Abstract
The revised 2022 European LeukemiaNet (ELN) AML risk stratification system requires validation in large, homogeneously treated cohorts. We studied 1118 newly diagnosed AML patients (median age, 58 years; range, 18–86 years) who received cytarabine-based induction chemotherapy between 1999 and 2012 and compared ELN-2022 to the previous ELN-2017 risk classification. Key findings were validated in a cohort of 1160 mostly younger patients. ELN-2022 reclassified 15% of patients, 3% into more favorable, and 12% into more adverse risk groups. This was mainly driven by patients reclassified from intermediate- to adverse-risk based on additional myelodysplasia-related mutations being included as adverse-risk markers. These patients (n = 79) had significantly better outcomes than patients with other adverse-risk genotypes (5-year OS, 26% vs. 12%) and resembled the remaining intermediate-risk group. Overall, time-dependent ROC curves and Harrel’s C-index controlling for age, sex, and AML type (de novo vs. sAML/tAML) show slightly worse prognostic discrimination of ELN-2022 compared to ELN-2017 for OS. Further refinement of ELN-2022 without including additional genetic markers is possible, in particular by recognizing TP53-mutated patients with complex karyotypes as “very adverse”. In summary, the ELN-2022 risk classification identifies a larger group of adverse-risk patients at the cost of slightly reduced prognostic accuracy compared to ELN-2017.
Similar content being viewed by others
Background
In 2010, an expert panel on behalf of the European LeukemiaNet (ELN) developed guidelines for diagnosis and management of acute myeloid leukemia (AML) in adults [1]. In 2017, an updated version was published [2], including a risk stratification system based on cytogenetic and molecular aberrations. The ELN-2017 risk classification has been validated in intensively treated AML patient cohorts [3,4,5,6,7,8,9], and found widespread adoption in routine practice and clinical trials. In 2022, another update of the ELN guidelines has been published [10]. This latest version introduced multiple changes to the risk stratification system. First, patients with internal tandem duplications of FLT3 (FLT3-ITD) in the absence of core binding factor (CBF) rearrangements or adverse-risk markers are now considered intermediate risk, regardless of FLT3-ITD-to-wild-type (wt) allelic ratio or NPM1 co-mutation. This change is not only intended to account for the impact of FLT3 inhibitors on outcomes, but also comes in the wake of several validation studies not showing different outcomes for patients with low vs. high FLT3-ITD allelic ratios [4, 11]. In contrast, we and others did confirm that consideration of FLT3-ITD allelic ratio improved risk stratification within ELN-2017 [3, 6, 9, 12]. Second, it has become clear that only in-frame mutations in the leucine zipper domain of CEBPA (CEBPAbZIP-inf) predict favorable outcomes [13], and consequently, only those are considered favorable-risk according to ELN-2022 – regardless of whether they occur alone or with a second CEBPA mutation. Finally, in the adverse-risk category, t(8;16)(p11;p13)/KAT6A::CREBBP and t(3q26.2;v)/MECOM(EVI1) rearrangements, and mutations in BCOR, EZH2, SF3B1, SRSF2, STAG2, U2AF1 or ZRSR2 in the absence of favorable-risk markers have been added as poor-risk markers.
While the proposed changes individually are supported by published data, the effects of these modifications on overall risk stratification have not yet been validated in large and homogeneously treated cohorts. We set out to test the prognostic relevance of the ELN-2022 classification in intensively treated AML patients, and to compare this revised risk stratification to the prior ELN-2017 system.
Patients and methods
We studied 1138 newly diagnosed AML patients (median age, 58 years [y]; range, 18–86 y) who received cytarabine-based induction chemotherapy in two subsequent multicenter phase III trials of the German AML Cooperative Group (AMLCG-1999, clinicaltrials.gov identifier NCT00266136, n = 864; and AMLCG-2008, NCT01382147, n = 274) between 1999 and 2012 [14,15,16,17]. Treatment regimens are summarized in the Supplementary Methods, and patient disposition is detailed in a previous report [3]. None of the patients received FLT3 inhibitors or gemtuzumab ozogamicin during first-line treatment. AML was diagnosed according to World Health Organization 2008 criteria [18]. Metaphase cytogenetics were analyzed centrally, and patients were profiled for mutations in 68 genes commonly mutated in myeloid neoplasms via targeted sequencing from bone marrow (BM) or peripheral blood (PB), as described previously [19]. The limit of detection was a variant allele frequency of ≥2%. Variants were classified in accordance with widely accepted consensus classifications [20, 21]. Twenty subjects were excluded due to missing genetic data. All study protocols were in accordance with the Declaration of Helsinki and approved by the institutional review boards of participating centers. All patients provided written informed consent for inclusion on the clinical trial and genetic analyses. Median follow-up of survivors was 98 months [22].
Key findings were validated in a published cohort of 1160 mostly younger AML patients (83% aged <60 y) treated with intensive induction chemotherapy on clinical trials of the Acute Myeloid Leukemia Study Group (AMLSG, Supp. Table 1) [23].
We studied associations between ELN genetic risk groups and other patient characteristics using Fisher’s exact test for categorical and the Wilcoxon rank-sum test for continuous variables. We used widely accepted definitions of common clinical endpoints (complete remission [CR], relapse-free survival [RFS], and overall survival [OS]) (Supplementary Methods) [24]. For time-to-event analyses, we calculated survival estimates using the Kaplan–Meier method except in the case of allogeneic stem cell transplant (alloSCT), where we used Simon–Makuch plots, as described in the Supplementary Methods. We compared groups by the log-rank test. We used multivariable logistic regression models to analyze factors associated with achievement of CR, and Cox proportional hazards models for survival endpoints. All multivariable models were stratified by trial arm to control for possible differences between cohorts. Potential models were tested using Akaike’s ‘An Information Criterion’. Collinearity of variables was tested using variance inflation factor. Statistical analyses were performed using R version 4.2.1 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Association of the ELN-2022 risk groups with baseline demographics and comparison to ELN-2017
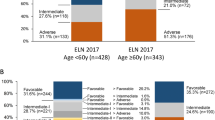
Out of 1118 patients stratified according to ELN-2022, 363 (32%) were classified as favorable, 302 (27%) as intermediate, and 453 (41%) as adverse-risk (Table 1). For those <60 y of age (n = 600), the distribution was 39%, 30%, and 31%, compared to 25%, 24%, and 52% for those aged ≥60 y (n = 518) (Fig. 1A). Similar to ELN-2017, ELN-2022 adverse risk significantly associated with older age (p < 0.0001 for ELN favorable/intermediate vs. adverse), male sex (p = 0.003), secondary AML (sAML; p = 0.0006), and a lower white blood cell (WBC) count at diagnosis (p < 0.0001). These associations persisted in patients aged <60 y or ≥60 y. We did not find a significant association between ELN-2022 adverse risk and tAML (p = 0.21). The significantly higher proportion of adverse-risk genetics among male patients (48% vs. 33%, p < 0.0001, Fig. 1B) was largely due to a lower prevalence of NPM1 mutations and higher prevalence of RUNX1 and ASXL1 mutations in males (p < 0.0001 and p = 0.009, respectively). In addition, some of the newly recognized adverse risk-defining mutations were also significantly associated with male sex (EZH2: p = 0.0002; SRSF2: p < 0,0001; STAG2: p = 0.0075; U2AF1: p = 0.0016; ZRSR2: p = 0.0005).
Compared to ELN-2017, 85% of patients remained in the same risk group. This substantial agreement between ELN-2017 and ELN-2022 was confirmed by Cohen’s kappa (unweighted kappa: 0.73 (95% CI: 0.77–0.80), weighted kappa: 0.84 (95% CI: 0.87–0.89)). Fifteen percent of patients (n = 171; 14% of male and 17% of female patients) were classified into a different ELN-2022 risk category, with 3% moving into a more favorable and 12% into a less favorable category (Fig. 1C). Reasons for reclassification are detailed in Supplementary Table 2.
Outcomes of AML patients classified according to the ELN-2022 risk stratification
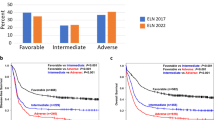
CR rates for patients in the ELN-2022 favorable, intermediate, and adverse risk groups were 73%, 66%, and 45% (Table 2). For the corresponding ELN-2017 categories, CR rates were 72%, 66%, and 41% (Supplementary Table 3). Five-year RFS was 52%, 32%, and 16%, respectively, for the ELN-2022 risk groups compared to 53%, 26%, and 12% for the corresponding ELN-2017 groups. Median RFS by ELN-2022 category was 7.2 y (95% CI, 3.7y-not reached), 1.0 y (95% CI, 0.8–1.4 y) and 0.7 y (95% CI, 0.6–0.9 y) for the favorable, intermediate, and adverse group (Fig. 2A), compared to 7.1 y (95% CI, 3.7y-not reached), 1.0 y (95% CI, 0.7–1.3 y), and 0.6 y (95% CI, 0.6–0.8 y) for the corresponding ELN-2017 categories.
Five-year OS by risk group was 55%, 34%, and 15% for ELN-2022 (Table 2), versus 54%, 31%, and 12% for the corresponding ELN-2017 categories. Median OS by ELN-2022 risk group was 9.5 y (95% confidence interval (CI), 4.8–12.3 y), 1.7 y (95% CI, 1.2–2.0 y), and 0.8 y (95% CI, 0.7–1.0 y) (Fig. 2B), compared to 8.2 y (95% CI, 4.6–11.9 y), 1.7 y (95% CI, 1.2–2.0 y), and 0.8 y (95% CI: 0.7–0.9 y) for the ELN-2017 categories. In analyses stratified by age (<60 y versus ≥60 y), the ELN-2022 classification maintained its prognostic impact in both age groups (Supplementary Fig. 1A–D).
In a multivariate model assessing factors associated with achievement of CR, ELN-2022 adverse risk, older age, higher WBC count, and a diagnosis of sAML or tAML were significantly associated with a lower likelihood of attaining CR (Fig. 3A). In a multivariate model for RFS, ELN-2022 favorable risk associated with longer RFS, while ELN-2022 adverse risk, older age and higher WBC count associated with shorter RFS (Fig. 3B). Those same factors also associated with survival in a multivariate model for OS (Fig. 3C). In addition, tAML associated with shorter OS.
A Forest plot showing odds ratios from a logistic regression model for achievement of complete remission. B Forest plot showing hazard ratios from a Cox proportional hazards model for relapse-free survival. C Forest plot showing hazard ratios from a Cox proportional hazards model for overall survival. Interaction P values refer to an interaction between the ELN-2022 risk groups and the respective variable.
Receiver operating characteristic (ROC) curves showed numerically lower areas under the curves for the associations of ELN-2022 with OS and RFS, compared to ELN-2017. ELN-2022 performed significantly worse at some timepoints (Supplementary Fig. 2A, B). Harrel’s C-index confirmed a slightly lower prognostic accuracy of ELN-2022 for OS, with values of 0.658 for ELN-2022, and 0.664 for ELN-2017, when controlling for age, sex, and presence of sAML or tAML.
Kaplan–Meier plots for the entire cohort classified using either ELN-2022 or ELN-2017 are shown in Fig. 4. We observed a trend towards better outcomes for the ELN-2022 adverse group compared to ELN-2017 adverse-risk patients. We therefore analyzed outcomes of reclassified subgroups in more detail.
Outcomes of reclassified subgroups
RFS and OS of reclassified patients are detailed in Supplementary Fig. 3. Only twelve patients were reclassified from the ELN-2017 adverse or intermediate groups into the ELN-2022 favorable risk-group, precluding formal outcome analyses for this subset (Supplementary Fig. 3A, B). The 61 patients reclassified from ELN-2017 favorable to ELN-2022 intermediate risk had a numerically higher 5 y OS rate than other ELN-2022 intermediate-risk patients (48% vs. 33%, Supplementary Fig. 3D; p = 0.307). In contrast, patients reclassified from ELN-2017 adverse to ELN-2022 intermediate risk (n = 21) had numerically worse 5 y OS than other intermediate risk patients (10% vs. 33%, Supplementary Fig. 3D; p = 0.068), and significantly worse OS than those reclassified from favorable to intermediate risk (p = 0.016). Finally, patients reclassified from ELN-2017 intermediate to ELN-2022 adverse risk (n = 68) achieved significantly better 5 y OS than other adverse-risk patients (25% vs. 12%, p = 0.007; Supplementary Fig. 3F).
Outcomes stratified by postremission therapy
Of all patients in our cohort, 665 reached CR. Of those, 109 underwent alloSCT in first CR (CR1; 97 aged <60 y and 12 aged ≥60 y). Since alloSCT in CR1 was rare in older patients, we analyzed outcomes according to postremission therapy only in patients <60 y who achieved CR1. This subgroup (n = 381) is characterized in detail in Supplementary Table 4. Even though our cohort was largely recruited before the widespread adoption of risk scores incorporating molecular genetics, we found a significant association of alloSCT in CR1 with ELN-2022 risk group (18% of favorable, 30% of intermediate, and 34% of adverse risk patients, p < 0.0001).
Supplementary Fig. 4 shows RFS and OS of ELN-2022 favorable, intermediate, and adverse-risk patients stratified by postremission therapy. In proportional hazards models calculated within each risk group using transplant status as a time-dependent covariable, RFS was numerically better for those receiving allogeneic transplant compared with those receiving chemotherapy or ASCT within all groups. However, this difference was significant only for favorable and adverse risk patients (p = 0.0028 and p = 0.024, respectively; Supplementary Fig. 4A, E). In the ELN-2022 favorable and intermediate groups, OS was not significantly different between patients receiving or not receiving an allogeneic transplant in CR1, while in the adverse group, those receiving alloSCT in CR1 had significantly better OS (p = 0.032, Supplementary Fig. 4F). Overall, these findings mirror our previous results using the ELN-2017 risk categories, where adverse risk patients also were the only group having a significant OS benefit from alloSCT. However, since postremission therapy assignment was not randomized, these results are likely to be biased by factors other than baseline genetic risk that have influenced treatment decisions (e.g., comorbidities and performance status). In addition, improved transplant protocols with lower treatment-related mortality may shift this balance in favor of allogeneic transplantation [25].
Outcomes of patients within genetic subsets of the ELN-2022 categories
Outcomes of specific subsets within the ELN-2022 risk categories are presented in detail in the Supplement (Supplementary Table 5, Supplementary Figs. 5, 6). The following observations seem particularly noteworthy: Due to the elimination of FLT3-ITD:wt allelic ratio from the classification, most patients with FLT3-ITD are now classified as intermediate-risk. In support of this modification, ELN-2022 intermediate-risk patients with FLT3-ITD had similar outcomes to other intermediate patients (5 y OS, 35% vs. 34%; Supplementary Fig. 5A, B). Next, we analyzed whether FLT3-ITD allelic ratio would add discriminatory power. While there was no significant OS difference between FLT3-ITDhigh versus FLT3-ITDlow patients within the intermediate risk group, those with FLT3-ITDlow (n = 78; 5 y OS, 45%) tended to have better survival than FLT3-ITDhigh patients (n = 126; 5 y OS, 27%) (Supplementary Fig. 5C, D, p = 0.097). Among patients with FLT3-ITD in the adverse-risk group, there was a significant difference between the FLT3-ITDhigh and FLT3-ITDlow subgroups (ITD-high: n = 43; 5 y OS, 10%; ITD-low: n = 42; 5 y OS, 25%; p = 0.027; Supplementary Fig. 5C, D). FLT3-ITD mutations were too rare in the ELN-2022 favorable-risk group (n = 14) to allow similar comparisons.
Outcomes of patients with myelodysplasia-related mutations
Seventy-nine patients, or 45% of all re-classified patients, were moved from ELN-2017 favorable (n = 11) or intermediate (n = 68) to ELN-2022 adverse risk based on the inclusion of additional myelodysplasia-related (MR) mutations (BCOR, EZH2, SF3B1, SRSF2, STAG2, U2AF1, ZRSR2) as poor-risk markers. The presence of MR mutations significantly correlated with older age (46/79 reclassified patients were aged ≥60 y) and male sex (48/79) (p < 0.0001 for both). These re-classified patients had significantly better RFS (5 y RFS, 25% vs. 12%; median RFS, 1.5 y vs. 0.6 y; p = 0.0035) and OS (5 y OS, 26% vs. 12%; median OS, 1.7 y vs. 0.7 y; p = 0.0004) than patients with other adverse-risk genotypes (including ASXL1, RUNX1 or TP53 mutations), and did not show a significant difference in RFS (p = 0.91) or OS (p = 0.34) compared to the ELN-2022 intermediate cohort (Fig. 5). Although patients with MR-associated mutations seemed to have a particularly strong benefit from alloSCT in CR1 (OS p = 0.0026; Supplementary Fig. 4G, H), the limited patient number (nalloSCT = 6) precludes definitive conclusions.
These findings were confirmed in an independent validation cohort of 1160 patients. Here, we also found significantly better OS for those reassigned to adverse risk based on MR mutation positivity (5 y OS, 30% vs. 18%; median OS, 1.6 vs. 1.0 y; p = 0.0052) compared to other adverse risk patients. In the validation cohort, OS of reassigned MR-mutated patients was significantly worse than for the remaining intermediate-risk group (p = 0.02).
Potential refinement of ELN-2022 without inclusion of additional markers
In our validation of the ELN-2017 risk classification, we proposed a refinement of the risk stratification system, without introducing additional genetic markers [3]. Within the novel ELN-2022 risk groups, patients with CBFB::MYH11 or CEBPAbZIP-inf mutations still had superior OS to other favorable-risk patients, with an estimated 5-year OS of 71% and 60%, respectively, compared to patients with RUNX1::RUNX1T1 or NPM1mut without FLT3mut who achieved 5-year OS rates of 50% and 51%, respectively (Supplementary Fig. 6A, B). On the other hand, patients with complex karyotypes in combination with mutated TP53 had particularly unfavorable outcomes, with a 5-year RFS and OS of 0% (Supplementary Fig. 6E, F). Based on these observations and previously published data, [23, 26,27,28,29,30] we maintain our proposal to refine the ELN risk groups by delineating a “very favorable” risk group including patients with either CBFB::MYH11 or CEBPAbZIP-inf without cytogenetic changes classified as intermediate or adverse risk (n = 89, or 8% of our cohort). On the other hand, patients harboring both a complex karyotype and mutated TP53 should be considered “very adverse” (n = 62; 6% of our cohort). According to this refined classification, CR rates for the very favorable, favorable, intermediate, adverse, and very adverse groups were 76, 72, 67, 47, and 27%, respectively (Supplementary Table 6). RFS and OS for this refined ELN-2022 classification are shown in Fig. 6, and RFS and OS stratified by age in Supplementary Fig. 7. Estimated 5-year OS was 65%, 51%, 34%, 17%, and 0%, respectively. In multivariable analyses adjusting for potential confounders (Supplementary Fig. 8), the “very adverse” group of this refined classification had inferior CR rate, RFS, and OS compared to the adverse group. The very favorable-risk subgroup had longer OS compared with the favorable subgroup, although CR rate and RFS were not significantly different. This OS difference was driven by survival after relapse (Supplementary Fig. 9), which was significantly longer for the very favorable compared with the favorable (p = 0.002) and all other subgroups, consistent with reports that patients with CEBPA mutations are particularly responsive to salvage therapies [31,32,33,34]. Our proposed refinement of the ELN-2022 risk stratification was validated in the AMLSG patient cohort. There, we observed a trend towards better OS of the very favorable compared to the favorable group (5 y OS, 77% vs. 58%; median OS, not reached vs. 8.5 y; p = 0.06), and significantly worse survival for the very adverse compared to the adverse group (5 y OS, 0% vs. 24%; median OS, 0.5 y vs. 1.2 y; p < 0.0001).
Discussion
The ELN-2017 recommendations for risk stratification of AML have achieved broad influence in clinical practice and were adopted worldwide [35]. Therefore, it is likely that the changes introduced by the ELN-2022 guidelines will also find their way into clinical trials and routine practice. Because only ~15% of AML patients are reclassified by the new recommendations, and outcomes of individual risk groups as well as overall prognostic accuracy remain largely similar, ELN-2022 represents an incremental change over the previous classification. However, for those patients affected by the proposed changes, it is still of utmost importance to evaluate whether that incremental change is a step towards more accurate risk prediction. Our analyses shed a mixed light on the newly introduced changes.
Like ELN-2017, ELN-2022 is a robust risk stratification system applicable in both younger and older patients who undergo intensive treatment. The association between male sex and adverse genetic risk, which we already observed for ELN-2017, still holds true for the new classifier. While this effect is largely due to sex differences in the frequency of mutations in ASXL1, NPM1, and RUNX1, it is augmented by the introduction of additional MR-related mutations as adverse-risk-defining, as presence of these mutations also associated with male sex. [19, 36,37,38] These findings are in line with a population-based analysis of U.S. SEER data, where male sex was an independent risk factor for worse OS [39]. However, gender did not associate with OS in a Swedish cohort study [40].
ELN-2022 recognizes MR mutations in BCOR, EZH2, SF3B1, SRSF2, STAG2, U2AF1, and ZRSR2 as independent markers of adverse risk. The idea that these mutations reflect myelodysplasia is reflected in the WHO-classification and the International-Consensus-Classification which – with the exemption of RUNX1 in the WHO-classification – also see these mutations as defining AML with myelodysplasia-related genetic changes [41, 42]. While these mutations mostly occur in the setting of sAML, their prognostic significance is not entirely clear [43].
In our cohort, recognition of these mutations as adverse-risk markers lead to reclassification of 79 patients, corresponding to about 7% of the entire cohort and 45% of all reclassified patients. However, our analysis does not support this modification to the risk classification, since RFS and OS of patients re-assigned to the adverse-risk group because of MR mutations were more favorable compared to other adverse risk patients, and not significantly worse than for the remaining intermediate-risk group. While the validation cohort confirms significantly better OS compared to other adverse-risk patients, it also shows worse outcomes compared to intermediate-risk. This outcome might nuance our finding but does not unequivocally support grouping MR-mutated with other adverse-risk patients. In summary, MR mutations in the absence of other, previously recognized adverse-risk markers do not constitute major independent drivers of poor outcomes in younger or elderly patients who receive intensive induction therapy. Hence, these patients might be better classified as intermediate-risk.
The new classification simplifies risk stratification by no longer considering FLT3-ITD:wt allelic ratio. This change is supported by our analyses as we did not find significant differences in survival when sub-stratifying the new ELN-2022 risk groups by the presence of FLT3-ITD, or by FLT3-ITD ratio, despite our cohort being treated in the pre-FLT3-inhibitor era. Given the disease-modifying effect of FLT3 inhibitors, the unfavorable prognostic impact of FLT3-ITDs is expected to be reduced further in patients receiving TKI treatment along with frontline intensive therapy [11].
While the ELN risk groups achieve reliable prognostic stratification, further refinement through identification of particularly favorable or adverse subgroups may be clinically beneficial, particularly if this can be achieved without including additional markers. To this end, we [3] and others [23, 26, 27] have reported that CBFB::MYH11 rearrangement associates with better outcomes than RUNX1::RUNX1T1, although a previous study from the UK did not find a difference between these subgroups [44]. In our validation of the ELN-2017 risk groups, we identified patients with biallelic CEBPA mutations as another subgroup with particularly favorable outcomes [23, 31]. Meanwhile, it has become clear that CEBPAbZIP-inf mutations, rather than biallelic mutations, are the CEBPA variants most specifically associated with good outcome [13]. In line with these data, our analysis show that in the context of ELN-2022, patients carrying CBFB::MYH11 or CEBPAbZIP-inf constitute a subset with “very favorable” outcomes that can be separated from the remaining “favorable” patients.
On the other side, patients with both a mutation in TP53 and a complex karyotype have dismal survival, with a 5-year OS of 0% [3, 28, 29]. Because of this grave unmet clinical need, the apparent lack of benefit from established intensive therapies, and inferior outcomes even compared to other ELN-2022 adverse risk patients, assigning these patients into a distinct “very unfavorable” risk group seems warranted. This group should be treated on clinical trials whenever possible.
The major strength of our study is the large patient cohort which was, across a broad age range, uniformly treated using cytarabine- and anthracycline- based induction regimens. Therefore, we can avoid biases potentially introduced by combining patient cohorts treated on different protocols with varying inclusion and exclusion criteria and treatment approaches. Furthermore, many intermediate- and adverse-risk patients received an allogeneic transplant in first remission, reflecting current standards of care. Limitations of our analysis include the fact that none of the patients in our cohort received novel agents recently introduced into the frontline standard of care, such as gemtuzumab-ozogamicin, midostaurin or CPX-351. These new therapies have been shown to improve outcomes in specific patient subgroups. Our validation study, and arguably the ELN risk stratification itself, do not reflect such subgroup-specific effects of novel, often genetically targeted, therapies.
Importantly, the ELN risk groups were developed based on data from cohorts of relatively young patients who were able to receive intensive induction chemotherapy, usually in the context of clinical trials or registries. While our study included patients fit for intensive therapy with no upper age limit, the median age of our cohort was 58 years – approximately 10 years below the age median of all AML patients. Our results, and again the ELN risk groups per se, should not be generalized to the large group of older AML patients receiving less-intensive treatment. In this context, a recent analysis in patients treated with azacitidine and venetoclax showed that the ELN-2017 risk categories appeared to achieve less clear prognostic separation than among intensively treated patients [45].
One reason for the widespread adoption of the ELN risk groups is their relative simplicity, as risk stratification is largely based on individual genetic alterations, while few gene:gene interactions and no non-genetic factors are considered. More comprehensive scores that also incluse clinical parameters such as performance status have been published, but are used less commonly, in part due to their higher complexity [46]. Machine learning approaches incorporating a broader spectrum of risk factors, and using complex mathematical models to derive more granular risk predictions, have been shown to refine prognostic discrimination and may help address some of the challenges outlined before, but are not yet broadly adopted [47, 48].
While the increasing number of approved therapeutics both in the first-line and relapse setting [11, 49,50,51,52,53,54,55,56,57,58] is good news for AML patients and clinicians, it also creates an urgent need to move from prognostic classifications that reflect historical outcomes of one specific therapeutic approach, to predictive models that will allow us to compare expected outcomes for different treatment strategies, and thereby select the most promising option. These models will also need to be able to account for (non-)accessibility of certain therapies due to local approval status or economic constraints, and evolve constantly based on the availability of newer therapies and updated clinical results.
In summary, our validation of the ELN-2022 risk stratification shows that more patients now fall in the adverse risk category, which trends towards having better outcomes than the adverse risk category of the previous ELN-2017 recommendations. Specifically, our data suggest the MR mutations newly classified as adverse-risk markers drive this change and should be more appropriately included in the intermediate-risk category. Further refinement of ELN-2022, especially to emphasize the unmet need of patients with a very poor prognosis, would be feasible by using markers already included in the classifier. Considering additional aspects of disease biology beyond gene mutations and incorporating the effects of new drugs as well as dynamic information along the disease course, can be expected to result in further improvements of AML prognostication.
Data availability
Legal restrictions prohibit us from publicly sharing raw sequencing data, which however can be made available upon reasonable request and permission of the local ethics committee.
References
Döhner H, Estey EH, Amadori S, Appelbaum FR, Büchner T, Burnett AK, et al. Diagnosis and management of acute myeloid leukemia in adults: recommendations from an international expert panel, on behalf of the European LeukemiaNet. Blood. 2010;115:453–74.
Döhner H, Estey E, Grimwade D, Amadori S, Appelbaum FR, Büchner T, et al. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood. 2017;129:424–47.
Herold T, Rothenberg-Thurley M, Grunwald VV, Janke H, Goerlich D, Sauerland MC, et al. Validation and refinement of the revised 2017 European LeukemiaNet genetic risk stratification of acute myeloid leukemia. Leukemia. 2020;34:3161–72.
Boddu PC, Kadia TM, Garcia-Manero G, Cortes J, Alfayez M, Borthakur G, et al. Validation of the 2017 European LeukemiaNet classification for acute myeloid leukemia with NPM1 and FLT3-internal tandem duplication genotypes. Cancer. 2019;125:1091–100.
Bataller A, Garrido A, Guijarro F, Oñate G, Diaz-Beyá M, Arnan M, et al. European LeukemiaNet 2017 risk stratification for acute myeloid leukemia: validation in a risk-adapted protocol. Blood Adv. 2022;6:1193–206.
Döhner K, Thiede C, Jahn N, Panina E, Gambietz A, Larson RA, et al. Impact of NPM1/FLT3-ITD genotypes defined by the 2017 European LeukemiaNet in patients with acute myeloid leukemia. Blood. 2020;135:371–80.
Hansen DK, Kim J, Thompson Z, Hussaini M, Nishihori T, Ahmad A, et al. ELN 2017 genetic risk stratification predicts survival of acute myeloid leukemia patients receiving allogeneic hematopoietic stem cell transplantation. Transplant Cell Ther. 2021;27:256.e251–256.e257.
Harada Y, Nagata Y, Kihara R, Ishikawa Y, Asou N, Ohtake S, et al. Prognostic analysis according to the 2017 ELN risk stratification by genetics in adult acute myeloid leukemia patients treated in the Japan Adult Leukemia Study Group (JALSG) AML201 study. Leuk Res. 2018;66:20–27.
Eisfeld A-K, Kohlschmidt J, Mims A, Nicolet D, Walker CJ, Blachly JS, et al. Additional gene mutations may refine the 2017 European LeukemiaNet classification in adult patients with de novo acute myeloid leukemia aged <60 years. Leukemia. 2020;34:3215–27.
Döhner H, Wei AH, Appelbaum FR, Craddock C, DiNardo CD, Dombret H, et al. Diagnosis and management of AML in adults: 2022 ELN recommendations from an international expert panel. Blood. 2022;140:1345–77.
Stone RM, Mandrekar SJ, Sanford BL, Laumann K, Geyer S, Bloomfield CD, et al. Midostaurin plus chemotherapy for acute myeloid leukemia with a FLT3 mutation. N Engl J Med. 2017;377:454–64.
Sakaguchi M, Yamaguchi H, Najima Y, Usuki K, Ueki T, Oh I, et al. Prognostic impact of low allelic ratio FLT3-ITD and NPM1 mutation in acute myeloid leukemia. Blood Adv. 2018;2:2744–54.
Taube F, Georgi JA, Kramer M, Stasik S, Middeke JM, Röllig C, et al. CEBPA mutations in 4708 patients with acute myeloid leukemia: differential impact of bZIP and TAD mutations on outcome. Blood. 2022;139:87–103.
Braess J, Amler S, Kreuzer KA, Spiekermann K, Lindemann HW, Lengfelder E, et al. Sequential high-dose cytarabine and mitoxantrone (S-HAM) versus standard double induction in acute myeloid leukemia-a phase 3 study. Leukemia. 2018;32:2558–71.
Büchner T, Berdel WE, Haferlach C, Haferlach T, Schnittger S, Müller-Tidow C, et al. Age-related risk profile and chemotherapy dose response in acute myeloid leukemia: a study by the German Acute Myeloid Leukemia Cooperative Group. J Clin Oncol. 2009;27:61–9.
Krug U, Berdel WE, Gale RP, Haferlach C, Schnittger S, Müller-Tidow C, et al. Increasing intensity of therapies assigned at diagnosis does not improve survival of adults with acute myeloid leukemia. Leukemia. 2016;30:1230–6.
Büchner T, Berdel WE, Schoch C, Haferlach T, Serve HL, Kienast J, et al. Double induction containing either two courses or one course of high-dose cytarabine plus mitoxantrone and postremission therapy by either autologous stem-cell transplantation or by prolonged maintenance for acute myeloid leukemia. J Clin Oncol. 2006;24:2480–9.
Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H, et al. WHO classification of tumours of haematopoietic and lymphoid tissues, vol. 2. Lyon: International agency for research on cancer; 2008.
Metzeler KH, Herold T, Rothenberg-Thurley M, Amler S, Sauerland MC, Görlich D, et al. Spectrum and prognostic relevance of driver gene mutations in acute myeloid leukemia. Blood. 2016;128:686–98.
Li MM, Datto M, Duncavage EJ, Kulkarni S, Lindeman NI, Roy S, et al. Standards and guidelines for the interpretation and reporting of sequence variants in cancer: a joint consensus recommendation of the Association for Molecular Pathology, American Society of Clinical Oncology, and College of American Pathologists. J Mol Diagn. 2017;19:4–23.
Froyen G, Le Mercier M, Lierman E, Vandepoele K, Nollet F, Boone E, et al. Standardization of somatic variant classifications in solid and haematological tumours by a two-level approach of biological and clinical classes: an initiative of the Belgian ComPerMed Expert Panel. Cancers. 2019;11:2030.
Korn EL. Censoring distributions as a measure of follow-up in survival analysis. Stat Med. 1986;5:255–60.
Papaemmanuil E, Gerstung M, Bullinger L, Gaidzik VI, Paschka P, Roberts ND, et al. Genomic classification and prognosis in acute myeloid leukemia. N Engl J Med. 2016;374:2209–21.
Cheson BD, Bennett JM, Kopecky KJ, Büchner T, Willman CL, Estey EH, et al. Revised recommendations of the International Working Group for Diagnosis, Standardization of Response Criteria, Treatment Outcomes, and Reporting Standards for Therapeutic Trials in Acute Myeloid Leukemia. J Clin Oncol. 2003;21:4642–9.
Loke J, Buka R, Craddock C. Allogeneic stem cell transplantation for acute myeloid leukemia: who, when, and how? Front Immunol. 2021;12:659595.
Brunner AM, Blonquist TM, Sadrzadeh H, Perry AM, Attar EC, Amrein PC, et al. Population-based disparities in survival among patients with core-binding factor acute myeloid leukemia: a SEER database analysis. Leuk Res. 2014;38:773–80.
Marcucci G, Mrózek K, Ruppert AS, Maharry K, Kolitz JE, Moore JO, et al. Prognostic factors and outcome of core binding factor acute myeloid leukemia patients with t(8;21) differ from those of patients with inv(16): a Cancer and Leukemia Group B study. J Clin Oncol. 2005;23:5705–17.
Mrózek K, Eisfeld AK, Kohlschmidt J, Carroll AJ, Walker CJ, Nicolet D, et al. Complex karyotype in de novo acute myeloid leukemia: typical and atypical subtypes differ molecularly and clinically. Leukemia. 2019;33:1620–34.
Rücker FG, Schlenk RF, Bullinger L, Kayser S, Teleanu V, Kett H, et al. TP53 alterations in acute myeloid leukemia with complex karyotype correlate with specific copy number alterations, monosomal karyotype, and dismal outcome. Blood. 2012;119:2114–21.
Pollyea DA, Pratz KW, Wei AH, Pullarkat V, Jonas BA, Recher C, et al. Outcomes in patients with poor-risk cytogenetics with or without TP53 mutations treated with venetoclax and azacitidine. Clin Cancer Res. 2022;28:5272–9.
Schlenk RF, Taskesen E, van Norden Y, Krauter J, Ganser A, Bullinger L, et al. The value of allogeneic and autologous hematopoietic stem cell transplantation in prognostically favorable acute myeloid leukemia with double mutant CEBPA. Blood. 2013;122:1576–82.
Tarlock K, Lamble AJ, Wang Y-C, Gerbing RB, Ries RE, Loken MR, et al. CEBPA-bZip mutations are associated with favorable prognosis in de novo AML: a report from the Children’s Oncology Group. Blood. 2021;138:1137–47.
Dufour A, Schneider F, Metzeler KH, Hoster E, Schneider S, Zellmeier E, et al. Acute myeloid leukemia with biallelic CEBPA gene mutations and normal karyotype represents a distinct genetic entity associated with a favorable clinical outcome. J Clin Oncol. 2010;28:570–7.
Konstandin NP, Pastore F, Herold T, Dufour A, Rothenberg-Thurley M, Hinrichsen T, et al. Genetic heterogeneity of cytogenetically normal AML with mutations of CEBPA. Blood Adv. 2018;2:2724–31.
Tallman MS, Wang ES, Altman JK, Appelbaum FR, Bhatt VR, Bixby D, et al. Acute myeloid leukemia, version 3.2019, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2019;17:721–49.
Döhner K, Schlenk RF, Habdank M, Scholl C, Rücker FG, Corbacioglu A, et al. Mutant nucleophosmin (NPM1) predicts favorable prognosis in younger adults with acute myeloid leukemia and normal cytogenetics: interaction with other gene mutations. Blood. 2005;106:3740–6.
Thiede C, Koch S, Creutzig E, Steudel C, Illmer T, Schaich M, et al. Prevalence and prognostic impact of NPM1 mutations in 1485 adult patients with acute myeloid leukemia (AML). Blood. 2006;107:4011–20.
Gaidzik VI, Teleanu V, Papaemmanuil E, Weber D, Paschka P, Hahn J, et al. RUNX1 mutations in acute myeloid leukemia are associated with distinct clinico-pathologic and genetic features. Leukemia. 2016;30:2160–8.
Acharya UH, Halpern AB, Wu QV, Voutsinas JM, Walter RB, Yun S, et al. Impact of region of diagnosis, ethnicity, age, and gender on survival in acute myeloid leukemia (AML). J Drug Assess. 2018;7:51–3.
Radkiewicz C, Johansson ALV, Dickman PW, Lambe M, Edgren G. Sex differences in cancer risk and survival: a Swedish cohort study. Eur J Cancer. 2017;84:130–40.
Khoury JD, Solary E, Abla O, Akkari Y, Alaggio R, Apperley JF, et al. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Myeloid and Histiocytic/Dendritic Neoplasms. Leukemia. 2022;36:1720–48
Arber DA, Orazi A, Hasserjian RP, Borowitz MJ, Calvo KR, Kvasnicka H-M, et al. International Consensus Classification of Myeloid Neoplasms and Acute Leukemias: integrating morphologic, clinical, and genomic data. Blood. 2022;140:1200–28.
Lindsley RC, Mar BG, Mazzola E, Grauman PV, Shareef S, Allen SL, et al. Acute myeloid leukemia ontogeny is defined by distinct somatic mutations. Blood. 2015;125:1367–76.
Grimwade D, Hills RK, Moorman AV, Walker H, Chatters S, Goldstone AH, et al. Refinement of cytogenetic classification in acute myeloid leukemia: determination of prognostic significance of rare recurring chromosomal abnormalities among 5876 younger adult patients treated in the United Kingdom Medical Research Council trials. Blood. 2010;116:354–65.
Döhner H, Pratz KW, DiNardo CD, Jonas BA, Pullarkat VA, Thirman MJ, et al. ELN risk stratification is not predictive of outcomes for treatment-naïve patients with acute myeloid leukemia treated with venetoclax and azacitidine. Blood. 2022;140:1441–4.
Pastore F, Dufour A, Benthaus T, Metzeler KH, Maharry KS, Schneider S, et al. Combined molecular and clinical prognostic index for relapse and survival in cytogenetically normal acute myeloid leukemia. J Clin Oncol. 2014;32:1586–94.
Gerstung M, Papaemmanuil E, Martincorena I, Bullinger L, Gaidzik VI, Paschka P, et al. Precision oncology for acute myeloid leukemia using a knowledge bank approach. Nat Genet. 2017;49:332–40.
Tazi Y, Arango-Ossa JE, Zhou Y, Bernard E, Thomas I, Gilkes A, et al. Unified classification and risk-stratification in Acute Myeloid Leukemia. Nat Commun. 2022;13:4622.
Burchert A, Bug G, Fritz LV, Finke J, Stelljes M, Röllig C, et al. Sorafenib maintenance after allogeneic hematopoietic stem cell transplantation for acute myeloid leukemia with FLT3–internal tandem duplication mutation (SORMAIN). J Clin Oncol. 2020;38:2993–3002.
Burnett AK, Hills RK, Milligan D, Kjeldsen L, Kell J, Russell NH, et al. Identification of patients with acute myeloblastic leukemia who benefit from the addition of gemtuzumab ozogamicin: results of the MRC AML15 trial. J Clin Oncol. 2011;29:369–77.
Castaigne S, Pautas C, Terré C, Raffoux E, Bordessoule D, Bastie JN, et al. Effect of gemtuzumab ozogamicin on survival of adult patients with de-novo acute myeloid leukaemia (ALFA-0701): a randomised, open-label, phase 3 study. Lancet. 2012;379:1508–16.
Wei AH, Döhner H, Sayar H, Ravandi F, Montesinos P, Dombret H, et al. Long-term overall survival (OS) with oral azacitidine (Oral-AZA) in patients with acute myeloid leukemia (AML) in first remission after intensive chemotherapy (IC): updated results from the phase 3 QUAZAR AML-001 trial. Blood. 2021;138:871.
Lancet JE, Uy GL, Cortes JE, Newell LF, Lin TL, Ritchie EK, et al. CPX-351 (cytarabine and daunorubicin) liposome for injection versus conventional cytarabine plus daunorubicin in older patients with newly diagnosed secondary acute myeloid leukemia. J Clin Oncol. 2018;36:2684–92.
DiNardo CD, Jonas BA, Pullarkat V, Thirman MJ, Garcia JS, Wei AH, et al. Azacitidine and venetoclax in previously untreated acute myeloid leukemia. N Engl J Med. 2020;383:617–29.
DiNardo CD, Pratz K, Pullarkat V, Jonas BA, Arellano M, Becker PS, et al. Venetoclax combined with decitabine or azacitidine in treatment-naive, elderly patients with acute myeloid leukemia. Blood. 2019;133:7–17.
DiNardo CD, Stein EM, de Botton S, Roboz GJ, Altman JK, Mims AS, et al. Durable remissions with ivosidenib in IDH1-mutated relapsed or refractory AML. N Engl J Med. 2018;378:2386–98.
Pollyea DA, Tallman MS, de Botton S, Kantarjian HM, Collins R, Stein AS, et al. Enasidenib, an inhibitor of mutant IDH2 proteins, induces durable remissions in older patients with newly diagnosed acute myeloid leukemia. Leukemia. 2019;33:2575–84.
Venugopal S, Takahashi K, Daver N, Maiti A, Borthakur G, Loghavi S, et al. Efficacy and safety of enasidenib and azacitidine combination in patients with IDH2 mutated acute myeloid leukemia and not eligible for intensive chemotherapy. Blood Cancer J. 2022;12:10.
Acknowledgements
The authors thank all patients and investigators who contributed data to our analysis. This work was supported by funding from the Deutsche José Carreras Leukämie-Stiftung (DJCLS 10R/2021) to TH and the German Cancer Consortium (Deutsches Konsortium für Translationale Krebsforschung, Heidelberg, Germany).
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
CR, TH and KHM conceived and designed the analysis. CR, MR-T, HG, AD, SS, KS, KHM and TH provided and analyzed data. MR-T, HG, AD, SS, KHM and KS characterized patient samples; CS, DG, WEB, BJW, UK, JB and WH coordinated the AMLCG clinical trials. CR, TH and KHM wrote the manuscript. All authors approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
HG is a current employee of Roche Pharma AG, Grenzach-Wyhlen, Germany. The other authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rausch, C., Rothenberg-Thurley, M., Dufour, A. et al. Validation and refinement of the 2022 European LeukemiaNet genetic risk stratification of acute myeloid leukemia. Leukemia 37, 1234–1244 (2023). https://doi.org/10.1038/s41375-023-01884-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41375-023-01884-2
- Springer Nature Limited
This article is cited by
-
Comparison of disease and risk classifications of AML before and after incorporation of NGS analysis of bone marrow samples
International Journal of Hematology (2024)
-
Novel Therapeutic Targets in Acute Myeloid Leukemia (AML)
Current Oncology Reports (2024)
-
CSE1L Silencing Enhances Cytarabine-mediated Cytotoxicity in Acute Myeloid Leukemia
Indian Journal of Hematology and Blood Transfusion (2024)
-
Validation of the 2022 European LeukemiaNet risk stratification for acute myeloid leukemia
Scientific Reports (2024)
-
Prognostic impact of secondary versus de novo ontogeny in acute myeloid leukemia is accounted for by the European LeukemiaNet 2022 risk classification
Leukemia (2023)