Abstract
MicroRNAs (miRNAs) have been demonstrated to have critical roles in regulating cancer cell proliferation, survival and sensitivity to chemotherapy. The potential application of using miRNAs to predict therapeutic response to cancer treatment holds high promise, but miRNAs with predictive value remain to be identified and underlying mechanisms have not been completely understood. Here, we show a strong correlation between miR-621 expression and chemosensitivity to paclitaxel plus carboplatin (PTX/CBP) regimen, an effective neoadjuvant chemotherapy for breast cancer patients. High level of miR-621 predicts better response to PTX/CBP regimen neoadjuvant chemotherapy in breast cancer patients, who also tend to achieve pathological complete response. Ectopic overexpression of miR-621 promoted apoptosis and increased chemosensitivity to PTX and CBP both in cultured breast cancer cells and in xenograft tumor model. We further show that FBXO11 is a direct functional target of miR-621 and miR-621 level is negatively correlated with FBXO11 expression in breast cancer patients. Ectopic expression of FBXO11 attenuated increased apoptosis in breast cancer cells overexpressing miR-621 upon PTX or CBP treatment. Consistently, high FBXO11 expression significantly correlated with poor survival in breast cancer patients. Mechanistically, we found in breast cancer cells FBXO11 interacts with p53 and promotes its neddylation, which suppressed the p53 transactivity. Accordingly, miR-621-dependent FBXO11 suppression enhanced p53 activity and increased apoptosis in breast cancer cells exposed to chemotherapeutics. Taken together, our data suggest that miR-621 enhances chemosensitivity of breast cancer cells to PTX/CBP chemotherapy by suppressing FBXO11-depedent inhibition of p53. miR-621 may serve as a predictive biomarker and a potential therapeutic target in breast cancer treatment.
Similar content being viewed by others
Introduction
Breast cancer is the most common malignancy in women. The mortality of breast cancer has decreased over the last several decades, likely because of a combination of mammographic screening and improvements in systemic therapy.1 Neoadjuvant systemic treatment before surgery for advanced breast cancer is considered one of the most crucial factors in reducing mortality.2, 3, 4, 5 Therefore, neoadjuvant chemotherapy (NAC) has become the standard treatment of locally advanced breast cancer, which can downstage the disease and improve the surgical option.6 Moreover, NAC permits observation of tumor responsiveness to the treatment, and analyzing residual disease after NAC may reveal novel therapeutic targets.7 To a specific NAC regimen, generally only a fraction of patients achieve full response, while others respond poorly and only suffer from side effects. Ideally, those patients who would benefit from a specific chemotherapy regimen can be identified which will allow the remainder to be spared from its side effects. Although clinical parameters such as tumor size, estrogen receptor (ER) or HER-2 receptor status, histologic or nuclear grade, or the expression of single molecular markers (Bcl-2, p53, MDR-1, Ki67 and so on) may have predictive value to therapeutic response, these are not regimen-specific, which limits their application in selecting NAC regimen.8, 9 The current development of predictive signatures to guide the use of chemotherapy has not yet yielded clinically reliable tests.10, 11, 12, 13, 14, 15
MicroRNAs (miRNA) are short, 20–22-nucleotide noncoding RNA molecules that negatively regulate gene expression by binding to the 3’-untranslated region (UTR) of their target genes with partial complementarity, leading to degradation of the target mRNAs, inhibition of their translation or both.16, 17 It has been found that miRNAs regulate various pathological behaviors of cancer cells, such as proliferation, motility and sensitivity to chemotherapy, by targeting multiple genes simultaneously or in parallel.18, 19, 20, 21, 22, 23, 24
Our previous study has shown that weekly PTX/CBP (paclitaxel plus carboplatin) regimen is effective nonanthracycline-containing NAC for breast cancer, with a pCR (pathologic complete response) rate of 19.4%.25 pCR indicates the disappearance of all invasive cancer cells in the breast (and/or in axillary lymph nodes) after neoadjuvant therapy, which is considered an early surrogate of prolonged survival.26, 27 However, the underlying mechanisms dictating patient response to PTX/CBP regimen are poorly understood. In this study, we demonstrated for the first time, a significant correlation between miR-621 expression and chemosensitivity to PTX/CBP regimen in breast cancer patients. High level of miR-621 predicts sensitivity to PTX/CBP NAC in breast cancer patients who tend to achieve pCR. The increased chemosensitivity could be mediated by miR-621-dependent downregulation of FBXO11, resulting in enhanced p53 transactivity and increased apoptosis upon chemotherapy. Therefore, miR-621 may be used as a predictive biomarker for PTX/CBP regimen and a potential therapeutic target in breast cancer treatment.
Results
High expression level of miR-621 correlates with pathological complete response and improves disease free survival
We previously identified 231 differentially expressed genes (DEGs) between 6 pCR cases and 25 non-pCR cases from gene profiling data.28 It was reported that CORNA algorism can be used to define significant microRNA-target correlation in DEGs, and to delineate significant associations between microRNAs and pathways with GO terms.29 Therefore, we analyzed the 231 DEGs by CORNA to identify significant DEG-targeted miRNAs. Interestingly, predicted miR-621-correlated genes were significantly enriched in these 231 DEGs, indicating that miR-621 may be a target-associated miRNA regulating breast cancer cell sensitivity to PTX/CBP regimen chemotherapy (Supplementary Table S1).
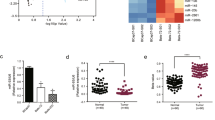
To validate the association between miR-621 and response to PTX/CBP regimen, an independent set of 50 patients (25 pCR patients and 25 non-pCR patients) was further analyzed by using quantitative RT-PCR. The clinicopathological features of this validation set are listed in Table 1. Consistently, we found the expression level of miR-621 was significantly higher in tumors from pCR patients than that in non-pCR patients (P=0.015) (Figure 1a), confirming that the higher expression of miR-621 is associated with a better response to PTX/CBP regimen chemotherapy. Furthermore, analyzing the disease free survival (DFS) of these 50 breast cancer patients after stratification by the level of miR-621 revealed that patients with higher miR-621 expression level have significantly longer DFS (P=0.04) (Figure 1b). Interestingly, the ER/progesterone receptor (PR) status also show significant inverse correlation with pCR, as patients with ER negative (P=0.02) or PR negative (P=0.03) tend to achieve pCR (Table 1). Nevertheless, multiple regression analysis showed that only the miR-621 level could be an independent predict factor for pCR (hazards ratio=0.142, 95% confidence interval=0.034–0.594; P=0.007) (Table 2). To evaluate this predictive value, we used the receiver operating characteristics (ROC) curve to analyze the sensitivity and specificity of miR-621. The area under the curve (AUC) was 0.859 (confidence interval=0.752–0.965; P<0.0001), which indicated high accuracy of predictive value. The sensitivity and specificity were 91.3 and 63.6%, respectively (Figure 1c). These findings suggest that increased miR-621 expression may be responsible for high sensitivity to PTX/CBP chemotherapy and predict pCR in breast cancer patients.
High expression level of miR-621 correlates with pathological complete response (pCR) and higher disease free survival (DFS). (a) Expression levels of miR-621 in breast cancer patients (n=50, 25 pCR vs 25 non-pCR), who received PTX/CBP regimen neoadjuvant chemotherapy, were quantitated by real-time PCR. RNU6 was used as an internal control. Data are mean±s.e.m. (b) Kaplan–Merier curves of 50 breast cancer patients after stratification by the level of miR-621 were used for depicting DFS. (c) Receiver operating characteristic (ROC) curve of 50 breast cancer patients’ level of miR-621 was used for analyzing the area under the curve (AUC) value.
MiR-621 enhances chemosensitivity to paclitaxel and carboplatin by promoting apoptosis in breast cancer cells
To validate the correlation between miR-621 and chemosensitivity, we examined the miR-621 expression level and IC50 (inhibitory concentration to produce 50% cell death) value of PTX or CBP in several breast cancer cell lines. MCF-7 cells appeared to be less sensitive to PTX or CBP treatment than MDA-MB-231 and MDA-MB-231HM cells (IC50 of PTX: 23.52, 15.43, 15.63 μg/ml, respectively, P=0.002) (IC50 of CBP: 0.69, 0.29, 0.32 mg/ml, respectively, P=0.001) (Figures 2a and b). In parallel, the endogenous expression level of miR-621 in MCF-7 cells was significantly lower than that in MDA-MB-231 and MDA-MB-231HM cells (P=0.001) (Figure 2c), suggesting a positive correlation between miR-621 expression level and sensitivity to PTX or CBP in breast cancer cells. Furthermore, overexpression of miR-621 (Supplementary Figure S1a) significantly increased chemosensitivity to PTX and CBP in both MCF-7 and MDA-MB-231HM cells (Figures 2d and e). Consistently, modulating miR-621 level in MCF-7 cells by transfecting pre-miR-621 or miR-621-sponge inhibitor (Supplementary Figure S1b) decreased or an enhanced cell survival in response to PTX plus CBP treatment, respectively (Figure 2f). These results collectively support that increasing expression of miR-621 might enhance drug sensitivity to PTX and CBP in breast cancer cells.
MiR-621 increases chemosensitivity to paclitaxel and carboplatin in breast cancer cells. (a–b) Cells were treated with paclitaxel (a) at a dose range of 1.16–100 μg/ml or carboplatin (b) from 0.0625–2.5 mg/ml for 24 h. IC50 was determined by survival fraction measured with Cell Counting Kit-8 (Dojindo Laboratories, Kumamoto, Japan). (c) Expression level of miR-621 in MCF-7, MDA-MB-231 and MDA-MB-231HM cells was quantified by real-time PCR. RNU6 was used as an internal control. (d–e) Cells were transiently transfected with pri-miR-621 or negative control vector. After transfection, cells were treated with carboplatin (d) or paclitaxel (e) for 24 hours. Then IC50 was analyzed as in (a–b). (f) MCF-7 cells were transiently transfected with control, pri-miR-621 or miR-621 sponge inhibitor, and treated with paclitaxel (20 μm) plus carboplatin (1 μg/ml) for times as indicated. The cell survival fraction data from three independent experiments were pooled and shown as mean±s.d. *P<0.05.
We next asked how miR-621 regulates drug sensitivity. Overexpression of miR-621 in MCF-7 and ZR-75-1 cells markedly increased the cleavage of Caspase 8 and PARP1 in response to PTX and CBP treatment, suggesting an enhanced apoptosis upon drug treatment (Figures 3a and b). The tumor suppressor p53 activates a transcriptional program that directs cells to undergo apoptosis or senescence in response to cellular stress.30, 31, 32 Interestingly, we found that the expression of p53 target genes, such as TNFRSF10B (DR5) and BBC3 (PUMA), were increased in response to PTX or CBP treatment, which was further enhanced by miR-621 overexpression (Figures 3a and b; Supplementary Figures S1c and d). In accordance, transfection of miR-621 sponge inhibitor suppressed the upregulation of PUMA and cleaved-PARP1 by PTX or CBP treatment (Supplementary Figures S1e and f). Moreover, increased mRNA levels of TNFRSF10B(DR5) and BBC3(PUMA) genes upon cytotoxic treatment were further augmented by overexpressing miR-621 (Figures 3c and d).
Ectopic expression of miR-621 enhances p53 transactivity and promotes apoptosis. (a–b) MCF-7 (a) and ZR-75-1 (b) cells were transfected with the control or pri-miR-621 and treated with paclitaxel (20 μm) or carboplatin (1 μg/ml) as shown. Whole cells extracts were analyzed by western blot by using indicated antibodies. *Non-specific band. (c–d) MCF-7 cell was transfected with control or pre-miR-621 and treated with paclitaxel (20 μm) or carboplatin (1 μg/ml). TNFRSF10B/DR5 (c) and BBC3/PUMA (d) expression were determined by quantitative real-time PCR. Fold induction of relative gene expression (normalized to glyceraldehyde-3-phosphate dehydrogenase (GAPDH) from three independent experiments were shown as mean±s.d. *P <0.05. **P <0.01. (e–f) HCT116 WT and HCT116 p53−/− cells were transfected with control or pri-miR-621 and treated with paclitaxel (20 μm) or carboplatin (1 μg/ml). Relative fold induction of p21 (e) and DR5 (f) expression was determined by quantitative real-time PCR and showed as mean±s.d. *P <0.05. (g) HCT116 WT and HCT116 p53−/− cells were transfected with control or pre-miR-621 and treated as in (e). Whole cells extracts were analyzed by western blot by using the indicated antibodies. *Non-specific band. (h) ZR-75-1 cells transfected with control or shRNA targeting p53 alone or along with miR-621 were treated and analyzed as in (g).
To further examine whether miR-621 can enhance p53 transactivity, we transfected pre-miR-621 into HCT116 WT and HCT116 p53−/− cells, and treated these cells with PTX or CBP. Expression of CDKN1A (p21) and TNFRSF10B (DR5) were significantly activated in HCT116 WT cells, but not in p53−/− cells, upon CBP or PTX treatment, and the effect was further enhanced by overexpressing miR-621. These findings support that p53 is essential for miR-621 to enhance p21/DR5 induction by CBP/PTX (Figures 3e–g). Consistently, we found the upregulation of p53-target genes p21, PUMA and DR5 was detected at both mRNA (Supplementary Figure S2) and protein levels (Figure 3h) in ZR-75-1 cells exposed to PTX or CBP treatment, which was absent in p53-depleted ZR-75-1 cells. Moreover, we found ectopic miR-621 further augmented induction of p53-target genes upon CBP or PTX treatment in wild-type ZR-75-1 cells. Altogether, our data suggest that miR-621 may regulate breast cancer cell chemosensitivity by enhancing p53 transactivity and thereby promoting apoptosis.
FBXO11 is a direct target of miR-621 in breast cancer cells
To elucidate the underlying mechanisms promoting chemosensitivity in human breast cancer cells by miR-621, we used several bioinformatics programs to identify potential target genes of miR-621. Among the target genes predicted by the miRanda, TargetScan and starBase, FBXO11 was indicated to be a potential target gene of miR-621, which was also found as a downregulated DEG in pCR patients in the training set (Supplementary Table S1). Accordingly, FBXO11 was also identified as a miR-621-associated DEG by CORNA analysis. Moreover, ectopic miR-621 substantially reduced FBXO11 level in HEK293T, MCF-7 and ZR-75-1 cells (Figures 4a and b; Figures 3a and b). Since Dicer is a central regulator of miRNA-mediated gene silencing by loading miRNA:mRNA into RISC complex, deletion of Dicer significantly represses miRNA-dependent gene repression.33 We found that overexpression of miR-621 could significantly decrease the expression of FBXO11 in HCT116 WT cells, but not in HCT116 DICER−/− cells (Figures 4c and d), suggesting that the repression of FBXO11 by miR-621 is Dicer-dependent. In accordance, miR-621 overexpression significantly repressed the activity of a luciferase reporter fused to FBXO11 3’-UTR, but not the one fused to FBXO11 3’-UTR mutant in which miR-621-recognition site was deleted (Figure 4e). All these data strongly support that miR-621 may repress FBXO11 expression through recognition of its binding site within FBXO11 3’-UTR in human cancer cells.
FBXO11 is a direct target of miR-621. (a) HEK293T cells were transfected with an increasing amount of pre-miR-621 plasmid as shown. FBXO11 expression was determined by western blot. Tubulin was used as loading control. (b) Similar experiment as in (a) was performed in MCF-7 cells. (c–d) HCT116 WT and HCT116 DICER−/− cells were transfected with pri-miR-621 or control vector. FBXO11 expression was determined by western blot (c) and quantitative real-time PCR (d). *P <0.05. (e) HEK293T cells were transfected with control or pri-miR-621 plasmid along with WT or mutant FBXO11-3’-UTR luciferase reporter as shown. The activity of both Renilla and Firefly luciferases was assayed by using the dual-luciferase reporter assay system and data from the triplicated experiments were showed as mean±s.d. *P =0.0026. Schematic representation of the putative miR-621 target site within the 3’-UTR of FBXO11 gene was shown on the top. (f) Expression levels of FBXO11 in 50 breast cancer patients who received PTX/CBP regimen neoadjuvant chemotherapy (validation set, stratified as in Figure 1a) were analyzed by quantitative PCR. GAPDH was used as an internal control. Data were shown as mean±s.e.m. (g) Spearman rank test of 50 breast cancer patients was used for depicting the correlation between FBXO11 and miR-621. (h) High FBXO11 level correlated to poor DFS in breast cancer patients received chemotherapy. Data obtained from GSE25055. (i) High expression of FBXO11 associated with poor relapse-free survival in breast cancer patients. Data were obtained from DRUGSUV by using GEO dataset GSE2034 (http://bioprofiling.de/cgi-bin/GEO/DRUGSURV/).
To confirm the negative correlation between the decreased FBXO11 and enhanced response to PTX/CBP regimen shown in our training set (Supplementary Table S1), we further analyzed expression of FBXO11 by quantitative PCR in patient samples from the validation set. We found that the expression of FBXO11 in pCR patient tumors was significantly lower than that in non-pCR patients (P=0.016, Figure 4f). Moreover, expression of FBXO11 presented a negative correlation with miR-621 level in these 50 patients (r=-0.355, P=0.021) (Figure 4g). Additionally, we found high FBXO11 level was associated with poor DFS in these 50 patients, although the correlation did not reach statistical significance likely because of small sample size (Supplementary Figure S3a). In support of this observation, we found lower FBXO11 expression also is significantly associated with better DFS in breast cancer patients received NAC in a recently published study (GSE25055, Figure 4h).34 Moreover, high FBXO11 level was found to correlate to shorter RFS (Figure 4i) and DMFS (Supplementary Figure S3b) in breast cancer patients. Collectively, these data indicate that the chemo-sensitizing effect of miR-621 in breast cancer may depend on FBXO11 downregulation.
TP53 transcriptional activity is involved in miR-621/FBXO11 axis to regulate chemosensitivity of breast cancer cells
The TP53 gene is the most frequently mutated gene in human cancers, and functional loss of TP53 gene is correlated with chemotherapy resistance and poor prognosis in breast cancer as well as several other cancer types.30 FBXO11 is a member of the F-box protein family which form SKP1-cullin-F-box ubiquitin ligase complex and promote protein ubiquitylation and proteasome-dependent degradation.35, 36 Although FBXO11 appeared not to directly promote p53 degradation, it was reported that FBXO11 could increase p53 neddylation, which abrogates p53 acetylation at Lys320 and leads to suppression of p53 transactivity.37 FBXO11 has two splicing isoforms (FBXO11-1 and -4), both of which can be inhibited by a siRNA duplex against FBXO11.35 We cloned FBXO11 isoform 1 (NP_079409.3) into pCMV backbone and transiently transfected into cells. Consistent with the previous study,37 we found that the p53-dependent transactivity was significantly reduced in cells overexpressing FBXO11, whereas deletion of F-box domain in FBXO11 (FBXO11 ΔN) abolished its suppressive effects (Figures 5a and b). Co-immunoprecipitation assay revealed that endogenous FBXO11 could interact with p53 in MCF-7 cells upon CBP or PTX treatment (Supplementary Figures S3c and d). Moreover, overexpression of FBXO11 substantially promoted p53 neddylation in MCF7 cells (Figure 5c), which correlated with decreased p53 transcriptional activity (Figure 5e). Accordingly, we found CBP treatment enhanced p53 neddylation in breast cancer cells, while overexpressing miR-621 suppressed both basal and drug-induced p53 neddylation, likely due to a decreased FBXO11 expression by miR-621 (Figure 5d). Interestingly, ectopic expression of FBXO11 also attenuated miR-621-depedent increase of p53 transactivation, suggesting a critical role of FBXO11 repression in enhancing p53 activity by miR-621 (Figure 5e). These results suggest that enhanced p53 transactivity by down-regulating FBXO11 may account for miR-621-depedent chemo-sensitizing effect in breast cancer cells.
miR-621/FBXO11 axis regulates chemosensitivity of breast cancer cells by modulating the p53 transcriptional activity. (a) HT1080 cells were transfected with control, FBXO11 WT (FBXO11-1) or FBXO11 ΔN (F-box deletion) along with the p53 luciferase reporter. The activity of both renilla and firefly luciferases was assayed by using the dual-luciferase reporter assay system and normalized luciferase activity was showed as mean±s.d. *P <0.05. (b) MCF7 cells were transfected as in (a) and treated with paclitaxel (20 μm) or carboplatin (1 μg/ml) for 8 h. p53 luciferase reporter activity was measured as in (a). (c) MCF-7 cells were transfected with control and FBXO11 WT. Neddylation of p53 was analyzed by p53 IP from SDS-denatured cell lysates followed by immunoblotting with antibodies as indicated. (d) MCF7 cells were transfected with miR-621 and treated with carboplatin (1 μg/ml) as indicated. P53 neddylation was examined by immunoblot as in (c). Longer exposure blot was used to show basal p53 neddylation. Non-relevant lanes were cropped and marked with a solid line. (e) MCF-7 cells were transfected with control, FBXO11 WT and/or pri-miR-621 along with p53 luciferase reporter as shown. The relative p53-dependent transactivity was assayed as in (a) and data from three independent experiments were shown as mean±s.d. *P <0.05. (f) MCF-7 cells were transfected with control or FBXO11 WT and treated with paclitaxel (20 μm) or carboplatin (1 μg/ml). Whole cells extracts were analyzed by Western blot using indicated antibodies. Two FBXO11 isoforms were indicated with arrowheads. *Non-specific band. (g) MCF-7 cells were transiently transfected with pri-miR-621 alone or along with FBXO11. Cells were treated with carboplatin (1 μg/ml) or paclitaxel (20 μm) for 24 h and analyzed by immunoblotting with indicated antibodies.
In line with these observation, overexpressing FBXO11 substantially reduced PTX or CBP-induced p21, DR5 or PUMA upregulation in MCF-7 cells without affecting p53 level, supporting a negative role of FBXO11 in regulating p53 transactivity (Figure 5f). Moreover, ectopic miR-621 increased the induction of DR5 and p21 as well as cleavage of Caspase 8 and PARP1 in MCF-7 cells upon PTX or CBP treatment, which was attenuated by co-transfecting FBXO11 (Figure 5g). Although the FBXO11-4 appeared to be the major isoform subjected to suppression by miR-621 in MCF-7 cells, reconstitution of FBXO11-1 was sufficient for functionally compensating the FBXO11 suppression in MCF-7 cells (Figures 5f and g), which is consistent with a previously report.35 Taken together, these results indicate that miR-621 may regulate chemosensitivity by down-regulating target gene FBXO11, resulting in enhanced p53 transactivity and apoptosis in breast cancer cells in response to PTX or CBP treatment.
MiR-621 upregulation enhances breast cancer chemosensitivity to paclitaxel and carboplatin in vivo
To validate the role of miR-621 in enhancing chemosensitivity of breast cancer to PTX/CBP treatment, we generated MCF-7 cells stably overexpressing miR-621 or control vector, and orthotopically injected these cells into the mammary gland fat pads of BALB/c nude mice. After 20 days, these mice were treated with PBS (Sham) or PTX (15mg/kg) plus CBP (5 mg/kg) every 4 days for six cycles. We found that miR-621 overexpression significantly delayed MCF-7 xenograft tumors growth. Moreover, the tumor volume of miR-621-overexpressing xenografts decreased to a greater extent than that of control xenografts, in response to PTX/CBP treatment, indicating a chemo-sensitizing effect of miR-621 (P=0.016; Figures 6a and b). Furthermore, consistent with in vitro analyses, miR-621-overexpressed tumors showed decreased expression of FBXO11, while no noticeable change of p53 expression was observed regardless of miR-621 level (Figures 6c and d). In accordance, we detected higher expression levels of DR5 and PUMA as well as increased Caspase 8 activation in miR-621-stable xenografts than that in control tumors upon PTX/CBP treatment, suggesting an enhanced apoptotic response (Figure 6e). All these observations from immunohistochemical staining were confirmed by western blot using xenograft tumor lysates (Figure 6f). Altogether, these data strongly support that miR-621 promotes chemosensitivity to PTX/CBP regimen in breast cancers, which is likely mediated by repressing FBXO11 and enhancing p53-regulated apoptosis upon chemotherapy (Figure 6g).
MiR-621 promotes apoptosis and increases chemosensitivity to paclitaxel and carboplatin in vivo. (a) Mice transplanted with MCF-7/miR-621 stable cells (n=5) or MCF-7/vector control cells (n=5) were mock treated or treated with paclitaxel plus carboplatin as shown. Xenograft tumor growth was monitored and showed as mean±s.d. *P <0.05. (b) Image of representative tumors from control or miR-621-stable MCF-7 xenografts harvested at end point. (c) Quantitative real-time PCR confirmed the increased expression level of miR-621 in MCF-7/miR-621 xenograft tumors. RNU6 was used as an internal control. Result was showed as mean±s.d (fold change 16.05). *P <0.05. (d–e) Images to visualize positive staining of FBXO11 (d), p53 (d), DR5 (e), PUMA (e) and cleaved-Caspase-8 (e) in xenograft tumor harvested at end point. Bars: 20 um, magnification × 400. (f) Xenograft tumors from two mice in each group were analyzed by immunoblot with indicated antibodies. (g) A diagram model depicting miR-621-promoted chemosensistivity in breast cancer cells.
Discussion
Increasing efforts to identify patients that are sensitive or resistant to regimen-specific treatment have highlighted the need for applicable biomarkers to predict response to specific drug, so as to achieve effective personalized treatment while sparing patients from side effects. In this study, we showed for the first time that miR-621 expression level might be useful for predicting response to PTX/CBP NAC in breast cancer patients. High miR-621 expression in breast cancer tissues strongly associated with increased sensitivity to PTX/CBP regimen and high probability to achieve pCR, which is corroborated by the significant association between high miR-621 and better DFS (Figures 1a and b).
It is well established that miRNAs can function as tumor suppressors or oncogenes depending on the cellular context and cancer types.18, 19, 20, 21 In breast cancer, aberrant miRNA expression has been implicated to affect the response to various treatments, including chemotherapy, anti-endocrine therapies, targeted therapies, and radiotherapy.38 MiR-621 is located in chromosome 13q, which also hosts tumor suppressor miR-16-1/-15a cluster. A previous study speculated that the loss of miR-621, miR-16-1 and miR-15a because of 13q deletion might induce the upregulation of critical cell cycle control genes, resulting in uncontrolled proliferation in dedifferentiated hepatocellular carcinoma.39 Data obtained from the TCGA invasive breast cancer dataset show that miR-621 is mostly deleted in a small number of patients who harbor miR-621 gene alteration (Supplementary Figure S4a), indicating that miR-621 likely acts as tumor suppressor in breast cancers. Here, we showed that miR-621 directly targeted FBXO11 3’-UTR and repressed its expression (Figure 4), which led to increased transcriptional activity of tumor suppressor p53 and breast cancer cell apoptosis upon PTX and CBP treatment both in vitro and in vivo (Figures 3, 5 and 6). These findings were further supported by clinical data indicating that breast cancer patients with low FBXO11 expression tend to achieve better DFS and OS (Figures 4h and i). Therefore, our study revealed a novel mechanism that may be involved in the chemo-sensitizing and tumor-suppressive effects of miR-621. It appeared miR-621 expressed in different breast cancer patients at a varying level, which may influence the sensitivity to the NAC regimen. Our in vitro overexpression studies likely mimicked the cell response to chemotherapeutics in those cancer cells with very high miR-621 level, which renders them sensitive to chemotherapy. The percentage of this high miR-621 population in the tumor mass likely dictated the overall response to NAC. As the pathophysiological functions and molecular mechanisms of a particular miRNA are highly dependent on cancer cell type, it is plausible additional molecular targets and the affected signaling cascades may be involved in mediating miR-621’s chemo-sensitizing effect. For instance, TP53 is mutated and loses it pro-apoptotic function in certain breast cancer cells. Nonetheless, we found upregulating miR-621 also increased the chemosensistivity in MDA-MB-231HM cells that harbor p53 mutation (Figures 2d and e). These data suggested that miR-621 may also sensitizing breast cancer cells to PTX/CBP regimen via additional p53-independent mechanism, which warrants further investigation.
FBXO11 is a member of the F-box protein family and a component of the SKP1-cullin-F-box ubiquitin E3 ligase complex. F-box proteins recruit substrates to SKP1-cullin-F-box ligase complexes, thereby promoting their ubiquitination and proteasome-dependent degradation.35, 36 FBXO11 mutation can be found in ~20% of diffuse large B-cell lymphoma patients, which correlates with increased levels of oncoprotein BCL6 and implies a tumor-suppressive role of FBXO11.40 Oncogenic miR-21-dependent suppression of FBXO11 was found to promote tumor progression in melanoma, glioblastoma and prostate cancer cells which may also mediated by increased BCL6 expression.41 A recent study also showed that FBXO11 promoted degradation of epithelial–mesenchymal transition (EMT) regulator SNAIL, indicating that FBXO11 may inhibit cancer metastasis by blocking EMT.42 However, genomic sequencing and expression array data from the TCGA dataset indicate that FBXO11 is predominantly amplified and/or upregulated in breast cancer patients who harbor FBXO11 gene alteration (Supplementary Figure S4b), suggesting that FBXO11 may be oncogenic in breast cancers. In addition, it was shown that FBXO11 could stabilize Set8 by downregulation of Cdt2 thereby restraining CRL4Cdt2 activity, resulting in increased breast cancer cell metastasis and potentially reduced apoptosis.35, 43, 44 We identified FBXO11 as a direct target of miR-621, which may mediate miR-621-dependent chemosensitivity to PTX/CBP regimen. Furthermore, we found that FBXO11 levels in primary tumors of breast cancer patients were inversely correlated with miR-621 expression, and the high expression of FBXO11 was associated with poor prognosis (Figure 4), further supporting an oncogenic function of FBXO11 in breast cancer. It is plausible that FBXO11 plays distinct roles in different cancer types and even in different stages of cancer development. In breast cancer, FBXO11 may promote primary tumor growth and suppress apoptosis in response to cytotoxic chemotherapy by inhibiting p53 activity in those cancer cells harboring functional p53, whereas it could repress metastasis via degrading SNAIL in p53-mutated cancer cells at late stages of breast cancer.
Consistent with a previous report, our data support that FBXO11 could suppress p53 transactivity and reduce breast cancer apoptosis upon chemotherapy (Figure 5). Besides, FBXO11 may also antagonize p53 by indirectly destabilizing p53 protein via Set8-depedent Numb acetylation, thereby promoting breast cancer progression.37, 44 Accordingly, we found CDK inhibitor p21 expression, as a primary gene target of p53, was decreased in breast cancer cells overexpressing FBXO11 but increased in miR-621-transfected cells upon CBP or PTX treatment (Figures 5f and g). However, FBXO11-mediated Cdt2 degradation was shown to stabilize p21, leading in increased p21 level.35 Therefore, it is likely that decreased p21 transcription due to FBXO11-dependent p53 inhibition overweighed the p21 protein stabilization due to FBXO11-mediated Cdt2 degradation in breast cancer cells exposed to chemotherapeutic drugs. Nevertheless, it is also possible there was additional miR-621-mediated mechanism promoting p21 upregulation which remains to be elucidated.
Although TP53 gene is frequently mutated in a variety of tumors, such as in ovarian (50%), colorectal (43%) and lung cancers (36%), only ~23% of breast cancer tumors harbor TP53 mutation, suggesting additional mechanisms for silencing p53 in breast cancer cells.45 The wild-type p53 tumor suppressor alters gene expression in response to diverse cellular stresses, including genotoxic drug treatment, leading to cell cycle arrest, apoptosis, senescence and DNA repair. Beside of inactivation of p53 by proteasome-dependent degradation, p53 neddylation was shown to inhibit its transactivity.37, 46 These findings are in agreement with a recent report showing that the over-activated neddylation pathway is a critical player promoting lung cancer development.47 Therefore, varying mechanisms developed in breast cancer cells to suppress wild-type p53 function may contribute substantially to therapeutic resistance to cytotoxic treatments which induce cancer cell apoptosis. FBXO11-dependent silencing of p53 transactivity may disrupt the apoptotic machinery, resulting in resistance to various anti-cancer drugs. Our results indicated that the miR-621-mediated suppression of FBXO11 could enhance the p53 transcriptional pathway, thereby promoting breast cancer cell apoptosis, especially in those harboring functional p53, in response to PTX and CBP treatment (Figures 3, 5 and 6). These findings suggest that upregulating miR-621 may be a promising chemo-sensitizing strategy, which may help to achieve better pCR rate in NAC as well as improve prognosis for systemic therapy in breast cancer patients.
In conclusion, our data demonstrate that elevated miR-621 expression levels may have important roles in enhancing sensitivity to PTX/CBP regimen chemotherapy in breast cancer patients. In addition, we show that miR-621 mediated-PTX and CBP sensitivity is, at least in part, mediated by downregulation of FBXO11 and subsequent increase of p53 transactivity. A single miRNA may regulates multiple genes within a signaling network or parallel networks which could work synergistically to modulate cellular processes. Although our present results established a link between miR-621 and FBXO11, it is plausible that additional targets of miR-621 may also contribute to miR-621’s function in promoting chemotherapy sensitivity. The efficacy of miR-621 in sensitizing breast cancer to chemotherapy may also support its application as potential chemo-sensitizing agents. Our studies may provide rationale for using miR-621 as predictive biomarker to stratify patients before receiving PTX/CBP NAC, and developing miR-621-based therapeutic agents for breast cancer treatment.
Materials and methods
Patient selection, evaluation of treatment response, sample collection and RNA extraction
A series of clinical data from 50 breast cancer patients was collected (Table 1). The design of this phase II neoadjuvant treatment study has been previously described.25 The protocol was reviewed and approved by an independent ethical committee/institutional review board, and all patients gave their written informed consent before inclusion in this study. All eligible patients received four cycles of weekly PTX/CBP regimen.25 Patients proceeded to surgery within 4 weeks of the last dose of chemotherapy. pCR was defined as the absence of invasive tumor cells in the final surgical breast and axillary lymph node samples. Residual ductal carcinoma in situ was included in the pCR category. Tumor tissue was obtained by a core biopsy prior to treatment and immediately stored at −80 °C. Total RNA was extracted with Trizol reagent (Invitrogen, Carlsbad, CA, USA) following the manufacturer’s instructions.
Cell lines, plasmids and transfection
MCF-7, MDA-MB-231, ZR-75-1, HT1080 and HEK293T cell lines were obtained from ATCC. The high pulmonary metastatic MDA-MB-231HM cell line, derived from MDA-MB-231, was established by our institute and reported previously.48, 49 HCT 116, HCT 116 Dicer−/− and HCT 116 p53−/− cells were obtained as a generous gift from Dr Bert Vogelstein (Johns Hopkins University). All these cells were cultured under standard conditions.
Control (26164) sponge construct obtained from Addgene and the strategy to generate miR-621 sponge has been described in a previous report.50 FBXO11 expression constructs were subcloned from ImageClone BC130445 (Thermo, Rockford, IL, USA) into pCMV backbone. FBXO11ΔN construct was generated by deleting FBXO11 N-terminal 1–127 amino acids (containing F-box). FBXO11 3-UTR was cloned from HeLa genomic DNA (NEB) and inserted into pmirGLO Dual-Luciferase Vector (Promega, Madison, WI, USA) for generating FBXO11 3-UTR luciferase reporter constructs. FBXO11 3-UTR miR-621 site mutant luciferase reporter was generated by Quick change site-direct mutagenesis. All plasmids were transfected with Lipofectamine 2000 (Invitrogen).
Stable expression of miR-621 with lentivirus vector
Human miR-621 gene was PCR amplified from normal genomic DNA and cloned into pCDH-CMV-MCS-EF1-Puro vector for ectopic expression of miR-621. MCF-7 cells were infected with vector control or miR-621-overexpression virus and selected by Puromycin. The expression levels of miR-621 between MCF-7/miR-621 and MCF-7/vector were confirmed by quantitative real-time PCR (fold change 10.39) (Supplementary Figure S5b).
Immunoprecipitation and immunoblotting
For co-immunoprecipitation experiments, cells were lysed in 10% PBS and 90% Lysis buffer (20 mm Tris (pH 7.0), 250 mm NaCl, 3 mm EDTA, 3 mm EGTA, 0.5% NP-40, 2 mm DTT, 0.5 mm PMSF, 20 mm β-glycerol phosphate, 1 mm sodium orthovanadate, 1 μg/ml leupeptin, 1 μg/ml aprotinin, 10 mm p-nitrophenyl phosphate, 10 mM sodium fluoride). In all, 1 μg of indicated antibody or IgG control was added into lysates and incubated with protein G-Sepharose at 4 °C overnight. Sepharose-enriched immunocomplexes were resolved on SDS-PAGE, transferred to polyvinylidene fluoride (PVDF) membrane and analyzed with immunoblotting.
To detect neddylated form of p53, cells were lyzed with 1% SDS in immunoprecipitation lysis buffer, incubated at 95 °C for 30 min, then diluted to 0.1% SDS with lysis buffer and subjected to immunoprecipitation with anti-p53 antibody. The transferred PVDF membrane was probed with antibody against NEDD8.
Animal study
All animal work was done in accordance with a protocol approved be the Institutional Animal Care and Use Committee at Fudan University Shanghai Cancer Center. MCF-7/miR-621; MCF-7/vector (1x107) cells were injected into the mammary fat pads of BALB/c nude mice. All mice were supplemented with estrogen pellets. 20 days after tumor cell transplantation, the mice were injected intraperitoneally with PTX (15 mg/kg; Sigma, St Louis, MO, USA) plus CBP (5 mg/kg; Sigma). The treatments were administered every 4 days for six cycles. Tumor sizes were measured with caliper and calculated by the formula V=(W)2xL/2. At end point, the mice were killed and the mammary tumors were harvested for quantitative real-time PCR, western blot and immunohistochemistry staining.
Statistical analyses
Statistical analysis was done by one-way ANOVA, and comparisons among groups were done by the independent sample two-sided Student t-test. Patient DFS analysis was estimated by the Kaplan–Meier method. Fisher’s exact test was used to analyze categorical variables. The Spearman rank test was used to identify the correlation between miR-621 and FBXO11. Multiple regression analysis was used to determine independent predict factors for achieving pCR. ROC curve was used to analyze the predictive value of miR-621. All statistical analyses were performed using SPSS 22.0 software (SPSS Inc, Chicago, IL, USA). P-value< 0.05 was considered as statistically significant.
References
Berry DA, Cronin KA, Plevritis SK, Fryback DG, Clarke L, Zelen M et al. Effect of screening and adjuvant therapy on mortality from breast cancer. New Engl J Med 2005; 353: 1784–1792.
Dieras V, Fumoleau P, Romieu G, Tubiana-Hulin M, Namer M, Mauriac L et al. Randomized parallel study of doxorubicin plus paclitaxel and doxorubicin plus cyclophosphamide as neoadjuvant treatment of patients with breast cancer. J Clin Oncol 2004; 22: 4958–4965.
Iwata H, Sato N, Masuda N, Nakamura S, Yamamoto N, Kuroi K et al. Docetaxel followed by fluorouracil/epirubicin/cyclophosphamide as neoadjuvant chemotherapy for patients with primary breast cancer. Jpn J Clinical Oncol 2011; 41: 867–875.
Lawrence G, Crawford J, Sherman F . Evaluating neoadjuvant chemotherapy in breast cancer. J Clin Oncol 2002; 20: 2906–2907.
Smith IC, Heys SD, Hutcheon AW, Miller ID, Payne S, Gilbert FJ et al. Neoadjuvant chemotherapy in breast cancer: significantly enhanced response with docetaxel. J Clin Oncol 2002; 20: 1456–1466.
Nowak AK, Wilcken NR, Stockler MR, Hamilton A, Ghersi D . Systematic review of taxane-containing versus non-taxane-containing regimens for adjuvant and neoadjuvant treatment of early breast cancer. Lancet Oncol 2004; 5: 372–380.
Balko JM, Giltnane JM, Wang K, Schwarz LJ, Young CD, Cook RS et al. Molecular profiling of the residual disease of triple-negative breast cancers after neoadjuvant chemotherapy identifies actionable therapeutic targets. Cancer Discov 2014; 4: 232–245.
Harris L, Fritsche H, Mennel R, Norton L, Ravdin P, Taube S et al. American Society of Clinical Oncology 2007 update of recommendations for the use of tumor markers in breast cancer. J Clin Oncol 2007; 25: 5287–5312.
Yerushalmi R, Woods R, Ravdin PM, Hayes MM, Gelmon KA . Ki67 in breast cancer: prognostic and predictive potential. Lancet Oncol 2010; 11: 174–183.
Borst P, Wessels L . Do predictive signatures really predict response to cancer chemotherapy? Cell Cycle 2010; 9: 4836–4840.
Lee JK, Coutant C, Kim YC, Qi Y, Theodorescu D, Symmans WF et al. Prospective comparison of clinical and genomic multivariate predictors of response to neoadjuvant chemotherapy in breast cancer. Clin Cancer Res 2010; 16: 711–718.
Weigelt B, Pusztai L, Ashworth A, Reis-Filho JS . Challenges translating breast cancer gene signatures into the clinic. Nat Rev Clin Oncol 2012; 9: 58–64.
Thuerigen O, Schneeweiss A, Toedt G, Warnat P, Hahn M, Kramer H et al. Gene expression signature predicting pathologic complete response with gemcitabine, epirubicin, and docetaxel in primary breast cancer. J Clin Oncol 2006; 24: 1839–1845.
Hess KR, Anderson K, Symmans WF, Valero V, Ibrahim N, Mejia JA et al. Pharmacogenomic predictor of sensitivity to preoperative chemotherapy with paclitaxel and fluorouracil, doxorubicin, and cyclophosphamide in breast cancer. J Clin Oncol 2006; 24: 4236–4244.
Tabchy A, Valero V, Vidaurre T, Lluch A, Gomez H, Martin M et al. Evaluation of a 30-gene paclitaxel, fluorouracil, doxorubicin, and cyclophosphamide chemotherapy response predictor in a multicenter randomized trial in breast cancer. Clin Cancer Res 2010; 16: 5351–5361.
Gregory RI, Chendrimada TP, Cooch N, Shiekhattar R . Human RISC couples microRNA biogenesis and posttranscriptional gene silencing. Cell 2005; 123: 631–640.
Lagos-Quintana M, Rauhut R, Lendeckel W, Tuschl T . Identification of novel genes coding for small expressed RNAs. Science 2001; 294: 853–858.
Bartel DP . MicroRNAs: target recognition and regulatory functions. Cell 2009; 136: 215–233.
Shivdasani RA . MicroRNAs: regulators of gene expression and cell differentiation. Blood 2006; 108: 3646–3653.
Ling H, Fabbri M, Calin GA . MicroRNAs and other non-coding RNAs as targets for anticancer drug development. Nat Rev Drug Discov 2013; 12: 847–865.
Blower PE, Chung JH, Verducci JS, Lin S, Park JK, Dai Z et al. MicroRNAs modulate the chemosensitivity of tumor cells. Mol Cancer Therapeut 2008; 7: 1–9.
Pan YZ, Morris ME, Yu AM . MicroRNA-328 negatively regulates the expression of breast cancer resistance protein (BCRP/ABCG2) in human cancer cells. Mol Pharmacol 2009; 75: 1374–1379.
Zhou M, Liu Z, Zhao Y, Ding Y, Liu H, Xi Y et al. MicroRNA-125b confers the resistance of breast cancer cells to paclitaxel through suppression of pro-apoptotic Bcl-2 antagonist killer 1 (Bak1) expression. J Biol Chem 2010; 285: 21496–21507.
Wang H, Tan G, Dong L, Cheng L, Li K, Wang Z et al. Circulating MiR-125b as a marker predicting chemoresistance in breast cancer. PloS One 2012; 7: e34210.
Chen XS, Nie XQ, Chen CM, Wu JY, Wu J, Lu JS et al. Weekly paclitaxel plus carboplatin is an effective nonanthracycline-containing regimen as neoadjuvant chemotherapy for breast cancer. Ann Oncol 2010; 21: 961–967.
Bonnefoi H, Litiere S, Piccart M, Macgrogan G, Fumoleau P, Brain E et al. Pathological complete response after neoadjuvant chemotherapy is an independent predictive factor irrespective of simplified breast cancer intrinsic subtypes: a landmark and two-step approach analyses from the EORTC 10994/BIG 1-00 phase III trial. Ann Oncol 2014; 25: 1128–1136.
Mamounas EP, Anderson SJ, Dignam JJ, Bear HD, Julian TB, Geyer CE Jr. et al. Predictors of locoregional recurrence after neoadjuvant chemotherapy: results from combined analysis of National Surgical Adjuvant Breast and Bowel Project B-18 and B-27. J Clin Oncol 2012; 30: 3960–3966.
Chen Y, Chen C, Yang B, Xu Q, Wu F, Liu F et al. Estrogen receptor-related genes as an important panel of predictors for breast cancer response to neoadjuvant chemotherapy. Cancer Lett 2011; 302: 63–68.
Wu X, Watson M . CORNA: testing gene lists for regulation by microRNAs. Bioinformatics 2009; 25: 832–833.
Petitjean A, Achatz MI, Borresen-Dale AL, Hainaut P, Olivier M . TP53 mutations in human cancers: functional selection and impact on cancer prognosis and outcomes. Oncogene 2007; 26: 2157–2165.
Powell E, Piwnica-Worms D, Piwnica-Worms H . Contribution of p53 to metastasis. Cancer Discov 2014; 4: 405–414.
Green DR, Kroemer G . Cytoplasmic functions of the tumour suppressor p53. Nature 2009; 458: 1127–1130.
Cummins JM, He Y, Leary RJ, Pagliarini R, Diaz LA Jr., Sjoblom T et al. The colorectal microRNAome. Proc Natl Acad Sci USA 2006; 103: 3687–3692.
Hatzis C, Pusztai L, Valero V, Booser DJ, Esserman L, Lluch A et al. A genomic predictor of response and survival following taxane-anthracycline chemotherapy for invasive breast cancer. JAMA 2011; 305: 1873–1881.
Abbas T, Mueller AC, Shibata E, Keaton M, Rossi M, Dutta A . CRL1-FBXO11 promotes Cdt2 ubiquitylation and degradation and regulates Pr-Set7/Set8-mediated cellular migration. Mol Cell 2013; 49: 1147–1158.
Skaar JR, Pagan JK, Pagano M . Mechanisms and function of substrate recruitment by F-box proteins. Nat Rev Mol Cell Biol 2013; 14: 369–381.
Abida WM, Nikolaev A, Zhao W, Zhang W, Gu W . FBXO11 promotes the Neddylation of p53 and inhibits its transcriptional activity. J Biol Chem 2007; 282: 1797–1804.
Mulrane L, McGee SF, Gallagher WM, O'Connor DP . miRNA dysregulation in breast cancer. Cancer Res 2013; 73: 6554–6562.
Skawran B, Steinemann D, Becker T, Buurman R, Flik J, Wiese B et al. Loss of 13q is associated with genes involved in cell cycle and proliferation in dedifferentiated hepatocellular carcinoma. Mod Pathol 2008; 21: 1479–1489.
Duan S, Cermak L, Pagan JK, Rossi M, Martinengo C, di Celle PF et al. FBXO11 targets BCL6 for degradation and is inactivated in diffuse large B-cell lymphomas. Nature 2012; 481: 90–93.
Yang CH, Pfeffer SR, Sims M, Yue J, Wang Y, Linga VG et al. The oncogenic microRNA-21 inhibits the tumor suppressive activity of FBXO11 to promote tumorigenesis. J Biol Chem 2015; 290: 6037–6046.
Zheng H, Shen M, Zha YL, Li W, Wei Y, Blanco MA et al. PKD1 phosphorylation-dependent degradation of SNAIL by SCF-FBXO11 regulates epithelial-mesenchymal transition and metastasis. Cancer Cell 2014; 26: 358–373.
Yang F, Sun L, Li Q, Han X, Lei L, Zhang H et al. SET8 promotes epithelial-mesenchymal transition and confers TWIST dual transcriptional activities. EMBO J 2012; 31: 110–123.
Dhami GK, Liu H, Galka M, Voss C, Wei R, Muranko K et al. Dynamic methylation of Numb by Set8 regulates its binding to p53 and apoptosis. Mol Cell 2013; 50: 565–576.
Walerych D, Napoli M, Collavin L, Del Sal G . The rebel angel: mutant p53 as the driving oncogene in breast cancer. Carcinogenesis 2012; 33: 2007–2017.
Xirodimas DP, Saville MK, Bourdon JC, Hay RT, Lane DP . Mdm2-mediated NEDD8 conjugation of p53 inhibits its transcriptional activity. Cell 2004; 118: 83–97.
Li L, Wang M, Yu G, Chen P, Li H, Wei D et al. Overactivated neddylation pathway as a therapeutic target in lung cancer. J Natl Cancer Inst 2014; 106: dju083.
Chang XZ, Li DQ, Hou YF, Wu J, Lu JS, Di GH et al. Identification of the functional role of AF1Q in the progression of breast cancer. Breast Cancer Res Treat 2008; 111: 65–78.
Li JY, Ou ZL, Yu SJ, Gu XL, Yang C, Chen AX et al. The chemokine receptor CCR4 promotes tumor growth and lung metastasis in breast cancer. Breast Cancer Res Treat 2012; 131: 837–848.
Ebert MS, Neilson JR, Sharp PA . MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells. Nat Methods 2007; 4: 721–726.
Acknowledgements
We thank Dr Bert Vogelstein (Johns Hopkins University) for wild-type, DICER−/− and p53−/− HCT116 cells. This work was supported in part by the National Natural Science Foundation of China (NSFC) (81272924 to JW), National Cancer Institute (R01 CA149251 to ZW), and American Cancer Society (RSG-13-186-01-CSM to ZW).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
About this article
Cite this article
Xue, J., Chi, Y., Chen, Y. et al. MiRNA-621 sensitizes breast cancer to chemotherapy by suppressing FBXO11 and enhancing p53 activity. Oncogene 35, 448–458 (2016). https://doi.org/10.1038/onc.2015.96
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2015.96
- Springer Nature Limited
This article is cited by
-
FBXW7 in breast cancer: mechanism of action and therapeutic potential
Journal of Experimental & Clinical Cancer Research (2023)
-
Genetic variations in tumor-suppressor miRNA-encoding genes and their target genes: focus on breast cancer development and possible therapeutic strategies
Clinical and Translational Oncology (2023)
-
miR-211-3p enhances induction chemotherapy insensitivity by upregulating CSF2/CCL20/TNF signaling in hypopharyngeal squamous cell carcinoma
Molecular Biology Reports (2022)
-
Ultrasound-targeted microbubble destruction-mediated silencing of FBXO11 suppresses development of pancreatic cancer
Human Cell (2022)
-
miRNAs as therapeutic predictors and prognostic biomarkers of neoadjuvant chemotherapy in breast cancer: a systematic review and meta-analysis
Breast Cancer Research and Treatment (2022)