B.B., Kale, R.R., Singh, R.K., Tiwari, V.K., 2009. Alkaloids: future prospective to combat leishmaniasis. Fitoterapia 80, 81–90.
Mosmann, T., 1983. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J. Immunol. Methods 65, 55–63.
Nenaah, G., 2010. Antibacterial and antifungal activities of (beta)-carboline alkaloids of Peganum harmala (L) seeds and their combination effects. Fitoterapia 81, 779–782.
Ngoc, P.B., Pham, T.B., Nguyen, H.D., Tran, T.T., Chu, H.H., Chau, V.M., Lee, J.H., Nguyen, T.D., 2016. A new antiinflammatory ß-carboline alkaloid from the hairy-root cultures of Eurycoma longifolia. Nat. Prod. Res. 30, 1360–1365.
Ohmoto, T., Sung, Y.I., Koike, K., Nikaido, T., 1985. Effect of alkaloids of simaroubaceous plants on the local blood flow rate. Japanese J. Pharmacogn. 39, 28–34.
Oryan, A., Akbari, M., 2016. Worldwide risk factors in leishmaniasis. Asian Pac. J. Trop. Med. 9, 925–932.
Rahimi-Moghaddam, P., Ebrahimi, S.A., Ourmazdi, H., Selseleh, M., Karjalian, M., HajHassani, G., Alimohammadian, M.H., Mahmoudian, M., Shafiei, M., 2011. In vitro and in vivo activities of Peganum harmala extract against Leishmania major. J. Res. Med. Sci. 16, 1032–1039.
Rodrigues, I.A., da Silva, B.A., dos Santos, A.L., Vermelho, A.B., Alviano, C.S., Dutra, P.M., Rosa, Mdo S., 2010. A new experimental culture medium for cultivation of Leishmania amazonensis: its efficacy for the continuous in vitro growth and differentiation of infective promastigote forms. Parasitol. Res. 106, 1249–1252.
Srivastava, S., Shankar, P., Mishra, J., Singh, S., 2016. Possibilities and challenges for developing a successful vaccine for leishmaniasis. Parasit. Vectors 9, 277.
Uliana, S.R., Trinconi, C.T., Coelho, A.C., 2017. Chemotherapy of leishmaniasis: present challenges. Parasitology 20, 1–17.
WHO, 2017. Leishmaniasis: Country Profiles — 2015, World Health Organization https://www.who.int/leishmaniasis/burden/Brazil_2015-hl3.pdf?ua=1/ accessed 1 March 2019.
WHO, 2016. Leishmaniasis in high-burden countries: an epidemiological update based on data reported in 2014. World Health Organization/Department of Control of Neglected Tropical Diseases, 2016. Weekly epidemiological record 91, 285–296.
Zhang, M., Sun, D., 2015. Recent advances of natural and synthetic ß-carbolines as anticancer agents. Anticancer Agents Med. Chem. 15, 537–547.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Authors’ contributions
IAR and DSA designed the work. IAR wrote the manuscript. RSG and ARG performed the antileishmanial assays. ACFA, ICL and JDC performed the phytochemical assays and data analysis. RSG and HASS performed the apoptosis assay and data analysis. CMA performed the transmission electron microscopy. ABV and CSA critically revised the manuscript. All the authors have read the final manuscript and approved the submission.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gabriel, R.S., Amaral, A.C.F., Lima, I.C. et al. β-Carboline-1-propionic acid alkaloid: antileishmanial and cytotoxic effects. Rev. Bras. Farmacogn. 29, 755–762 (2019). https://doi.org/10.1016/j.bjp.2019.08.002
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1016/j.bjp.2019.08.002