Abstract
We investigated the risk of stroke related to long-term ambient air pollution exposure, in particular the role of various exposure time windows, using four cohorts from Stockholm County, Sweden. In total, 22,587 individuals were recruited from 1992 to 2004 and followed until 2011. Yearly air pollution levels resulting from local road traffic emissions were assessed at participant residences using dispersion models for particulate matter (PM10) and nitrogen oxides (NOX). Cohort-specific hazard ratios were estimated for time-weighted air pollution exposure during different time windows and the incidence of stroke, adjusted for common risk factors, and then meta-analysed. Overall, 868 subjects suffered a non-fatal or fatal stroke during 238,731 person-years of follow-up. An increment of 20 μg/m3 in estimated annual mean of road-traffic related NOX exposure at recruitment was associated with a hazard ratio of 1.16 (95% CI 0.83–1.61), with evidence of heterogeneity between the cohorts. For PM10, an increment of 10 μg/m3 corresponded to a hazard ratio of 1.14 (95% CI 0.68–1.90). Time-window analyses did not reveal any clear induction-latency pattern. In conclusion, we found suggestive evidence of an association between long-term exposure to NOX and PM10 from local traffic and stroke at comparatively low levels of air pollution.
Similar content being viewed by others
Introduction
Stroke is a leading cause of disease and death in the Western world although the incidence has decreased in recent decades.1 The body of evidence regarding long-term air pollution exposure, especially to respirable particles, and various cardiovascular risk factors and diseases is growing.2 For example, associations have been reported with the development of atherosclerosis, hypertension, ischemic heart disease and cardiovascular mortality. Proposed pathophysiological mechanisms include systemic inflammation and oxidative stress, imbalance in the autonomic nervous system, endothelial dysfunction, vasoconstriction and thrombosis. Although some studies show associations with cerebrovascular incidence or mortality in relation to long-term air pollution exposure, evidence is limited and conflicting.3, 4, 5, 6 Data regarding the role of air pollution exposure for ischemic vs hemorrhagic stroke and the influence of potential effect modifiers are sparse and ambiguous.
In understanding the mechanisms behind effects of long-term exposure to ambient air pollution on cardiovascular disease, as well as to predict consequences of preventive measures, it is important to assess the role of timing of exposure in relation to occurrence of the adverse outcomes. It has been shown, that cardiovascular and other mortality decreased within a year after a coal ban in Dublin, Ireland, which drastically lowered the black smoke levels.7 A follow-up of the Harvard Six City study and a study of myocardial infarction survivors, suggested that the relevant exposure period for mortality was the past few years8, 9 Studies of air pollution effects on cardiovascular disease or mortality have generally not detected marked associations with timing of exposure during follow-up periods of up to two decades.3, 10, 11 Most of the studies on cerebrovascular effects of air pollution, however, did not address this issue.
The aim of the current study was to assess individual long-term exposure to air pollution from road traffic in relation to stroke incidence, in an area with relatively low air pollution levels. We took changes in residence and in annual exposure levels during follow-up into account and investigated different types of stroke, in a combined analysis of four cohorts from Stockholm County, Sweden.
Methods
Study Population
The study included four cohorts based in Stockholm County, Sweden. The Stockholm Diabetes Preventive Program (SDPP),12 a population-based prospective study, recruited 3128 men in 1992–1994 and 4821 women in 1996–1998 from five municipalities in Stockholm County. The study participants were 35–56 years old at recruitment. None had previously diagnosed diabetes and half of the cohort (53%) had a family history of diabetes (one first degree relative or two second degree relatives), while the other half was selected to match on age and sex of the first half. The cohort study of 60 year olds (SIXTY)13 invited a random population sample consisting of one-third of all men and women who were living in Stockholm County and turned 60 years of age between August 1997 and March 1999. The SIXTY study included 4232 subjects. The Screening Across the Lifespan Twin Study (SALT)14 screened all twins born in Sweden before 1958 for the most common complex diseases with a focus on cardiovascular diseases. Recruitment took place during 1998–2002. In the present study, the SALT participants residing in Stockholm County at recruitment were included, resulting in 7043 subjects with an age range of 42–100 years of age at recruitment. The Swedish National study of Aging and Care in Kungsholmen (SNAC-K)15 included randomly sampled individuals >=60 years of age between March 2001 and June 2004 from a central area in Stockholm City. A total of 3363 subjects of 60–104 years of age were recruited.
In all four cohorts, individual data collected at enrolment on socio-demographic characteristics such as occupation status and education were obtained from questionnaires that also provided information on lifestyle factors, including smoking status, levels of physical activity and alcohol consumption. Information on diabetes and hypertension was obtained either from questionnaires or clinical data. Socio-economic variables aggregated at neighborhood level were retrieved from Statistics Sweden. Neighborhoods consisted of small geographical units with an average population of 1000–2000 subjects considered to be homogenous with regard to socio-economic characteristics. The study was approved by the Ethical Review Board Stockholm, Sweden.
Outcome Data
Data on individual stroke events were retrieved from the National Hospital Discharge Registry and the National Cause of Death Registry, including data from February 1964 to December 2011. A stroke event was defined based on the International Classification of Diseases (ICD) version 9 and 10: hospitalizations with principal diagnosis of ischemic stroke (ICD9: 433; 434; ICD10: I63), hemorrhagic stroke (ICD9: 431; ICD10: I61), unspecified stroke (ICD9: 436; ICD10: I64) and out-of-hospital deaths from cerebrovascular diseases (ICD9: 431–436; ICD10: I61-I64). If the person passed away within 28 days after a stroke event, the event was classified as fatal. Only stroke events after recruitment of the respective cohort were included in analyses, whereas earlier events were used to classify later events as non-incident.
Exposure Assessment
Long-term exposure to ambient air pollution from road traffic was estimated based on a methodology described in detail elsewhere.16 Briefly, residential histories retrieved from the Swedish tax authorities were available for all cohort participants from 1991 until 2010, including data on all residential addresses and the date from which the person resided at each particular address. The residential address was known also earlier than 1991 for those moving in before this year. In case of emigration from Sweden or Stockholm County, this was recorded with a specific date used for censoring (see below). The residential addresses within Stockholm County were geocoded, 90% by automatic matching against the Swedish Mapping Cadastral and Land Registration Authority Databases, and an additional 9% manually. One percent of the addresses could not be geocoded, mainly because of insufficient address details.
Annual mean concentrations of nitrogen oxides (NOx) and particulate matter with an aerodynamic diameter of less than 10 micrograms (PM10) were calculated using a wind model and a Gaussian air quality dispersion model, both part of the Airviro Air Quality Management System (SMHI, Norrköping, Sweden; http://airviro.smhi.se). The emission inventory of the Stockholm and Uppsala County Air Quality Management Association supplied the input to the model. Information in this database has been updated yearly by the municipalities in the region since 1993. It is a geographic information system and contains detailed information about emissions from, for example, road and ferry traffic, petrol stations, industrial areas and households.17 For the present study, only emissions from local road traffic were included as it is the dominating source of both NOx and PM10.18, 19 NOx was used as a marker of road traffic-derived combustion pollutants, while PM10 was used as a marker for road wear. Emission factors for NOx and exhaust particles from road traffic were obtained from the EVA model of the Swedish Transport Administration. Emission factors for non-exhaust PM (mainly road wear but including some contributions from brake and tyre wear) were obtained using NOx as tracer for traffic emissions.20 In Stockholm, road wear increases drastically because of the use of studded tyres and traction sand on streets during winter; up to 90% of the locally emitted PM10 may be due to road abrasion.19, 21 The contributions from road, tyre and brake wear in Stockholm is further analysed in a modelling study which clearly shows the dominance of road wear and that the surface moisture, and subsequent retention and suppression of suspension, also influences the PM10 levels.22
The model resolution for inner Stockholm and the urban parts of the municipalities of Solna, Järfälla and Södertälje was 25 m grid cells. The rest of Stockholm County had a resolution of 100 m or 500 m, respectively, in urban and rural areas. The model estimates air pollution concentrations 2 m above ground level and handles buildings by using a roughness parameter.23 This results in underestimated concentrations in street canyons with heavy traffic. Therefore, a street canyon contribution was calculated using the SMHI-Airviro street canyon model (http://airviro.smhi.se). This contribution was added to all addresses with multistory houses on both sides within 30 m of the most polluted street segments in the inner city of Stockholm, and corresponded to air pollution levels at half the building height at those addresses.
The SMHI-Airviro Gaussian model has been validated in a number of previous studies, for example, Johansson et al.24 and Eneroth et al.25 The comparison between time series calculations of annual mean NOx concentrations and urban background measurements from a continuous monitoring station in Stockholm during the period 1998–2005 provided R2 of 0.74–0.80 for different years.26 Within the European Study of Cohorts for Air Pollution Effects (ESCAPE), differences between dispersion model estimates and monitoring results were explored.27 In Stockholm, SMHI-Airviro Gaussian model calculations were compared with measured concentrations of NO2 and PM10, respectively, at 39 and 19 monitoring sites, providing Spearman rank correlations of 0.755 for NO2 and 0.580 for PM10.
Levels of NOx and PM10 resulting from local road traffic emissions were calculated for all geocoded addresses for every year from 1987 until the end of follow-up. Reduced emissions of exhaust particulates and NOx due to stricter European vehicle regulations are included in the EVA emission model. The emission factors for non-exhaust PM were assumed to be constant as the proportion of cars with studded tyres in Stockholm has been relatively stable during the period of the present study. To compensate for trends in traffic volumes, the calculated levels of NOx and PM10 were re-scaled based on measured traffic flow in and out of the regional centre of Stockholm on an annual basis. Concentrations of NO2 were calculated based on modelled concentrations of NOx. An empirical non-linear relationship was derived for each year based on measured concentrations of NOx and NO2 in Stockholm and its surroundings.The annual subject-specific exposure estimates were used to construct study entry and time-weighted exposure concentrations during different time windows.
Statistics
Cohort-specific Cox proportional hazard regression analysis was used to estimate hazard ratios (HRs) of stroke associated with long-term ambient air pollution exposure. NOx and PM10 were analysed separately. Person-time at risk was calculated from enrolment into the study until stroke, death from another cause, emigration (i.e., to an address without information on air pollution exposure) or end of study (31 December 2011), whichever event occurred first. Age was used as the underlying time scale in all models. Risk estimates were calculated as HRs with 95% confidence intervals using increments of 20 μg/m3 for NOx and 10 μg/m3 for PM10. The data were divided into 6-month periods, allowing us to use exposure to PM10 and NOx as time-varying covariates and to adjust for calendar year in 5-year periods. First, concentrations of NOx and PM10 at the study entry address of each individual were used as exposure variables in cohort-specific analyses. Second, the NOx and PM10 exposures were calculated for each subject for each subject-specific 6-month period during the follow-up. Exposure time windows were then created for 6–10-, 4–6-, 2–4- and 0–2-year intervals prior the end date of every 6-month interval during the follow-up.
Adjustment models were defined a priori and covariates were chosen based on the literature and available data from the cohorts. The fully adjusted models had a common set of individual-level covariates including gender, education level, smoking status, smoking intensity among current smokers and socio-economic index. This index was based on current or last (if retired) profession and categorized into low (blue collar worker), medium (low and intermediate level white collar worker, and self-employed) and high (high-level white-collar worker). Additional variables were included if present for at least two cohorts.
The proportional-hazard assumption for all covariates was investigated. If any variable in the individual cohort models violated this assumption, effect estimates were compared with a stratified Cox analysis for that cohort.3 In the analyses, we included only individuals with complete data on exposure estimates and confounders. We did not include confounders with missing data for more than 20% of the individuals; hence for SALT, alcohol consumption and occupational status and for SNAC-K physical activity was excluded.
To increase power, we combined the effect estimates of the four cohorts using a random effect meta-analysis model.28 We investigated heterogeneity between the cohorts using the Higgins I2 statistics.29 Furthermore, we investigated a linear trend between subsequent exposure time windows in time and the risk of stroke. We used the time-window-specific effect estimates as a dependent variable and the time-window intervals as a categorical explanatory variable in a meta-regression model.
Potential effect modification by gender, smoking, hypertension (defined as ≥140 mm Hg systolic or ≥90 mm Hg diastolic BP, or intake of blood pressure-lowering medication, or in the SALT cohort, on self-reported data on prevalent hypertension) and diabetes were investigated by cohort-specific stratified analysis adjusted for the full set of covariates, and then combined into meta-analysis. When analysing effect modification by diabetes, calendar year was recoded into two periods instead of three periods in the model for SIXTY owing to the lack of model convergence when using 5-year intervals. Furthermore, the impact of family history of diabetes on the association between exposure and risk of stroke was investigated in SDPP and SIXTY by stratified analysis. Such data were lacking for the other two cohorts.
Sensitivity analyses were performed by restricting events to (i) only ischemic stroke, (ii) only non-fatal cases and (iii) only including incident cases after study enrolment. In a separate analysis, we explored the influence of contextual confounding by adding neighborhood mean income as an area-level socio-economic variable to the fully adjusted model
All analyses were performed using Stata version 11.0 (StataCorp LP, College Station, TX, USA).
Results
A total of 22,587 subjects were recruited into the four cohorts (Table 1). After exclusion of the subjects recruited into more than one cohort or with missing data in any of the exposures or covariates, 20,070 subjects remained for the analysis. Overall, 6–13% of the subjects in each cohort were excluded owing to missing data.
At study entry, the mean age of all subjects was 60 years (range 35–104), and varying between cohorts (Table 2). Most participants had education up to secondary school or equivalent and were predominantly working or retired. Across cohorts, 15% to 25% of the participants were current smokers and about half reported regular alcohol consumption (daily/weekly). Diabetes prevalence was low, ranging from 1.6% to 8.6% across cohorts, while the proportion of hypertensive individuals was between 22.2% and 69.8% in the different cohorts. Most participants were either in the high or medium socio-economic category, and the average household income in the neighborhood was similar for three cohorts but higher for the cohort located in Stockholm city.
Air pollution linkage was successfully made for 99% of all individual addresses in Stockholm county (n=43,344 addresses).The base-line exposure concentrations of NOx and PM10 were similar for the two cohorts with recruitment in the whole of Stockholm County (SIXTY and SALT), whereas the SNAC-K cohort from Stockholm city had higher exposure levels. The SDPP cohort had the lowest levels and least variability for both PM10 and NOx because of the recruitment of study participants from five suburban and semi-urban municipalities (Figure 1). Average concentrations varied somewhat across the exposure windows and followed the same between-cohort variability as for the study entry exposure data (Supplementary Table 1).
In general, NOx concentrations were reduced during the observation period, whereas levels of PM10 were relatively constant. Modeled NOx and PM10 were highly correlated for all cohorts, where SDPP, SIXTY and SALT had a high Pearson correlation (r ~0.9), whereas SNAC-K had a slightly lower correlation (r=0.75).
A total of 868 subjects suffered a stroke during the 238,731 person-years at risk. Of the subjects, 775 (89%) were first ever cases of stroke after study entry, 755 (87%) were non-fatal and 737 (84%) were ischemic. The occurrence of stroke events were distributed over cohorts accordingly; 130 events in SDPP, 160 events in SIXTY, 314 events in SALT and 264 events in SNAC-K (Table 2).
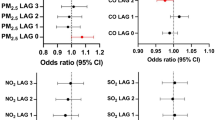
The cohort-specific HR for total stroke per 20 μg/m3 increment of road-traffic-related exposure to NOx at enrolment address ranged between 0.84 and 1.78 (statistically significant only in the SIXTY cohort) when adjusting for all covariates (Figure 2, Supplementary Table 2). The combined HR was 1.16 (0.83–1.61). Similar risk estimates were seen per 10 μg/m3 increase of traffic-related PM10, where the cohort specific HR ranged between 0.59 and 2.21, also significant only in the SIXTY cohort. The combined analysis gave an overall HR of 1.14 (0.68–1.90). Moderate heterogeneity was suggested by the Higgin’s I2 statistic: 53.7% for NOx and 66.9% for PM10. There were no major or consistent differences in risk estimates between crude and adjusted models, indicating only limited confounding by the risk factors under study (Supplementary Table 2). Furthermore, the cohort specific and combined HRs were very similar using NO2 and NOX (Supplementary Figure 1).
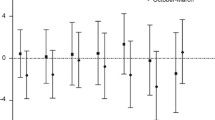
In the time-window analysis for the fully adjusted model, no clear trend in the effect estimates could be detected after meta-analysis (Figure 3, Supplementary Table 3), but the confidence intervals were wide. Results were similar for NOx and PM10 exposure, although a close to statistically significant excess risk was seen for NOx exposure 6–10 years prior to the event. Moderate heterogeneity was also found in all separate time-window meta-analyses, ranging from I2=35.4% to 67.0% for NOx and 58.3% to 67.0% for PM10. There was no significant trend between the effect estimates from the meta-analysed exposure time windows and the time interval they covered. The P-value for time-window category as an explanatory variable for the meta-analysed time-window HRs was not significant (P=0.75). There were rather high correlations in individual exposure between different time windows, that is, 0.7–1 for PM10 and 0.5–0.9 for NOX, which contributes to the similar risk estimates. Only, about half of the study subjects moved during the follow-up period.
No strong effect modification of the association between NOx or PM10 and stroke was observed by gender, smoking, hypertension or diabetes (Supplementary Figure 2). The HRs for NOx and stroke for individuals with heredity for diabetes in SDPP and SIXTY were 2.07 (0.72–5.96) and 1.27 (0.38–4.29), respectively, compared with 1.36 (0.33–5.64) and 1.63 (1.10–2.40) for individuals without heredity for diabetes. Corresponding results for PM10 were 4.10 (0.98–17.11)) and 1.73 (0.39–7.71) for those with heredity for diabetes, respectively, compared with 0.8 (0.10–7.13) and 1.84 (1.10–3.09) for those without (data not shown). Restricting stroke events to non-fatal, ischemic or incident cases as well as adjusting the main model for mean income as area-level socio-economic indicator did not have any major impact on the risk estimates (Supplementary Figure 3). Further sensitivity analyses assessed the potential effect modification by age in the SALT and SNACK cohorts, which included a sizable fraction of older subjects. When individuals older than 75 or 85 years of age were excluded, there was no clear or consistent pattern of changes in the HRs (data not shown).
Discussion
We found suggestive evidence of an association between air pollution from local road traffic, using NOx and PM10 as indicators, and incidence of stroke in a region with comparatively low air pollution levels. No clear differences were indicated in effect estimates between various exposure time windows, but the power was limited in these analyses.
Some heterogeneity between cohorts in risk estimates for stroke related to long-term air pollution exposure was observed. Other studies have shown mixed results with borderline significant associations primarily for fatal stroke, with NO2 (from dispersion modelling with high spatial detail) in Denmark,5 and statistically significant associations for both ischemic and hemorrhagic stroke and NO2 (exposure based on land-use regression models) in Japan.30 The ESCAPE study on incidence of cerebrovascular events found suggestive evidence of an association for stroke and PM2.5, PM 10 and Coarse PM but not for NO2 or NOx (exposure based on land-use regression models).6 On the contrary, studies from England,31 Oslo3 and North America4, 10, 32 did not see any elevated stroke risks associated with air pollution (mainly based on exposure assessment with less geographic detail). A study on women in USA, based on vicinity to urban background monitors, found larger effect estimates for PM2.5 on cerebrovascular events (and death) for within-city exposure differences than for between-city differences, but no effects of NO2.33 These differences indicate that high spatial resolution is needed to describe the air pollution contrasts that may be associated with stroke risk.
One technical difference between our study and others' is that we have estimated the partial contribution to air pollution levels from road traffic only, whereas most other studies have estimated total levels. Within the Stockholm region, however, the local spatial differences in residential levels of both PM10 and NOx are dominated by the emissions from road traffic.34 Adding a regional background effect to all estimated values would not have changed our results, which were based on absolute rather than relative differences in exposure.
It is of particular interest to compare our results with those of the recently published study on cerebrovascular events from the ESCAPE project in which 20% of the stroke cases were in the four cohorts in the present study. Our exposure assessment was based on dispersion modeling, whereas ESCAPE used land use regression and our region constituted the lowest exposed area in ESCAPE, particularly for NOx. We found suggestive evidence of associations for both NOx and PM10, whereas no association was observed for NOx in ESCAPE based on exposure at residential address at study enrolment. Furthermore, the magnitude of the effect was similar for PM10 in the two studies (14% and 11% per 10 μg/m3). Notably, the cohort-specific effect estimates for long-term air pollution exposure and stroke in this study differed somewhat from the estimates presented for the same cohorts in the ESCAPE study (ESCAPE data retrieved through author correspondence). These variations could to some extent be explained by slightly diverse model adjustments, but a more probable explanation is the choice of exposure modeling technique. A major uncertainty with employment of both exposure assessment methodologies in most epidemiological studies is the failure to consider exposure contributions from occupational locations and commuting.35 This generally would be expected to contribute to dilution of the associations.
Consistently with other European studies on long-term effects of air pollution on stroke, we did not find a difference between associating ischemic stroke and all stroke cases to PM10 and NOx. Although short-term studies generally suggest a stronger association with ischemic stroke,5, 36, 37, 38, 39, 40 the literature on chronic effects of air pollution and types of stroke is sparse and results are mixed. A case–control study of ischemic stroke hospitalization in southern Sweden and yearly mean NOx averages prior to events showed no significant association.41 In a Danish study using address-specific NO2 concentrations weighted over 9.8 years, the strongest associations were found for non-specified and ischemic strokes whereas no association was found for hemorrhagic stroke.5 A US study did not find a significant association between stroke type and the interquartile range (4 μg/m3) change in average PM2.5, PM10–2.5 or PM10 exposure in the 12 months prior event in a cohort including only men.10 Studies from Asia have linked air pollution to both ischemic and hemorrhagic stroke30, 42 where one long-term study found a significant association of yearly mean NO2 levels from 1, 2 and 3 fiscal years prior a ischemic stroke and 2–3 years prior a hemorrhagic event.30 In general, hemorrhagic stroke is less common than ischemic stroke, which leads to lower statistical precision and power in detecting risks for this type of stroke.
A recent review of epidemiological evidence on long-term exposure to air pollution and cardio-respiratory mortality found significant heterogeneity in PM2.5 effect estimates across studies.43 It was suggested that this was related to differences in particle composition, infiltration of particles indoors, population characteristics and methodological differences in exposure assessment and confounder control. In the ESCAPE study on cerebrovascular events based on 11 cohorts from 7 European countries,6 heterogeneity was found for all exposure metrics but NOx and the coarse PM fraction. Age was proposed as a major heterogeneity source but was suggested to correlate with other cohort characteristics.
On the other hand, studies within the ESCAPE project on acute coronary events44 using the same cohorts, and on mortality adding 11 cohorts,45 failed to detect such heterogeneity. In our study, we detected between-cohort heterogeneity in the effect estimates for long-term exposure to NOX and PM10 on stroke incidence, even though the cohorts were based in only one region. The two cohorts in our study not showing associations differed from the other in certain aspects. One (SALT) lacked information on occupation status and alcohol consumption, available in all or most other cohorts, which probably led to poorer confounding control. The other (SNAC-K) was considerably older, with ages up to 105 years at recruitment, where less susceptible “survivors” may have been enriched
SNAC-K also differed considerably from the other cohorts in regard to prevalence of hypertension. On the other hand, the SDPP cohort (where associations were suggested) was selected so that diabetes heredity was more common, and those with such heredity appeared at higher risk of stroke associated with air pollution exposure. This cohort also had a longer observation period than the other because of earlier recruitment. However, taken together, we cannot find explanations that fully account for the observed heterogeneity.
A strength of our study consisted in the detailed assessment of air pollution for each subject, which took changes in residential address and calendar time into account. In addition, individual data on many cardiovascular risk factors were available along with information on potential contextual confounders. National health registries that are validated, for example regarding stroke,46 were used to obtain the outcome data which minimized the risk of misclassification, although the quality of differentiation between ischemic and hemorrhagic stroke was lower during the earlier years of follow-up. Furthermore, the individual cohorts were rather small limiting the statistical power to detect associations, particularly in analyses of subgroups and interactions.
In conclusion, our findings indicated a possible association between local air pollution from road traffic and incidence of stroke in a combined analysis of four cohorts from Stockholm County. No clear differences in risk related to time windows of exposure were seen, but the interpretation was hampered by a limited statistical power in these analyses.
References
Feigin VL, Forouzanfar MH, Krishnamurthi R, Mensah GA, Connor M, Bennett DA et al. Global and regional burden of stroke during 1990-2010: findings from the Global Burden of Disease Study 2010. Lancet 2014; 383: 245–254.
Brook RD, Rajagopalan S, Pope CA, 3rd, Brook JR, Bhatnagar A, Diez-Roux AV et al. Particulate matter air pollution and cardiovascular disease: An update to the scientific statement from the American Heart Association. Circulation 2010; 121: 2331–2378.
Nafstad P, Haheim LL, Wisloff T, Gram F, Oftedal B, Holme I et al. Urban air pollution and mortality in a cohort of Norwegian men. Environ Health Perspect 2004; 112: 610–615.
Pope CA, 3rd, Burnett RT, Thurston GD, Thun MJ, Calle EE, Krewski D et al. Cardiovascular mortality and long-term exposure to particulate air pollution: epidemiological evidence of general pathophysiological pathways of disease. Circulation 2004; 109: 71–77.
Andersen ZJ, Kristiansen LC, Andersen KK, Olsen TS, Hvidberg M, Jensen SS et al. Stroke and long-term exposure to outdoor air pollution from nitrogen dioxide: a cohort study. Stroke 2012; 43: 320–325.
Stafoggia M, Cesaroni G, Peters A, Andersen ZJ, Badaloni C, Beelen R et al. Long-term exposure to ambient air pollution and incidence of cerebrovascular rvents: Results from 11 European cohorts within the ESCAPE Project. Environ Health Perspect 2014; 122: 919–925.
Clancy L, Goodman P, Sinclair H, Dockery DW . Effect of air-pollution control on death rates in Dublin, Ireland: an intervention study. Lancet 2002; 360: 1210–1214.
Laden F, Schwartz J, Speizer FE, Dockery DW . Reduction in fine particulate air pollution and mortality: Extended follow-up of the Harvard Six Cities study. Am J Respir Crit Care Med 2006; 173: 667–672.
Zanobetti A, Schwartz J . Particulate air pollution, progression, and survival after myocardial infarction. Environ Health Perspect 2007; 115: 769–775.
Puett RC, Hart JE, Suh H, Mittleman M, Laden F . Particulate matter exposures, mortality, and cardiovascular disease in the health professionals follow-up study. Environ Health Perspect 2011; 119: 1130–1135.
Chen R, Zhang Y, Yang C, Zhao Z, Xu X, Kan H . Acute effect of ambient air pollution on stroke mortality in the China air pollution and health effects study. Stroke 2013; 44: 954–960.
Eriksson AK, Ekbom A, Granath F, Hilding A, Efendic S, Ostenson CG . Psychological distress and risk of pre-diabetes and Type 2 diabetes in a prospective study of Swedish middle-aged men and women. Diabet Med 2008; 25: 834–842.
Wandell PE, Wajngot A, de Faire U, Hellenius ML . Increased prevalence of diabetes among immigrants from non-European countries in 60-year-old men and women in Sweden. Diabetes Metab 2007; 33: 30–36.
Lichtenstein P, Sullivan PF, Cnattingius S, Gatz M, Johansson S, Carlstrom E et al. The Swedish Twin Registry in the third millennium: an update. Twin Res Hum Genet 2006; 9: 875–882.
Meinow B, Kareholt I, Lagergren M . According to need? Predicting the amount of municipal home help allocated to elderly recipients in an urban area of Sweden. Health Soc Care Community 2005; 13: 366–377.
Bellander T, Berglind N, Gustavsson P, Jonson T, Nyberg F, Pershagen G et al. Using geographic information systems to assess individual historical exposure to air pollution from traffic and house heating in Stockholm. Environ Health Perspect 2001; 109: 633–639.
Johansson C, Hadenius A, Johansson P-Å, Jonson T . SHAPE: The Stockholm study on health effects of air pollution and their economic consequences. Part 1: NO2 and particulate matter in Stockholm [Internet] 1999[cited 2015 Jan 23] 1999: 41. Available from http://slb.nu/slb/rapporter/pdf8/lvf1998_006.pdf.
Johansson C, Eneroth K . TESS–Traffic Emissions, Socioeconomic valuation and Socioeconomic measures, Part 1: Emissions and exposure of particles and NOx in Greater Stockholm. Stockholm and Uppsala County Air Quality Management Association Report LVF [Internet]. 2007:2. [cited 2015 Jan 23] Available from http://slb.nu/slb/rapporter/pdf8/lvf2007_002.pdf.
Johansson C, Norman M, Gidhagen L . Spatial & temporal variations of PM10 and particle number concentrations in urban air. Environ Monit Assess 2007; 127: 477–487.
Omstedt G, Bringfelt B, Johansson C . A model for vehicle-induced non-tailpipe emissions of particles along Swedish roads. Atmospheric Environment 2005; 39: 6088–6097.
Sjödin Å, Ferm M, Björk A, Rahmberg M, Gudmundsson A, Swietlicki E et al. Wear particles from road traffic-a field, laboratory, and modelling study. Goteborg. IVL Swedish Environmental Research Institute Ltd.[ Internet]: Sweden, 2010 [cited 2015 Jan 23] Available from http://www.ivl.se/webdav/files/B-rapporter/B1830.pdf.
Denby B, Sundvor I, Johansson C, Pirjola L, Ketzel M, Norman M et al. A coupled road dust and surface moisture model to predict non-exhaust road traffic induced particle emissions (NORTRIP). Part 1: Road dust loading and suspension modelling. Atmospheric Environment 2013; 77: 283–300.
Gidhagen L, Johansson C, Langner J, Foltescu V . Urban scale modeling of particle number concentration in Stockholm. Atmospheric Environment 2005; 39: 1711–1725.
Johansson C, Hadenius A, Johansson P-Å, Jonson T . SHAPE: The Stockholm study on health effects of air pollution and their economic consequences. Part 1: NO2 and particulate matter in Stockholm VAEGVERKET. PUBLIKATION [Internet]. 1999: 41[cited 2015 Jan 23] 1401-9612 Available from http://www.ivl.se/webdav/files/B-rapporter/B1830.pdf.
Eneroth K, Johansson C, Bellander T . Exposure comparison between measurements and calculations based on dispersion modelling (EXPOSE). Stockholm Environment and Health Protection Administration[Internet]. 2006. [cited 2015 Jan 23] Available from http://slb.nu/slb/rapporter/pdf8/lvf2006_012.pdf.
Johansson C, Andersson C, Bergström R, Krecl P . Exposure to particles due to local and non-local sources in Stockholm: estimates based on modelling and measurements 1997-2006. ITM RAPPORT 2008. Department of Applied Environmental Science, Stockholm University: Stockholm (Sweden), ITM report 175.
de Hoogh K, Korek M, Vienneau D, Keuken M, Kukkonen J, Nieuwenhuijsen MJ et al. Comparing land use regression and dispersion modelling to assess residential exposure to ambient air pollution for epidemiological studies. Environ Int 2014; 73: 382–392.
DerSimonian R, Laird N . Meta-analysis in clinical trials. Control Clin Trials 1986; 7: 177–188.
Higgins JP, Thompson SG . Quantifying heterogeneity in a meta-analysis. Stat Med 2002; 21: 1539–1558.
Yorifuji T, Kashima S, Tsuda T, Ishikawa-Takata K, Ohta T, Tsuruta K et al. Long-term exposure to traffic-related air pollution and the risk of death from hemorrhagic stroke and lung cancer in Shizuoka, Japan. Sci Total Environ 2013; 443: 397–402.
Atkinson RW, Carey IM, Kent AJ, van Staa TP, Anderson HR, Cook DG . Long-term exposure to outdoor air pollution and incidence of cardiovascular diseases. Epidemiology 2013; 24: 44–53.
Chen H, Goldberg MS, Burnett RT, Jerrett M, Wheeler AJ, Villeneuve PJ . Long-term exposure to traffic-related air pollution and cardiovascular mortality. Epidemiology 2013; 24: 35–43.
Miller KA, Siscovick DS, Sheppard L, Shepherd K, Sullivan JH, Anderson GL et al. Long-term exposure to air pollution and incidence of cardiovascular events in women. N Engl J Med 2007; 356: 447–458.
Täppefur M . Air pollution in Stockholm and Uppsala Counties, and in Gävle and Sandviken municipalities. Emission data for year 2011. SLB-ANALYS. 2011; APRIL 2013.
Ozkaynak H, Baxter LK, Dionisio KL, Burke J . Air pollution exposure prediction approaches used in air pollution epidemiology studies. J Expo Sci Environ Epidemiol 2013; 23: 566–572.
Vidale S, Bonanomi A, Guidotti M, Arnaboldi M, Sterzi R . Air pollution positively correlates with daily stroke admission and in hospital mortality: a study in the urban area of Como, Italy. Neurol Sci 2010; 31: 179–182.
Wellenius GA, Schwartz J, Mittleman MA . Air pollution and hospital admissions for ischemic and hemorrhagic stroke among medicare beneficiaries. Stroke 2005; 36: 2549–2553.
Chan CC, Chuang KJ, Chien LC, Chen WJ, Chang WT . Urban air pollution and emergency admissions for cerebrovascular diseases in Taipei, Taiwan. Eur Heart J 2006; 27: 1238–1244.
Andersen ZJ, Olsen TS, Andersen KK, Loft S, Ketzel M, Raaschou-Nielsen O . Association between short-term exposure to ultrafine particles and hospital admissions for stroke in Copenhagen, Denmark. Eur Heart J 2010; 31: 2034–2040.
Oudin A, Stromberg U, Jakobsson K, Stroh E, Bjork J . Estimation of short-term effects of air pollution on stroke hospital admissions in southern Sweden. Neuroepidemiology 2010; 34: 131–142.
Oudin A, Stroh E, Stromberg U, Jakobsson K, Bjork J . Long-term exposure to air pollution and hospital admissions for ischemic stroke. A register-based case-control study using modelled NO(x) as exposure proxy. BMC Public Health 2009; 9: 301.
Yamazaki S, Nitta H, Ono M, Green J, Fukuhara S . Intracerebral haemorrhage associated with hourly concentration of ambient particulate matter: case-crossover analysis. Occup Environ Med 2007; 64: 17–24.
Hoek G, Krishnan RM, Beelen R, Peters A, Ostro B, Brunekreef B et al. Long-term air pollution exposure and cardio- respiratory mortality: a review. Environ Health 2013; 12: 43.
Cesaroni G, Forastiere F, Stafoggia M, Andersen ZJ, Badaloni C, Beelen R et al. Long term exposure to ambient air pollution and incidence of acute coronary events: prospective cohort study and meta-analysis in 11 European cohorts from the ESCAPE Project. BMJ 2014; 348: f7412.
Beelen R, Raaschou-Nielsen O, Stafoggia M, Andersen ZJ, Weinmayr G, Hoffmann B et al. Effects of long-term exposure to air pollution on natural-cause mortality: an analysis of 22 European cohorts within the multicentre ESCAPE project. Lancet 2014; 383: 785–795.
Koster M, Asplund K, Johansson A, Stegmayr B . Refinement of Swedish administrative registers to monitor stroke events on the national level. Neuroepidemiology 2013; 40: 240–246.
Acknowledgements
This project was funded by the Swedish Environmental Protection Agency, the Swedish Council for Working Life and Social Research, and the Swedish Heart-Lung Foundation. The SDPP cohort was additionally funded by the Stockholm County Council, the Swedish Research Council, the Swedish Diabetes Association and the Novo Nordisk Scandinavia. The SIXTY cohort was additionally funded by the Stockholm County Council and the Swedish Research Council (longitudinal research and 0593). The SALT cohort was additionally supported by NIH grant AG-08724. We acknowledge the Stockholm and Uppsala County Air Quality Management Association for enabling use of their emission inventory.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Additional information
Supplementary Information accompanies the paper on the Journal of Exposure Science and Environmental Epidemiology website
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
About this article
Cite this article
Korek, M., Bellander, T., Lind, T. et al. Traffic-related air pollution exposure and incidence of stroke in four cohorts from Stockholm. J Expo Sci Environ Epidemiol 25, 517–523 (2015). https://doi.org/10.1038/jes.2015.22
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jes.2015.22
- Springer Nature America, Inc.
Keywords
This article is cited by
-
Associations between short-term exposure to ambient PM2.5 and incident cases of cerebrovascular disease in Yantai, China
Environmental Science and Pollution Research (2022)
-
Association between exposure to ambient air pollution and hospital admission, incidence, and mortality of stroke: an updated systematic review and meta-analysis of more than 23 million participants
Environmental Health and Preventive Medicine (2021)
-
Health and economic impacts from PM2.5 pollution transfer attributed to domestic trade in China: a provincial-level analysis
Environmental Science and Pollution Research (2021)
-
Ambient fine particulate matter of diameter ≤ 2.5 μm and risk of hemorrhagic stroke: a systemic review and meta-analysis of cohort studies
Environmental Science and Pollution Research (2021)
-
A comprehensive evaluation of the association between ambient air pollution and adverse health outcomes of major organ systems: a systematic review with a worldwide approach
Environmental Science and Pollution Research (2019)