Abstract
Cholangiocarcinoma (CCA), which is a poor prognosis malignancy that arises from the malignant transformation of cholangiocytes, is associated with chronic inflammation of the biliary epithelium. Thus far, the molecular mechanisms of the origin and neoplastic processes of CCA that are promoted by inflammation are still unclear and need to be fully elucidated. Here using small RNA sequencing to determine the microRNA (miRNA) expression profiles in CCA, we found that let-7c, miR-99a and miR-125b, which are three miRNAs of the same cluster, were downregulated in CCA and targeted interleukin 6 (IL-6), IL-6R and type 1 insulin-like growth factor, which are important cytokines and receptors of the IL-6/signal transducer and activator 3 (STAT3) pathway and have key roles in inflammation and CCA initiation. We also found that enforced expression of let-7c, miR-99a or miR-125b could reduce the activity of STAT3 and further suppress CCA tumorigenicity in vivo and inhibit the migration and invasion of CCA cells in vitro. Surprisingly, let-7c/miR-99a/miR-125b cluster also significantly decreased the ability of CCA cells for cancer stem cell-like mammosphere generation by downregulating CD133 and CD44, which suggests the pivotal roles of let-7c, miR-99a and miR-125b in CCA by regulating both inflammation and stem-like properties. Our findings showed potential links between miRNAs and inflammation, and provide a potential treatment strategy for developing an miRNA-based therapy via IL-6/STAT3 targeting for CCA.
Similar content being viewed by others
Introduction
Cholangiocarcinoma (CCA) is the second-most common primary hepatobiliary malignancy and is generally believed to be a result of malignant transformation of cholangiocytes that line the intrahepatic and extrahepatic biliary tract.1 The incidence of CCA is increasing worldwide, and the overall survival rate is <5% of patients surviving to 5 years.2 Currently, conventional chemotherapy and radiation therapy have been notably ineffective in improving long-term survival, and the only curative treatment for CCA is surgical resection of the tumor.2 Therefore, an understanding of the cellular mechanisms underlying CCA pathogenesis is urgently needed to develop efficient treatments. The known risk factors account for only a few cases of CCA and are associated with chronic inflammation of the biliary system; these include hepatolithiasis, liver fluke infection, primary sclerosing cholangitis and choledochal cysts.3 In recent years, accumulating evidences also suggest the potential roles of CCA stem cells in CCA pathology. Some membrane glycoproteins, including CD133 and CD44, have pivotal roles in cancer stem cells of hepatobiliary malignancy, and have been used as stem cell biomarkers for stem-like cells, however, the underlying mechanisms of CCA stem cells are still not clear yet.4, 5, 6, 7
Inflammation, one of the most studied risk factors of CCA, favors tumorigenesis by stimulating angiogenesis, damaging DNA, maintaining stem cells in tumor microenvironment and chronically stimulating cell proliferation.8, 9 Thus, studies of inflammation-associated cytokines on the pathogenesis and growth of CCA are of great significance.10 Interleukin 6 (IL-6) is one of the most important multifunctional cytokines in the response of hepatobiliary epithelia to inflammation.11 An increased concentration of IL-6 in the bile and serum has been identified as contributing to cholangeitis and abscessus.12, 13, 14, 15 The production of IL-6 through an autocrine–paracrine mechanism by biliary epithelial cells or cholangiocytes mediates the acute phase and immune responses,16 and can influence the growth of normal and tumor cells and contribute to tumor formation.17 IL-6 imparts its signal into the cell by forming a complex with the plasma membrane proteins IL-6R and gp130, which results in the activation of Janus kinases (JAKs). The JAKs phosphorylate signal transducer and activator (STAT) proteins, predominantly STAT3 for IL-6, at residue tyrosine 705. The phosphorylated STAT3 dimerizes and translocates to the nucleus, where it binds to DNA and triggers the transcription of target genes, including oncogenes, survival genes, cell cycle regulators, cytokines and molecular modulators of angiogenesis and migration.18, 19, 20, 21 The activation of STAT3 promotes tumorigenesis and progression to aggressiveness. The receptor for type 1 insulin-like growth factor (IGF1R) has also been identified as an important factor for cellular transformation, malignant progression and cancer cell survival.22 Cross-activation of IGF1R signaling and the IL-6/STAT3 pathway have also been described.23, 24, 25 Several studies suggest an important role for an activated IL-6/STAT3 pathway in the pathogenesis or progression of CCA.10, 11, 20, 26 However, the mechanism by which aberrant activation of the IL-6/STAT3 pathway contributes to tumorigenesis is largely unknown.
MicroRNAs (miRNAs) are a class of small, noncoding RNAs of approximately 22 nucleotides in length that can silence specific target genes through translational repression or direct mRNA degradation by binding to 3′ untranslated regions (3′ UTR).27 These exist stably in various tissues and control a number of fundamental biological processes. In addition, aberrant expression of miRNAs is reported in various diseases, including CCA. However, the deregulation of miRNAs in CCA remains unclear, and the mechanisms that regulate the inflammatory pathway in the pathogenesis and progression of CCA are poorly understood. In this study, we investigated the miRNA expression profiles in human CCA using small RNA sequencing and found that downregulation of let-7c, miR-99a and miR-125b, which belong to the same miRNA cluster, were associated with activation of the IL-6/STAT3 pathway. Exogenous expression of these miRNAs reduced the activity of STAT3 and inhibited the malignant transformation of CCA cells and tumorigenicity in mice. These results suggest that these miRNAs may serve as pivotal pathogenesis factors, at least in part, by regulating the IL-6/STAT3 signaling pathway.
Results
RNA sequencing identified a novel set of miRNAs that were differentially expressed in CCA
To assess the deregulation of global miRNA expression, we investigated the miRNA spectrum from the pools of three moderately poorly differentiated and three well-differentiated CCA tumors (T), as well as pools of their peritumoral (N) tissues. Overall, 1733 miRNAs, including miRNA's, were evaluated, and the relative expression of each miRNA was examined: 652 were detected in sequencing samples, and only 125 miRNAs had >200 r.p.m. in at least one sample. To confirm the library sequencing, we tested the expression of some randomly selected miRNAs in the pooled samples using quantitative reverse transcriptase–PCR. The results from the two detection methods were coherent (Supplementary Figure 1).
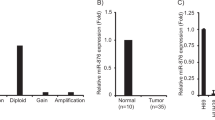
Of the 125 miRNAs, with fold changes >2.0 and P-values <0.001 when compared with the normal group, only 10 or 33 miRNAs were upregulated and 21 or 42 miRNAs were downregulated in the well- or moderately poorly differentiated tumor groups, respectively (Supplementary Table 2). Eight miRNAs were differentially expressed in the well and moderately poorly differentiated CCA samples compared with the normal sample: three of the miRNAs (miR-31, miR-200c and miR-141) were upregulated, and five miRNAs (miR-451, miR-486-5p, miR-101, miR-130a and miR-122) were downregulated in the well- and moderately poorly differentiated CCA. To further investigate the expression patterns of miRNAs in CCA, we used quantitative reverse transcriptase–PCR to determine the expression of the miRNAs with important roles in carcinoma, especially in hepatobiliary cancer, which were differentially expressed based on the sequencing data. We found that miR-122, miR-32, miR-101, let-7c, miR-99a and miR-125b were significantly (P<0.05) downregulated, whereas miR-200c, miR-21 and miR-221 were significantly (P<0.05) upregulated in 24 paired tumor tissues compared with normal tissue (Figure 1). Therefore, these miRNAs may have important roles in CCA.
miRNAs are differentially expressed in CCA and adjacent tissues. miR-32, miR-122, miR-101, let-7c, miR-99a and miR-125b were significantly downregulated (P<0.05), whereas miR-200c, miR-21 and miR-221 were significantly upregulated (P<0.05). dCt=CtmicroRNA – Ctinternal control. All indicated P-values were determined using a two-tailed, paired t-test.
The IL-6/ STAT3 pathway was found to be a prime target for deregulated miRNAs in CCA
Inflammation-associated cytokines and their receptors have been reported having pivotal roles in CCA and most of them serve as important risk factors.10, 28 To screen and investigate whether the differentially expressed miRNAs identified above directly target to the inflammation-associated genes or pathway, we focused on the most common CCA-associated inflammation cytokines include IL-6,10, 26 IL-8,29 IGF1,30 transforming growth factor-β31, 32, 33 and VEGF,33 and predicted the targeting relationship between the deregulated miRNAs in CCA and these cytokines and receptors using Starbase v2.0, which is a database that combines prediction algorithms of miRNA-mRNA interactions and large-scale CLIP-Seq data.34 The results showed that most of these miRNAs could interact with at least one of the cytokines or receptors, as shown in Figure 2, which implies that many miRNAs may be involved in CCA by regulating these inflammation-associated cytokine pathways. Interestingly, among the deregulated miRNAs, we found that miR-99a, let-7c and miR-125b, which belong to the same cluster located on chromosome 21q21,35 could regulate all of the mentioned inflammation-associated cytokine pathways that are involved in CCA. As miRNAs that originate from the same genomic cluster may be co-expressed and involved in pathways that cooperatively contribute to cell progression, we speculated that the let-7c/miR-99a/miR-125b cluster may be an important regulator involved in CCA by regulating cytokine-induced inflammatory pathways.
To investigate whether the let-7c/miR-99a/miR-125b cluster acts synergistically in CCA, we first detected the expression levels of miR-99a, let-7c and miR-125b in cancerous tissues of CCA patients, and analyzed the correlation relationships of three miRNAs using Pearson correlation test. We found that these miRNAs were concordantly expressed, that is, a patient with a high expression level of any one of let-7c/miR-99a/miR-125b cluster also showed high expression levels of the other two (Figure 3a). Next, we investigate whether these three co-expressed miRNAs were involved in cooperative pathways. As IL-6 is one of most important inflammation cytokines involved in pathogenesis and growth of CCA,10, 11 and the interaction of IL-6 with IL-6R that results in the activation of STAT3 have been demonstrated importance in inflammation and cancer initiation,18 we chose the IL-6/STAT3 inflammatory pathways to experimentally address the miRNA target interactions with inflammation cytokines and receptors. As shown in Figure 2, the inflammation factor IL-6 was predicted to be a target of let-7c, and its receptor, IL-6R, was a potential target of let-7c and miR-125b. In addition, IGF1R was a predicted target of miR-99a. IGF signaling could activate the STAT3 signaling pathway.22, 23, 24 We co-transfected the individual miRNAs (miR-99a, let-7c or miR-125b) with a Renilla luciferase reporter construct containing the wild type (WT) or mutated versions of the putative mRNA 3′ UTR fragment into HEK-293T cells (RNA sequences in Figure 3b). Relative to the nonspecific control miRNA, co-transfection with each miRNA significantly suppressed luciferase activity of the vector containing the WT 3′ UTR compared with the empty vector and the mutated version (histograms in Figure 3b). Moreover, overexpression of let-7c, miR-99a or miR-125b in lentivirus-infected CCA cell lines (Supplementary Figure 2) reduced the protein expression of their corresponding targets (let-7c targets IL-6, let-7c/miR-125b targets IL-6R and miR-99a targets IGF1R) (Figure 3c). Together, these data demonstrate that let-7c and miR-125b are able to regulate IL-6R expression, let-7c can also target IL-6, and IGF1R is the bona fide target of miR-99a.
IGF1R, IL-6R and IL-6 are direct targets of let-7c/miR-99a/miR-125b. (a) The positive correlation among miR-99a, let-7c and miR-125b, correlation analysis was determined using the Pearson product–moment correlation. (b) Schematic representation of the interaction of an miRNA with the 3′ UTR of its corresponding target. Each predicted MRE 3′ UTR was inserted into a psiCHECK-2 vector immediately downstream from the Renilla luciferase gene. Luciferase reporter assays analyzing the putative targets of the three miRNAs. HEK-293T cells were co-transfected with psiCHECK-2 plasmids with the 3′ UTR of the target genes or the target genes with mutated miRNA-binding sites (Mut) and miRNA mimics or negative control mimics (NC) using Lipofectamine 2000. Firefly luciferase activity was normalized to Renilla luciferase activity, and the results were expressed relative to the control. (c) QBC939, MZ-cha-1 and SK-cha-1 cells were infected by lentivirus overexpressing miRNA or negative controls, and cells lysates were prepared for western blotting with antibodies against IGF1R, IL-6R or IL-6. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) expression served as a loading control, and densitometric ratios were recorded. Experiments were performed in triplicate, and all indicated P-values were determined using t-test. *P<0.05; **P<0.01.
As IL-6, IL-6R and IGF1R are associated with IL-6/STAT3 signaling, we further tested whether let-7c, miR-99a and miR-125b could affect the activity of STAT3. Treatment of CCA cells with IL-6 for the indicated time induced and maintained STAT3 phosphorylation in control CCA cells. Previous studies suggested that phosphorylated STAT3 is not expressed without IL-6 stimulation and peaked on 0.5 h after IL-6 stimulation,20, 36 thus in the study we only detected STAT3 phosphorylation level on 0.5, 1 and 2 h after IL-6 stimulation. We found that exogenous let-7c, miR-99a and miR-125b promoted the reduction of phosphorylated STAT3 and eliminated the activity of STAT3 (Figure 4a). These experiments further support let-7c/miR-99a/miR-125b having important roles in the IL-6/STAT3 signaling pathways.
The IL-6/STAT3 pathway is regulated by let-7c/miR-99a/miR-125b in CCA cell lines and clinical samples. (a) Treatment of QBC939, MZ-cha-1 and SK-cha-1 cells with IL-6 for 0.5, 1, or 2 h, respectively, induced and maintained STAT3 phosphorylation; and exogenous let-7c, miR-99a and miR-125b eliminated the activity of STAT3. (b) Downregulation of let-7c/miR-99a/miR-125b in clinical CCA samples resulted in the upregulation of IL-6R and IGF1R. GADPH expression served as a loading control and densitometric ratios were recorded. Experiments were performed in triplicate.
We next investigated whether downregulation of miRNAs in clinical CCA samples resulted in the upregulation of their targets. We evaluated the protein expression levels of IGF1R, IL-6R and IL-6 as well as STAT3 phosphorylation in 18 randomly paired protein available CCA samples using western blotting. The results showed that IL-6R, IGF1R and pSTAT3 were elevated in approximately 77.8% (14/18), 55.6% (10/18) and 72.2% (13/18) of tumor tissues, respectively (Figure 4b and Supplementary Figure 3). Unfortunately, the expression of IL-6 was barely detectable, which could be caused by excretion out of the cell and maintenance in tumor tissues.
All together, these data demonstrated that the IL-6/STAT3 pathway is under the control of let-7c/miR-99a/miR-125b, and that aberrant activation of the IL-6/STAT3 pathway by the downregulation of let-7c/miR-99a/miR-125b may cause the pathogenesis of CCA.
Enforced expression of let-7c/miR-99a/miR-125b inhibited CCA mammosphere formation in vitro
Many studies have revealed that the activation of IL-6/STAT3 signaling is an important contributor to inflammation-induced cancer.18, 19, 20 To verify the impact of let-7c/miR-99a/miR-125b on the pathogenesis of CCA, we first used a mammosphere culture system to evaluate the function of these miRNAs on the capability of CCA cells in malignancy transformation.37 Single-cell suspensions of CCA cells were grown in conditions that do not allow for adherence to a substratum. Most CCA cells died under these conditions, but a small number survived and generated floating, spherical colonies, and these cells showed increased potential for self-renewal and differentiation and presented more stem-like, malignant cells.
MZ-cha-1 and SK-cha-1 cells were cultured in suspension (plated on ultra-low attachment plates) to generate mammospheres, but not under adherent conditions (Figure 5a). The expression levels of let-7c/miR-99a/miR-125b were all substantially downregulated in mammosphere cells compared with non-mammosphere cells (Figure 5a) in both cell lines, implying the inhibitory roles of let-7c/miR-99a/miR-125b in mammosphere generation. To further test whether low levels of let-7c/miR-99a/miR-125b could control mammosphere generation and, thus, regulate cancer cell self-renewal, we studied the effect of enforced let-7c/miR-99a/miR-125b expression using the mammosphere assay. Consistent with our expectations, the sphere-forming capacity was significantly reduced by miRNAs compared with the negative controls (26.7% for let-7c, 71.1% for miR-99a and 44.4% for miR-125b in MZ-cha-1; and 38.1% for let-7c, 24.8% for miR-99a and 31.4% for miR-125b in SK-cha-1; Figures 5b and c), and the volume of the mammospheres was smaller in let-7c/miR-99a/miR-125b overexpressing MZ-cha-1 and SK-cha-1 cells than in control cells (Figures 5b and c). To address whether the effects on mammosphere formation are indeed because of the suppression of stem cell-like properties, we measured the level of CD133 and CD44, markers of CCA stem cells, in mammospheres. In addition, we found that the percentage of CD133+CD44+ cells is reduced in MZ-cha-1 and SK-cha-1 cells with let-7c/miR-99a/miR-125b overexpression (Figures 5b and c, right lanes). These data demonstrate that let-7c/miR-99a/miR-125b has an important suppressing role in CCA cell malignancy transformation, and these miRNAs may also be involved in the maintenance and proliferation of tumor stem cells in CCA, which further provides potential effective targets for CCA treatment.
let-7c/miR-99a/miR-125b inhibited CCA cell transformation and migration in vitro. (a) The let-7c/miR-99a/miR-125b cluster is significant downregulated in mammospheres cultured in suspension (t-test). (b) Enforced let-7c/miR-99a/miR-125b expression suppressed mammosphere generation and proportion of CD133+CD44+ cells in MZ-cha-1 cells. (c) Enforced let-7c/miR-99a/miR-125b expression suppressed mammosphere generation and proportion of CD133+CD44+ cells in SK-cha-1 cells. An average of three times of experiments was showed in each experiment, and one-way analysis of variance test was used. *P<0.05; **P<0.01; ***P<0.001.
Enforced expression of let-7c/miR-99a/miR-125b inhibited CCA cell migration and invasion in vitro
The activation of STAT3 has also been linked to the progression of cancer by promoting tumor migration.38, 39 As let-7c/miR-99a/miR-125b regulated the activation of IL-6/STAT3 pathway in CCA cells, we postulated that ectopic expression of let-7c/miR-99a/miR-125b would suppress malignancy transformation and impede the migratory and invasive abilities of CCA cells. We therefore used cell migration and cell invasion assays to confirm this speculation. Consistent with our expectation, overexpressed let-7c/miR-99a/miR-125b significantly decreased the migration ability of MZ-cha-1 and SK-cha-1 cells (Figure 6a) and decreased the cell invasion of MZ-cha-1 and SK-cha-1 cells (Figure 6b). We performed cellular viability and apoptosis assays to determine whether the inhibition on the migratory and invasive potential is due to a reduction in survival and proliferation, and no significant repression was observed with these miRNAs overexpression (Supplementary Figure 4), which suggested that re-forced let-7c/miR-99a/miR-125b expression helped inhibit the migration and invasion capabilities of the cells, and these inhibitions are not due to altered proliferation or apoptosis rates. It is also noteworthy that overexpressed let-7c/miR-99a/miR-125b showed similar functions as STAT3 inhibitors in CCA,39 which further suggested that let-7c/miR-99a/miR-125b helps to inhibit the capability of tumor migration by affecting the IL-6/STAT3 pathway.
let-7c/miR-99a/miR-125b inhibited CCA cell migration and invasion in vitro. Enforced let-7c/miR-99a/miR-125b expression decreased the ability of (a) migration and (b) invasion of MZ-cha-1 and SK-cha-1 cells. Five random sights in each sample were selected to analyze cell count, and the mean of triplicate experiments was exhibited. **P<0.01; ***P<0.001.
Let-7c/miR-99a/miR-125b suppress tumorigenicity in vivo
To examine whether let-7c/miR-99a/miR-125b also inhibited the capability of malignant transformation and tumor migration of CCA cells and tumor growth in vivo, let-7c/miR-99a/miR-125b-overexpressing and control MZ-cha-1 and SK-cha-1 cells were injected subcutaneously into the flanks of nude mice, and the animals were closely monitored for tumor growth.
Consistent with the in vitro results, a significant and sustained suppression of tumor growth was found in animals injected with let-7c/miR-99a/miR-125b-overexpressing cells compared with the control group, suggesting tumor-suppressing roles for all three miRNAs (Figures 7a and b and Supplementary Figures 5A and B). Let-7c/miR-99a/miR-125b also significantly reduced the tumor weight compared with that of the control group in mice (Figure 7c and Supplementary Figure 5C), whereas no significant changes in body weight among groups. All these data further confirmed the tumor-suppressing roles of the let-7c/miR-99a/miR-125b cluster in CCA pathogenesis.
Let-7c/miR-99a/miR-125b suppressed tumorigenicity of CCA cells in vivo. let-7c, miR-99a and miR-125b reduced (a, b) tumor volume (mean±s.e.m., one-way analysis of variance) and (c) tumor weight in nude mice (mean±s.d., one-way analysis of variance, five mice per group). (d) In tumors overexpressing the miRNAs, IL-6, IL-6R and IGF1R as well as the phosphorylation of STAT3 were suppressed. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) and β-actin served as loading controls, and densitometric ratios of objective protein/GAPDH were recorded. *P<0.05.
As we found that the let-7c/miR-99a/miR-125b cluster controlled tumorigenesis by targeting IL-6, IL-6R and IGF1R and affecting the IL-6/STAT3 inflammatory pathway in vitro, we asked whether this mechanism also worked in vivo. To examine this, we then evaluated expression of the inflammatory cytokine IL-6, as well as the two cytokine receptors IL-6R and IGF1R, in mice tumor tissues. The results showed that the protein level of IL-6, IL-6R and IGF1R was repressed in most miRNA overexpressing tumor tissues and, more importantly, that phosphorylation of STAT3 as well as activated phosphorylated IGF1R were also reduced in these tissues (Figure 7d, Supplementary Figures 5D and 6). These findings indicated that these inflammatory cytokines and receptors were regulated by the overexpressed miRNAs, which led to inhibition of the IL-6/STAT3 pathway in CCA cells. Further immunofluorescence analysis of CD44 demonstrated the stemness of CCA cells was repressed by let-7c/miR-99a/miR-125b in xenograft tumor specimens (Supplementary Figure 7). Therefore, the regulated inflammatory pathway blocked the pathogenesis and progression of CCA in vivo.
Discussion
The importance of some deregulated miRNAs have been recognized in CCA;40 however, little is known about the link between miRNAs and inflammation of the biliary epithelium, which is one of the most important risk factors of CCA.41 Here, small RNA sequencing was selected to screen the deregulated miRNAs in CCA tissues. Among the ectopic miRNAs, the family members of the let-7c/miR-99a/miR-125b cluster were found similarly downregulated and targeted the IL-6/STAT3 pathway. Furthermore, overexpressed let-7c/miR-99a/miR-125b reduced the activation of STAT3 and inhibited the malignant transformation of CCA cells in vitro and tumorigenicity in vivo.
MiRNAs have critical roles in carcinogenesis, and also have been regarded as potential biomarkers for cancer diagnosis and targets for anticancer therapeutics in various cancers.42, 43 However, not all of the ectopic miRNAs in cancer tissues have roles in oncogenesis; only the most abundant miRNAs in a cell mediate target suppression, and miRNAs that are expressed below ~100 copies per cell have little regulatory capacity.44, 45 Therefore, our work revealed the deregulation of miRNAs with >200 r.p.m. in well- and moderately poorly differentiated CCA tissues compared with normal tissues using high-throughput sequencing. These data provide global and, more importantly, promising functional profiling of miRNAs in the pathogenesis and progression of CCA.
In this article, we found that that let-7c, miR-99a and miR-125b are downregulated in CCA patients. Overexpression of let-7c/miR-99a/miR-125b suppresses CCA tumorigenicity in vivo and inhibits the migration and invasion by targeting important cytokines and receptors (IL-6, IL-6R and IGF1R) of the IL-6/STAT3 inflammation pathway. Surprisingly, let-7c/miR-99a/miR-125b also significantly decreased the ability of CCA cells for cancer stem cell-like mammosphere generation. The members of the let-7, miR-99/100 and miR-125 family are organized into three clusters in the human chromosome. These miRNAs are often co-expressed and participate in the same biological pathways. Our previous studies have revealed that miR-125b and miR-100 are upregulated in acute myeloid leukemia and block myeloid cell differentiation.46, 47, 48 A recent study also indicated that miR-99a and miR-100 are downregulated in acute lymphoblastic leukemia and promote cell apoptosis induced by glucocorticoids.49 Henson et al.50 reported that miR-125b and miR-100 are downregulated in oral squamous cell carcinoma, and exogenous expression of miR-125b and miR-100 significantly reduced cell proliferation. In prostate cancer, the androgen receptor binds to the host gene of the let-7c/miR-99a/miR-125b cluster, LINC00478, and this cluster is repressed by the chromatin remodeler EZH2 when the androgen receptor is stimulated.35 Meng et al.51 reported that miR-99a is reduced by overexpression of IL-6 in CCA cells, which indicates that the let-7c/miR-99a/miR-125b cluster may be repressed by the IL-6 inflammation factor and may be involved in inflammatory signaling pathways.
Inflammation is a complex response, and inflammatory cytokines, including ILs, T-cell growth factor and chemokines, contribute to control of the magnitude of the inflammatory responses. These cytokines are often regarded as important mediators in CCA. Here, we found many miRNAs that were deregulated in CCA could regulate various inflammatory cytokines, inflammation-related cytokines and their receptors, which implies that these deregulated miRNAs are involved in multiple inflammatory signaling pathways. Among these pathways, IL-6-mediated STAT3 activation, which is aberrantly sustained in CCA cells,26 is one of the well-established, classical pathways. To our knowledge, this is the first report to demonstrate the contribution of the let-7c/miR-99a/miR-125b cluster to the pathogenesis of CCA.
Notably, the let-7c/miR-99a/miR-125b cluster showed significant inhibition of tumorigenicity in vivo and of cell progression in vitro, which further raised the potential of the let-7c/miR-99a/miR-125b cluster serving as molecular targets for CCA treatment. Furthermore, pSTAT3 inhibition was prolonged when cells were treated with let-7c/miR-99a/miR-125b, which may counteract many biological effects of STAT3-mediated tumorigenesis pathways. Moreover, previous reports have shown that inhibition of STAT3 by small interfering RNA effectively inhibited tumor growth and overcame drug resistance,52, 53, 54, 55 thus, the endogenous miRNAs that target STAT3 may provide a more natural and secure choice for treatment, and our research provides an alternative strategy for developing an miRNA-based therapy via IL-6/STAT3 targeting for CCA.
In conclusion, this study screened the miRNA profiles of CCA and provided a first glimpse of the functional role of the let-7c/miR-99a/miR-125b cluster by targeting pivotal inflammatory factors such as IL-6, IL-6R and IGF1R, which further suppress the activation of STAT3 and inhibit tumor pathogenesis and progression. These results indicate the potential roles of the let-7c/miR-99a/miR-125b cluster as serving as therapeutic targets in CCA.
Materials and methods
Patient samples and cell lines
Human CCA and peritumoral (designated as normal) tissues were obtained with informed consent between 2011 and 2013 from Sun Yat-sen Memorial Hospital. Sample collection was approved by the Hospital’s Protection of Human Subjects Committee. Twenty-four pairs of normal peritumoral specimens and pathologically diagnosed biopsy specimens were obtained from the same patients with CCA who underwent surgical resection. The clinicopathological characteristics of the CCA patients are summarized in Table 1.
QBC939 human CCA cells were obtained from Shuguang Wang (The Third Military Medical University, Chongqing, China), whereas SK-cha-1 and MZ-cha-1 human CCA cells were kindly provided by Dr Chundong Yu (Xiamen University, Fujian, China).
Library preparation, quantitative reverse transcription real-time PCR and RNA sequencing
Total RNA was isolated using TRIzol (Invitrogen, Carlsbad, CA, USA), and quantitative reverse transcription real-time PCR was performed as reported previously.46, 56 dCt (delta Ct) values of specific gene were normalized with those obtained from the amplification of the internal control, in detail, dCt=Ctgene – Ctinternal control, and the relative expression of gene=2-(dCtgene – dCtcontrol). The primers for real-time PCR are shown in Supplementary Table 1. Small RNA library preparation and sequencing were performed using Solexa sequencing Technology (BGI, Shenzhen, China), and sequencing data analysis was performed as previously described.57
Luciferase assays
HEK-293T cells were co-transfected with 200 ng of psiCHECK-2-derived reporter vectors and 50 nM of miRNA mimics or the mimic control and then applied to a luciferase reporter assay, as previously described.46
Lentiviral vector production and stably transduced cells
For lentiviral vector construction, a genomic fragment containing the pre-miRNA (~200 bp) was inserted into the pGreenPuro vector (System Biosciences, Johnstown, PA, USA). A fragment that could form a hairpin structure that was processed into a small interfering RNA that targeted firefly luciferase was inserted into the same vector as a negative control. Primers and other oligonucleotides are shown in Supplementary Table 1.
Immunoblotting and immunofluorescence analysis
Cells were lysed in RIPA buffer. The proteins were detected with anti-IL-6 (Santa Cruz, Dallas, TX, USA, sc-7920), anti-IL-6R (Abcam, Cambridge, UK, ab128008), IGF1R (Sigma-Aldrich, St Louis, MO, USA, SAB1104892), anti-STAT3 (Santa Cruz, sc-482), anti-phoshpo-STAT3 (Cell Signaling, Danvers, MA, USA, 9145), anti-phoshpo-IGF1R (Cell Signaling, 2969), anti-beta-actin (Cell Signaling, 4970) or anti-glyceraldehyde 3-phosphate dehydrogenase (Protein Tech Group, Chicago, IL, USA, 10494-1-AP). Immunoreactivity was determined using the ECL method (Millipore, Boston, MA, USA) according to the manufacturer’s instructions.
Migration and invasion assays
A 24-well transwell plate (8-μm pore size, Corning, NY, USA) was used to measure migratory and invasive ability. For the transwell migration assays, 5 × 104 lentiviral stably transduced MZ-cha-1 cells were plated in the top chamber that was lined with a non-coated membrane. For the invasion assays, the chamber inserts were coated with a 1:8 deliquation of Matrigel (BD Biosciences, Franklin Lakes, NJ, USA). Then, 2.5 × 105 cells were plated in the top chamber. After incubation at 37 °C for 24 h, the cells located in the lower chamber were fixed, stained with 0.1% crystal violet and counted using a microscope. The mean of triplicate assays for each experimental condition was determined.
Proliferation and apoptosis assays
Cell proliferation was assessed using the Cell Counting Kit-8 (CCK-8). After transfection, 5 × 103 cells per well were plated in 96-well sterile plastic culture plates, and the CCK-8 assay (Dojindo Molecular Technologies, Shanghai, China) was performed after 0, 24, 48, 72, and 96 h. To assess the rate of apoptosis, the Annexin V-PI Kit (Nanjing Keygen, Nanjing, China) was used according to the manufacturer’s guidelines. The detection was performed with a FACS Calibur using CellQuest software (BDIS, San Jose, CA, USA).
Mammosphere culture
Cells (1500 cells/ml) were cultured for 8 days in serum-free DMEM-F12 (BioWhittaker, Radnor, PA, USA) supplemented with B27 (1:50, Invitrogen), 20 ng/ml EGF (Invitrogen), 20 ng/ml bFGF (Invitrogen) and Antibiotic-antimycotic (1:100, Invitrogen) in suspension in Corning Costar 3471 6-well plates.
Flow cytometry
Flow cytometry for CD133 and CD44 expression was performed using phycoerythrin-conjugated monoclonal mouse anti-human CD133/1 (Miltenyi Biotec, Bergisch Gladbach, Germany) and antigen-presenting cell-conjugated mouse anti-human CD44 (Miltenyi Biotec). Samples were analyzed on a BD FACS Calibur (BD Biosciences) and data were analyzed using Flowjo software (BD Biosciences).
Animal model
Six-week-old male nude mice were maintained under specific, pathogen-free conditions in the Laboratory Animal Center of Sun Yat-sen University. All experimental procedures involving animals were in accordance with the Guide for the Care and Use of Laboratory Animals (NIH publications nos. 80-23, revised 1996) and were performed according to the institutional ethical guidelines for animal experiments. Mice were randomly assigned to four groups with five mice, and investigators were blinded to the group allocation. In each group, lentiviral stably transduced MZ-cha-1 or SK-cha-1 cells (2.5 × 106) were subcutaneously injected into the dorsal right flanks of the mice, and the mice were monitored each 3 days for tumor growth.
Statistical analysis
Two-tailed Student’s t-test was performed to determine statistically significant differences between two groups, and multiple comparisons were done using one-way analysis of variance, and a P-value of <0.05 was considered significant. *P<0.05, **P<0.01, ***P<0.001. Variation within each group were estimated, and the statistically comparison would not perform unless the variance was similar between groups. If not mentioned, all data were expressed as the mean±s.d. of three independent experiments. The linear correlation between miRNAs was measured by Pearson product–moment correlation test.
References
Gores GJ . Cholangiocarcinoma: current concepts and insights. Hepatology 2003; 37: 961–969.
Shaib Y, El-Serag HB . The epidemiology of cholangiocarcinoma. Semin Liver Dis 2004; 24: 115–125.
Lazaridis KN, Gores GJ . Cholangiocarcinoma. Gastroenterology 2005; 128: 1655–1667.
Kokuryo T, Yokoyama Y, Nagino M . Recent advances in cancer stem cell research for cholangiocarcinoma. J Hepatobiliary Pancreat Sci 2012; 19: 606–613.
Li Z . CD133: a stem cell biomarker and beyond. Exp Hematol Oncol 2013; 2: 17.
Thanan R, Pairojkul C, Pinlaor S, Khuntikeo N, Wongkham C, Sripa B et al. Inflammation-related DNA damage and expression of CD133 and Oct3/4 in cholangiocarcinoma patients with poor prognosis. Free Radic Biol Med 2013; 65: 1464–1472.
Zhang KZ, Zhang QB, Zhang QB, Sun HC, Ao JY, Chai ZT et al. Arsenic trioxide induces differentiation of CD133+ hepatocellular carcinoma cells and prolongs posthepatectomy survival by targeting GLI1 expression in a mouse model. J Hematol Oncol 2014; 7: 28.
Landi S, Moreno V, Gioia-Patricola L, Guino E, Navarro M, de Oca J et al. Association of common polymorphisms in inflammatory genes interleukin (IL)6, IL8, tumor necrosis factor alpha, NFKB1, and peroxisome proliferator-activated receptor gamma with colorectal cancer. Cancer Res 2003; 63: 3560–3566.
Sun Z, Wang S, Zhao RC . The roles of mesenchymal stem cells in tumor inflammatory microenvironment. J Hematol Oncol 2014; 7: 14.
Fava G, Lorenzini I . Molecular pathogenesis of cholangiocarcinoma. Int J Hepatol 2012; 2012: 630543.
Johnson C, Han Y, Hughart N, McCarra J, Alpini G, Meng F . Interleukin-6 and its receptor, key players in hepatobiliary inflammation and cancer. Transl Gastrointest Cancer 2012; 1: 58–70.
Rosen HR, Winkle PJ, Kendall BJ, Diehl DL . Biliary interleukin-6 and tumor necrosis factor-alpha in patients undergoing endoscopic retrograde cholangiopancreatography. Dig Dis Sci 1997; 42: 1290–1294.
Akiyama T, Hasegawa T, Sejima T, Sahara H, Seto K, Saito H et al. Serum and bile interleukin 6 after percutaneous transhepatic cholangio-drainage. Hepatogastroenterology 1998; 45: 665–671.
Kimura F, Miyazaki M, Suwa T, Sugiura T, Shinoda T, Itoh H et al. Serum interleukin-6 levels in patients with biliary obstruction. Hepatogastroenterology 1999; 46: 1613–1617.
Scotte M, Daveau M, Hiron M, Delers F, Lemeland JF, Teniere P et al. Interleukin-6 (IL-6) and acute-phase proteins in rats with biliary sepsis. Eur Cytokine Netw 1991; 2: 177–182.
Park J, Gores GJ, Patel T . Lipopolysaccharide induces cholangiocyte proliferation via an interleukin-6-mediated activation of p44/p42 mitogen-activated protein kinase. Hepatology 1999; 29: 1037–1043.
Maione D, Di Carlo E, Li W, Musiani P, Modesti A, Peters M et al. Coexpression of IL-6 and soluble IL-6R causes nodular regenerative hyperplasia and adenomas of the liver. EMBO J 1998; 17: 5588–5597.
Hirano T, Ishihara K, Hibi M . Roles of STAT3 in mediating the cell growth, differentiation and survival signals relayed through the IL-6 family of cytokine receptors. Oncogene 2000; 19: 2548–2556.
Groner B, Lucks P, Borghouts C . The function of Stat3 in tumor cells and their microenvironment. Semin Cell Dev Biol 2008; 19: 341–350.
Isomoto H, Kobayashi S, Werneburg NW, Bronk SF, Guicciardi ME, Frank DA et al. Interleukin 6 upregulates myeloid cell leukemia-1 expression through a STAT3 pathway in cholangiocarcinoma cells. Hepatology 2005; 42: 1329–1338.
Furqan M, Akinleye A, Mukhi N, Mittal V, Chen Y, Liu D . STAT inhibitors for cancer therapy. J Hematol Oncol 2013; 6: 90.
Baserga R, Peruzzi F, Reiss K . The IGF-1 receptor in cancer biology. Int J Cancer 2003; 107: 873–877.
Rojas A, Liu G, Coleman I, Nelson PS, Zhang M, Dash R et al. IL-6 promotes prostate tumorigenesis and progression through autocrine cross-activation of IGF-IR. Oncogene 2011; 30: 2345–2355.
Zong CS, Chan J, Levy DE, Horvath C, Sadowski HB, Wang LH . Mechanism of STAT3 activation by insulin-like growth factor I receptor. J Biol Chem 2000; 275: 15099–15105.
Li S, Wang N, Brodt P . Metastatic cells can escape the proapoptotic effects of TNF-alpha through increased autocrine IL-6/STAT3 signaling. Cancer Res 2012; 72: 865–875.
Isomoto H . Epigenetic alterations in cholangiocarcinoma-sustained IL-6/STAT3 signaling in cholangio- carcinoma due to SOCS3 epigenetic silencing. Digestion 2009; 79: 2–8.
Bartel DP . MicroRNAs: genomics, biogenesis, mechanism, and function. Cell 2004; 116: 281–297.
Sirica AE . Cholangiocarcinoma: molecular targeting strategies for chemoprevention and therapy. Hepatology 2005; 41: 5–15.
Boonyanugomol W, Chomvarin C, Hahnvajanawong C, Sripa B, Kaparakis-Liaskos M, Ferrero RL . Helicobacter pylori cag pathogenicity island (cagPAI) involved in bacterial internalization and IL-8 induced responses via NOD1- and MyD88-dependent mechanisms in human biliary epithelial cells. PLoS One 2013; 8: e77358.
Alvaro D, Barbaro B, Franchitto A, Onori P, Glaser SS, Alpini G et al. Estrogens and insulin-like growth factor 1 modulate neoplastic cell growth in human cholangiocarcinoma. Am J Pathol 2006; 169: 877–888.
Yazumi S, Ko K, Watanabe N, Shinohara H, Yoshikawa K, Chiba T et al. Disrupted transforming growth factor-beta signaling and deregulated growth in human biliary tract cancer cells. Int J Cancer 2000; 86: 782–789.
Zen Y, Harada K, Sasaki M, Chen TC, Chen MF, Yeh TS et al. Intrahepatic cholangiocarcinoma escapes from growth inhibitory effect of transforming growth factor-beta1 by overexpression of cyclin D1. Lab Invest 2005; 85: 572–581.
Benckert C, Jonas S, Cramer T, Von Marschall Z, Schafer G, Peters M et al. Transforming growth factor beta 1 stimulates vascular endothelial growth factor gene transcription in human cholangiocellular carcinoma cells. Cancer Res 2003; 63: 1083–1092.
Yang JH, Li JH, Shao P, Zhou H, Chen YQ, Qu LH . StarBase: a database for exploring microRNA-mRNA interaction maps from Argonaute CLIP-Seq and Degradome-Seq data. Nucleic Acids Res 2011; 39: D202–D209.
Sun D, Layer R, Mueller AC, Cichewicz MA, Negishi M, Paschal BM et al. Regulation of several androgen-induced genes through the repression of the miR-99a/let-7c/miR-125b-2 miRNA cluster in prostate cancer cells. Oncogene 2014; 33: 1448–1457.
Isomoto H, Mott JL, Kobayashi S, Werneburg NW, Bronk SF, Haan S et al. Sustained IL-6/STAT-3 signaling in cholangiocarcinoma cells due to SOCS-3 epigenetic silencing. Gastroenterology 2007; 132: 384–396.
Dontu G, Abdallah WM, Foley JM, Jackson KW, Clarke MF, Kawamura MJ et al. In vitro propagation and transcriptional profiling of human mammary stem/progenitor cells. Genes Dev 2003; 17: 1253–1270.
Xie TX, Huang FJ, Aldape KD, Kang SH, Liu M, Gershenwald JE et al. Activation of stat3 in human melanoma promotes brain metastasis. Cancer Res 2006; 66: 3188–3196.
Zheng T, Hong X, Wang J, Pei T, Liang Y, Yin D et al. Gankyrin promotes tumor growth and metastasis through activation of IL-6/STAT3 signaling in human cholangiocarcinoma. Hepatology 2014; 59: 935–946.
Munoz-Garrido P, Garcia-Fernandez DBM, Hijona E, Carracedo M, Marin JJ, Bujanda L et al. MicroRNAs in biliary diseases. World J Gastroenterol 2012; 18: 6189–6196.
Khan SA, Thomas HC, Davidson BR, Taylor-Robinson SD . Cholangiocarcinoma. Lancet 2005; 366: 1303–1314.
Smith AD, Roda D, Yap TA . Strategies for modern biomarker and drug development in oncology. J Hematol Oncol 2014; 7: 70.
Wang WT, Chen YQ . Circulating miRNAs in cancer: from detection to therapy. J Hematol Oncol 2014; 7: 86.
Mullokandov G, Baccarini A, Ruzo A, Jayaprakash AD, Tung N, Israelow B et al. High-throughput assessment of microRNA activity and function using microRNA sensor and decoy libraries. Nat Methods 2012; 9: 840–846.
Brown BD, Gentner B, Cantore A, Colleoni S, Amendola M, Zingale A et al. Endogenous microRNA can be broadly exploited to regulate transgene expression according to tissue, lineage and differentiation state. Nat Biotechnol 2007; 25: 1457–1467.
Lin KY, Zhang XJ, Feng DD, Zhang H, Zeng CW, Han BW et al. miR-125b, a target of CDX2, regulates cell differentiation through repression of the core binding factor in hematopoietic malignancies. J Biol Chem 2011; 286: 38253–38263.
Sun YM, Lin KY, Chen YQ . Diverse functions of miR-125 family in different cell contexts. J Hematol Oncol 2013; 6: 6.
Zheng YS, Zhang H, Zhang XJ, Feng DD, Luo XQ, Zeng CW et al. MiR-100 regulates cell differentiation and survival by targeting RBSP3, a phosphatase-like tumor suppressor in acute myeloid leukemia. Oncogene 2012; 31: 80–92.
Li XJ, Luo XQ, Han BW, Duan FT, Wei PP, Chen YQ . MicroRNA-100/99a, deregulated in acute lymphoblastic leukaemia, suppress proliferation and promote apoptosis by regulating the FKBP51 and IGF1R/mTOR signalling pathways. Br J Cancer 2013; 109: 2189–2198.
Henson BJ, Bhattacharjee S, O'Dee DM, Feingold E, Gollin SM . Decreased expression of miR-125b and miR-100 in oral cancer cells contributes to malignancy. Genes Chromosomes Cancer 2009; 48: 569–582.
Meng F, Wehbe-Janek H, Henson R, Smith H, Patel T . Epigenetic regulation of microRNA-370 by interleukin-6 in malignant human cholangiocytes. Oncogene 2008; 27: 378–386.
Falamarzian A, Aliabadi HM, Molavi O, Seubert JM, Lai R, Uludag H et al. Effective down-regulation of signal transducer and activator of transcription 3 (STAT3) by polyplexes of siRNA and lipid-substituted polyethyleneimine for sensitization of breast tumor cells to conventional chemotherapy. J Biomed Mater Res A 2014; 102: 3216–3228.
Leong PL, Andrews GA, Johnson DE, Dyer KF, Xi S, Mai JC et al. Targeted inhibition of Stat3 with a decoy oligonucleotide abrogates head and neck cancer cell growth. Proc Natl Acad Sci USA 2003; 100: 4138–4143.
Barton BE, Murphy TF, Shu P, Huang HF, Meyenhofer M, Barton A . Novel single-stranded oligonucleotides that inhibit signal transducer and activator of transcription 3 induce apoptosis in vitro and in vivo in prostate cancer cell lines. Mol Cancer Ther 2004; 3: 1183–1191.
Ling X, Arlinghaus RB . Knockdown of STAT3 expression by RNA interference inhibits the induction of breast tumors in immunocompetent mice. Cancer Res 2005; 65: 2532–2536.
Wang WT, Zhao YN, Yan JX, Weng MY, Wang Y, Chen YQ et al. Differentially expressed microRNAs in the serum of cervical squamous cell carcinoma patients before and after surgery. J Hematol Oncol 2014; 7: 6.
Zhang H, Yang JH, Zheng YS, Zhang P, Chen X, Wu J et al. Genome-wide analysis of small RNA and novel MicroRNA discovery in human acute lymphoblastic leukemia based on extensive sequencing approach. PLoS One 2009; 4: e6849.
Acknowledgements
This work was supported by the funds from National Science and Technology Department (973, 2011CB8113015 and 2011CBA0110), National Science Foundation of China (no. 81270629) and Guangdong (2014A020212135). This work is also supported in part by the Guangdong Province Key Laboratory of Computational Science and the Guangdong Province Computational Science Innovative Research Team.
Author contributions
K-YL, HY and B-WH conceived and carried out the experiment, wrote the paper; W-TW, P-PW and X-JL performed mammosphere formation experiments and migration and invasion assays; Y-QC conceived the experiment and cowrote the paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
About this article
Cite this article
Lin, KY., Ye, H., Han, BW. et al. Genome-wide screen identified let-7c/miR-99a/miR-125b regulating tumor progression and stem-like properties in cholangiocarcinoma. Oncogene 35, 3376–3386 (2016). https://doi.org/10.1038/onc.2015.396
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2015.396
- Springer Nature Limited
This article is cited by
-
MicroRNA-1182 and let-7a exert synergistic inhibition on invasion, migration and autophagy of cholangiocarcinoma cells through down-regulation of NUAK1
Cancer Cell International (2021)
-
miR-125b-5p and miR-99a-5p downregulate human γδ T-cell activation and cytotoxicity
Cellular & Molecular Immunology (2019)
-
Global identification and characterization of lncRNAs that control inflammation in malignant cholangiocytes
BMC Genomics (2018)
-
Let-7c inhibits cholangiocarcinoma growth but promotes tumor cell invasion and growth at extrahepatic sites
Cell Death & Disease (2018)
-
A miR-125b/CSF1-CX3CL1/tumor-associated macrophage recruitment axis controls testicular germ cell tumor growth
Cell Death & Disease (2018)