Abstract
Parkinson’s disease (PD) is the second most common neurodegenerative disease worldwide, and its treatment remains a big challenge. The pathogenesis of PD may be related to environmental and genetic factors, and exposure to toxins and gene mutations may be the beginning of brain lesions. The identified mechanisms of PD include α-synuclein aggregation, oxidative stress, ferroptosis, mitochondrial dysfunction, neuroinflammation, and gut dysbiosis. The interactions among these molecular mechanisms complicate the pathogenesis of PD and pose great challenges to drug development. At the same time, the diagnosis and detection of PD are also one of obstacles to the treatment of PD due to its long latency and complex mechanism. Most conventional therapeutic interventions for PD possess limited effects and have serious side effects, heightening the need to develop novel treatments for this disease. In this review, we systematically summarized the pathogenesis, especially the molecular mechanisms of PD, the classical research models, clinical diagnostic criteria, and the reported drug therapy strategies, as well as the newly reported drug candidates in clinical trials. We also shed light on the components derived from medicinal plants that are newly identified for their effects in PD treatment, with the expectation to provide the summary and outlook for developing the next generation of drugs and preparations for PD therapy.
Similar content being viewed by others
Introduction
PD is the second most common neurodegenerative disease worldwide, with global prevalence increasing by 74.3% between 1990 and 2016.1 In 1817, James Parkinson published his monograph titled An Essay on the Shaking Palsy which represents the first description of PD as a neurological disorder.2 Beginning with Jean-Martin Charcot, a succession of scientists contributed to the comprehensive description of the clinical range and anatomopathological basis of PD, including motor, non-motor symptoms, the neuropathological changes in the substantia nigra (SN), Lewy bodies, and the role of dopamine (DA).3,4 Following these discoveries (Fig. 1), highly efficacious therapies like pharmacological DA substitution (levodopa treatment) and deep brain stimulation have become available to effectively control the symptoms. However, none of these treatments can stop PD from being a progressive disorder especially the increasing severity of treatment-resistant motor and non-motor symptoms still carry PD patient’s painful life.5 In this review, we described the clinical features and diagnostic criteria of PD, summarized the molecular mechanisms underlying PD and the research models, we also listed drugs used in market and clinical practice, and discussed available PD treatments like natural compounds, with the expectation to provide the summary and outlook for developing the next generation of drugs and treatments for PD therapy.
Basic research and drug development history for PD disease and therapy. A2a adenosine receptor type 2a, mGlu metabotropic glutamate receptor, NAM negative allosteric modulator, PAM positive allosteric modulator, EDN embryonic dopamine neuron, PDDPC personalized iPSC-derived dopamine progenitor cell, iPSC induced pluripotent stem cell, DBS deep brain stimulation
Epidemiology, clinical features, and diagnostic criteria
The incidence of PD increases after 60 years of age, rising sharply to more than 3% among individuals of over 80 years old.6,7,8,9 In most populations, men have higher PD incidence than women.8,10,11 Variation in living habits and environment likely explains the difference of prevalence across regions and races. Environmental toxins may trigger PD symptoms, while dietary habits may alter disease incidence, notable examples include greater risk in smokers and people who regularly consume caffeine.12,13,14
Motor symptoms and non-motor symptoms make up the majority of the clinical characteristics of PD. Patients with PD have a variety of motor symptoms, including bradykinesia, muscle stiffness, rest tremor, and postural and gait difficulties.15,16 There are two main kinds of PD: tremor-dominant PD and non-tremor-dominant PD, based on clinical findings. In comparison to non-tremor-dominant PD, tremor-dominant PD is frequently linked to a slower pace of development and less functional impairment.17 Olfactory dysfunction, cognitive decline, constipation, depression, sleep problems such as excessive daytime drowsiness and rapid eye movement sleep behavior disorder, autonomic dysfunction, pain, and exhaustion are examples of non-motor characteristics. Non-motor symptoms frequently appear in the early stages of PD before the motor symptoms.18,19 PD progression will have certain problems, such as dyskinesia, psychosis, and motor and non-motor fluctuations, as well as a deterioration of the motor characteristics and long-term symptomatic therapy.20 According to reports, up to 80% of PD patients have freezing of the gait and falls after roughly 17 years of the disease, and up to 50% of patients say they have experienced choking. Dementia is also said to be more common in individuals who have had PD for at least 20 years.20 The primary pathogenic characteristics of PD include the steady degradation of just a subset of neurons within particular brain areas, such as the SN, as the illness progresses. Dopaminergic neurons are lost exclusively in the ventrolateral SN in the early stages; however, this damage spreads in the late stages.21,22,23,24 Furthermore, in several different areas of the brain, certain neurons have abnormally high levels of α-synuclein accumulated in their cytoplasm.25 In cholinergic and monoaminergic brainstem neurons as well as olfactory neurons, aggregated α-synuclein produces Lewy bodies, a frequent hallmark of neuropathology. Lewy bodies multiply as Parkinson’s disease advances, impacting non-dopaminergic neurons in other brain regions in addition to the limbic and neocortical regions.26,27 Finally, neurons outside of the central nervous system (CNS), such those in the olfactory bulb or mesenteric system, deteriorate as a result of PD. Overall, 10% of identified patients were initially categorized as other illnesses due to the possibility that PD symptoms might manifest early and the complexity of the disease presentation.28 In order to increase diagnostic precision, the International Parkinson and Movement Disorder Society has established criteria. According to these recommendations, the presence of bradykinesia and at least one other cardinal motor characteristic constitutes Parkinson’s syndrome (4–6-Hz rest tremor or limb rigidity). Additionally included were “red flags” for alternate diagnosis and excluding clinical characteristics. DA transporter-single-photon emission computed tomography (DAT-SPECT), structural magnetic resonance imaging (MRI), magnetic resonance diffusion-weighted imaging (MR-DWI), and genetic testing are frequently utilized to make a clinical diagnosis of PD. Because hyposmia or anosmia are present in around 90% of individuals with PD, olfactory function testing with the UPSIT or Sniffin Stick tests is occasionally a component of the initial clinical examination.29,30,31 With the use of cutting-edge MRI methods, certain MRI characteristics that are highly specific for atypical parkinsonism have been identified. These include quantitative susceptibility mapping (QSM), which allows for the determination of iron accumulation in the SN, and neuromelanin imaging (NMI), which takes use of the paramagnetic characteristics of neuromelanin.32,33 Notably, NMI has the potential to show alterations in prodromal PD.34 A further technique used to distinguish between PD and clinical mimics unrelated to presynaptic nigrostriatal terminal dysfunction is 123I-ioflupane single-photon emission CT (SPECT).35,36 Despite these developments, one area of clinical diagnosis still needs work: the use of genetic testing, which is presently reserved for situations in which a particular hereditary etiology is suspected. However, we also notice a rise in the number of genes linked to complex symptoms that include parkinsonism as PD worsens. For such circumstances, routine genetic testing may be helpful.
Etiology and pathogenesis of PD
Environment genetic factors
The complex etiology of PD involves both environmental and genetic factors.37,38 Environmental causes are situations such as pesticide exposure, physical inactivity, head injury, and stress.39 People intoxicated with 1-methyl-4-phenyl-1,2,3,6-tetrahydrodropyridine (MPTP) developed a syndrome nearly identical to PD and its active metabolite, 1-methyl-4-phenylpyridinium (MPP+), is similar to paraquat in structure.40 Human epidemiological studies have implicated residence in a rural environment and related exposure to herbicides and pesticides with an elevated risk of PD.41 However, it is still necessary to get convincing data to implicate the link between specific toxins and PD.
Autosomal-dominant PD genes
It has been demonstrated in patients with SNCA mutations whose brains showed the aggregation of α-synuclein, represented as the occurrence of Lewy bodies and the loss of DA neurons, that the PARK1/PARK4 gene for the expression of α-synuclein is related to the abnormal pathological aggregation of insoluble α-synuclein fibril.42,43 The most frequent genetic cause of PD is a mutation in the leucine-rich repeat kinase 2 gene (LRRK2), known as PARK8. LRRK2 mutations have primarily been found in late-onset individuals older than 50 years.44,45 The most common variations of this mutation are G2019S, R1441C, R1441G, and R1441H, which can cause DA neurons to die and degenerate by interfering with a variety of physiological processes, including vesicle transport, cytoskeletal function, protein synthesis, and the lysosomal system.45,46 It has been discovered that PARK13, the HTRA2 serine peptidase 2 gene (HTRA2), is released into the cytoplasm from damaged mitochondria and is crucial to maintaining normal mitochondrial function. It has also been demonstrated to play a neuroprotective role under stressful conditions, with PARK13 knockout mice exhibiting elevated levels of reactive oxygen species (ROS), mitochondrial dysfunction, and PD phenotypes.47,48,49 Its activity is controlled by PINK1-mediated phosphorylation, which is crucial for maintaining mitochondrial integrity under stress. PARK13 targets the destruction of misfolded SNCA.50,51 PARK17, a gene that makes up the reverse transcriptome complex (VPS35), is required for the retrotransfer of proteins from endosomes in the pre-lysosomal compartment network to the trans-Golgi network.52 The cation-independent mannose 6-phosphate receptor (CI-MPR) may bind with VPS35 in an endosomal compartment and get sequestered in recycling tubules, preventing it from being sent to vacuoles or lysosomes.53 In response to diverse conditions, the PARK18 gene, which encodes the eukaryotic translation initiation factor 4 gamma 1 (EIF4G1), controls the commencement of the translation of mRNAs encoding mitochondrial, cell survival, and growth proteins.54,55 The eIF4G1-eIF4E or eIF4G1-eIF3e binding, which is assumed to serve as the molecular bridge between the mRNA cap-binding complex and the 40S subunit and causes mitochondria-related imbalance, has been discovered to be affected by two mutations, EIF4G1 p.A502V, and EIF4G1 p.R1205H.54,56
Autosomal-recessive PD genes
The most frequent cause of autosomal-recessive early-onset Parkinson’s syndrome is PARK2, which is encoded by the parkin RBR E3 ubiquitin protein ligase gene (PRKN). According to studies, the PARK2 mutation is present in up to 7% of PD patients between 30 and 35 years old and as much as 50% of PD cases over 25 years old.57,58 In the ubiquitin–proteasome system, which is thought to be a multipurpose neuroprotective agent against a variety of toxic injuries, including mitochondrial poisons, and is thought to be essential for the survival of DA neurons, Parkin plays a significant role as an E3 ubiquitin ligase, working in conjunction with E1 ubiquitin-activating enzyme and E2 ubiquitin-conjugating enzyme to degrade targeted proteins.59 It has been demonstrated that PARK6, a serine/threonine protein kinase encoded by the PTEN-induced putative kinase 1 gene (PINK1), interacts with parkin to encourage selective autophagy in depolarized mitochondria and maintain mitochondrial integrity.60 Parkin is typically recruited to depolarized mitochondria to start autophagy and eliminate the damaged or malfunctioning mitochondria after being phosphorylated by PINK1 to activate its E3 ligase activity.37 In inefficient mitochondria, PINK1 builds up in the outer mitochondrial membrane to start the removal of damaged mitochondria from the cell because it cannot be transmitted to the inner mitochondrial membrane to be broken down.61 PARK7, also known as DJ-1, the parkinsonism-associated deglycase gene, guards DA neurons in the model system from harm brought on by mutant synuclein, rotenone, 6-hydroxydopamine (6-OHDA), and hydrogen peroxide.62 The primary function of the widely expressed protein DJ-1 is to protect cells against oxidative stress through a variety of ways.61,63 According to reports, PARK2, PARK6, and PARK7 are all involved in the same biological process. Transmembrane endo-/lysosomal related proteins are encoded by PARK9, the ATPase 13A2 gene (ATP13A2). The lysosomal signaling lipids phosphatidylic acid and phosphatidylinositol (3,5) biphosphate interact with the N-terminus of ATP13A2, which serves as the catalyst for ATP13A2 action and controls endo/lysosomal cargo sorting.64,65 The majority of ATP13A2 mutations affect its functional domains, particularly its transmembrane and E1-E2 adenosine triphosphatase domains. If ATP13A2 is functionally lost, this may lead to Zn2+ dysregulation and abnormal cell metabolism, including dysfunctional energy production and decreased lysosomal proteolysis.66,67,68 In addition, it has been demonstrated that ATP13A2 reduces the neurotoxicity of α-synuclein.69 The PARK15 protein, encoded by the F-box protein 7 gene (FBXO7), is a subunit of the F-box protein that functions as an adapter protein in the SKP1/cullin-1/F-box protein E3 ubiquitin ligase complex to recognize and mediate the non-degrading ubiquitination of glycogen synthase kinase (GSK)-3β and the translocase of outer mitochondrial membrane 20 to control mitophagy, mitochondria motility, mitochondria membrane potential, mitochondria bioenergetics, mitochondria biogenesis and mitochondria-associated apoptosis.70,71,72,73,74 In addition, because FBXO7 is a stress-responsive protein, malfunction may lower complex-I’s activity in the electron transport chain, lowering mitochondrial membrane potential and ATP levels while raising cytoplasmic ROS.75
Molecular mechanisms of PD
α-synuclein aggregation
Neuronal degeneration has been linked to numerous molecular and cellular changes, including α-synuclein aggregation, aberrant protein handling, excitotoxicity, oxidative stress, apoptosis, and mitochondrial dysfunction. Abnormal α-synuclein aggregation is one of the most important hypotheses explaining the death of nigrostriatal neurons in PD.76 Localized to the cytosol, mitochondria, and nucleus, α-synuclein is a potential chaperone that plays a role in the dynamics of synaptic vesicles, intracellular trafficking, and mitochondrial function.77,78,79 Some evidence suggests that the protein participates in lipid metabolism of the brain, a process that contributes to PD pathogenesis.80 α-synuclein itself can become neurotoxic when soluble α-synuclein monomers form oligomers, which combine into tiny protofibrils and eventually form large, insoluble fibrils.81,82 Age-related decline in proteolytic defense mechanisms of the brain may play an important role in α-synuclein accumulation.83,84 Specifically, intracellular α-synuclein homeostasis is maintained by the ubiquitin–proteasome and lysosomal autophagy systems. Extracellular proteases not part of either system are also implicated cleaving α-synuclein. Thus, impairment of these degradation systems may contribute to α-synuclein accumulation.
Oxidative stress
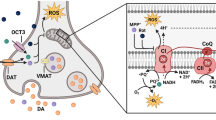
Oxidative stress (OS) is a major process in aging that directly harms the CNS. Under physiological conditions, free radicals or ROS are important to host defense, gene transcription, synaptic plasticity regulation, and apoptosis.85 However, OS occurs when ROS overwhelms cellular antioxidant activity. Cytotoxic compounds then accumulate to cause protein collapse, enzyme failure, lipid breakdown,86 and cell death in various neurons, including DA-neuronal tissue (Fig. 2).87 These dysfunctions contribute to PD pathogenesis, and may also be a cause of Alzheimer’s disease (AD).88,89 Currently, NADPH oxidase (NOX) is considered the most important ROS generator,90 playing a crucial role in triggering OS and neurotoxicity.91 Mitochondria are also major producers of ROS.92,93 The electron transport chain’s complexes I and III are thought to be where most ROS is produced in mitochondria. Superoxide radical, the main ROS generated in mitochondria, was created when the one electron was transported from oxygen to oxygen. Superoxide dismutase 2 or MnSOD may convert the superoxide radical to hydrogen peroxide, which the catalase enzyme can subsequently detoxify. However, the Fenton reaction, which severely oxidizes DNA or lipids, can cause hydrogen peroxide to transform into a highly reactive hydroxyl radical in the presence of metal ions like Fe2+.93,94 The mechanism of ferroptosis is connected to the imbalance of iron ion homeostasis, implying a connection between ferroptosis and OS.95 Lipids may be oxidized by the Fenton reaction’s hydroxyl radicals to produce lipid peroxides, which can cause ferroptotic cell death.96,97 Depletion of glutathione, which worsens intracellular OS by promoting the buildup of lipid peroxides to trigger ferroptosis, is another biochemical sign of ferroptosis. In addition, increased OS can decrease lysosomes and harm the lysosomal autophagy system, connecting OS to the buildup of α-synuclein. Another hypothesis contends that extra cytosolic DA can simply be oxidized to create DA-quinones. Then, the DA quinine-modified α-synuclein may partially inhibit chaperone-mediated autophagy, causing α-synuclein to self-assemble.98,99 Meanwhile, the aggregate formation of intracellular α-synuclein increased mitochondrial OS.100
Intracellular α-synuclein homeostasis is maintained via the ubiquitin–proteasome and lysosomal autophagy systems. Impairment of these degradation systems by OS, mitochondrial dysfunction, or neuroinflammation could contribute to α-synuclein accumulation. Furthermore, mutations of genes like LRRK2, DJ-1, Parkin, and Pink1 cause mitochondrial dysfunction and increase cell death. Finally, OS and neuroinflammation appear to be connected
Ferroptosis
An aberrant iron metabolism and severe lipid peroxidation trigger ferroptosis, an iron-dependent kind of controlled cell death that results in OS and cell death (Fig. 3).101 It was also found to be involved in DA neuron death in PD.102 The enzyme acyl-CoA synthetase long-chain family member 4 (ACSL4) converts coenzyme A (CoA) to free polyunsaturated fatty acids (PUFA) in the cytosol. PUFA-CoA can then be incorporated into phospholipids, which are then oxidized by lipoxygenases 12/15 and cause lipid peroxidation. By halting lipid peroxidation, glutathione (GSH), an antioxidant that the body produces from glutamate and cysteine, can suppress ferroptosis.103,104 The rate-limiting substrate, cysteine, can either be produced from methionine via the transsulfuration route or taken up by the xCT antiporter as an oxidized cystine dimer. The xCT antiporter is inhibited by erastin, depleting the intracellular cysteine pool and impairing GSH production as a result. Another cellular antioxidant enzyme called DJ-1 prevents the transsulfuration pathway from being destroyed, protecting the production of cysteine and GSH and acting as a ferroptosis inhibitor.105 The only member of the glutathione peroxidase family capable of reducing lipid hydroperoxides under physiological circumstances is glutathione peroxidase 4 (GPX4).106,107 To convert lipid hydroperoxides to lipid alcohols, GPX4 requires decreased GSH, and one of the most popular methods to induce ferroptosis experimentally is the direct inactivation of GPX4 by RAS-selective lethal 3. The traits of ferroptosis induction are remarkably compatible with the pathogenic alterations seen in PD patients, and ferroptosis genes themselves can be connected to PD. Coexistence of iron and α-synuclein in Lewy bodies in the midbrain in PD patients.108 α-synuclein as a metal-binding protein will change the conformation while binding iron leading to the aggregation of α-synuclein.109 Iron homeostasis in neural networks is regulated by microglia and astrocytes. As a result of iron accumulation in activated microglia and subsequent production of proinflammatory cytokines, iron deposition in the CNS may rise. Divalent metal transporter 1 (DMT1), iron regulatory protein 1, and transferrin receptor 1 (TfR1) considerably elevated expression, but ferroportin 1 (FPN1) dramatically downregulated expression, which aggravated neuronal iron deposition.110,111,112,113,114,115 In addition to producing ROS, NOX in active microglia led to OS that caused DA neurons to undergo ferroptosis.116 Furthermore, inducible nitric oxide synthase (iNOS) was markedly elevated in microglia in response to inflammatory signals, which allowed it to inhibit 15-lipoxygenase activity and thwart ferroptosis.117 Various forms of iron are transported by astrocytes mostly through protein interaction cascades, particularly ceruloplasmin (CP). Although CP’s ferroxidase activity may efficiently oxidize Fe2+ into Fe3+, enabling iron efflux from cells, almost 80% of this activity was reduced in the SN of PD patients, suggesting that decreased CP expression and consequent iron buildup play a role in neuronal mortality in PD.118,119,120,121 Iron buildup in the brain that is neurotoxic will also be aided by microglia and astrocytes overexpressing heme oxygenase-1.122,123 To remove ROS from DA neurons, reactive astrocytes may also produce different antioxidant molecules like GSH and metallothioneins. Nrf2 activation in astrocytes can also upregulate antioxidant enzymes including GSH production enzymes and MTs to protect the DA neurons from OS.124
First, inflammatory cytokines (IL-1β, TNF-α, IL-6) released by activated microglia and astrocytes promote iron accumulation in neurons by upregulating DMT1 and downregulating FPN1. BDNF and GDNF secreted by activated astrocytes reduce iron accumulation in neurons by downregulating DMT1. Second, ROS released from activated microglia promote neuronal OS. Upregulation of Nrf2 and the release of metallothioneins in astrocytes contribute to neuronal resistance to OS. BDNF brain-derived neurotrophic factor, GDNF glial cell line-derived neurotrophic factor, HO-1 heme oxygenase-1, IL-1β interleukin-1β, IL-6 interleukin 6, iNOS inducible nitric oxide synthase, NOX NADPH oxidase, Nrf2 nuclear factor-erythroid factor-2, Tf transferrin, TNF-α tumor necrosis factor α, 12/15-LOX lipoxygenases 12/15, LOOH-PL lipid hydroperoxide–phospholipid, LOH-PL lipid alcohol–phospholipid
Mitochondrial dysfunction
Mitochondrial dysfunction is increasingly understood to be important in PD pathogenesis. Indeed, many studies have found that mitochondrial dysfunction induces dopaminergic neurodegeneration and chronic ROS production. The first indication of this connection arose from observations after infusions of MPTP selectively inhibited mitochondrial complex I.40,125 The same negative outcomes occurred with other inhibitors of this complex, including rotenone, pyridaben, fenpyroximate, and trichloroethylene.126,127,128 In addition, mice overexpressing α-synuclein were more susceptible to toxins than α-synuclein-knockout mice, suggesting that mitochondrial α-synuclein worsens toxicity.129,130,131 Transcription-factor dysregulation and the resultant changes to mitochondrial biogenesis are hypothesized to be major causes of mitochondrial dysfunction. In particular, transcription-factor coactivator peroxisome proliferator-activated receptor gamma coactivator-1a (PGC-1a) is a key regulator of mitochondrial biogenesis. In PGC-1a knockout mice, dopaminergic cells are more sensitive to MPTP, whereas PGC-1a overexpression protects against neurotoxicity.132,133 Another source of mitochondrial dysfunction is pathogenic mutations in certain genes, such as Parkin, DJ-1, LRRK2, and PINK1.126,131,134,135 Parkin encodes E3, a ubiquitin protease ligase. Animals lacking Parkin are highly susceptible to rotenone, a mitochondrial complex-I inhibitor.131,135,136 Mutations to PINK1 cause an autosomal-recessive form of PD, likely through decreasing mitochondria respiration and ATP synthesis, while increasing α-synuclein aggregation.137 PINK1 dysfunction also appears to cause defects in mitochondria localization and impairs mitophagy.138 Research on combined Parkin and PINK1 knockouts in Drosophila showed that they belong in the same pathway, with PINK1 being upstream to Parkin.59 When mitochondria are damaged and depolarized, the cytosol recruits Parkin to mediate selective autophagic removal.139 For Parkin translocation, dysfunctional mitochondria must accumulate PINK1 and activate kinases.140,141,142,143 Research suggests that Src homology 2 domain-containing tyrosine phosphatase-2 (SHP2) is important to mitochondrial translocation and ubiquitination of Parkin, given that SHP2 knockdown inhibits the process (Sun et al. 144). Tyr dephosphorylation may be the mechanism underlying SHP2 regulation of Parkin activity. The drug lovastatin enhances SHP2 activity and thus is a candidate for PD treatment. Loss-of-function mutations in the DJ-1 locus also cause a rare autosomal-recessive form of PD and increase susceptibility to OS-induced cell death. Both DJ-1 knockout mice and humans carrying DJ-1 mutations have mitochondria with impaired respiration.145,146,147 In contrast, autosomal-dominant PD is associated with mutations in LRRK2. The striatum of older homozygous LRRK2G2019S knock-in mice exhibited mitochondrial abnormalities, as did the DA neurons of Caenorhabditis elegans harboring G2019SLRRK2 mutations.148,149 In general, mitochondrial fission is associated with LRRK2 mutations mediated by dynamin-like proteins.150
Neuroinflammation
Cellular and molecular investigations of postmortem human brains revealed neuroinflammation-related damage in patients with PD.151,152,153,154 Both innate and adaptive immune responses are involved in PD progression.155,156,157,158 As brain-resident innate immune cells, activated microglia upregulate nuclear factor kappa-B (NF-κB) and NLR family pyrin domain-containing 3 (NLRP3), triggering an increase of cytokines, such as IL-1β and TNF-α.159,160 In patients with early PD, the midbrain and putamen are more densely populated with activated microglia,161,162 correlating with decreased activity of DA transporter ligands. Despite the widespread acceptance of chronic inflammation in PD, we remain uncertain regarding how neuroinflammation occurs. As a damage-associated molecular pattern (DAMP), α-synuclein may cause a proinflammatory shift when entering cells via toll-like receptor (TLR)-2.159,163,164,165,166 Dying or damaged cells may also release DAMPs, IL-1α, or mitochondrial ROS that trigger an innate immune response upon interaction with pattern recognition receptors (PRRs). Consecutive NLRP3 activation then elevates IL-1β synthesis, initiating further innate immune responses.167 Therefore, microglial activation in PD168,169 may result from PRR-mediated responses to DAMPs. In animal models with 6-OHDA-induced neurodegeneration, microglia were gradually repolarized from an anti-inflammatory M2 to a proinflammatory M1 phenotype.170 After repolarization, NF-κB initiates cytokine production in M1 cells,159,171,172 leading to interleukin and procaspase-1 transcription. These processes form the inflammasome NLRP3, which works with caspase-1 to activate proinflammatory IL-1β. Other proinflammatory proteins released from M1 cells (e.g., iNOS and TNF) also contribute to neurodegeneration in PD.173 Finally, Noelker et al. observed that TLR-4 knockout lowered the number of activated microglial cells and protected against SN dopaminergic degeneration, demonstrating that TLR-4 contributes to neuroinflammation.174 Adaptive immune responses also factor into neuroinflammation during PD. Several studies indicate that T-cell subpopulations contribute to PD pathophysiology.175 For example, CD4 and CD8 T cells significantly infiltrated into the SN of patients with PD,160 with CD8 T-cell concentrations particularly high.176,177 Because this infiltration occurs in early-stage PD and subsides with disease progression, CD8 T cells seem to be important at the beginning of the disease. Further evidence of CD4 T-cell contribution to neurodegeneration include their population and activity shifts in patients with PD, along with an increase in human leukocyte antigen-DR positive antigen-presenting microglia.178 Studies on the role of Th17 cells in PD have confirmed this hypothesis. Neurons appear to be more susceptible to IL-17 or autologous Th17 cells and are eventually subjected to NF-κB-dependent cell death.179 In addition, knockout or pharmacological inhibition of CD4 T cells downregulates major histocompatibility complex (MHC) II expression in CNS myeloid cells and protects against tyrosine hydroxylase (TH) neuron loss in the ipsilateral SN pars compacta (SNpc).179
Gut dysbiosis
The role of gut microbiota in neurological diseases has attracted considerable interest. Gut–brain microbiota signaling encompasses the CNS, enteric nervous system, autonomic nervous system, and hypothalamic–pituitary–adrenal axis. Signaling pathways between the CNS and enteric nervous system involve metabolites, hormones, the immune system, and afferent nerves.180,181 Microbiota can mediate inflammation of the enteric nervous system (Fig. 4). Intestinal inflammation is a contributing factor in PD pathogenesis, as patients exhibit high levels of zonulin and alpha-1-antitrypsin, markers of intestinal barrier dysfunction, as well as of calprotectin, a marker of intestinal inflammation.182 Specific microbial taxa have been closely associated with systemic inflammatory responses. For instance, Verrucomicrobiaceae abundance was correlated with plasma interferon (IFN)-γ levels, while Bacteroides abundance was associated with plasma TNF levels. Roseburia upregulated innate immune genes and negatively regulated the NF-κB pathway, thus promoting immune homeostasis.183 Through these effects on intestinal inflammation, gut microbiota and their metabolites could influence PD pathophysiology.184 A notable metabolite in this regard is lipopolysaccharide (LPS), which increases both α-synuclein accumulation in the enteric nervous system and intestinal permeability.185 Moreover, LPS intervention in Thy1-α-synuclein mice significantly decreased two tight junction proteins (zona occludens 1 and E-cadherin) in intestinal epithelial cells, highlighting a relationship between gut microbiota and PD pathogenesis.186 An example of evidence supporting microbiota involvement in PD is the finding that exposure to curli-producing bacteria increases α-synuclein deposition and accumulation in intestinal ganglion cells and the brain, leading to inflammation.187 Multiple animal experiments have also demonstrated that α-synuclein pathology can spread from the intestine to the brain along the gut–brain axis, and injecting α-synuclein into the intestinal wall causes pathological changes in the CNS.188,189,190,191
DAMPs (e.g., α-synuclein) trigger an innate immune response upon interaction with pattern recognition receptors in microglial cells. Microglial activation then increases the amount of NF-κB and NLRP3, leading to subsequent cytokine upregulation. Gut dysbiosis sends signals to the CNS and enteric nervous system via metabolites, hormones, and the immune system, thus mediating neuroinflammation
However, pathological processes are not necessarily limited to proceeding along the brain–gut or gut–brain axes. Pathologies may develop separately in the enteric nervous system and CNS during disease progression. In consideration of this possibility, Arotcarena et al. proposed a mechanism involving the general circulation acting as a path for long-distance, bidirectional transmission of endogenous α-synuclein between the intestinal tract and CNS.192
Research models of PD
Our understanding of the pathophysiology, etiology, and molecular processes of PD has improved thanks to a variety of models.193 The SNpc dopaminergic neurons constitute the foundation of the SNpc, and toxins like 6-OHDA, MPP+, and MPTP may quickly degenerate the SNpc, resulting in strong, well-defined motor impairments. Toxin models are the traditional models for PD research.194,195,196 Animal models of PD span invertebrate and vertebrate animals. Although C. elegans overexpressing α-synuclein damages dopaminergic neurons, the degeneration is not progressive, and α-synuclein inclusions are lacking.197,198 Drosophila overexpressing wild-type (WT), A53T, and A30P α-synuclein showed many PD characteristics, including age-dependent, selective loss of dopaminergic neurons and Lewy body-like filamentous inclusions.199 Nevertheless, they do not express α-synuclein with the complexity of vertebrates, nor can these models exhibit key clinical features, such as resting tremors, bradykinesia, and rigidity. While transgenic PD mice do not exhibit overt degenerative pathology in dopaminergic neurons, functional abnormalities are present in their nigrostriatal system.200 Of the mouse models available, only the mouse prion promoter A53T α-synuclein transgenic mice (MitoPark) recapitulated the full range of α-synuclein pathology observed in humans.200,201,202,203 These MitoPark mice are thus particularly promising as PD models. They are generated through selective disruption of the gene encoding mitochondrial transcription-factor A (Tfam) in dopaminergic neurons. The mutation lowers mitochondrial DNA copy number, similar to characteristics observed in human PD.204,205 In addition, Tfam disruption causes a respiratory chain deficiency that results in a progressive degenerative phenotype. Both these features of mitochondrial dysfunction are present in human PD, again emphasizing the model’s usefulness. Recently, a novel model emphasizing the aggregated, misfolded forms of α-synuclein seen in Lewy bodies has emerged as a crucial tool in PD research. Preformed fibrils (PFFs), which resemble the structural components of Lewy bodies and Lewy neurites, were produced by researchers incubating recombinant α-synuclein monomeric proteins under certain circumstances. In primary neuronal cultures from WT mice as well as cell lines overexpressing disease-related proteins, the PFFs can cause synaptic dysfunction, changes in cell excitability, and cell death.206,207 In mice overexpressing disease-related proteins or non-transgenic animals, intracerebral injection of PFFs into the dorsal striatum causes dysregulation of striatal DA release, neurodegeneration in the SNpc, and behavioral impairments.208,209,210,211 This type of model exhibits a longer time course of degeneration than other models, with early α-synuclein pathology in PD-relevant brain regions and the development of DA dysfunction, nigral degeneration, and motor deficits months after induction, suggesting a progression that is similar to that of the human condition. The repeatability of the results and the investment in a model with a time course of disease that takes several months to develop have been hampered by the model’s inability to reliably generate pathogenic PFFs.
Therapeutic strategies for PD
Commercially available drugs for PD
Many drugs have emerged as appropriate treatments, we differentiate the drugs according to their pharmacological targets and list them in Table 1.
Levodopa
Although a classic treatment for PD,212,213 levodopa (L-DOPA) has several undesirable side effects, including motor-response oscillations and drug-induced dyskinesias. Both presynaptic and postsynaptic mechanisms are involved in the development of these motor complications, which eventually arise from non-physiological pulsatile striatal DA receptor stimulation.214,215 The key cause of maladaptive neuronal responses is discontinuous drug delivery, stemming from L-DOPA’s short half-life, as well as variability in gastrointestinal absorption and blood brain barrier transport. To address these challenges, novel sustained-release formulations of L-DOPA and continuous delivery techniques are continuously being developed. These include intestinal delivery via percutaneous endoscopic gastrojejunostomy tubes and subcutaneous delivery via minipumps.216
DA agonists
Striatal medium spiny neurons have two types of DA receptors. Receptor agonists that directly target the D2 receptor family include dopaminomimetics such as the ergot alkaloid bromocriptine.217,218 Ergot alkaloids are ergoline derivatives that also activate 5-hydroxytryptamine (5-HT) receptors, including the 5-HT2B subtype. However, they have been implicated in cardiac valvular fibrosis and pleuropulmonary fibrosis, raising important safety concerns. In contrast, non-ergoline drugs do not have this issue and are thus preferred for PD treatment. DA agonists have a longer half-life than L-DOPA, making them strong candidates for adjunct therapy in patients with motor fluctuations.218,219,220 However, they have a lower overall effect than L-DOPA, as well as a higher tendency to cause sleepiness and hamper impulse control.221 Apomorphine is unique among DA agonists in having combined action on both D1 and D2 receptors, along with an equal affinity for L-DOPA.222 Continuous subcutaneous apomorphine infusions have been linked to a decrease in pre-existing L-DOPA-induced dyskinesias and limiting motor-response variations.223 Currently, new apomorphine formulations for sublingual use are undergoing clinical development.224
Catechol-O-methyltransferase and monoamine oxidase type B inhibitors
During the peripheral metabolism of L-DOPA, catechol-O-methyltransferase (COMT) ortho-methylates the drug via a secondary metabolic route. When COMT is inhibited, L-DOPA bioavailability and half-life are improved.225 Given this effect, COMT inhibitors combined with L-DOPA have become part of the first-line treatment for patients with PD. Currently, three COMT inhibitor preparations are available for clinical use, including entacapone and opicapone.217,226 Monoamine oxidase type B (MAO-B) is a primary clearance mechanism for synaptically released DA in glial cells.227 Inhibiting MAO-B (e.g., through selective inhibitor selegiline) prolongs DA’s effect and increases its synaptic concentrations.228 However, because selegiline is irreversible, safinamide has emerged as a reversible MAO-B inhibitor for use in PD treatments.229
Non-dopaminergic targets
Despite the remarkable effect of dopaminergic therapy on PD symptoms, therapies involving other targets remain a necessity. First, novel treatments are required to address complications of L-DOPA therapy, such as motor fluctuations, L-DOPA-induced dyskinesia, and L-DOPA-resistant (“non-dopaminergic”) motor features (e.g., treatment-resistant tremors, postural instability, frozen gait, swallowing difficulties, and speech disturbances). Currently, the only accessible and effective pharmacological tool for L-DOPA-induced dyskinesia is amantadine, hypothesized to be a N-methyl-D-aspartate receptor antagonist.217,220 Second, novel treatments must also address non-motor symptoms of PD, particularly depression, cognitive dysfunction, and autonomic failure. A major problem with non-motor symptoms is that many are unresponsive to DA replacement therapy; some are even precipitated or aggravated by this treatment.230 Cholinesterase inhibitors have beneficial effects on cognitive disturbances in patients with PD and dementia. This positive outcome is possibly associated with a significant loss of cholinergic projections from the nucleus basalis of Meynert in dementia.217,231 For psychotic symptoms in PD, clozapine is the most effective therapy. Finally, autonomic dysfunction is quite common in PD, especially during the late stage. A number of pharmacological therapies are available that predominantly focus on the autonomic nervous system. These include mineralocorticoid fludro cortisone; adrenergic agents (e.g., midodrine and etilefrine); anti-muscarinics (e.g., tolterodine, oxybutynin, or trospium chloride) for urinary urgency or incontinence; noradrenaline precursor (droxidopa) to treat orthostatic hypotension; and prokinetic drugs (e.g., macrogol or lubiprostone) to improve constipation.218,219,232
Drugs for PD treatment under clinical trials
Numerous clinical trials are underway to test the novel drugs that have been developed (Table 2). Some of them have shown potential candidates for PD. Tavapadon is a potent, highly selective, orally administered, DA D1/D5 receptor partial agonist and in the clinical trial (NCT02847650), compared to placebo, Tavapadon showed a better improvement effect which provided the research basis for the current clinical phase III trial (NCT04760769).233 IRL790 could interact with DA D3 receptor, and it was developed as an experimental treatment for L-DOPA-induced dyskinesia, impulse control disorder, and psychosis in PD. In the research,234 patients with advanced PD on IRL790 experienced a reduction in motor symptoms and no serious adverse effects and in the follow-up phase II clinical trials are still in the early stages (NCT03368170). Deferiprone in Phase II randomized double-blind placebo-controlled clinical trials (NCT00943748, NCT01539837) reduced SN iron deposition and progression of motor handicap in PD patients.235,236 Cu(II)ATSM exerted a positive effect by preventing lipid peroxidation in a Phase I dose-escalation study in early PD patients (NCT03204929) which suggests the potential for PD treatment.237 Prasinezumab is a monoclonal antibody directed against α-synuclein. As a potential therapeutic approach against a key target in PD, its development has received much attention but in a new phase II clinical study (NCT03100149), it showed no therapeutic effect compared to placebo and has safety concerns.238 But its effects may require more experiments to verify, and new clinical trials are underway (NCT04777331). DNL151 is an LRRK2 inhibitor and has shown a relatively obvious therapeutic effect on PD in the double-blind randomized clinical phase I trial (NCT04056689) hosted by Denali Therapeutics, and now it has launched clinical phase II (NCT05348785) and III phase trials (NCT05418673). If the results are good, it may prove the great potential of LRRK2 in PD treatment.
Botanical medications for PD treatment
Given the side effects of Western medicine and the invasiveness of external physical interventions, new treatments should be developed. In the past few years, many researchers have investigated the role of various natural products from medicinal plants and their formulations in the treatment of PD. Numerous natural products have been identified for the molecular regulation of PD (Table 3 and Fig. 5).
Phenol
Using a 6-OHDA-induced PD model, Yang et al. demonstrated the protective effects of curcumin on the injured hippocampus. These benefits included significant improvement in mental status, increases to DA and norepinephrine levels, neural regeneration of hippocampal tissue, and activation of proteins involved in cell survival-related pathways, such as BDNF, tropomyosin receptor kinase (Trk) B, and phosphoinositide 3-kinase (PI3K).239 Corroborating this study, other experiments with 6-OHDA PD models have shown that curcumin restores neuronal regeneration via stimulating Trk/PI3K signaling, which limits TNF-α and caspase activity while increasing BDNF levels.240 Further research into curcumin’s mechanism of action indicates that it at least partially involves interacting with a α7 nicotinic acetylcholine receptor. Through this mechanism, curcumin enhances the survival of striatal TH fibers and neurons in the SNpc and decreases abnormal turning behavior.241 Curcumin also inhibits a wide range of inflammatory compounds, including cytokines, ILs, chemokines, inflammatory enzymes, cycloxygenase-2, glial fibrillary acidic protein, and cyclin D1. Furthermore, curcumin blocks numerous participants in apoptotic pathways, such as iNOS, LPS-induced TNF-α, IL-1β, and IL-6, and c-Jun N-terminal kinase (JNK).242,243,244,245 These anti-inflammatory properties are further verified with data showing that curcumin modulates of the effects of various inflammatory mediators.246,247 In vitro and in vivo studies have shown that resveratrol exerts neuroprotective effects in PD models generated from 6-OHDA, MPP+, and rotenone.248,249,250,251 Resveratrol activates the pro-survival PI3K/protein kinase B (Akt) pathway, increases the B-cell lymphoma (Bcl-2)/Bcl-2-associated X (Bax) ratio, and decreases cytochrome C release so that caspase-3 remains inactive, thereby blocking apoptosis.252 Furthermore, resveratrol decreases ROS production and increases antioxidant defenses after exposure to MPTP/MPP+ 253 and rotenone.254,255 In experimentally induced PD, resveratrol protects against mitochondrial dysfunction, counteracting changes to mitochondrial morphology and mitochondrial membrane potential,250,254 while increasing mitochondrial biogenesis and complex-I activity.255,256 In several animal models, resveratrol stimulates autophagic degradation of α-synuclein after sirtuin (SIRT) 1 activation and decreases α-synuclein expression in the striatum.257,258 Another neuroprotective mechanism of resveratrol is similar to gastrodin action, activating HO-1 and mitogen-activated protein kinase (MAPK) pathways to increase autophagic flux.254,259 Regulation of astroglial activation also plays a role in neuroprotection. In a promising sign for clinical application, resveratrol presents synergistic effects when administered with L-DOPA.258
Alkaloids
Experiments applying berberine on MPTP-treated mice significantly ameliorated dopaminergic neuronal degeneration in the SN compacta and improved motor impairment. Berberine also decreased α-synuclein levels, enhanced microtubule-associated protein light chain 3 (LC3-II)-associated autophagy. Furthermore, berberine also activated adenosine 5′-monophosphate (AMP)-activated protein kinase (AMPK), and a notable benefit is that AMPK lowers α-synuclein-induced toxicity and protects cells from rotenone.260,261,262 Another study investigating the effects of berberine in mouse models showed that NLRP3-associated neuroinflammation was significantly mitigated and decreased the level of NLRP3 inflammasome in mice treated with berberine.263 The specific mechanism of berberine action may be its effect on TH, the rate-limiting enzyme in the phenylalanine–tyrosine–DA pathway. This pathway provides DA to the brain and generates L-DOPA with tetrahydrobiopterin as a coenzyme. In the gut, bacterial nitroreductase has been shown to transform berberine into dihydroberberine. This reaction supplies H• and increases tetrahydrobiopterin concentrations, which in turn enhances TH activity. The end result is an acceleration of L-DOPA production by gut bacteria.144,264,265 Through the autophagy–lysosome pathway, isorhynchophylline (IRN) promotes the clearance of WT, A53T, and A30P α-synuclein aggregates in neuronal cells. While independent of the mTOR pathway, IRN-induced autophagy is dependent on Beclin 1 function.266 Treatment with IRN markedly reduced MPP+-induced endoplasmic-reticulum stress responses. In addition, IRN inhibition of the apoptosis signal-regulating kinase 1 (ASK1)/JNK pathway appears to suppress mitochondria-dependent apoptosis which suggests the protection of neurons.267,268,269,270
Flavonoids
The flavonoid puerarin has anti-parkinsonian effects that are dependent on Nrf2. In a study using MPTP-treated mice, puerarin regulated Fyn and GSK-3β phosphorylation in the ventral midbrain. The Fyn/GSK-3β pathway facilitates Nrf2 accumulation in the nucleus, leading to de novo glutathione synthesis.271 Available evidence indicates that puerarin promotes dopaminergic neuron survival, proliferation, and differentiation via progesterone receptors.272,273 Puerarin also acts on the PI3K/Akt pathway to alleviate inflammatory responses274 and inhibit GSK-3 activity in neurons, thus limiting caspase-3 production and associated apoptosis.275,276 These interactions, along with inhibition of nuclear p53 accumulation, explain how puerarin protects against MPP+-induced neuroblastoma SH-SY5Y cell death.277 Baicalein administration reversed MPTP-induced motor dysfunction, dopaminergic neuronal loss, and proinflammatory cytokine elevation. In addition, baicalein inhibited the activation and proliferation of disease-associated proinflammatory microglia. The underlying mechanism of this protective effect is probably inhibition of the NLRP3/caspase-1/gasdermin D pathway.278 This pathway is associated with pyroptosis, a type of programmed cell death that participates in the loss of dopaminergic neurons.279,280,281,282 Pytoptosis occurs first through activating the NLRP3 inflammasome, which then promotes caspase-1 maturation.283 Caspase-1 then mediates the oligomerization of gasdermin D, a pyroptosis executive protein, thus stimulating proinflammatory IL-1β and IL-18 secretion.284,285 Research also showed that baicalein acts on the BDNF/TrkB/Cyclic AMP response-element binding protein (CREB) pathway to reduce α-synuclein aggregation and protect synaptic plasticity.286 Several studies using baicalein under different conditions have further highlighted its therapeutic value. For instance, combining baicalein and low-dose L-DOPA significantly recovered gait function in patients to a level comparable with results from high-dose L-DOPA treatment, although some L-DOPA side effects were also present.287 Finally, baicalein antagonized rotenone-induced ROS overproduction, upregulated Bax and cleaved caspase-3, downregulated Bcl-2, and phosphorylated extracellular signal-regulated kinases (ERK) 1/2.288
Terpenoids
Through acting on the Nrf2-NLRP3-caspase-1 pathway to inhibit the NLRP3 inflammasome, celastrol relieves motor deficits and nigrostriatal dopaminergic degeneration.289 In neurons, celastrol promotes autophagy, autophagosome biogenesis, and mitophagy, probably in association with MAPK pathways. In MPP+-induced PD cell models, celastrol inhibits dopaminergic neuronal death, ATP loss, and mitochondrial membrane depolarization. Research using these models also suggest that celastrol maintains mitochondrial quality by sequestering defective mitochondria into autophagosomes for degradation.290 After α-synuclein preformed fibril-induced microglial activation, triptolide treatment suppressed microglial activation and attenuated proinflammatory cytokine release. Specifically, the drug targeted the miR155-5p/Src homology 2 (SH2) domain in NF-κB, suppressing its activity in the inositol polyphosphate 5-phosphatase (SHIP)1 pathway.291 Studies suggest that miR155-5p overexpression provokes NF-κB activity through SHIP1 suppression.292 Triptolide acts to disrupt miR155-5p repression of SHIP1, thereby mitigating the inflammatory reaction. Research in an LPS-induced PD model demonstrated that blocking metabotropic glutamate receptor subtype 5 (mGlu5) attenuated the anti-inflammatory effects of triptolide. In addition, mGlu5 appears to mediate the effect of triptolide on microglia-induced astrocyte activation in vitro and in vivo.293 Triptolide has also been described as a potent autophagy inducer in neuronal cells, helping to clear various forms of α-synuclein via the autophagic pathway.294
Saponins
The underlying neuroprotective mechanism of ginsenoside Rb1-improvements to synaptic plasticity involves promoting hippocampal CA3 α-synuclein expression, restoring glutamate in the CA3‐schaffer collateral-CA1 pathway, and sequentially increasing postsynaptic density-95 expression.295 In LPS-treated rats, ginsenoside Rb1 treatment considerably lowered apomorphine-induced rotations, SN inflammation, and DA (plus metabolites) depletion in the striatum. These effects may be related to the inhibition of the NF-κB signaling pathway.296 The ginsenoside Rg3 augmented TH-positive neuron count in the SN, mean density of TH-positive nerve fibers, and DA content in the striatum, while also lowering ROS levels in the SN.297
Discussion and perspectives
PD is a debilitating neurodegenerative disease with pathological hallmarks of α-synuclein accumulation and loss of dopaminergic neurons in SN. The mechanisms involved in PD are very complex, such as the aggregation of α-synuclein, OS, neuroinflammation, ferroptosis, mitochondrial dysfunction, gut dysbiosis, etc. Interactions also have an important impact on the occurrence and progression of PD. The deep mechanism of the impact of these mechanisms on PD still needs to be explored, and for the models needed for experimental research, in addition to the toxin model, transgenic model and PFFs model that we have summarized, new models are needed to promote the research of PD. In the strategy for the treatment of PD, the main drug is still L-DOPA, and other drugs such as MAO-B inhibitors, COMT inhibitors, etc. are more commonly used in combination with L-DOPA. Moreover, the existing drugs often have no effective therapeutic effect on patients with advanced PD, which greatly reduces the quality of life of PD patients. Therefore, new drugs are still needed for treatment. Exploring new drugs can be considered from the mechanism of PD, such as targeting α-synuclein aggregation, ferroptosis and OS etc. The related drugs currently under development have not shown good effects, and may still require a long period of exploration. As for the new PD treatment drugs, besides the current hot chemical drugs, biological drugs, etc., some researchers have approved the therapeutic efficacy of traditional Chinese medicines which show tremendous potential in the field of PD treatment. The compounds derived from traditional Chinese medicines have complicated pharmacological effects and reverse the pathological mechanisms of PD such as the OS, neuroinflammation, and aggregation of α-synuclein. In the review of Sun et al., numerous natural drugs such as tanshinone and andrographolide that are reported for the anti-inflammatory effects and may be potential drugs for the treatment of PD have been proposed for further investigations.265 Moreover, based on the complex mechanisms, multi-drug combinations may offer a new perspective on PD treatment such as the combination of biological and chemical drugs or natural small molecules.
Besides the drug interventions, some surgical treatment methods also carry patients' new optimal approach. Deep brain interference induced by high-frequency (100–200 Hz) electrodes can replicate the impact of a lesion without impairing brain tissue.219 Taking advantage of this technology, a clinical trial has combined transcranial direct current stimulation and treadmill gait training to improve gait-function recovery in PD patients (NCT04591236). Transcutaneous magnetic stimulation of the spinal cord has also emerged recently as a possible therapeutic option for gait disorders, capable of stimulating neural elements non-invasively (NCT05008289). Another promising area of research is the use of stem cells. A clinical study first differentiated patient-derived midbrain dopaminergic progenitor cells from autologous iPSCs. These cells were then implanted into the putamen. The PD symptoms of treated patients then improved, allowing for a 6% decrease in L-DOPA-equivalent daily dose.298 These suggest that researchers may discovery the novel treatment approach in the area of stem cell transplant.
References
Vos, T. et al. Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990–2016: a systematic analysis for the Global Burden of Disease Study 2016. The Lancet 390, 1211–1259 (2017).
Parkinson, J. An essay on the shaking palsy. 1817. J. Neuropsychiatry Clin. Neurosci. 14, 223–236 (2002).
Charcot, J. M. Lecons sur, les maladies du système nerveux, Vol. 1 (Lecrosnier et Babé, 1886).
Przedborski, S. The two-century journey of Parkinson disease research. Nat. Rev. Neurosci. 18, 251–259 (2017).
Poewe, W. et al. Parkinson disease. Nat. Rev. Dis. Prim. 3, 17013 (2017).
Twelves, D., Perkins, K. S. & Counsell, C. Systematic review of incidence studies of Parkinson’s disease. Mov. Disord. 18, 19–31 (2003).
Savica, R., Grossardt, B. R., Bower, J. H., Ahlskog, J. E. & Rocca, W. A. Incidence and pathology of synucleinopathies and tauopathies related to parkinsonism. JAMA Neurol. 70, 859–866 (2013).
Van Den Eeden, S. K. et al. Incidence of Parkinson’s disease: variation by age, gender, and race/ethnicity. Am. J. Epidemiol. 157, 1015–1022 (2003).
Pringsheim, T., Jette, N., Frolkis, A. & Steeves, T. D. The prevalence of Parkinson’s disease: a systematic review and meta-analysis. Mov. Disord. 29, 1583–1590 (2014).
Baldereschi, M. et al. Parkinson’s disease and parkinsonism in a longitudinal study: two-fold higher incidence in men. ILSA Working Group. Italian Longitudinal Study on Aging. Neurology 55, 1358–1363 (2000).
Kusumi, M., Nakashima, K., Harada, H., Nakayama, H. & Takahashi, K. Epidemiology of Parkinson’s disease in Yonago City, Japan: comparison with a study carried out 12 years ago. Neuroepidemiology 15, 201–207 (1996).
Gordon, P. H., Mehal, J. M., Holman, R. C., Rowland, A. S. & Cheek, J. E. Parkinson’s disease among American Indians and Alaska natives: a nationwide prevalence study. Mov. Disord. 27, 1456–1459 (2012).
Morens, D. M. et al. Epidemiologic observations on Parkinson’s disease: incidence and mortality in a prospective study of middle-aged men. Neurology 46, 1044–1050 (1996).
Ascherio, A. & Schwarzschild, M. A. The epidemiology of Parkinson’s disease: risk factors and prevention. Lancet Neurol. 15, 1257–1272 (2016).
Gibb, W. R. & Lees, A. J. The relevance of the Lewy body to the pathogenesis of idiopathic Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 51, 745–752 (1988).
Marras, C. & Lang, A. Parkinson’s disease subtypes: lost in translation? J. Neurol. Neurosurg. Psychiatry 84, 409–415 (2013).
Jankovic, J. et al. Variable expression of Parkinson’s disease: a base-line analysis of the DATATOP cohort. The Parkinson Study Group. Neurology 40, 1529–1534 (1990).
Khoo, T. K. et al. The spectrum of nonmotor symptoms in early Parkinson disease. Neurology 80, 276–281 (2013).
Postuma, R. B. et al. Identifying prodromal Parkinson’s disease: pre-motor disorders in Parkinson’s disease. Mov. Disord. 27, 617–626 (2012).
Hely, M. A., Morris, J. G., Reid, W. G. & Trafficante, R. Sydney Multicenter Study of Parkinson’s disease: non-L-dopa-responsive problems dominate at 15 years. Mov. Disord. 20, 190–199 (2005).
Fearnley, J. M. & Lees, A. J. Ageing and Parkinson’s disease: substantia nigra regional selectivity. Brain 114, 2283–2301 (1991).
Damier, P., Hirsch, E. C., Agid, Y. & Graybiel, A. M. The substantia nigra of the human brain. II. Patterns of loss of dopamine-containing neurons in Parkinson’s disease. Brain 122, 1437–1448 (1999).
Dijkstra, A. A. et al. Stage-dependent nigral neuronal loss in incidental Lewy body and Parkinson’s disease. Mov. Disord. 29, 1244–1251 (2014).
Iacono, D. et al. Parkinson disease and incidental Lewy body disease: Just a question of time? Neurology 85, 1670–1679 (2015).
Braak, H. et al. Staging of brain pathology related to sporadic Parkinson’s disease. Neurobiol. Aging 24, 197–211 (2003).
Hirsch, E., Graybiel, A. M. & Agid, Y. A. Melanized dopaminergic neurons are differentially susceptible to degeneration in Parkinson’s disease. Nature 334, 345–348 (1988).
Hirsch, E. C., Orieux, G., Muriel, M. P., Francois, C. & Feger, J. Nondopaminergic neurons in Parkinson’s disease. Adv. Neurol. 91, 29–37 (2003).
Rizzo, G. et al. Accuracy of clinical diagnosis of Parkinson disease: a systematic review and meta-analysis. Neurology 86, 566–576 (2016).
Postuma, R. B. et al. Validation of the MDS clinical diagnostic criteria for Parkinson’s disease. Mov. Disord. 33, 1601–1608 (2018).
Krismer, F. et al. Sniffing the diagnosis: Olfactory testing in neurodegenerative parkinsonism. Parkinsonism Relat. Disord. 35, 36–41 (2017).
Katzenschlager, R. & Lees, A. J. Olfaction and Parkinson’s syndromes: its role in differential diagnosis. Curr. Opin. Neurol. 17, 417–423 (2004).
Ohtsuka, C. et al. Changes in substantia nigra and locus coeruleus in patients with early-stage Parkinson’s disease using neuromelanin-sensitive MR imaging. Neurosci. Lett. 541, 93–98 (2013).
Langkammer, C. et al. Quantitative susceptibility mapping (QSM) as a means to measure brain iron? A post mortem validation study. Neuroimage 62, 1593–1599 (2012).
Ehrminger, M. et al. The coeruleus/subcoeruleus complex in idiopathic rapid eye movement sleep behaviour disorder. Brain 139, 1180–1188 (2016).
Stoessl, A. J., Lehericy, S. & Strafella, A. P. Imaging insights into basal ganglia function, Parkinson’s disease, and dystonia. Lancet 384, 532–544 (2014).
Politis, M. Neuroimaging in Parkinson disease: from research setting to clinical practice. Nat. Rev. Neurol. 10, 708–722 (2014).
Kalinderi, K., Bostantjopoulou, S. & Fidani, L. The genetic background of Parkinson’s disease: current progress and future prospects. Acta Neurol. Scand. 134, 314–326 (2016).
Tambasco, N. et al. A53T in a parkinsonian family: a clinical update of the SNCA phenotypes. J. Neural Transm. (Vienna) 123, 1301–1307 (2016).
Marras, C., Canning, C. G. & Goldman, S. M. Environment, lifestyle, and Parkinson’s disease: Implications for prevention in the next decade. Mov. Disord. 34, 801–811 (2019).
Langston, J. W., Ballard, P., Tetrud, J. W. & Irwin, I. Chronic Parkinsonism in humans due to a product of meperidine-analog synthesis. Science 219, 979–980 (1983).
Tanner, C. M. Epidemiology of Parkinson’s disease. Neurol. Clin. 10, 317–329 (1992).
Ikeuchi, T. et al. Patients homozygous and heterozygous for SNCA duplication in a family with parkinsonism and dementia. Arch. Neurol. 65, 514–519 (2008).
Morgado, H. V., Vilatela, M. E. A. & López, M. L. The role of genetics in Parkinson’s disease. Salud Ment. 30, 16–24 (2007).
Monfrini, E. & Di Fonzo, A. Leucine-rich repeat kinase (LRRK2) genetics and Parkinson’s disease. Adv. Neurobiol. 14, 3–30 (2017).
Deng, H., Wang, P. & Jankovic, J. The genetics of Parkinson disease. Ageing Res Rev. 42, 72–85 (2018).
Hedrich, K. et al. Recurrent LRRK2 (Park8) mutations in early-onset Parkinson’s disease. Mov. Disord. 21, 1506–1510 (2006).
Martins, L. M. et al. Neuroprotective role of the Reaper-related serine protease HtrA2/Omi revealed by targeted deletion in mice. Mol. Cell Biol. 24, 9848–9862 (2004).
Plun-Favreau, H. et al. The mitochondrial protease HtrA2 is regulated by Parkinson’s disease-associated kinase PINK1. Nat. Cell Biol. 9, 1243–1252 (2007).
Moisoi, N. et al. Mitochondrial dysfunction triggered by loss of HtrA2 results in the activation of a brain-specific transcriptional stress response. Cell Death Differ. 16, 449–464 (2009).
Fitzgerald, J. C. et al. Phosphorylation of HtrA2 by cyclin-dependent kinase-5 is important for mitochondrial function. Cell Death Differ. 19, 257–266 (2012).
Basak, I. et al. Arabidopsis AtPARK13, which confers thermotolerance, targets misfolded proteins. J. Biol. Chem. 289, 14458–14469 (2014).
Bonifacino, J. S. & Hurley, J. H. Retromer. Curr. Opin. Cell Biol. 20, 427–436 (2008).
Arighi, C. N., Hartnell, L. M., Aguilar, R. C., Haft, C. R. & Bonifacino, J. S. Role of the mammalian retromer in sorting of the cation-independent mannose 6-phosphate receptor. J. Cell Biol. 165, 123–133 (2004).
Chartier-Harlin, M. C. et al. Translation initiator EIF4G1 mutations in familial Parkinson disease. Am. J. Hum. Genet. 89, 398–406 (2011).
Deng, H., Wu, Y. & Jankovic, J. The EIF4G1 gene and Parkinson’s disease. Acta Neurol. Scand. 132, 73–78 (2015).
Sonenberg, N. & Hinnebusch, A. G. Regulation of translation initiation in eukaryotes: mechanisms and biological targets. Cell 136, 731–745 (2009).
Hattori, N. & Mizuno, Y. Twenty years since the discovery of the parkin gene. J. Neural Transm. 124, 1037–1054 (2017).
Klein, C., Lohmann-Hedrich, K., Rogaeva, E., Schlossmacher, M. G. & Lang, A. E. Deciphering the role of heterozygous mutations in genes associated with parkinsonism. Lancet Neurol. 6, 652–662 (2007).
Pickrell, A. M. & Youle, R. J. The roles of PINK1, parkin, and mitochondrial fidelity in Parkinson’s disease. Neuron 85, 257–273 (2015).
Seirafi, M., Kozlov, G. & Gehring, K. Parkin structure and function. FEBS J. 282, 2076–2088 (2015).
van der Merwe, C., Jalali Sefid Dashti, Z., Christoffels, A., Loos, B. & Bardien, S. Evidence for a common biological pathway linking three Parkinson’s disease-causing genes: parkin, PINK1 and DJ-1. Eur. J. Neurosci. 41, 1113–1125 (2015).
Wilhelmus, M. M., Nijland, P. G., Drukarch, B., de Vries, H. E. & van Horssen, J. Involvement and interplay of Parkin, PINK1, and DJ1 in neurodegenerative and neuroinflammatory disorders. Free Radic. Biol. Med. 53, 983–992 (2012).
Zondler, L. et al. DJ-1 interactions with α-synuclein attenuate aggregation and cellular toxicity in models of Parkinson’s disease. Cell Death Dis. 5, e1350 (2014).
Demirsoy, S. et al. ATP13A2/PARK9 regulates endo-/lysosomal cargo sorting and proteostasis through a novel PI(3, 5)P2-mediated scaffolding function. Hum. Mol. Genet. 26, 1656–1669 (2017).
Holemans, T. et al. A lipid switch unlocks Parkinson’s disease-associated ATP13A2. Proc. Natl Acad. Sci. USA 112, 9040–9045 (2015).
Park, J. S., Koentjoro, B., Veivers, D., Mackay-Sim, A. & Sue, C. M. Parkinson’s disease-associated human ATP13A2 (PARK9) deficiency causes zinc dyshomeostasis and mitochondrial dysfunction. Hum. Mol. Genet. 23, 2802–2815 (2014).
Tsunemi, T. & Krainc, D. Zn2+ dyshomeostasis caused by loss of ATP13A2/PARK9 leads to lysosomal dysfunction and alpha-synuclein accumulation. Hum. Mol. Genet. 23, 2791–2801 (2014).
Park, J. S., Blair, N. F. & Sue, C. M. The role of ATP13A2 in Parkinson’s disease: clinical phenotypes and molecular mechanisms. Mov. Disord. 30, 770–779 (2015).
Kong, S. M. et al. Parkinson’s disease-linked human PARK9/ATP13A2 maintains zinc homeostasis and promotes α-Synuclein externalization via exosomes. Hum. Mol. Genet. 23, 2816–2833 (2014).
Teixeira, F. R. et al. Gsk3β and Tomm20 are substrates of the SCFFbxo7/PARK15 ubiquitin ligase associated with Parkinson’s disease. Biochem J. 473, 3563–3580 (2016).
Ordureau, A. et al. Defining roles of PARKIN and ubiquitin phosphorylation by PINK1 in mitochondrial quality control using a ubiquitin replacement strategy. Proc. Natl Acad. Sci. USA 112, 6637–6642 (2015).
Duarte, F. V. et al. Adenosine receptors: regulatory players in the preservation of mitochondrial function induced by ischemic preconditioning of rat liver. Purinergic Signal 13, 179–190 (2017).
Baines, C. P. et al. Loss of cyclophilin D reveals a critical role for mitochondrial permeability transition in cell death. Nature 434, 658–662 (2005).
Zhou, Z. D., Lee, J. C. T. & Tan, E. K. Pathophysiological mechanisms linking F-box only protein 7 (FBXO7) and Parkinson’s disease (PD). Mutat. Res Rev. Mutat. Res. 778, 72–78 (2018).
Delgado-Camprubi, M., Esteras, N., Soutar, M. P., Plun-Favreau, H. & Abramov, A. Y. Deficiency of Parkinson’s disease-related gene Fbxo7 is associated with impaired mitochondrial metabolism by PARP activation. Cell Death Differ. 24, 120–131 (2017).
Maries, E., Dass, B., Collier, T. J., Kordower, J. H. & Steece-Collier, K. The role of alpha-synuclein in Parkinson’s disease: insights from animal models. Nat. Rev. Neurosci. 4, 727–738 (2003).
Vekrellis, K., Xilouri, M., Emmanouilidou, E., Rideout, H. J. & Stefanis, L. Pathological roles of α-synuclein in neurological disorders. Lancet Neurol. 10, 1015–1025 (2011).
Wales, P., Pinho, R., Lázaro, D. F. & Outeiro, T. F. Limelight on alpha-synuclein: pathological and mechanistic implications in neurodegeneration. J. Parkinsons Dis. 3, 415–459 (2013).
Burré, J. The synaptic function of α-synuclein. J. Parkinsons Dis. 5, 699–713 (2015).
Ruipérez, V., Darios, F. & Davletov, B. Alpha-synuclein, lipids and Parkinson’s disease. Prog. Lipid Res. 49, 420–428 (2010).
Kim, C. & Lee, S. J. Controlling the mass action of alpha-synuclein in Parkinson’s disease. J. Neurochem 107, 303–316 (2008).
Melki, R. Role of different alpha-synuclein strains in synucleinopathies, similarities with other neurodegenerative diseases. J. Parkinsons Dis. 5, 217–227 (2015).
Kaushik, S. & Cuervo, A. M. Proteostasis and aging. Nat. Med. 21, 1406–1415 (2015).
Xilouri, M., Brekk, O. R. & Stefanis, L. α-Synuclein and protein degradation systems: a reciprocal relationship. Mol. Neurobiol. 47, 537–551 (2013).
Brieger, K., Schiavone, S., Miller, F. J. Jr & Krause, K. H. Reactive oxygen species: from health to disease. Swiss Med. Wkly 142, w13659 (2012).
Lee, D. H., Gold, R. & Linker, R. A. Mechanisms of oxidative damage in multiple sclerosis and neurodegenerative diseases: therapeutic modulation via fumaric acid esters. Int. J. Mol. Sci. 13, 11783–11803 (2012).
Shukla, V., Mishra, S. K. & Pant, H. C. Oxidative stress in neurodegeneration. Adv. Pharm. Sci. 2011, 572634 (2011).
Kemppainen, S. et al. Cerebral dopamine neurotrophic factor improves long-term memory in APP/PS1 transgenic mice modeling Alzheimer’s disease as well as in wild-type mice. Behav. Brain Res 291, 1–11 (2015).
Melief, E. J. et al. Partial depletion of striatal dopamine enhances penetrance of cognitive deficits in a transgenic mouse model of Alzheimer’s disease. J. Neurosci. Res. 93, 1413–1422 (2015).
Bedard, K. & Krause, K. H. The NOX family of ROS-generating NADPH oxidases: physiology and pathophysiology. Physiol. Rev. 87, 245–313 (2007).
Halliwell, B. Oxidative stress and neurodegeneration: where are we now? J. Neurochem. 97, 1634–1658 (2006).
Kovac, S. et al. Nrf2 regulates ROS production by mitochondria and NADPH oxidase. Biochim Biophys. Acta 1850, 794–801 (2015).
Subramaniam, S. R. & Chesselet, M. F. Mitochondrial dysfunction and oxidative stress in Parkinson’s disease. Prog. Neurobiol. 106-107, 17–32 (2013).
Starkov, A. A. The role of mitochondria in reactive oxygen species metabolism and signaling. Ann. N. Y Acad. Sci. 1147, 37–52 (2008).
Dionísio, P. A., Amaral, J. D. & Rodrigues, C. M. P. Oxidative stress and regulated cell death in Parkinson’s disease. Ageing Res. Rev. 67, 101263 (2021).
Guiney, S. J., Adlard, P. A., Bush, A. I., Finkelstein, D. I. & Ayton, S. Ferroptosis and cell death mechanisms in Parkinson’s disease. Neurochem. Int. 104, 34–48 (2017).
Kagan, V. E. et al. Oxidized arachidonic and adrenic PEs navigate cells to ferroptosis. Nat. Chem. Biol. 13, 81–90 (2017).
Dehay, B. et al. Pathogenic lysosomal depletion in Parkinson’s disease. J. Neurosci. 30, 12535–12544 (2010).
Hernandes, M. S., Café-Mendes, C. C. & Britto, L. R. NADPH oxidase and the degeneration of dopaminergic neurons in parkinsonian mice. Oxid. Med. Cell Longev. 2013, 157857 (2013).
Dryanovski, D. I. et al. Calcium entry and α-synuclein inclusions elevate dendritic mitochondrial oxidant stress in dopaminergic neurons. J. Neurosci. 33, 10154–10164 (2013).
Dixon, S. J. et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell 149, 1060–1072 (2012).
Do Van, B. et al. Ferroptosis, a newly characterized form of cell death in Parkinson’s disease that is regulated by PKC. Neurobiol. Dis. 94, 169–178 (2016).
Friedmann Angeli, J. P. et al. Inactivation of the ferroptosis regulator Gpx4 triggers acute renal failure in mice. Nat. Cell Biol. 16, 1180–1191 (2014).
Skouta, R. et al. Ferrostatins inhibit oxidative lipid damage and cell death in diverse disease models. J. Am. Chem. Soc. 136, 4551–4556 (2014).
Cao, J. et al. DJ-1 suppresses ferroptosis through preserving the activity of S-adenosyl homocysteine hydrolase. Nat. Commun. 11, 1251 (2020).
Forcina, G. C. & Dixon, S. J. GPX4 at the crossroads of lipid homeostasis and ferroptosis. Proteomics 19, e1800311 (2019).
Cozza, G. et al. Glutathione peroxidase 4-catalyzed reduction of lipid hydroperoxides in membranes: the polar head of membrane phospholipids binds the enzyme and addresses the fatty acid hydroperoxide group toward the redox center. Free Radic. Biol. Med. 112, 1–11 (2017).
Castellani, R. J., Siedlak, S. L., Perry, G. & Smith, M. A. Sequestration of iron by Lewy bodies in Parkinson’s disease. Acta Neuropathol. 100, 111–114 (2000).
Golts, N. et al. Magnesium inhibits spontaneous and iron-induced aggregation of alpha-synuclein. J. Biol. Chem. 277, 16116–16123 (2002).
Guo, J. J. et al. Intranasal administration of α-synuclein preformed fibrils triggers microglial iron deposition in the substantia nigra of Macaca fascicularis. Cell Death Dis. 12, 81 (2021).
Kenkhuis, B. et al. Iron loading is a prominent feature of activated microglia in Alzheimer’s disease patients. Acta Neuropathol. Commun. 9, 27 (2021).
Friedrich, I. et al. Cell specific quantitative iron mapping on brain slices by immuno-µPIXE in healthy elderly and Parkinson’s disease. Acta Neuropathol. Commun. 9, 47 (2021).
Healy, S., McMahon, J., Owens, P. & FitzGerald, U. Significant glial alterations in response to iron loading in a novel organotypic hippocampal slice culture model. Sci. Rep. 6, 36410 (2016).
Wang, J., Song, N., Jiang, H., Wang, J. & Xie, J. Pro-inflammatory cytokines modulate iron regulatory protein 1 expression and iron transportation through reactive oxygen/nitrogen species production in ventral mesencephalic neurons. Biochim Biophys. Acta 1832, 618–625 (2013).
Urrutia, P. et al. Inflammation alters the expression of DMT1, FPN1 and hepcidin, and it causes iron accumulation in central nervous system cells. J. Neurochem. 126, 541–549 (2013).
Sharma, N. & Nehru, B. Apocyanin, a microglial NADPH oxidase inhibitor prevents dopaminergic neuronal degeneration in lipopolysaccharide-induced Parkinson’s disease model. Mol. Neurobiol. 53, 3326–3337 (2016).
Kapralov, A. A. et al. Redox lipid reprogramming commands susceptibility of macrophages and microglia to ferroptotic death. Nat. Chem. Biol. 16, 278–290 (2020).
Xu, H. et al. New progress on the role of glia in iron metabolism and iron-induced degeneration of dopamine neurons in Parkinson’s disease. Front. Mol. Neurosci. 10, 455 (2017).
Jeong, S. Y. & David, S. Glycosylphosphatidylinositol-anchored ceruloplasmin is required for iron efflux from cells in the central nervous system. J. Biol. Chem. 278, 27144–27148 (2003).
Ayton, S. et al. Ceruloplasmin dysfunction and therapeutic potential for Parkinson disease. Ann. Neurol. 73, 554–559 (2013).
Wang, J., Bi, M. & Xie, J. Ceruloplasmin is involved in the nigral iron accumulation of 6-OHDA-lesioned rats. Cell Mol. Neurobiol. 35, 661–668 (2015).
Fernández-Mendívil, C. et al. Protective role of microglial HO-1 blockade in aging: implication of iron metabolism. Redox Biol. 38, 101789 (2021).
Song, W. et al. Parkinsonian features in aging GFAP.HMOX1 transgenic mice overexpressing human HO-1 in the astroglial compartment. Neurobiol. Aging 58, 163–179 (2017).
Wang, Z. L., Yuan, L., Li, W. & Li, J. Y. Ferroptosis in Parkinson’s disease: glia-neuron crosstalk. Trends Mol. Med. 28, 258–269 (2022).
Burns, R. S., LeWitt, P. A., Ebert, M. H., Pakkenberg, H. & Kopin, I. J. The clinical syndrome of striatal dopamine deficiency. Parkinsonism induced by 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP). N. Engl. J. Med. 312, 1418–1421 (1985).
Chaturvedi, R. K. & Beal, M. F. Mitochondrial approaches for neuroprotection. Ann. N. Y Acad. Sci. 1147, 395–412 (2008).
Panov, A. et al. Rotenone model of Parkinson disease: multiple brain mitochondria dysfunctions after short term systemic rotenone intoxication. J. Biol. Chem. 280, 42026–42035 (2005).
Borland, M. K. et al. Chronic, low-dose rotenone reproduces Lewy neurites found in early stages of Parkinson’s disease, reduces mitochondrial movement and slowly kills differentiated SH-SY5Y neural cells. Mol. Neurodegener. 3, 21 (2008).
Dauer, W. & Przedborski, S. Parkinson’s disease: mechanisms and models. Neuron 39, 889–909 (2003).
Klivenyi, P. et al. Mice lacking alpha-synuclein are resistant to mitochondrial toxins. Neurobiol. Dis. 21, 541–548 (2006).
Thomas, B. & Beal, M. F. Parkinson’s disease. Hum. Mol. Genet. 16, R183–R194 (2007).
Mudò, G. et al. Transgenic expression and activation of PGC-1α protect dopaminergic neurons in the MPTP mouse model of Parkinson’s disease. Cell Mol. Life Sci. 69, 1153–1165 (2012).
St-Pierre, J. et al. Suppression of reactive oxygen species and neurodegeneration by the PGC-1 transcriptional coactivators. Cell 127, 397–408 (2006).
Lin, M. T. & Beal, M. F. Mitochondrial dysfunction and oxidative stress in neurodegenerative diseases. Nature 443, 787–795 (2006).
Casarejos, M. J. et al. Susceptibility to rotenone is increased in neurons from parkin null mice and is reduced by minocycline. J. Neurochem. 97, 934–946 (2006).
Palacino, J. J. et al. Mitochondrial dysfunction and oxidative damage in parkin-deficient mice. J. Biol. Chem. 279, 18614–18622 (2004).
Liu, G. et al. alpha-Synuclein is differentially expressed in mitochondria from different rat brain regions and dose-dependently down-regulates complex I activity. Neurosci. Lett. 454, 187–192 (2009).
Bertolin, G. et al. The TOMM machinery is a molecular switch in PINK1 and PARK2/PARKIN-dependent mitochondrial clearance. Autophagy 9, 1801–1817 (2013).
Narendra, D., Tanaka, A., Suen, D. F. & Youle, R. J. Parkin is recruited selectively to impaired mitochondria and promotes their autophagy. J. Cell Biol. 183, 795–803 (2008).
Geisler, S. et al. PINK1/Parkin-mediated mitophagy is dependent on VDAC1 and p62/SQSTM1. Nat. Cell Biol. 12, 119–131 (2010).
Matsuda, N. et al. PINK1 stabilized by mitochondrial depolarization recruits Parkin to damaged mitochondria and activates latent Parkin for mitophagy. J. Cell Biol. 189, 211–221 (2010).
Narendra, D. P. et al. PINK1 is selectively stabilized on impaired mitochondria to activate Parkin. PLoS Biol. 8, e1000298 (2010).
Vives-Bauza, C. et al. PINK1-dependent recruitment of Parkin to mitochondria in mitophagy. Proc. Natl Acad. Sci. USA 107, 378–383 (2010).
Liu, W. et al. SHP2-mediated mitophagy boosted by lovastatin in neuronal cells alleviates parkinsonism in mice. Signal Transduct. Target Ther. 6, 34 (2021).
Lavara-Culebras, E. & Paricio, N. Drosophila DJ-1 mutants are sensitive to oxidative stress and show reduced lifespan and motor deficits. Gene 400, 158–165 (2007).
Irrcher, I. et al. Loss of the Parkinson’s disease-linked gene DJ-1 perturbs mitochondrial dynamics. Hum. Mol. Genet. 19, 3734–3746 (2010).
Krebiehl, G. et al. Reduced basal autophagy and impaired mitochondrial dynamics due to loss of Parkinson’s disease-associated protein DJ-1. PLoS ONE 5, e9367 (2010).
Saha, S. et al. LRRK2 modulates vulnerability to mitochondrial dysfunction in Caenorhabditis elegans. J. Neurosci. 29, 9210–9218 (2009).
Yue, M. et al. Progressive dopaminergic alterations and mitochondrial abnormalities in LRRK2 G2019S knock-in mice. Neurobiol. Dis. 78, 172–195 (2015).
Wang, X. et al. LRRK2 regulates mitochondrial dynamics and function through direct interaction with DLP1. Hum. Mol. Genet. 21, 1931–1944 (2012).
Mogi, M. et al. Interleukin-1 beta, interleukin-6, epidermal growth factor and transforming growth factor-alpha are elevated in the brain from Parkinsonian patients. Neurosci. Lett. 180, 147–150 (1994).
Mogi, M. et al. Tumor necrosis factor-alpha (TNF-alpha) increases both in the brain and in the cerebrospinal fluid from parkinsonian patients. Neurosci. Lett. 165, 208–210 (1994).
Mogi, M. et al. Transforming growth factor-beta 1 levels are elevated in the striatum and in ventricular cerebrospinal fluid in Parkinson’s disease. Neurosci. Lett. 193, 129–132 (1995).
Mogi, M., Harada, M., Kondo, T., Riederer, P. & Nagatsu, T. Interleukin-2 but not basic fibroblast growth factor is elevated in parkinsonian brain. Short communication. J. Neural Transm. 103, 1077–1081 (1996).
Sommer, A. et al. Infiltrating T lymphocytes reduce myeloid phagocytosis activity in synucleinopathy model. J. Neuroinflammation 13, 174 (2016).
Brochard, V. et al. Infiltration of CD4+ lymphocytes into the brain contributes to neurodegeneration in a mouse model of Parkinson disease. J. Clin. Investig. 119, 182–192 (2009).
Stone, D. K., Reynolds, A. D., Mosley, R. L. & Gendelman, H. E. Innate and adaptive immunity for the pathobiology of Parkinson’s disease. Antioxid. Redox Signal 11, 2151–2166 (2009).
Kustrimovic, N. et al. Parkinson’s disease patients have a complex phenotypic and functional Th1 bias: cross-sectional studies of CD4+ Th1/Th2/T17 and Treg in drug-naïve and drug-treated patients. J. Neuroinflammation 15, 205 (2018).
Panicker, N. et al. Fyn kinase regulates misfolded α-synuclein uptake and NLRP3 inflammasome activation in microglia. J. Exp. Med. 216, 1411–1430 (2019).
Ferreira, S. A. & Romero-Ramos, M. Microglia response during Parkinson’s disease: alpha-synuclein intervention. Front. Cell Neurosci. 12, 247 (2018).
Ouchi, Y. et al. Microglial activation and dopamine terminal loss in early Parkinson’s disease. Ann. Neurol. 57, 168–175 (2005).
Ouchi, Y., Yagi, S., Yokokura, M. & Sakamoto, M. Neuroinflammation in the living brain of Parkinson’s disease. Parkinsonism Relat. Disord. 15, S200–S204 (2009).
Haque, M. E. et al. Targeting the microglial NLRP3 inflammasome and its role in Parkinson’s disease. Mov. Disord. 35, 20–33 (2020).
Labzin, L. I., Heneka, M. T. & Latz, E. Innate immunity and neurodegeneration. Annu. Rev. Med. 69, 437–449 (2018).
Earls, R. H. et al. Intrastriatal injection of preformed alpha-synuclein fibrils alters central and peripheral immune cell profiles in non-transgenic mice. J. Neuroinflammation 16, 250 (2019).
Kuhbandner, K. et al. Alpha-synuclein: a modulator during inflammatory CNS demyelination. J. Mol. Neurosci. 70, 1038–1049 (2020).
Dinarello, C. A. Overview of the IL-1 family in innate inflammation and acquired immunity. Immunol. Rev. 281, 8–27 (2018).
McGeer, P. L., Itagaki, S., Boyes, B. E. & McGeer, E. G. Reactive microglia are positive for HLA-DR in the substantia nigra of Parkinson’s and Alzheimer’s disease brains. Neurology 38, 1285–1291 (1988).
McGeer, P. L. & McGeer, E. G. Inflammation and the degenerative diseases of aging. Ann. N. Y Acad. Sci. 1035, 104–116 (2004).
Ambrosi, G. et al. Complex changes in the innate and adaptive immunity accompany progressive degeneration of the nigrostriatal pathway induced by intrastriatal injection of 6-hydroxydopamine in the rat. Neurotox. Res. 32, 71–81 (2017).
Singh, S. S. et al. NF-κB-mediated neuroinflammation in Parkinson’s disease and potential therapeutic effect of polyphenols. Neurotox. Res 37, 491–507 (2020).
Tang, Y. & Le, W. Differential roles of M1 and M2 microglia in neurodegenerative diseases. Mol. Neurobiol. 53, 1181–1194 (2016).
Hunot, S. et al. Nitric oxide synthase and neuronal vulnerability in Parkinson’s disease. Neuroscience 72, 355–363 (1996).
Noelker, C. et al. Toll like receptor 4 mediates cell death in a mouse MPTP model of Parkinson disease. Sci Rep 3, 1393 (2013).
Sommer, A. et al. Th17 lymphocytes induce neuronal cell death in a human iPSC-based model of Parkinson’s disease. Cell Stem Cell 23, 123–131.e126 (2018).
Galiano-Landeira, J., Torra, A., Vila, M. & Bové, J. CD8 T cell nigral infiltration precedes synucleinopathy in early stages of Parkinson’s disease. Brain 143, 3717–3733 (2020).
Karikari, A. A. et al. Neurodegeneration by α-synuclein-specific T cells in AAV-A53T-α-synuclein Parkinson’s disease mice. Brain Behav. Immun. 101, 194–210 (2022).
Karikari, A. A. et al. Neurodegeneration by α-synuclein-specific T cells in AAV-A53T-α-synuclein Parkinson’s disease mice. Brain Behav Immun 101, 194-210 (2022).
Williams, G. P. et al. CD4 T cells mediate brain inflammation and neurodegeneration in a mouse model of Parkinson’s disease. Brain 144, 2047–2059 (2021).
Heiss, C. N. & Olofsson, L. E. The role of the gut microbiota in development, function and disorders of the central nervous system and the enteric nervous system. J. Neuroendocrinol. 31, e12684 (2019).
Lahiri, S. et al. The gut microbiota influences skeletal muscle mass and function in mice. Sci. Transl. Med. 11, eaan5662 (2019).
Schwiertz, A. et al. Fecal markers of intestinal inflammation and intestinal permeability are elevated in Parkinson’s disease. Parkinsonism Relat. Disord. 50, 104–107 (2018).
Patterson, A. M. et al. Human gut symbiont Roseburia hominis promotes and regulates innate immunity. Front. Immunol. 8, 1166 (2017).
Matheoud, D. et al. Intestinal infection triggers Parkinson’s disease-like symptoms in Pink1(-/-) mice. Nature 571, 565–569 (2019).
Kelly, L. P. et al. Progression of intestinal permeability changes and alpha-synuclein expression in a mouse model of Parkinson’s disease. Mov. Disord. 29, 999–1009 (2014).
Gorecki, A. M. et al. Altered gut microbiome in Parkinson’s disease and the influence of lipopolysaccharide in a human α-synuclein over-expressing mouse model. Front. Neurosci. 13, 839 (2019).
Chen, S. G. et al. Exposure to the functional bacterial amyloid protein curli enhances alpha-synuclein aggregation in aged Fischer 344 rats and Caenorhabditis elegans. Sci. Rep. 6, 34477 (2016).
Challis, C. et al. Gut-seeded α-synuclein fibrils promote gut dysfunction and brain pathology specifically in aged mice. Nat. Neurosci. 23, 327–336 (2020).
Kim, S. et al. Transneuronal propagation of pathologic α-synuclein from the gut to the brain models Parkinson’s disease. Neuron 103, 627–641.e627 (2019).
Uemura, N. et al. Inoculation of α-synuclein preformed fibrils into the mouse gastrointestinal tract induces Lewy body-like aggregates in the brainstem via the vagus nerve. Mol. Neurodegener. 13, 21 (2018).
Van Den Berge, N. et al. Evidence for bidirectional and trans-synaptic parasympathetic and sympathetic propagation of alpha-synuclein in rats. Acta Neuropathol. 138, 535–550 (2019).
Arotcarena, M. L. et al. Bidirectional gut-to-brain and brain-to-gut propagation of synucleinopathy in non-human primates. Brain 143, 1462–1475 (2020).
Dickson, D. W. Neuropathology of Parkinson disease. Parkinsonism Relat. Disord. 46, S30–s33 (2018).
Schober, A. Classic toxin-induced animal models of Parkinson’s disease: 6-OHDA and MPTP. Cell Tissue Res. 318, 215–224 (2004).
Vandamme, T. F. Use of rodents as models of human diseases. J. Pharm. Bioallied Sci. 6, 2–9 (2014).
Ding, F. et al. Development of a stable, early stage unilateral model of Parkinson’s disease in middle-aged rhesus monkeys. Exp. Neurol. 212, 431–439 (2008).
Kuwahara, T. et al. Familial Parkinson mutant alpha-synuclein causes dopamine neuron dysfunction in transgenic Caenorhabditis elegans. J. Biol. Chem. 281, 334–340 (2006).
Lakso, M. et al. Dopaminergic neuronal loss and motor deficits in Caenorhabditis elegans overexpressing human alpha-synuclein. J. Neurochem 86, 165–172 (2003).
Feany, M. B. & Bender, W. W. A Drosophila model of Parkinson’s disease. Nature 404, 394–398 (2000).
Chesselet, M. F. In vivo alpha-synuclein overexpression in rodents: a useful model of Parkinson’s disease? Exp. Neurol. 209, 22–27 (2008).
Dawson, T., Mandir, A. & Lee, M. Animal models of PD: pieces of the same puzzle? Neuron 35, 219–222 (2002).
Giasson, B. I. et al. Neuronal alpha-synucleinopathy with severe movement disorder in mice expressing A53T human alpha-synuclein. Neuron 34, 521–533 (2002).
Lee, M. K. et al. Human alpha-synuclein-harboring familial Parkinson’s disease-linked Ala-53 -> Thr mutation causes neurodegenerative disease with alpha-synuclein aggregation in transgenic mice. Proc. Natl Acad. Sci. USA 99, 8968–8973 (2002).
Ekstrand, M. I. et al. Mitochondrial transcription factor A regulates mtDNA copy number in mammals. Hum. Mol. Genet. 13, 935–944 (2004).
Grünewald, A., Kumar, K. R. & Sue, C. M. New insights into the complex role of mitochondria in Parkinson’s disease. Prog. Neurobiol. 177, 73–93 (2019).
Luk, K. C. et al. Exogenous alpha-synuclein fibrils seed the formation of Lewy body-like intracellular inclusions in cultured cells. Proc. Natl Acad. Sci. USA 106, 20051–20056 (2009).
Volpicelli-Daley, L. A. et al. Exogenous α-synuclein fibrils induce Lewy body pathology leading to synaptic dysfunction and neuron death. Neuron 72, 57–71 (2011).
Luk, K. C. et al. Intracerebral inoculation of pathological α-synuclein initiates a rapidly progressive neurodegenerative α-synucleinopathy in mice. J. Exp. Med. 209, 975–986 (2012).
Luk, K. C. et al. Pathological α-synuclein transmission initiates Parkinson-like neurodegeneration in nontransgenic mice. Science 338, 949–953 (2012).
Paumier, K. L. et al. Intrastriatal injection of pre-formed mouse α-synuclein fibrils into rats triggers α-synuclein pathology and bilateral nigrostriatal degeneration. Neurobiol. Dis. 82, 185–199 (2015).
Abdelmotilib, H. et al. α-Synuclein fibril-induced inclusion spread in rats and mice correlates with dopaminergic Neurodegeneration. Neurobiol. Dis. 105, 84–98 (2017).
Gray, R. et al. Long-term effectiveness of dopamine agonists and monoamine oxidase B inhibitors compared with levodopa as initial treatment for Parkinson’s disease (PD MED): a large, open-label, pragmatic randomised trial. Lancet 384, 1196–1205 (2014).
LeWitt, P. A. & Fahn, S. Levodopa therapy for Parkinson disease: a look backward and forward. Neurology 86, S3–S12 (2016).
Olanow, C. W., Obeso, J. A. & Stocchi, F. Continuous dopamine-receptor treatment of Parkinson’s disease: scientific rationale and clinical implications. Lancet Neurol. 5, 677–687 (2006).
Cenci, M. A. Presynaptic mechanisms of l-DOPA-induced dyskinesia: the findings, the debate, and the therapeutic implications. Front Neurol. 5, 242 (2014).
Poewe, W. & Antonini, A. Novel formulations and modes of delivery of levodopa. Mov. Disord. 30, 114–120 (2015).
Fox, S. H. et al. The movement disorder society evidence-based medicine review update: treatments for the motor symptoms of Parkinson’s disease. Mov. Disord. 26, S2–S41 (2011).
Connolly, B. S. & Lang, A. E. Pharmacological treatment of Parkinson disease: a review. J. Am. Med. Assoc. 311, 1670–1683 (2014).
Seppi, K. et al. The movement disorder society evidence-based medicine review update: treatments for the non-motor symptoms of Parkinson’s disease. Mov. Disord. 26, S42–S80 (2011).
Jankovic, J. & Poewe, W. Therapies in Parkinson’s disease. Curr. Opin. Neurol. 25, 433–447 (2012).
Voon, V., Mehta, A. R. & Hallett, M. Impulse control disorders in Parkinson’s disease: recent advances. Curr. Opin. Neurol. 24, 324–330 (2011).
Frankel, J. P., Lees, A. J., Kempster, P. A. & Stern, G. M. Subcutaneous apomorphine in the treatment of Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 53, 96–101 (1990).
Katzenschlager, R. et al. Continuous subcutaneous apomorphine therapy improves dyskinesias in Parkinson’s disease: a prospective study using single-dose challenges. Mov. Disord. 20, 151–157 (2005).
Hauser, R. A. et al. Sublingual apomorphine (APL-130277) for the acute conversion of OFF to ON in Parkinson’s disease. Mov. Disord. 31, 1366–1372 (2016).
Müller, T. Catechol-O-methyltransferase inhibitors in Parkinson’s disease. Drugs 75, 157–174 (2015).
Ferreira, J. J. et al. Opicapone as an adjunct to levodopa in patients with Parkinson’s disease and end-of-dose motor fluctuations: a randomised, double-blind, controlled trial. Lancet Neurol. 15, 154–165 (2016).
Schapira, A. H. Monoamine oxidase B inhibitors for the treatment of Parkinson’s disease: a review of symptomatic and potential disease-modifying effects. CNS Drugs 25, 1061–1071 (2011).
Birkmayer, W., Riederer, P., Ambrozi, L. & Youdim, M. B. Implications of combined treatment with ‘Madopar’ and L-deprenil in Parkinson’s disease. A long-term study. Lancet 1, 439–443 (1977).
Schapira, A. H. et al. Assessment of safety and efficacy of safinamide as a levodopa adjunct in patients with parkinson disease and motor fluctuations: a randomized clinical trial. JAMA Neurol. 74, 216–224 (2017).
Chaudhuri, K. R. & Schapira, A. H. Non-motor symptoms of Parkinson’s disease: dopaminergic pathophysiology and treatment. Lancet Neurol. 8, 464–474 (2009).
Connolly, B. & Fox, S. H. Treatment of cognitive, psychiatric, and affective disorders associated with Parkinson’s disease. Neurotherapeutics 11, 78–91 (2014).
Perez-Lloret, S., Rey, M. V., Pavy-Le Traon, A. & Rascol, O. Emerging drugs for autonomic dysfunction in Parkinson’s disease. Expert Opin. Emerg. Drugs 18, 39–53 (2013).
Riesenberg, R., Werth, J., Zhang, Y., Duvvuri, S. & Gray, D. PF-06649751 efficacy and safety in early Parkinson’s disease: a randomized, placebo-controlled trial. Ther. Adv. Neurol. Disord. 13, 1756286420911296 (2020).
Svenningsson, P. et al. Safety and tolerability of IRL790 in Parkinson’s disease with levodopa-induced dyskinesia-a phase 1b trial. NPJ Parkinsons Dis. 4, 35 (2018).
Devos, D. et al. Targeting chelatable iron as a therapeutic modality in Parkinson’s disease. Antioxid. Redox Signal 21, 195–210 (2014).
Martin-Bastida, A. et al. Brain iron chelation by deferiprone in a phase 2 randomised double-blinded placebo controlled clinical trial in Parkinson’s disease. Sci. Rep. 7, 1398 (2017).
Hung, L. W. et al. The hypoxia imaging agent CuII(atsm) is neuroprotective and improves motor and cognitive functions in multiple animal models of Parkinson’s disease. J Exp Med 209, 837-854 (2012).
Pagano, G. et al. Trial of prasinezumab in early-stage Parkinson’s disease. N. Engl. J. Med. 387, 421–432 (2022).
Yang, J., Song, S., Li, J. & Liang, T. Neuroprotective effect of curcumin on hippocampal injury in 6-OHDA-induced Parkinson’s disease rat. Pathol. Res. Pr. 210, 357–362 (2014).
Baj, T. & Seth, R. Role of curcumin in regulation of TNF-α mediated brain inflammatory responses. Recent Pat. Inflamm. Allergy Drug Discov. 12, 69–77 (2018).
El Nebrisi, E., Javed, H., Ojha, S. K., Oz, M. & Shehab, S. Neuroprotective effect of curcumin on the nigrostriatal pathway in a 6-hydroxydopmine-induced rat model of Parkinson’s disease is mediated by α7-nicotinic receptors. Int. J. Mol. Sci. 21, 7329 (2020).
Creţu, E., Trifan, A., Vasincu, A. & Miron, A. Plant-derived anticancer agents—curcumin in cancer prevention and treatment. Rev. Med. Chir. Soc. Med Nat. Iasi 116, 1223–1229 (2012).
Ghasemi, F., Bagheri, H., Barreto, G. E., Read, M. I. & Sahebkar, A. Effects of curcumin on microglial cells. Neurotox. Res. 36, 12–26 (2019).
Fan, C. et al. Neuroprotective effects of curcumin on il-1β-induced neuronal apoptosis and depression-like behaviors caused by chronic stress in rats. Front. Cell Neurosci. 12, 516 (2018).
Ghosh, S., Banerjee, S. & Sil, P. C. The beneficial role of curcumin on inflammation, diabetes and neurodegenerative disease: a recent update. Food Chem. Toxicol. 83, 111–124 (2015).
Gupta, S. C., Patchva, S., Koh, W. & Aggarwal, B. B. Discovery of curcumin, a component of golden spice, and its miraculous biological activities. Clin. Exp. Pharm. Physiol. 39, 283–299 (2012).
Sharma, N. & Nehru, B. Curcumin affords neuroprotection and inhibits α-synuclein aggregation in lipopolysaccharide-induced Parkinson’s disease model. Inflammopharmacology 26, 349–360 (2018).
Albani, D. et al. The SIRT1 activator resveratrol protects SK-N-BE cells from oxidative stress and against toxicity caused by alpha-synuclein or amyloid-beta (1-42) peptide. J. Neurochem 110, 1445–1456 (2009).
Zhang, J. et al. Resveratrol protects PC12 cell against 6-OHDA damage via CXCR4 signaling pathway. Evid. Based Complement Altern. Med. 2015, 730121 (2015).
Zeng, W., Zhang, W., Lu, F., Gao, L. & Gao, G. Resveratrol attenuates MPP(+)-induced mitochondrial dysfunction and cell apoptosis via AKT/GSK-3β pathway in SN4741 cells. Neurosci. Lett. 637, 50–56 (2017).
Wang, H. et al. Resveratrol suppresses rotenone-induced neurotoxicity through activation of SIRT1/Akt1 signaling pathway. Anat. Rec. 301, 1115–1125 (2018).
Huang, N. et al. Resveratrol delays 6-hydroxydopamine-induced apoptosis by activating the PI3K/Akt signaling pathway. Exp. Gerontol. 124, 110653 (2019).
Abolaji, A. O., Adedara, A. O., Adie, M. A., Vicente-Crespo, M. & Farombi, E. O. Resveratrol prolongs lifespan and improves 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced oxidative damage and behavioural deficits in Drosophila melanogaster. Biochem. Biophys. Res. Commun. 503, 1042–1048 (2018).
Lin, K. L. et al. Resveratrol provides neuroprotective effects through modulation of mitochondrial dynamics and ERK1/2 regulated autophagy. Free Radic. Res. 52, 1371–1386 (2018).
Palle, S. & Neerati, P. Improved neuroprotective effect of resveratrol nanoparticles as evinced by abrogation of rotenone-induced behavioral deficits and oxidative and mitochondrial dysfunctions in rat model of Parkinson’s disease. Naunyn Schmiedebergs Arch. Pharm. 391, 445–453 (2018).
Peng, K. et al. Resveratrol regulates mitochondrial biogenesis and fission/fusion to attenuate rotenone-induced neurotoxicity. Oxid. Med. Cell Longev. 2016, 6705621 (2016).
Guo, Y. J. et al. Resveratrol alleviates MPTP-induced motor impairments and pathological changes by autophagic degradation of α-synuclein via SIRT1-deacetylated LC3. Mol. Nutr. Food Res. 60, 2161–2175 (2016).
Liu, Q. et al. Resveratrol synergizes with low doses of L-DOPA to improve MPTP-induced Parkinson disease in mice. Behav. Brain Res. 367, 10–18 (2019).
Lin, T. K. et al. Resveratrol partially prevents rotenone-induced neurotoxicity in dopaminergic SH-SY5Y cells through induction of heme oxygenase-1 dependent autophagy. Int. J. Mol. Sci. 15, 1625–1646 (2014).
Deng, H. & Ma, Z. Protective effects of berberine against MPTP-induced dopaminergic neuron injury through promoting autophagy in mice. Food Funct. 12, 8366–8375 (2021).
Dulovic, M. et al. The protective role of AMP-activated protein kinase in alpha-synuclein neurotoxicity in vitro. Neurobiol. Dis. 63, 1–11 (2014).
Hou, Y. S. et al. Sestrin2 protects dopaminergic cells against rotenone toxicity through AMPK-dependent autophagy activation. Mol. Cell Biol. 35, 2740–2751 (2015).
Huang, S. et al. Berberine protects against NLRP3 inflammasome via ameliorating autophagic impairment in MPTP-induced Parkinson’s disease model. Front. Pharm. 11, 618787 (2020).
Wang, Y. et al. Oral berberine improves brain dopa/dopamine levels to ameliorate Parkinson’s disease by regulating gut microbiota. Signal Transduct. Target Ther. 6, 77 (2021).
Zhu, Y. et al. New opportunities and challenges of natural products research: when target identification meets single-cell multiomics. Acta Pharm. Sin. B 12, 4011–4039 (2022).
Lu, J. H. et al. Isorhynchophylline, a natural alkaloid, promotes the degradation of alpha-synuclein in neuronal cells via inducing autophagy. Autophagy 8, 98–108 (2012).
Li, X. M., Zhang, X. J. & Dong, M. X. Isorhynchophylline attenuates MPP(+)-induced apoptosis through endoplasmic reticulum stress- and mitochondria-dependent pathways in PC12 cells: involvement of antioxidant activity. Neuromolecular Med 19, 480–492 (2017).
Hu, X. et al. Peroxiredoxin-2 protects against 6-hydroxydopamine-induced dopaminergic neurodegeneration via attenuation of the apoptosis signal-regulating kinase (ASK1) signaling cascade. J. Neurosci. 31, 247–261 (2011).
Kim, S. M. et al. Neuroprotective effects of black soybean anthocyanins via inactivation of ASK1-JNK/p38 pathways and mobilization of cellular sialic acids. Life Sci. 90, 874–882 (2012).
Lee, K. W. et al. Apoptosis signal-regulating kinase 1 mediates MPTP toxicity and regulates glial activation. PLoS ONE 7, e29935 (2012).
Li, X., Zhang, J., Zhang, X. & Dong, M. Puerarin suppresses MPP(+)/MPTP-induced oxidative stress through an Nrf2-dependent mechanism. Food Chem. Toxicol. 144, 111644 (2020).
Shiying, L. et al. Puerarin promoted proliferation and differentiation of dopamine-producing cells in Parkinson’s animal models. Biomed. Pharmacother. 106, 1236–1242 (2018).
Zhao, Y. et al. Botanical drug puerarin promotes neuronal survival and neurite outgrowth against MPTP/MPP(+)-induced toxicity via progesterone receptor signaling. Oxid. Med. Cell Longev. 2020, 7635291 (2020).
Ji, Y., Wang, D., Zhang, B. & Lu, H. Bergenin ameliorates MPTP-induced Parkinson’s disease by activating PI3K/Akt signaling pathway. J. Alzheimers Dis. 72, 823–833 (2019).
Luo, S. et al. Akt phosphorylates NQO1 and triggers its degradation, abolishing its antioxidative activities in Parkinson’s disease. J. Neurosci. 39, 7291–7305 (2019).
Yang, L., Wang, H., Liu, L. & Xie, A. The role of insulin/IGF-1/PI3K/Akt/GSK3β signaling in Parkinson’s disease dementia. Front. Neurosci. 12, 73 (2018).
Zhu, G., Wang, X., Wu, S. & Li, Q. Involvement of activation of PI3K/Akt pathway in the protective effects of puerarin against MPP+-induced human neuroblastoma SH-SY5Y cell death. Neurochem Int. 60, 400–408 (2012).
Rui, W., Li, S., Xiao, H., Xiao, M. & Shi, J. Baicalein attenuates neuroinflammation by inhibiting NLRP3/caspase-1/GSDMD pathway in MPTP induced mice model of Parkinson’s disease. Int. J. Neuropsychopharmacol. 23, 762–773 (2020).
Cookson, B. T. & Brennan, M. A. Pro-inflammatory programmed cell death. Trends Microbiol. 9, 113–114 (2001).
Bergsbaken, T., Fink, S. L. & Cookson, B. T. Pyroptosis: host cell death and inflammation. Nat. Rev. Microbiol. 7, 99–109 (2009).
Gordon, R. et al. Inflammasome inhibition prevents α-synuclein pathology and dopaminergic neurodegeneration in mice. Sci. Transl. Med. 10, eaah4066 (2018).
Franchi, L., Eigenbrod, T., Muñoz-Planillo, R. & Nuñez, G. The inflammasome: a caspase-1-activation platform that regulates immune responses and disease pathogenesis. Nat. Immunol. 10, 241–247 (2009).
Zhao, D., Wu, Y., Zhuang, J., Xu, C. & Zhang, F. Activation of NLRP1 and NLRP3 inflammasomes contributed to cyclic stretch-induced pyroptosis and release of IL-1β in human periodontal ligament cells. Oncotarget 7, 68292–68302 (2016).
Chen, X. et al. Pyroptosis is driven by non-selective gasdermin-D pore and its morphology is different from MLKL channel-mediated necroptosis. Cell Res. 26, 1007–1020 (2016).
Wu, D. M. et al. Salidroside protection against oxidative stress injury through the Wnt/β-catenin signaling pathway in rats with Parkinson’s disease. Cell Physiol. Biochem. 46, 1793–1806 (2018).
Zhao, X. et al. Baicalein alleviates depression-like behavior in rotenone- induced Parkinson’s disease model in mice through activating the BDNF/TrkB/CREB pathway. Biomed. Pharmacother. 140, 111556 (2021).
Zheng, Z. V. et al. Baicalein enhances the effect of low dose Levodopa on the gait deficits and protects dopaminergic neurons in experimental Parkinsonism. J. Clin. Neurosci. 64, 242–251 (2019).
Song, J. X. et al. Baicalein antagonizes rotenone-induced apoptosis in dopaminergic SH-SY5Y cells related to Parkinsonism. Chin. Med. 7, 1 (2012).
Zhang, C. et al. The Nrf2-NLRP3-caspase-1 axis mediates the neuroprotective effects of Celastrol in Parkinson’s disease. Redox Biol. 47, 102134 (2021).
Lin, M. W., Lin, C. C., Chen, Y. H., Yang, H. B. & Hung, S. Y. Celastrol inhibits dopaminergic neuronal death of Parkinson’s disease through activating mitophagy. Antioxidants 9, 37 (2019).
Feng, Y. et al. Triptolide inhibits preformed fibril-induced microglial activation by targeting the microRNA155-5p/SHIP1 pathway. Oxid. Med Cell Longev. 2019, 6527638 (2019).
Lu, S., Liao, Q. S. & Tang, L. MiR-155 affects osteosarcoma cell proliferation and invasion through regulating NF-κB signaling pathway. Eur. Rev. Med. Pharm. Sci. 22, 7633–7639 (2018).
Huang, Y. Y. et al. Triptolide up-regulates metabotropic glutamate receptor 5 to inhibit microglia activation in the lipopolysaccharide-induced model of Parkinson’s disease. Brain Behav. Immun. 71, 93–107 (2018).
Hu, G. et al. Triptolide promotes the clearance of α-synuclein by enhancing autophagy in neuronal cells. Mol. Neurobiol. 54, 2361–2372 (2017).
Qu, S., Meng, X., Liu, Y., Zhang, X. & Zhang, Y. Ginsenoside Rb1 prevents MPTP-induced changes in hippocampal memory via regulation of the α-synuclein/PSD-95 pathway. Aging 11, 1934–1964 (2019).
Li, D. W. et al. Ginsenoside Rb1 protects dopaminergic neurons from inflammatory injury induced by intranigral lipopolysaccharide injection. Neural Regen. Res. 14, 1814–1822 (2019).
Han, Y. et al. Ginsenoside Rg3 exerts a neuroprotective effect in rotenone-induced Parkinson’s disease mice via its anti-oxidative properties. Eur. J. Pharm. 909, 174413 (2021).
Schweitzer, J. S. et al. Personalized iPSC-derived dopamine progenitor cells for Parkinson’s disease. N. Engl. J. Med. 382, 1926–1932 (2020).
Acknowledgements
The study was supported by the National Key Research and Development Program of China (2022YFC3501904, 2021YFC1712805), Zhejiang province commonweal projects (TGY23H090038, LG F22H280001), the Key Project at Central Government Level (2060302), and the Macau Science and Technology Development Fund, Macau Special Administrative Region, China.
Author information
Authors and Affiliations
Contributions
P.L.-H. conceived the article. X.D.-C., P.L.-Hand C.Y. mainly wrote the manuscript. X.Y., S.T.C.-Y. assisted in the writing of the manuscript. All authors have read, discussed and approved the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Dong-Chen, X., Yong, C., Yang, X. et al. Signaling pathways in Parkinson’s disease: molecular mechanisms and therapeutic interventions. Sig Transduct Target Ther 8, 73 (2023). https://doi.org/10.1038/s41392-023-01353-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41392-023-01353-3
- Springer Nature Limited
This article is cited by
-
Broad proteomics analysis of seeding-induced aggregation of α-synuclein in M83 neurons reveals remodeling of proteostasis mechanisms that might contribute to Parkinson’s disease pathogenesis
Molecular Brain (2024)
-
Hypoxia inducible factor-1α regulates microglial innate immune memory and the pathology of Parkinson’s disease
Journal of Neuroinflammation (2024)
-
Systematic analysis of multi-omics data reveals component-specific blood-based biomarkers for Parkinson’s disease
Translational Medicine Communications (2024)
-
COVID-19 related neurological manifestations in Parkinson’s disease: has ferroptosis been a suspect?
Cell Death Discovery (2024)
-
Identifying MSMO1, ELOVL6, AACS, and CERS2 related to lipid metabolism as biomarkers of Parkinson's disease
Scientific Reports (2024)