Abstract
Background
The treatment and management of patients suffering from hypophosphatemic rickets (HR) remain a major challenge for dental practitioners and affected patients.
Objectives
To report a case of HR presenting with specific dental findings and to review the dental manifestations and treatment of HR patients.
Methods
Case: A 32-year-old male presented with multiple dental abscesses and short stature. A thorough history was taken followed by clinical oral examination, and relevant radiological investigation was done. Literature research: In 2020, electronic literature searches were carried out in PubMed and complemented by a careful assessment of the reference lists of the identified relevant papers. Articles and reports fulfilled the inclusion criteria: indexed reviews, case series and case reports in English and restricted to human studies were considered.
Results
The intraoral examination revealed multiple dental abscesses and general periodontal disease; the radiographic examination showed poorly defined lamina dura, large pulp chambers and periapical lesions. Based on the contents of the 43 articles identified in the search, the current knowledge of dental manifestations, treatment and management of HR was summarized.
Conclusions
As HR is a multisystem disease, multidisciplinary care is needed. By summarizing current evidences, we proposed an evidence-based dental management and provided recommendations on diagnosis and treatment of the disease.
It is of profound clinical significance to acquire knowledge of the dental manifestations and provide optimal treatment options for patients.
Similar content being viewed by others
Introduction
Hypophosphatemic rickets (HR) is a group of hereditary metabolic bone diseases caused by renal phosphate wasting, which has no response of high doses of vitamin D [1]. HR is characterized by short stature, delayed walking, bow legs, bone/joint pain, spontaneous dental abscess, hypophosphatemia, and inappropriately normal serum 1,25(OH)2D level [2]. Genetic defects in factors necessary for phosphate handling is the main cause of phosphate wasting [3]. HR can be mainly divided into three types, including X-linked hypophosphatemic rickets (XLHR) due to a loss function of phosphate regulating gene with homologies to endopeptidases on the X chromosome (PHEX) [4], autosomal dominant hypophosphatemic rickets (ADHR) caused by mutation in the fibroblast growth factor 23 (FGF23) gene [5] and autosomal recessive hypophosphatemic rickets (ARHR) as a result of mutation in dentin matrix protein 1 (DMP1) gene, ecto-nucleotide pyrophos- phatase/phosphodiesterase 1 (ENNP1) gene, or family with sequence similarity 20, member C (FAM20C) [6,7,8].
XLHR, also named as vitamin D resistant rickets, familial hypophosphatemic rickets, or phosphate diabetes, is the most common form of HR, with an incidence of around 1:20000 [9]. PHEX, mainly expressed in osteocytes and odontoblasts, is located on chromosome Xp22.1 and encodes for an endopeptidase [10]. Inactivating PHEX mutations contribute to the increasing secretion of FGF23 [11]. As a key circulating factor that directs sodium-dependent phosphate transporters in the kidney and intestine, the high level FGF23 results in impaired proximal renal phosphate reabsorption, causing hypophosphatemia and systemic regulation of mineralization [12]. Meanwhile, the PHEX mutations contribute to the local accumulation of mineralization inhibitors such as OPN (osteopontin) and ASARM (the acidic serine- and aspartate-rich motif) peptides at the level of the extracellular matrix [12]. Likewise, the certain genetic mutation emphasizes the importance of FGF23 and DMP1 in the pathogenesis of ADHR and ARHR, respectively [5, 6]. As the same result of phosphate wasting, it is found that different types of HR cause similar, though not identical, clinical and radiographical parameters, which mostly depend on the duration of hypophosphatemia [1].
Early diagnosis of HR is of great significance, since early treatment promotes growth, reduces bone pain and improves dental health [13, 14]. The diagnosis of HR is on the basis of combination of clinical, radiological and biochemical findings. Clinically, patients of HR feature leg bowing, delayed walking, waddling gait, bone/joint pain, short stature and dental abscesses [2]. Especially, any form of leg bowing and widening of the metaphysis acquire a further radiological and biomedical examination [15]. Radiographic manifestations show the long bone deformities and abnormal growth plates with widened and frayed metaphyses [15]. Biomedical criteria for the diagnosis of HR includes serum phosphate below the normal threshold for age, elevated alkaline phosphatase, normal or upper normal parathyroid hormone levels, normal serum calcium, and low urinary calcium excretion [16]. In addition, a family history of the disease is benefit for diagnosis due to an X-linked dominant inheritance pattern of PHEX mutation. Genetic analysis, which identifies mutations in the PHEX gene in about 70% of patients with HR, is the final confirmation [15]. If genetic analysis is not available, elevated plasma levels of intact FGF23 supports the diagnosis [17].
Treatment initiation is recommended as soon as diagnosis is established [17]. Conventional treatment with oral phosphate supplements and active vitamin D (calcitriol or alfacalcidol), has been proposed for decades [3, 18, 19]. This kind of therapy has been shown to result in the positive outcomes of decrease in bone pain, normalization of alkaline phosphatase level, increase in growth velocity, straightening of legs and improving dental mineralization [3, 13, 20, 21]. However, due to the limitations, such as increasing risk of nephrocalcinosis, urolithiasis or hyperparathyroidism, the dose of phosphate supplements and vitamin D analogues should be strictly controlled according to individual situation [22, 23]. Recently, a novel therapy with burosumab, a kind of humanized monoclonal anti-FGF23 antibody, has been approved for the treatment of XLHR patients over 1 year of age in Europe and US, with the function of increasing renal phosphate reabsorption and normalizing serum phosphate [15, 24]. Nevertheless, all of those therapies focus on regulation of systemic serum phosphate, the defects in mineral quality and quantity during formation of mineralized tissues such as tooth and bone due to the local dysfunction of PHEX intend to be treated [11]. Given that, the disease follow-up and treatment management are essential.
For some patients, dental manifestations might be the first symptom diagnosed [25]. Furthermore, early prophylaxis and treatment can improve dental status [14]. Hence, broadening knowledge of various dental manifestations of HR is required to provide timely treatment options, which does help to improve the quality of HR patients’ life [26]. This article aimed at reporting a case of male with HR, reviewing the dental manifestations and treatment of HR patients and providing suggestions of dental management.
Case report
A 32-year-old male was referred to the Department of Prosthodontics with the complaint of “several abscesses that appeared in the mouth”. The patient was diagnosed with HR at the age of 2, but his parents didn’t have metabolic disorder. Since been diagnosed, he was treated with oral phosphate supplementation, calcitriol and vitamin D2. In addition, he was undergoing pharmaceutical therapy with burosumab at a dose of 60 mg/month by hypodermic injection at the age of 31.
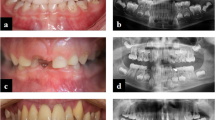
On physical examination, the patient showed short stature, bow legs, achilles tendinitis and mild hearing loss. The intraoral examination revealed multiple fistulas and abscesses at the periapical regions of maxillary right lateral incisor, mandibular left lateral incisor and first molar, and mandibular right central incisor (Fig. 1a–e). The pulpal vitality test was negative at maxillary right lateral incisor, maxillary right canine and second molar, mandibular left canine, mandibular left lateral incisor and first molar, and mandibular right central incisor and third molar. Periodontal examination revealed the absence of many teeth, and general periodontal disease resulting in some teeth increased probing pocket depth of 10 mm or more. Radiographic examination in Fig. 1f showed poorly defined lamina dura and enlarged pulp chambers and radicular canals. Radiolucencies were detected at periapical regions of maxillary right lateral incisor, maxillary right canine and second molar, mandibular left lateral incisor and canine, mandibular left lateral incisor, mandibular left second and third molar, mandibular right central and lateral incisor, and mandibular right canine and third molar. Several teeth (i.e., maxillary right lateral incisor and canine, maxillary left canine, and mandibular left lateral incisor) had received endodontic treatment before.
Intraoral views (a–e) showed multiple fistulas and abscesses, malocclusion, and dental attrition in the left lower incisors; Panoramic radiograph (f) revealed enlarged pulp chambers, the radiolucencies at periapical regions of teeth # 12, 13, 17, 22, 23, 32, 36, 37, 41, 42, 43 and 48, and the presence of endodontically treated teeth # 12, 13, 23 and 32.
Literature search
A literature search was carried out to identify all cases of HR associated dental manifestations. The PubMed database was searched until 10 March 2020 with the following MESH term: “Rickets, Hypophosphatemic” and a keyword: “dental”. It was supplemented by manual searches in the reference lists of relevant articles. The search retrieved 198 results. Articles and reports including indexed reviews, case series and case reports published in English and in peer-reviewed journals were considered, and restricted to human studies. After screening the title and abstract, 43 relevant full-length articles were included. This search identified about 416 reported cases [12, 14, 21, 25, 27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65], as presented in Table 1.
Dental manifestation
Clinically, recurrent spontaneous dental abscesses both in primary and permanent dentition without carries, periodontal problems, traumatic injuries or restorations are common findings in HR patients [12, 14, 21, 25, 27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44]. Meanwhile, with the increasing age, the number of endodontically affected teeth significantly rises, and the incisors and canines usually get affected prior to molars and premolars [45]. HR patients are more prone to suffer periodontal bone loss than the general population, while the percentage of BOP seems to be similar without commensurate increase [33, 39, 41, 46,47,48]. Another main dental finding is the smaller dental arches which always accompany with crowded dentition and class III occlusion [29, 46, 49, 50]. Other infrequent dental findings are ectopic permanent canines [34], delayed eruption of both primary and permanent teeth [39], delayed dental development [41, 51] and dental hypersensitivity [52].
Radiologically, both primary and permanent teeth exhibit enlarged pulp chambers with high pulp horns which sometimes extend up to or beyond the dentino-enamel junction [12, 21, 27, 29, 30, 32,33,34, 37, 39,40,41,42,43,44,45, 49, 52,53,54,55,56]. Zones of hypomineralized early forming coronal dentin appear as a “halo” at the dentino-enamel junction around the circumpulpal dentin [52, 55]. Hypoplastic alveolar ridge, reduced radiopacity of lamina dura and varying degree of alveolar bone loss are also radiological characteristics of HR [25, 27, 32, 41, 44, 46,47,48, 53, 54]. Other common manifestations are thinner enamel and dentin [21, 30, 40, 53], primary tooth resorption [44, 56] and short root [43, 57].
Histopathologically, hypomineralized dentin, featured by a widened predentin with fewer well-defined dentinal tubules and numerous unmerged calcospherites creating interglobular spaces, is commonly seen in teeth of HR patients, while the mantle dentin is unaffected [12, 14, 21, 30,31,32, 37, 40, 52, 53, 55,56,57,58,59,60]. The organization and polarization of odontoblasts are also impaired [66]. Enamel alternation, such as numerous crater-shaped depressions and deep microcleavages penetrating into the enamel thickness, can be seen in some patients [21, 32, 35, 37, 39, 49, 58, 61, 62]. Due to the dentin and enamel alternation, bacteria can invade easily from the oral cavity to the dental pulp, causing pulp necrosis in caries-free tooth [39]. Cementum is thinner, with roughly granular with hypomineralized interglobular patterns [48, 67].
Dental treatment
Prophylaxis
Preventing bacteria invading dentin and pulp is important to reduce dental abscesses [17]. Stainless steel crown was used to cover the crown of primary molar in the past [30, 55]. However, the limitations of large pulp chamber, high pulp horn, thin enamel and hypomineralized dentin should be taken into strict consideration. Sealing occlusal surfaces of primary and permanent teeth with composite resin is suggested, the properties of which have greatly improved over the last decade [26, 68]. It is worth noting that self-etch adhesive system is recommended to minimize risk of pulp irritation, and the sealing must be repeated every year due to gradual wear of the resin [3]. The early monitored use of topical applications of fluoride is critical for preventing subsequent serious dental infections [33, 36,37,38,39, 42]. Tooth attrition is easier to be seen in HR patients for the enamel alternation, so a nightguard acrylic splint is also suggested [53, 55].
Endodontic treatment
For the endodontically affected teeth, root canal treatment is the conventional choice, while extraction is necessary for those of abscesses spread quickly in jaw bone especially in primary teeth [3]. The procedures of endodontic treatment should try to be sterile. Using sodium hypochlorite to irrigate canals and Ca(OH)2 as intracanal medicament for a 10-day interval is recommended, and the determination of working length is preferred to combine electronic apex locators and radiograph [38]. Avoiding any voids and achieving the best possible density of the root canal filling are the goal of the obturation of the root canal system [26]. Meanwhile, the filling of root canal might be suggested using thermoplasticized obturation techniques with a virtually insoluble sealer [26]. Apical curettage procedure and apical barriers with mineral trioxide aggregate might be performed in sever abscesses and open apical foramens, respectively [28, 33].
Prosthodontic treatment
For some mild or moderate damages, adhesive procedures can be used, but in sever situation, such as dominant enamel fracturing and rapid dental wear, full coverage restoration should be chosen [68]. Regarding to the coronal restoration of endodontic treatment teeth, occlusal coverage with fitted stainless steel crowns is suggested to protect teeth from recurrent infections, however, posts cannot be supported due to the thin dentin [40, 55]. As for the ceramic crown, it is not recommended for teeth with prominent pulp horns, since the preparation costs a greater loss of dentin than metal crown [26]. Therefore, all-ceramic occlusal veneer might be another choice not only as a minimal invasive approach but for aesthetic reason [26].
Periodontal treatment
Conventional supportive periodontal therapy is of great benefit for HR patients [3]. Hence, twice-yearly visits to perform conventional supportive periodontal therapy aimed to decrease gingival inflammation and suppress periodontal pockets for adults is suggested [17].
Orthodontic treatment
Although it is confirmed that the periodontium of HR patients is less prone to orthodontic treatment [67], orthodontic treatment is not contraindicated especially for those treated with conventional systemic treatment, and could trigger extensive remodeling of the alveolar bone [68].
Implant treatment
Implants are acceptable for HR patients and several successful cases have been reported [63, 64]. However, the successful rate is declined in those who are not under conventional treatment, hence, implant surgery is recommended to be performed after 3-6 months of conventional treatment, which should be continued for 6 months following implant surgery [17]. It might be better to prolong the healing time up to 6 months to obtain a good stability [64].
Dental management
In summary, the lifetime dental management for a particular patient is as followed. (1) Once the diagnosis is established, the conventional treatment should be initiated and last a lifetime if possible [17]. (2) The dental examination should be performed twice a year regularly for adults and children, including dental orthopantomogram, of which the first time is suggested at age 5 [17, 68]. (3) Typical fluoride application and pit and fissure sealing both in primary and permanent teeth should be carried out as soon as acquired [40, 42]. (4) We recommend a thorough dental examination clinically and radiologically searching for all of the enlarged pulp chambers, periapical bone loss and pulp necrosis [17]. (5) In adults, it is suggested to perform conventional supportive periodontal therapy, including periodontal risk assessment as well as supragingival and subgingival debridement twice a year if necessary [17]. (6) If orthodontic and implant treatment are required, it must be based on the premise that conventional therapy is correctly treated [26, 63, 64].
Discussion
HR is genetic disorders, whose main symptoms are hypomineralized skeleton and dentition [12]. Dental abscesses without caries are observed most frequently in HR, even in some cases as the first symptom diagnosed [25]. In the present report (Fig. 1), the symptoms were enlarged pulp chambers and multiple dental abscesses. The cause of dental abscesses is the enamel alternation and hypomineralized dentin [39]. However, the relationship between HR and hypomineralized dentin remains unclear. In the past, hypophosphatemia was thought to be responsible for dysplastic and poorly mineralized circumpulpal dentin with wide areas of interglobular dentin, which limited growth and fusion of calcospherites [69]. Recent researches have shown that the mineralization induced by human cells is disturbed independently of hypophosphatemia and supported a local role for PHEX, DMP1 or FAM20C in matrix mineralization [11, 70, 71]. This finding might illustrate that conventional therapy improve dental complications but not prevent [35]. Due to different mechanisms of three forms of HR, the article mainly focused on XLHR. Several studies have confirmed that PHEX regulates the mineralization of the extracellular matrix at the local level in mineralized bone and dentin and maintains mineralized matrix homeostasis by cleaving of acidic, non-collagenous SIBLING proteins and peptides of the extracellular matrix such as OPN, MEPE (matrix extracellular phosphoglycoprotein) and ARARM [66, 72, 73]. Furthermore, it is suggested that OPN and MEPE inhibit tooth mineralization through different ways, with OPN acting at the mineralized calcospherites and MEPE at the region of the unmineralized interglobular dentin [66].
From the literature review (Table 1), we can find that the majority of reported patients are females, which seems to indicate a higher HR prevalence in female. However, male patients are usually shown severer dental complications, such as taurodontism [69]. Since most cases are determined by PHEX, an X-linked dominant mutant gene, it might be a gene dose effect [32]. The severity of complications might depend on three factors, including family history, medical history and age. Patients born to affected mother are inclined to bear poorer dental status for primary dentition than those born to healthy mother, due to the lower phosphate and vitamin D obtained from the affected mother during fetal odontogenesis [21]. Nevertheless, a family history contributes to an earlier diagnosis of HR, which might take an earlier treatment for patients, resulting a better outcome for permanent dentition [15]. Although conventional treatment is not able to prevent oral complications, its beneficial effects on dental and periodontal tissues cannot be underestimated [35]. It has already been confirmed that conventional treatment for HR patients improves mineralization of dentin and decreases the incidence of endodontic infection in children [14, 21]. Meanwhile, the benefit of continuing treatment for adults has been proved in permanent teeth and periodontal health recent years [48]. However, the dose of medicine should depend on the age and stage of development, for excessive phosphate might result in hyperparathyroidism [17]. The number of endodontically affected teeth raises significantly with age, due to the exposure of defective dentin [45]. Thus, only incisors or canines are affected in younger patients and affected posterior teeth are more commonly seen in older patients [45]. Similar situation is present in the case report (Fig. 1).
In order to decrease the risk of dental abscess, preventive approaches, such as topical fluoride application, pit and fissure sealing, stainless steel crown and nightguard acrylic splint are proposed for decades [30, 33, 55]. As the using of conventional steel crown requires tooth preparation which might cause the irritation of pulp, a crown conservative technique that using separating elastic and non-removal tooth structure has been recommended [74]. Resulting from the progress in bonding dentistry and the principle of sealing the wells and grooves of permanent teeth, pit and fissure sealing using fluid composite resin with self-etching bonding system might be a better choice [36]. And it is suggested that all occlusal surfaces, including principal and secondary grooves should be covered [36]. However, this preventive approach needs to be reperformed annually because of the loss or attrition of resin [3]. The self-etching bonding system is recommended because of its simple implementation and a less aggressive etching on the enamel, avoiding further damaging the cracks and irritating pulp [36].
Dental abscesses might persist in life, although patients are under conventional and preventive treatment [38]. Endodontic management is necessary to maintain a functional dentition [28]. The risk of reinfections is increased due to the altered dentin, even though there is no literature that reports a higher rate of failure in HR patients [75]. When it comes to the longstanding sinus tract and/or periradicular radiolucency after endodontic treatment, periradicular curettage might be a choice other than extraction [33]. It is still a challenge for post-endodontic coronal restorations. Some authors choose composite resin to restore the cavities for the little damage of teeth [33, 43]. However, the microleakage in the dentin-restoration interfaces might be a risk of reinfection. Metal crowns are generally recommended for restorations [68]. Meanwhile, the post might increase the risk of root fracture due to the thin dentin, which is not recommended [40]. Since the adhesive bonding to the unaltered enamel is reliable, all-ceramic occlusal veneers is advisable [26].
HR patients are susceptible to periodontitis [33, 39, 41]. Periodontitis is an inflammatory disease which is initiated by microbial plaque and leads to attachment and alveolar bone loss [47]. As reported, Dmp1-null mice develops severe periodontal defects without obvious infection or inflammation, while in human the percentage of BOP shows no difference between patients and general population, which indicates that the periodontal defects in patient are different form traditional periodontitis [47, 76]. The cause of periodontitis in HR patients is probably the reduced and hypoplastic cementum, which is sensitive to disturbances in mineral metabolism and increased attachment loss of PDL [77]. Thus, conventional management initiated from childhood and continued during adulthood can prevent the periodontal defects to some extent [48]. Decreasing occlusal loads, abandoning smoking and maintaining a good oral hygiene also play important roles in preventing periodontal defects [47].
It has been found that majority of HR patients have performed orthodontic treatment [75]. Small dental arches are commonly seen in HR patients [50]. As reported, there are significant differences in arch dimensions especially the maxillary between HR patients and general population [50]. The different degrees of craniofacial alterations bring about a class III skeletal relationship, which appear milder due to the downward and backward position of the condyles [78]. And ectopic permanent canines with crowded dentition is shown in HR patients [29, 39]. Those characteristics combined together lead a major demand for orthodontic treatment. Meanwhile, conventional treatment makes it no longer contraindicate [68]. Implant treatment is also acceptable. The successful rate might be increased after conventional treatment [17]. Prolonging the healing time and continuing conventional treatment after implant surgery can help to obtain a better stability of implant [64].
Conclusion
HR is associated with marked dental manifestations and patients with them exhibit a tendency of poorer quality of life. Early diagnosis, treatment and management are the keys to successful outcomes. It is of great essence to carry out frequent and regular dental care for HR patients. Since HR is a multisystem disease, multidisciplinary care is needed. Hence, it is important for dentists to master the knowledge of various dental manifestations and provide optimal treatment options along with other specialists in pediatric and adult fields.
References
Bitzan M, Goodyer PR. Hypophosphatemic Rickets. Pediatr Clin North Am. 2019;66:179–207.
Durmaz E, Zou M, Al-Rijjal RA, Baitei EY, Hammami S, Bircan I, et al. Novel and de novo PHEX mutations in patients with hypophosphatemic rickets. Bone. 2013;52:286–91.
Linglart A, Biosse-Duplan M, Briot K, Chaussain C, Esterle L, Guillaume-Czitrom S, et al. Therapeutic management of hypophosphatemic rickets from infancy to adulthood. Endocr Connect. 2014;3:R13–30.
Francis F, Hennig S, Korn B, Reinhardt R, de Jong P, Poustka A, et al. A gene (PEX) with homologies to endopeptidases is mutated in patients with X-linked hypophosphatemic rickets. Nat Genet. 1995;11:130–6.
White KE, Evans WE, O'Riordan JLH, Speer MC, Econs MJ, Lorenz-Depiereux B, et al. Autosomal dominant hypophosphataemic rickets is associated with mutations in FGF23. Nat Genet. 2000;26:345–8.
Feng JQ, Ward LM, Liu S, Lu Y, Xie Y, Yuan B, et al. Loss of DMP1 causes rickets and osteomalacia and identifies a role for osteocytes in mineral metabolism. Nat Genet. 2006;38:1310–5.
Levy-Litan V, Hershkovitz E, Avizov L, Leventhal N, Bercovich D, Chalifa-Caspi V, et al. Autosomal-recessive hypophosphatemic rickets is associated with an inactivation mutation in the ENPP1 gene. Am J Hum Genet. 2010;86:273–8.
Rafaelsen SH, Raeder H, Fagerheim AK, Knappskog P, Carpenter TO, Johansson S, et al. Exome sequencing reveals FAM20c mutations associated with fibroblast growth factor 23-related hypophosphatemia, dental anomalies, and ectopic calcification. J Bone Min Res. 2013;28:1378–85.
Zhao Y, Yang F, Wang L, Che H. Familial hypophosphatemic rickets caused by a PHEX gene mutation accompanied by a NPR2 missense mutation. J Pediatr Endocrinol Metab. 2020;33:305–11.
Clausmeyer S, Hesse V, Clemens PC, Engelbach M, Kreuzer M, Becker-Rose P, et al. Mutational analysis of the PHEX gene: novel point mutations and detection of large deletions by MLPA in patients with X-linked hypophosphatemic rickets. Calcif Tissue Int. 2009;85:211–20.
Coyac BR, Hoac B, Chafey P, Falgayrac G, Slimani L, Rowe PS, et al. Defective Mineralization in X-Linked Hypophosphatemia Dental Pulp Cell Cultures. J Dent Res. 2017;97:184–91.
McKee MD, Hoac B, Addison WN, Barros NM, Millan JL, Chaussain C. Extracellular matrix mineralization in periodontal tissues: Noncollagenous matrix proteins, enzymes, and relationship to hypophosphatasia and X-linked hypophosphatemia. Periodontology. 2013;63:102–22.
Makitie O, Doria A, Kooh SW, Cole WG, Daneman A, Sochett E. Early treatment improves growth and biochemical and radiographic outcome in X-linked hypophosphatemic rickets. J Clin Endocrinol Metab. 2003;88:3591–7.
Chaussain-Miller C, Sinding C, Septier D, Wolikow M, Goldberg M, Garabedian M. Dentin structure in familial hypophosphatemic rickets: Benefits of vitamin D and phosphate treatment. Oral Dis. 2007;13:482–9.
Rothenbuhler A, Schnabel D, Hogler W, Linglart A. Diagnosis, treatment-monitoring and follow-up of children and adolescents with X-linked hypophosphatemia (XLH). Metabolism. 2019;103s:153892.
Emma F, Cappa M, Antoniazzi F, Bianchi ML, Chiodini I, Eller Vainicher C, et al. X-linked hypophosphatemic rickets: an Italian experts’ opinion survey. Pediatr Nephrol. 2019;45:67.
Haffner D, Emma F, Eastwood DM, Duplan MB, Bacchetta J, Schnabel D, et al. Clinical practice recommendations for the diagnosis and management of X-linked hypophosphataemia. Nat Rev Nephrol. 2019;15:435–55.
Ovejero D, Gafni RI, Collins MT. 1,25-Dihydroxyvitamin D as Monotherapy for XLH: Back to the Future? J Bone Min Res. 2016;31:925–8.
Thabet MA, Truchina O, Chan JC. X-linked hypophosphatemia: Molecular biology and treatment controversies. Acta Paediatrica Sin. 1994;35:180–7.
Rasmussen H, Pechet M, Anast C, Mazur A, Gertner J, Broadus A. Long-term treatment of familial hypophosphatemic rickets with oral phosphate and 1 alpha-hydroxyvitamin D3. J Pediatr. 1981;99:16–25.
Chaussain-Miller C, Sinding C, Wolikow M, Lasfargues JJ, Godeau G, Garabedian M. Dental abnormalities in patients with familial hypophosphatemic vitamin D-resistant rickets: prevention by early treatment with 1-hydroxyvitamin D. J Pediatr. 2003;142:324–31.
Mäkitie O, Kooh S, Sochett E. Prolonged high-dose phosphate treatment: a risk factor for tertiary hyperparathyroidism in X-linked hypophosphatemic rickets. Clin Endocrinol. 2003;58:163–8.
Taylor A, Sherman N, Norman M. Nephrocalcinosis in X-linked hypophosphatemia: Effect of treatment versus disease. Pediatr Nephrol. 1995;9:173–5.
Insogna KL, Briot K, Imel EA, Kamenický P, Ruppe MD, Portale AA, et al. A randomized, double-blind, placebo-controlled, phase 3 trial evaluating the efficacy of Burosumab, an Anti-FGF23 Antibody, in Adults With X-Linked Hypophosphatemia: Week 24 Primary Analysis. J Bone Mi Res. 2018;33:1383–93.
Souza AP, Kobayashi TY, Lourenco Neto N, Silva SM, Machado MA, Oliveira TM. Dental manifestations of patient with vitamin D-resistant rickets. J Appl Oral Sci. 2013;21:601–6.
Sabandal MM, Robotta P, Burklein S, Schafer E. Review of the dental implications of X-linked hypophosphataemic rickets (XLHR). Clin Oral Investig. 2015;19:759–68.
Sauk JJ Jr, Witkop CJ Jr. Electron optic analysis of human dentin in hypophosphatemic vitamin D-resistant rickets (report of a kindred with consanguinity). J Oral Pathol. 1973;2:203–14.
Cohen S, Becker GL. Origin, diagnosis, and treatment of the dental manifestations of vitamin D-resistant rickets: review of the literature and report of case. J Am Dent Assoc. 1976;92:120–9.
Ainley JE Jr. Manifestations of familial hypophosphatemia. J Endod. 1978;4:26–8.
Rakocz M, Keating JR, Johnson R. Management of the primary dentition in vitamin D-resistant rickets. Oral Surg Oral Med Oral Pathol. 1982;54:166–71.
Chadwick BL, Aldred MJ. An unusual giant cell lesion in a child with vitamin D-resistant rickets. Int J Paediatr Dent. 1992;2:41–5.
Goodman JR, Gelbier MJ, Bennett JH, Winter GB. Dental problems associated with hypophosphataemic vitamin D resistant rickets. Int J Paediatr Dent. 1998;8:19–28.
Pereira CM, de Andrade CR, Vargas PA, Coletta RD, de Almeida OP, Lopes MA. Dental alterations associated with X-linked hypophosphatemic rickets. J Endod. 2004;30:241–5.
Batra P, Tejani Z, Mars M. X-linked hypophosphatemia: dental and histologic findings. J Can Dent Assoc. 2006;72:69–72.
Baroncelli GI, Angiolini M, Ninni E, Galli V, Saggese R, Giuca MR. Prevalence and pathogenesis of dental and periodontal lesions in children with X-linked hypophosphatemic rickets. Eur J Paediatr Dent. 2006;7:61–6.
Douyere D, Joseph C, Gaucher C, Chaussain C, Courson F. Familial hypophosphatemic vitamin D-resistant rickets-prevention of spontaneous dental abscesses on primary teeth: a case report. Oral Surg Oral Med Oral Pathol Oral Radio Endod. 2009;107:525–30.
Souza MA, Soares Junior LA, Santos MA, Vaisbich MH. Dental abnormalities and oral health in patients with Hypophosphatemic rickets. Clin (Sao Paulo). 2010;65:1023–6.
Beltes C, Zachou E. Endodontic management in a patient with vitamin D-resistant Rickets. J Endod. 2012;38:255–8.
Rabbani A, Rahmani P, Ziaee V, Ghodoosi S. Dental problems in hypophosphatemic rickets, a cross sectional study. Iran J Pediatr. 2012;22:531–4.
Cremonesi I, Nucci C, D’Alessandro G, Alkhamis N, Marchionni S, Piana G. X-linked hypophosphatemic rickets: Enamel abnormalities and oral clinical findings. Scanning. 2014;36:456–61.
Ayesha Thabusum D, Stinton NM, Uston KA, Davis CD. Hypophosphatemic rickets and pre-eruptive spontaneous dental abscess. Case Rep Dent. 2016;83:46–50.
Paredes SEY, Segato RAB, Moreira LD, Moreira A, Serrano K, Rodrigues CT, et al. Dentoalveolar abscesses not associated with caries or trauma: A diagnostic hallmark of hypophosphatemic rickets initially misdiagnosed as hypochondroplasia. Head Neck Pathol. 2017;12:604–9.
Murayama T, Iwatsubo R, Akiyama S, Amano A, Morisaki I. Familial hypophosphatemic vitamin D-resistant rickets: dental findings and histologic study of teeth. Oral Surg Oral Med Oral Pathol Oral Radio Endod. 2000;90:310–6.
Soares EC, Costa FW, Ribeiro TR, Alves AP, Fonteles CS. Clinical approach in familial hypophosphatemic rickets: report of three generations. Spec Care Dent. 2013;33:304–7.
Andersen MG, Beck-Nielsen SS, Haubek D, Hintze H, Gjorup H, Poulsen S. Periapical and endodontic status of permanent teeth in patients with hypophosphatemic rickets. J Oral Rehabil. 2012;39:144–50.
Al-Jundi SH, Hammad MM, Dabous I. Case report: hypophosphatemic rickets and aggressive periodontitis: a review of the role of dentine matrix protein 1 in the pathogenesis. Eur Arch Paediatr Dent. 2011;12:46–50.
Ye L, Liu R, White N, Alon US, Cobb CM. Periodontal status of patients with hypophosphatemic rickets: a case series. J Periodontol. 2011;82:1530–5.
Biosse Duplan M, Coyac BR, Bardet C, Zadikian C, Rothenbuhler A, Kamenicky P, et al. Phosphate and Vitamin D Prevent Periodontitis in X-Linked Hypophosphatemia. J Dent Res. 2016;96:388–95.
Schwartz S, Scriver CR, Reade TM, Shields ED. Oral findings in patients with autosomal dominant hypophosphatemic bone disease and X-linked hypophosphatemia: further evidence that they are different diseases. Oral Surg Oral Med Oral Pathol. 1988;66:310–4.
Al-Jundi SH, Al-Naimy YF, Alsweedan S. Dental arch dimensions in children with hypophosphataemic Vitamin D resistant rickets. Eur Arch Paediatr Dent. 2010;11:83–7.
Sh AJ, Am H. Dental Development in Patients with Hypophosphatemia Rickets. Int J Clin Pediatr Dent. 2010;3:1–4.
Hintze H, Wenzel A, Kruhoffer F. Dental hypersensitivity due to hypophosphataemia? Dentomaxillofac Radio. 1990;19:81–3.
Bender IB, Naidorf IJ. Dental observations in vitamin D-resistant rickets with special reference to periapical lesions. J Endod. 1985;11:514–20.
Gigliotti R, Harrison H, Reveley RA, Drabkowski AJ. Familial vitamin D-refractory rickets. J Am Dent Assoc. 1971;82:383–7.
Alexander S, Moloney L, Kilpatrick N. Endodontic management of a patient with X-linked hypophosphataemic rickets. Aust Endod J. 2001;27:57–61.
Rathore R, Nalawade TM, Pateel D, Mallikarjuna R. Oral manifestations of vitamin D resistant rickets in orthopantomogram. BMJ Case Rep. 2013;2013:bcr2012008318.
Hillmann G, Geurtsen W. Pathohistology of undecalcified primary teeth in vitamin D-resistant rickets: review and report of two cases. Oral Surg Oral Med Oral Pathol Oral Radio Endod. 1996;82:218–24.
Lyles KW, Burkes EJ Jr, McNamara CR, Harrelson JM, Pickett JP, Drezner MK. The concurrence of hypoparathyroidism provides new insights to the pathophysiology of X-linked hypophosphatemic rickets. J Clin Endocrinol Metab. 1985;60:711–7.
Abe K, Ooshima T, Lily TS, Yasufuku Y, Sobue S. Structural deformities of deciduous teeth in patients with hypophosphatemic vitamin D-resistant rickets. Oral Surg Oral Med Oral Pathol. 1988;65:191–8.
Daley TD, Jarvis A, Wysocki GP, Kogon SL. X-ray microanalysis of teeth from healthy patients and patients with familial hypophosphatemia. Calcif Tissue Int. 1990;47:350–5.
Nikiforuk G, Fraser D. Chemical determinants of enamel hypoplasia in children with disorders of calcium and phosphate homeostasis. J Dent Res. 1979;58:1014–5.
Yuan L, Wu S, Xu H, Xiao J, Yang Z, Xia H, et al. Identification of a novel PHEX mutation in a Chinese family with X-linked hypophosphatemic rickets using exome sequencing. Biol Chem. 2015;396:27–33.
Resnick D. Implant placement and guided tissue regeneration in a patient with congenital vitamin D-resistant rickets. J Oral Implantol. 1998;24:214–8.
Friberg B. Branemark system implants and rare disorders: a report of six cases. Int J Periodontics Restor Dent. 2013;33:139–48.
Shields ED, Scriver CR, Reade T, Fujiwara TM, Morgan K, Ciampi A, et al. X-linked hypophosphatemia: the mutant gene is expressed in teeth as well as in kidney. Am. J. Hum. Genet. 1990;46:434–42.
Salmon B, Bardet C, Coyac BR, Baroukh B, Naji J, Rowe PS, et al. Abnormal osteopontin and matrix extracellular phosphoglycoprotein localization, and odontoblast differentiation, in X-linked hypophosphatemic teeth. Connect Tissue Res. 2014;55:79–82.
Coyac BR, Falgayrac G, Baroukh B, Slimani L, Sadoine J, Penel G, et al. Tissue-specific mineralization defects in the periodontium of the Hyp mouse model of X-linked hypophosphatemia. Bone. 2017;103:334–46.
Opsahl Vital S, Gaucher C, Bardet C, Rowe PS, George A, Linglart A, et al. Tooth dentin defects reflect genetic disorders affecting bone mineralization. Bone. 2012;50:989–97.
Connor J, Olear EA, Insogna KL, Katz L, Baker S, Kaur R, et al. Conventional Therapy in Adults With X-Linked Hypophosphatemia: Effects on enthesopathy and dental disease. J Clin Endocrinol Metab. 2015;100:3625–32.
Wang X, Wang J, Liu Y, Yuan B, Ruest LB, Feng JQ, et al. The specific role of FAM20C in dentinogenesis. J Dent Res. 2015;94:330–6.
Rangiani A, Cao ZG, Liu Y, Voisey Rodgers A, Jiang Y, Qin CL, et al. Dentin matrix protein 1 and phosphate homeostasis are critical for postnatal pulp, dentin and enamel formation. Int J Oral Sci. 2012;4:189–95.
Boukpessi T, Hoac B, Coyac BR, Leger T, Garcia C, Wicart P, et al. Osteopontin and the dento-osseous pathobiology of X-linked hypophosphatemia. Bone. 2017;95:151–61.
Coyac BR, Falgayrac G, Penel G, Schmitt A, Schinke T, Linglart A, et al. Impaired mineral quality in dentin in X-linked hypophosphatemia. Connect Tissue Res. 2018;59:91–6.
Seow WK. Diagnosis and management of unusual dental abscesses in children. Aust Dent J. 2003;48:156–68.
Hanisch M, Bohner L, Sabandal MMI, Kleinheinz J, Jung S. Oral symptoms and oral health-related quality of life of individuals with x-linked hypophosphatemia. Head Face Med. 2019;15:8.
Ye L, Zhang S, Ke H, Bonewald LF, Feng JQ. Periodontal breakdown in the Dmp1 null mouse model of hypophosphatemic rickets. J Dent Res. 2008;87:624–9.
Zhang H, Chavez MB, Kolli TN, Tan MH, Fong H, Chu EY, et al. Dentoalveolar defects in the Hyp Mouse Model of X-linked Hypophosphatemia. Physiol Rep. 2020;99:419–28.
Al-Jundi SH, Dabous IM, Al-Jamal GA. Craniofacial morphology in patients with hypophosphataemic vitamin-D-resistant rickets: A cephalometric study. J Oral Rehabil. 2009;36:483–90.
Acknowledgements
This work was supported by National Natural Science Foundation of China (81970982). XJ and YX contributed equally to this work and should be considered as co-first authors. BF and LZ contributed equally to this work and should be considered as co-corresponding authors.
Author information
Authors and Affiliations
Contributions
BF and LZ coordinated the project. XJ and YX performed the basic literature research and drafted the first version of the manuscript. WL, ZS, YS, and XP added further literature identified by manual search. BF, LZ, XJ, YX, WL, ZS, YS, and XP discussed the successive versions.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no conflict of interest. The patient involved in the study have signed a written informed consent that the health-related data would be used for research purposes and photographs would be published in this way. This study was approved by the Institutional Ethic Board of the Affiliated Stomatology Hospital, Zhejiang University School of Medicine, Hangzhou, China (No. 2020-16).
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jin, X., Xu, Y., Liu, W. et al. Dental manifestations and treatment of hypophosphatemic rickets: A case report and review of literature. BDJ Open 9, 2 (2023). https://doi.org/10.1038/s41405-023-00129-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41405-023-00129-9
- Springer Nature Limited
This article is cited by
-
Characteristics of oral health of patients with X-linked hypophosphatemia: case reports and literature review
BDJ Open (2024)
-
Ce-MOFs Fluorescent Probes with Different Ligands for Phosphate Detection
Journal of Analysis and Testing (2024)