Abstract
In the MASTER study (NCT03224507), daratumumab+carfilzomib/lenalidomide/dexamethasone (D-KRd) demonstrated promising efficacy in transplant-eligible newly diagnosed multiple myeloma (NDMM). In GRIFFIN (NCT02874742), daratumumab+lenalidomide/bortezomib/dexamethasone (D-RVd) improved outcomes for transplant-eligible NDMM. Here, we present a post hoc analysis of patients with high-risk cytogenetic abnormalities (HRCAs; del[17p], t[4;14], t[14;16], t[14;20], or gain/amp[1q21]). Among 123 D-KRd patients, 43.1%, 37.4%, and 19.5% had 0, 1, or ≥2 HRCAs. Among 120 D-RVd patients, 55.8%, 28.3%, and 10.8% had 0, 1, or ≥2 HRCAs. Rates of complete response or better (best on study) for 0, 1, or ≥2 HRCAs were 90.6%, 89.1%, and 70.8% for D-KRd, and 90.9%, 78.8%, and 61.5% for D-RVd. At median follow-up (MASTER, 31.1 months; GRIFFIN, 49.6 months for randomized patients/59.5 months for safety run-in patients), MRD-negativity rates as assessed by next-generation sequencing (10–5) were 80.0%, 86.4%, and 83.3% for 0, 1, or ≥2 HRCAs for D-KRd, and 76.1%, 55.9%, and 61.5% for D-RVd. PFS was similar between studies and superior for 0 or 1 versus ≥2 HRCAs: 36-month PFS rates for D-KRd were 89.9%, 86.2%, and 52.4%, and 96.7%, 90.5%, and 53.5% for D-RVd. These data support the use of daratumumab-containing regimens for transplant-eligible NDMM with HCRAs; however, additional strategies are needed for ultra-high–risk disease (≥2 HRCAs).

Video Abstract
Similar content being viewed by others
Introduction
Daratumumab is a human IgGκ monoclonal antibody targeting CD38 with a direct on-tumor [1,2,3,4] and immunomodulatory [5,6,7] mechanism of action, demonstrating greater cytotoxicity toward multiple myeloma (MM) cells ex vivo compared with analogs of other CD38 antibodies [8]. Daratumumab is approved as a monotherapy and in combination with standard-of-care regimens for patients with relapsed or refractory MM and a part of combination therapy for patients with newly diagnosed MM (NDMM) [9].
Induction therapy with a triplet regimen consisting of a proteasome inhibitor, immunomodulatory drug, and dexamethasone is standard of care for transplant-eligible patients with NDMM [10]. Recent studies have examined the addition of daratumumab to these triplet regimens. The single-arm, phase 2 MASTER study (ClinicalTrials.gov Identifier: NCT03224507) evaluated daratumumab plus carfilzomib, lenalidomide, and dexamethasone (D-KRd) and demonstrated promising clinical efficacy in transplant-eligible patients with NDMM [11]. The primary analysis (median follow-up, 25.1 months) showed that minimal residual disease (MRD) negativity at the 10–5 threshold was achieved by 80% of D-KRd patients, as determined at the end of MRD-directed treatment [11]. The final analysis of MASTER was recently reported, and data continued to demonstrate a benefit of D-KRd in this setting in the overall population and suggested a possible pathway for treatment cessation among responding patients exhibiting sustained MRD negativity [12]. The randomized, phase 2 GRIFFIN study (ClinicalTrials.gov Identifier: NCT02874742) evaluated daratumumab plus lenalidomide, bortezomib, and dexamethasone (D-RVd) or lenalidomide, bortezomib, and dexamethasone (RVd) alone in transplant-eligible patients with NDMM [13]. The primary analysis (median follow-up, 13.5 months) showed that the rate of stringent complete response (CR) by the end of post-autologous stem cell transplant (ASCT) consolidation was significantly higher for D-RVd versus RVd (42.4% vs 32.0%; 1-sided p = 0.068, which met the pre-specified 1-sided α of 0.10) [13]. At the time of GRIFFIN final analysis, which occurred after all patients completed ≥1 year of long-term follow-up after the end of study treatment, death, or withdrawal (median follow-up, 49.6 months), responses continued to deepen over time for D-RVd versus RVd, and there was an improvement in progression-free survival (PFS) for the D-RVd group versus the RVd group (hazard ratio, 0.45; 95% confidence interval, 0.21–0.95; p = 0.032) [14]. The safety profiles of D-KRd and D-RVd were previously reported [11, 13]. No unexpected safety concerns occurred for these daratumumab-based quadruplet therapies, and adverse events in each regimen were consistent with previous reports of the individual regimen components.
Patients with MM may have high-risk disease characteristics, such as the presence of extramedullary disease, International Staging System stage III disease, advanced age, and/or the presence of high-risk cytogenetic abnormalities (HRCAs) [15,16,17]. These high-risk disease characteristics are associated with a poor overall prognosis and shorter survival, and patients with high-risk features constitute a population with high unmet medical need [15, 18, 19]. The consensus from the International Myeloma Working Group (IMWG) advises that cytogenetic risk should be evaluated using bone marrow aspirate–based fluorescence in situ hybridization panels for t(4;14), del(17p), and t(14;16), with an extended panel for clinical trials that includes t(11;14), t(14;20), gain(1q), del(1p), del(13q), and ploidy status [18]. While risk stratification is important for understanding overall prognosis, much remains to be learned about how HRCAs impact clinical outcomes and influence optimal therapy selection and treatment sequencing. The objective of this study is to better understand clinical outcomes for daratumumab-based treatment among patients with NDMM by HRCA risk stratifications, according to a revised definition inclusive of the cytogenetic abnormalities del(17p), t(4;14), t(14;16), t(14;20), and/or gain/amp(1q21) (≥3 copies of chromosome 1q21). Here, we present a post hoc analysis of side-by-side results including patients from MASTER (D-KRd) and GRIFFIN (D-RVd) with 0, 1, or ≥2 HRCAs, noting the goal is not to compare D-KRd and D-RVd but rather to evaluate the overall value of frontline daratumumab-based therapy.
Methods
Patients and study design
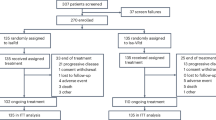
The full details of the MASTER (ClinicalTrials.gov Identifier: NCT03224507) [11] and GRIFFIN (ClinicalTrials.gov Identifier: NCT02874742) [13] studies have been previously reported. Briefly, in the multicenter, single-arm, phase 2 MASTER study, D-KRd was evaluated in transplant-eligible patients with NDMM. Patients had no upper age limit and an Eastern Cooperative Oncology Group performance status score of ≤2. Patients received up to 4 D-KRd induction cycles; high-dose therapy and ASCT; and up to 2 phases of D-KRd consolidation therapy (Cycles 5–8 and 9–12). MRD assessments in MASTER occurred post-induction, post-ASCT, and after each consolidation phase, and patients achieving 2 consecutive MRD negative (10–5) assessments transitioned to treatment-free observation. Patients who completed consolidation without 2 consecutive MRD-negative assessments transitioned to lenalidomide maintenance. The study design included enrichment for patients with MM harboring HRCAs to meet the criteria that ≥35% of participants would have t(4;14), t(14;16), and/or del(17p). In both studies, cytogenetic risk was assessed at baseline by fluorescence in situ hybridization via local testing.
In the multicenter, randomized, open-label, phase 2 GRIFFIN study, D-RVd was evaluated versus RVd alone in transplant-eligible patients with NDMM. Patients were 18–70 years of age and had an Eastern Cooperative Oncology Group performance status score of ≤2. Prior to the randomized phase of GRIFFIN, a safety run-in was conducted in 16 patients to assess D-RVd dose-limiting toxicities [20]. Following completion of the safety run-in, the study proceeded to the randomization phase in which patients were randomized 1:1 to the D-RVd or the RVd group. In this phase, patients received 4 D-RVd or RVd induction cycles, followed by high-dose therapy and ASCT, then 2 D-RVd or RVd consolidation cycles, followed by up to 2 years of maintenance therapy consisting of daratumumab plus lenalidomide or lenalidomide alone. Patients in the safety run-in phase of GRIFFIN received the same treatment as patients in the randomized phase in the D-RVd group. MRD negativity was measured at baseline, at first evidence of suspected CR or stringent CR (including patients with very good partial response or better and suspected daratumumab interference), after induction therapy, at the post-transplant consolidation disease evaluation, and after 1 and 2 years of maintenance therapy.
For both studies, the protocols and appropriate related documents were approved by the institutional review board or independent ethics committee at each participating site, and all patients gave written informed consent. The studies were conducted in accordance with the International Conference on Harmonisation Good Clinical Practice guidelines, the principles originating from the Declaration of Helsinki, as well as study site–specific regulations. The MASTER study followed the University of Alabama at Birmingham O’Neal Comprehensive Cancer Center Data and Safety Monitoring Plan. Each study established an independent Data Monitoring Committee for oversight for study conduct.
Endpoints, objectives, and analyses
The primary endpoint of the MASTER study was the achievement of MRD negativity at any time during therapy and was previously reported [11]. The primary endpoint of GRIFFIN was the stringent CR rate by the end of post-ASCT consolidation treatment and was also previously published [13]. Both studies also assessed additional endpoints, including response rates, MRD-negativity rates (minimum sensitivity threshold of 1 in 100,000 cells [10−5]), and PFS. In GRIFFIN, response to study treatment and PFS were evaluated using a validated computer algorithm in alignment with IMWG criteria [21, 22]. Patients were considered MRD positive if the MRD assessment was positive, indeterminate, or unavailable. Best response on study for MASTER was also evaluated using IMWG criteria.
The objective of this post hoc analysis was to evaluate the clinical efficacy of the daratumumab-based quadruplet therapies D-KRd (from MASTER) and D-RVd (from GRIFFIN) in patients with NDMM with HRCAs, defined as having ≥1 of the following genetic abnormalities: del(17p), t(4;14), t(14;16), t(14;20), and/or gain/amp(1q21) (≥3 copies of chromosome 1q21). Cytogenetic abnormalities (fluorescence in situ hybridization [FISH]) were assessed by the local labs, normally accessed at the study sites, on bone marrow aspirates in both MASTER and GRIFFIN. Patients with evaluable data were grouped into standard risk, high risk, or ultra-high risk based on the presence of 0, 1, or ≥2 HRCAs, respectively. In addition, MRD-negativity rates were also presented for patients achieving ≥CR in each cytogenetic subgroup. This study descriptively presents the results for the D-KRd and D-RVd groups side by side, and thus no statistical or treatment comparisons between the 2 groups were performed. Kaplan–Meier plots and estimates of PFS were provided for each HRCA group in each study.
Results
Patient characteristics
A total of 123 patients with NDMM were enrolled in the MASTER study; most patients had ≥1 HRCA (1 HRCA, 37.4%, n = 46; ≥2 HRCAs, 19.5%, n = 24) and the remainder had 0 HRCA (43.1%, n = 53). There was no difference in the median duration of study treatment for D-KRd induction, ASCT, and consolidation among patients with NDMM with 0, 1, or ≥2 HRCAs, which was 11.5, 11.5, and 11.7 months, respectively (Table 1). Among 120 patients with NDMM in GRIFFIN who received D-RVd therapy (n = 104 randomized phase and n = 16 safety run-in), most had 0 HRCA (55.8%, n = 67) or 1 HRCA (28.3%, n = 34), and a smaller number of patients had ≥2 HRCAs (10.8%, n = 13). Six (5.0%) GRIFFIN patients were not evaluable for cytogenetic abnormalities because cytogenetic testing was not done or data were not captured; therefore, these patients were not included in this analysis. The median duration of study treatment for D-RVd induction, ASCT, and consolidation was 8.1, 8.1, and 7.4 months among patients with NDMM with 0, 1, or ≥2 HRCAs, respectively, and the median duration of study maintenance therapy was 24.4, 24.2, and 23.9 months for patients with 0, 1, or ≥2 HRCAs, respectively (Table 2). In both MASTER and GRIFFIN, the median age of patients was 60 years. Among patients with ≥2 HRCAs, a relatively high proportion had International Staging System stage III disease (45.8% [n = 11] of D-KRd patients and 30.8% [n = 4] of D-RVd patients) or extramedullary disease (7.7% [n = 1] of D-RVd; data on extramedullary disease were not available in MASTER).
Efficacy
In an analysis of best response at any time point during the study, rates of ≥CR were highest for patients with 0 or 1 HRCA in both GRIFFIN and MASTER (Fig. 1). In MASTER, rates of ≥CR among D-KRd patients were 90.6%, 89.1%, and 70.8% for 0, 1, or ≥2 HRCAs, respectively. In GRIFFIN, rates of ≥CR among D-RVd patients were 90.9%, 78.8%, and 61.5% for 0, 1, or ≥2 HRCAs.
Rates of ≥CR were assessed based on International Uniform Response Criteria Consensus Recommendations, and percentages were calculated with the number of patients in the treatment group as the denominator. ≥CR complete response or better, D-KRd daratumumab plus carfilzomib/lenalidomide/dexamethasone, D-RVd daratumumab plus lenalidomide/bortezomib/dexamethasone, HRCA high-risk cytogenetic abnormality. *HRCAs include any of the following genetic abnormalities: del(17p), t(4;14), t(14;16), t(14;20), and gain/amp(1q21) (≥3 copies of chromosome 1q21). Patients were grouped into categories: standard risk (0 HRCA), high risk (1 HRCA), or ultra-high risk (≥2 HRCAs). †Evaluable patients in MASTER included all enrolled patients (0 HRCA, n = 53; 1 HRCA, n = 46; ≥2 HRCAs, n = 24). ‡Evaluable patients in GRIFFIN were the response-evaluable population (0 HRCA, n = 66; 1 HRCA, n = 33; ≥2 HRCAs, n = 13).
MRD-negativity (both 10–5 and 10–6) rates were generally similar for D-KRd across patients with 0, 1, or ≥2 HRCAs, but were highest for D-KRd in patients with 1 HRCA (Table 3). In MASTER, MRD-negativity rates following D-KRd were 80.0%, 86.4%, and 83.3% at the 10–5 threshold for 0, 1, or ≥2 HRCAs, respectively, and 68.0%, 79.5%, and 66.7% at the 10–6 threshold. In D-KRd patients who achieved ≥CR, MRD-negativity (10–5) rates were 84.4%, 89.7%, and 94.1% for 0, 1, or ≥2 HRCAs, respectively. Rates of sustained MRD negativity (10–5) lasting ≥12 months were 64.0%, 72.7%, and 50.0% among D-KRd patients with 0, 1, or ≥2 HRCAs. Median time to MRD negativity (10–5) was similar across HRCA groups (0 HRCA, 7.5 months; 1 HRCA, 7.1 months; ≥2 HRCAs, 7.6 months). In GRIFFIN, MRD-negativity (10–5) rates in D-RVd patients were 76.1%, 55.9%, and 61.5% for 0, 1, or ≥2 HRCAs, respectively. MRD-negativity (10–6) rates were higher for D-RVd patients with 0 HRCA (44.8%) and 1 HRCA (26.5%) compared with ≥2 HRCAs (15.4%). In patients who achieved ≥CR, MRD-negativity (10–5) rates in D-RVd patients were 83.3%, 69.2%, and 87.5% among patients with 0, 1, or ≥2 HRCAs, respectively. Rates of sustained MRD negativity (10–5) lasting ≥12 months were 53.7%, 38.2%, and 30.8% among D-RVd patients with 0, 1, or ≥2 HRCAs. Median time to MRD negativity (10–5) was 8.5, 8.6, and 19.6 months among D-RVd patients with 0, 1, or ≥2 HRCAs.
PFS rates in MASTER and GRIFFIN were superior for patients with 0 or 1 HRCA compared with ≥2 HRCAs (Fig. 2). In MASTER, at a median follow-up of 31.1 months, estimated 24-month PFS rates for D-KRd patients were 92.4%, 95.7%, and 65.5% for 0, 1, or ≥2 HRCAs, respectively, and estimated 36-month rates were 89.9%, 86.2%, and 52.4%. In GRIFFIN, PFS analyses were conducted for D-RVd patients in a combined analysis of randomized patients (median follow-up at final analysis among all randomized patients, 49.6 months) and among patients from the safety run-in phase (median follow-up, 59.5 months). Estimated 36-month rates for all D-RVd patients were 96.7%, 90.5%, and 53.5% for 0, 1, or ≥2 HRCAs, respectively, and estimated 48-month PFS rates were 93.7%, 90.5%, and 53.5%.
Kaplan–Meier estimates of PFS among patients in the ITT population by cytogenetic risk status (0 HRCA, 1 HRCA, or ≥2 HRCAs) are shown for (A) MASTER and (B) GRIFFIN. Median PFS was not reached for any group. PFS progression-free survival, D-KRd daratumumab plus carfilzomib/lenalidomide/dexamethasone, D-RVd daratumumab plus lenalidomide/bortezomib/dexamethasone, HRCA high-risk cytogenetic abnormality. *HRCAs include any of the following genetic abnormalities: del(17p), t(4;14), t(14;16), t(14;20), and gain/amp(1q21) (≥3 copies of chromosome 1q21). Patients were grouped into categories: standard risk (0 HRCA), high risk (1 HRCA), or ultra-high risk (≥2 HRCAs).
In MASTER, 3 patients died while on D-KRd study therapy (0 HRCA, n = 2 [both from sudden death]; ≥2 HRCAs, n = 1 [metapneumovirus during transplant]). After discontinuation of D-KRd therapy and during follow-up, 2 patients died, but neither death was preceded by progressive disease (PD; 1 HRCA, n = 2 [fall and COVID-19 pneumonia]). In GRIFFIN, 8 D-RVd patients died (0 HRCA, n = 2 [due to an adverse event (bronchopneumonia) and PD]; 1 HRCA, n = 1 [due to PD]; ≥2 HRCAs, n = 4 [all due to PD]; not evaluable for cytogenetics, n = 1 [due to respiratory failure]).
Discussion
This analysis of transplant-eligible patients with NDMM by cytogenetic risk status from the MASTER and GRIFFIN studies showed that patients with high cytogenetic risk derive clinical benefit from frontline daratumumab-based quadruplet therapy. In both MASTER and GRIFFIN, patients with 0 or 1 HRCA achieved higher rates of ≥CR than patients with ≥2 HRCAs. PFS results by cytogenetic risk were similar between the MASTER and GRIFFIN studies and showed high estimated PFS rates for patients with 0 or 1 HRCA for both D-KRd and D-RVd therapy; these were superior to PFS rates for patients with ≥2 HRCAs. In general, rates of MRD negativity and durable MRD negativity were highest among patients with 0 or 1 HRCA, similar to the results for ≥CR and PFS. Together, these observations indicate that patients with standard-risk and high-risk disease (≤1 HRCA) respond well to frontline daratumumab-based quadruplet therapy, specifically to MRD-directed therapy in MASTER, and have similar outcomes. In fact, it is remarkable that the outcomes of patients with 1 HRCA in the setting of quadruplet induction/consolidation and ASCT greatly resemble the outcomes of patients with standard-risk disease. It should also be noted that, in both studies, all patients had improvement in clinical outcomes by MRD assessment following ASCT, sustaining the benefit of this portion of the treatment regimens. However, the relatively rapid progression seen among patients with >2 HRCAs in both studies indicates that innovations beyond the addition of CD38 antibody therapy are needed to improve outcomes among ultra-high−risk patients (≥2 HRCAs). In aggregate, this observation is a testament to the notion that risk is context dependent and potentially modifiable by therapy.
It has been established that MM patients with HRCAs generally have a worse prognosis, and that the prognosis of these patients varies depending on a number of factors, including the presence and the amount of specific genetic abnormalities as well as choice of therapy [18]. To this end, there is considerable interest in understanding whether emerging MM therapies provide clinical benefit among patients with HRCAs and how upfront therapy can be optimally used to target high-risk groups. Several phase 3 clinical trials of daratumumab-based regimens among patients with NDMM have shown that the addition of daratumumab improves outcomes for patients with high-risk cytogenetics compared with non–daratumumab-based standard-of-care regimens [23,24,25]. A meta-analysis including these randomized studies of daratumumab (MAIA [daratumumab plus lenalidomide and dexamethasone], ALCYONE [daratumumab plus bortezomib, melphalan, and prednisone], and CASSIOPEIA [daratumumab plus bortezomib, thalidomide, and dexamethasone]) in patients with NDMM with high cytogenetic risk further validated the outcomes reported in the individual studies. Specifically, Giri et al. demonstrated that the addition of daratumumab to standard-of-care regimens reduced the risk of disease progression or death by 33% for patients with high-risk disease versus standard of care alone (hazard ratio, 0.67; 95% confidence interval, 0.47–0.95) [26]. To be noted, the methods for the assessment of high-risk MM varied across the included studies. For ALCYONE and MAIA, high-risk patients were defined as having ≥1 of the following HRCAs at baseline: del(17p), t(4;14), and t(14;16) [23, 24]. For CASSIOPEIA, high-risk MM was defined as having del(17p) and/or t(4;14), without the inclusion of t(14;16) [25]. A pooled analysis of patient-level data from MAIA and ALCYONE, which included transplant-ineligible patients with high-risk NDMM defined as having del(17p), t(4;14), and/or t(14;16), demonstrated that, with a median follow-up of 43.7 months, daratumumab-based therapy reduced the risk of disease progression or death by 41% (adjusted hazard ratio, 0.59; 95% confidence interval, 0.41–0.85) versus the standard-of-care regimen without daratumumab (bortezomib, melphalan, and prednisone or lenalidomide and dexamethasone) [27]. In this analysis, the estimated proportion of patients who did not progress and were still alive at 36 months was 41.3% among those who received daratumumab-based therapy versus 19.9% among those who received standard of care [27]. Our present report of HRCAs from the MASTER and GRIFFIN studies includes transplant-eligible patients with NDMM who received daratumumab-based quadruplet therapy plus ASCT, and HRCAs were defined according to a newer, revised definition (≥1 of the following: del[17p], t[4;14], t[14;16], t[14;20], and/or gain/amp[1q21]).
In this analysis, we performed post hoc analyses by the number of HRCAs (0, 1, or ≥2). As noted, ≥CR rates and PFS were similar for patients with 0 or 1 HRCA and worse for patients with ≥2 HRCAs; however, this should be interpreted in the context that the groups were not stratified (eg, by revised International Staging System). Additionally, in this study, gain/amp(1q21) accounted for a relatively high proportion of patients with only 1 HRCA in both the D-KRd (52.2%) and D-RVd (76.5%) groups. This analysis does not evaluate individual HRCAs as the number of patients with any specific HRCA is very limited. Other studies have demonstrated that the number and type of HRCA can be an indicator of prognosis. An analysis of patients with NDMM in the Medical Research Council (MRC) Myeloma IX trial [28] who received cyclophosphamide, vincristine, doxorubicin, and dexamethasone or cyclophosphamide, thalidomide, and dexamethasone followed by transplant showed that the presence of ≥2 chromosomal abnormalities (defined as t[4;14], t[14;16], t[14;20], gain[1q], and/or del[17p]) was associated with more aggressive disease [29]. In this analysis, median PFS was 30.8, 21.9, and 14.4 months for patients with 0, 1, and ≥2 HRCAs, respectively [29]. Acknowledging the limitations of cross-study comparisons (eg, differences in study designs and treatment backbones), it is notable that the poor prognosis of patients with ≥2 HRCAs seen in GRIFFIN and MASTER is consistent with the findings from the MRC Myeloma IX study. Interestingly, patients with 0 and 1 HRCA who received daratumumab-based quadruplet therapy in GRIFFIN or MASTER had similar PFS outcomes, which contrasts with the MRC Myeloma IX study, where patients with 1 HRCA had better intermediate PFS outcomes than those with ≥2 HRCAs, but not as high as those with 0 HRCA. It should also be considered that PFS outcomes overall were lower in the MRC Myeloma IX study versus PFS outcomes in MASTER and GRIFFIN. Our observations from this combined MASTER and GRIFFIN analysis showed that daratumumab-based quadruplet therapy had similar efficacy among patients with 0 and 1 HRCA. Further and more in-depth investigations of the optimal use of daratumumab-based therapy in high-risk transplant-eligible patients with NDMM, based on the number and identity of specific HRCAs, are warranted. For patients with ultra-high−risk disease, promising outcomes were observed in the OPTIMUM/MUKnine study, which evaluated frontline daratumumab-containing induction and extended/intensified consolidation with transplant for patients with ≥2 of the following: t(4;14), t(14;16), t(14;20), gain(1q), del(1p), del(17p); or gene expression SKY92 (SkylineDx, Rotterdam, The Netherlands) profiling; or with primary plasma cell leukemia (circulating plasma cells >20%) [30].
It is important to note that the study designs of MASTER and GRIFFIN are different, so direct comparisons should be avoided. Neither MASTER nor GRIFFIN collected information on clone size for individual HRCAs or copy number for 1q21 abnormalities. Given the potential heterogeneity in cut-offs used to determine the presence of cytogenetic abnormalities across local laboratories, this lack of information on clone size may be a notable limitation. Per the recommendations of the European Myeloma Network [31], the appropriate cut-off level is 10% for fusion abnormalities and 20% for numerical abnormalities. Additionally, 1q21 abnormalities were assessed as a single "gain/amp(1q21)" group, without further differentiation, making it difficult to assess the prognostic value of gain 1q21 versus the more prognostic adverse amplification of 1q21. Additionally, MASTER provided enrichment for patients with HRCAs, while GRIFFIN did not. The goal of this analysis was not to compare D-KRd and D-RVd treatments, but rather to evaluate the overall value of frontline daratumumab-based treatment. The results reported here demonstrate that daratumumab-based quadruplet frontline therapy with ASCT provides clinical benefit to patients with ≤1 HRCA, but new approaches are needed for patients with ≥2 HRCAs, which is an area of significant unmet need. It will be necessary to explore the use of novel agents and treatment combinations for this group, including bispecific T-cell engagers and chimeric antigen receptor T cells. In addition, prolonged or indefinite multiagent maintenance strategies may be required to extend PFS in such patients. In summary, continued research, longer follow-up, and larger studies are needed to fully understand and identify the optimal therapies for patients with HRCAs, especially for patients with ultra-high−risk disease (≥2 HRCAs).
Data availability
For the GRIFFIN study, the data sharing policy of Janssen Pharmaceutical Companies of Johnson & Johnson is available at https://www.janssen.com/clinical-trials/transparency. As noted on this site, requests for access to the study data can be submitted through Yale Open Data Access (YODA) Project site at http://yoda.yale.edu. For the investigator-initiated MASTER study, although these data are not currently publicly available, requests for sharing can be sent to the corresponding author of this manuscript and will be evaluated on an individual basis.
References
de Weers M, Tai YT, van der Veer MS, Bakker JM, Vink T, Jacobs DCH, et al. Daratumumab, a novel therapeutic human CD38 monoclonal antibody, induces killing of multiple myeloma and other hematological tumors. J Immunol. 2011;186:1840–8.
Lammerts van Bueren J, Jakobs D, Kaldenhoven N, Roza M, Hiddingh S, Meesters J, et al. Direct in vitro comparison of daratumumab with surrogate analogs of CD38 antibodies MOR03087, SAR650984 and Ab79. Blood. 2014;124:3474.
Overdijk MB, Verploegen S, Bögels M, van Egmond M, Lammerts van Bueren JJ, et al. Antibody-mediated phagocytosis contributes to the anti-tumor activity of the therapeutic antibody daratumumab in lymphoma and multiple myeloma. MAbs. 2015;7:311–21.
Overdijk MB, Jansen JHM, Nederend M, Lammerts van Bueren JJ, Groen RWJ, Parren PWHI, et al. The therapeutic CD38 monoclonal antibody daratumumab induces programmed cell death via Fcγ receptor–mediated cross-linking. J Immunol. 2016;197:807–13.
Krejcik J, Casneuf T, Nijhof IS, Verbist B, Bald J, Plesner T, et al. Daratumumab depletes CD38+ immune regulatory cells, promotes T-cell expansion, and skews T-cell repertoire in multiple myeloma. Blood. 2016;128:384–94.
Adams HC III, Stevenaert F, Krejcik J, Van der Borght K, Smets T, Bald J, et al. High-parameter mass cytometry evaluation of relapsed/refractory multiple myeloma patients treated with daratumumab demonstrates immune modulation as a novel mechanism of action. Cytometry A. 2019;95:279–89.
Casneuf T, Adams HC III, van de Donk NWCJ, Abraham Y, Bald J, Vanhoof G, et al. Deep immune profiling of patients treated with lenalidomide and dexamethasone with or without daratumumab. Leukemia. 2021;35:573–84.
Kinder M, Bahlis NJ, Malavasi F, De Goeij B, Babich A, Sendecki J, et al. Comparison of CD38 antibodies in vitro and ex vivo mechanisms of action in multiple myeloma. Haematologica. 2021;106:2004–8.
DARZALEX® (daratumumab) injection, for intravenous use [package insert]. Horsham, PA: Janssen Biotech, Inc.; 2023.
Dimopoulos MA, Moreau P, Terpos E, Mateos MV, Zweegman S, Cook G, et al. Multiple myeloma: EHA-ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Hemasphere. 2021;5:e528.
Costa LJ, Chhabra S, Medvedova E, Dholaria BR, Schmidt TM, Godby KN, et al. Daratumumab, carfilzomib, lenalidomide, and dexamethasone with minimal residual disease response-adapted therapy in newly diagnosed multiple myeloma. J Clin Oncol. 2022;40:2901–12.
Costa LJ, Chhabra S, Medvedova E, Dholaria BR, Schmidt TM, Godby KN, et al. Minimal residual disease response-adapted therapy in newly diagnosed multiple myeloma (MASTER): final report of the multicentre, single-arm, phase 2 trial. Lancet Haematol. 2023;10:e890–e901.
Voorhees PM, Kaufman JL, Laubach J, Sborov DW, Reeves B, Rodriguez C, et al. Daratumumab, lenalidomide, bortezomib, and dexamethasone for transplant-eligible newly diagnosed multiple myeloma: the GRIFFIN trial. Blood. 2020;136:936–45.
Voorhees PM, Sborov DW, Laubach J, Kaufman JL, Reeves B, Rodriguez C, et al. Addition of daratumumab to lenalidomide, bortezomib, and dexamethasone for transplantation-eligible patients with newly diagnosed multiple myeloma (GRIFFIN): final analysis of an open-label, randomised, phase 2 trial. Lancet Haematol. 2023;10:e825–e37.
Mateos MV, Martínez BP, González-Calle V. High-risk multiple myeloma: how to treat at diagnosis and relapse? Hematology Am Soc Hematol Educ Program. 2021;2021:30–36.
Costa LJ, Usmani SZ. Defining and managing high-risk multiple myeloma: current concepts. J Natl Compr Canc Netw. 2020;18:1730–7.
Chng WJ, Dispenzieri A, Chim CS, Fonseca R, Goldschmidt H, Lentzsch S, et al. IMWG consensus on risk stratification in multiple myeloma. Leukemia. 2014;28:269–77.
Sonneveld P, Avet-Loiseau H, Lonial S, Usmani S, Siegel D, Anderson KC, et al. Treatment of multiple myeloma with high-risk cytogenetics: a consensus of the International Myeloma Working Group. Blood. 2016;127:2955–62.
Walker BA, Mavrommatis K, Wardell CP, Ashby TC, Bauer M, Davies F, et al. A high-risk, Double-Hit, group of newly diagnosed myeloma identified by genomic analysis. Leukemia. 2019;33:159–70.
Voorhees PM, Rodriguez C, Reeves B, Nathwani N, Costa LJ, Lutska Y, et al. Daratumumab plus RVd for newly diagnosed multiple myeloma: final analysis of the safety run-in cohort of GRIFFIN. Blood Adv. 2021;5:1092–6.
Rajkumar SV, Dimopoulos MA, Palumbo A, Blade J, Merlini G, Mateos MV, et al. International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol. 2014;15:e538–e48.
Kumar S, Paiva B, Anderson KC, Durie B, Landgren O, Moreau P, et al. International Myeloma Working Group consensus criteria for response and minimal residual disease assessment in multiple myeloma. Lancet Oncol. 2016;17:e328–e46.
Mateos MV, Dimopoulos MA, Cavo M, Suzuki K, Jakubowiak A, Knop S, et al. Daratumumab plus bortezomib, melphalan, and prednisone for untreated myeloma. N Engl J Med. 2018;378:518–28.
Facon T, Kumar SK, Plesner T, Orlowski RZ, Moreau P, Bahlis N, et al. Daratumumab, lenalidomide, and dexamethasone versus lenalidomide and dexamethasone alone in newly diagnosed multiple myeloma (MAIA): overall survival results from a randomised, open-label, phase 3 trial. Lancet Oncol. 2021;22:1582–96.
Moreau P, Attal M, Hulin C, Arnulf B, Belhadj K, Benboubker L, et al. Bortezomib, thalidomide, and dexamethasone with or without daratumumab before and after autologous stem-cell transplantation for newly diagnosed multiple myeloma (CASSIOPEIA): a randomised, open-label, phase 3 study. Lancet. 2019;394:29–38.
Giri S, Grimshaw A, Bal S, Godby K, Kharel P, Djulbegovic B, et al. Evaluation of daratumumab for the treatment of multiple myeloma in patients with high-risk cytogenetic factors: a systematic review and meta-analysis. JAMA Oncol. 2020;6:1759–65.
Jakubowiak AJ, Kumar S, Medhekar R, Pei H, Lefebvre P, Kaila S, et al. Daratumumab improves depth of response and progression-free survival in transplant-ineligible, high-risk, newly diagnosed multiple myeloma. Oncologist. 2022;27:e589–e96.
Morgan GJ, Davies FE, Gregory WM, Cocks K, Bell SE, Szubert AJ, et al. First-line treatment with zoledronic acid as compared with clodronic acid in multiple myeloma (MRC Myeloma IX): a randomised controlled trial. Lancet. 2010;376:1989–99.
Shah V, Sherborne AL, Walker BA, Johnson DC, Boyle EM, Ellis S, et al. Prediction of outcome in newly diagnosed myeloma: a meta-analysis of the molecular profiles of 1905 trial patients. Leukemia. 2018;32:102–10.
Kaiser MF, Hall A, Walker K, Newnham N, de Tute RM, Roberts S, et al. Daratumumab, cyclophosphamide, bortezomib, lenalidomide, dexamethasone (Dara-CVRd), V-augmented autologous stem cell transplant (V-ASCT) and Dara-Vrd consolidation in ultra-high risk (UHiR) newly diagnosed myeloma (NDMM) and primary plasma cell leukemia (pPCL) compared with Myeloma XI/XI+ trial treatment for Uhir MM: the UK Optimum/Muknine trial. Blood. 2021;138:465.
Ross FM, Avet-Loiseau H, Ameye G, Gutierrez NC, Liebisch P, O’Connor S, et al. Report from the European Myeloma Network on interphase FISH in multiple myeloma and related disorders. Haematologica. 2012;97:1272–7.
Acknowledgements
The authors thank the patients who volunteered to participate in these studies, their families, and the staff members at the study sites who cared for them. For the MASTER study (ClinicalTrials.gov Identifier: NCT03224507), the authors would also like to acknowledge the Academic Consortium to Overcome Multiple Myeloma through Innovative Trials (COMMIT) and the partnership with Adaptive Biotechnologies for assistance with IDE. The investigator-initiated MASTER study received support from Amgen and Janssen Pharmaceuticals. The GRIFFIN study (ClinicalTrials.gov Identifier: NCT02874742) was supported in part by the Alliance Foundation Trials, LLC, also in part by funds from Janssen Oncology Research and Development. The content is solely the responsibility of the authors and does not necessarily represent the official views of the Alliance Foundation Trials, LLC. https://acknowledgments.alliancefound.org. We also recognize the contributions of Shira Dinner, the Executive Officer for Alliance Foundation Trials for Clinical Trials. Medical writing and editorial support were provided by Charlotte D. Majerczyk, PhD, of Lumanity Communications Inc., and were funded by Janssen Global Services, LLC.
Author information
Authors and Affiliations
Contributions
NSC, JLK, JL, PH, RZO, HP, TSL, SG, LJC, PGR, and PMV contributed to the study design. NSC, RS, JLK, JL, DWS, EM, BR, CR, SC, A Chari, SB, LDA, BRD, NN, PH, NS, NB, SAH, CC, AJ, TMW, RZO, KHS, AJC, HP, SG, LJC, SZU, PGR, and PMV contributed to the investigation and data collection. NSC, RS, JLK, JL, TMS, DWS, BD, CR, SC, A Chari, SB, LDA, BRD, PH, NS, NB, SAH, AJ, RZO, KHS, AJC, HP, A Cortoos, SP, TSL, SG, LJC, SZU, PGR, and PMV contributed to the analysis and/or interpretation of data. All authors contributed to the drafting and/or reviewing of the manuscript and approve this manuscript for submission. All authors confirm access to the primary clinical trial data.
Corresponding authors
Ethics declarations
Competing interests
RS served as a consultant or in an advisory role for Sanofi-Aventis, Janssen Oncology, and Oncopeptides; and received research funding from Sanofi. JLK consulted for AbbVie, Janssen, Roche/Genentech, Bristol Myers Squibb, Celgene, and Tecnopharma; received research funding from AbbVie, Amgen, Bristol Myers Squibb, Fortis Therapeutics, Heidelberg Pharma, Janssen, Novartis, Roche/Genentech, Sutro Biopharma, and Takeda; received honoraria from AbbVie, Janssen, Roche/Genentech, and Tecnopharma; and holds a membership on a board or advisory committee for Incyte and TG Therapeutics. JL received honoraria from Great Debates & Updates – Hematologic Malignancies. TMS received consulting fees from BioLineRx, Janssen, and Sanofi; and received support for attending meetings and/or travel from Sanofi. DWS consulted for, holds membership on an entity’s board of directors for, or served on advisory committees for Arcellx, GSK, and Janssen; and consulted for Bristol Myers Squibb, Pfizer, AbbVie, and Sanofi. BR received honoraria from Incyte, Bristol Myers Squibb, and PharmaEssentia. BD received consulting fees from Janssen, Sanofi, Genentech, and AbbVie; received payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing, or educational events from Sanofi, Janssen, Karyopharm Therapeutics, Bristol Myers Squibb, and GSK; and participated on a data safety monitoring board or advisory board for Amgen, Takeda, Janssen, Sanofi, Arcellx, and Natera. CR consulted for Janssen, Bristol Myers Squibb, Takeda, AbbVie, Karyopharm Therapeutics, and Artiva; and served on a speakers bureau for Janssen, Bristol Myers Squibb, Takeda, AbbVie, Karyopharm Therapeutics, and Artiva. SC received research funding from Janssen, AbbVie, C4 Therapeutics, Takeda, and CARSgen Therapeutics; and received honoraria from Janssen, Sanofi, and GSK. SB received grants or contracts from an entity from the Amyloidosis Foundation; and received payment or honoraria for lectures, presentations, speakers bureaus, manuscript writing, or educational events from Adaptive Biotechnologies. LDA holds a membership on an entity’s board of directors or advisory committees for, served as a consultant for, and received honoraria from GSK, Bristol Myers Squibb, Celgene, Janssen, Amgen, Oncopeptides, Karyopharm Therapeutics, AbbVie, BeiGene, Sanofi, and Cellectar Biosciences. BRD received research funding from Pfizer, Janssen, Takeda, MEI, Angiocrine, and Poseida; and served on a speakers bureau for Jazz Pharmaceuticals and Celgene. PH served as a consultant for and received honoraria from Sanofi, Amgen, Janssen, Karyopharm Therapeutics, Takeda, and Kite; was a consultant for and received honoraria and research funding from Bristol Myers Squibb; received research funding from Millennium and Spectrum Pharmaceuticals; received honoraria from Novartis, Incyte, GSK, and AbbVie; and was a consultant for Pharmacyclics. NS received research funding from Bristol Myers Squibb/Celgene, Janssen, bluebird bio, Sutro Biopharma, TeneoBio, Poseida, Nektar, and Precision Biosciences; served as a consultant for GSK, Amgen, Indapta Therapeutics, Sanofi, CareDx, Kite, Karyopharm Therapeutics, Oncopeptides, and CSL Behring; and is a current employee and equity holder of AstraZeneca. NB served on a speakers bureau for Amgen, Sanofi, and Genzyme; and served on an advisory board for Sanofi, Genzyme, and Janssen. SAH served as a consultant for Bristol Myers Squibb/Celgene, Janssen, Takeda, Pfizer, Oncopeptides, GSK, Secura Bio, and Sanofi; and received research funding from Bristol Myers Squibb and Oncopeptides. CC received honoraria from Takeda, Bristol Myers Squibb, Pfizer, and Janssen. AJ served as a consultant or in an advisory role for and received honoraria from AbbVie, Amgen, Bristol Myers Squibb, Celgene, GSK, Janssen, Karyopharm Therapeutics, and Sanofi. TMW served as a consultant for Carevive, Janssen, Seagen, and Sanofi. RZO received research funding from Asylia Therapeutics, Biotheryx, Heidelberg Pharma, CARsgen Therapeutics, Bristol Myers Squibb/Celgene, Exelixis, Janssen Biotech, Sanofi-Aventis, and Takeda Pharmaceuticals North America; received honoraria from and holds a membership on an entity’s board of directors or advisory committees for AbbVie, Biotheryx, Bristol Myers Squibb, Janssen Biotech, Karyopharm Therapeutics, Meridian Therapeutics, Monte Rosa Therapeutics, Neoleukin Corporation, Oncopeptides AB, Regeneron Pharmaceuticals, Sanofi-Aventis, and Takeda Pharmaceuticals North America; and is a current stockholder of Asylia Therapeutics. KHS received honoraria from Bristol Myers Squibb, Janssen, GSK, Adaptive Biotechnologies, Sanofi, Takeda, and Amgen; served as an ad hoc member of an advisory committee for GSK, Janssen, and Bristol Myers Squibb; served on a speakers bureau for GSK, Bristol Myers Squibb, Sanofi, Karyopharm Therapeutics, Takeda, Janssen, Adaptive Biotechnologies, and Amgen; received research funding from AbbVie and Karyopharm Therapeutics; and is the principal investigator of clinical trials sponsored by Janssen and Bristol Myers Squibb, with all research outside the scope of the submitted work. AJC served as a consultant for and received research funding from Janssen, Bristol Myers Squibb, and AbbVie; received research funding from Harpoon, Sanofi-Aventis, and Nektar; was a consultant for Allogene, EUSA, GSK, and Secura Bio; and received research funding from and holds a membership on an entity’s board of directors or advisory committees for Adaptive Biotechnologies. SG received honoraria from Carevive and OncLive; and received research funding from Carevive and Pack Health. LJC served as a consultant or in an advisory role for AbbVie, Amgen, Celgene, Karyopharm Therapeutics, and Sanofi; served on a speakers bureau for Amgen and Sanofi; received honoraria from Amgen, Celgene, Janssen, Karyopharm Therapeutics, and Sanofi; and received research funding from Amgen and Janssen. SZU served as a consultant for Celgene, Amgen, Janssen Oncology, Seattle Genetics, Takeda, GSK, Karyopharm Therapeutics, AbbVie, SkylineDx, Merck, Oncopeptides, Genentech, Gilead Sciences, and Bristol Myers Squibb/Celgene; served on a speakers bureau for Takeda, Amgen, Janssen Oncology, Sanofi, and Bristol Myers Squibb/Celgene; and received research funding from Celgene and Array BioPharma. PGR received research funding from Oncopeptides, Celgene/Bristol Myers Squibb, Takeda, and Karyopharm Therapeutics; and served on advisory committees for Oncopeptides, Celgene/Bristol Myers Squibb, Takeda, Karyopharm Therapeutics, Janssen, Sanofi, Secura Bio, GSK, Regeneron Pharmaceuticals, AstraZeneca, and Protocol Intelligence. PMV served as a consultant for, received honoraria from, and holds a membership on an entity’s board of directors or advisory committees for AbbVie, Bristol Myers Squibb, Karyopharm Therapeutics, Regeneron Pharmaceuticals, and Sanofi. HP, A Cortoos, SP, and TSL are current equity holders and employees of Janssen. NSC, KNG, EM, A Chari, and NN have nothing to disclose.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Callander, N.S., Silbermann, R., Kaufman, J.L. et al. Daratumumab-based quadruplet therapy for transplant-eligible newly diagnosed multiple myeloma with high cytogenetic risk. Blood Cancer J. 14, 69 (2024). https://doi.org/10.1038/s41408-024-01030-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41408-024-01030-w
- Springer Nature Limited