Abstract
MYC translocation occurs in 8–14% of diffuse large B-cell lymphoma (DLBCL), and may concur with BCL2 and/or BCL6 translocation, known as double-hit (DH) or triple-hit (TH). DLBCL-MYC/BCL2-DH/TH are largely germinal centre B-cell like subtype, but show variable clinical outcome, with IG::MYC fusion significantly associated with inferior survival. While DLBCL-MYC/BCL6-DH are variable in their cell-of-origin subtypes and clinical outcome. Intriguingly, only 40-50% of DLBCL with MYC translocation show high MYC protein expression (>70%). We studied 186 DLBCLs with MYC translocation including 32 MYC/BCL2/BCL6-TH, 75 MYC/BCL2-DH and 26 MYC/BCL6-DH. FISH revealed a MYC/BCL6 fusion in 59% of DLBCL-MYC/BCL2/BCL6-TH and 27% of DLBCL-MYC/BCL6-DH. Targeted NGS showed a similar mutation profile and LymphGen genetic subtype between DLBCL-MYC/BCL2/BCL6-TH and DLBCL-MYC/BCL2-DH, but variable LymphGen subtypes among DLBCL-MYC/BCL6-DH. MYC protein expression is uniformly high in DLBCL with IG::MYC, but variable in those with non-IG::MYC including MYC/BCL6-fusion. Translocation breakpoint analyses of 8 cases by TLC-based NGS showed no obvious genomic configuration that enables MYC transactivation in 3 of the 4 cases with non-IG::MYC, while a typical promoter substitution or IGH super enhancer juxtaposition in the remaining cases. The findings potentially explain variable MYC expression in DLBCL with MYC translocation, and also bear practical implications in its routine assessment.
Similar content being viewed by others
Introduction
Diffuse large B-cell lymphoma (DLBCL) is a group of heterogeneous aggressive B-cell lymphoma with variable cell-of-origin (COO), genetic changes, molecular mechanisms and clinical outcomes. Based on COO, DLBCL can be broadly classified into activated B-cell like (ABC) and germinal centre B-cell like (GCB) subtype, with a subset of the latter further identified as molecular high grade (MHG)/double-hit signature (DHITsig) due to their enriched MYC expression and centroblast signatures [1, 2]. Based on genetic alterations, DLBCL can be subdivided into distinct subgroups using LymphGen algorithm or other: MCD (MYD88L265P and CD79B mutations), N1 (NOTCH1 mutation), A53 (aneuploidy with TP53 inactivation), BN2 (BCL6 translocation and NOTCH2 mutation), ST2 (SGK1 and TET2 mutated) and EZB (EZH2 mutation and BCL2 translocation), with the latter subgroup further divided into EZB-MYC+ and EZB-MYC- according to MYC signature [3, 4]. There is a broad correlation between COO molecular subtypes and genetic subgroups. ABC-DLBCL largely comprises of MCD, N1 and A53, while GCB-DLBCL is primarily composed of EZB and ST2, with BN2 seen in both ABC and GCB-DLBCL. These subgroups are further underpinned by their distinct molecular mechanisms and different clinical outcomes.
Despite the steady progress in molecular characterization and sub-classification of DLBCL, few of these advances are applied in a routine clinical setting. For routine diagnosis and prognostication of DLBCL, only MYC, BCL2 and BCL6 translocations are investigated along with international prognostic index. MYC translocation occurs in 8–14% of DLBCL. This translocation can occur together with BCL2 and/or BCL6 translocation, known as double-hit (DH) or triple-hit (TH). Among MYC translocation positive DLBCL, ~9% are MYC/BCL2/BCL6-TH, ~40% and ~18% are MYC/BCL2-DH and MYC/BCL6-DH respectively [5,6,7]. Most of cases with MYC/BCL2-DH/TH are GCB subtype or EZB-MYC+ [3, 5]. In contrast, those with MYC/BCL6-DH are rather heterogeneous in their molecular subtypes, with 30% each being GCB or ABC subtype respectively, 15% due to MHG, and the remaining cases unclassifiable [5]. These cases showed a mutation profile remarkably different from those with MYC/BCL2-DH/TH, but do not exhibit any prominent signatures although a proportion of these cases are associated with NOTCH2 mutation, thus BN2 subtype [5]. For these reasons, the 5th edition of the World Health Organization Classification of Haematolymphoid Tumours (WHO-HAEM5) excludes the cases with concomitant MYC and BCL6 rearrangements (without BCL2 rearrangement) from the DH entity and renames the entity as diffuse large B-cell lymphoma/high-grade B-cell lymphoma with MYC and BCL2 rearrangements (DLBCL/HGBL-MYC/BCL2) to recognise their variable morphology [8].
The clinical outcome of DLBCL/HGBL-MYC/BCL2-DH is also heterogeneous. Cases with IG::MYC are significantly associated with worse progression-free survival (PFS) and overall survival (OS), particularly within the first two years of diagnosis, while those with non-IG::MYC showed no significant difference in both PFS and OS from DLBCL without MYC translocation [7, 9]. The molecular mechanisms underlying the different clinical impacts by MYC translocation partner are unclear. In addition, MYC protein expression varies considerably in DLBCL with MYC translocation, ranging from negative to 100% positivity in lymphoma cells [10,11,12]. In DLBCL with IGH::MYC, the breakpoint commonly occurs in region spanning the 5’UTR and intron 1 of the MYC gene and the switch region of the IGH locus respectively, thus placing the MYC gene in close proximity of the highly active IGH super enhancer, causing MYC constitutive over-expression [13]. Moreover, DLBCL with IGH::MYC often acquire MYC mutations that impair MYC protein degradation, consequently sustaining its expression and function [5]. However, the impact of non-IG partner on MYC expression is unclear. Among the known non-IG partners of MYC translocation including BCL6, ZCCHC7 and RFTN1, BCL6 is the most frequent [13, 14]. It also remains unclear how often non-IG::MYC translocation involves BCL6 as a partner, and how non-IG::MYC impacts on MYC activation given their clear difference in clinical impact from the IG::MYC translocation. To investigate these, we studied 186 cases of DLBCL with MYC translocation including 32 MYC/BCL2/BCL6-TH, 75 MYC/BCL2-DH and 26 MYC/BCL6-DH by combined analyses of MYC translocation partner and MYC protein expression, mutation profiling and breakpoint analysis of MYC translocation in selected cases to understand their transactivation potential.
Materials and methods
The study was performed in accordance with local ethical guidelines for the research use of tissue materials with the approval of the ethics committees of the involved institutions (05-Q1604-10, 04-Q1205-125, 10-H0504-79).
A total of 186 cases of DLBCL with MYC translocation were retrieved from surgical files of Addenbrookes Hospital, University of Cambridge and HMDS, St James’ University Hospital, Leeds, UK. These cases comprised of 32 MYC/BCL2/BCL6-TH, 75 MYC/BCL2-DH, 26 cases with MYC/BCL6-DH, and 53 cases MYC-single hit (SH) (Fig. 1).
Interphase fluorescence in situ hybridisation (FISH)
Chromosome translocation status at the MYC, BCL2 and BCL6 locus was available from routine haematopathological diagnosis or previous studies [5]. Further interphase FISH with MYC/BCL6 (Cytocell), MYC/IGH (Abbott), MYC/IGK and MYC/IGL (Cytocell) dual fusion probes were performed on FFPE tissue slides where indicated in the present study.
Immunohistochemistry
MYC (Abcam clone Y69) and BCL6 (Leica Clone LN22) immunohistochemistry were performed where possible in all cases where tissue materials remained available using the Bond-III system (Leica Biosystems) with the Bond Polymer Refine Detection Kit as the same condition of routine histopathological diagnosis. This was carried out centrally in the Cambridge lab and the staining intensity (weak, moderate, strong) and percentage in tumour cells (>70% or <70%) were scored [11].
DNA extraction and quality assessment
Histology was reviewed and areas containing confluent lymphoma cells (>40%) in each specimen were microdissected on consecutive tissue sections. DNA was extracted using the QIAamp DNA Micro Kit (QIAGEN, Crawly, UK), quantified with a Qubit® Fluorometer (Life Technologies, UK) and assessed for quality by PCR [5, 15].
Mutation analysis by targeted sequencing
The mutation data in 125 cases were from a previous study, in which a panel of B-cell lymphoma associated genes (n = 70) were sequenced using HaloPlexHS target enrichment and Illumina HiSeq4000 platform, with a well-validated in house variant calling pipeline [5]. In 53 cases, mutation data were similarly obtained but using TWIST capture target enrichment of a much larger gene panel (n = 191) (Table S1) [16].
LymphGen genetic subtypes were assigned where possible according to Wright et al [3].
Targeted locus capture next generation sequencing (TLC-NGS)
TLC-NGS was essentially carried out as previously described [17]. FFPE tissue sections were deparaffinised, followed by a 30 min pretreatment step at 90 °C, digestion with NlaIII restriction enzyme and ligation with T4 DNA ligase. The sample was incubated at 80°C overnight to reverse crosslinking and then subjected to DNA purification. A total of 100 ng DNA was fragmented and used for NGS library preparation, hybridization with capture probes using Roche HyperCap reagents according to the manufacturer’s instructions. Paired-end sequencing was performed using an Illumina Novaseq 6000. TLC-NGS reads were mapped to the human genome (hg19) using BWA-MEM (version: 0.7.17-r1188; settings: -SP -k12 -A2 -B3) in paired-end mode, and gene rearrangements were identified using PLIER (Proximity-Ligation based IdEntification of Rearrangements) according to previously validated pipeline [17].
Statistical analysis
Associations among MYC translocation, translocation partner and MYC protein expression were analysed using the Fisher’s exact test. All quoted P values are two-sided.
Results
BCL6 frequently involves MYC translocation in DLBCL with MYC/BCL6/BCL2-TH or MYC/BCL6-DH
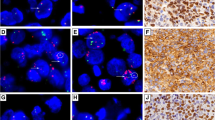
Interphase FISH with the BCL6/MYC fusion probe was performed in 54 cases of DLBCL with MYC/BCL2/BCL6-TH (n = 32) or MYC/BCL6-DH (n = 22). Among these cases, 25 (46.3%) had evidence of genomic fusion between the MYC and BCL6 loci by FISH (Fig. 2A), and the frequency of MYC/BCL6 fusion was significantly higher in the MYC/BCL2/BCL6-TH (19/32 = 59%) than the MYC/BCL6-DH (6/22 = 27%) group (Fig. 2B).
A Example of interphase FISH in a case with a triple hit (TH), in which the MYC and BCL6 translocation detected by their breakapart probes (BAP) are due to MYC/BCL6 fusion. B The frequency of MYC/BCL6 fusion is significantly higher in cases with MYC/BCL6/BCL2-”TH” than those with MYC/BCL6 “double hit” (DH).
Among the 25 cases with FISH evidence of MYC/BCL6 fusion, 24 had complete data on IG/MYC fusion by interphase FISH with the MYC/IGH fusion probe, and additional MYC/IGK(L) fusion probe if no evidence of MYC/IGH fusion. Six of these cases had an IGH::MYC fusion, and this is consistent with previous observation of a three way translocation involving the MYC, BCL6 and IGH loci by cytogenetic studies [18].
Genetic features of DLBCL with MYC/BCL6 fusion
Mutation profiling by targeted NGS was carried out in 178 cases, and 135 of these cases were successfully subtyped using the LymphGen algorithm [3].
Overall, the mutation profile of the MYC/BCL2/BCL6-TH group is very similar to that of the MYC/BCL2-DH group (Fig. 3A), characterised by frequent mutations in follicular lymphoma associated genes (BCL2, CREBBP, KMT2D, EZH2, TNFRSF14). Our previous study shows that most cases with MYC/BCL2/BCL6-TH, like those with MYC/BCL2-DH, are GCB, with a subset being MHG [5]. In support of this, the present study further demonstrated that both MYC/BCL2/BCL6-TH (22/24 = 92%) and MYC/BCL2-DH (65/67 = 97%) groups were predominantly the EZB-MYC+ genetic subtype. Within the MYC/BCL2/BCL6-TH group, there were no apparent differences in the mutation profile and LymphGen genetic subtype between MYC/BCL6 fusion positive and negative cases (Fig. 3A).
In contrast, the mutation profile of DLBCL-MYC/BCL6-DH was of less characteristic, but clearly differed from that of the MYC/BCL2/BCL6-TH or MYC/BCL2-DH group (Fig. 3A). The MYC/BCL6-DH cases vary in their COO subtype as shown in our previous study [5]. The present study further demonstrated that these cases varied in their LymphGen genetic subtypes although more frequently being the BN2 subtype or unclassifiable (Fig. 3B). Within the MYC/BCL6-DH group, there were also no apparent differences in the mutation profile and genetic subtype between MYC/BCL6 fusion positive and negative cases albeit based on few cases.
MYC protein expression is uniformly high in cases with IG::MYC but varies in those with non-IG::MYC
Given that MYC translocation is thought to dysregulate its transcription control, we compared MYC protein expression according to MYC translocation partner. High MYC expression was defined when the protein is expressed in 70% of lymphoma cells with moderate to strong staining by immunohistochemistry as such high MYC protein expression has been previously shown to identify high risk cases [11].
High MYC protein expression was seen in each of the 20 cases of DLBCL with IG::MYC translocation investigated (Fig. 4). Among DLBCL with non-IG::MYC translocation including those with MYC/BCL6 fusion, MYC expression was variable, with only up to 50% cases showing a high MYC protein expression (Fig. 4B). There was no difference in the proportion of cases with high MYC protein expression between the MYC/BCL6 fusion positive and negative groups (Fig. 4). These findings suggest that non-IG::MYC translocations may have variable effects on MYC transcription control and not every non-IG::MYC translocation can cause constitutive MYC expression.
Breakpoint analysis of MYC translocation reveal insights explaining variable MYC expression
To investigate why MYC protein expression was variable in cases with non-IG::MYC translocation, we performed TLC-NGS and breakpoint analyses in 8 cases, including 4 with non-IG::MYC (3 with MYC/BCL6 fusion) and 4 with IGH::MYC respectively. In each case, TLC-NGS investigation confirmed the findings of FISH analyses, and importantly unravelled the breakpoints and orientation of the involved genes, thus helping to understand their transcriptional potential (Table 1).
Among the three cases with MYC/BCL6 fusion, two (DLBCL-134, DLBCL-173) involved direct juxtaposition between the MYC and BCL6 loci, with the breakpoints occurring downstream or at the 3ʹUTR of the MYC gene, but upstream or within the intron 1 of the BCL6 gene (Fig. 5). In both cases, the rearranged MYC and BCL6 genes were in an opposite orientation, thus no structural changes in the 5ʹ region of MYC transcriptional control albeit uncertain on any potential effect of the super enhancers downstream of the MYC and also at the 5ʹ region of the BCL6 gene [19, 20]. In both cases, the MYC protein expression was weak in <40% lymphoma cells. In the remaining case with MYC/BCL6 fusion (DLBCL-123), an insertion of a segment of chromosome 3 sequence neighbouring to the BCL6 locus together with a segment of the IGH switch region occurred within the intron 1 of the MYC gene (Fig. 6). Although the precise breakpoints of the inserted IGH sequence could not be accurately defined, the involved region spanned the switch super enhancer, which could potentially drive MYC expression. In keeping with this, MYC protein was strongly expressed in most lymphoma cells in this case (Fig. 6).
Among the 5 cases without MYC/BCL6 fusion by FISH, TLC-NGS analyses confirmed the FISH observations in each case, and further identified their translocation partners (Table 1). Two cases showed a novel MYC translocation: one fused with TOX at 8q12 in an opposite orientation (DLBCL-136), the other fused with HNRNPA1 at 12q13 in the same orientation (DLBCL-154) (Figs. 5, 6). In both cases, the MYC breakpoint occurred either upstream (in the case with TOX) or in the intron 1 (in the case with HNRNPA1) of the MYC gene. In the case of TOX/MYC fusion, MYC transcription was unlikely driven directly by the TOX gene as the translocated TOX was in opposite orientation with MYC and loose its 5’ transcriptional regulatory region, but MYC protein expression was moderately high. Interestingly, both TLC-NGS and interphase FISH in this case showed increased copies of both the rearranged (3–6 copies by interphase FISH) and non-rearranged (2 copies by interphase FISH) MYC alleles, in keeping with the variable staining extensity among lymphoma cells (Figs. 5, S1). In the case with HNRNPA1::MYC fusion, MYC was in the same orientation with HNRNPA1, and placed under the transcription control of HNRNPA1. HNRNPA1 encodes a heterogeneous nuclear ribonucleoprotein that is ubiquitously expressed, and strong MYC protein expression was uniformly seen in lymphoma cells of this case (Fig. 6).
In the remaining three cases (L0318, DLBCL-96, DLBCL-178) without MYC/BCL6 fusion, MYC translocation was associated with IGH (Table 1).
Apart from the above novel MYC translocations, TLC-NGS also identified previously known LCP1::BCL6 (DLBCL-178, Fig. 7) and CIITA::BCL6 fusion each in one case (DLBCL-136, Fig. 5). In both cases, the genomic fusion was in the same orientation and the breakpoint was in the intron 1 of both BCL6 gene and its partner gene, and these genomic configurations are typical of BCL6 promoter substitution by its translocation which causes enhanced BCL6 expression (Figs. 5, 7). In the case with HNRNPA1::MYC fusion (DLBCL-154), TLC-NGS revealed additionally a complex fusion among BCL6, IGH and BCL2 (Fig. 6), with the IGH segment (from the joining to the switch region) in between the BCL6 and BCL2 gene on derivative chromosome 3. In this case, the presence of IGH super enhancers (at both joining and switch region) most likely drive constitutive BCL6 and BCL2 transactivation, hence the strong expression of both proteins in lymphoma cells (Fig. 6).
Discussion
The present study reports several significant novel findings, and they include: (1) MYC and BCL6 translocation in a significant proportion of DLBCL, particularly those with MYC/BCL2/BCL6-TH, are due to a direct juxtaposition between the MYC and BCL6 loci, rather than being an independent event; (2) MYC protein expression is uniformly high in DLBCL with IG::MYC, but varies in those with non-IG::MYC, including BCL6/MYC fusion; (3) MYC translocation with non-IG partner may not always acquire a genomic configuration that enables MYC constitutive transactivation, resulting in high MYC expression. These findings provide molecular insights, which explain several perplexing features of DLBCL with MYC translocation, and also bear practical implications in routine prognostic assessment.
MYC and BCL6 translocation detected by interphase FISH with their respective break-apart probes was commonly referred as independent oncogenic events, thus recorded as DH or TH when additional BCL2 translocation is present. Remarkably, 59% of the so-called MYC/BCL2/BCL6-TH and 27% of MYC/BCL6-DH DLBCL are actually due to a direct genomic fusion between the MYC and BCL6 loci. The finding is not totally unexpected as MYC is one of the many promiscuous translocation partners of BCL6, and t(3;8)(q27;q24)/BCL6::MYC and t(3;8;14)(q27;q24;q32)/IGH::BCL6/MYC have been previously reported [18, 21].
A major molecular mechanism underpinning the oncogenic potential of MYC translocation is its transactivation due to juxtaposition to a super enhancer, such as those at the IGH joining and switch region or promoter substitution. The IGH super enhancers are expected to be highly active in all mature B-cells as they express high levels of immunoglobulin. Such super-enhancer mediated transcriptional activation, unlike promoter substitution, is independent of the genomic orientation of the MYC and IG genes and to a certain extent also of the “linear” distance between the two genes [22], thus explaining the uniform high MYC protein expression seen in DLBCL with IG::MYC, and also Burkitt lymphoma.
Among the 4 cases of DLBCL with non-IG::MYC investigated by TLC-NGS, 3 showed MYC gene in an opposite orientation with its translocation partner (BCL6, TOX), without affecting the MYC promoter region. The moderate variable MYC expression in the case with TOX::MYC (DLBCL-136) is most likely the result of MYC gene amplification (Fig. 5, Fig. S1). Otherwise, there was no evidence of constitutive MYC expression in these cases. There were potential super enhancers downstream of the MYC gene and in the translocated BCL6 region [19, 20], the potential impact on these super enhancers by these translocations is unclear. As the transactivation potential of super enhancers depend on cell type and differentiation stage and is regulated by a range of factors, such as genetic/epigenetic modifications and transcriptional factor binding [20, 23, 24], different translocations may give rise to variable potentials of MYC transactivation, from low levels of dysregulation to utmost constitutive activation. Nonetheless, lack of high MYC expression in these cases suggests these translocations do not cause MYC constitutive transactivation. This speculation is in keeping with the previous observation that a proportion of DLBCL with MYC translocation lack high MYC mRNA and protein expression [11, 12]. In contrast, the remaining case (DLBCL-154) with HNRNPA1::MYC is a typical promoter substitution, and shows strong uniform MYC expression as expected since HNRNPA1, encoding for an RNA binding protein, is ubiquitously expressed (Fig. 6).
The above findings potentially explain why IGH::MYC, but not non-IG::MYC confers significantly inferior survival in patients with DLBCL-MYC/BCL2-DH [7], and also why ~25% of DLBCL with MYC translocation, including those with a MYC/BCL2-DH, are conventional GCB, but not MHG subtype [1]. Our observations also highlight the heterogeneous MYC expression in DLBCL with non-IG::MYC translocation. Of note, 44% of DLBCL with non-IG::MYC/BCL2-DH lacked high MYC protein expression above 70% (Fig. 3A). It remains to be investigated whether there is any potential difference in clinical outcome between non-IG::MYC translocation positive DLBCL with high and low MYC protein expression, and whether those with high MYC expression are similar to cases with IG::MYC in their clinical outcome. To address this pivotal question, a large cohort of genetic subtype matched DLBCL with MYC translocation, such as those with MYC/BCL2-DH, is required.
In DLBCL, MYC and BCL6 translocation are most likely acquired due to relentless exposure to somatic hypermutation and class switch activities during B-cell expansion in germinal centres, and are likely a secondary event [13, 25]. This is particularly evident in cases with BCL2 translocation, which is the primary genetic event, occurring as a consequence of erroneous VDJ recombination at the pre-B stage of B-cell development in the bone marrow. The secondary structural changes may not be always a driver event, similar to the point mutations in many well-known lymphoma genes acquired due to somatic hypermutation activities [26]. In view of this and the above discussion, it is pertinent to question whether every non-IG::MYC translocation in DLBCL is an activation event, albeit to be attested in future studies.
In routine clinical practice, interphase FISH is used for detection of MYC, BCL2 and BCL6 translocation, together with their translocation partners, although commonly only including IGH. Among MYC translocation positive DLBCL, IG::MYC accounts for ~55% of cases [7, 9]. The full spectrum of non-IG partners of MYC translocation remains to be characterised although BCL6 may account for a majority. A major challenge to delineate whether a non-IG/MYC translocation is a constitutive activation event, thus clinically important, is to characterise its genomic configuration, search for evidence that enables MYC constitutive transactivation. This cannot be resolved by interphase FISH even when the translocation partner is known, but requires breakpoint analyses such as by TLC-NGS which is not yet available in a routine clinical setting. In the absence of any knowledge of genomic configuration of the translocation, the pathogenic potential and the prognostic value of non-IG/MYC translocation need to be interpreted in conjunction with MYC protein expression.
Our findings also raise the debate whether all DLBCL should be investigated for MYC translocation with regard to risk stratification in routine histopathological diagnosis by interphase FISH or first screened by MYC immunohistochemistry (where necessary immunohistochemistry with an alternative antibody to rule out potential false negative due to mutation impairing the antibody binding site [12]), and only cases with MYC protein expression above a certain level (to be determined) selected for further FISH analyses. Further breakpoint analysis of non-IG/MYC translocation and their correlation with the level of MYC protein expression in a large cohort should help to resolve these practical issues. Nonetheless, it is important to routinely investigate whether MYC translocation is associated with IG (both heavy and light chain) loci and MYC protein expression as both have been shown to be associated with adverse clinical outcome.
In summary, a significant proportion of DLBCL with both MYC and BCL6 translocations are due to direct juxtaposition between the two genomic loci. MYC translocation involving non-IG loci including BCL6 varies in their genomic configurations, and may not often gain genomic configuration that can cause constitute MYC transactivation, leading to its enhanced protein expression. The prognostic value of MYC translocation needs to be interpreted in conjunction with its translocation partner and MYC protein expression level.
Data availability
All core data generated or analysed during this study are included in this published article, and additional raw data are available from the corresponding author on reasonable request.
References
Sha C, Barrans S, Cucco F, Bentley MA, Care MA, Cummin T, et al. Molecular high-grade B-cell lymphoma: defining a poor-risk group that requires different approaches to therapy. J Clin Oncol. 2019;37:202–12.
Ennishi D, Jiang A, Boyle M, Collinge B, Grande BM, Ben-Neriah S, et al. Double-hit gene expression signature defines a distinct subgroup of germinal center B-cell-like diffuse large B-cell lymphoma. J Clin Oncol. 2019;37:190–201.
Wright GW, Huang DW, Phelan JD, Coulibaly ZA, Roulland S, Young RM, et al. A probabilistic classification tool for genetic subtypes of diffuse large B cell lymphoma with therapeutic implications. Cancer Cell. 2020;37:551–68.e14.
Lacy SE, Barrans SL, Beer PA, Painter D, Smith AG, Roman E, et al. Targeted sequencing in DLBCL, molecular subtypes, and outcomes: a Haematological Malignancy Research Network report. Blood. 2020;135:1759–71.
Cucco F, Barrans S, Sha C, Clipson A, Crouch S, Dobson R, et al. Distinct genetic changes reveal evolutionary history and heterogeneous molecular grade of DLBCL with MYC/BCL2 double-hit. Leukemia. 2020;34:1329–41.
Clipson A, Barrans S, Zeng N, Crouch S, Grigoropoulos NF, Liu H, et al. The prognosis of MYC translocation positive diffuse large B-cell lymphoma depends on the second hit. J Pathol Clin Res. 2015;1:125–33.
Rosenwald A, Bens S, Advani R, Barrans S, Copie-Bergman C, Elsensohn MH, et al. Prognostic significance of MYC rearrangement and translocation partner in diffuse large B-cell lymphoma: a study by the Lunenburg lymphoma biomarker consortium. J Clin Oncol. 2019;37:3359–68.
Alaggio R, Amador C, Anagnostopoulos I, Attygalle AD, Araujo IBO, Berti E, et al. The 5th edition of the World Health Organization Classification of Haematolymphoid Tumours: Lymphoid Neoplasms. Leukemia. 2022;36:1720–48.
Copie-Bergman C, Cuillière-Dartigues P, Baia M, Briere J, Delarue R, Canioni D, et al. MYC-IG rearrangements are negative predictors of survival in DLBCL patients treated with immunochemotherapy: a GELA/LYSA study. Blood. 2015;126:2466–74.
Ambrosio MR, Lazzi S, Bello GL, Santi R, Porro LD, de Santi MM, et al. MYC protein expression scoring and its impact on the prognosis of aggressive B-cell lymphoma patients. Haematologica. 2019;104:e25–e8.
Ziepert M, Lazzi S, Santi R, Vergoni F, Granai M, Mancini V, et al. A 70% cut-off for MYC protein expression in diffuse large B cell lymphoma identifies a high-risk group of patients. Haematologica. 2020;105:2667–70.
Collinge B, Ben-Neriah S, Chong L, Boyle M, Jiang A, Miyata-Takata T, et al. The impact of MYC and BCL2 structural variants in tumors of DLBCL morphology and mechanisms of false-negative MYC IHC. Blood 2021;137:2196–208.
Chong LC, Ben-Neriah S, Slack GW, Freeman C, Ennishi D, Mottok A, et al. High-resolution architecture and partner genes of MYC rearrangements in lymphoma with DLBCL morphology. Blood Adv. 2018;2:2755–65.
Bertrand P, Bastard C, Maingonnat C, Jardin F, Maisonneuve C, Courel MN, et al. Mapping of MYC breakpoints in 8q24 rearrangements involving non-immunoglobulin partners in B-cell lymphomas. Leukemia. 2007;21:515–23.
Wang M, Escudero-Ibarz L, Moody S, Zeng N, Clipson A, Huang Y, et al. Somatic mutation screening using archival formalin-fixed, paraffin-embedded tissues by fluidigm multiplex PCR and illumina sequencing. J Mol Diagn. 2015;17:521–32.
Tzioni MM, Wotherspoon A, Chen Z, Cucco F, Makker J, Du MQ. Divergent evolution of metachronous follicular lymphoma and extranodal marginal zone lymphoma of mucosa-associated lymphoid tissue from a common precursor. J Pathol. 2023;261:11–8.
Allahyar A, Pieterse M, Swennenhuis J, Los-de Vries GT, Yilmaz M, Leguit R, et al. Robust detection of translocations in lymphoma FFPE samples using targeted locus capture-based sequencing. Nat Commun. 2021;12:3361.
De Paoli E, Bandiera L, Ravano E, Cesana C, Grillo G, Mancini V, et al. A double-hit High-grade B-cell lymphoma with three-way translocation t(3;8;14)(q27;q24;q32) involving BCL6, MYC, and IGH. Clin Case Rep. 2018;6:2411–5.
Bahr C, von Paleske L, Uslu VV, Remeseiro S, Takayama N, Ng SW, et al. A Myc enhancer cluster regulates normal and leukaemic haematopoietic stem cell hierarchies. Nature. 2018;553:515–20.
Bal E, Kumar R, Hadigol M, Holmes AB, Hilton LK, Loh JW, et al. Super-enhancer hypermutation alters oncogene expression in B cell lymphoma. Nature. 2022;607:808–15.
Johnson SM, Umakanthan JM, Yuan J, Fedoriw Y, Bociek RG, Kaiser-Rogers K, et al. Lymphomas with pseudo-double-hit BCL6-MYC translocations due to t(3;8)(q27;q24) are associated with a germinal center immunophenotype, extranodal involvement, and frequent BCL2 translocations. Hum Pathol. 2018;80:192–200.
Pinaud E, Marquet M, Fiancette R, Péron S, Vincent-Fabert C, Denizot Y, et al. The IgH locus 3’ regulatory region: pulling the strings from behind. Adv Immunol 2011;110:27–70.
Ryan RJ, Drier Y, Whitton H, Cotton MJ, Kaur J, Issner R, et al. Detection of enhancer-associated rearrangements reveals mechanisms of oncogene dysregulation in B-cell Lymphoma. Cancer Discov. 2015;5:1058–71.
Iyer AR, Gurumurthy A, Kodgule R, Aguilar AR, Saari T, Ramzan A et al. Selective enhancer dependencies in MYC -intact and MYC -rearranged germinal center B-cell diffuse large B-cell lymphoma. bioRxiv. 2023. https://doi.org/10.1101/2023.05.02.538892.
Dobson R, Wotherspoon A, Liu SA, Cucco F, Chen Z, Tang Y, et al. Widespread in situ follicular neoplasia in patients who subsequently developed follicular lymphoma. J Pathol. 2022;256:369–77.
Hübschmann D, Kleinheinz K, Wagener R, Bernhart SH, López C, Toprak UH, et al. Mutational mechanisms shaping the coding and noncoding genome of germinal center derived B-cell lymphomas. Leukemia. 2021;35:2002–16.
Acknowledgements
We would like to thank Wanfeng Zhao for her help on immunohistochemistry.
Funding
The research was supported by grants from Blood Cancer UK (19010, 19011) UK and Cancer Research UK (C8333/A29707), MMT was supported by a BBSRC DTP PhD studentship (BBSRC BB/M011194/1). The Human Research Tissue Bank is supported by the NIHR Cambridge Biomedical Research Centre.
Author information
Authors and Affiliations
Contributions
FISH, targeted NGS, data collection and analyses: CZ, SB, FC, DJ, MMT, ZC, YL, JM, MQD; TLC-NGS: ES, JFS, HF, MQD; FISH and pathology: LRB, HL, HED, ES, MQD; Clinical data: NS, SKN, MK, MPP; Study design, case contribution, coordination and research funding: MQD, RT, DRW, AJD, CB, PWMJ; Manuscript writing and preparation: MQD with contributions from all authors. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
ES, JFS and HF are employees of Cergentis, which owns patents on the TLC-NGS method. The authors declare no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, C., Stelloo, E., Barrans, S. et al. Non-IG::MYC in diffuse large B-cell lymphoma confers variable genomic configurations and MYC transactivation potential. Leukemia 38, 621–629 (2024). https://doi.org/10.1038/s41375-023-02134-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41375-023-02134-1
- Springer Nature Limited
This article is cited by
-
Mature B, T and NK-cell, plasma cell and histiocytic/dendritic cell neoplasms: classification according to the World Health Organization and International Consensus Classification
Journal of Hematology & Oncology (2024)
-
What is new in the 5th edition of the World Health Organization classification of mature B and T/NK cell tumors and stromal neoplasms?
Journal of Hematopathology (2024)
-
Cytogenetic and pathologic characterization of MYC-rearranged B-cell lymphomas in pediatric and young adult patients
Journal of Hematopathology (2024)